Expression of Piwi, MMP, TIMP, and Sox during Gut Regeneration in Holothurian Eupentacta fraudatrix (Holothuroidea, Dendrochirotida)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Collection, Maintenance, and Evisceration
2.2. Real-Time PCR
2.3. RNA Probe Synthesis
2.4. Whole Mount In Situ Hybridization
2.5. Sequence Searching and Phylogenetic Analysis
2.6. Immunocytochemistry
3. Results
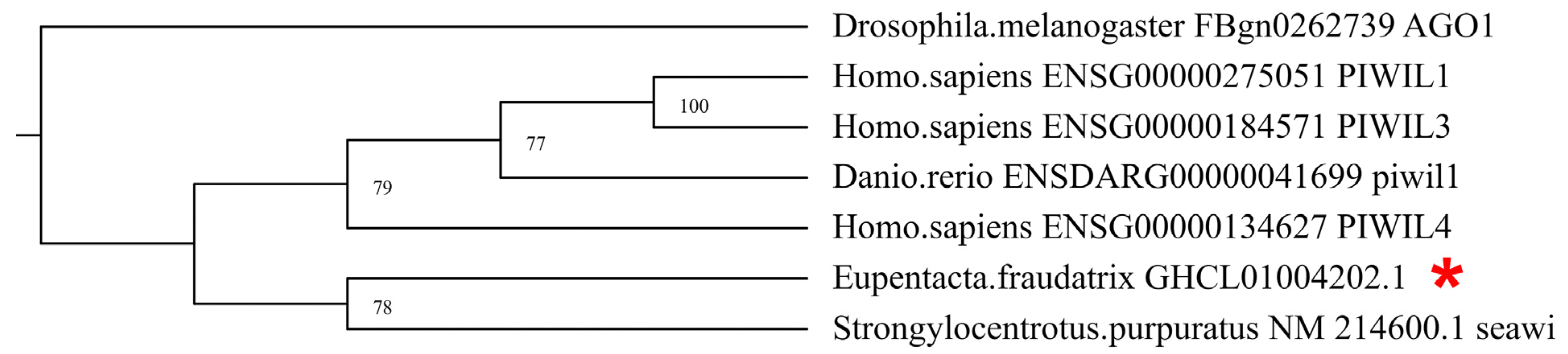
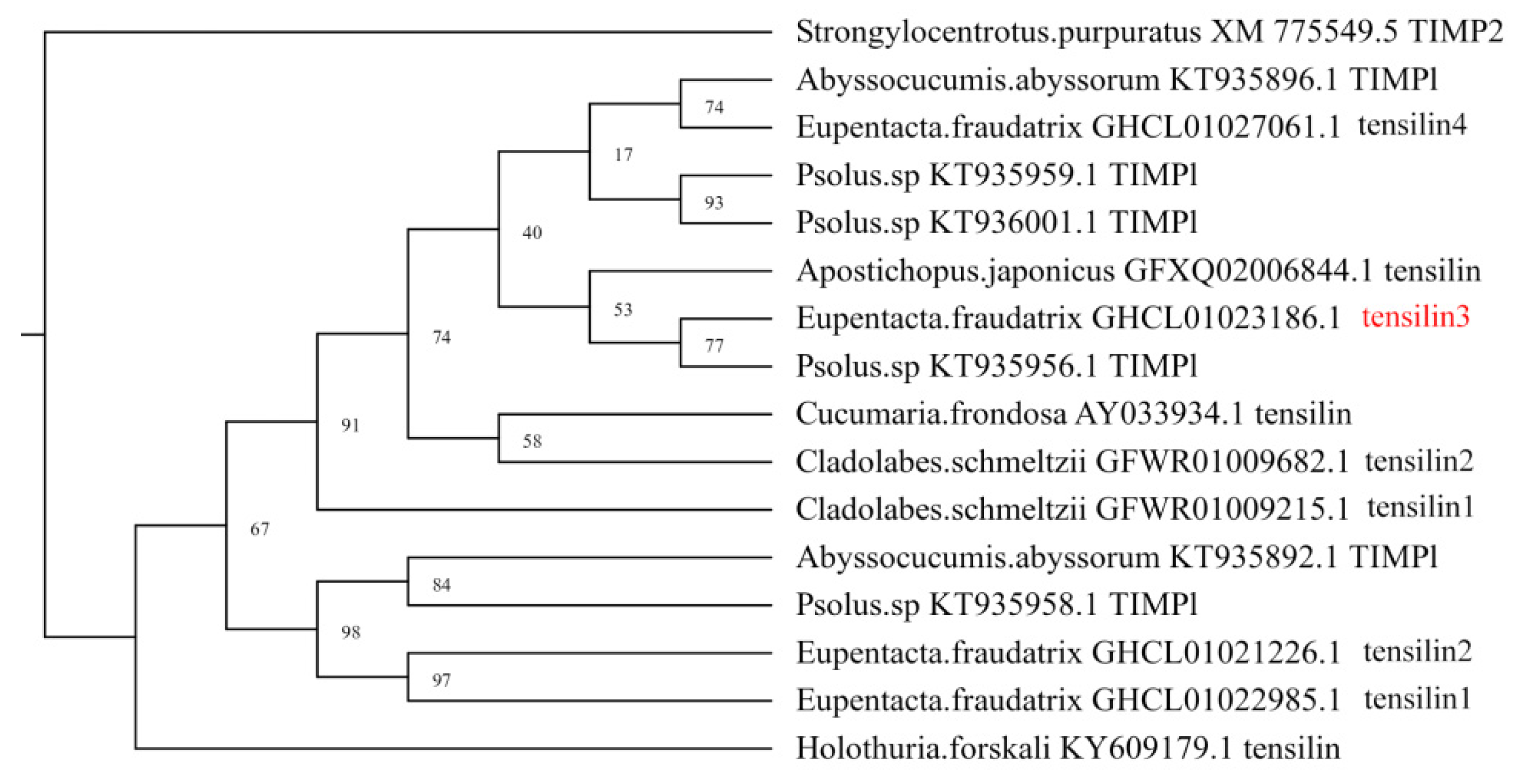
3.1. Identification and Characterization of DEGs of Piwi, MMP, TIMP, and Sox Orthologs
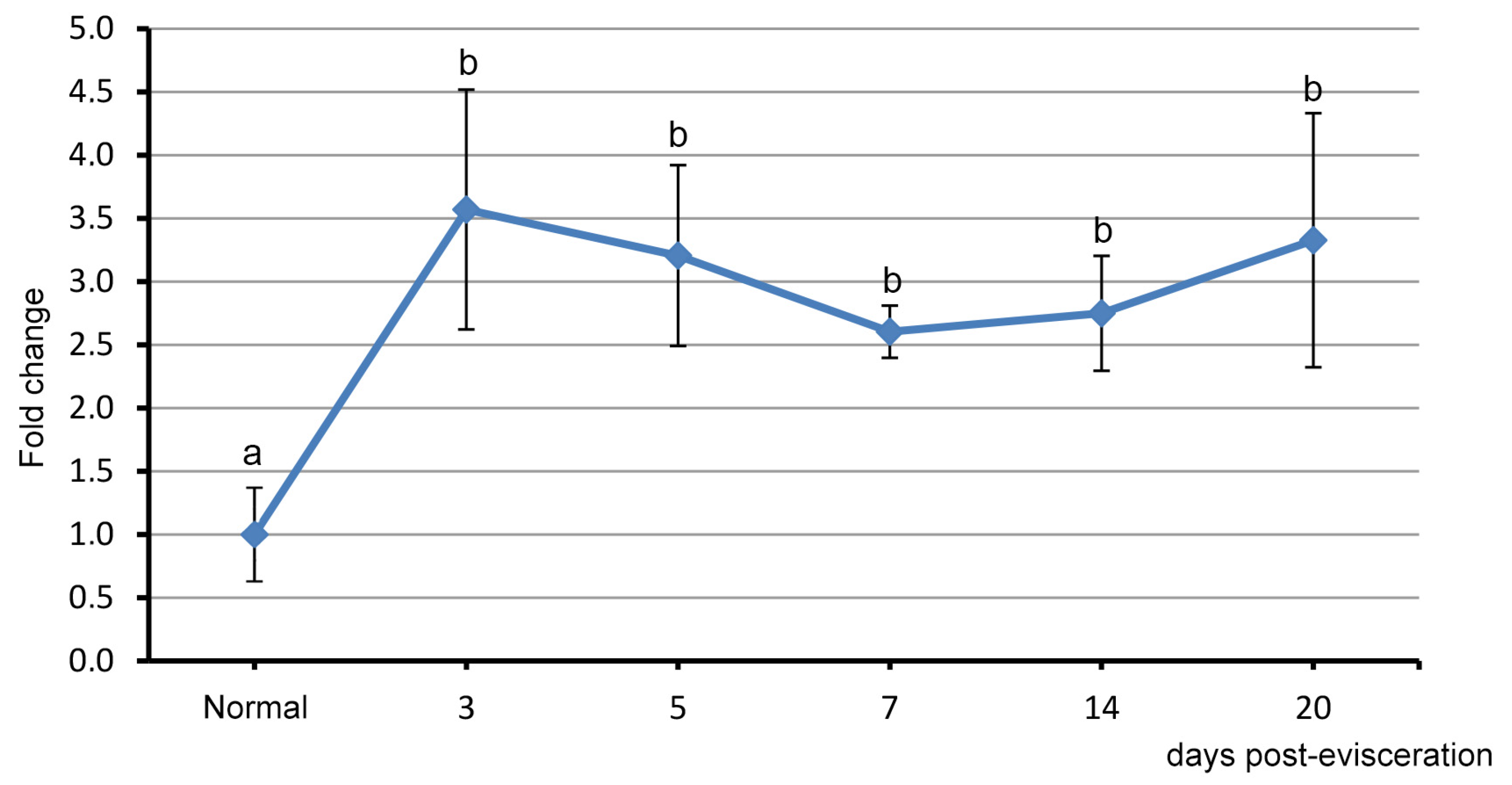
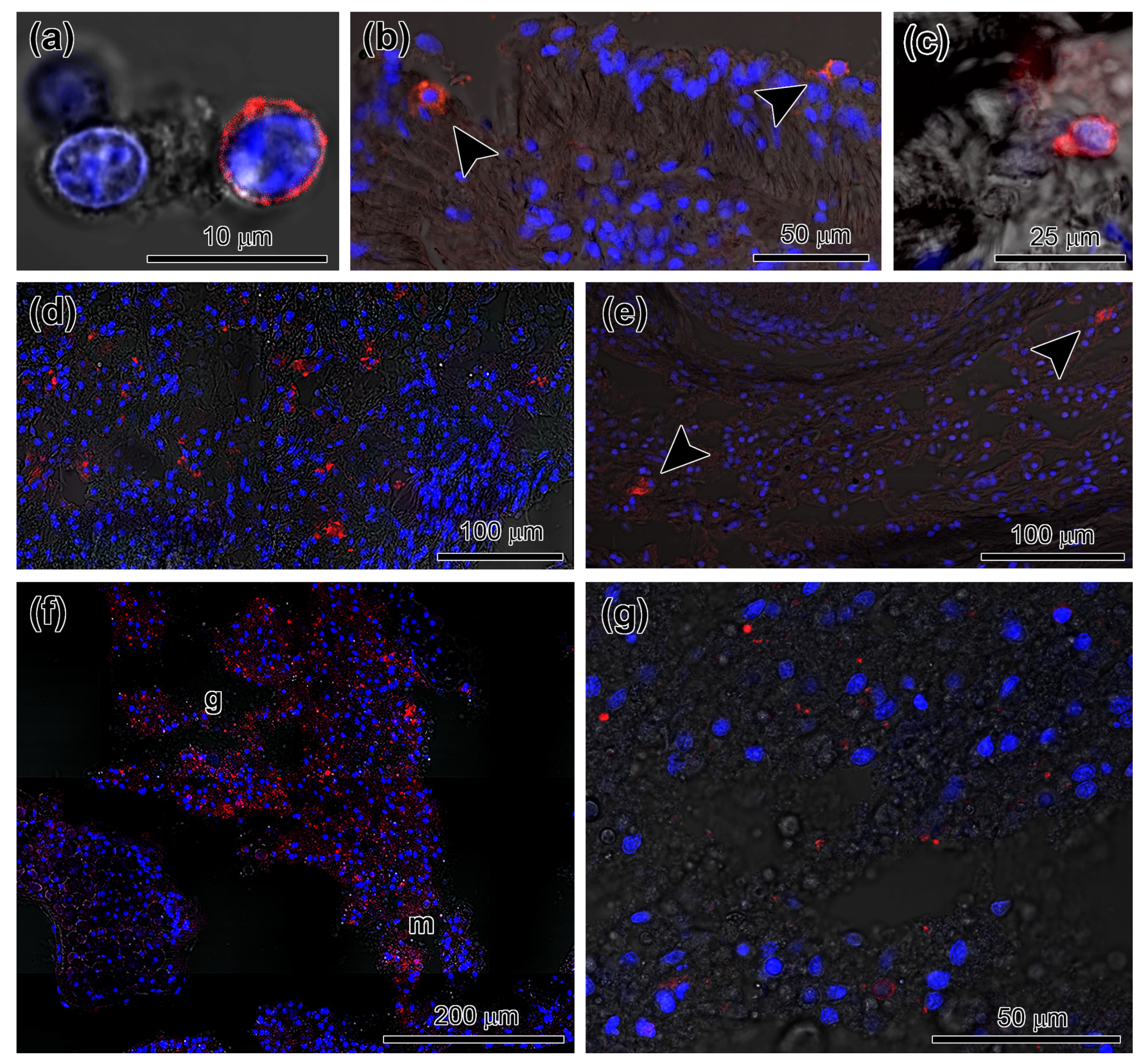
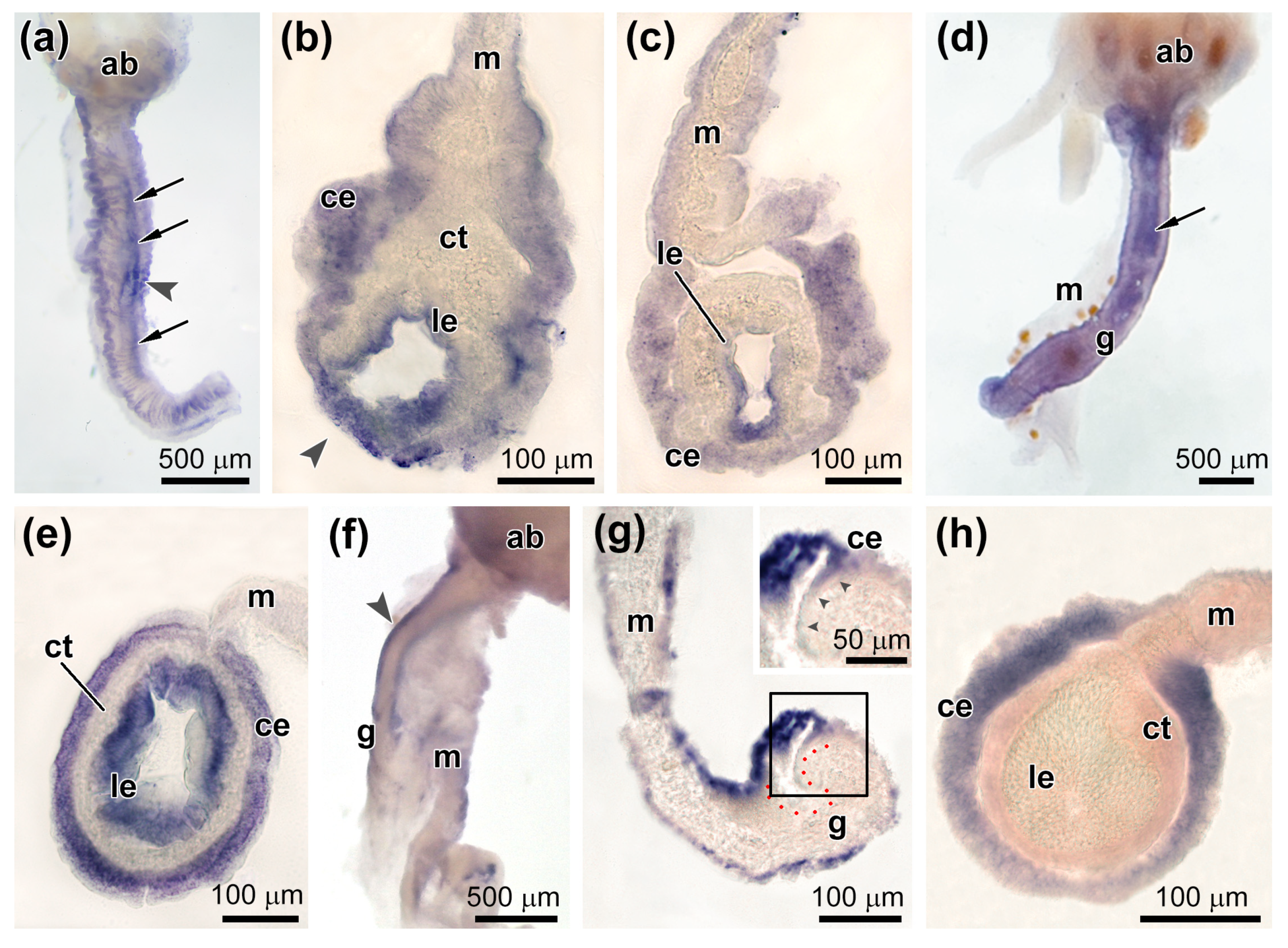
3.2. Spatial Distribution of Piwi-Positive Cells
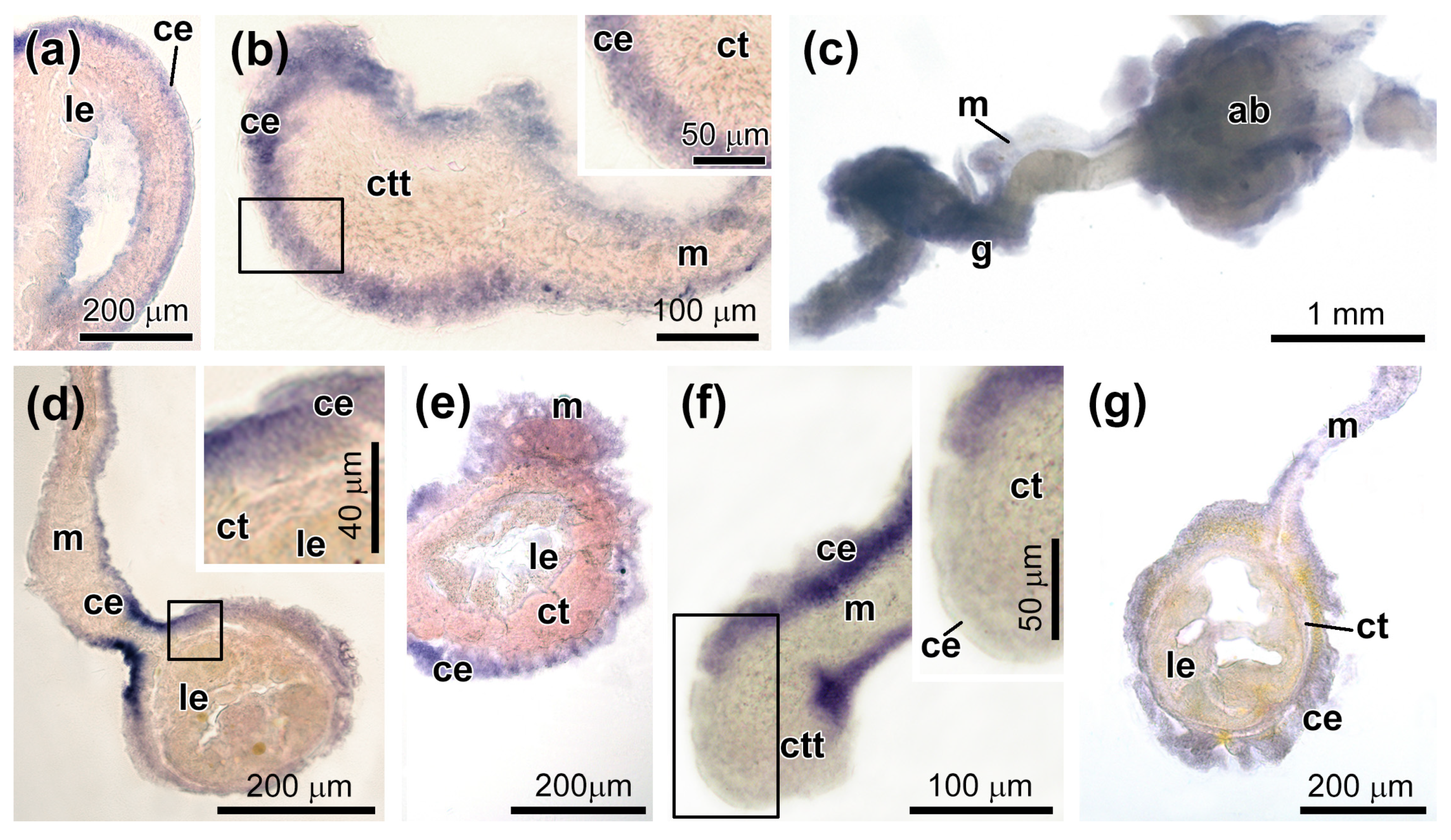
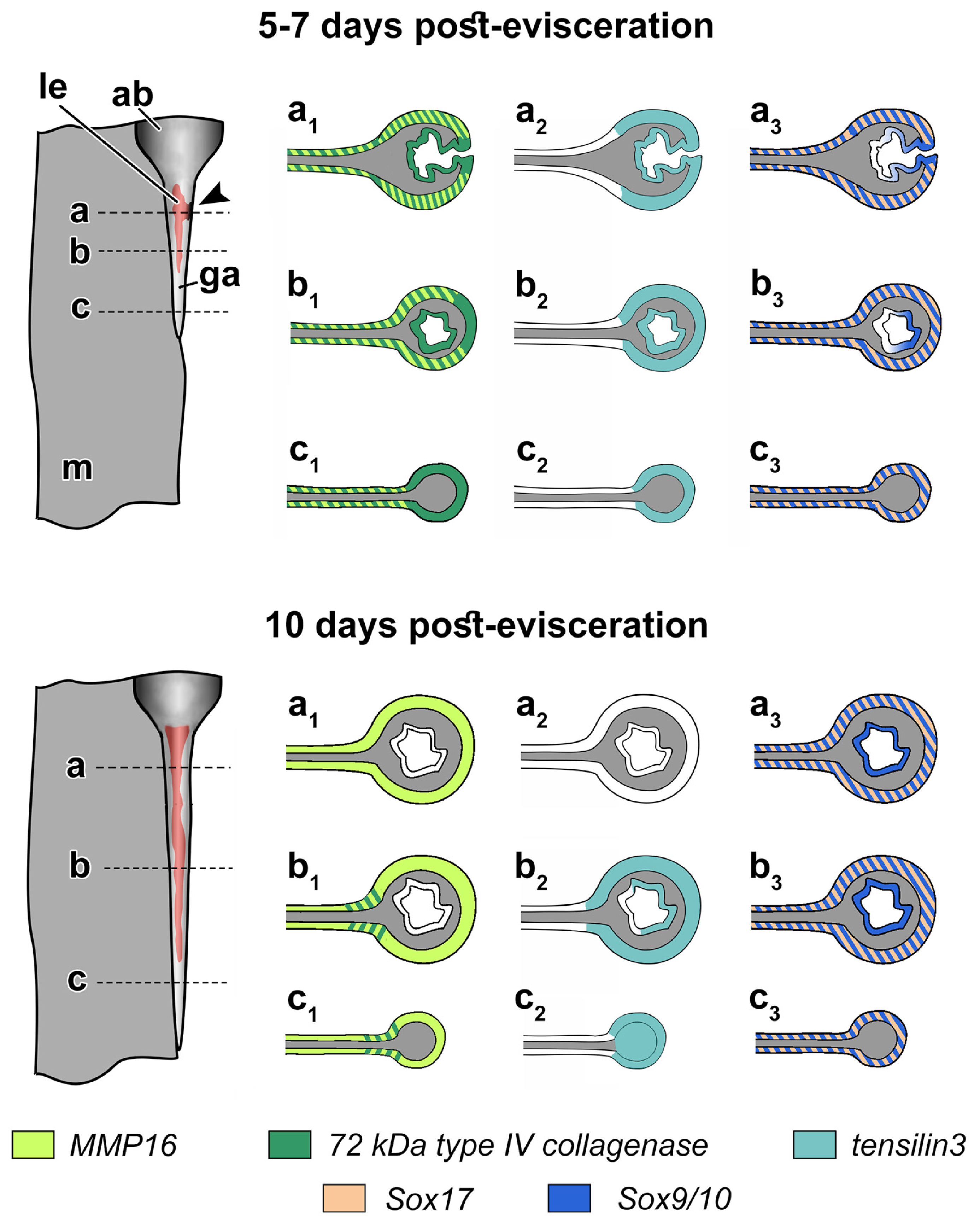
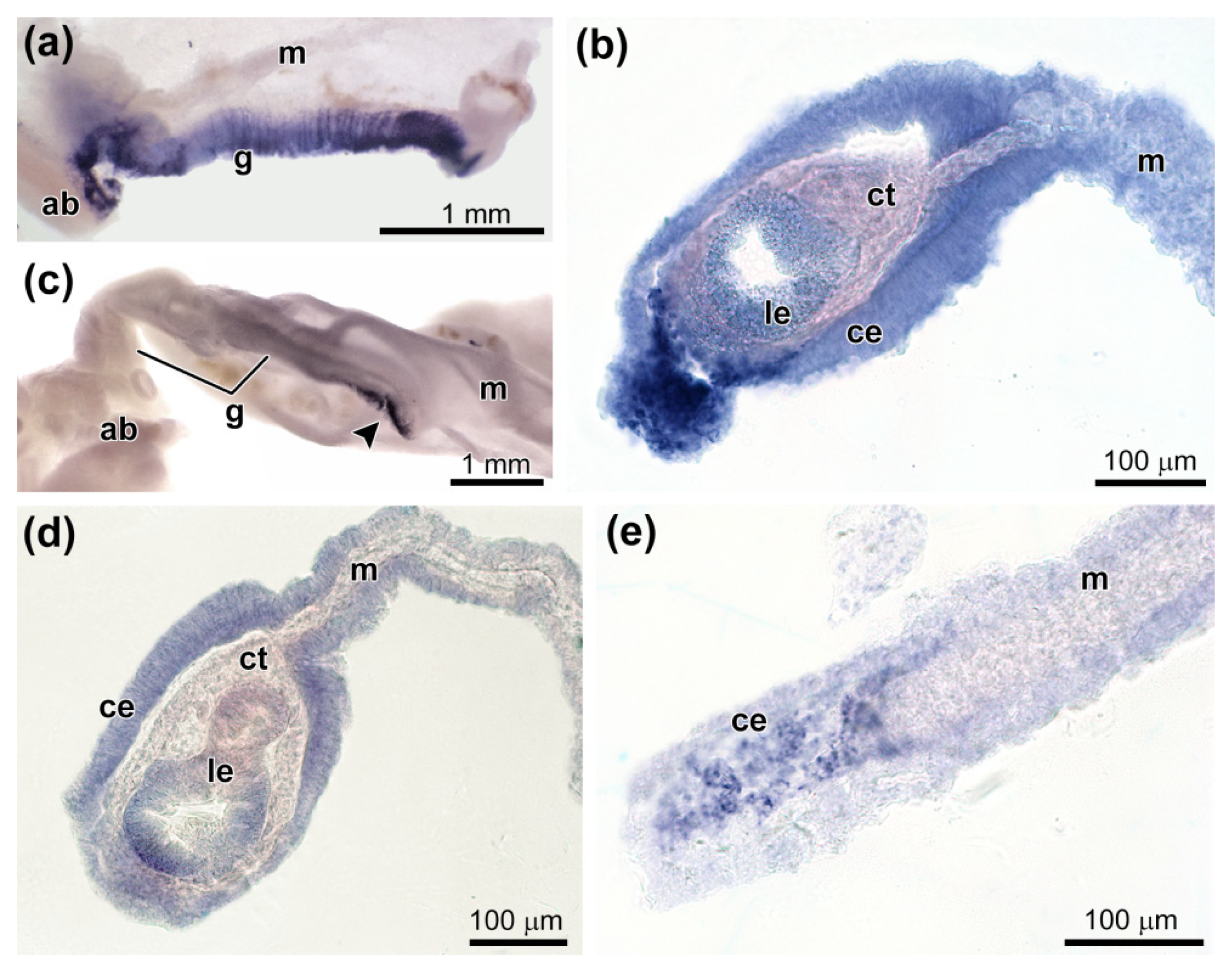
3.3. Spatial Distribution of MMPs Transcripts
3.4. Spatial Distribution of Ef-Tensilin3 Transcripts
3.5. Spatial Distribution of Ef-Sox9/10 and Ef-Sox17 Transcripts
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Candia Carnevali, M.D. Regeneration in echinoderms: Repair, regrowth, cloning. Invertebr. Surv. J. 2006, 3, 64–76. [Google Scholar]
- Dolmatov, I.Y. Regeneration in echinoderms. Russ. J. Mar. Biol. 1999, 25, 225–233. [Google Scholar]
- Dolmatov, I.Y.; Eliseikina, M.G.; Bulgakov, A.A.; Ginanova, T.T.; Lamash, N.E.; Korchagin, V.P. Muscle regeneration in the holothurian Stichopus japonicus. Roux Arch. Dev. Biol. 1996, 205, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Dolmatov, I.Y.; Ginanova, T.T. Muscle regeneration in holothurians. Microsc. Res. Tech. 2001, 55, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Dolmatov, I.Y.; Ginanova, T.T. Post-autotomy regeneration of the respiratory trees in the holothurian Apostichopus japonicus (Holothurioidea, Aspidochirotida). Cell Tissue Res. 2009, 336, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Frolova, L.T.; Dolmatov, I.Y. Microscopic anatomy of the digestive system in normal and regenerating specimens of the brittlestar Amphipholis kochii. Biol. Bull. 2010, 218, 303–316. [Google Scholar] [CrossRef]
- García-Arrarás, J.E.; Dolmatov, I.Y. Echinoderms: Potential model systems for studies on muscle regeneration. Curr. Pharm. Des. 2010, 16, 942–955. [Google Scholar] [CrossRef] [Green Version]
