Characterization of the MMP9 Gene and Its Association with Cryptocaryon irritans Resistance Traits in Trachinotus ovatus (Linnaeus, 1758)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Infection Experiment and Sample Collection
2.2. DNA/RNA Extraction and cDNA Synthesis
2.3. Bioinformatics Analysis
2.4. SNP Screening and Genotyping
2.5. T. ovatus MMP9 Gene Expression Analysis
3. Results
3.1. ToMMP9 Sequence Characteristics
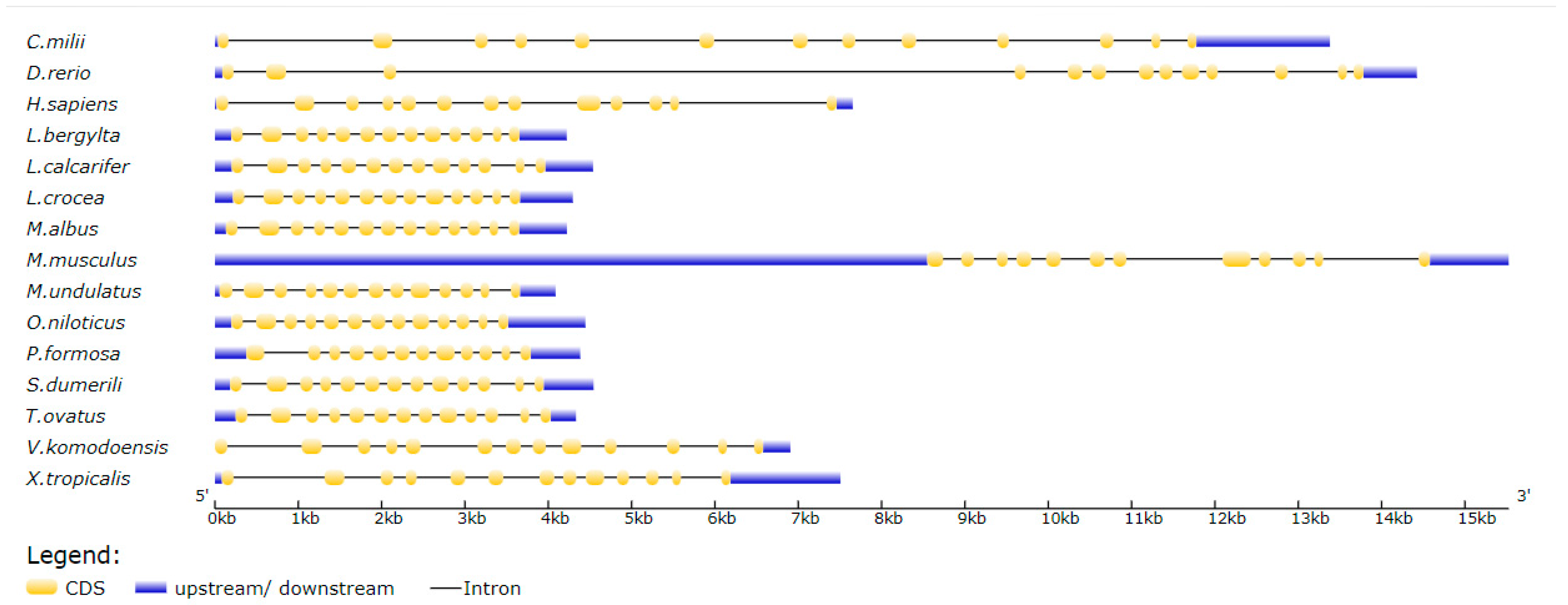
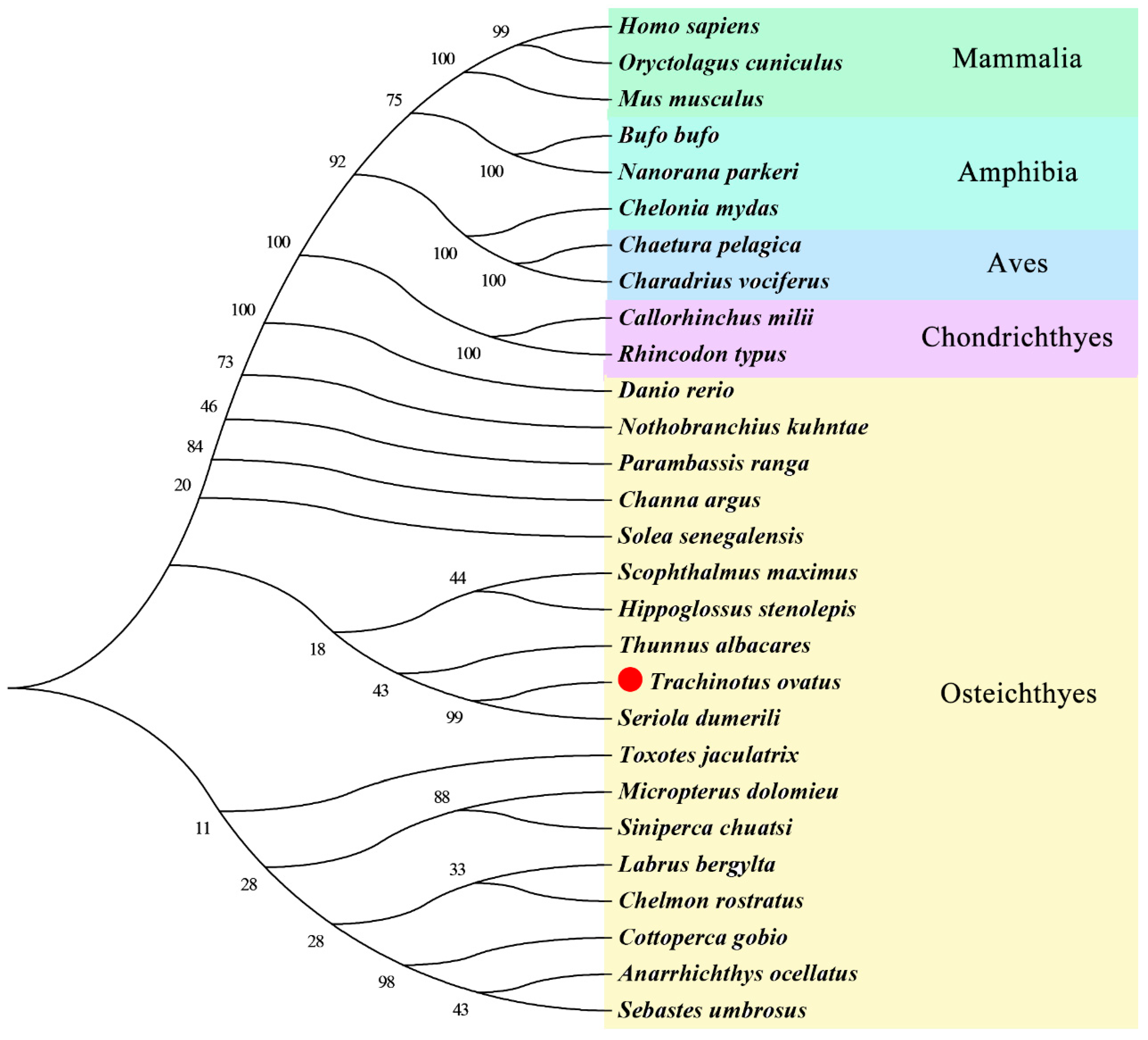
3.2. ToMMP9 Phylogeny and Structural Analysis
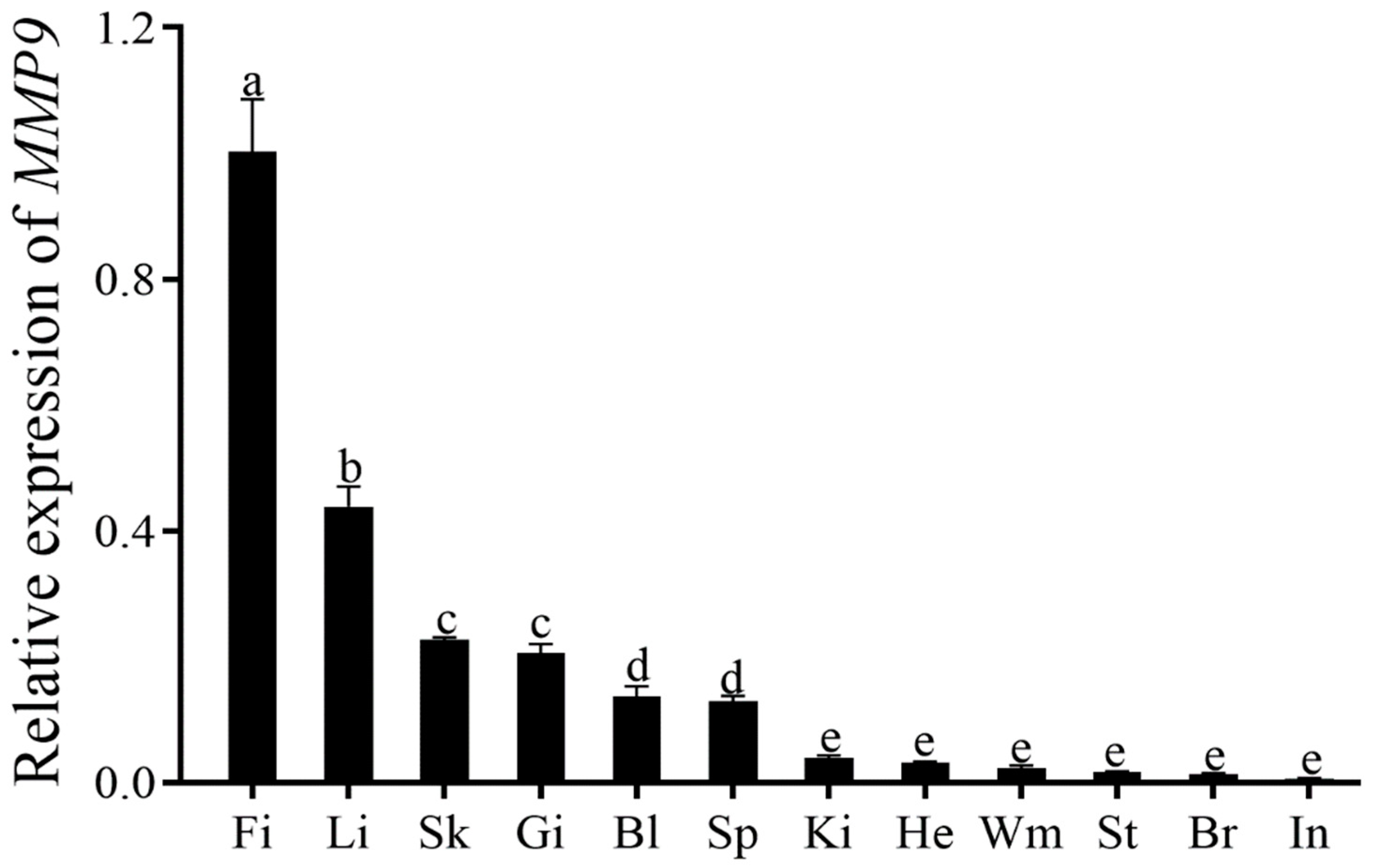
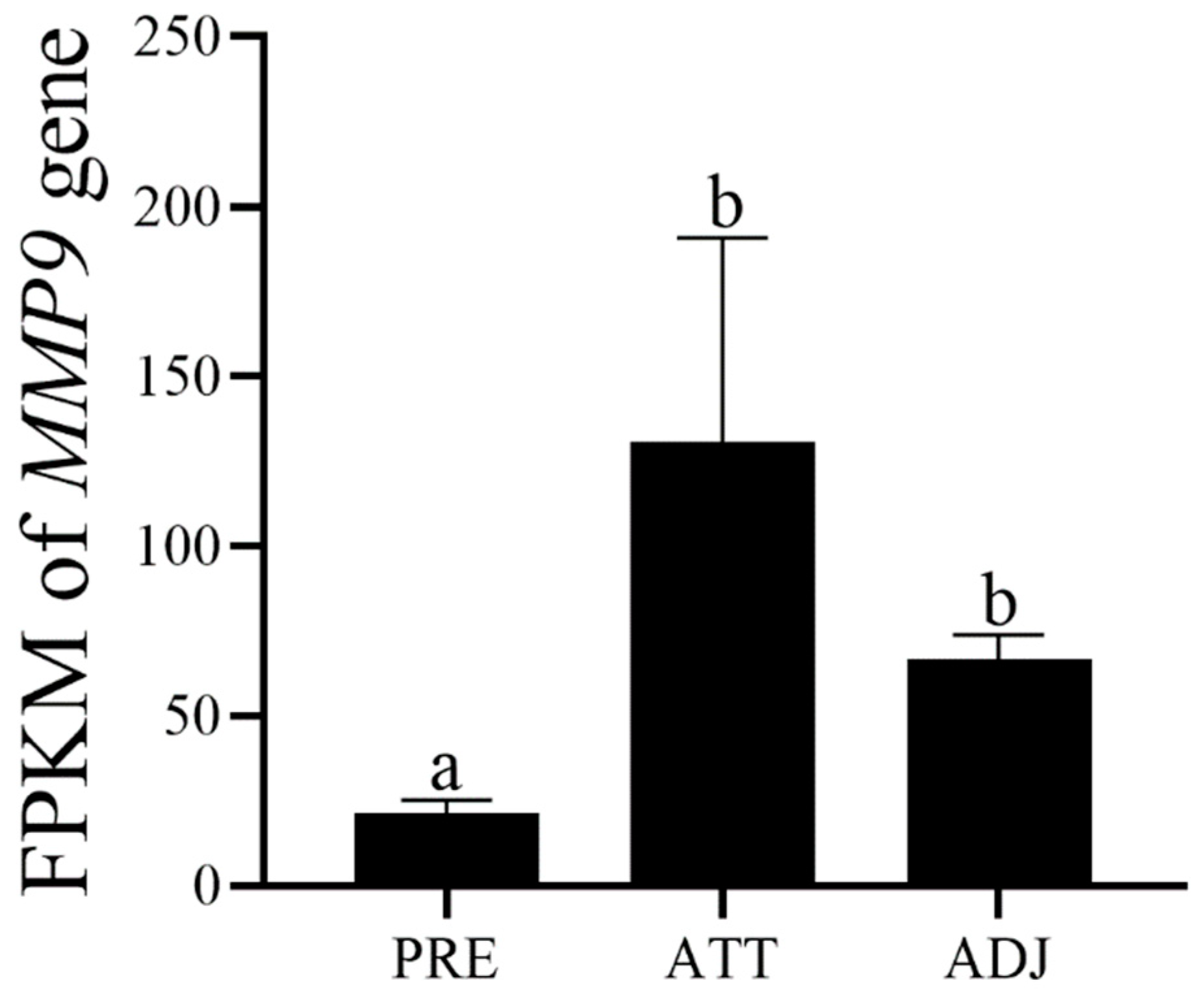
3.3. ToMMP9 Expression Profile in Healthy Tissues and Expression Pattern after C. irritans Infection
3.4. Correlation Analysis of ToMMP9 SNPs and Resistance Characteristics of C. irritans
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Maral, S.; Acar, M.; Balcik, O.S.; Uctepe, E.; Hatipoglu, O.F.; Akdeniz, D.; Altun, H.U.; Kosar, A.; Gunduz, M.; Gunduz, E. Matrix Metalloproteinases 2 and 9 Polymorphism in Patients with Myeloproliferative Diseases: A STROBE-Compliant Observational Study. Medicine 2015, 94, e732. [Google Scholar] [CrossRef]
- Qorri, B.; Kalaydina, R.V.; Velickovic, A.; Kaplya, Y.; Decarlo, A.; Szewczuk, M.R. Agonist-Biased Signaling via Matrix Metalloproteinase-9 Promotes Extracellular Matrix Remodeling. Cells 2018, 7, 117. [Google Scholar] [CrossRef]
- Rath, T.; Roderfeld, M.; Graf, J.; Roeb, E. Matrix metalloproteinases in inflammatory bowel disease-from basic research to clinical significance. Z. Gastroenterol. 2009, 47, 758–769. [Google Scholar] [CrossRef] [PubMed]
- Amin, S.A.; Adhikari, N.; Jha, T. Is dual inhibition of metalloenzymes HDAC-8 and MMP-2 a potential pharmacological target to combat hematological malignancies? Pharmacol. Res. 2017, 122, 8–19. [Google Scholar] [CrossRef]
