KAT8 beyond Acetylation: A Survey of Its Epigenetic Regulation, Genetic Variability, and Implications for Human Health
Abstract
1. Introduction
2. KAT8 SNPs: Genetic Variability and Implications
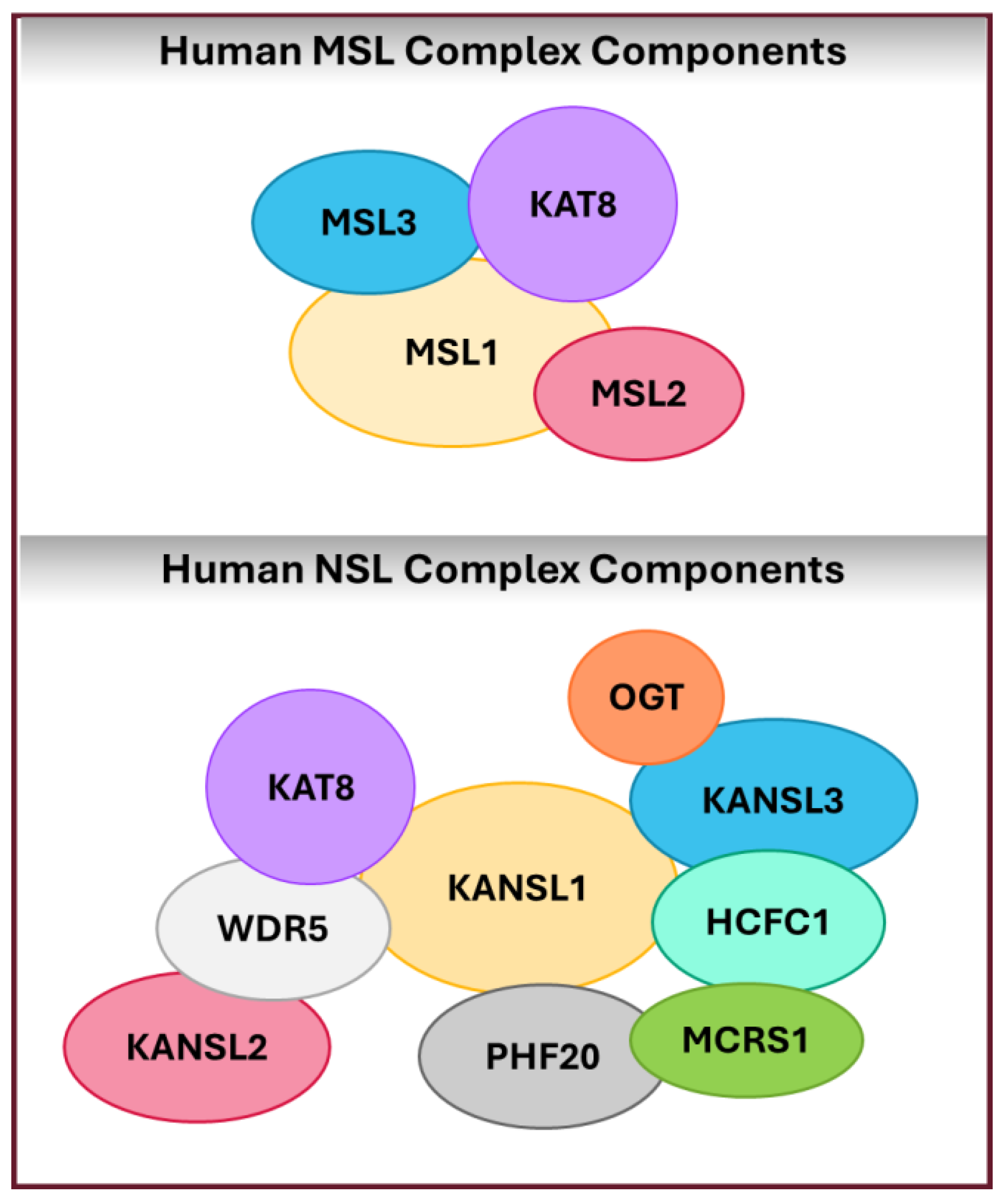
3. Structural Insights into KAT8 and Its Complexes
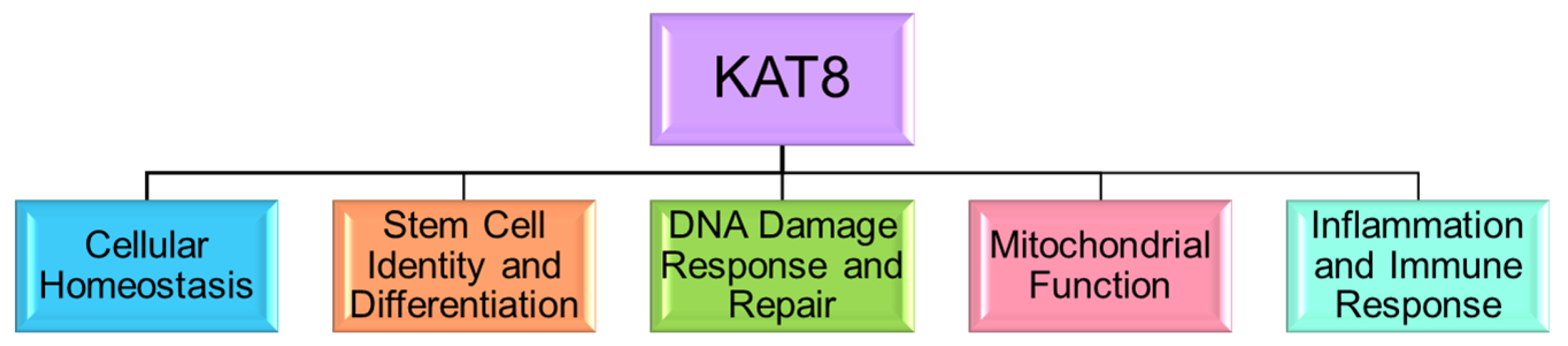
4. KAT8 in Cellular Homeostasis and Transcriptional Regulation
5. KAT8’s Role in Stem Cell Identity and Differentiation
6. KAT8 in DNA Damage Response and Repair
7. KAT8 in Mitochondrial Function
8. KAT8 in Inflammation and Immune Response
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| 2023 | The Genetic Basis of Childhood Obesity: A Systematic Review. | Vourdoumpa A, Paltoglou G, Charmandari E. | [66] |
| 2022 | Mendelian Randomization Analysis Reveals No Causal Relationship Between Nonalcoholic Fatty Liver Disease and Severe COVID-19. | Li J, Tian A, Zhu H, Chen L, et al. | [15] |
| 2022 | Simulated distributions from negative experiments highlight the importance of the body mass index distribution in explaining depression-body mass index genetic risk score interactions. | Casanova F, O’Loughlin J, Lewis C, Frayling TM, et al. | [67] |
| 2021 | Obesity Genes and Weight Loss During Lifestyle Intervention in Children With Obesity. | Heitkamp M, Siegrist M, Molnos S, Brandmaier S, et al. | [68] |
| 2020 | Genetic risk of obesity as a modifier of associations between neighbourhood environment and body mass index: an observational study of 335 046 UK Biobank participants. | Mason KE, Palla L, Pearce N, Phelan J, et al. | [69] |
| 2019 | Bidirectional Mendelian randomization to explore the causal relationships between body mass index and polycystic ovary syndrome. | Brower MA, Hai Y, Jones MR, Guo X, et al. | [70] |
| 2019 | Height and Body Mass Index as Modifiers of Breast Cancer Risk in BRCA1/2 Mutation Carriers: A Mendelian Randomization Study. | Qian F, Wang S, Mitchell J, McGuffog L, et al. | [71] |
| 2018 | Computational analyses of obesity associated loci generated by genome-wide association studies. | Cheng M, Mei B, Zhou Q, Zhang M, et al. | [72] |
| 2018 | Associations of adult genetic risk scores for adiposity with childhood abdominal, liver and pericardial fat assessed by magnetic resonance imaging. | Monnereau C, Santos S, van der Lugt A, Jaddoe VWV, et al. | [73] |
| 2018 | A systematic analysis highlights multiple long non-coding RNAs associated with cardiometabolic disorders. | Ghanbari M, Peters MJ, de Vries PS, Boer CG, et al. | [74] |
| 2018 | Assessing the causal role of body mass index on cardiovascular health in young adults: Mendelian randomization and recall-by-genotype analyses. | Wade KH, Chiesa ST, Hughes AD, Chaturvedi N, et al. | [75] |
| 2018 | A Large Multiethnic Genome-Wide Association Study of Adult Body Mass Index Identifies Novel Loci. | Hoffmann TJ, Choquet H, Yin J, Banda Y, et al. | [76] |
| 2018 | A high throughput, functional screen of human Body Mass Index GWAS loci using tissue-specific RNAi Drosophila melanogaster crosses. | Baranski TJ, Kraja AT, Fink JL, Feitosa M, et al. | [77] |
| 2017 | Genome-wide meta-analysis of 241,258 adults accounting for smoking behaviour identifies novel loci for obesity traits. | Justice AE, Winkler TW, Feitosa MF, Graff M, et al. | [78] |
| 2017 | Gene-obesogenic environment interactions in the UK Biobank study. | Tyrrell J, Wood AR, Ames RM, Yaghootkar H, et al. | [79] |
| 2017 | Association of Body Mass Index With Cardiometabolic Disease in the UK Biobank: A Mendelian Randomization Study. | Lyall DM, Celis-Morales C, Ward J, Iliodromiti S, et al. | [80] |
| 2016 | Body mass index and psychiatric disorders: a Mendelian randomization study. | Hartwig FP, Bowden J, Loret de Mola C, Tovo-Rodrigues L, et al. | [81] |
| 2016 | Genetic Evidence for a Link Between Favorable Adiposity and Lower Risk of Type 2 Diabetes, Hypertension, and Heart Disease. | Yaghootkar H, Lotta LA, Tyrrell J, Smit RA, et al. | [82] |
| 2016 | Genetics of Obesity. | Srivastava A, Srivastava N, Mittal B. | [83] |
| 2016 | Height, body mass index, and socioeconomic status: mendelian randomisation study in UK Biobank. | Tyrrell J, Jones SE, Beaumont R, Astley CM, et al. | [84] |
| 2016 | Associations of genetic risk scores based on adult adiposity pathways with childhood growth and adiposity measures. | Monnereau C, Vogelezang S, Kruithof CJ, Jaddoe VW, et al. | [85] |
| 2016 | Genome-Wide Association Analyses in 128,266 Individuals Identifies New Morningness and Sleep Duration Loci. | Jones SE, Tyrrell J, Wood AR, Beaumont RN, et al. | [86] |
| 2016 | Obesity and Multiple Sclerosis: A Mendelian Randomization Study. | Mokry LE, Ross S, Timpson NJ, Sawcer S, et al. | [87] |
| 2015 | Physical activity, smoking, and genetic predisposition to obesity in people from Pakistan: the PROMIS study. | Ahmad S, Zhao W, Renstrom F, Rasheed A, et al. | [16] |
| 2015 | The Influence of Age and Sex on Genetic Associations with Adult Body Size and Shape: A Large-Scale Genome-Wide Interaction Study. | Winkler TW, Justice AE, Graff M, Barata L, et al. | [88] |
| 2015 | Genetic studies of body mass index yield new insights for obesity biology. | Locke AE, Kahali B, Berndt SI, Justice AE, et al. | [17] |
| 2015 | Obesity genetics in mouse and human: back and forth, and back again. | Yazdi FT, Clee SM, Meyre D. | [89] |
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| 2023 | Genome-wide association studies reveal shared genetic haplotypes of autoimmune rheumatic and endocrine diseases with psychiatric disorders. | Voskarides K, Giannopoulou N, Eid R, Parperis K, et al. | [90] |
| 2023 | Brain structure and allelic associations in Alzheimer’s disease. | Moon SW, Zhao L, Matloff W, Hobel S, et al. | [91] |
| 2022 | Cell type-specific histone acetylation profiling of Alzheimer’s disease subjects and integration with genetics. | Ramamurthy E, Welch G, Cheng J, Yuan Y, et al. | [18] |