- Mashanov, V.S.; García-Arrarás, J.E. Gut regeneration in holothurians: A snapshot of recent developments. Biol. Bull. 2011, 221, 93–109. [Google Scholar] [CrossRef]
- Mladenov, P.V.; Bisgrove, B.; Asotra, S.; Burke, R.D. Mechanisms of arm tip regeneration in the sea star, Leptasterias hexactis. Roux Arch. Dev. Biol. 1989, 198, 19–28. [Google Scholar] [CrossRef]
- Eliseikina, M.G.; Magarlamov, T.Y.; Dolmatov, I.Y. Stem cells of holothuroid coelomocytes. In Echinoderms: Durham; Harris, L.G., Bottger, S.A., Walker, C.W., Lesser, M.P., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 163–166. [Google Scholar]
- Zavalnaya, E.G.; Shamshurina, E.V.; Eliseikina, M.G. The Immunocytochemical Identification of PIWI-positive cells during the recovery of a coelomocyte population after evisceration in the holothurian Eupentacta fraudatrix (Djakonov et Baranova, 1958) (Holothuroidea: Dendrochirota). Russ. J. Mar. Biol. 2020, 46, 97–104. [Google Scholar] [CrossRef]
- Dolmatov, I.Y.; Mashanov, V.S. Regeneration in Holothurians; Dalnauka: Vladivostok, Russia, 2007; p. 212. [Google Scholar]
- Vogt, G. Hidden treasures in stem cells of indeterminately growing bilaterian invertebrates. Stem Cell Rev. Rep. 2012, 8, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Mashanov, V.; Zueva, O.; Mashanova, D.; Garcia-Arraras, J.E. Expression of stem cell factors in the adult sea cucumber digestive tube. Cell Tissue Res 2017, 370, 427–440. [Google Scholar] [CrossRef]
- Mashanov, V.S.; Zueva, O.R.; Garcia-Arraras, J.E. Expression of pluripotency factors in echinoderm regeneration. Cell Tissue Res. 2015, 359, 521–536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mashanov, V.S.; Zueva, O.R.; García-Arrarás, J.E. Myc regulates programmed cell death and radial glia dedifferentiation after neural injury in an echinoderm. BMC Dev. Biol. 2015, 15, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aravin, A.A.; Sachidanandam, R.; Girard, A.; Fejes-Toth, K.; Hannon, G.J. Developmentally regulated piRNA clusters implicate MILI in transposon control. Science 2007, 316, 744–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juliano, C.; Wang, J.; Lin, H. Uniting germline and stem cells: The function of Piwi proteins and the piRNA pathway in diverse organisms. Ann. Rev. Genet 2011, 45, 447–469. [Google Scholar] [CrossRef] [Green Version]
