Genetics of Diabetic Retinopathy, a Leading Cause of Irreversible Blindness in the Industrialized World
Abstract
1. Background
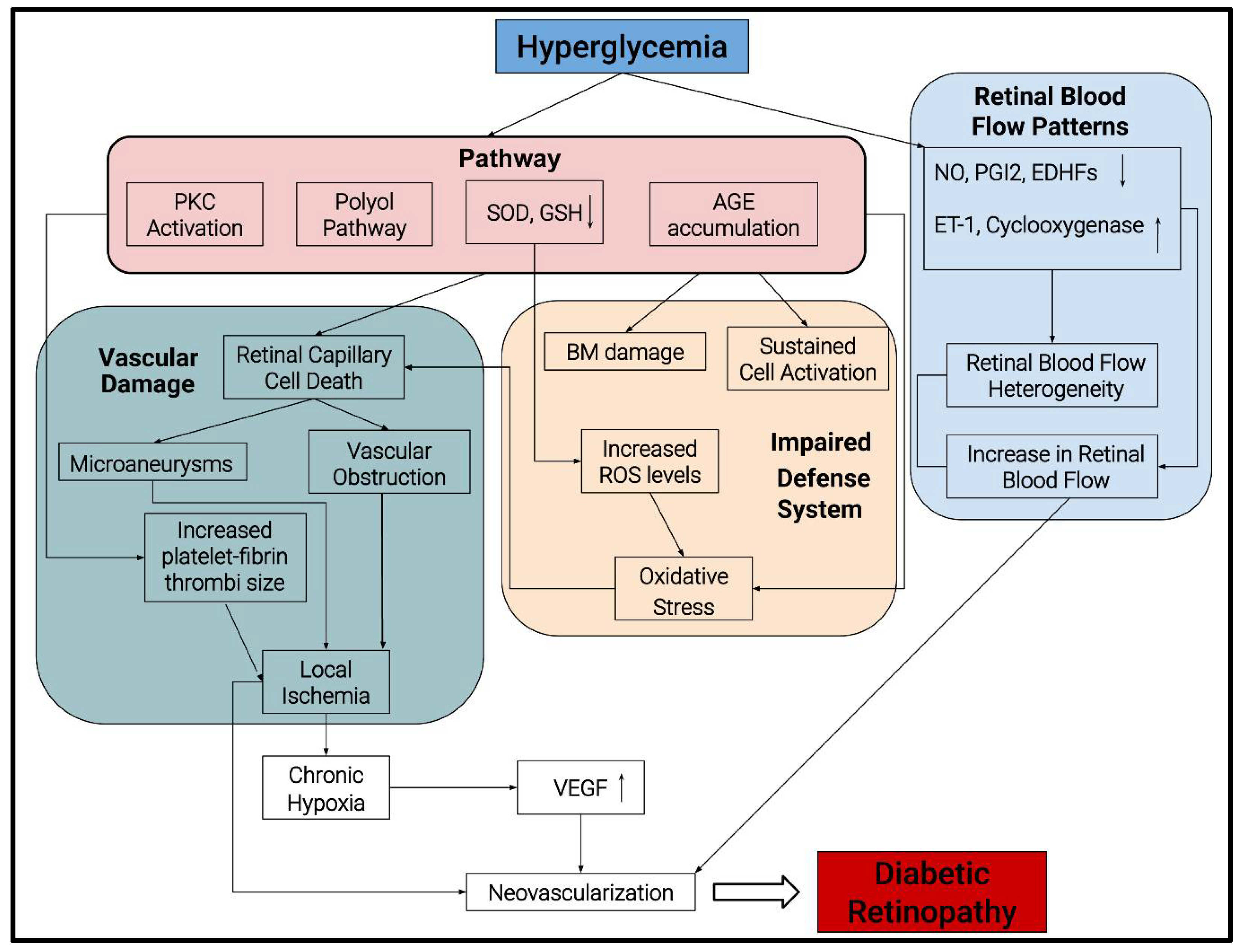
2. Pathogenesis of DR
3. The Genetic Basis of DR
3.1. Genetics in the Prevalence of DR
3.2. Familial Risk of DR
3.3. Progression of DR
4. Genetic Studies
4.1. Candidate Gene Studies
4.1.1. Aldose Reductase (ALR)
4.1.2. Receptor for Advanced Glycation End Products (RAGE)
4.1.3. VEGF Gene
4.1.4. eNOS Gene
4.1.5. Angiotensin-Converting Enzyme Gene
4.1.6. Erythropoietin Gene
4.2. Linkage Studies
4.3. Genome-Wide Association Studies
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ciulla, T.A.; Amador, A.G.; Zinman, B. Diabetic retinopathy and diabetic macular edema: Pathophysiology, screening, and novel therapies. Diabetes Care 2003, 26, 2653–2664. [Google Scholar] [CrossRef] [PubMed]
- Diabetic Retinopathy. In Textbook of Diabetes; Wiley Online Library: Hoboken, NJ, USA, 2020.