- Fields, G.B. New strategies for targeting matrix metalloproteinases. Matrix Biol. 2015, 44–46, 239–246. [Google Scholar] [CrossRef]
- Fanjul-Fernandez, M.; Folgueras, A.R.; Cabrera, S. Matrix metalloproteinases: Evolution, gene regulation and functional analysis in mouse models. Biochim. Biophys. Acta. 2010, 1803, 3–19. [Google Scholar] [CrossRef]
- Kapoor, C.; Vaidya, S.; Wadhwan, V. Seesaw of matrix metalloproteinases (MMPs). J. Cancer Res. Ther. 2016, 12, 28–35. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchi, H. Loop-mediated isothermal amplification of DNA. Nucl. Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef] [PubMed]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nature Reviews Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Barksby, H.E.; Milner, J.M.; Patterson, A.M. Matrix metalloproteinase 10 promotion of collagenolysis via procollagenase activation: Implications for cartilage degradation in arthritis. Arthritis Rheum. 2006, 54, 3244–3253. [Google Scholar] [CrossRef]
- Geurts, N.; Martens, E.; Van Aelst, I. β-hematin interaction with the hemopexin domain of gelatinase B/MMP-9 provokes autocatalytic processing of the propeptide, thereby priming activation by MMP-3. Biochemistry 2008, 47, 2689–2699. [Google Scholar] [CrossRef] [PubMed]
- Velasco, G.; Cal, S.; Merlos-Suarez, A. Human MT6-matrix metalloproteinase: Identification, progelatinase A activation, and expression in brain tumors. Cancer Res. 2000, 60, 877–882. [Google Scholar] [PubMed]
- Bronisz, E.; Kurkowska-Jastrzębska, I. Matrix Metalloproteinase 9 in Epilepsy: The Role of Neuroinflammation in Seizure Development. Mediators Inflamm. 2016, 2016, 7369020. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, H.; Luo, X.; Deng, J.; Pan, Y.; Liang, H. Overexpression of SMYD3 and matrix metalloproteinase-9 are associated with poor prognosis of patients with gastric cancer. Tumour. Biol. 2015, 36, 4377–4386. [Google Scholar] [CrossRef]