- Vagin, V.V. A distinct small RNA pathway silences selfish genetic elements in the germline. Science 2006, 313, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, E.A.; Wessel, G.M. Vasa genes: Emerging roles in the germ line and in multipotent cells. BioEssays 2010, 32, 626–637. [Google Scholar] [CrossRef] [Green Version]
- Praher, D.; Zimmermann, B.; Genikhovich, G.; Columbus-Shenkar, Y.; Modepalli, V.; Aharoni, R.; Moran, Y.; Technau, U. Characterization of the piRNA pathway during development of the sea anemone Nematostella vectensis. RNA Biol. 2017, 14, 1727–1741. [Google Scholar] [CrossRef] [Green Version]
- Juliano, C.E.; Reich, A.; Liu, N.; Götzfried, J.; Zhong, M.; Uman, S.; Reenan, R.A.; Wessel, G.M.; Steele, R.E.; Lin, H. PIWI proteins and PIWI-interacting RNAs function in Hydra somatic stem cells. Proc. Natl. Acad. Sci. USA 2014, 111, 337–342. [Google Scholar] [CrossRef] [Green Version]
- Solana, J.; Kao, D.; Mihaylova, Y.; Jaber-Hijazi, F.; Malla, S.; Wilson, R.; Aboobaker, A. Defining the molecular profile of planarian pluripotent stem cells using a combinatorial RNAseq, RNA interference and irradiation approach. Genome Biol. 2012, 13, R19. [Google Scholar] [CrossRef] [Green Version]
- Cary, G.A.; Wolff, A.; Zueva, O.; Pattinato, J.; Hinman, V.F. Analysis of sea star larval regeneration reveals conserved processes of whole-body regeneration across the metazoa. BMC Biol. 2019, 17, 16. [Google Scholar] [CrossRef] [Green Version]
- Oulhen, N.; Heyland, A.; Carrier, T.J.; Zazueta-Novoa, V.; Fresques, T.; Laird, J.; Onorato, T.M.; Janies, D.; Wessel, G. Regeneration in bipinnaria larvae of the bat star Patiria miniata induces rapid and broad new gene expression. Mech. Dev. 2016, 142, 10–21. [Google Scholar] [CrossRef]
- Reinardy, H.C.; Emerson, C.E.; Manley, J.M.; Bodnar, A.G. Tissue regeneration and biomineralization in sea urchins: Role of Notch signaling and presence of stem cell markers. PLoS ONE 2015, 10, e0133860. [Google Scholar] [CrossRef] [Green Version]
- Dolmatov, I.Y. Variability of regeneration mechanisms in echinoderms. Russ. J. Mar. Biol. 2020, 46, 391–404. [Google Scholar] [CrossRef]
- Dolmatov, I.Y. Molecular aspects of regeneration mechanisms in holothurians. Genes 2021, 12, 250. [Google Scholar] [CrossRef] [PubMed]
- García-Arrarás, J.E.; Lázaro-Peña, M.I.; Díaz-Balzac, C.A. Holothurians as a model system to study regeneration. Results Probl. Cell Differ. 2018, 65, 255–283. [Google Scholar]
- Quispe-Parra, D.; Valentín, G.; García-Arrarás, J.E. A roadmap for intestinal regeneration. Int. J. Dev. Biol. 2021, 65, 427–437. [Google Scholar] [CrossRef]
