Bioinformatics Analysis of WRKY Family Genes in Erianthus fulvus Ness
Abstract
1. Introduction
2. Materials and Methods
2.1. Identification of the EfWRKY Gene Family Members
2.2. Predicted Physicochemical Properties and Secondary Structure Analysis of WRKY Protein in E. fulvus
2.3. Construction of a Phylogenetic Tree
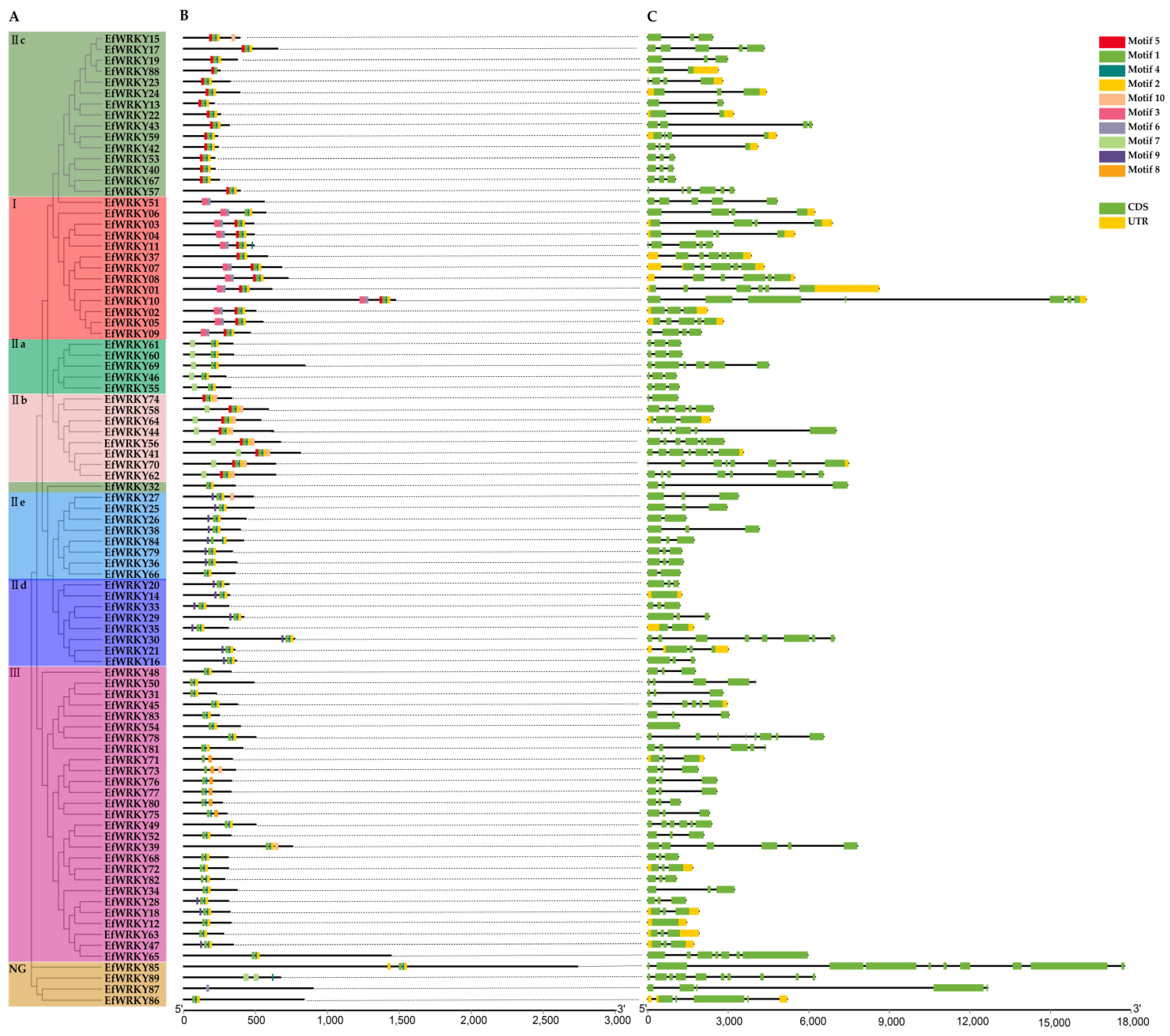
2.4. Analysis of Conserved Motifs, Gene Structure, and Chromosomal Localization of WRKY Proteins in E. fulvus
2.5. Chromosome Localization, Gene Duplication, and Covariance Analysis
2.6. Analysis of Cis-Acting Elements of Promoters
2.7. Gene Ontology (GO) Annotation
3. Results
3.1. Identification and Prediction of Physicochemical Properties of EfWRKY Gene Family Members
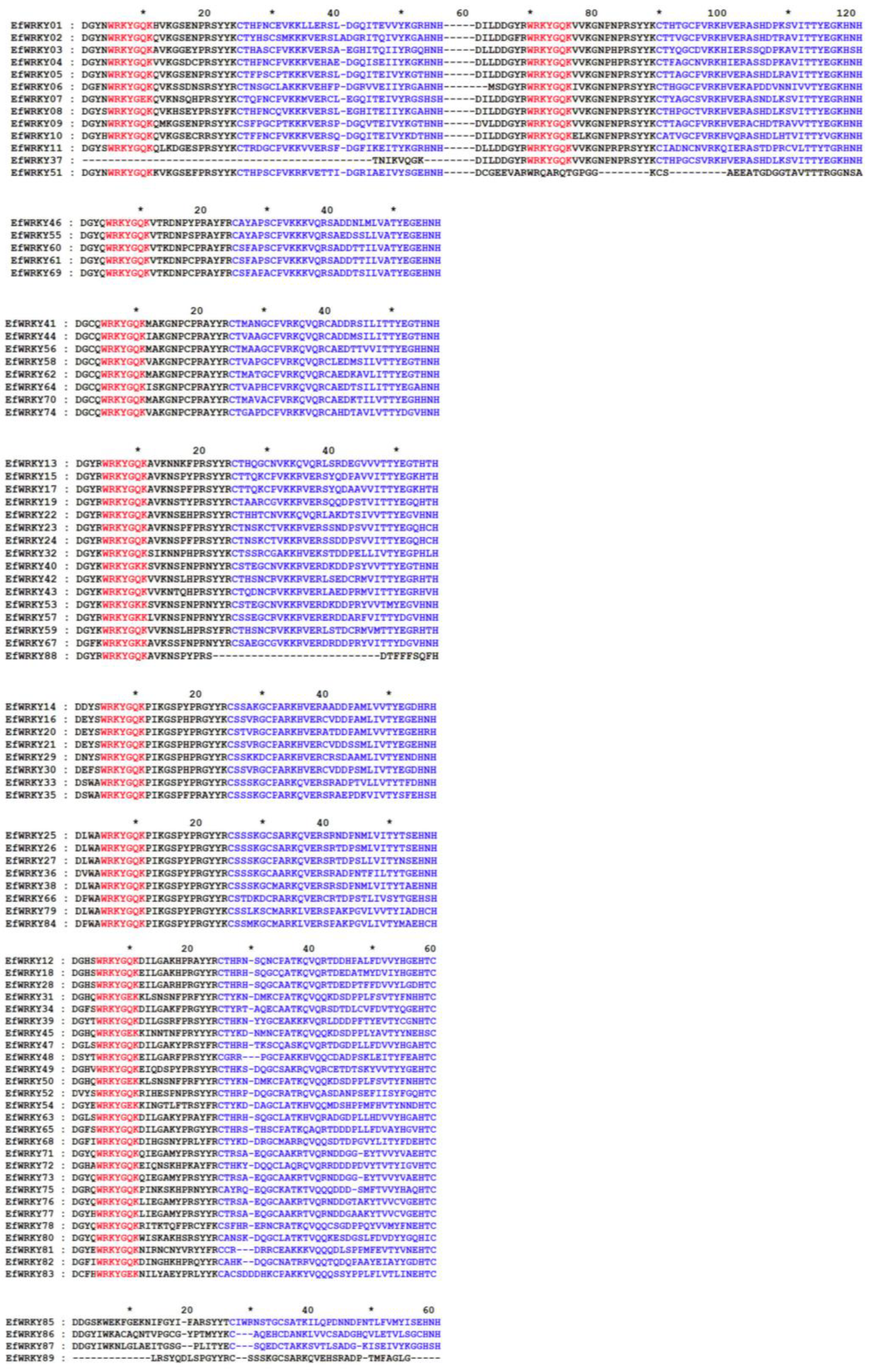
3.2. Secondary Structure Analysis of the89 WRKY Protein in E. fulvus
3.3. Phylogenetic, Conserved Motif, and Gene Structure Analysis of WRKY Gene Family Members in E. fulvus
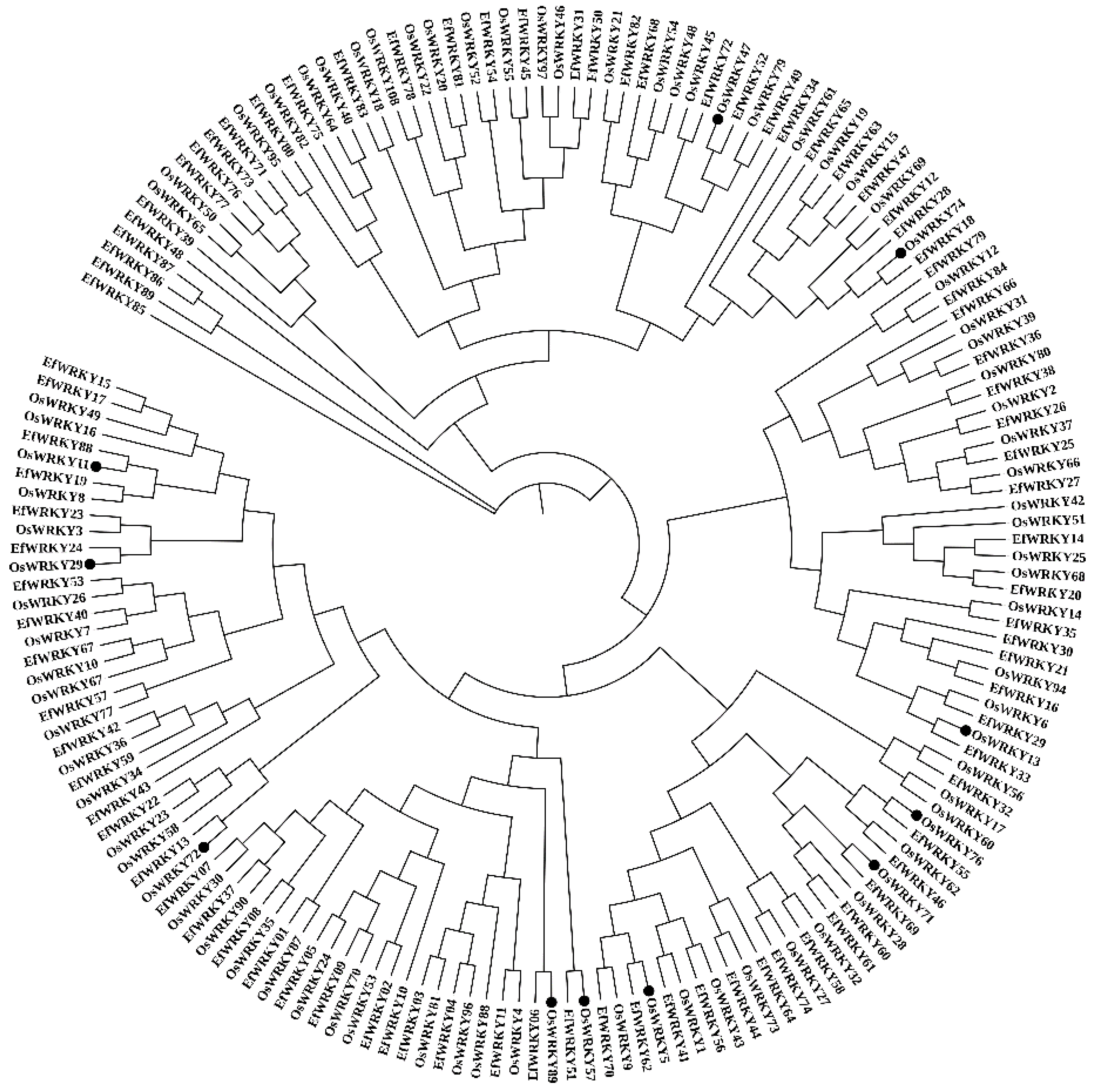
3.4. Phylogenetic and Functional Analysis of the OsWRKY and EfWRKY Gene Families
3.5. Analysis of Cis-Acting Elements of 89 EfWRKY Gene Promoters
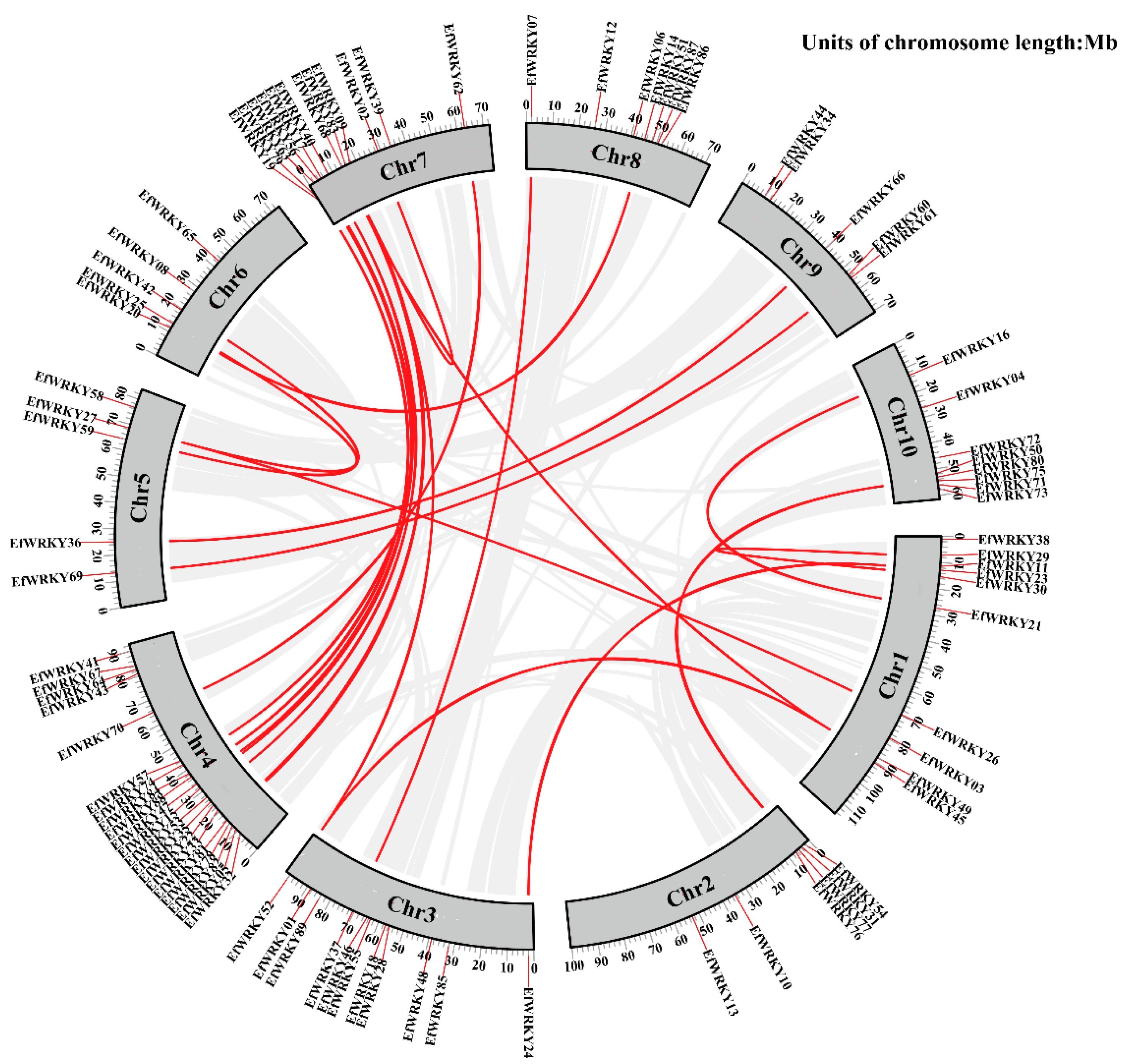
3.6. Chromosomal Localization Analysis of 89 EfWRKY Genes
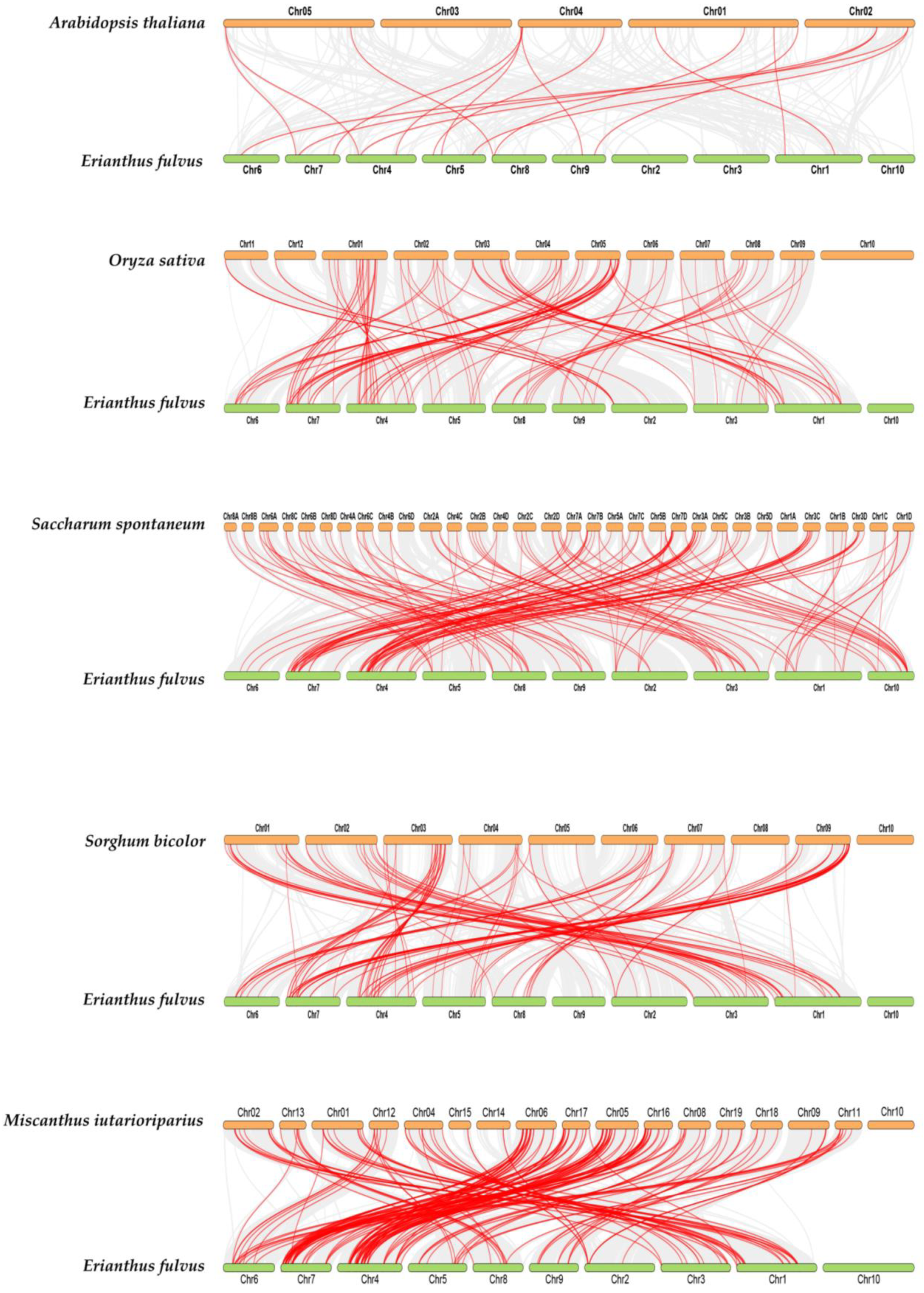
3.7. Synteny Relationship of EfWRKY Genes
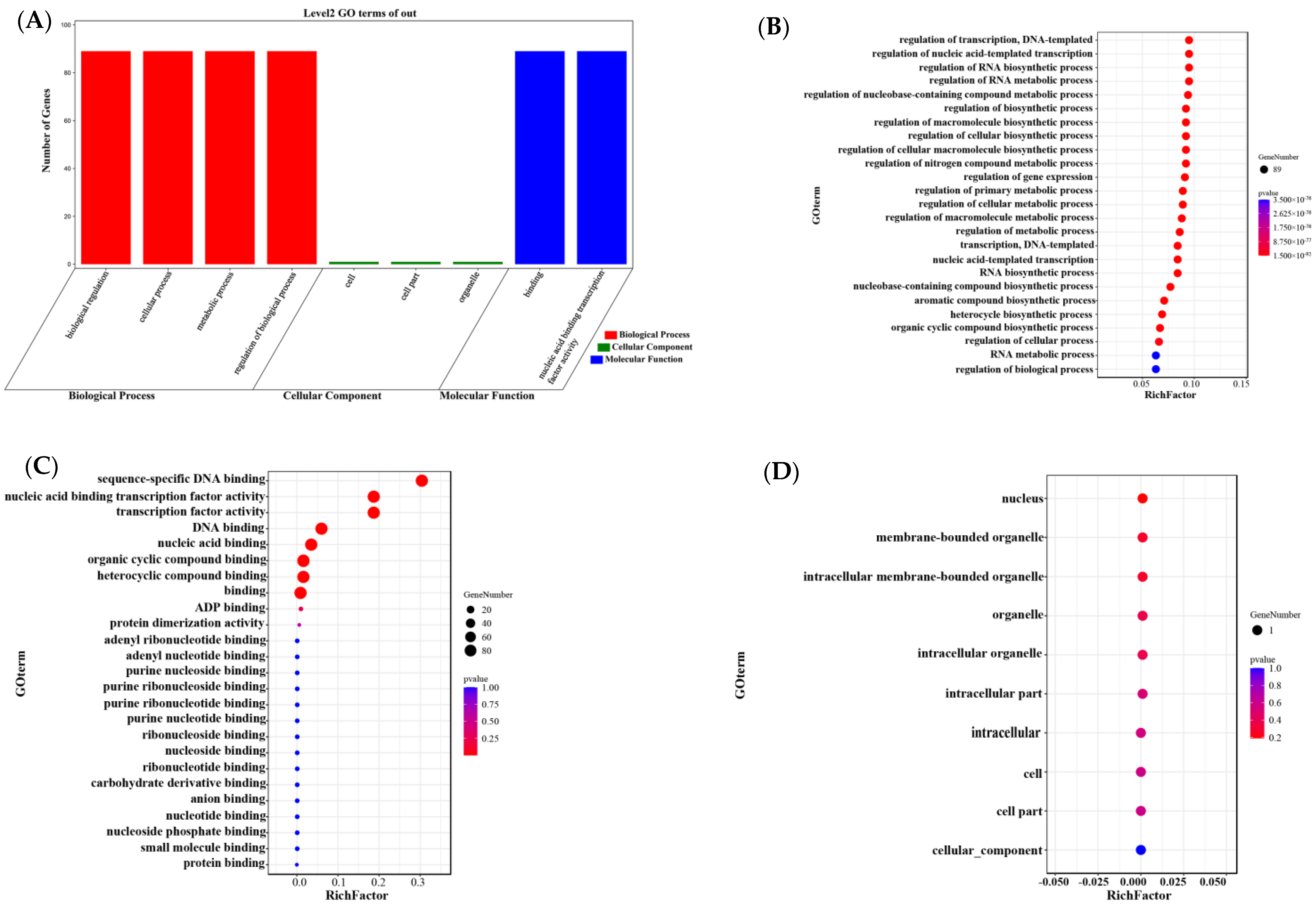
3.8. Gene Ontology (GO) Annotation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ji, Q.; Zhang, Y.; Deng, J. Research Progress of Frost on Sugarcane. Sugar Crop. China 2018, 40, 74–77. [Google Scholar]
- Gui-Hua, W. Value of Sugarcane as Feed and Its Developmental Prospect of Industrialization. Sugar Crop. China 2004, 54–56. [Google Scholar]
- Gentile, A.; Dias, L.I.; Mattos, R.S.; Ferreira, T.H.; Menossi, M. MicroRNAs and drought responses in sugarcane. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, S.; Dao, J.; Deng, J.; Shahzad, A.N.; Fan, X.; Li, R.; Quan, Y.; Bukhari, S.A.H.; Zeng, Z. Drought-induced alterations in photosynthetic, ultrastructural and biochemical traits of contrasting sugarcane genotypes. PLoS ONE 2020, 15, e0235845. [Google Scholar] [CrossRef]