- Thomas, R.; Halim, S.; Gurudas, S.; Sivaprasad, S.; Owens, D. IDF Diabetes Atlas: A review of studies utilising retinal photography on the global prevalence of diabetes related retinopathy between 2015 and 2018. Diabetes Res. Clin. Pract. 2019, 157, 107840. [Google Scholar] [CrossRef] [PubMed]
- Cdcgov Watch Out for Diabetic Retinopathy|Features|CDC. Available online: https://www.cdc.gov/features/diabetic-retinopathy/index.html (accessed on 30 June 2021).
- Diabetic Retinopathy Data and Statistics|National Eye Institute. Available online: https://www.ncbi.nlm.nih.gov/pubmed/ (accessed on 30 June 2021).
- Hammes, H.-P.; Lin, J.; Renner, O.; Shani, M.; Lundqvist, A.; Betsholtz, C.; Brownlee, M.; Deutsch, U. Pericytes and the pathogenesis of diabetic retinopathy. Diabetes 2002, 51, 3107–3112. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Xin, X.; Jee, K.; Babapoor-Farrokhran, S.; Kashiwabuchi, F.; Ma, T.; Bhutto, I.; Hassan, S.J.; Daoud, Y.; Baranano, D. VEGF secreted by hypoxic Müller cells induces MMP-2 expression and activity in endothelial cells to promote retinal neovascularization in proliferative diabetic retinopathy. Diabetes 2013, 62, 3863–3873. [Google Scholar] [CrossRef]
- Roy, S.; Kern, T.S.; Song, B.; Stuebe, C. Mechanistic insights into pathological changes in the diabetic retina: Implications for targeting diabetic retinopathy. Am. J. Pathol. 2017, 187, 9–19. [Google Scholar] [CrossRef]
- Shoshani, Y.; Harris, A.; Shoja, M.M.; Arieli, Y.; Ehrlich, R.; Primus, S.; Ciulla, T.; Cantor, A.; Wirostko, B.; Siesky, B.A. Impaired ocular blood flow regulation in patients with open-angle glaucoma and diabetes. Clin. Exp. Ophthalmol. 2012, 40, 697–705. [Google Scholar] [CrossRef]
- Ciulla, T.A.; Harris, A.; Latkany, P.; Piper, H.C.; Arend, O.; Garzozi, H.; Martin, B. Ocular perfusion abnormalities in diabetes. Acta Ophthalmol. Scand. 2002, 80, 468–477. [Google Scholar] [CrossRef]
- Curtis, T.; Gardiner, T.; Stitt, A. Microvascular lesions of diabetic retinopathy: Clues towards understanding pathogenesis? Eye 2009, 23, 1496–1508. [Google Scholar] [CrossRef]
- Cai, J.; Boulton, M. The pathogenesis of diabetic retinopathy: Old concepts and new questions. Eye 2002, 16, 242–260. [Google Scholar] [CrossRef]
- Aiello, L.P.; Avery, R.L.; Arrigg, P.G.; Keyt, B.A.; Jampel, H.D.; Shah, S.T.; Pasquale, L.R.; Thieme, H.; Iwamoto, M.A.; Park, J.E. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N. Engl. J. Med. 1994, 331, 1480–1487. [Google Scholar] [CrossRef]
- Harris, M.I.; Klein, R.; Cowie, C.C.; Rowland, M.; Byrd-Holt, D.D. Is the risk of diabetic retinopathy greater in non-Hispanic blacks and Mexican Americans than in non-Hispanic whites with type 2 diabetes?: A US population study. Diabetes Care 1998, 21, 1230–1235. [Google Scholar] [CrossRef]
- Prevalence of Diagnosed Diabetes|Diabetes|CDC. Available online: https://www.cdc.gov/diabetes/data/statistics-report/diagnosed-diabetes.html (accessed on 30 June 2021).