- Boyko, A.V.; Girich, A.S.; Tkacheva, E.S.; Dolmatov, I.Y. The Eupentacta fraudatrix transcriptome provides insights into regulation of cell transdifferentiation. Sci. Rep. 2020, 10, 1522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, K.; Zhang, L.B.; Sun, L.N.; Lin, C.G.; Feng, Q.M.; Zhang, S.Y.; Yang, H.S.; Brinkman, R.; Lin, G.; Huang, Z. Transcriptome analysis provides insights into the molecular mechanisms responsible for evisceration behavior in the sea cucumber Apostichopus japonicus. Comp. Biochem. Physiol. Part D Genom. Proteom. 2019, 30, 143–157. [Google Scholar] [CrossRef]
- Zhang, L.; Feng, Q.; Sun, L.; Ding, K.; Huo, D.; Fang, Y.; Zhang, T.; Yang, H. Differential gene expression in the intestine of sea cucumber (Apostichopus japonicus) under low and high salinity conditions. Comp. Biochem. Physiol. Part D Genom. Proteom. 2017, 25, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Lamash, N.E.; Dolmatov, I.Y. Proteases from the regenerating gut of the holothurian Eupentacta fraudatrix. PLoS ONE 2013, 8, e58433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quiñones, J.L.; Rosa, R.; Ruiz, D.L.; García-Arrarás, J.E. Extracellular matrix remodeling and metalloproteinase involvement during intestine regeneration in the sea cucumber Holothuria glaberrima. Dev. Biol. 2002, 250, 181–197. [Google Scholar] [CrossRef] [Green Version]
- Miao, T.; Wan, Z.; Sun, L.; Li, X.; Xing, L.; Bai, Y.; Wang, F.; Yang, H. Extracellular matrix remodeling and matrix metalloproteinases (ajMMP-2 like and ajMMP-16 like) characterization during intestine regeneration of sea cucumber Apostichopus japonicus. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2017, 212, 12–23. [Google Scholar] [CrossRef]
- Clouse, R.M.; Linchangco, G.V.; Kerr, A.M.; Reid, R.W.; Janies, D.A. Phylotranscriptomic analysis uncovers a wealth of tissue inhibitor of metalloproteinases variants in echinoderms. R. Soc. Open Sci. 2015, 2, 150377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dolmatov, I.Y.; Afanasyev, S.V.; Boyko, A.V. Molecular mechanisms of fission in echinoderms: Transcriptome analysis. PLoS ONE 2018, 13, e0195836. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, A.R.; Barbaglio, A.; Oliveira, M.J.; Ribeiro, C.C.; Wilkie, I.C.; Candia Carnevali, M.D.; Barbosa, M.A. Matrix metalloproteinases in a sea urchin ligament with adaptable mechanical properties. PLoS ONE 2012, 7, e49016. [Google Scholar] [CrossRef] [Green Version]