| 2022 | The genetic architecture of Alzheimer disease risk in the Ohio and Indiana Amish. | Osterman MD, Song YE, Adams LD, Laux RA, et al. | [92] |
| 2022 | Identifying genetic markers enriched by brain imaging endophenotypes in Alzheimer’s disease. | Kim M, Wu R, Yao X, Saykin AJ, et al. | [93] |
| 2022 | Genome-wide association of polygenic risk extremes for Alzheimer’s disease in the UK Biobank. | Gouveia C, Gibbons E, Dehghani N, Eapen J, et al. | [94] |
| 2022 | Mining High-Level Imaging Genetic Associations via Clustering AD Candidate Variants with Similar Brain Association Patterns. | Wu R, Bao J, Kim M, Saykin AJ, et al. | [95] |
| 2021 | Genetic Variability in Molecular Pathways Implicated in Alzheimer’s Disease: A Comprehensive Review. | Vogrinc D, Goricar K, Dolzan V. | [96] |
| 2021 | A transcriptome-wide association study of Alzheimer’s disease using prediction models of relevant tissues identifies novel candidate susceptibility genes. | Sun Y, Zhu J, Zhou D, Canchi S, et al. | [97] |
| 2021 | A transcriptome-wide association study identifies novel blood-based gene biomarker candidates for Alzheimer’s disease risk. | Sun Y, Zhou D, Rahman MR, Zhu J, et al. | [98] |
| 2021 | Mapping the proteo-genomic convergence of human diseases. | Pietzner M, Wheeler E, Carrasco-Zanini J, Cortes A, et al. | [99] |
| 2020 | Identification of Novel Alzheimer’s Disease Loci Using Sex-Specific Family-Based Association Analysis of Whole-Genome Sequence Data. | Prokopenko D, Hecker J, Kirchner R, Chapman BA, et al. | [100] |
| 2020 | Interpretation of risk loci from genome-wide association studies of Alzheimer’s disease. | Andrews SJ, Fulton-Howard B, Goate A. | [101] |
| 2020 | Polygenic mediation analysis of Alzheimer’s disease implicated intermediate amyloid imaging phenotypes. | Yingxuan E, Yao X, Liu K, Risacher SL, et al. | [102] |
| 2020 | The association of clinical phenotypes to known AD/FTD genetic risk loci and their inter-relationship. | Li QS, Tian C, 23andMe Research Team, Hinds D, et al. | [103] |
| 2020 | Genomic mechanisms in Alzheimer’s disease. | Bertram L, Tanzi RE. | [104] |
| 2019 | Evaluation of the Common Molecular Basis in Alzheimer’s and Parkinson’s Diseases. | Rana P, Franco EF, Rao Y, Syed K, et al. | [105] |
| 2019 | Genome-wide meta-analysis identifies new loci and functional pathways influencing Alzheimer’s disease risk. | Jansen IE, Savage JE, Watanabe K, Bryois J, et al. | [106] |
| 2018 | Meta-analysis of genome-wide association studies for neuroticism in 449,484 individuals identifies novel genetic loci and pathways. | Nagel M, Jansen PR, Stringer S, Watanabe K, et al. | [19] |
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| 2022 | Association of Novel Loci With Keratoconus Susceptibility in a Multitrait Genome-Wide Association Study of the UK Biobank Database and Canadian Longitudinal Study on Aging. | He W, Han X, Ong JS, Hewitt AW, et al. | [107] |
| 2020 | Genome-Wide Association Study of VKORC1 and CYP2C9 on acenocoumarol dose, stroke recurrence and intracranial haemorrhage in Spain. | Cullell N, Carrera C, Muino E, Torres-Aguila NP, et al. | [108] |
| 2018 | Pharmacogenetic studies with oral anticoagulants. Genome-wide association studies in vitamin K antagonist and direct oral anticoagulants. | Cullell N, Carrera C, Muino E, Torres N, et al. | [109] |
| 2017 | Penetrance of Polygenic Obesity Susceptibility Loci across the Body Mass Index Distribution. | Abadi A, Alyass A, Robiou du Pont S, Bolker B, et al. | [110] |
| 2017 | Cohort-specific imputation of gene expression improves prediction of warfarin dose for African Americans. | Gottlieb A, Daneshjou R, DeGorter M, Bourgeois S, et al. | [111] |
| 2016 | Longitudinal relationships between glycemic status and body mass index in a multiethnic study: evidence from observational and genetic epidemiology. | Ishola AF, Gerstein HC, Engert JC, Mohan V, et al. | [112] |
| 2013 | Genomics of ADME gene expression: mapping expression quantitative trait loci relevant for absorption, distribution, metabolism and excretion of drugs in human liver. | Schroder A, Klein K, Winter S, Schwab M, et al. | [113] |
| 2012 | Liver expression quantitative trait loci: a foundation for pharmacogenomic research. | Glubb DM, Dholakia N, Innocenti F. | [114] |
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| 2022 | Mendelian Randomization Analysis Reveals No Causal Relationship Between Nonalcoholic Fatty Liver Disease and Severe COVID-19. | Li J, Tian A, Zhu H, Chen L, et al. | [15] |
| 2022 | Genomics and phenomics of body mass index reveals a complex disease network. | Huang J, Huffman JE, Huang Y, Do Valle I, et al. | [115] |
| 2022 | Pleiotropic genetic architecture and novel loci for C-reactive protein levels. | Koskeridis F, Evangelou E, Said S, Boyle JJ, et al. | [21] |
| 2019 | Mapping Genome Variants Sheds Light on Genetic and Phenotypic Differentiation in Chinese. | Guo L, Ye K. | [116] |
| 2019 | Whole Genome Analyses of Chinese Population and De Novo Assembly of A Northern Han Genome. | Du Z, Ma L, Qu H, Chen W, et al. | [20] |
| 2015 | New genetic loci link adipose and insulin biology to body fat distribution. | Shungin D, Winkler TW, Croteau-Chonka DC, Ferreira T, et al. | [117] |
| 2008 | Prostasin: a possible candidate gene for human hypertension. | Zhu H, Guo D, Li K, Yan W, et al. | [118] |
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| 2021 | A cross-population atlas of genetic associations for 220 human phenotypes. | Sakaue S, Kanai M, Tanigawa Y, Karjalainen J, et al. | [119] |
| 2020 | Lung Development Genes and Adult Lung Function. | Portas L, Pereira M, Shaheen SO, Wyss AB, et al. | [120] |
| 2020 | Polygenic Hyperlipidemias and Coronary Artery Disease Risk. | Ripatti P, Ramo JT, Mars NJ, Fu Y, et al. | [121] |