- Shengsheng, L.; Weiwei, L.; Haiqiu, W.; Hongliang, H.; Yaqin, J.; Liqiu, T. Research Progress of Drought Stress on Sugarcane Growth. China Trop. Agric. 2020, 5, 70–73. [Google Scholar]
- Haibi, L.; Yiyun, G.; Ronghua, Z.; Jinju, W.; Rongzhong, Y.; Xiaoqiu, Z.; Rui, L.; Hui, Z.; Xihui, L. Research Progress on Drought Resistance and Drought-Resistant Breeding of Sugarcane. Mol. Plant Breed. 2019, 17, 3406–3415. [Google Scholar]
- Chao-Hua, X.; Xin, L.; Xin-Long, L.; Huo-Sheng, S.; Li, M.; Jun, M.; Xiu-Qin, L.; Xu-Juan, L.; Qing, C. Exploration and Phenotypic Traits Analysis of Erianthus fulvus. J. Plant Genet. Resour. 2014, 15, 962–966. [Google Scholar]
- Paul, J.R.; Imre, E.S.; Patricia, R.; Qingxi, J.S. WRKY transcription factors. Trends Plant Sci. 2010, 15, 247–258. [Google Scholar]
- Thomas, E.; Paul, J.R.; Silke, R.; Imre, E.S. The WRKY superfamily of plant transcription factors. Trends Plant Sci. 2000, 5, 199–206. [Google Scholar]
- Rushton, P.J.; Macdonald, H.; Huttly, A.K.; Lazarus, C.M.; Hooley, R. Members of a new family of DNA-binding proteins bind to a conserved cis-element in the promoters of α-Amy2 genes. Plant Mol. Biol. 1995, 29, 691–702. [Google Scholar] [CrossRef]
- Eulgem, T.; Rushton, P.J.; Schmelzer, E.; Hahlbrock, K.; Somssich, I.E. Early nuclear events in plant defence signalling: Rapid gene activation by WRKY transcription factors. Embo J. 1999, 18, 4689–4699. [Google Scholar] [CrossRef] [PubMed]
- Rushton, P.J.; Torres, J.T.; Parniske, M.; Wernert, P.; Hahlbrock, K.; Somssich, I.E. Interaction of elicitor-induced DNA-binding proteins with elicitor response elements in the promoters of parsley PR1 genes. Embo J. 1996, 15, 5690–5700. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.M.; Shi, Y. The galvanization of biology: A growing appreciation for the roles of zinc. Science 1996, 271, 1081–1085. [Google Scholar] [CrossRef]
