Daptomycin-Nonsusceptible Staphylococcus aureus: The Role of Combination Therapy with Daptomycin and Gentamicin
Abstract
:1. Introduction
2. Experimental Section
2.1. Bacterial Strains
| Patient | Strains | Clinical Syndrome | Multi-Locus Sequence Type | MIC (μg/mL) | References | ||
|---|---|---|---|---|---|---|---|
| Daptomycin | Vancomycin | Gentamicin | |||||
| 1 | A6224 | Bacteremia | 5 | 0.25 | 2 | 0.5 | [10] |
| A6226 | 2 | 4 | 0.5 | ||||
| 2 | A9719 | Endocarditis | 5 | 0.25 | 2 | 0.5 | [7] |
| A9744 | 2 | 2 | 1 | ||||
| 3 | A8819 | Osteomyelitis, septic arthritis | 105 | 0.25 | 1 | 0.5 | [7] |
| A8817 | 2 | 1 | 0.5 | ||||
| 4 | A8796 | Bacteremia, osteomyelitis | 105 | 0.25 | 1 | 0.5 | [2,7] |
| 5 | A9639 | Bacteremia, osteomyelitis | 1892 | 2 | 4 | 1 | [10,30] |
| Control | ATCC 29213 | − | − | 0.25 | 1 | 0.5 | − |
2.2. Antibiotic Susceptibility Testing
2.3. Time-Kill Analyses
3. Results and Discussion
3.1. Synergistic Bactericidal Effects of Gentamicin and Daptomycin at the Concentration Mimicking That in Tissue Compartments
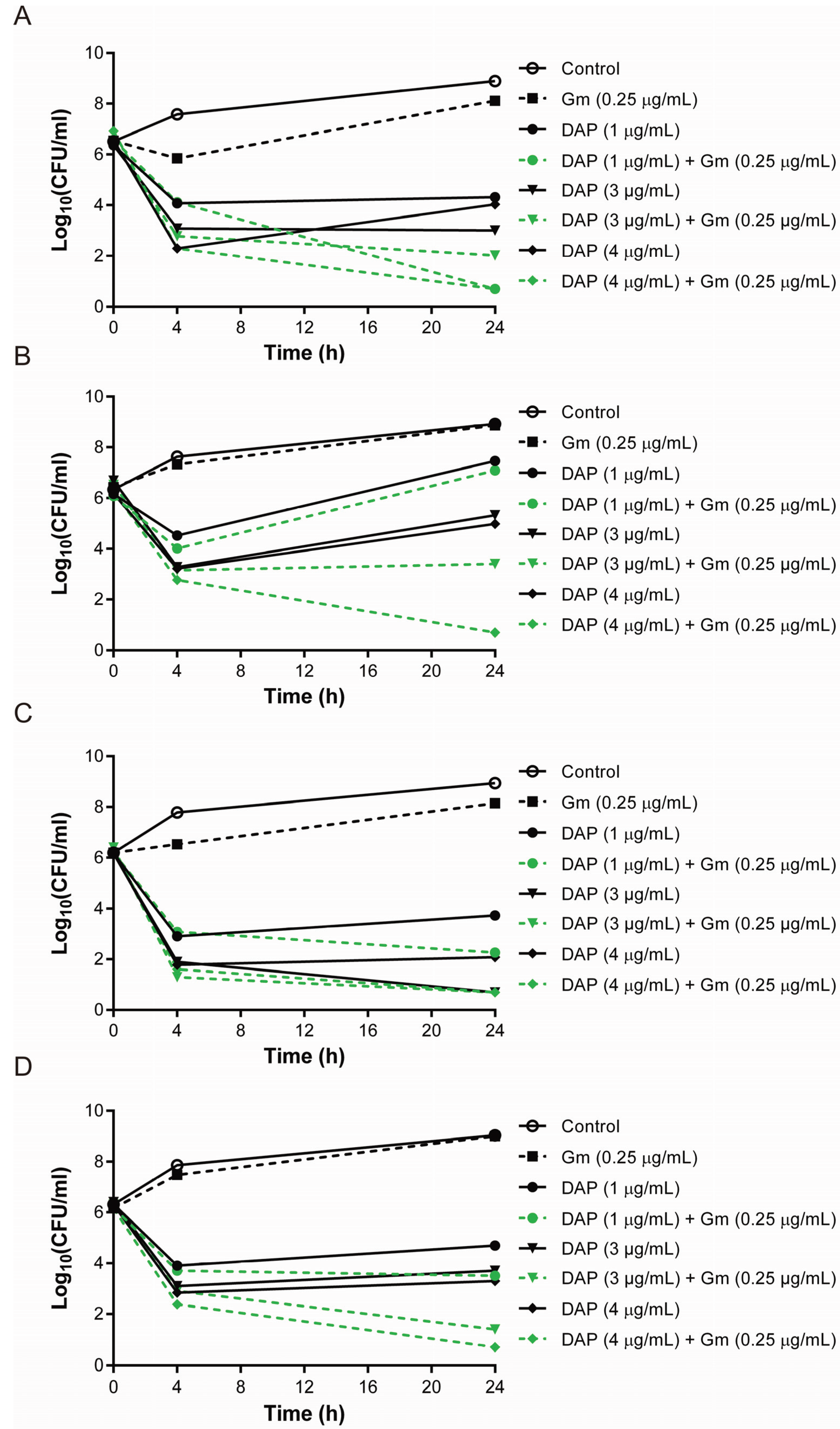
3.2. The Combination of Daptomycin and Gentamicin Effectively Eradicated Daptomycin-Exposed or Vancomycin-Exposed DAP-R S. aureus Isolates
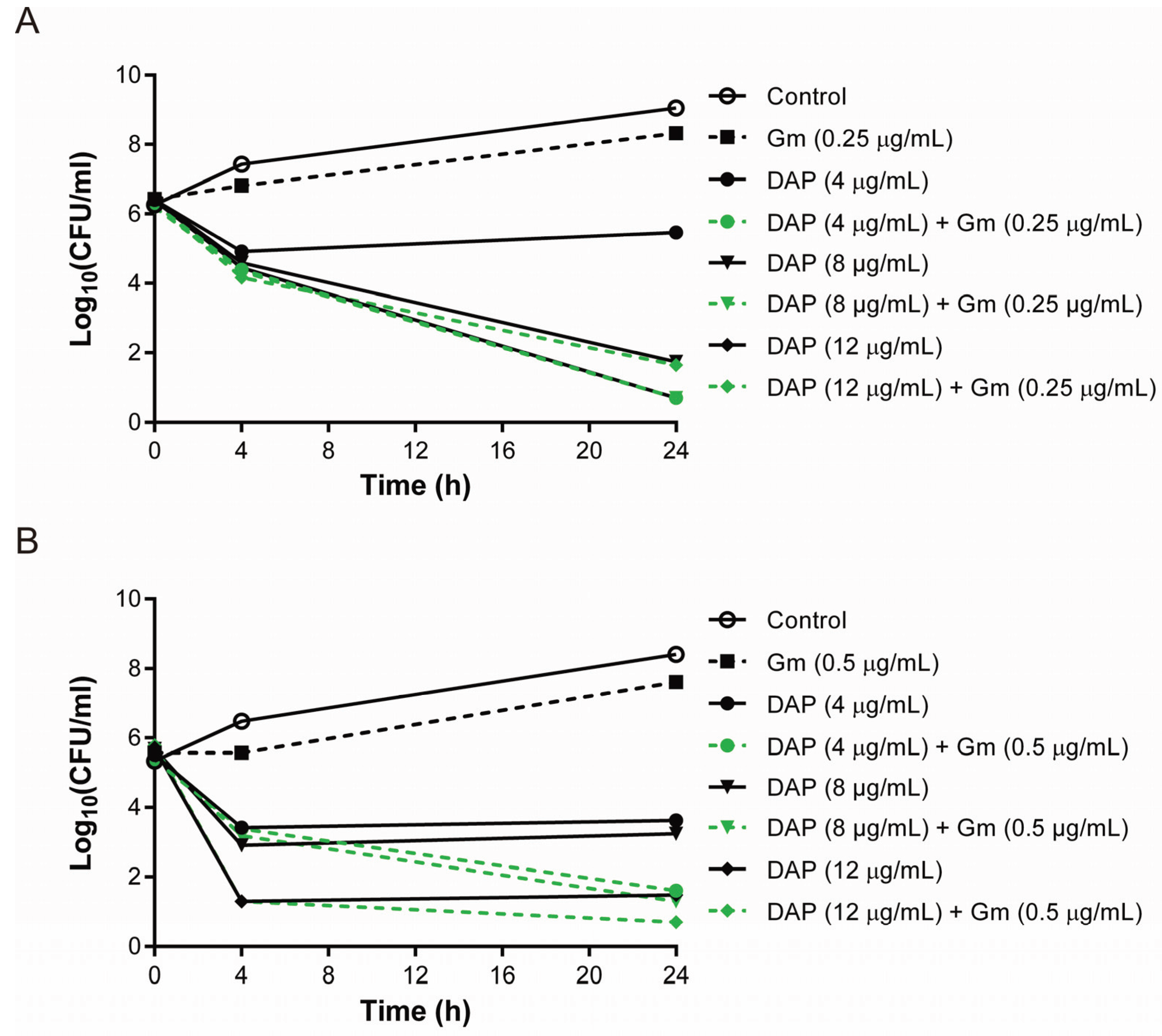
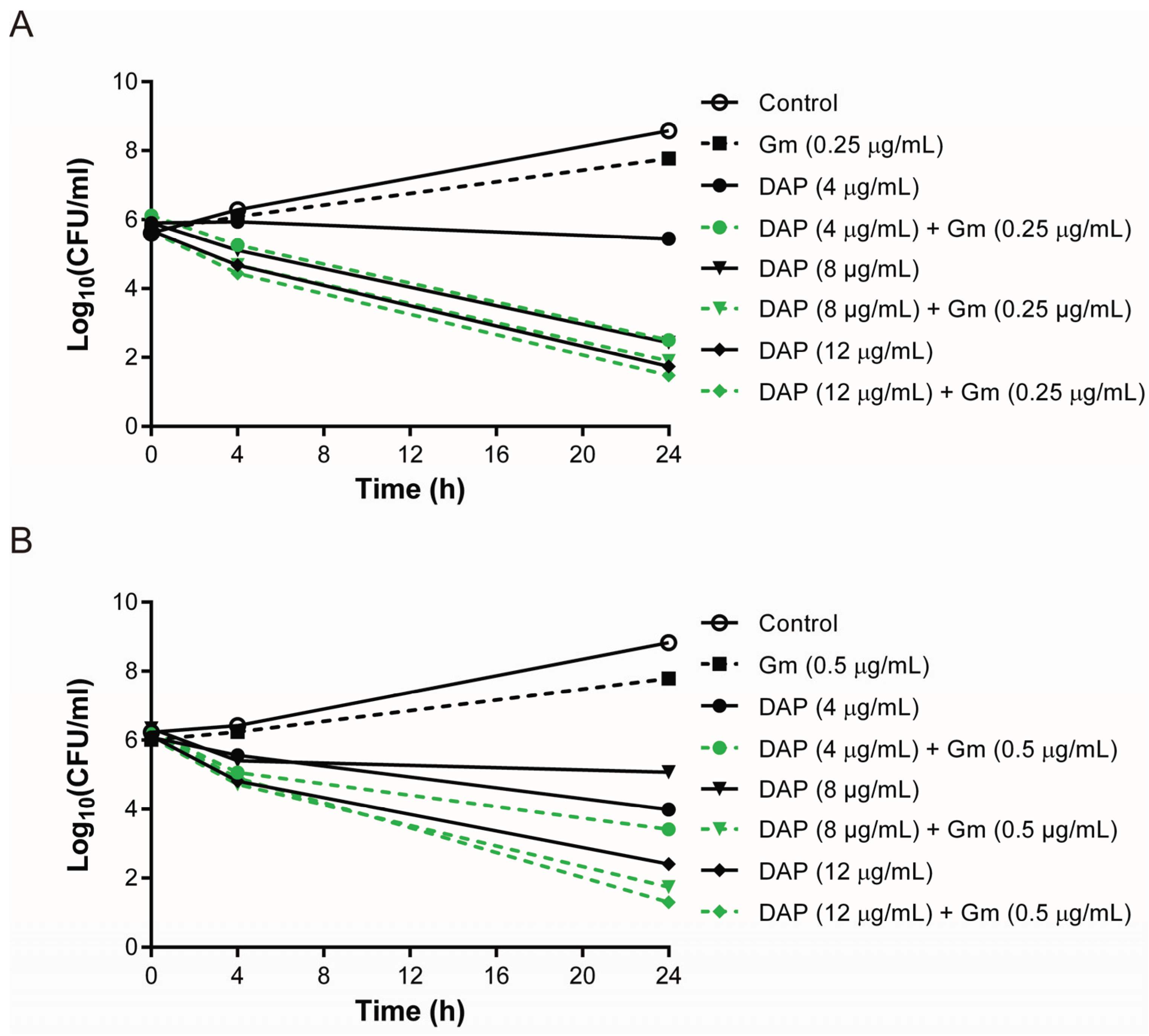
3.3. Lower Bactericidal Rates of Daptomycin and Gentamicin against Vancomycin-Exposed DAP-R Isolates Compared to the Rates against Daptomycin-Exposed DAP-R Isolates
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fowler, V.G., Jr.; Boucher, H.W.; Corey, G.R.; Abrutyn, E.; Karchmer, A.W.; Rupp, M.E.; Levine, D.P.; Chambers, H.F.; Tally, F.P.; Vigliani, G.A.; et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by staphylococcus aureus. N. Engl. J. Med. 2006, 355, 653–665. [Google Scholar] [CrossRef] [PubMed]
- Marty, F.M.; Yeh, W.W.; Wennersten, C.B.; Venkataraman, L.; Albano, E.; Alyea, E.P.; Gold, H.S.; Baden, L.R.; Pillai, S.K. Emergence of a clinical daptomycin-resistant staphylococcus aureus isolate during treatment of methicillin-resistant staphylococcus aureus bacteremia and osteomyelitis. J. Clin. Microbiol. 2006, 44, 595–597. [Google Scholar] [CrossRef] [PubMed]