- Bursell, S.-E.; Fonda, S.J.; Lewis, D.G.; Horton, M.B. Prevalence of diabetic retinopathy and diabetic macular edema in a primary care-based teleophthalmology program for American Indians and Alaskan Natives. PLoS ONE 2018, 13, e0198551. [Google Scholar]
- Nagi, D.; Pettitt, D.; Bennett, P.; Klein, R.; Knowler, W. Diabetic retinopathy assessed by fundus photography in Pima Indians with impaired glucose tolerance and NIDDM. Diabet. Med. 1997, 14, 449–456. [Google Scholar] [CrossRef]
- Lee, E.T.; Russell, D.; Morris, T.; Warn, A.; Kingsley, R.; Ogola, G. Visual impairment and eye abnormalities in Oklahoma Indians. Arch. Ophthalmol. 2005, 123, 1699–1704. [Google Scholar] [CrossRef][Green Version]
- Hirsch, I.B.; Brownlee, M. Beyond hemoglobin A1c—need for additional markers of risk for diabetic microvascular complications. JAMA 2010, 303, 2291–2292. [Google Scholar] [CrossRef]
- Control, D.; Group, C.T.R. Clustering of long-term complications in families with diabetes in the diabetes control and complications trial. Diabetes 1997, 46, 1829. [Google Scholar]
- Arar, N.H.; Freedman, B.I.; Adler, S.G.; Iyengar, S.K.; Chew, E.Y.; Davis, M.D.; Satko, S.G.; Bowden, D.W.; Duggirala, R.; Elston, R.C. Heritability of the severity of diabetic retinopathy: The FIND-Eye study. Investig. Ophthalmol. Vis. Sci. 2008, 49, 3839–3845. [Google Scholar] [CrossRef]
- Hietala, K.; Forsblom, C.; Summanen, P.; Groop, P.-H. Heritability of proliferative diabetic retinopathy. Diabetes 2008, 57, 2176–2180. [Google Scholar] [CrossRef]
- Leslie, R.; Pyke, D. Diabetic retinopathy in identical twins. Diabetes 1982, 31, 19–21. [Google Scholar] [CrossRef]
- Bhatwadekar, A.D.; Duan, Y.; Chakravarthy, H.; Korah, M.; Caballero, S.; Busik, J.V.; Grant, M.B. Ataxia Telangiectasia Mutated Dysregulation Results in Diabetic Retinopathy. Stem Cells 2016, 34, 405–417. [Google Scholar] [CrossRef][Green Version]
- Jarajapu, Y.P.; Bhatwadekar, A.D.; Caballero, S.; Hazra, S.; Shenoy, V.; Medina, R.; Kent, D.; Stitt, A.W.; Thut, C.; Finney, E.M.; et al. Activation of the ACE2/angiotensin-(1-7)/Mas receptor axis enhances the reparative function of dysfunctional diabetic endothelial progenitors. Diabetes 2013, 62, 1258–1269. [Google Scholar] [CrossRef] [PubMed]
- Abhary, S.; Hewitt, A.W.; Burdon, K.P.; Craig, J.E. A systematic meta-analysis of genetic association studies for diabetic retinopathy. Diabetes 2009, 58, 2137–2147. [Google Scholar] [CrossRef]
- Sharma, A.; Valle, M.L.; Beveridge, C.; Liu, Y.; Sharma, S. Unraveling the role of genetics in the pathogenesis of diabetic retinopathy. Eye 2019, 33, 534–541. [Google Scholar] [CrossRef]
- Cabrera, A.P.; Mankad, R.N.; Marek, L.; Das, R.; Rangasamy, S.; Monickaraj, F.; Das, A. Genotypes and Phenotypes: A Search for Influential Genes in Diabetic Retinopathy. Int. J. Mol. Sci. 2020, 21, 2712. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Tian, Z.; Zhang, L.; Liu, R.; Guan, Q.; Jiang, J. Genetic association of AKR1B1 gene polymorphism rs759853 with diabetic retinopathy risk: A meta-analysis. Gene 2018, 676, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Abhary, S.; Burdon, K.P.; Laurie, K.J.; Thorpe, S.; Landers, J.; Goold, L.; Lake, S.; Petrovsky, N.; Craig, J.E. Aldose reductase gene polymorphisms and diabetic retinopathy susceptibility. Diabetes Care 2010, 33, 1834–1836. [Google Scholar] [CrossRef]