- Cormack, R.S.; Eulgem, T.; Rushton, P.J.; Kochner, P.; Hahlbrock, K.; Somssich, I.E. Leucine zipper-containing WRKY proteins widen the spectrum of immediate early elicitor-induced WRKY transcription factors in parsley. Biochim. Biophys. Acta 2002, 1576, 92–100. [Google Scholar] [CrossRef]
- Ishiguro, S.; Nakamura, K. Characterization of a cDNA encoding a novel DNA-binding protein, SPF1, that recognizes SP8 sequences in the 5′ upstream regions of genes coding for sporamin and β-amylase from sweet potato. Mol. Gen. Genet. 1994, 244, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, X.; Han, J.; Lu, W.; Ren, Z. Genome-wide analysis of the WRKY gene family in the cucumber genome and transcriptome-wide identification of WRKY transcription factors that respond to biotic and abiotic stresses. BMC Plant Biol. 2020, 20, 443. [Google Scholar] [CrossRef] [PubMed]
- Suhas, G.K.; Ranjit, S.G.; Ashutosh, R.; Moin, A.; Major, S.; Bijendra, S. Genome wide expression analysis of WRKY genes in tomato (Solanum lycopersicum) under drought stress. Plant Gene 2018, 13, 8–17. [Google Scholar]
- Waqas, M.; Azhar, M.T.; Rana, I.A.; Azeem, F.; Ali, M.A.; Nawaz, M.A.; Chung, G.; Atif, R.M. Genome-wide identification and expression analyses of WRKY transcription factor family members from chickpea (Cicer arietinum L.) reveal their role in abiotic stress-responses. Genes Genom. 2019, 41, 467–481. [Google Scholar] [CrossRef]
- Skibbe, M.; Qu, N.; Galis, I.; Baldwin, I.T. Induced plant defenses in the natural environment: Nicotiana attenuata WRKY3 and WRKY6 coordinate responses to herbivory. Plant Cell 2008, 20, 1984–2000. [Google Scholar] [CrossRef]