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| 2022 | A saturated map of common genetic variants associated with human height. | Yengo L, Vedantam S, Marouli E, Sidorenko J, et al. | [22] |
| 2016 | Association of Forced Vital Capacity with the Developmental Gene NCOR2. | Minelli C, Dean CH, Hind M, Alves AC, et al. | [122] |
| Year | Title | Author(s) | Reference |
|---|---|---|---|
| rs61320757 | |||
| 2021 | The genetic architecture of human cortical folding. | van der Meer D, Kaufmann T, Shadrin AA, Makowski C, et al. | [123] |
| rs368991827 | |||
| 2021 | Mapping the proteo-genomic convergence of human diseases. | Pietzner M, Wheeler E, Carrasco-Zanini J, Cortes A, et al. | [99] |
| rs748699921 | |||
| 2020 | Lysine acetyltransferase 8 is involved in cerebral development and syndromic intellectual disability. | Li L, Ghorbani M, Weisz-Hubshman M, Rousseau J, et al. | [23] |
References
- Rea, S.; Xouri, G.; Akhtar, A. Males Absent on the First (MOF): From Flies to Humans. Oncogene 2007, 26, 5385–5394. [Google Scholar] [CrossRef]
- Schunter, S.; Villa, R.; Flynn, V.; Heidelberger, J.B.; Classen, A.K.; Beli, P.; Becker, P.B. Ubiquitylation of the Acetyltransferase MOF in Drosophila Melanogaster. PLoS ONE 2017, 12, e0177408. [Google Scholar] [CrossRef]
- Taipale, M.; Rea, S.; Richter, K.; Vilar, A.; Lichter, P.; Imhof, A.; Akhtar, A. HMOF Histone Acetyltransferase Is Required for Histone H4 Lysine 16 Acetylation in Mammalian Cells. Mol. Cell. Biol. 2005, 25, 6798–6810. [Google Scholar] [CrossRef]
- Thomas, T.; Voss, A.K. The Diverse Biological Roles of MYST Histone Acetyltransferase Family Proteins. Cell Cycle 2007, 6, 696–704. [Google Scholar] [CrossRef]
- Burrell, J.A.; Stephens, J.M. KAT8, Lysine Acetyltransferase 8, Is Required for Adipocyte Differentiation in Vitro. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 166103. [Google Scholar] [CrossRef]
- Su, J.; Wang, F.; Cai, Y.; Jin, J. The Functional Analysis of Histone Acetyltransferase MOF in Tumorigenesis. Int. J. Mol. Sci. 2016, 17, 99. [Google Scholar] [CrossRef]
- Kiran, S.; Kumar, V.; Kumar, S.; Price, R.L.; Singh, U.P. Adipocyte, Immune Cells, and Mirna Crosstalk: A Novel Regulator of Metabolic Dysfunction and Obesity. Cells 2021, 10, 1004. [Google Scholar] [CrossRef]
- Ling, C.; Rönn, T. Epigenetics in Human Obesity and Type 2 Diabetes. Cell Metab. 2019, 29, 1028–1044. [Google Scholar] [CrossRef]
- Gupta, A.; Guerin-Peyrou, T.G.; Sharma, G.G.; Park, C.; Agarwal, M.; Ganju, R.K.; Pandita, S.; Choi, K.; Sukumar, S.; Pandita, R.K.; et al. The Mammalian Ortholog of Drosophila MOF That Acetylates Histone H4 Lysine 16 Is Essential for Embryogenesis and Oncogenesis. Mol. Cell. Biol. 2008, 28, 397–409. [Google Scholar] [CrossRef]
- Han, Z.; Wu, H.; Kim, S.; Yang, X.; Li, Q.; Huang, H.; Cai, H.; Bartlett, M.G.; Dong, A.; Zeng, H.; et al. Revealing the Protein Propionylation Activity of the Histone Acetyltransferase MOF (Males Absent on the First). J. Biol. Chem. 2018, 293, 3410–3420. [Google Scholar] [CrossRef]
- Liu, X.; Wei, W.; Liu, Y.; Yang, X.; Wu, J.; Zhang, Y.; Zhang, Q.; Shi, T.; Du, J.X.; Zhao, Y.; et al. MOF as an Evolutionarily Conserved Histone Crotonyltransferase and Transcriptional Activation by Histone Acetyltransferase-Deficient and Crotonyltransferase-Competent CBP/P300. Cell Discov. 2017, 3, 17016. [Google Scholar] [CrossRef]
- Sapountzi, V.; Côté, J. MYST-Family Histone Acetyltransferases: Beyond Chromatin. Cell. Mol. Life Sci. 2011, 68, 1147–1156. [Google Scholar] [CrossRef]
- Sykes, S.M.; Mellert, H.S.; Holbert, M.A.; Li, K.; Marmorstein, R.; Lane, W.S.; Mcmahon, S.B. Acetylation of the P53 DNA Binding Domain Regulates Apoptosis Induction. Mol. Cell 2006, 24, 841–851. [Google Scholar] [CrossRef]
- Li, X.; Wu, L.; Corsa, C.A.S.; Kunkel, S.; Dou, Y. Two Mammalian MOF Complexes Regulate Transcription Activation by Distinct Mechanisms. Mol. Cell 2009, 36, 290–301. [Google Scholar] [CrossRef]
- Li, J.; Tian, A.; Zhu, H.; Chen, L.; Wen, J.; Liu, W.; Chen, P. Mendelian Randomization Analysis Reveals No Causal Relationship Between Nonalcoholic Fatty Liver Disease and Severe COVID-19. Clin. Gastroenterol. Hepatol. 2022, 20, 1553–1560.e78. [Google Scholar] [CrossRef]
- Ahmad, S.; Zhao, W.; Renström, F.; Rasheed, A.; Samuel, M.; Zaidi, M.; Shah, N.; Mallick, N.H.; Zaman, K.S.; Ishaq, M.; et al. Physical Activity, Smoking, and Genetic Predisposition to Obesity in People from Pakistan: The PROMIS Study. BMC Med. Genet. 2015, 16, 114. [Google Scholar] [CrossRef]
- Locke, A.E.; Kahali, B.; Berndt, S.I.; Justice, A.E.; Pers, T.H.; Day, F.R.; Powell, C.; Vedantam, S.; Buchkovich, M.L.; Yang, J.; et al. Genetic Studies of Body Mass Index Yield New Insights for Obesity Biology. Nature 2015, 518, 197–206. [Google Scholar] [CrossRef]
- Ramamurthy, E.; Welch, G.; Cheng, J.; Yuan, Y.; Gunsalus, L.; Bennett, D.A.; Tsai, L.H.; Pfenning, A.R. Cell Type-Specific Histone Acetylation Profiling of Alzheimer’s Disease Subjects and Integration with Genetics. Front. Mol. Neurosci. 2023, 15, 948456. [Google Scholar] [CrossRef]
- Nagel, M.; Jansen, P.R.; Stringer, S.; Watanabe, K.; De Leeuw, C.A.; Bryois, J.; Savage, J.E.; Hammerschlag, A.R.; Skene, N.G.; Muñoz-Manchado, A.B.; et al. Meta-Analysis of Genome-Wide Association Studies for Neuroticism in 449,484 Individuals Identifies Novel Genetic Loci and Pathways. Nat. Genet. 2018, 50, 920–927. [Google Scholar] [CrossRef]
- Du, Z.; Ma, L.; Qu, H.; Chen, W.; Zhang, B.; Lu, X.; Zhai, W.; Sheng, X.; Sun, Y.; Li, W.; et al. Whole Genome Analyses of Chinese Population and De Novo Assembly of a Northern Han Genome. Genom. Proteom. Bioinform. 2019, 17, 229–247. [Google Scholar] [CrossRef]
- Koskeridis, F.; Evangelou, E.; Said, S.; Boyle, J.J.; Elliott, P.; Dehghan, A.; Tzoulaki, I. Pleiotropic Genetic Architecture and Novel Loci for C-Reactive Protein Levels. Nat. Commun. 2022, 13, 6939. [Google Scholar] [CrossRef]