- Alexander, J.; Stainier, D.Y.R. A molecular pathway leading to endoderm formation in zebrafish. Curr. Biol. 1999, 9, 1147–1157. [Google Scholar] [CrossRef] [Green Version]
- Shivdasani, R.A. Molecular regulation of vertebrate early endoderm development. Dev. Biol. 2002, 249, 191–203. [Google Scholar] [CrossRef] [Green Version]
- Bankaitis, E.D.; Ha, A.; Kuo, C.J.; Magness, S.T. Reserve stem cells in intestinal homeostasis and injury. Gastroenterology 2018, 155, 1348–1361. [Google Scholar] [CrossRef] [Green Version]
- Gracz, A.D.; Magness, S.T. Defining hierarchies of stemness in the intestine: Evidence from biomarkers and regulatory pathways. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, G260–G273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, H.; Park, K.H. Regulation and function of SOX9 during cartilage development and regeneration. Semin. Cancer Biol. 2020, 67, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Ma, Y.; Dai, X.; Ren, T.; Fu, Y.; Liu, W.; Han, Y.; Wu, Y.; Cheng, Y.; Zhang, T.; et al. Regeneration of functional alveoli by adult human SOX9+ airway basal cell transplantation. Protein Cell 2018, 9, 267–282. [Google Scholar] [CrossRef] [Green Version]
- Hojo, H.; Chung, U.I.; Ohba, S. Identification of the gene-regulatory landscape in skeletal development and potential links to skeletal regeneration. Regen. Ther. 2017, 6, 100–107. [Google Scholar] [CrossRef]
- Mashanov, V.S.; Dolmatov, I.Y.; Heinzeller, T. Transdifferentiation in holothurian gut regeneration. Biol. Bull. 2005, 209, 184–193. [Google Scholar] [CrossRef]
- Dolmatov, I.Y. Regeneration of the aquapharyngeal complex in the holothurian Eupentacta fraudatrix (Holothuroidea, Dendrochirota). Monogr. Dev. Biol. 1992, 23, 40–50. [Google Scholar] [PubMed]
- Leibson, N.L. Regeneration of digestive tube in holothurians Stichopus japonicus and Eupentacta fraudatrix. Monogr. Dev. Biol. 1992, 23, 51–61. [Google Scholar] [PubMed]
- Dolmatov, I.Y.; Nizhnichenko, V.A.; Dolmatova, L.S. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in echinoderms: Structure and possible functions. Cells 2021, in press. [Google Scholar]
- Lai, A.G.; Aboobaker, A.A. EvoRegen in animals: Time to uncover deep conservation or convergence of adult stem cell evolution and regenerative processes. Dev. Biol. 2018, 433, 118–131. [Google Scholar] [CrossRef]