- Grunewald, W.; Karimi, M.; Wieczorek, K.; Van de Cappelle, E.; Wischnitzki, E.; Grundler, F.; Inze, D.; Beeckman, T.; Gheysen, G. A role for AtWRKY23 in feeding site establishment of plant-parasitic nematodes. Plant Physiol. 2008, 148, 358–368. [Google Scholar] [CrossRef]
- Zou, X.; Seemann, J.R.; Neuman, D.; Shen, Q.J. A WRKY gene from creosote bush encodes an activator of the abscisic acid signaling pathway. J. Biol. Chem. 2004, 279, 55770–55779. [Google Scholar] [CrossRef] [PubMed]
- Yuping, Q.; Diqiu, Y. Over-expression of the stress-induced OsWRKY45 enhances disease resistance and drought tolerance in Arabidopsis. Environ. Exp. Bot. 2008, 65, 35–47. [Google Scholar]
- Jiang, Y.; Deyholos, M.K. Functional characterization of Arabidopsis NaCl-inducible WRKY25 and WRKY33 transcription factors in abiotic stresses. Plant Mol. Biol. 2009, 69, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Lagacé, M.; Matton, D.P. Characterization of a WRKY transcription factor expressed in late torpedo-stage embryos of Solanum chacoense. Planta 2004, 219, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Krassimira, S.A.; Conger, B.V. Isolation of two somatic embryogenesis-related genes from orchardgrass (Dactylis glomerata). Plant Sci. 2002, 162, 301–307. [Google Scholar]
- Robatzek, S.; Somssich, I.E. Targets of AtWRKY6 regulation during plant senescence and pathogen defense. Gene. Dev. 2002, 16, 1139–1149. [Google Scholar] [CrossRef]
- Robatzek, S.; Somssich, I.E. A new member of the Arabidopsis WRKY transcription factor family, AtWRKY6, is associated with both senescence- and defence-related processes: AtWRKY6 is associated with senescence and defence. Plant J. 2001, 28, 123–133. [Google Scholar] [CrossRef]
- Qian, Z.; Li, X.; He, L.; Gu, S.; Shen, Q.; Rao, X.; Zhang, R.; Di, Y.; Xie, L.; Wang, X.; et al. EfGD: The Erianthus fulvus genome database. Database 2022, 2022, baac076. [Google Scholar] [CrossRef]