- Yengo, L.; Vedantam, S.; Marouli, E.; Sidorenko, J.; Bartell, E.; Sakaue, S.; Graff, M.; Eliasen, A.U.; Jiang, Y.; Raghavan, S.; et al. A Saturated Map of Common Genetic Variants Associated with Human Height. Nature 2022, 610, 704–712. [Google Scholar] [CrossRef]
- Li, L.; Ghorbani, M.; Weisz-Hubshman, M.; Rousseau, J.; Thiffault, I.; Schnur, R.E.; Breen, C.; Oegema, R.; Weiss, M.M.M.; Waisfisz, Q.; et al. Lysine Acetyltransferase 8 Is Involved in Cerebral Development and Syndromic Intellectual Disability. J. Clin. Investig. 2020, 130, 1431–1445. [Google Scholar] [CrossRef]
- Kadlec, J.; Hallacli, E.; Lipp, M.; Holz, H.; Sanchez-Weatherby, J.; Cusack, S.; Akhtar, A. Structural Basis for MOF and MSL3 Recruitment into the Dosage Compensation Complex by MSL1. Nat. Struct. Mol. Biol. 2011, 18, 142–150. [Google Scholar] [CrossRef]
- Villa, R.; Forné, I.; Müller, M.; Imhof, A.; Straub, T.; Becker, P.B. MSL2 Combines Sensor and Effector Functions in Homeostatic Control of the Drosophila Dosage Compensation Machinery. Mol. Cell 2012, 48, 647–654. [Google Scholar] [CrossRef]
- Kim, D.; Blus, B.J.; Chandra, V.; Huang, P.; Rastinejad, F.; Khorasanizadeh, S. Corecognition of DNA and a Methylated Histone Tail by the MSL3 Chromodomain. Nat. Struct. Mol. Biol. 2010, 17, 1027–1029. [Google Scholar] [CrossRef]
- Keller, C.I.; Akhtar, A. The MSL Complex: Juggling RNA-Protein Interactions for Dosage Compensation and Beyond. Curr. Opin. Genet. Dev. 2015, 31, 1–11. [Google Scholar] [CrossRef]
- Radzisheuskaya, A.; Shliaha, P.V.; Grinev, V.V.; Shlyueva, D.; Damhofer, H.; Koche, R.; Gorshkov, V.; Kovalchuk, S.; Zhan, Y.; Rodriguez, K.L.; et al. Complex-Dependent Histone Acetyltransferase Activity of KAT8 Determines Its Role in Transcription and Cellular Homeostasis. Mol. Cell 2021, 81, 1749–1765.e8. [Google Scholar] [CrossRef]
- Smith, E.R.; Cayrou, C.; Huang, R.; Lane, W.S.; Côté, J.; Lucchesi, J.C. A Human Protein Complex Homologous to the Drosophila MSL Complex Is Responsible for the Majority of Histone H4 Acetylation at Lysine 16. Mol. Cell. Biol. 2005, 25, 9175–9188. [Google Scholar] [CrossRef]
- Morra, R.; Yokoyama, R.; Ling, H.; Lucchesi, J.C. Role of the ATPase/Helicase Maleless (MLE) in the Assembly, Targeting, Spreading and Function of the Male-Specific Lethal (MSL) Complex of Drosophila. Epigenet. Chromatin 2011, 4, 6–13. [Google Scholar] [CrossRef]
- Sheikh, B.N.; Guhathakurta, S.; Akhtar, A. The Non-specific Lethal (NSL) Complex at the Crossroads of Transcriptional Control and Cellular Homeostasis. EMBO Rep. 2019, 20, e47630. [Google Scholar] [CrossRef] [PubMed]
- Meunier, S.; Shvedunova, M.; Van Nguyen, N.; Avila, L.; Vernos, I.; Akhtar, A. An Epigenetic Regulator Emerges as Microtubule Minus-End Binding and Stabilizing Factor in Mitosis. Nat. Commun. 2015, 6, 7889. [Google Scholar] [CrossRef]
- Klein, B.J.; Wang, X.; Cui, G.; Yuan, C.; Botuyan, M.V.; Lin, K.; Lu, Y.; Wang, X.; Zhao, Y.; Bruns, C.J.; et al. PHF20 Readers Link Methylation of Histone H3K4 and P53 with H4K16 Acetylation. Cell Rep. 2016, 17, 1158–1170. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Jiang, X.; Jiang, H.; Gao, Q.; Wang, F.; Fan, S.; Khan, T.; Jabeen, N.; Khan, M.; Ali, A.; et al. Histone Acetyltransferase KAT8 Is Essential for Mouse Oocyte Development by Regulating Reactive Oxygen Species Levels. Development 2017, 144, 2165–2174. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, B.N.; Bechtel-Walz, W.; Lucci, J.; Karpiuk, O.; Hild, I.; Hartleben, B.; Vornweg, J.; Helmstädter, M.; Sahyoun, A.H.; Bhardwaj, V.; et al. MOF Maintains Transcriptional Programs Regulating Cellular Stress Response. Oncogene 2016, 35, 2698–2710. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.C.; Mühlpfordt, F.; Vaquerizas, J.M.; Raja, S.J.; Holz, H.; Luscombe, N.M.; Manke, T.; Akhtar, A. The NSL Complex Regulates Housekeeping Genes in Drosophila. PLoS Genet. 2012, 8, e1002736. [Google Scholar] [CrossRef] [PubMed]
- Karoutas, A.; Szymanski, W.; Rausch, T.; Guhathakurta, S.; Rog-Zielinska, E.A.; Peyronnet, R.; Seyfferth, J.; Chen, H.R.; de Leeuw, R.; Herquel, B.; et al. The NSL Complex Maintains Nuclear Architecture Stability via Lamin A/C Acetylation. Nat. Cell Biol. 2019, 21, 1248–1260. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Klionsky, D.J. BRD4 Is a Newly Characterized Transcriptional Regulator That Represses Autophagy and Lysosomal Function. Autophagy 2017, 13, 1801–1803. [Google Scholar] [CrossRef]
- Koolen, D.A.; Kramer, J.M.; Neveling, K.; Nillesen, W.M.; Moore-Barton, H.L.; Elmslie, F.V.; Toutain, A.; Amiel, J.; Malan, V.; Tsai, A.C.H.; et al. Mutations in the Chromatin Modifier Gene KANSL1 Cause the 17q21.31 Microdeletion Syndrome. Nat. Genet. 2012, 44, 639–641. [Google Scholar] [CrossRef]
- Koolen, D.A.; Sharp, A.J.; Hurst, J.A.; Firth, H.V.; Knight, S.J.L.; Goldenberg, A.; Saugier-Veber, P.; Pfundt, R.; Vissers, L.E.L.M.; Destrée, A.; et al. Clinical and Molecular Delineation of the 17q21.31 Microdeletion Syndrome. J. Med. Genet. 2008, 45, 710–720. [Google Scholar] [CrossRef]
- Zhao, X.; Su, J.; Wang, F.; Liu, D.; Ding, J.; Yang, Y.; Conaway, J.W.; Conaway, R.C.; Cao, L.; Wu, D.; et al. Crosstalk between NSL Histone Acetyltransferase and MLL/SET Complexes: NSL Complex Functions in Promoting Histone H3K4 Di-Methylation Activity by MLL/SET Complexes. PLoS Genet. 2013, 9, e1003940. [Google Scholar] [CrossRef] [PubMed]
- Sha, L.; Ayoub, A.; Cho, U.S.; Dou, Y. Insights on the Regulation of the MLL/SET1 Family Histone Methyltransferases. Biochim. Biophys. Acta Gene Regul. Mech. 2020, 1863, 194561. [Google Scholar] [CrossRef]
- Chelmicki, T.; Dündar, F.; Turley, M.J.; Khanam, T.; Aktas, T.; Ramirez, F.; Gendrel, A.V.; Wright, P.R.; Videm, P.; Backofen, R.; et al. MOF-Associated Complexes Ensure Stem Cell Identity and Xist Repression. eLife 2014, 3, e02024. [Google Scholar] [CrossRef] [PubMed]