- Lindholm, E.; Bakhtadze, E.; Sjögren, M.; Cilio, C.; Agardh, E.; Groop, L.; Agardh, C.-D. The−374 T/A polymorphism in the gene encoding RAGE is associated with diabetic nephropathy and retinopathy in type 1 diabetic patients. Diabetologia 2006, 49, 2745–2755. [Google Scholar] [CrossRef]
- Balasubbu, S.; Sundaresan, P.; Rajendran, A.; Ramasamy, K.; Govindarajan, G.; Perumalsamy, N.; Hejtmancik, J.F. Association analysis of nine candidate gene polymorphisms in Indian patients with Type 2 diabetic retinopathy. BMC Med. Genet. 2010, 11, 158. [Google Scholar] [CrossRef]
- Yang, Q.; Zhang, Y.; Zhang, X.; Li, X.; Liu, J. Association of VEGF Gene Polymorphisms with Susceptibility to Diabetic Retinopathy: A Systematic Review and Meta-Analysis. Horm. Metab. Res. 2020, 52, 264–279. [Google Scholar]
- Midani, F.; Ben Amor, Z.; El Afrit, M.A.; Kallel, A.; Feki, M.; Soualmia, H. The Role of Genetic Variants (rs869109213 and rs2070744) of the eNOS Gene and Bgl II in the α2 Subunit of the α2β1 Integrin Gene in Diabetic Retinopathy in a Tunisian Population, Seminars in Ophthalmology; Taylor & Francis: Oxford, UK, 2019; pp. 365–374. [Google Scholar]
- Luo, S.; Shi, C.; Wang, F.; Wu, Z. Association between the Angiotensin-Converting Enzyme (ACE) Genetic Polymorphism and Diabetic Retinopathy-A Meta-Analysis Comprising 10,168 Subjects. Int. J. Environ. Res. Public Health 2016, 13, 1142. [Google Scholar] [CrossRef]
- Liang, S.; Pan, M.; Hu, N.; Wu, Y.Y.; Chen, H.; Zhu, J.H.; Guan, H.J.; Sang, A.M. Association of angiotensin-converting enzyme gene 2350 G/A polymorphism with diabetic retinopathy in Chinese Han population. Mol. Biol. Rep. 2013, 40, 463–468. [Google Scholar] [CrossRef]
- Fan, Y.; Fu, Y.Y.; Chen, Z.; Hu, Y.Y.; Shen, J. Gene-gene interaction of erythropoietin gene polymorphisms and diabetic retinopathy in Chinese Han. Exp. Biol. Med. 2016, 241, 1524–1530. [Google Scholar] [CrossRef]
- Abhary, S.; Burdon, K.P.; Casson, R.J.; Goggin, M.; Petrovsky, N.P.; Craig, J.E. Association between erythropoietin gene polymorphisms and diabetic retinopathy. Arch. Ophthalmol. 2010, 128, 102–106. [Google Scholar] [CrossRef]
- Mankoč Ramuš, S.; Pungeršek, G.; Petrovič, M.G.; Petrovič, D. The GG genotype of erythropoietin rs1617640 polymorphism affects the risk of proliferative diabetic retinopathy in Slovenian subjects with Type 2 diabetes mellitus: Enemy or ally? Acta Ophthalmol. 2021. [Google Scholar] [CrossRef]
- Vuori, N.; Sandholm, N.; Kumar, A.; Hietala, K.; Syreeni, A.; Forsblom, C.; Juuti-Uusitalo, K.; Skottman, H.; Imamura, M.; Maeda, S. CACNB2 is a novel susceptibility gene for diabetic retinopathy in type 1 diabetes. Diabetes 2019, 68, 2165–2174. [Google Scholar] [CrossRef]
- Grassi, M.A.; Tikhomirov, A.; Ramalingam, S.; Below, J.E.; Cox, N.J.; Nicolae, D.L. Genome-wide meta-analysis for severe diabetic retinopathy. Hum. Mol. Genet. 2011, 20, 2472–2481. [Google Scholar] [CrossRef]