- Mistry, J.; Finn, R.D.; Eddy, S.R.; Bateman, A.; Punta, M. Challenges in homology search: HMMER3 and convergent evolution of coiled-coil regions. Nucleic Acids Res. 2013, 41, e121. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Bailey, T.L.; Boden, M.; Buske, F.A.; Frith, M.; Grant, C.E.; Clementi, L.; Ren, J.; Li, W.W.; Noble, W.S. MEME SUITE: Tools for motif discovery and searching. Nucleic Acids Res. 2009, 37, w202–w208. [Google Scholar] [CrossRef]
- Chengjie, C.; Hao, C.; Yi, Z.; Hannah, R.T.; Margaret, H.F.; Yehua, H.; Rui, X. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar]
- He, C.; Teixeira, D.S.J.A.; Tan, J.; Zhang, J.; Pan, X.; Li, M.; Luo, J.; Duan, J. A Genome-Wide Identification of the WRKY Family Genes and a Survey of Potential WRKY Target Genes in Dendrobium officinale. Sci. Rep. 2017, 7, 9200. [Google Scholar] [CrossRef]
- Wang, H.; Hao, J.; Chen, X.; Hao, Z.; Wang, X.; Lou, Y.; Peng, Y.; Guo, Z. Overexpression of rice WRKY89 enhances ultraviolet B tolerance and disease resistance in rice plants. Plant Mol. Biol. 2007, 65, 799–815. [Google Scholar] [CrossRef] [PubMed]
- Chi-Yeol, K.; Kieu, T.X.V.; Cong, D.N.; Dong-Hoon, J.; Sang-Kyu, L.; Manu, K.; Sung-Ryong, K.; Su-Hyun, P.; Ju-Kon, K.; Jong-Seong, J. Functional analysis of a cold-responsive rice WRKY gene, OsWRKY71. Plant Biotechnol. Rep. 2016, 10, 13–23. [Google Scholar]
- Taehoon, K.; Kiyoon, K.; Suk-Hwan, K.; Gynheung, A.; Nam-Chon, P. OsWRKY5 Promotes Rice Leaf Senescence via Senescence-Associated NAC and Abscisic Acid Biosynthesis Pathway. Int. J. Mol. Sci. 2019, 20, 4437. [Google Scholar]
- Zhou, C.; Lin, Q.; Lan, J.; Zhang, T.; Liu, X.; Miao, R.; Mou, C.; Nguyen, T.; Wang, J.; Zhang, X.; et al. WRKY Transcription Factor OsWRKY29 Represses Seed Dormancy in Rice by Weakening Abscisic Acid Response. Front. Plant Sci. 2020, 11, 691. [Google Scholar] [CrossRef]
- Wu, X.; Shiroto, Y.; Kishitani, S.; Ito, Y.; Toriyama, K. Enhanced heat and drought tolerance in transgenic rice seedlings overexpressing OsWRKY11 under the control of HSP101 promoter. Plant Cell Rep. 2009, 28, 21–30. [Google Scholar] [CrossRef]