- Ravens, S.; Fournier, M.; Ye, T.; Stierle, M.; Dembele, D.; Chavant, V.; Tora, L. Mof-Associated Complexes Have Overlapping and Unique Roles in Regulating Pluripotency in Embryonic Stem Cells and during Differentiation. eLife 2014, 3, e02104. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Zhang, Y.; Liu, Y.; Chen, J.; Zong, C.; Yu, C.; Cui, S.; Gao, W.; Qin, D.; Sun, W.; et al. Signal Transducer and Activator of Transcription 5B (STAT5B) Modulates Adipocyte Differentiation via MOF. Cell Signal 2015, 27, 2434–2443. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, D.; Chen, B.; Yang, Y.; Zhu, H.; Li, D.; Liu, K.; Zhu, L.; Liu, H.; Li, M.; et al. The Histone Acetyltransferase Mof Regulates Runx2 and Osterix for Osteoblast Differentiation. Cell Tissue Res. 2023, 393, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Hunt, C.R.; Pandita, R.K.; Pae, J.; Komal, K.; Singh, M.; Shay, J.W.; Kumar, R.; Ariizumi, K.; Horikoshi, N.; et al. T-Cell-Specific Deletion of Mof Blocks Their Differentiation and Results in Genomic Instability in Mice. Mutagenesis 2013, 28, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Hong Tsang, T.; Wiese, M.; Helmstädter, M.; Stehle, T.; Seyfferth, J.; Shvedunova, M.; Holz, H.; Walz, G.; Akhtar, A. Transcriptional Regulation by the NSL Complex Enables Diversification of IFT Functions in Ciliated versus Nonciliated Cells. Sci. Adv. 2023, 9, eadh5598. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.B.; Wang, M.; Gao, S.; Shaikh, A.S.; Chen, J.; Li, X.Z. The Histone Acetyltranseferase KAT8 Regulates Cell Differentiation by Suppression of MN1 in AML. Br. J. Haematol. 2018, 182, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Füllgrabe, J.; Lynch-Day, M.A.; Heldring, N.; Li, W.; Struijk, R.B.; Ma, Q.; Hermanson, O.; Rosenfeld, M.G.; Klionsky, D.J.; Joseph, B. The Histone H4 Lysine 16 Acetyltransferase HMOF Regulates the Outcome of Autophagy. Nature 2013, 500, 468–471. [Google Scholar] [CrossRef]
- Sharma, G.G.; So, S.; Gupta, A.; Kumar, R.; Cayrou, C.; Avvakumov, N.; Bhadra, U.; Pandita, R.K.; Porteus, M.H.; Chen, D.J.; et al. MOF and Histone H4 Acetylation at Lysine 16 Are Critical for DNA Damage Response and Double-Strand Break Repair. Mol. Cell Biol. 2010, 30, 3582–3595. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Seyfferth, J.; Lucci, J.; Gilsbach, R.; Preissl, S.; Böttinger, L.; Mårtensson, C.U.; Panhale, A.; Stehle, T.; Kretz, O.; et al. MOF Acetyl Transferase Regulates Transcription and Respiration in Mitochondria. Cell 2016, 167, 722–738.e23. [Google Scholar] [CrossRef] [PubMed]
- Guhathakurta, S.; Erdogdu, N.U.; Hoffmann, J.J.; Grzadzielewska, I.; Schendzielorz, A.; Seyfferth, J.; Mårtensson, C.U.; Corrado, M.; Karoutas, A.; Warscheid, B.; et al. COX17 Acetylation via MOF–KANSL Complex Promotes Mitochondrial Integrity and Function. Nat. Metab. 2023, 5, 1931. [Google Scholar] [CrossRef] [PubMed]
- De Talhouët, C.; Gallego, N.E.; Soutar, M.; O’callaghan, B.; Plun-Favreau, H. KAT8 Compound Inhibition Inhibits the Initial Steps of PINK1-Dependant Mitophagy. bioRxiv 2023. [Google Scholar] [CrossRef]
- Chen, C.; Liu, T.; Tang, Y.; Luo, G.; Liang, G.; He, W. Epigenetic Regulation of Macrophage Polarization in Wound Healing. Burns Trauma. 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, K.A.; Joshi, A.; Carson, W.F.; Schaller, M.; Allen, R.; Mukerjee, S.; Kittan, N.; Feldman, E.L.; Henke, P.K.; Hogaboam, C.; et al. Epigenetic Changes in Bone Marrow Progenitor Cells Influence the Inflammatory Phenotype and Alter Wound Healing in Type 2 Diabetes. Diabetes 2015, 64, 1420–1430. [Google Scholar] [CrossRef]
- Ohradanova-Repic, A.; Ericsson, A.C.; Yang, M.; Zhang, C. Function of Macrophages in Disease: Current Understanding on Molecular Mechanisms. Front. Immunol. 2021, 1, 620510. [Google Scholar] [CrossRef]
- Koh, T.J.; DiPietro, L.A. Inflammation and Wound Healing: The Role of the Macrophage. Expert. Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef]
- Hamidzadeh, K.; Christensen, S.M.; Dalby, E.; Chandrasekaran, P.; Mosser, D.M. Macrophages and the Recovery from Acute and Chronic Inflammation. Annu. Rev. Physiol. 2017, 79, 567–592. [Google Scholar] [CrossRef]
- den Dekker, A.D.; Davis, F.M.; Joshi, A.D.; Wolf, S.J.; Allen, R.; Lipinski, J.; Nguyen, B.; Kirma, J.; Nycz, D.; Bermick, J.; et al. TNF-α Regulates Diabetic Macrophage Function through the Histone Acetyltransferase MOF. JCI Insight 2020, 5, e132306. [Google Scholar] [CrossRef]
- Liu, Y.; Du, J.; Liu, X.; Wang, L.; Han, Y.; Huang, C.; Liang, R.; Zheng, F.; Shi, G.; Li, B. MG149 Inhibits Histone Acetyltransferase KAT8-Mediated IL-33 Acetylation to Alleviate Allergic Airway Inflammation and Airway Hyperresponsiveness. Signal Transduct. Target. Ther. 2021, 6. [Google Scholar] [CrossRef] [PubMed]
- van den Bosch, T.; Leus, N.G.J.; Wapenaar, H.; Boichenko, A.; Hermans, J.; Bischoff, R.; Haisma, H.J.; Dekker, F.J. A 6-Alkylsalicylate Histone Acetyltransferase Inhibitor Inhibits Histone Acetylation and pro-Inflammatory Gene Expression in Murine Precision-Cut Lung Slices. Pulm. Pharmacol. Ther. 2017, 44, 88. [Google Scholar] [CrossRef] [PubMed]
- Mikula, M.; Majewska, A.; Ledwon, J.K.; Dzwonek, A.; Ostrowski, J. Obesity Increases Histone H3 Lysine 9 and 18 Acetylation at Tnfa and Ccl2 Genes in Mouse Liver. Int. J. Mol. Med. 2014, 34, 1647–1654. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Li, Y.; Zheng, Q.; Ding, M.; Zhou, H.; Li, X.; Association, C.P. Epigenetic Modification in Liver Fibrosis: Promising Therapeutic Direction with Significant Challenges Ahead. Acta Pharm. Sin. B 2024, 14, 1009–1029. [Google Scholar] [CrossRef] [PubMed]
- Huai, W.; Liu, X.; Wang, C.; Zhang, Y.; Chen, X.; Chen, X.; Xu, S.; Thomas, T.; Li, N.; Cao, X. KAT8 Selectively Inhibits Antiviral Immunity by Acetylating IRF3. J. Exp. Med. 2019, 216, 772–785. [Google Scholar] [CrossRef] [PubMed]
- Vourdoumpa, A.; Paltoglou, G.; Charmandari, E. The Genetic Basis of Childhood Obesity: A Systematic Review. Nutrients 2023, 15, 1416. [Google Scholar] [CrossRef] [PubMed]
