Abstract
Some of the key genes and regulatory mechanisms controlling drought response in durum wheat have been identified. One of the major challenges for breeders is how to use this knowledge for the achievement of drought stress tolerance. In the present study, we report the expression profiles of the TdDRF1 gene, at consecutive plant growth stages, from different durum wheat genotypes evaluated in two different field environments. The expression of a possible target gene (Wdnh13) of the TdDRF1 gene was also investigated and analogies with the transcript profiles were found. The results of the qRT-PCR highlighted differences in molecular patterns, thus suggesting a genotype dependency of the TdDRF1 gene expression in response to the stress induced. Furthermore, a statistical association between the expression of TdDRF1 transcripts and agronomic traits was also performed and significant differences were found among genotypes, suggesting a relationship. One of the genotypes was found to combine molecular and agronomic characteristics.
Keywords:
durum wheat; expression profiles; field trials; qRT-PCR; TdDRF1 gene; transcription factors; Wdhn13 1. Introduction
Unmitigated climate change due to increasing greenhouse gas emissions will have an adverse impact on plant growth and crop yield in some areas of the world, including through the more frequent occurrences of drought stress [1,2]. To mitigate against water scarcity and/or irregular availability and to enhance the sustainability of global food production, it is necessary to explore avenues for producing more food with proportionally less water [3]. Cereals are our dominant source of food, with wheat playing a major contribution to human diet and health [4]. Durum wheat (Triticum turgidum var. durum) is largely cultivated in the Mediterranean basin and other semi-arid and marginal areas, with the milled product being used mainly for making pasta and other staple foods.
Plants deploy complex mechanisms to cope with stresses like dehydration. Several genes have been described that are activated at the transcriptional level, with cis- and trans-acting factors involved in the expression of dehydration responsive genes [5]. The dehydration responsive element binding (DREB) family of transcription factors (TFs) represents one of the major players involved in abiotic (dehydration, cold, high salinity) stress responses [6]. The DREB proteins interact with the drought-responsive element (DRE) motif in the promoter regions of many stress-inducible genes and belong to the larger AP2/ERF (APETALA2/ethylene-responsive factor) family, as the DNA binding and recognition is mediated by the Apetala2 (AP2) domain [6,7]. TFs are therefore good candidates for improving crop tolerance to drought because of their role as master regulators of several clusters of genes [8,9,10].
A DREB2-related gene, namely TdDRF1 (Triticum durum dehydration responsive factor 1), was isolated in durum wheat and reported as producing three forms of transcript through alternative splicing (AS): TdDRF1.1 and TdDRF1.3, encoding putative TFs containing the AP2/EREBP DNA-binding domain and the nuclear localization signal (NLS), and TdDRF1.2, encoding a putative abortive protein lacking both the AP2 domain and the NLS. TdDRF1 gene expression was linked to the plant response to water deficit [11] and its analysis in different genotypes of durum wheat and one triticale cultivar under greenhouse conditions and subjecting plants to a moderate dehydration stress resulted in different genotypic behaviours [12]. Furthermore, a preliminary study of the field expression of TdDRF1 was also carried out, analysing durum wheat and triticale lines in a short time-course with five sampling points [13]. The above-mentioned studies revealed that TdDRF1 gene expression had an important genotype-dependence, as also found for other transcription factor genes controlling plant response to abiotic stress [14,15] or key metabolic pathways [16,17]. The relationships among the expression patterns of the three transcripts and the phenotype response to water stress in a complex field environment are still largely unknown, highlighting the need for further research to gain more insight into the gene expression under realistic environmental conditions.
The aim of the present work was to investigate the molecular behaviour of the TdDRF1 gene in the field during a time-course drought stress experiment in six durum wheat genotypes, and to find whether there was a relationship between the expression of the TdDRF1 gene and one possible downstream target (Wdhn13 gene). The association between transcript profile analysis and agronomic performance was also explored.
2. Materials and Methods
2.1. Plant Materials
Six durum wheat genotypes were used in the field experiments (Table 1).

Table 1.
Pedigree information, date of release/development, and origin of six durum wheat genotypes (Country of origin: IT, Italy; MX, Mexico).
Duilio, Creso, and Colosseo are Italian commercial varieties. The other three genotypes were developed by the CIMMYT program in Mexico, with Barnacla and AEL being advanced experimental lines and Altar C84 being a high yielding variety commercially released in Mexico and other countries.
2.2. Field Experiments
The field trial was conducted at the CIMMYT experimental station (Campo Experimental Norman Ernest Borlaug, CENEB) near Cd. Obregón (Sonora, Mexico) during the cropping season of 2010. The experiment was arranged in a randomized complete block design with 4 replicates and plots of 3.36 m2 for each of the two irrigation treatments or testing environments. The two different irrigation conditions were full irrigation (FI), with 550–600 mm of total water supplied by gravity irrigation during the full crop cycle, and reduced irrigation (RI), with 220–250 mm of total water applied through a drip system, all before heading. In both irrigation conditions plots were fertilized optimally as per the site-specific agronomic recommendations using a total of 250 units of nitrogen in the form of urea (50 units at sowing, 100 units at first node, and 100 units at the end of tillering) and phosphorus (50 units applied at sowing). Plots were maintained free of diseases and pests via the uniform application of fungicide and insecticide.
2.3. Agronomic Traits
After mechanical harvest of the whole plots, grain yield and thousand kernel weight were determined and considered in relation with the TdDRF1 expression profiles.
2.4. RNA Extraction
A time-course experiment was designed with a sampling schedule consisting of 7 collection stages (T1 to T7), as reported in Table 2.

Table 2.
Leaf collection schedule implemented during the time-course experiment to establish the expression profile of the TdDRF1 gene.
This schedule was planned to include the whole growing period, from heading to harvest. For each sampling, ten representative flag leaves were harvested for each plot, pooled together, immediately frozen in liquid nitrogen, and subsequently stored at −80 °C prior to RNA extraction. Total RNA was extracted from the leaves using the TRIzol® Reagent (Invitrogen, Carlsbad, CA, USA) in accordance with the manufacturer’s instructions, and lyophilized. Lyophilized RNA samples (each yielding being approximately 25–35 µg of total RNA) were then resuspended in nuclease-free sterile water, qualitatively assessed by agarose gel electrophoresis, and quantified with a NanoDrop ND-1000 Spectrophotometer (NanoDrop Technologies, Wilmington, NC, USA).
2.5. Reverse Transcription, Pre-Amplification, and qRT-PCR
A set of specific primers, designed using the Assay-By-Design software (Applied Biosystems) with a view to obtain three specific and distinguishable fragments corresponding to each TdDRF1 transcript, were used [11] (Supplementary Figure S1).