- Friedman, L.; Alder, J.D.; Silverman, J.A. Genetic changes that correlate with reduced susceptibility to daptomycin in staphylococcus aureus. Antimicrob. Agents Chemother. 2006, 50, 2137–2145. [Google Scholar] [CrossRef] [PubMed]
- Ernst, C.M.; Staubitz, P.; Mishra, N.N.; Yang, S.J.; Hornig, G.; Kalbacher, H.; Bayer, A.S.; Kraus, D.; Peschel, A. The bacterial defensin resistance protein mprf consists of separable domains for lipid lysinylation and antimicrobial peptide repulsion. PLoS Pathogens 2009, 5, e1000660. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.N.; Yang, S.J.; Sawa, A.; Rubio, A.; Nast, C.C.; Yeaman, M.R.; Bayer, A.S. Analysis of cell membrane characteristics of in vitro-selected daptomycin-resistant strains of methicillin-resistant staphylococcus aureus. Antimicrob. Agents Chemother. 2009, 53, 2312–2318. [Google Scholar] [CrossRef] [PubMed]
- Rubio, A.; Moore, J.; Varoglu, M.; Conrad, M.; Chu, M.; Shaw, W.; Silverman, J.A. LC-MS/MS characterization of phospholipid content in daptomycin-susceptible and -resistant isolates of staphylococcus aureus with mutations in Mprf. Mol. Membr. Biol. 2012, 29, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Peleg, A.Y.; Miyakis, S.; Ward, D.V.; Earl, A.M.; Rubio, A.; Cameron, D.R.; Pillai, S.; Moellering, R.C., Jr.; Eliopoulos, G.M. Whole genome characterization of the mechanisms of daptomycin resistance in clinical and laboratory derived isolates of staphylococcus aureus. PloS ONE 2012, 7, e28316. [Google Scholar] [CrossRef] [PubMed]
- Dubrac, S.; Boneca, I.G.; Poupel, O.; Msadek, T. New insights into the walk/walr (YycG/YycF) essential signal transduction pathway reveal a major role in controlling cell wall metabolism and biofilm formation in staphylococcus aureus. J. Bacteriol. 2007, 189, 8257–8269. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Tominaga, E.; Neoh, H.M.; Hiramatsu, K. Correlation between reduced daptomycin susceptibility and vancomycin resistance in vancomycin-intermediate staphylococcus aureus. Antimicrob. Agents Chemother. 2006, 50, 1079–1082. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.R.; Ward, D.V.; Kostoulias, X.; Howden, B.P.; Moellering, R.C., Jr.; Eliopoulos, G.M.; Peleg, A.Y. Serine/threonine phosphatase stp1 contributes to reduced susceptibility to vancomycin and virulence in staphylococcus aureus. J. Infectious Dis. 2012, 205, 1677–1687. [Google Scholar] [CrossRef] [PubMed]