- Casanova, F.; O’loughlin, J.; Lewis, C.; Frayling, T.M.; Wood, A.R.; Tyrrell, J. Simulated Distributions from Negative Experiments Highlight the Importance of the Body Mass Index Distribution in Explaining Depression-Body Mass Index Genetic Risk Score Interactions. Int. J. Epidemiol. 2022, 51, 1581–1592. [Google Scholar] [CrossRef] [PubMed]
- Heitkamp, M.; Siegrist, M.; Molnos, S.; Brandmaier, S.; Wahl, S.; Langhof, H.; Grallert, H.; Halle, M. Obesity Genes and Weight Loss during Lifestyle Intervention in Children with Obesity. JAMA Pediatr. 2021, 175, e205142. [Google Scholar] [CrossRef] [PubMed]
- Mason, K.E.; Palla, L.; Pearce, N.; Phelan, J.; Cummins, S. Genetic Risk of Obesity as a Modifier of Associations between Neighbourhood Environment and Body Mass Index: An Observational Study of 335 046 UK Biobank Participants. BMJ Nutr. Prev. Health 2020, 3, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Brower, M.A.; Hai, Y.; Jones, M.R.; Guo, X.; Chen, Y.D.I.; Rotter, J.I.; Krauss, R.M.; Legro, R.S.; Azziz, R.; Goodarzi, M.O. Bidirectional Mendelian Randomization to Explore the Causal Relationships between Body Mass Index and Polycystic Ovary Syndrome. Human. Reproduction 2019, 34, 127–136. [Google Scholar] [CrossRef]
- Qian, F.; Wang, S.; Mitchell, J.; McGuffog, L.; Barrowdale, D.; Leslie, G.; Oosterwijk, J.C.; Chung, W.K.; Gareth Evans, D.; Engel, C.; et al. Height and Body Mass Index as Modifiers of Breast Cancer Risk in BRCA1/2 Mutation Carriers: A Mendelian Randomization Study. J. Natl. Cancer Inst. 2019, 111, 350–364. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Mei, B.; Zhou, Q.; Zhang, M.; Huang, H.; Han, L.; Huang, Q. Computational Analyses of Obesity Associated Loci Generated by Genome-Wide Association Studies. PLoS ONE 2018, 13, e0199987. [Google Scholar] [CrossRef] [PubMed]
- Monnereau, C.; Vogelezang, S.; Kruithof, C.J.; Jaddoe, V.W.V.; Felix, J.F. Associations of Genetic Risk Scores Based on Adult Adiposity Pathways with Childhood Growth and Adiposity Measures. BMC Genet. 2016, 17, 120. [Google Scholar] [CrossRef] [PubMed]
- Ghanbari, M.; Peters, M.J.; De Vries, P.S.; Boer, C.G.; Van Rooij, J.G.J.; Lee, Y.C.; Kumar, V.; Uitterlinden, A.G.; Ikram, M.A.; Wijmenga, C.; et al. A Systematic Analysis Highlights Multiple Long Non-Coding RNAs Associated with Cardiometabolic Disorders. J. Hum. Genet. 2018, 63, 431–446. [Google Scholar] [CrossRef] [PubMed]
- Wade, K.H.; Chiesa, S.T.; Hughes, A.D.; Chaturvedi, N.; Charakida, M.; Rapala, A.; Muthurangu, V.; Khan, T.; Finer, N.; Sattar, N.; et al. Assessing the Causal Role of Body Mass Index on Cardiovascular Health in Young Adults: Mendelian Randomization and Recall-by-Genotype Analyses. Circulation 2018, 138, 2187–2201. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, T.J.; Choquet, H.; Yin, J.; Banda, Y.; Kvale, M.N.; Glymour, M.; Schaefer, C.; Risch, N.; Jorgenson, E. A Large Multiethnic Genome-Wide Association Study of Adult Body Mass Index Identifies Novel Loci. Genetics 2018, 210, 499–515. [Google Scholar] [CrossRef] [PubMed]
- Baranski, T.J.; Kraja, A.T.; Fink, J.L.; Feitosa, M.; Lenzini, P.A.; Borecki, I.B.; Liu, C.T.; Cupples, L.A.; North, K.E.; Province, M.A. A High Throughput, Functional Screen of Human Body Mass Index GWAS Loci Using Tissue-Specific RNAi Drosophila Melanogaster Crosses. PLoS Genet. 2018, 14, e1007222. [Google Scholar] [CrossRef] [PubMed]
- Justice, A.E.; Winkler, T.W.; Feitosa, M.F.; Graff, M.; Fisher, V.A.; Young, K.; Barata, L.; Deng, X.; Czajkowski, J.; Hadley, D.; et al. Genome-Wide Meta-Analysis of 241,258 Adults Accounting for Smoking Behaviour Identifies Novel Loci for Obesity Traits. Nat. Commun. 2017, 8, 14977. [Google Scholar] [CrossRef]
- Tyrrell, J.; Wood, A.R.; Ames, R.M.; Yaghootkar, H.; Beaumont, R.N.; Jones, S.E.; Tuke, M.A.; Ruth, K.S.; Freathy, R.M.; Smith, G.D.; et al. Gene–Obesogenic Environment Interactions in the UK Biobank Study. Int. J. Epidemiol. 2017, 46, 559–575. [Google Scholar] [CrossRef]
- Lyall, D.M.; Celis-Morales, C.; Ward, J.; Iliodromiti, S.; Anderson, J.J.; Gill, J.M.R.; Smith, D.J.; Ntuk, U.E.; Mackay, D.F.; Holmes, M.V.; et al. Association of Body Mass Index with Cardiometabolic Disease in the UK Biobank: A Mendelian Randomization Study. JAMA Cardiol. 2017, 2, 882–889. [Google Scholar] [CrossRef]
- Hartwig, F.P.; Bowden, J.; Loret De Mola, C.; Tovo-Rodrigues, L.; Davey Smith, G.; Horta, B.L. Body Mass Index and Psychiatric Disorders: A Mendelian Randomization Study. Sci. Rep. 2016, 6, 32730. [Google Scholar] [CrossRef]
- Yaghootkar, H.; Lotta, L.A.; Tyrrell, J.; Smit, R.A.J.; Jones, S.E.; Donnelly, L.; Beaumont, R.; Campbell, A.; Tuke, M.A.; Hayward, C.; et al. Genetic Evidence for a Link between Favorable Adiposity and Lower Risk of Type 2 Diabetes, Hypertension, and Heart Disease. Diabetes 2016, 65, 2448–2460. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Srivastava, N.; Mittal, B. Genetics of Obesity. Indian J. Clin. Biochem. 2016, 31, 361–371. [Google Scholar] [CrossRef]
- Tyrrell, J.; Jones, S.E.; Beaumont, R.; Astley, C.M.; Lovell, R.; Yaghootkar, H.; Tuke, M.; Ruth, K.S.; Freathy, R.M.; Hirschhorn, J.N.; et al. Height, Body Mass Index, and Socioeconomic Status: Mendelian Randomisation Study in UK Biobank. BMJ 2016, 352, i582. [Google Scholar] [CrossRef] [PubMed]
- Monnereau, C.; Santos, S.; Van Der Lugt, A.; Jaddoe, V.W.V.; Felix, J.F. Associations of Adult Genetic Risk Scores for Adiposity with Childhood Abdominal, Liver and Pericardial Fat Assessed by Magnetic Resonance Imaging. Int. J. Obes. 2018, 42, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.E.; Tyrrell, J.; Wood, A.R.; Beaumont, R.N.; Ruth, K.S.; Tuke, M.A.; Yaghootkar, H.; Hu, Y.; Teder-Laving, M.; Hayward, C.; et al. Genome-Wide Association Analyses in 128,266 Individuals Identifies New Morningness and Sleep Duration Loci. PLoS Genet. 2016, 12, e1006125. [Google Scholar] [CrossRef]