Supplementary Figure S2 is a schematic representation of the complete procedure of reverse transcription, pre-amplification, and qRT-PCR. The pre-amplification step was included to optimize the real-time reactions. A High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) was used for the reverse transcription reactions. Samples (20 µL) contained 2 µL of 10× RT Buffer, 0.8 µL of 25× dNTP Mix (100 mM), 2 µL of 10× RT Random Primers, 1 µL of RNase Inhibitor (20 U/µL), and 2 µg of total RNA in nuclease-free water. The thermal cycling conditions were 10 min at 25 °C, 2 h at 37 °C, and 5 min at 85 °C and at 4 °C. Pre-amplification reactions (50 µL) contained 2 µL of TaqMan® PreAmp Master Mix 2× (Applied Biosystems), 12.5 µL of pooled assay mix (0.2×, each assay), and 250 ng of cDNA sample in nuclease-free water. Reactions were held at 95 °C for 10 min and then at 95 °C for 15 s and 60 °C for 4 min 14 times. The resulting pre-amplified reactions were then diluted (1:20) in 1× TE buffer and used as the starting material for the subsequent Custom TaqMan® Gene Expression Assays (Applied Biosystems) for the three target transcripts and the endogenous control carried out in the Applied Biosystems 7300 Real-Time PCR System. The final volume (20 µL) of a single PCR reaction contained 10 µL of 2× TaqMan® Universal PCR Master Mix with AmpErase® UNG (Applied Biosystems), 1 µL of 20× Custom TaqMan® Gene Expression Assay (Applied Biosystems), and 2 µL of diluted pre-amplified product as a template. Samples were run in three biological replicates (from three randomized plots) and three technical replicates.
For endogenous control of relative quantification, different wheat genes were tested: 18S rRNA, TaSNK1 [18], actin, Ta2291, and Ta2776 [19]. The GeNorm algorithm was used to calculate the gene-stability value (M) for all reference genes according to:
where Vjk represents the arithmetic mean of all pairwise variations [19]. As the gene with the lowest M values showed the most stable expression, TaSNK1 was used in our qRT-PCR assays (Supplementary Table S1).
A relative quantification of the TdDRF1 transcripts was obtained using the ΔΔCT method [20] for each sample and results were expressed as normalized relative quantity (NRQ). Furthermore, each expression profile was also calculated as the log2 value of the fold change (FC) (abundance under stress/abundance under control) for each transcript and time.
2.6. Transcripts of the Wdhn13 Gene
Based on the literature, the Wdhn13 gene was chosen as a putative target of the regulation by the TdDRF1 transcription factor [21,22], using the sequence from Triticum aestivum, locus AB297677, deposited in NCBI in 2007. A qRT-PCR analysis was performed on RNA samples of AEL and of Barnacla collected from both the FI and RI conditions during the time-course. The following pair of primers was used: FOR 5′-GATGGCAACTACGGGAAGTC-3′ and REV 5′-GCAGCTTGTCCTTGATCTTG-3′, amplifying an 88 bp cDNA fragment, which was cloned for the setup of the standard curve. qRT-PCR reactions were performed using SYBR green technology in accordance with the procedure reported by Vítámvás and colleagues [23].
2.7. Statistical Analyses
All statistical analyses were carried out with IBM SSPS Statistics 23. The analysis of variance (ANOVA) for each parameter was performed at a 95% confidence level and the significant difference between means was tested using Tukey’s method when applicable. Furthermore, the significance of contrast between the up-regulated and down-regulated TdDRF1 transcripts was also calculated using molecular data as a fixed factor and agronomic data (GY and TKW) as the variable ones.
3. Results
3.1. Agronomic Data
The grain yield (GY) and thousand kernel weight (TKW) averaged over four replicates in the two irrigation conditions are reported in Table 3.

Table 3.
Grain yield (GY) in ton/ha and thousand kernel weight (TKW) in g observed for the six durum wheat genotypes, evaluated under full (FI) and reduced (RI) irrigation conditions. Data reported are means ± standard deviations.; * p < 0.05; ** p < 0.01.
3.2. Expression Profiles of TdDRF1 Gene
For each transcript, ANOVA was carried out using a log transformation of normalized relative quantities (NRQs) of transcript between the two irrigation conditions (FI and RI) for each time. Significant differences are summarized in Table 4. With regard to the putative transcription factors, significant differences were found in the TdDRF1.3 transcript at T1, in both TdDRF1.1 and TdDRF1.3 transcripts at T4, in the TdDRF1.1 transcript at T5, and in the TdDRF1.3 transcript at T7.

Table 4.
ANOVA summery table. Significant differences in TdDRF1 transcripts between FI and RI conditions during the time-course are shown.
For each genotype, NRQs of each transcript calculated during the time-course and rescaled to its own T1 value are shown in Figure 1. Unfortunately, the samples of Altar C84 under the RI condition were lost. As each profile value referred to its own T1, the analyses of transcripts were carried out at a trend level throughout the time-course. Furthermore, the strategy of relative quantification is only suitable for comparing results from the same transcript between treatments, so that results obtained with primer pairs different to each other could not be directly compared [20].
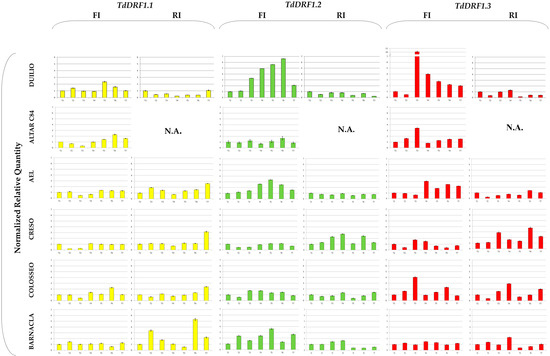
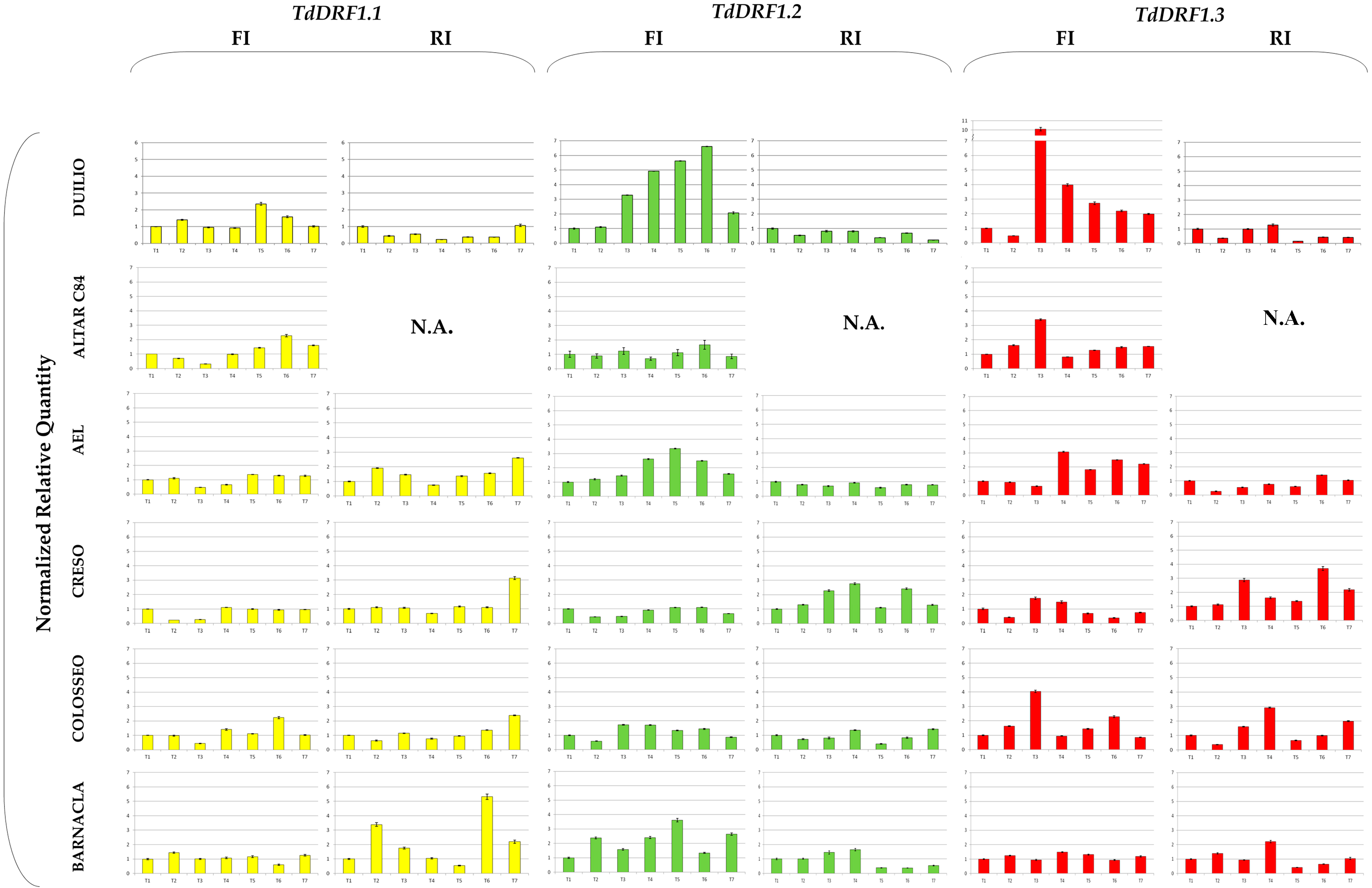
Figure 1.