- Howden, B.P.; McEvoy, C.R.; Allen, D.L.; Chua, K.; Gao, W.; Harrison, P.F.; Bell, J.; Coombs, G.; Bennett-Wood, V.; Porter, J.L.; et al. Evolution of multidrug resistance during staphylococcus aureus infection involves mutation of the essential two component regulator walkr. PLoS Pathogens 2011, 7, e1002359. [Google Scholar] [CrossRef] [PubMed]
- Debbia, E.; Pesce, A.; Schito, G.C. In vitro activity of LY146032 alone and in combination with other antibiotics against gram-positive bacteria. Antimicrob. Agents Chemother. 1988, 32, 279–281. [Google Scholar] [CrossRef] [PubMed]
- Agence francaise de securite sanitaire des produits de, s. Update on good use of injectable aminoglycosides, gentamycin, tobramycin, netilmycin, amikacin. Pharmacological properties, indications, dosage, and mode of administration, treatment monitoring. Med. Mal. Infectieuses 2012, 42, 301–308. [Google Scholar]
- Snydman, D.R.; McDermott, L.A.; Jacobus, N.V. Evaluation of in vitro interaction of daptomycin with gentamicin or beta-lactam antibiotics against staphylococcus aureus and enterococci by FIC index and timed-kill curves. J. Chemother. 2005, 17, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, B.T.; Rybak, M.J. Etest synergy testing of clinical isolates of staphylococcus aureus demonstrating heterogeneous resistance to vancomycin. Diagn. Microbiol. Infectious Dis. 2006, 54, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, B.T.; Rybak, M.J. Short-course gentamicin in combination with daptomycin or vancomycin against staphylococcus aureus in an in vitro pharmacodynamic model with simulated endocardial vegetations. Antimicrob. Agents Chemother. 2005, 49, 2735–2745. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Credito, K.; Lin, G.; Appelbaum, P.C. Activity of daptomycin alone and in combination with rifampin and gentamicin against staphylococcus aureus assessed by time-kill methodology. Antimicrob. Agents Chemother. 2007, 51, 1504–1507. [Google Scholar] [CrossRef] [PubMed]