- Yabluchanskiy, A.; Ma, Y.; Iyer, R.P.; Hall, M.E.; Lindsey, M.L. Matrix metalloproteinase-9: Many shades of function in cardiovascular disease. Physiology 2013, 28, 391–403. [Google Scholar] [CrossRef]
- Yoshinari, N.; Ishida, T.; Kudo, A.; Kawakami, A. Gene expression and functional analysis of zebrafish larval fin fold regeneration. Dev. Biol. 2009, 325, 71–81. [Google Scholar] [CrossRef]
- LeBert, D.C.; Squirrell, J.M.; Rindy, J.; Broadbridge, E.; Lui, Y.; Zakrzewska, A.; Eliceiri, K.W.; Meijer, A.H.; Huttenlocher, A. Matrix metalloproteinase 9 modulates collagen matrices and wound repair. Development 2015, 142, 2136–2146. [Google Scholar] [CrossRef] [PubMed]
- Xiong, N.X.; Luo, S.W.; Fan, L.F.; Mao, Z.W.; Luo, K.K.; Liu, S.J.; Wu, C.; Hu, F.Z.; Wang, S.; Wen, M.; et al. Comparative analysis of erythrocyte hemolysis, plasma parameters and metabolic features in red crucian carp (Carassius auratus red var) and triploid hybrid fish following Aeromonas hydrophila challenge. Fish Shellfish Immunol. 2021, 118, 369–384. [Google Scholar] [CrossRef]
- Liyanage, T.D.; Nikapitiya, C.; Lee, J.; De, Z.M. Potential immune regulatory role of miR-146a upon Aeromonas hydrophila and Edwardsiella piscicida infections in zebrafish. Braz. J. Microbiol. 2020, 51, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.Y.; Shen, Y.B.; Fu, J.J.; Liu, F.; Guo, S.Z.; Li, J.L. Characterization of MMP-9 gene from grass carp (Ctenopharyngodon idella): An Aeromonas hydrophila-inducible factor in grass carp immune system. Fish Shellfish Immunol. 2013, 35, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Ke, F.; Wang, Y.; Hong, J.; Xu, C.; Chen, H.; Zhou, S.B. Characterization of MMP-9 gene from a normalized cDNA library of kidney tissue of yellow catfish (Pelteobagrus fulvidraco). Fish Shellfish Immunol. 