Expression profiles of the TdDRF1 gene transcripts. Each expression profile is shown as normalized relative quantity rescaled to its own T1 value. Results referred to the six durum wheat genotypes analysed under both FI and RI conditions (TdDRF1.1 in yellow, TdDRF1.2 in green and TdDRF1.3 in red, respectively). The time course consisted of seven points (T1 to T7). N.A., data not available. Data are represented as mean + SE.
Comparing the genotypes shown in Figure 1, the TdDRF1.1 transcript under the FI condition showed a slightly variable trend with some exceptions: Altar C84, AEL, Creso, and Colosseo showed a decrease at T3, while Duilio, Altar C84, AEL, and Colosseo showed an increase at T6. On the other hand, under the RI condition, each genotype showed a more distinct behaviour: Duilio showed a clear decrease at each time, AEL, Creso, and Colosseo remained almost constant at their T1 values, with a substantial increase at T7, while Barnacla showed a higher variability during the time-course with two large increases at T2 and T6.
With regard to the TdDRF1.2 transcript under the FI condition, there were differences mainly at T3, T4, and T5. Duilio, AEL, and Barnacla displayed a large increase in comparison with their T1 values, while Altar C84 and Creso remained almost constant and Colosseo showed a slight increase. Under the RI condition, only Creso showed appreciable variations during the time-course.
As regards the TdDRF1.3 transcript under the FI condition, all genotypes were found to be appreciably variable during the time-course with the exception of Barnacla. In particular, at T3 there was a large increase in Duilio, Altar C84, and Colosseo and, to a lesser extent, Creso, while AEL was late, showing an increase at T4. Under the RI condition, Duilio and AEL were almost constant or less than their initial values, while the other three genotypes showed variations, mainly in Creso and Colosseo and, to a minor extent, in Barnacla.
In addition, a tentative comparison of the trends among the three transcripts was carried out for each genotype. Duilio showed an overall expression of the three transcripts under full irrigation, while under reduced irrigation it seemed to be down-regulated by the water stress. Under full irrigation, Altar C84 showed a complementary activation of the transcription factors (TdDRF1.1 and TdDRF1.3) at different times. AEL showed similar behaviours for the TdDRF1.2 and TdDRF1.3 transcripts under full irrigation, while under reduced irrigation neither transcript showed any appreciable changes, with TdDRF1.1 being variable. Creso showed an almost constant expression of the three transcripts under full irrigation, while under reduced irrigation the TdDRF1.2 and TdDRF1.3 transcripts were found to respond to the water stress. On the other hand, Colosseo expressed mainly the TdDRF1.3 transcript under both conditions, while Barnacla expressed only the TdDRF1.2 transcript under full irrigation and the TdDRF1.1 transcript under reduced irrigation with a single peak in the TdDRF1.3 transcript at T4.
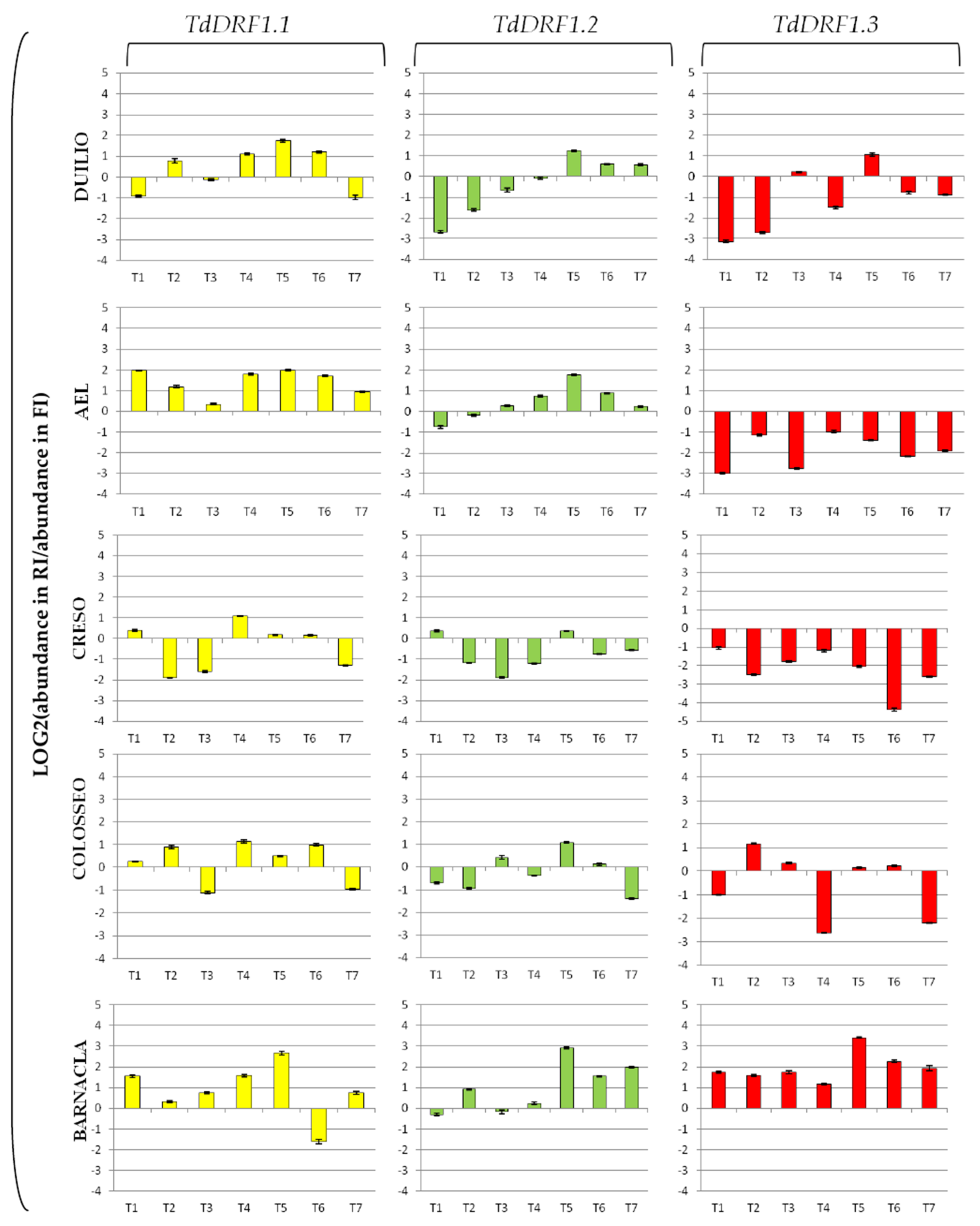
The expression profiles represented as fold change (log2 value of the ratio for abundance under stress and abundance under control) for each transcript are shown in Figure 2. The TdDRF1.1 transcript turned out to be mostly up-regulated in the genotypes, particularly at T4, T5, and T6, to different extents, with Creso showing the lowest values. The TdDRF1.2 transcript showed the greatest variability among genotypes, even if all of them were up-regulated at T5. Both AEL and Duilio showed a similar trend, initially down-regulated and then shifting to up-regulated at different time points (T3 and T5, respectively). Creso was mostly down-regulated, while Colosseo showed a swinging trend (up- and down-regulated at different times). Barnacla was largely up-regulated from T5 onwards. The TdDRF1.3 transcript was largely up-regulated in Barnacla during the whole time-course with a peak at T5 (in fact detectable in all its transcripts). On the contrary, all other genotypes showed a down-regulation throughout the time-course, apart from a light up-regulation in Colosseo and Duilio at T2 and T5, respectively.