- Xiao, J.; Cheng, H.; Li, X.; Xiao, J.; Xu, C.; Wang, S. Rice WRKY13 regulates cross talk between abiotic and biotic stress signaling pathways by selective binding to different cis-elements. Plant Physiol. 2013, 163, 1868–1882. [Google Scholar] [CrossRef]
- Raineri, J.; Wang, S.; Peleg, Z.; Blumwald, E.; Chan, R.L. The rice transcription factor OsWRKY47 is a positive regulator of the response to water deficit stress. Plant Mol. Biol. 2015, 88, 401–413. [Google Scholar] [CrossRef]
- Holub, E.B. The arms race is ancient history in Arabidopsis, the wildflower. Nat. Rev. Genet. 2001, 2, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Hou, X.; Xie, K.; Xiong, L. Genome-wide identification of BURP domain-containing genes in rice reveals a gene family with diverse structures and responses to abiotic stresses. Planta 2009, 230, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Xiao, J.; Li, X.; Zhang, Q.; Lian, X. Sequence and expression analysis of the C3HC4-type RING finger gene family in rice. Gene 2009, 444, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Yue, G.; Hu, X.; He, Y.; Yang, A.; Zhang, J. Identification and characterization of two members of the FtsH gene family in maize (Zea mays L.). Mol. Biol. Rep. 2010, 37, 855–863. [Google Scholar] [CrossRef]
- Yang, L.; Zhu, H.; Guo, W.; Zhang, T. Molecular cloning and characterization of five genes encoding pentatricopeptide repeat proteins from Upland cotton (Gossypium hirsutum L.). Mol. Biol. Rep. 2010, 37, 801–808. [Google Scholar] [CrossRef]
- Xu, P.; Zhu, H.L.; Xu, H.B.; Zhang, Z.Z.; Zhang, C.Q.; Zhang, L.X.; Ma, Z.Q. Composition and phylogenetic analysis of wheat cryptochrome gene family. Mol. Biol. Rep. 2010, 37, 825–832. [Google Scholar] [CrossRef]