2015, 45, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Fast, M.D. Fish immune responses to parasitic copepod (namely sea lice) infection. Dev. Comp. Immunol. 2014, 43, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Skugor, S.; Glover, K.A.; Nilsen, F.; Krasnov, A. Local and systemic gene expression responses of Atlantic salmon (Salmo salar L.) to infection with the salmon louse (Lepeophtheirus salmonis). BMC Genom. 2008, 9, 498. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, J.; Liu, J.; Guo, J.; Wei, Y.; Li, J.; He, P.; Lan, T.; Peng, L.; Li, H. MicroRNA miR-155-5p knockdown attenuates Angiostrongylus cantonensis-induced eosinophilic meningitis by downregulating MMP9 and TSLP proteins. Int. J. Parasitol. 2021, 51, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Komar, A.A. Genetics: SNPs, silent but not invisible. Science 2007, 315, 466–467. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.Y.; Guo, H.Y.; Zhu, C.Y.; Ma, Z.H.; Jiang, S.G.; Zhang, D.C. Genetic polymorphism of breeding populations of golden pompano (Trachinotus ovatus). South China Fish. 2014, 10, 67–71. (In Chinese) [Google Scholar]
- Dan, X.M.; Li, A.X.; Lin, X.T.; Bo, J.S.; Zhang, H.F. Immune response and immunoprotection of pompanos (Trachinotus ovatus) against Cryptocaryon irritans. Acta Hydrobiol. Sin. 2008, 32, 13–18. [Google Scholar] [CrossRef]
- Zhang, D.C.; Guo, L.; Guo, H.Y.; Zhu, K.C.; Li, S.Q.; Zhang, Y.; Zhang, N.; Liu, B.S.; Jiang, S.G.; Li, J.T. Chromosome-level genome assembly of golden pompano (Trachinotus ovatus) in the family Carangidae. Sci Data 2019, 6, 216. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Matrisian, L.M. Metalloproteinases and their inhibitors in matrix remodeling. Trends Genet. 1990, 6, 121–125. [Google Scholar] [CrossRef]