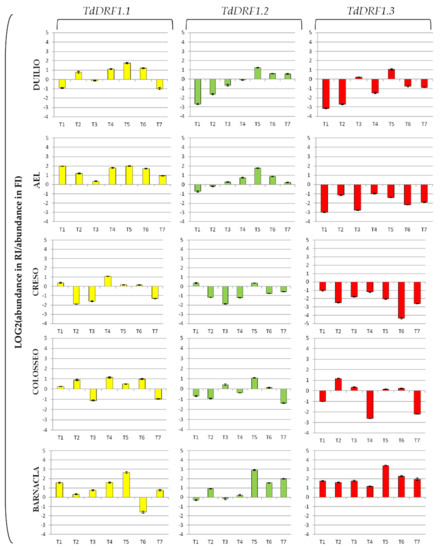
Figure 2.
Expression profiles of the three TdDRF1 transcripts. Each expression profile is shown as fold change (log2 value of the ratio for abundance under stress and abundance under control). Results referred to five durum wheat genotypes analysed under both FI and RI conditions (TdDRF1.1 in yellow, TdDRF1.2 in green, and TdDRF1.3 in red, respectively). The time course consisted of seven points (T1 to T7). Data are represented as mean ± SE.
3.3. Statistical Association between Fold Change of TdDRF1 Transcripts and Traits
With the aim of finding an association between the agronomical data and the fold change in the three transcripts under the reduced irrigation condition, molecular data were represented using a binary code. For each genotype, we assigned a value = 0 representing a down-regulation and a value = 1 representing an up-regulation at each time point, whatever the size (see Figure 2). The analysis was carried out at the most relevant times for each trait analysed, that is, T2 for grain yield (GY) and T6 for thousand kernel weight (TKW), and was extended to neighbour points T1 and T7, respectively. The contrast between the molecular data (up- and down-regulation of transcripts) and the traits analysed is shown in Table 5.

Table 5.
Statistical association between molecular and agronomical data under RI condition. Molecular data were represented as a binary code and agronomical traits are referred to as GY and TKW.
At T2, the TdDRF1.1 transcript was significantly associated to GY, with p < 0.01 and all up-regulated genotypes showing a mean GY significantly higher than Creso (down-regulated). TdDRF1.2 was also significantly associated (p < 0.01) to the yield and Barnacla (up-regulated) showed a GY value significantly higher than the mean GY of all others. On widening the analysis to T1, Barnacla, characterized by up-regulation of the TdDRF1.3 transcript, again showed a GY value significantly higher than all the others.