- Fu, Y.-P.; Hallman, D.M.; Gonzalez, V.H.; Klein, B.E.; Klein, R.; Hayes, M.G.; Cox, N.J.; Bell, G.I.; Hanis, C.L. Identification of diabetic retinopathy genes through a genome-wide association study among Mexican-Americans from Starr County, Texas. J. Ophthalmol. 2009, 2010, 861291. [Google Scholar] [CrossRef]
- Burdon, K.P.; Fogarty, R.D.; Shen, W.; Abhary, S.; Kaidonis, G.; Appukuttan, B.; Hewitt, A.W.; Sharma, S.; Daniell, M.; Essex, R.W. Genome-wide association study for sight-threatening diabetic retinopathy reveals association with genetic variation near the GRB2 gene. Diabetologia 2015, 58, 2288–2297. [Google Scholar] [CrossRef]
- Pollack, S.; Igo, R.P.; Jensen, R.A.; Christiansen, M.; Li, X.; Cheng, C.-Y.; Ng, M.C.; Smith, A.V.; Rossin, E.J.; Segrè, A.V. Multiethnic genome-wide association study of diabetic retinopathy using liability threshold modeling of duration of diabetes and glycemic control. Diabetes 2019, 68, 441–456. [Google Scholar] [CrossRef]
- Imamura, M.; Takahashi, A.; Matsunami, M.; Horikoshi, M.; Iwata, M.; Araki, S.-I.; Toyoda, M.; Susarla, G.; Ahn, J.; Park, K.H. Genome-wide association studies identify two novel loci conferring susceptibility to diabetic retinopathy in Japanese patients with type 2 diabetes. Hum. Mol. Genet. 2021, 30, 716–726. [Google Scholar] [CrossRef]
- Mathebula, S.D. Polyol pathway: A possible mechanism of diabetes complications in the eye. Afr. Vis. Eye Health 2015, 74, 5. [Google Scholar] [CrossRef]
- Dvornik, D. Aldose reductase inhibitors as pathobiochemical probes. J. Diabetes Complicat. 1992, 6, 25–34. [Google Scholar] [CrossRef]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Leto, G.; Pricci, F.; Amadio, L.; Iacobini, C.; Cordone, S.; Diaz-Horta, O.; Romeo, G.; Barsotti, P.; Rotella, C.M.; di Mario, U. Increased retinal endothelial cell monolayer permeability induced by the diabetic milieu: Role of advanced non-enzymatic glycation and polyol pathway activation. Diabetes/Metab. Res. Rev. 2001, 17, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Moczulski, D.; Scott, L.; Antonellis, A.; Rogus, J.; Rich, S.; Warram, J.; Krolewski, A. Aldose reductase gene polymorphisms and susceptibility to diabetic nephropathy in type 1 diabetes mellitus. Diabet. Med. 2000, 17, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Ko, B.; Lam, K.L.; Watt, N.M.-S.; Chung, S.S.-M. An (AC) n dinucleotide repeat polymorphic marker at the 5′ end of the aldose reductase gene is associated with early-onset diabetic retinopathy in NIDDM patients. Diabetes 1995, 44, 727–732. [Google Scholar] [CrossRef]
- Li, W.; Chen, S.; Mei, Z.; Zhao, F.; Xiang, Y. Polymorphisms in sorbitol-aldose reductase (Polyol) pathway genes and their influence on risk of diabetic retinopathy among Han Chinese. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2019, 25, 7073. [Google Scholar] [CrossRef]
- Yang, B.; Millward, A.; Demaine, A. Functional differences between the susceptibility Z−2/C−106 and protective Z+2/T−106 promoter region polymorphisms of the aldose reductase gene may account for the association with diabetic microvascular complications. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2003, 1639, 1–7. [Google Scholar] [CrossRef]