- Wang, Y.; Deng, D.; Bian, Y.; Lv, Y.; Xie, Q. Genome-wide analysis of primary auxin-responsive Aux/IAA gene family in maize (Zea mays. L.). Mol. Biol. Rep. 2010, 37, 3991–4001. [Google Scholar] [CrossRef]
- Gollan, P.J.; Bhave, M. Genome-wide analysis of genes encoding FK506-binding proteins in rice. Plant Mol. Biol. 2010, 72, 1. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, X.; Yu, H.; Ouyang, Y.; Wang, L.; Zhang, Q. The ankyrin repeat gene family in rice: Genome-wide identification, classification and expression profiling. Plant Mol. Biol. 2009, 71, 207–226. [Google Scholar] [CrossRef]
- Dai, X.; You, C.; Wang, L.; Chen, G.; Zhang, Q.; Wu, C. Molecular characterization, expression pattern, and function analysis of the OsBC1L family in rice. Plant Mol. Biol. 2009, 71, 469–481. [Google Scholar] [CrossRef]
- Ma, Z.; Li, W.; Wang, H.; Yu, D. WRKY transcription factors WRKY12 and WRKY13 interact with SPL10 to modulate age-mediated flowering. J. Integr. Plant Biol. 2020, 62, 1659–1673. [Google Scholar] [CrossRef] [PubMed]
- Zhong-Qi, F.; Xiao-Li, T.; Wei, S.; Jian-Fei, K.; Wang-Jin, L.; Jian-Ye, C.; Hikmet, B. BrWRKY65, a WRKY Transcription Factor, Is Involved in Regulating Three Leaf Senescence-Associated Genes in Chinese Flowering Cabbage. Int. J. Mol. Sci. 2017, 18, 1228. [Google Scholar]
- Lei, R.; Li, X.; Ma, Z.; Lv, Y.; Hu, Y.; Yu, D. Arabidopsis WRKY2 and WRKY34 transcription factors interact with VQ20 protein to modulate pollen development and function. Plant J. Cell Mol. Biol. 2017, 91, 962–976. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Yue, C.; Chen, D.; Zheng, Y.; Zhang, Q.; Yang, J.; Ye, N. Genome-wide identification of WRKY family genes and their response to abiotic stresses in tea plant (Camellia sinensis). Genes Genom. 2019, 41, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Cha, J.; Choi, C.; Choi, N.; Ji, H.; Park, S.R.; Lee, S.; Hwang, D. Rice WRKY11 Plays a Role in Pathogen Defense and Drought Tolerance. Rice 2018, 11, 5. [Google Scholar] [CrossRef] [PubMed]