- Chia, F.-S.; Xing, J. Echinoderm coelomocytes. Zool. Stud. 1996, 35, 231–254. [Google Scholar]
- Rinkevich, Y.; Matranga, V.; Rinkevich, B. Stem cells in aquatic invertebrates: Common premises and emerging unique themes. In Stem Cells in Marine Organisms; Rinkevich, B., Matranga, V., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2009; pp. 61–103. [Google Scholar]
- García-Arrarás, J.E.; Estrada-Rodgers, L.; Santiago, R.; Torres, I.I.; Díaz-Miranda, L.; Torres-Avillán, I. Cellular mechanisms in the regeneration of the intestine of the sea cucumber, Holothuria glaberrima Selenka (Holothuroidea: Echinodermata). J. Exp. Zool. 1998, 281, 288–304. [Google Scholar] [CrossRef]
- Page-McCaw, A.; Ewald, A.J.; Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat. Rev. Mol. Cell Bio. 2007, 8, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Nagase, H. Progress in matrix metalloproteinase research. Mol. Aspects Med. 2008, 29, 290–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dolmatov, I.Y.; Shulga, A.P.; Ginanova, T.T.; Eliseikina, M.G.; Lamash, N.E. Metalloproteinase inhibitor GM6001 delays regeneration in holothurians. Tissue Cell 2019, 59, 1–9. [Google Scholar] [CrossRef]
- Shulga, A.P.; Lamash, N.E. Proteinases with gelatinase activity and their role in ambulacrum regeneration in holothurians Eupentacta fraudatrix (D’yakonov and Baranova, 1958) and Cucumaria japonica (Semper, 1868) (Echinodermata: Holothuroidea). Russ. J. Mar. Biol. 2020, 46, 461–471. [Google Scholar] [CrossRef]
- Angerer, L.; Hussain, S.; Wei, Z.; Livingston, B.T. Sea urchin metalloproteases: A genomic survey of the BMP-1/tolloid-like, MMP and ADAM families. Dev. Biol. 2006, 300, 267–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Arrarás, J.E.; Bello, S.A.; Malavez, S. The mesentery as the epicenter for intestinal regeneration. Semin. Cell Dev. Biol. 2019, 92, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G. Tissue inhibitors of metalloproteinases. Genome Biol. 2011, 12, 233. [Google Scholar] [CrossRef]
- Woessner, J.F., Jr. Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J. 1991, 5, 2145–2154. [Google Scholar] [CrossRef] [Green Version]
- Vázquez-Vélez, G.E.; Rodríguez-Molina, J.F.; Quiñones-Frías, M.C.; Pagán, M. A proteoglycan-like molecule offers insights into ground substance changes during holothurian intestinal regeneration. J. Histochem. Cytochem. 2016, 64, 381–393. [Google Scholar] [CrossRef] [Green Version]