- Katakami, N.; Kaneto, H.; Takahara, M.; Matsuoka, T.; Imamura, K.; Ishibashi, F.; Kanda, T.; Kawai, K.; Osonoi, T.; Matsuhisa, M. Aldose reductase C-106T gene polymorphism is associated with diabetic retinopathy in Japanese patients with Type 2 diabetes. Diabetes Res. Clin. Pract. 2011, 92, e57–e60. [Google Scholar] [CrossRef]
- Rezaee, M.R.; Amiri, A.A.; Hashemi-Soteh, M.B.; Daneshvar, F.; Emady-Jamaly, R.; Jafari, R.; Soleimani, B.; Haghiaminjan, H. Aldose reductase C-106T gene polymorphism in Type 2 diabetics with microangiopathy in Iranian individuals. Indian J. Endocrinol. Metab. 2015, 19, 95–99. [Google Scholar]
- Kaur, N.; Vanita, V. Association of aldose reductase gene (AKR1B1) polymorphism with diabetic retinopathy. Diabetes Res. Clin. Pract. 2016, 121, 41–48. [Google Scholar] [CrossRef]
- SIu, M.; Bushuieva, O.; Ziablitsev, S.; Natrus, L. Relationship of the AKR1B1 rs759853 and rs9640883 with the development of diabetic retinopathy. J. Ophthalmol. 2017, 2, 3–7. [Google Scholar]
- Mogilevskyy, S.Y.; Panchenko, I.O.; Ziablitsev, S.V. Predicting the risk of diabetic retinopathy-associated macular edema in patients with type 2 diabetes mellitus. Oftalmol. Zhurnal 2019, 3–8. [Google Scholar] [CrossRef]
- Mrozikiewicz-Rakowska, B.; Łukawska, M.; Nehring, P.; Szymański, K.; Sobczyk-Kopcioł, A.; Krzyżewska, M.; Maroszek, P.; Płoski, R.; Czupryniak, L. Genetic predictors associated with diabetic retinopathy in patients with diabetic foot. Pol. Arch. Intern. Med. 2018, 128, 35–42. [Google Scholar]
- Lin, S.; Peng, Y.; Cao, M.; Chen, R.; Hu, J.; Pu, Z.; Cai, Z.; Mou, L. Association between Aldose Reductase Gene C (-106) T Polymorphism and Diabetic Retinopathy: A Systematic Review and Meta-Analysis. Ophthalmic Res. 2020, 63, 224–233. [Google Scholar] [CrossRef]
- Bushuyeva, O.V. New factors of diabetic retinopathy progression in Type 2 diabetes mellitus patients. East. Eur. Sci. J. Med. Sci. 2020, 1, 54–57. [Google Scholar]
- Abu-Hassan, D.; Al-Bdour, M.; Saleh, I.; Freihat, M.; El-Khateeb, M. The relationship between aldose reductase gene C106T polymorphism and the severity of retinopathy in Type 2 diabetic patients: A case–control study. J. Res. Med. Sci. 2021, 26, 2. [Google Scholar] [CrossRef]
- Deng, Y.; Yang, X.-F.; Gu, H.; Lim, A.; Ulziibat, M.; Snellingen, T.; Xu, J.; Ma, K.; Liu, N.-p. Association of C (-106) T polymorphism in aldose reductase gene with diabetic retinopathy in Chinese patients with Type 2 diabetes mellitus. Chin. Med. Sci. J. 2014, 29, 1–6. [Google Scholar] [CrossRef]
- Song, Z.-D.; Tao, Y.; Han, N.; Wu, Y.-Z. Association of the aldose reductase-106TT genotype with increased risk for diabetic retinopathy in the Chinese han population: An updated meta-analysis. Curr. Eye Res. 2016, 41, 1087–1091. [Google Scholar] [CrossRef]
- Shawki, H.A.; Elzehery, R.; Abo-hashem, E.M.; Shahin, M.; Youssef, M.M. Gene polymorphism of C106T “rs759853” is not associated with diabetic retinopathy in Egyptian patients with Type 2 diabetes mellitus. Gene Rep. 2020, 21, 100865. [Google Scholar] [CrossRef]
- Iskhakova, A.; Toropovsky, A.; Zolotarev, A.; Pavlova, O.; Komarova, M. Molecular and genetic aspects of early diagnosis of diabetic retinopathy. Int. Res. J. 2021, 3, 79–85. [Google Scholar]