- Vandooren, J.; Geurts, N.; Martens, E.; VandenSteen, P.E.; Jonghe, S.D.; Herdewijn, P.; Opdenakker, G. Gelatin degradation assay reveals MMP-9 inhibitors and function of O-glycosylated domain. World J. Biol. Chem. 2011, 2, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Dufour, A.; Sampson, N.S.; Li, J.; Kuscu, C.; Rizzo, R.C.; Deleon, J.L.; Zhi, J.; Jaber, N.; Liu, E.; Zucker, S.; et al. Small-molecule anticancer compounds selectively target the hemopexin domain of matrix metalloproteinase-9. Cancer Res. 2011, 71, 4977–4988. [Google Scholar] [CrossRef] [PubMed]
- Fagerberg, L.; Hallström, B.M.; Oksvold, P.; Kampf, C.; Djureinovic, D.; Odeberg, J.; Habuka, M.; Tahmasebpoor, S.; Danielsson, A.; Edlund, K.; et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol. Cell Proteom. 2014, 13, 397–406. [Google Scholar] [CrossRef]
- Yue, F.; Cheng, Y.; Breschi, A.; Vierstra, J.; Wu, W.; Ryba, T.; Sandstrom, R.; Ma, Z.; Davis, C.; Pope, B.D.; et al. A comparative encyclopedia of DNA elements in the mouse genome. Nature 2014, 515, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Fuscoe, J.C.; Zhao, C.; Guo, C.; Jia, M.; Qing, T.; Bannon, D.I.; Lancashire, L.; Bao, W.; Du, T.; et al. A rat RNA-Seq transcriptomic BodyMap across 11 organs and 4 developmental stages. Nat. Commun. 2014, 5, 3230. [Google Scholar] [CrossRef] [PubMed]
- Yoong, S.; O’Connell, B.; Soanes, A.; Crowhurst, M.O.; Lieschke, G.J.; Ward, A.C. Characterization of the zebrafish matrix metalloproteinase 9 gene and its developmental expression pattern. Gene Expr. Patterns 2007, 7, 39–46. [Google Scholar] [CrossRef]
- Brawand, D.; Wagner, C.E.; Li, Y.I.; Malinsky, M.; Keller, I.; Fan, S.; Simakov, O.; Ng, A.Y.; Lim, Z.W.; Bezault, E.; et al. The genomic substrate for adaptive radiation in African cichlid fish. Nature 2014, 513, 375–381. [Google Scholar] [CrossRef]
- Colorni, A.; Burgess, P. Cryptocaryon irritans Brown 1951, the cause of ‘white spot disease’ in marine fish: An update. Aquar. Sci. Conserv. 1997, 1, 217–238. [Google Scholar] [CrossRef]