- Verstappen, J.; Von den Hoff, J.W. Tissue inhibitors of metalloproteinases (TIMPs): Their biological functions and involvement in oral disease. J. Dent. Res. 2006, 85, 1074–1084. [Google Scholar] [CrossRef] [PubMed]
- Lambert, E.; Dasse, E.; Haye, B.; Petitfrere, E. TIMPs as multifacial proteins. Crit. Rev. Oncol. Hematol. 2004, 49, 187–198. [Google Scholar] [CrossRef]
- Hayakawa, T.; Yamashita, K.; Ohuchi, E.; Shinagawa, A. Cell growth-promoting activity of tissue inhibitor of metalloproteinases-2 (TIMP-2). J. Cell Sci. 1994, 107, 2373–2379. [Google Scholar] [CrossRef]
- Jiang, Y.; Goldberg, I.D.; Shi, Y.E. Complex roles of tissue inhibitors of metalloproteinases in cancer. Oncogene 2002, 21, 2245–2252. [Google Scholar] [CrossRef] [Green Version]
- Ricca, T.I.; Liang, G.; Suenaga, A.P.M.; Han, S.W.; Jones, P.A.; Jasiulionis, M.G. Tissue inhibitor of metalloproteinase 1 expression associated with gene demethylation confers anoikis resistance in early phases of melanocyte malignant transformation. Transl. Oncol. 2009, 2, 329–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, K.-K.; Liu, X.-W.; Chirco, R.; Fridman, R.; Kim, H.-R.C. Identification of CD63 as a tissue inhibitor of metalloproteinase-1 interacting cell surface protein. EMBO J. 2006, 25, 3934–3942. [Google Scholar] [CrossRef] [Green Version]
- Tang, J.; Kang, Y.; Huang, L.; Wu, L.; Peng, Y. TIMP1 preserves the blood–brain barrier through interacting with CD63/integrin β1 complex and regulating downstream FAK/RhoA signaling. Acta Pharm. Sin. B. 2020, 10, 987–1003. [Google Scholar] [CrossRef] [PubMed]
- Mashanov, V.S.; Zueva, O.R.; Rojas-Catagena, C.; Garcia-Arraras, J.E. Visceral regeneration in a sea cucumber involves extensive expression of survivin and mortalin homologs in the mesothelium. BMC Dev. Biol. 2010, 10, 117. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Yang, H.; Chen, M.; Ma, D.; Lin, C. RNA-Seq reveals dynamic changes of gene expression in key stages of intestine regeneration in the sea cucumber Apostichopus japonicus. [corrected]. PLoS ONE 2013, 8, e69441. [Google Scholar]
- Zorn, A.M.; Wells, J.M. Vertebrate endoderm development and organ formation. Annu. Rev. Cell Dev. Biol. 2009, 25, 221–251. [Google Scholar] [CrossRef] [Green Version]