- Shah, V.O.; Scavini, M.; Nikolic, J.; Sun, Y.; Vai, S.; Griffith, J.K.; Dorin, R.I.; Stidley, C.; Yacoub, M.; Vander Jagt, D.L. Z−2 microsatellite allele is linked to increased expression of the aldose reductase gene in diabetic nephropathy. J. Clin. Endocrinol. Metab. 1998, 83, 2886–2891. [Google Scholar] [CrossRef] [PubMed]
- Demaine, A.; Cross, D.; Millward, A. Polymorphisms of the aldose reductase gene and susceptibility to retinopathy in Type 1 diabetes mellitus. Investig. Ophthalmol. Vis. Sci. 2000, 41, 4064–4068. [Google Scholar]
- Wang, Y.; Ng, M.C.; Lee, S.-C.; So, W.-Y.; Tong, P.C.; Cockram, C.S.; Critchley, J.A.; Chan, J.C. Phenotypic heterogeneity and associations of two aldose reductase gene polymorphisms with nephropathy and retinopathy in Type 2 diabetes. Diabetes Care 2003, 26, 2410–2415. [Google Scholar] [CrossRef][Green Version]
- Mi, W.; Xia, Y.; Bian, Y. Meta-analysis of the association between aldose reductase gene (CA)n microsatellite variants and risk of diabetic retinopathy. Exp. Ther. Med. 2019, 18. [Google Scholar] [CrossRef]
- Fan, W.-Y.; Gu, H.; Yang, X.-F.; She, C.-Y.; Liu, X.-P.; Liu, N.-P. Association of candidate gene polymorphisms with diabetic retinopathy in Chinese patients with type 2 diabetes. Int. J. Ophthalmol. 2020, 13, 301. [Google Scholar] [CrossRef]
- Bucala, R.; Vlassara, H. Advanced glycosylation end products in diabetic renal and vascular disease. Am. J. Kidney Dis. 1995, 26, 875–888. [Google Scholar] [CrossRef]
- Vlassara, H.; Palace, M.R. Diabetes and advanced glycation endproducts. J. Intern. Med. 2002, 251, 87–101. [Google Scholar] [CrossRef]
- Stitt, A.W.; Li, Y.M.; Gardiner, T.A.; Bucala, R.; Archer, D.B.; Vlassara, H. Advanced glycation end products (AGEs) co-localize with AGE receptors in the retinal vasculature of diabetic and of AGE-infused rats. Am. J. Pathol 1997, 150, 523–531. [Google Scholar]
- Hughes, S.J.; Wall, N.; Scholfield, C.N.; McGeown, J.G.; Gardiner, T.A.; Stitt, A.W.; Curtis, T.M. Advanced glycation endproduct modified basement membrane attenuates endothelin-1 induced [Ca2+]i signalling and contraction in retinal microvascular pericytes. Mol. Vis. 2004, 10, 996–1004. [Google Scholar]
- Chen, B.H.; Jiang, D.Y.; Tang, L.S. Advanced glycation end-products induce apoptosis involving the signaling pathways of oxidative stress in bovine retinal pericytes. Life Sci. 2006, 79, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Neeper, M.; Schmidt, A.M.; Brett, J.; Yan, S.D.; Wang, F.; Pan, Y.C.; Elliston, K.; Stern, D.; Shaw, A. Cloning and expression of a cell surface receptor for advanced glycosylation end products of proteins. J. Biol. Chem. 1992, 267, 14998–15004. [Google Scholar] [CrossRef]
- Yamagishi, S.; Fujimori, H.; Yonekura, H.; Yamamoto, Y.; Yamamoto, H. Advanced glycation endproducts inhibit prostacyclin production and induce plasminogen activator inhibitor-1 in human microvascular endothelial cells. Diabetologia 1998, 41, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, N.; Kim, Y.S.; Lanting, L.; Natarajan, R. Regulation of cyclooxygenase-2 expression in monocytes by ligation of the receptor for advanced glycation end products. J. Biol. Chem. 2003, 278, 34834–34844. [Google Scholar] [CrossRef]