- Baltch, A.L.; Ritz, W.J.; Bopp, L.H.; Michelsen, P.B.; Smith, R.P. Antimicrobial activities of daptomycin, vancomycin, and oxacillin in human monocytes and of daptomycin in combination with gentamicin and/or rifampin in human monocytes and in broth against staphylococcus aureus. Antimicrob. Agents Chemother. 2007, 51, 1559–1562. [Google Scholar] [CrossRef] [PubMed]
- Baltch, A.L.; Ritz, W.J.; Bopp, L.H.; Michelsen, P.; Smith, R.P. Activities of daptomycin and comparative antimicrobials, singly and in combination, against extracellular and intracellular staphylococcus aureus and its stable small-colony variant in human monocyte-derived macrophages and in broth. Antimicrob. Agents Chemother. 2008, 52, 1829–1833. [Google Scholar] [CrossRef] [PubMed]
- Miro, J.M.; Garcia-de-la-Maria, C.; Armero, Y.; Soy, D.; Moreno, A.; del Rio, A.; Almela, M.; Sarasa, M.; Mestres, C.A.; Gatell, J.M.; et al. Addition of gentamicin or rifampin does not enhance the effectiveness of daptomycin in treatment of experimental endocarditis due to methicillin-resistant staphylococcus aureus. Antimicrob. Agents Chemother. 2009, 53, 4172–4177. [Google Scholar] [CrossRef] [PubMed]
- Entenza, J.M.; Giddey, M.; Vouillamoz, J.; Moreillon, P. In vitro prevention of the emergence of daptomycin resistance in staphylococcus aureus and enterococci following combination with amoxicillin/clavulanic acid or ampicillin. Int. J. Antimicrob. Agents 2010, 35, 451–456. [Google Scholar] [CrossRef] [PubMed]
- LaPlante, K.L.; Woodmansee, S. Activities of daptomycin and vancomycin alone and in combination with rifampin and gentamicin against biofilm-forming methicillin-resistant staphylococcus aureus isolates in an experimental model of endocarditis. Antimicrob. Agents Chemother. 2009, 53, 3880–3886. [Google Scholar] [CrossRef] [PubMed]
- LaPlante, K.L.; Rybak, M.J. Impact of high-inoculum staphylococcus aureus on the activities of nafcillin, vancomycin, linezolid, and daptomycin, alone and in combination with gentamicin, in an in vitro pharmacodynamic model. Antimicrob. Agents Chemother. 2004, 48, 4665–4672. [Google Scholar] [CrossRef] [PubMed]