- Furuyama, K.; Kawaguchi, Y.; Akiyama, H.; Horiguchi, M.; Kodama, S.; Kuhara, T.; Hosokawa, S.; Elbahrawy, A.; Soeda, T.; Koizumi, M.; et al. Continuous cell supply from a Sox9-expressing progenitor zone in adult liver, exocrine pancreas and intestine. Nat. Genet. 2011, 43, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Mead, T.J.; Wang, Q.; Bhattaram, P.; Dy, P.; Afelik, S.; Jensen, J.; Lefebvre, V. A far-upstream (-70 kb) enhancer mediates Sox9 auto-regulation in somatic tissues during development and adult regeneration. Nucleic Acids Res. 2013, 41, 4459–4469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Asakura, M.; Inoue, H.; Nakamura, T.; Sano, M.; Niu, Z.; Chen, M.; Schwartz, R.J.; Schneider, M.D. Sox17 is essential for the specification of cardiac mesoderm in embryonic stem cells. Proc. Natl. Acad. Sci. USA 2007, 104, 3859–3864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakraborty, S.; Combs, M.D.; Yutzey, K.E. Transcriptional regulation of heart valve progenitor cells. Pediatr. Cardiol. 2010, 31, 414–421. [Google Scholar] [CrossRef] [PubMed] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dolmatov, I.Y.; Kalacheva, N.V.; Tkacheva, E.S.; Shulga, A.P.; Zavalnaya, E.G.; Shamshurina, E.V.; Girich, A.S.; Boyko, A.V.; Eliseikina, M.G. Expression of Piwi, MMP, TIMP, and Sox during Gut Regeneration in Holothurian Eupentacta fraudatrix (Holothuroidea, Dendrochirotida). Genes 2021, 12, 1292. https://doi.org/10.3390/genes12081292
Dolmatov IY, Kalacheva NV, Tkacheva ES, Shulga AP, Zavalnaya EG, Shamshurina EV, Girich AS, Boyko AV, Eliseikina MG. Expression of Piwi, MMP, TIMP, and Sox during Gut Regeneration in Holothurian Eupentacta fraudatrix (Holothuroidea, Dendrochirotida). Genes. 2021; 12(8):1292. https://doi.org/10.3390/genes12081292
Chicago/Turabian StyleDolmatov, Igor Yu., Nadezhda V. Kalacheva, Ekaterina S. Tkacheva, Alena P. Shulga, Eugenia G. Zavalnaya, Ekaterina V. Shamshurina, Alexander S. Girich, Alexey V. Boyko, and Marina G. Eliseikina. 2021. "Expression of Piwi, MMP, TIMP, and Sox during Gut Regeneration in Holothurian Eupentacta fraudatrix (Holothuroidea, Dendrochirotida)" Genes 12, no. 8: 1292. https://doi.org/10.3390/genes12081292
APA StyleDolmatov, I. Y., Kalacheva, N. V., Tkacheva, E. S., Shulga, A. P., Zavalnaya, E. G., Shamshurina, E. V., Girich, A. S., Boyko, A. V., & Eliseikina, M. G. (2021). Expression of Piwi, MMP, TIMP, and Sox during Gut Regeneration in Holothurian Eupentacta fraudatrix (Holothuroidea, Dendrochirotida). Genes, 12(8), 1292. https://doi.org/10.3390/genes12081292