- Mokry, L.E.; Ross, S.; Timpson, N.J.; Sawcer, S.; Davey Smith, G.; Richards, J.B. Obesity and Multiple Sclerosis: A Mendelian Randomization Study. PLoS Med. 2016, 13, e1002053. [Google Scholar] [CrossRef]
- Winkler, T.W.; Justice, A.E.; Graff, M.; Barata, L.; Feitosa, M.F.; Chu, S.; Czajkowski, J.; Esko, T.; Fall, T.; Kilpeläinen, T.O.; et al. The Influence of Age and Sex on Genetic Associations with Adult Body Size and Shape: A Large-Scale Genome-Wide Interaction Study. PLoS Genet. 2015, 11, e1005378, Erratum in PLoS Genet. 2016, 12, e1006166. [Google Scholar] [CrossRef] [PubMed]
- Yazdi, F.T.; Clee, S.M.; Meyre, D. Obesity Genetics in Mouse and Human: Back and Forth, and Back Again. PeerJ 2015, 3, e856. [Google Scholar] [CrossRef]
- Voskarides, K.; Giannopoulou, N.; Eid, R.; Parperis, K.; Chatzittofis, A. Genome-Wide Association Studies Reveal Shared Genetic Haplotypes of Autoimmune Rheumatic and Endocrine Diseases with Psychiatric Disorders. Brain Behav. 2023, 13, e2955. [Google Scholar] [CrossRef]
- Moon, S.W.; Zhao, L.; Matloff, W.; Hobel, S.; Berger, R.; Kwon, D.; Kim, J.; Toga, A.W.; Dinov, I.D. Brain Structure and Allelic Associations in Alzheimer’s Disease. CNS Neurosci. Ther. 2023, 29, 1034–1048. [Google Scholar] [CrossRef] [PubMed]
- Osterman, M.D.; Song, Y.E.; Adams, L.D.; Laux, R.A.; Caywood, L.J.; Prough, M.B.; Clouse, J.E.; Herington, S.D.; Slifer, S.H.; Lynn, A.; et al. The Genetic Architecture of Alzheimer Disease Risk in the Ohio and Indiana Amish. Hum. Genet. Genom. Adv. 2022, 3, 100114. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Wu, R.; Yao, X.; Saykin, A.J.; Moore, J.H.; Shen, L. Identifying Genetic Markers Enriched by Brain Imaging Endophenotypes in Alzheimer’s Disease. BMC Med. Genom. 2022, 15, 168. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, C.; Gibbons, E.; Dehghani, N.; Eapen, J.; Guerreiro, R.; Bras, J. Genome-Wide Association of Polygenic Risk Extremes for Alzheimer’s Disease in the UK Biobank. Sci. Rep. 2022, 12, 8404. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Bao, J.; Kim, M.; Saykin, A.J.; Moore, J.H.; Shen, L. Mining High-Level Imaging Genetic Associations via Clustering AD Candidate Variants with Similar Brain Association Patterns. Genes. 2022, 13, 1520. [Google Scholar] [CrossRef] [PubMed]
- Vogrinc, D.; Goričar, K.; Dolžan, V. Genetic Variability in Molecular Pathways Implicated in Alzheimer’s Disease: A Comprehensive Review. Front. Aging Neurosci. 2021, 13, 646901. [Google Scholar] [CrossRef]
- Sun, Y.; Zhu, J.; Zhou, D.; Canchi, S.; Wu, C.; Cox, N.J.; Rissman, R.A.; Gamazon, E.R.; Wu, L. A Transcriptome-Wide Association Study of Alzheimer’s Disease Using Prediction Models of Relevant Tissues Identifies Novel Candidate Susceptibility Genes. Genome Med. 2021, 13, 141. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, D.; Rahman, M.R.; Zhu, J.; Ghoneim, D.; Cox, N.J.; Beach, T.G.; Wu, C.; Gamazon, E.R.; Wu, L. A Transcriptome-Wide Association Study Identifies Novel Blood-Based Gene Biomarker Candidates for Alzheimer’s Disease Risk. Hum. Mol. Genet. 2022, 31, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Pietzner, M.; Wheeler, E.; Carrasco-Zanini, J.; Cortes, A.; Koprulu, M.; Wörheide, M.A.; Oerton, E.; Cook, J.; Stewart, I.D.; Kerrison, N.D.; et al. Mapping the Proteo-Genomic Convergence of Human Diseases. Science 2021, 374, eabj1541. [Google Scholar] [CrossRef]
- Prokopenko, D.; Hecker, J.; Kirchner, R.; Chapman, B.A.; Hoffman, O.; Mullin, K.; Hide, W.; Bertram, L.; Laird, N.; DeMeo, D.L.; et al. Identification of Novel Alzheimer’s Disease Loci Using Sex-Specific Family-Based Association Analysis of Whole-Genome Sequence Data. Sci. Rep. 2020, 10, 5029. [Google Scholar] [CrossRef]
- Andrews, S.J.; Fulton-Howard, B.; Goate, A. Interpretation of Risk Loci from Genome-Wide Association Studies of Alzheimer’s Disease. Lancet Neurol. 2020, 19, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Yingxuan, E.; Yao, X.; Liu, K.; Risacher, S.L.; Saykin, A.J.; Long, Q.; Zhao, Y.; Shen, L.; ADNI. Polygenic Mediation Analysis of Alzheimer’s Disease Implicated Intermediate Amyloid Imaging Phenotypes. AMIA Annu. Symp. Proc. 2021, 2020, 422–431. [Google Scholar] [PubMed]
- Li, Q.S.; Tian, C.; Hinds, D.; Seabrook, G.R. The Association of Clinical Phenotypes to Known AD/FTD Genetic Risk Loci and Their Interrelationship. PLoS ONE 2020, 15, e0241552. [Google Scholar] [CrossRef] [PubMed]
- Bertram, L.; Tanzi, R.E. Genomic Mechanisms in Alzheimer’s Disease. Brain Pathology 2020, 30, 966–977. [Google Scholar] [CrossRef] [PubMed]
- Rana, P.; Franco, E.F.; Rao, Y.; Syed, K.; Barh, D.; Azevedo, V.; Ramos, R.T.J.; Ghosh, P. Evaluation of the Common Molecular Basis in Alzheimer’s and Parkinson’s Diseases. Int. J. Mol. Sci. 2019, 20, 3730. [Google Scholar] [CrossRef] [PubMed]
- Jansen, I.E.; Savage, J.E.; Watanabe, K.; Bryois, J.; Williams, D.M.; Steinberg, S.; Sealock, J.; Karlsson, I.K.; Hägg, S.; Athanasiu, L.; et al. Genome-Wide Meta-Analysis Identifies New Loci and Functional Pathways Influencing Alzheimer’s Disease Risk. Nat. Genet. 2019, 51, 404–413. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Han, X.; Ong, J.S.; Hewitt, A.W.; MacKey, D.A.; Gharahkhani, P.; MacGregor, S.; Aung, T.; Burdon, K.P.; Cheng, C.Y.; et al. Association of Novel Loci With Keratoconus Susceptibility in a Multitrait Genome-Wide Association Study of the UK Biobank Database and Canadian Longitudinal Study on Aging. JAMA Ophthalmol. 2022, 140, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Cullell, N.; Carrera, C.; Muiño, E.; Torres-Aguila, N.P.; Cárcel-Márquez, J.; González-Sánchez, J.; Gallego-Fabrega, C.; Molina, J.; Besora, S.; Sotoca, J.; et al. Genome-Wide Association Study of VKORC1 and CYP2C9 on Acenocoumarol Dose, Stroke Recurrence and Intracranial Haemorrhage in Spain. Sci. Rep. 2020, 10, 2806. [Google Scholar] [CrossRef] [PubMed]