- Diggles, B.K.; Lester, R.J.G. Infections of Cryptocaryon irritans wild fish from southeast Queensland, Australia. Dis. Aquat. Org. 1996, 25, 159–167. [Google Scholar] [CrossRef]
- Wang, Y.G.; Liu, Z.W.; Lin, C.Y.; Cheng, X.; Wang, L.; Li, H. Cryptocaryoniosis in cultured turbot and its treatment. J. Fish. China 2011, 35, 1105–1112. [Google Scholar] [CrossRef]
- Zeinali, F.; Homaei, A.; Kamrani, E. Sources of marine superoxide dismutases: Characterist and applications. Int. J. Biol. Macromol. 2015, 79, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Hadwan, M.H. Simple spectrophotometric assay for measuring catalase activity in biologi tissues. BMC Biochem. 2018, 19, 7–15. [Google Scholar] [CrossRef]
- Yin, F.; Gong, Q.; Li, Y.; Dan, X.; Sun, P.; Gao, Q.; Shi, Z.; Peng, S.; Li, A. Effects of Cryptocaryon irritans infection on the survival, feeding, respiratory rate and ionic regulation of the marbled rockfish Sebastiscusmarmoratus. Parasitology 2014, 141, 279–286. [Google Scholar] [CrossRef]
- Dan, X.M.; Li, A.X.; Lin, X.T.; Teng, N.; Zhu, X.Q. A standardized method to propagate Cryptocaryon irritans on a susceptible host pompano Trachinotus ovatus. Aquaculture 2006, 258, 127–133. [Google Scholar] [CrossRef]
- Zhu, K.C.; Zhang, N.; Liu, B.S.; Guo, L.; Guo, H.Y.; Jiang, S.G.; Zhang, D.C. Functional Analysis of IRF1 Reveals its Role in the Activation of the Type I IFN Pathway in Golden Pompano, Trachinotus ovatus (Linnaeus 1758). Int. J. Mol. Sci. 2020, 21, 2652. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.W.; Jiang, B.; Dan, X.M.; Li, A.X. Advances in the research on mucosal immune response offish against Cryptocaryon irritans infection. J. Fish. China 2019, 43, 156–167. [Google Scholar]
- Wang, F.H.; Xie, M.Q.; Li, A.X. A novel protein isolated from the serum of rabbitfish (Siganus oramin) is lethal to Cryptocaryon irritans. Fish Shellfish Immunol. 2010, 29, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.S.; Guyer, M.S.; Charkravarti, A. Variations on a theme: Cataloging human DNA sequence variation. Science 1997, 278, 1580–1581. [Google Scholar] [CrossRef]