Considering the other agronomic trait, TKW, a significant association was found at T6 for TdDRF1.2, and the down-regulated genotypes (Creso and Barnacla) showed a TKW value significantly higher than that of the up-regulated group (Duilio, AEL, and Colosseo). On the other hand, widening the analysis to T7, TdDRF1.1 was also significantly associated to TKW and the group of down-regulated genotypes (Duilio, Creso, and Colosseo) showed a mean TKW value significantly higher than the up-regulated group (AEL and Barnacla).
3.4. Expression Profile of the Wdhn13 Gene
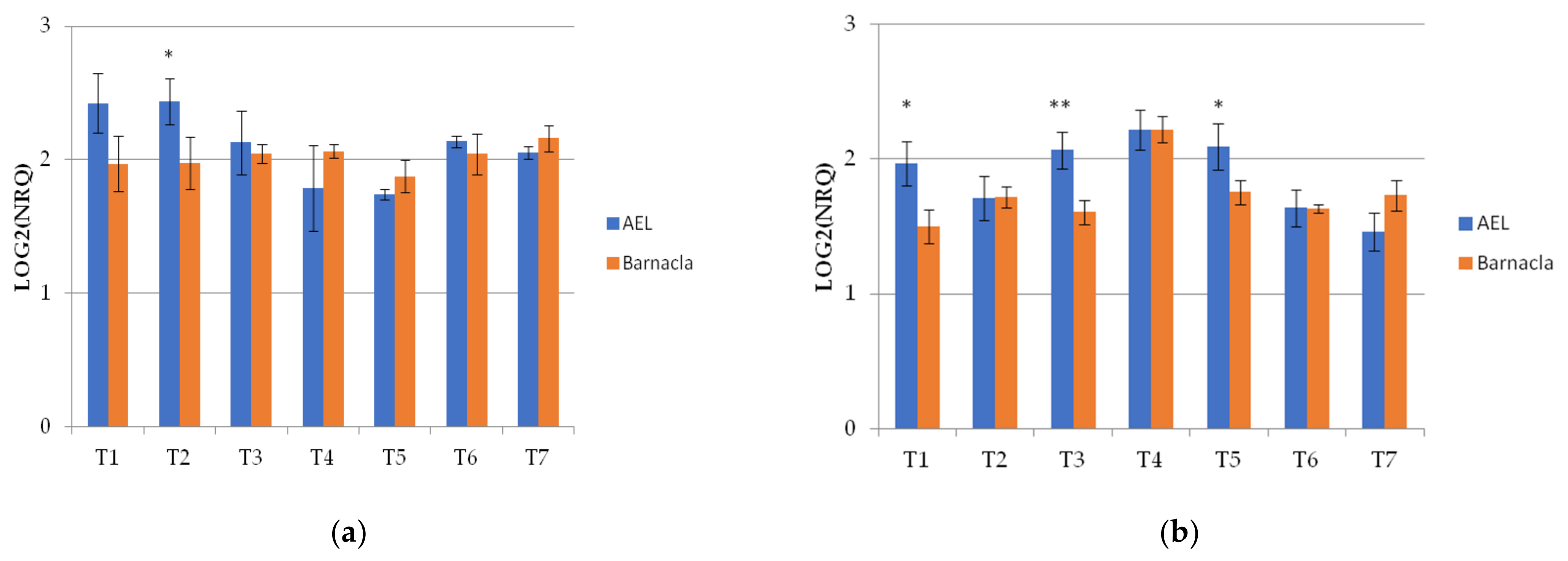
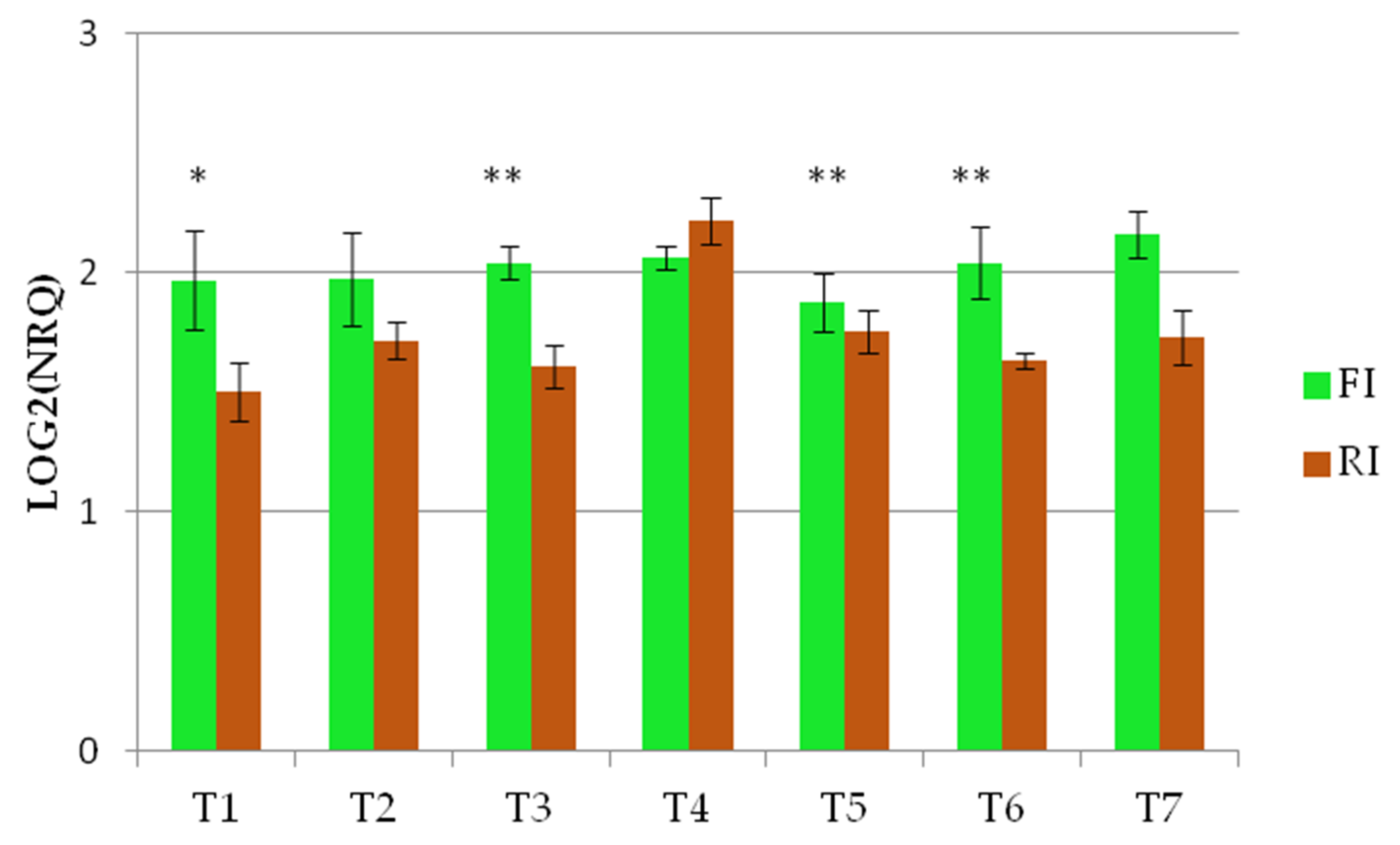
The Wdhn13 gene encodes for LEA D-11 DHN (dehydrin), which has been reported to respond to water stress in wheat plants [21,24]. The Wdhn13 gene transcript was analysed in two genotypes, AEL and Barnacla, under both irrigation conditions. Regardless of genotypes, significant differences of the Wdhn13 gene transcript were found between FI and RI conditions at T1, T2, T4, T6, and T7, as shown in Table 6. The expression profiles of the Wdhn13 gene transcript obtained analysing the environments separately in two genotypes are reported in Figure 3. Figure 3a refers to the full irrigation condition, while Figure 3b refers to the reduced irrigation condition. No significant differences were found between genotypes under FI, with the exception of T2 (p < 0.05), while significant differences were observed under RI (at T1, T3, and T5).

Table 6.
ANOVA summery table. Significant differences in Wdhn13 gene transcript between FI and RI conditions during the time-course.
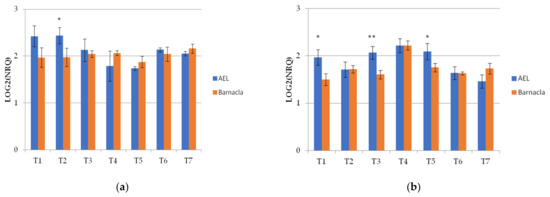
Figure 3.
The expression profiles of Wdhn13 gene transcript in the two genotypes measured under FI and RI conditions. (a) The expression profiles of Wdhn13 transcript in AEL and Barnacla observed under FI condition. (b) The expression profiles of Wdhn13 transcript in AEL and Barnacla observed under RI condition. Data are represented as mean ± SE. Significance codes: ** p < 0.01, * p < 0.05.