- Cullell, N.; Carrera, C.; Muiño, E.; Torres, N.; Krupinski, J.; Fernandez-Cadenas, I. Pharmacogenetic Studies with Oral. Anticoagulants. Genome-Wide Association Studies in Vitamin K. Antagonist and Direct Oral. Anticoagulants 2018, 9, 29238–29258. [Google Scholar] [CrossRef]
- Abadi, A.; Alyass, A.; Robiou du Pont, S.; Bolker, B.; Singh, P.; Mohan, V.; Diaz, R.; Engert, J.C.; Yusuf, S.; Gerstein, H.C.; et al. Penetrance of Polygenic Obesity Susceptibility Loci across the Body Mass Index Distribution. Am. J. Hum. Genet. 2017, 101, 925–938. [Google Scholar] [CrossRef]
- Gottlieb, A.; Daneshjou, R.; DeGorter, M.; Bourgeois, S.; Svensson, P.J.; Wadelius, M.; Deloukas, P.; Montgomery, S.B.; Altman, R.B. Cohort-Specific Imputation of Gene Expression Improves Prediction of Warfarin Dose for African Americans. Genome Med. 2017, 9, 98. [Google Scholar] [CrossRef] [PubMed]
- Ishola, A.F.; Gerstein, H.C.; Engert, J.C.; Mohan, V.; Diaz, R.; Anand, S.S.; Meyre, D. Longitudinal Relationships between Glycemic Status and Body Mass Index in a Multiethnic Study: Evidence from Observational and Genetic Epidemiology. Sci. Rep. 2016, 6, 30744. [Google Scholar] [CrossRef] [PubMed]
- Schröder, A.; Klein, K.; Winter, S.; Schwab, M.; Bonin, M.; Zell, A.; Zanger, U.M. Genomics of ADME Gene Expression: Mapping Expression Quantitative Trait Loci Relevant for Absorption, Distribution, Metabolism and Excretion of Drugs in Human Liver. Pharmacogenomics J. 2013, 13, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Glubb, D.M.; Dholakia, N.; Innocenti, F. Liver Expression Quantitative Trait Loci: A Foundation for Pharmacogenomic Research. Front. Genet. 2012, 3, 153. [Google Scholar] [CrossRef]
- Huang, J.; Huffman, J.E.; Huang, Y.; Do Valle, Í.; Assimes, T.L.; Raghavan, S.; Voight, B.F.; Liu, C.; Barabási, A.L.; Huang, R.D.L.; et al. Genomics and Phenomics of Body Mass Index Reveals a Complex Disease Network. Nat. Commun. 2022, 13, 7973. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Ye, K. Mapping Genome Variants Sheds Light on Genetic and Phenotypic Differentiation in Chinese. Genom. Proteom. Bioinform. 2019, 17, 226–228. [Google Scholar] [CrossRef]
- Shungin, D.; Winkler, T.; Croteau-Chonka, D.C.; Ferreira, T.; Locke, A.E.; Mägi, R.; Strawbridge, R.J.; Pers, T.H.; Fischer, K.; Justice, A.E.; et al. New Genetic Loci Link Adipose and Insulin Biology to Body Fat Distribution. Nature 2015, 518, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Guo, D.; Li, K.; Yan, W.; Tan, Y.; Wang, X.; Treiber, F.A.; Chao, J.; Snieder, H.; Dong, Y. Prostasin: A Possible Candidate Gene for Human Hypertension. Am. J. Hypertens. 2008, 21, 1028–1033. [Google Scholar] [CrossRef]
- Sakaue, S.; Kanai, M.; Tanigawa, Y.; Karjalainen, J.; Kurki, M.; Koshiba, S.; Narita, A.; Konuma, T.; Yamamoto, K.; Akiyama, M.; et al. A Cross-Population Atlas of Genetic Associations for 220 Human Phenotypes. Nat. Genet. 2021, 53, 1415–1424. [Google Scholar] [CrossRef]
- Portas, L.; Pereira, M.; Shaheen, S.O.; Wyss, A.B.; London, S.J.; Burney, P.G.J.; Hind, M.; Dean, C.H.; Minelli, C. Lung Development Genes and Adult Lung Function. Am. J. Respir. Crit. Care Med. 2020, 202, 853–865. [Google Scholar] [CrossRef]
- Ripatti, P.; Rämö, J.T.; Mars, N.J.; Fu, Y.; Lin, J.; Söderlund, S.; Benner, C.; Surakka, I.; Kiiskinen, T.; Havulinna, A.S.; et al. Polygenic Hyperlipidemias and Coronary Artery Disease Risk. Circ. Genom. Precis. Med. 2020, 13, E002725. [Google Scholar] [CrossRef] [PubMed]
- Minelli, C.; Dean, C.H.; Hind, M.; Alves, A.C.; Amaral, A.F.S.; Siroux, V.; Huikari, V.; Artigas, M.S.; Evans, D.M.; Loth, D.W.; et al. Association of Forced Vital Capacity with the Developmental Gene NCOR2. PLoS ONE 2016, 11, e0147388. [Google Scholar] [CrossRef] [PubMed]
- van der Meer, D.; Kaufmann, T.; Shadrin, A.A.; Makowski, C.; Frei, O.; Roelfs, D.; Monereo-Sánchez, J.; J Linden, D.E.; Rokicki, J.; Alnaes, D.; et al. The Genetic Architecture of Human Cortical Folding. Sci. Adv. 2021, 7, eabj9446. [Google Scholar] [CrossRef] [PubMed]
| SNP | Variant Type | Phenotypic Consequence(s) | Total Number of References |
|---|---|---|---|
| rs9925964 | Splice Region | Body mass index, longitudinal BMI measurement, physical activity measurement | 27 |
| rs59735493 | Intron | Alzheimer’s disease, anxiety | 19 |
| rs749767 | Intergenic (BCKDK, KAT8) | central corneal thickness, corneal resistance factor | 8 |
| rs1549293 | 3 Prime UTR | Body mass index, longitudinal BMI measurement, physical activity measurement, waist circumference, triglyceride measurement | 7 |
| rs138259061 | Intron | Triglyceride levels | 3 |
| rs11865499 | Noncoding Transcript Exon | Body height | 2 |
| rs61320757 | Intron | Brain measurement—vertex-wise sulcal depth | 1 |
| rs368991827 | Intron | Prostasin levels | 1 |
| rs748699921, 6+ | Missense | Li–Ghorbani–Weisz-Hubshman syndrome | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoo, L.; Mendoza, D.; Richard, A.J.; Stephens, J.M. KAT8 beyond Acetylation: A Survey of Its Epigenetic Regulation, Genetic Variability, and Implications for Human Health. Genes 2024, 15, 639. https://doi.org/10.3390/genes15050639
Yoo L, Mendoza D, Richard AJ, Stephens JM. KAT8 beyond Acetylation: A Survey of Its Epigenetic Regulation, Genetic Variability, and Implications for Human Health. Genes. 2024; 15(5):639. https://doi.org/10.3390/genes15050639
Chicago/Turabian StyleYoo, Lindsey, David Mendoza, Allison J. Richard, and Jacqueline M. Stephens. 2024. "KAT8 beyond Acetylation: A Survey of Its Epigenetic Regulation, Genetic Variability, and Implications for Human Health" Genes 15, no. 5: 639. https://doi.org/10.3390/genes15050639
APA StyleYoo, L., Mendoza, D., Richard, A. J., & Stephens, J. M. (2024). KAT8 beyond Acetylation: A Survey of Its Epigenetic Regulation, Genetic Variability, and Implications for Human Health. Genes, 15(5), 639. https://doi.org/10.3390/genes15050639