- Wei, K.Y.; Xie, S.M.; Wang, S.T.; Chen, Y.K.; Niu, D.H.; Li, J.L. Polymorphism of SNPs in EGFR intron 1 and its association with growth traits in Sinonovacula constricta. J. Fish. China 2019, 43, 483–491. [Google Scholar]
- Guo, L.; He, P.Y.; Zhu, K.C.; Guo, H.Y.; Liu, B.S.; Zhang, N.; Jiang, S.G.; Zhang, D.C. Functional identification of ToLAAO genes and polymorphism association analysis of Cryptocaryon irritans resistance in Trachinotus ovatus. Aquac. Res. 2022, 53, 208–220. [Google Scholar] [CrossRef]
- Wang, J.X.; Luo, H.; Sun, Q.H.; Wang, H.L.; Liu, H. Characterization of β2m gene and its association with antibacterial trait in Megalobrama amblycephala. Aquaculture 2021, 541, 736802. [Google Scholar] [CrossRef]
- Zhu, K.C.; Liu, J.; Liu, B.S.; Guo, H.Y.; Zhang, N.; Guo, L.; Jiang, S.G.; Zhang, D.C. Functional characterization of four ToRac genes and their association with anti-parasite traits in Trachinotus ovatus (Linnaeus, 1758). Aquaculture 2022, 560, 738514. [Google Scholar] [CrossRef]
- Morgan, J.T.; Fink, G.R.; Bartel, D.P. Excised linear introns regulate growth in yeast. Nature 2019, 565, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Parenteau, J.; Maignon, L.; Berthoumieux, M.; Catala, M.; Gagnon, V.; Abou Elela, S. Introns are mediators of cell response to starvation. Nature 2019, 565, 612–617. [Google Scholar] [CrossRef] [PubMed]

| Primer | Sequence (5′–3′) |
|---|---|
| PMMP9-F | ATGGCTGCGGCTCTT |
| PMMP9-R | TCCTGCGTGCCTATCC |
| MMP9-F | GAGCATTCCCCTCAAGTCCG |
| MMP9-R | GCCGCAGATTCCCAGGTTTA |
| S-PMMP9-F | TCCATTGCAGCCGCTTGTA |
| S-PMMP9-R | TGCATGTTGCCTAATCCATAATCT |
| S-MMP9-F | GGCAGAAGTGAGTAGCA |
| S-MMP9-R | TGACACCATAGACTGGAAGC |
| qPCR-MMP9-F | CACCAGTGAGGGACGAG |