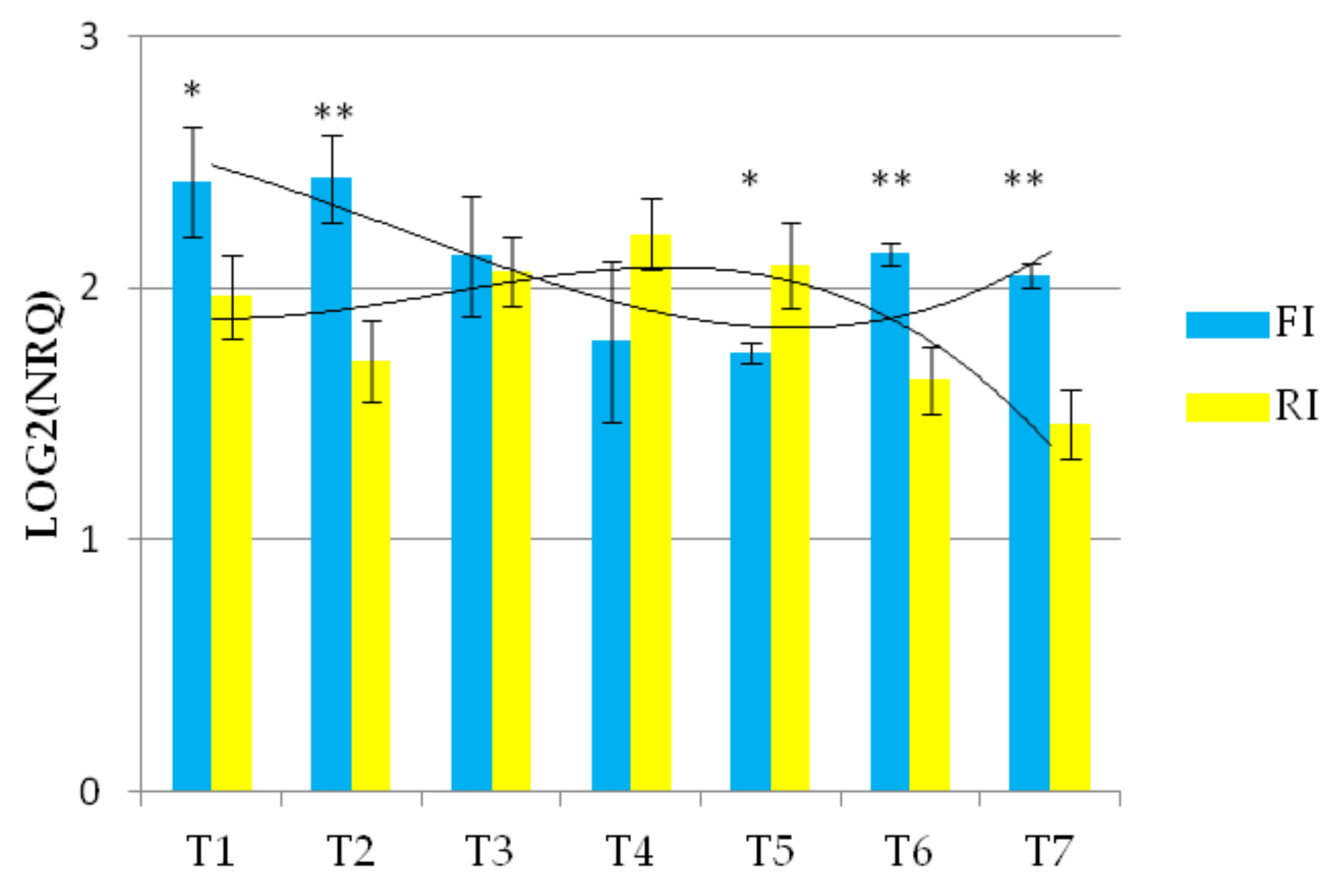
The expression of the Wdhn13 gene transcript in AEL is shown in Figure 4 and demonstrated significant differences under the two conditions, showing almost opposite concavities crossing at T3 and T6, suggesting up-regulation under reduced irrigation.
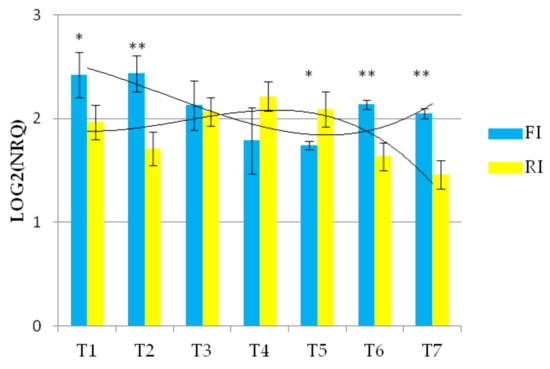
Figure 4.
The expression profile of Wdhn13 gene transcript of AEL under FI and RI conditions. A third-order polynomial trendline was added to highlight the opposite concavities; ** p < 0.01, * p < 0.05.
The Wdhn13 gene transcript profiles were also different between the two conditions in Barnacla, showing a larger variability under reduced irrigation, while the expression levels under full irrigation were almost stable (Figure 5).
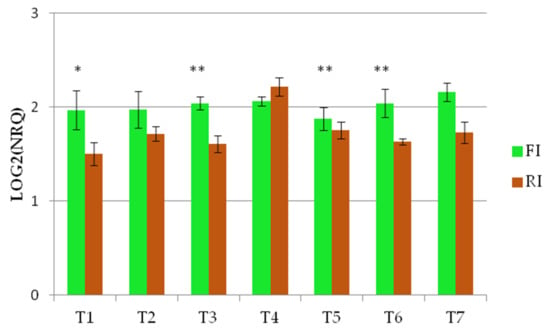
Figure 5.
The expression profile of Wdhn13 transcript of Barnacla under FI and RI conditions ** p < 0.01, * p < 0.05.
4. Discussion
This study was aimed at analysing and comparing the expression levels of the TdDRF1 gene in the field under two different irrigation conditions, i.e., full and reduced irrigation (FI and RI, respectively), taking into account the reported involvement of its transcripts under water stress. Six different durum wheat genotypes were studied with the aim of finding any different behaviours among the transcripts. Our results revealed significant differences in the TdDRF1 transcripts between the two irrigation conditions at different time points (Table 4). Both the TdDRF1.1 and TdDRF1.3 transcripts showed significant differences between the two conditions at T4, suggesting their full involvement in drought response, which continued with TdDRF1.1 at T5 and TdDRF1.3 at T7, pointing to their possible different role in the trans-activation throughout the time. On the other hand, these results could reflect the activity of the alternative splicing mechanism during the time-course, producing and cumulating the three transcripts in different ways as a response to the environment [25]. The modulation of the TdDRF1.2 transcript is particularly interesting, as the latter is not directly involved in the downstream gene modulation but, with high probability, plays an important role in gene regulation through its expression and degradation via nonsense-mediated mRNA decay or other RNA surveillance mechanisms during transcript maturation, as reported for the abortive forms produced by the alternative splicing mechanism [26,27].
Looking at the trends shown in Figure 1 as a whole, every genotype seemed to have its own transcripts profile, depending mainly on genetics rather than environmental effects, as was previously observed [11,12,28]. The TdDRF1.1 transcript remained almost steady across the time-course in both conditions, with few exceptions other than the notable behaviour of Barnacla under the reduced irrigation condition. Since the TdDRF1.1 transcript results from the junction of all four exons (E1-E2-E3-E4) present in the gene sequence, it could be speculated that the quantity of this transcript, as the first product obtained, remains lower than the other ones, taking into account another mechanism very common in plants, known as intron retention [29], which could explain the formation of TdDRF1.2 (E1-E2-E4) and TdDRF1.3 (E1-E4) transcripts from an immature TdDRF1.1 transcript. In addition to a relatively high value for the TdDRF1.1 transcript, Barnacla also showed an opposite behaviour to the others under reduced irrigation. In the other genotypes, the TdDRF1.3 transcript under full irrigation increased throughout the time-course, with some exceptions and to different extents, with Duilio up to 10-fold. Under reduced irrigation, Duilio and AEL did not change their initial expression levels, while both Creso and Colosseo showed substantial increases. It is important to underline that all the results are based on a time-course spanning 124 days from sowing with a seven-day window (Table 2); it could be possible that, in some cases and particularly under reduced irrigation, the window was too large to capture the expression modulations based on early and short responses. The a priori selection of an experimental design aimed at analysing gene expression in the field is a challenging task, particularly when the study involves transcription factors whose few molecules are sufficient to respond promptly to the stress by producing a large transcriptional burst of stress-responsive gene expression in a short period. Furthermore, in the field, plants may experience several distinct abiotic stresses either concurrently or at different times through the growing season [30], and consequently plant responses to water scarcity are very complex [31,32]. Finally, durum wheat is an allopolyploid (tetraploid) species and, due to its large genome size and the high levels of sequence similarity between the chromosomes duplicated, it is traditionally difficult to analyse [33]. Undeniably, for all the above-mentioned reasons, the number of gene expression studies using plant materials from the field is much less covered by the scientific literature than those under controlled environmental conditions.