- Yokotani, N.; Sato, Y.; Tanabe, S.; Chujo, T.; Shimizu, T.; Okada, K.; Yamane, H.; Shimono, M.; Sugano, S.; Takatsuji, H.; et al. WRKY76 is a rice transcriptional repressor playing opposite roles in blast disease resistance and cold stress tolerance. J. Exp. Bot. 2013, 64, 5085–5097. [Google Scholar] [CrossRef]
- Tripathi, P.; Rabara, R.C.; Langum, T.J.; Boken, A.K.; Rushton, D.L.; Boomsma, D.D.; Rinerson, C.I.; Rabara, J.; Reese, R.N.; Chen, X.; et al. The WRKY transcription factor family in Brachypodium distachyon. BMC Genom. 2012, 13, 270. [Google Scholar] [CrossRef]
- van Verk, M.C.; Pappaioannou, D.; Neeleman, L.; Bol, J.F.; Linthorst, H.J.M. A Novel WRKY transcription factor is required for induction of PR-1a gene expression by salicylic acid and bacterial elicitors. Plant Physiol. 2008, 146, 1983–1995. [Google Scholar] [CrossRef]
- Tao, X.; Chengjie, C.; Chuhao, L.; Jiarou, L.; Chaoyang, L.; Yehua, H. Genome-wide investigation of WRKY gene family in pineapple: Evolution and expression profiles during development and stress. BMC Genom. 2018, 19, 490. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Li, X.; Li, F.; Li, D.; Dong, Y.; Fan, Y. Bioinformatics Analysis of WRKY Family Genes in Erianthus fulvus Ness. Genes 2022, 13, 2102. https://doi.org/10.3390/genes13112102
Chen H, Li X, Li F, Li D, Dong Y, Fan Y. Bioinformatics Analysis of WRKY Family Genes in Erianthus fulvus Ness. Genes. 2022; 13(11):2102. https://doi.org/10.3390/genes13112102
Chicago/Turabian StyleChen, Haowen, Xuzhen Li, Fusheng Li, Dengyu Li, Yang Dong, and Yuanhong Fan. 2022. "Bioinformatics Analysis of WRKY Family Genes in Erianthus fulvus Ness" Genes 13, no. 11: 2102. https://doi.org/10.3390/genes13112102
APA StyleChen, H., Li, X., Li, F., Li, D., Dong, Y., & Fan, Y. (2022). Bioinformatics Analysis of WRKY Family Genes in Erianthus fulvus Ness. Genes, 13(11), 2102. https://doi.org/10.3390/genes13112102