- DeRyke, C.A.; Sutherland, C.; Zhang, B.; Nicolau, D.P.; Kuti, J.L. Serum bactericidal activities of high-dose daptomycin with and without coadministration of gentamicin against isolates of staphylococcus aureus and enterococcus species. Antimicrob. Agents chemother. 2006, 50, 3529–3534. [Google Scholar] [CrossRef] [PubMed]
- Rose, W.E.; Leonard, S.N.; Rybak, M.J. Evaluation of daptomycin pharmacodynamics and resistance at various dosage regimens against staphylococcus aureus isolates with reduced susceptibilities to daptomycin in an in vitro pharmacodynamic model with simulated endocardial vegetations. Antimicrob. Agents Chemother. 2008, 52, 3061–3067. [Google Scholar] [CrossRef] [PubMed]
- Steenbergen, J.N.; Mohr, J.F.; Thorne, G.M. Effects of daptomycin in combination with other antimicrobial agents: A review of in vitro and animal model studies. J. Antimicrob. Chemother. 2009, 64, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Dvorchik, B.H.; Brazier, D.; DeBruin, M.F.; Arbeit, R.D. Daptomycin pharmacokinetics and safety following administration of escalating doses once daily to healthy subjects. Antimicrob. Agents Chemother. 2003, 47, 1318–1323. [Google Scholar] [CrossRef] [PubMed]
- Montange, D.; Berthier, F.; Leclerc, G.; Serre, A.; Jeunet, L.; Berard, M.; Muret, P.; Vettoretti, L.; Leroy, J.; Hoen, B.; et al. Penetration of daptomycin into bone and synovial fluid in joint replacement. Antimicrob. Agents Chemother. 2014, 58, 3991–3996. [Google Scholar] [CrossRef] [PubMed]
- Traunmuller, F.; Schintler, M.V.; Metzler, J.; Spendel, S.; Mauric, O.; Popovic, M.; Konz, K.H.; Scharnagl, E.; Joukhadar, C. Soft tissue and bone penetration abilities of daptomycin in diabetic patients with bacterial foot infections. J. Antimicrob. Chemother. 2010, 65, 1252–1257. [Google Scholar] [CrossRef] [PubMed]
- Peleg, A.Y.; Monga, D.; Pillai, S.; Mylonakis, E.; Moellering, R.C., Jr.; Eliopoulos, G.M. Reduced susceptibility to vancomycin influences pathogenicity in staphylococcus aureus infection. J. Infectious Dis. 2009, 199, 532–536. [Google Scholar] [CrossRef] [PubMed]
- National Committee for Clinical Laboratory Standards. Methods for Dilution Antimicrobial Susceptibility Testing for Bacteria that Grow Aerobically. In Approved Standard M7–A5, 5th ed.; Committee for Clinical Laboratory Standards: Wayne, PA, USA, 2000. [Google Scholar]