With the results represented as the fold change, it is possible to state the expression levels in terms of down- and up-regulation of transcripts. It is worth noting that Duilio and AEL showed similar trends for all transcripts with very few differences: TdDRF1.1 was always up-regulated, while TdDRF1.3 was always down-regulated at all times. This molecular behaviour may suggest that the early stress response, aimed at activating in trans downstream genes, is mainly based on the 1.1 isoform. On the whole, the three transcripts of TdDRF1 in Creso were strongly inhibited by water stress, possibly suggesting that this genotype employs other genes to cope with drought stress. Colosseo showed a certain similarity with Duilio and AEL with regard to the TdDRF1.1 transcript. The molecular behaviour of the TdDRF1 gene in Barnacla was unique, as all transcripts were largely up-regulated, suggesting a continuous transcription process that cumulates the three transcripts through a very articulated and complex control of alternative splicing [34]. Interestingly, in the contrast between the molecular and agronomic data, the up-regulation of the TdDRF1.2 and TdDRF1.3 transcripts, at T2 and T1, respectively, was significantly associated with grain yield, with Barnacla showing a value higher than the mean of all the others. The significant associations found by the statistical analyses are intriguing, but further studies at a molecular level and with a larger panel of different genotypes are necessary to better clarify the role of the transcripts in field stress response and to monitor their possible effects on the different stages of plant development and maturation.
A further aim of this work was to highlight the possible relationship between the TdDRF1 transcripts and the Wdhn13 gene, involved in environmental stress tolerance. In a recent study, Mehrabad Pour-Benab and colleagues [35] investigated dehydrin expression in different species of Triticum and Aegilops under well-watered and drought stress treatments in the greenhouse. Thirty days after applying the water stress, they observed the doubling of the Wdhn13 expression level in water-stressed plants in the different wheat species. Our analysis in the field showed significant differences in the Wdhn13 transcript levels between FI and RI conditions, at every time point except T3 and T5. Similarly, the TdDRF1 transcripts were also found to be significantly different between the two conditions throughout the time-course. It could be speculated that the largest Wdhn13 differences at T6 and T7 reflect the activity of transcription factors (TdDRF1.1 at T5 and TdDRF1.3 at T7) on the promoter of the Wdhn13 gene, resulting finally in the dehydrin protein, involved in the drought stress tolerance at a cellular level.
Our results showed that the molecular behaviour of specific plant genes is highly dynamic and much more complex than a direct on/off switching. Furthermore, they confirm the main effect of genotype on the TdDRF1 gene expression profile and, in this regard, the molecular behaviour of Barnacla appears particularly interesting, as it is the only genotype showing both transcription factors active under reduced irrigation during the time-course, even if to different extents. It is particularly intriguing that the grain yield value of Barnacla was the highest and was statistically associated with the up-regulation of two transcripts under reduced irrigation, thus conferring an added value to this genotype.
In conclusion, notwithstanding the intrinsic difficulty of identifying the effect of a single factor in the complex picture of gene expression, this work represents a remarkable contribution to highlighting the role of the TdDRF1 gene in field conditions. Further experiments are necessary with a larger panel of genotypes and set of genes (transcription factors and target genes) to better understand the molecular plant response to drought, with the final aim of setting up a support tool to assist in orienting breeders’ decisions.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes13030555/s1, Figure S1: Specific amplification and detection of the three TdDRF1 transcripts, Figure S2: Schematic representation of the complete procedure of reverse transcription, pre-amplification, and qRT-PCR, Table S1: Gene-stability values (M) of the analysed reference genes.
Author Contributions
P.G. coordinated the project; A.L., K.A., and P.G. designed the experimental work; K.A. managed the field experiment and elaborated the agronomic data; A.L. and K.T. carried out the molecular analyses; C.C. carried out bioinformatics and the statistical analyses; A.L., C.C., and P.G. analysed the data and interpreted the results; C.C. and P.G. developed the first draft; K.A. and A.L. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This study was partially supported by the High Relevance Mexico–Italy Project of the Italian Foreign Affairs Ministry and by the Italian RIADE project (Integrated research for applying new technologies and processes for combating desertification). A.L. was supported by Fellowships of the Government of Mexico, “Secretaría de Relaciones Exteriores” for two internships at CIMMYT (Mexico). K.T. was supported by an ENEA International 2-year postdoc Fellowship at ENEA Casaccia Research Centre, Italy.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
All authors gave the consent to the acknowledgement. The authors are sincerely grateful to Chiara Rasi, Domenico Di Bianco, and Fabio Felici for their valuable work in the field during the time-course experiment; to the technical staff of CIMMYT experimental station (CENEB) near Cd. Obregón for their care and the valuable field work; to Giulio Marconi (ENEA library service) for his constant assistance; and to Ian Pace, professional mother-tongue reviewer, for the revision of the English text.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study or in the collection, analyses, and interpretation of data.
Abbreviations
AEL, Advanced experimental line; DRE, drought-responsive element; DREB, dehydration responsive element binding gene; FC, fold change; FI, full irrigation; GY, grain yield; NRQ, normalized relative quantity of transcript; RI, reduced irrigation; TdDRF1, Triticum durum dehydration responsive factor 1 gene; TKW, thousand kernel weight; TF, transcription factor; Wdhn13, wheat dehydrin 13 gene.