| qPCR-MMP9-R | GGCTGCCACCAGAAACA |
| EF-1α F | CCCCTTGGTCGTTTTGCC |
| EF-1α R | GCCTTGGTTGTCTTTCCGCTA |
| Gene | Locus | Tybe | Changes of DNA Base |
|---|---|---|---|
| MMP9 | promoter | transversion | A/C |
| intron | transition | A/G |
| Gene | Position | Genotype | Susceptible | Resistant | X2 (p) | Allele | Susceptible | Resistant | X2 (p) |
|---|---|---|---|---|---|---|---|---|---|
| MMP-9 | A/C | AA | 17 | 21 | 0.68 (0.71) | A | 58 | 63 | 0.52 (0.47) |
| CC | 9 | 8 | C | 42 | 37 | ||||
| AC | 24 | 21 | |||||||
| A/G | AA | 14 | 3 | 9.07 (0.01) | A | 50 | 31 | 7.49 (0.006) | |
| GG | 14 | 22 | G | 50 | 69 | ||||
| AG | 22 | 25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Zhu, K.-C.; Pan, J.-M.; Guo, H.-Y.; Liu, B.-S.; Zhang, N.; Yang, J.-W.; Zhang, D.-C. Characterization of the MMP9 Gene and Its Association with Cryptocaryon irritans Resistance Traits in Trachinotus ovatus (Linnaeus, 1758). Genes 2023, 14, 475. https://doi.org/10.3390/genes14020475
Liu J, Zhu K-C, Pan J-M, Guo H-Y, Liu B-S, Zhang N, Yang J-W, Zhang D-C. Characterization of the MMP9 Gene and Its Association with Cryptocaryon irritans Resistance Traits in Trachinotus ovatus (Linnaeus, 1758). Genes. 2023; 14(2):475. https://doi.org/10.3390/genes14020475
Chicago/Turabian StyleLiu, Jun, Ke-Cheng Zhu, Jin-Min Pan, Hua-Yang Guo, Bao-Suo Liu, Nan Zhang, Jing-Wen Yang, and Dian-Chang Zhang. 2023. "Characterization of the MMP9 Gene and Its Association with Cryptocaryon irritans Resistance Traits in Trachinotus ovatus (Linnaeus, 1758)" Genes 14, no. 2: 475. https://doi.org/10.3390/genes14020475
APA StyleLiu, J., Zhu, K.-C., Pan, J.-M., Guo, H.-Y., Liu, B.-S., Zhang, N., Yang, J.-W., & Zhang, D.-C. (2023). Characterization of the MMP9 Gene and Its Association with Cryptocaryon irritans Resistance Traits in Trachinotus ovatus (Linnaeus, 1758). Genes, 14(2), 475. https://doi.org/10.3390/genes14020475