- Pillai, S.K.; Moellering, R.C., Jr.; Eliopoulos, G.M. Antimicrobial Combinations. In Antibiotics in Laboratory Medicine, 5th ed.; Lorian, V., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2005; pp. 365–440. [Google Scholar]
- Zammit, M.C.; Fiorentino, L.; Cassar, K.; Azzopardi, L.M.; LaFerla, G. Factors affecting gentamicin penetration in lower extremity ischemic tissues with ulcers. Int. J. Lower Extremity Wounds 2011, 10, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Lorentzen, H.; Kallehave, F.; Kolmos, H.J.; Knigge, U.; Bulow, J.; Gottrup, F. Gentamicin concentrations in human subcutaneous tissue. Antimicrob. Agents Chemother. 1996, 40, 1785–1789. [Google Scholar] [PubMed]
- Gill, M.A.; Cohen, J.L.; Chenella, F.C.; Hisayasu, G.H.; Chandrasoma, P.; Warnecke, G.M.; Chung, H.; Heseltine, P.N.; Yellin, A.E.; Berne, T.V. Gentamicin penetration into diseased appendix tissue. Ther. Drug Monit. 1984, 6, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Beraud, G.; le Moal, G.; Elsendoorn, A.; Tattevin, P.; Godet, C.; Alfandari, S.; Couet, W.; Roblot, P.; Roblot, F. A Survey on the use of gentamicin in infective endocarditis. Eur. J. Clin. Microbiol. Infectious Dis. 2012, 31, 1413–1418. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Suecof, L.A.; Sutherland, C.A.; Gao, L.; Kuti, J.L.; Nicolau, D.P. In vivo microdialysis study of the penetration of daptomycin into soft tissues in diabetic versus healthy volunteers. Antimicrob. Agents Chemother. 2008, 52, 3941–3946. [Google Scholar] [CrossRef] [PubMed]
- Gika, H.G.; Michopoulos, F.; Divanis, D.; Metalidis, S.; Nikolaidis, P.; Theodoridis, G.A. Daptomycin determination by liquid chromatography-mass spectrometry in peritoneal fluid, blood plasma, and urine of clinical patients receiving peritoneal dialysis treatment. Anal. Bioanal. Chem. 2010, 397, 2191–2197. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-H.; Peleg, A.Y.; Monash University, Clayton, Australia. Unpublished results. 2015.