References
- Raza, A.; Razzaq, A.; Mehmood, S.S.; Zou, X.; Zhang, X.; Lv, Y.; Xu, J. Impact of climate change on crops adaptation and strategies to tackle its outcome: A review. Plants 2019, 8, 34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pareek, A.; Dhankher, O.P.; Foyer, C.H. Mitigating the impact of climate change on plant productivity and ecosystem sustainability. J. Exp. Bot. 2020, 71, 451–456. [Google Scholar] [CrossRef] [PubMed]
- FAO. The State of Food and Agriculture 2020. Overcoming Water Challenges in Agriculture; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Shewry, P.R.; Hey, S.J. The contribution of wheat to human diet and health. Food Energy Secur. 2015, 4, 178–202. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Ann. Review Plant Biol. 2006, 57, 781–803. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, P.K.; Gupta, K.; Lopato, S.; Agarwal, P. Dehydration responsive element binding transcription factors and their applications for the engineering of stress tolerance. J. Exp. Bot. 2017, 68, 2135–2148. [Google Scholar] [CrossRef]
- Faraji, S.; Filiz, E.; Kazemitabar, S.K.; Vannozzi, A.; Palumbo, F.; Barcaccia, G.; Heidari, P. The AP2/ERF gene family in Triticum durum: Genome-wide identification and expression analysis under drought and salinity stresses. Genes 2020, 11, 1464. [Google Scholar] [CrossRef]
- Lata, C.; Prasad, M. Role of DREBs in regulation of abiotic stress responses in plants. J. Exp. Bot. 2011, 62, 4731–4748. [Google Scholar] [CrossRef] [Green Version]
- Rabara, R.C.; Tripathi, P.; Rushton, P.J. The potential of transcription factor-based genetic engineering in improving crop tolerance to drought. OMICS 2014, 18, 601–614. [Google Scholar] [CrossRef] [Green Version]
- El-Esawi, M.A.; Al-Ghamdi, A.A.; Ali, H.M.; Ahmad, M. Overexpression of AtWRKY30 Transcription Factor Enhances Heat and Drought Stress Tolerance in Wheat (Triticum aestivum L.). Genes 2019, 10, 163. [Google Scholar] [CrossRef] [Green Version]
- Latini, A.; Rasi, C.; Sperandei, M.; Cantale, C.; Iannetta, M.; Dettori, M.; Ammar, K.; Galeffi, P. Identification of a DREB-related gene in Triticum durum and its expression under water stress conditions. Ann. Appl. Biol. 2007, 150, 187–195. [Google Scholar] [CrossRef]
- Latini, A.; Sperandei, M.; Cantale, C.; Arcangeli, C.; Ammar, K.; Galeffi, P. Variability and expression profile of the DRF1 gene in four cultivars of durum wheat and one triticale under moderate water stress conditions. Planta 2013, 237, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Latini, A.; Sperandei, M.; Sharma, S.; Cantale, C.; Iannetta, M.; Dettori, M.; Ammar, K.; Galeffi, P. Molecular analyses of a DREB-related gene in durum wheat and triticale. In Biosaline Agriculture and High Salinity Tolerance; Abdelly, C., Öztürk, M., Ashraf, M., Grignon, C., Eds.; Birkäuser Verlag AG: Basel, Switzerland, 2008; pp. 287–295. [Google Scholar]
- Catacchio, C.R.; Alagna, F.; Perniola, R.; Bergamini, C.; Rotunno, S.; Calabrese, F.M.; Crupi, P.; Antonacci, D.; Ventura, M.; Cardone, M.F. Transcriptomic and genomic structural variation analyses on grape cultivars reveal new insights into the genotype-dependent responses to water stress. Sci. Rep. 2019, 9, 2809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baker, C.R.; Stewart, J.J.; Amstutz, C.L.; Ching, L.G.; Johnson, J.D.; Niyogi, K.K.; Adams, W.W., 3rd; Demmig-Adams, B. Genotype-dependent contribution of CBF transcription factors to long-term acclimation to high light and cool temperature. Plant Cell Environ. 2021, 45, 392–411. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, V.M.; Santiago, R.; Malvar, R.A.; Butrón, A. Inducible maize defense mechanisms against the corn borer Sesamia nonagrioides: A transcriptome and biochemical approach. Mol. Plant Microbe Interact. 2012, 25, 61–68. [Google Scholar] [CrossRef] [Green Version]
- Sari, E.; Bhadauria, V.; Vandenberg, A.; Banniza, S. Genotype-dependent interaction of lentil lines with Ascochyta lentis. Front. Plant Sci. 2017, 8, 764. [Google Scholar] [CrossRef] [Green Version]
- Kam, J.; Gresshoff, P.; Shorter, R.; Xue, G.-P. Expression analysis of RING zinc finger genes from Triticum aestivum and identification of TaRZF70 that contains four RING-H2 domains and differentially responds to water deficit between leaf and root. Plant Sci. 2007, 173, 650–659. [Google Scholar] [CrossRef]
- Paolacci, A.R.; Tanzarella, O.A.; Porceddu, E.; Ciaffi, M. Identification and validation of reference genes for quantitative RT-PCR normalization in wheat. BMC Mol. Biol. 2009, 10, 11. [Google Scholar] [CrossRef] [Green Version]
- Rieu, I.; Powers, S.J. Real-time quantitative RT-PCR: Design, calculations, and statistics. Plant Cell 2009, 21, 1031–1033. [Google Scholar] [CrossRef] [Green Version]
- Ohno, R.; Takumi, S.; Nakamura, C. Kinetics of transcript and protein accumulation of a low-molecular-weight wheat LEA D-11 dehydrin in response to low temperature. J. Plant Physiol. 2003, 160, 193–200. [Google Scholar] [CrossRef]
- Kobayashi, F.; Takumi, S.; Egawa, C.; Ishibashi, M.; Nakamura, C. Expression patterns of low temperature responsive genes in a dominant ABA-less-sensitive mutant line of common wheat. Physiol. Plant. 2006, 127, 612–623. [Google Scholar] [CrossRef] [Green Version]
- Vítámvás, P.; Kosová, K.; Musilová, J.; Holková, L.; Mařík, P.; Smutná, P.; Klíma, M.; Prášil, I.T. Relationship between dehydrin accumulation and winter survival in winter wheat and barley grown in the field. Front. Plant Sci. 2019, 10, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Egawa, C.; Kobayashi, F.; Ishibashi, M.; Nakamura, T.; Nakamura, C.; Takumi, S. Differential regulation of transcript accumulation and alternative splicing of a DREB2 homolog under abiotic stress conditions in common wheat. Genes Genet. Syst. 2006, 81, 77–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, L.; Pan, Z.; Chen, L.; Dai, Y.; Wan, J.; Ye, H.; Nguyen, H.T.; Zhang, G.; Chen, H. Analysis of Whole Transcriptome RNA-seq Data Reveals Many Alternative Splicing Events in Soybean Roots under Drought Stress Conditions. Genes 2020, 11, 1520. [Google Scholar] [CrossRef] [PubMed]
- Dubrovina, A.S.; Kiseklev, K.V.; Zhuravlev, Y.N. The role of canonical and noncanonical pre-mRNA splicing in plant stress responses. Biomed Res Int. 2013, 264314. [Google Scholar] [CrossRef]
- Göhring, J.; Jacak, J.; Barta, A. Imaging of endogenous messenger RNA splice variants in living cells reveals nuclear retention of transcripts inaccessible to nonsense-mediated decay in Arabidopsis. Plant Cell 2014, 26, 754–764. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Able, A.J.; Able, J.A. Integrated analysis of small RNA, transcriptome, and degradome sequencing reveals the water-deficit and heat stress response network in durum wheat. Int. J. Mol. Sci. 2020, 21, 6017. [Google Scholar] [CrossRef]
- Iñiguez, L.P.; Hernández, G. The evolutionary relationship between alternative splicing and gene duplications. Front. Genet. 2017, 8, 14. [Google Scholar] [CrossRef] [Green Version]
- Tester, M.; Bacic, A. Abiotic stress tolerance in grasses. From model plants to crop plants. Plant Physiol. 2005, 137, 791–793. [Google Scholar] [CrossRef] [Green Version]
- Chaves, M.M.; Pereira, J.S.; Maroco, J.; Rodrigues, M.L.; Ricardo, C.P.P.; Osório, M.L.; Carvalho, I.; Faria, T.; Pinheiro, C. How plants cope with water stress in the field? Photosynthesis and growth. Ann. Bot. 2002, 89, 907–916. [Google Scholar] [CrossRef] [Green Version]
- Osakabe, Y.; Osakabe, K.; Shinozaki, K.; Tran, L.-S.P. Response of plants to water stress. Front. Plant Sci. 2014, 5, 86. [Google Scholar] [CrossRef] [Green Version]
- Kuo, T.; Hatakeyama, M.; Tameshige, T.; Shimizu, K.K.; Sese, J. Homeolog expression quantification methods for allopolyploids. Brief. Bioinform. 2020, 21, 395–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, M.; Manley, J.M. Mechanisms of alternative splicing regulation: Insights from molecular and genomics approaches. Nat. Rev. Mol. Cell Biol. 2009, 10, 741–754. [Google Scholar] [CrossRef] [PubMed]
- Mehrabad Pour-Benab, S.; Fabriki-Ourang, S.; Mehrabi, A.-A. Expression of dehydrin and antioxidant genes and enzymatic antioxidant defense under drought stress in wild relatives of wheat. Biotechnol. Biotechnol. Equip. 2019, 33, 1063–1073. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).