- Yang, S.J.; Kreiswirth, B.N.; Sakoulas, G.; Yeaman, M.R.; Xiong, Y.Q.; Sawa, A.; Bayer, A.S. Enhanced expression of dltabcd is associated with the development of daptomycin nonsusceptibility in a clinical endocarditis isolate of staphylococcus aureus. J. Infectious Dis. 2009, 200, 1916–1920. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.J.; Bayer, A.S.; Mishra, N.N.; Meehl, M.; Ledala, N.; Yeaman, M.R.; Xiong, Y.Q.; Cheung, A.L. The staphylococcus aureus two-component regulatory system, grars, senses and confers resistance to selected cationic antimicrobial peptides. Infection Immunity 2012, 80, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Camargo, I.L.; Neoh, H.M.; Cui, L.; Hiramatsu, K. Serial daptomycin selection generates daptomycin-nonsusceptible staphylococcus aureus strains with a heterogeneous vancomycin-intermediate phenotype. Antimicrob. Agents Chemother. 2008, 52, 4289–4299. [Google Scholar] [CrossRef] [PubMed]
- Blake, K.L.; O’Neill, A.J. Transposon library screening for identification of genetic loci participating in intrinsic susceptibility and acquired resistance to antistaphylococcal agents. J. Antimicrob. Chemother. 2013, 68, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Akins, R.L.; Rybak, M.J. In vitro activities of daptomycin, arbekacin, vancomycin, and gentamicin alone and/or in combination against glycopeptide intermediate-resistant staphylococcus aureus in an infection model. Antimicrob. Agents Chemother. 2000, 44, 1925–1929. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, J.-H.; Peleg, A.Y. Daptomycin-Nonsusceptible Staphylococcus aureus: The Role of Combination Therapy with Daptomycin and Gentamicin. Genes 2015, 6, 1256-1267. https://doi.org/10.3390/genes6041256
Jiang J-H, Peleg AY. Daptomycin-Nonsusceptible Staphylococcus aureus: The Role of Combination Therapy with Daptomycin and Gentamicin. Genes. 2015; 6(4):1256-1267. https://doi.org/10.3390/genes6041256
Chicago/Turabian StyleJiang, Jhih-Hang, and Anton Y. Peleg. 2015. "Daptomycin-Nonsusceptible Staphylococcus aureus: The Role of Combination Therapy with Daptomycin and Gentamicin" Genes 6, no. 4: 1256-1267. https://doi.org/10.3390/genes6041256
APA StyleJiang, J.-H., & Peleg, A. Y. (2015). Daptomycin-Nonsusceptible Staphylococcus aureus: The Role of Combination Therapy with Daptomycin and Gentamicin. Genes, 6(4), 1256-1267. https://doi.org/10.3390/genes6041256