- Schmidt, A.M.; Hori, O.; Chen, J.X.; Li, J.F.; Crandall, J.; Zhang, J.; Cao, R.; Yan, S.D.; Brett, J.; Stern, D. Advanced glycation endproducts interacting with their endothelial receptor induce expression of vascular cell adhesion molecule-1 (VCAM-1) in cultured human endothelial cells and in mice. A potential mechanism for the accelerated vasculopathy of diabetes. J. Clin. Investig. 1995, 96, 1395–1403. [Google Scholar] [CrossRef]
- Wautier, J.L.; Schmidt, A.M. Protein glycation: A firm link to endothelial cell dysfunction. Circ. Res. 2004, 95, 233–238. [Google Scholar] [CrossRef]
- Chen, L.; Cui, Y.; Li, B.; Weng, J.; Wang, W.; Zhang, S.; Huang, X.; Guo, X.; Huang, Q. Advanced glycation end products induce immature angiogenesis in in vivo and ex vivo mouse models. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, H519–H533. [Google Scholar] [CrossRef]
- Gui, F.; You, Z.; Fu, S.; Wu, H.; Zhang, Y. Endothelial Dysfunction in Diabetic Retinopathy. Front. Endocrinol. 2020, 11. [Google Scholar] [CrossRef]
- Erna, R.; Sastrawan, D.; Saleh, M.I. Effect of RAGE -429T/C and Gly82Ser Gene Polymorphism in Diabetic Retinopathy in General Hospital Mohammad Hoesin Palembang. J. Res. Med. Dent. Sci. 2017, 5, 19–23. [Google Scholar]
- Yuan, D.; Yuan, D.; Liu, Q. Association of the receptor for advanced glycation end products gene polymorphisms with diabetic retinopathy in type 2 diabetes: A meta-analysis. Ophthalmologica 2012, 227, 223–232. [Google Scholar] [CrossRef]
- Qayyum, S.; Afzal, M.; Naveed, A.K. Association analysis of 374T/A (rs1800624) receptor for advanced glycation end-products (RAGE) gene polymorphism with diabetic retinopathy in Pakistani patients. Pak. J. Med. Sci. 2021, 37, 733. [Google Scholar] [CrossRef]
- Tao, D.; Mai, X.; Zhang, T.; Mei, Y. Association between the RAGE (receptor for advanced glycation end-products)-374T/A gene polymorphism and diabetic retinopathy in T2DM. Rev. Assoc. MÉDica Bras. 2017, 63, 971–977. [Google Scholar] [CrossRef]
- Yu, W.; Yang, J.; Sui, W.; Qu, B.; Huang, P.; Chen, Y. Association of genetic variants in the receptor for advanced glycation end products gene with diabetic retinopathy: A meta-analysis. Medicine 2016, 95, e4463. [Google Scholar] [CrossRef]
- Lu, W.; Feng, B. The-374A allele of the RAGE gene as a potential protective factor for vascular complications in type 2 diabetes: A meta-analysis. Tohoku J. Exp. Med. 2010, 220, 291–297. [Google Scholar] [CrossRef]
- Ng, Z.X.; Kuppusamy, U.R.; Tajunisah, I.; Fong, K.C.S.; Chua, K.H. Association analysis of− 429T/C and− 374T/A polymorphisms of receptor of advanced glycation end products (RAGE) gene in Malaysian with type 2 diabetic retinopathy. Diabetes Res. Clin. Pract. 2012, 95, 372–377. [Google Scholar] [CrossRef]
- Vanita, V. Association of RAGE (p. Gly82Ser) and MnSOD (p. Val16Ala) polymorphisms with diabetic retinopathy in T2DM patients from north India. Diabetes Res. Clin. Pract. 2014, 104, 155–162. [Google Scholar] [CrossRef]
- Jia, C.; Yong, Y.; Tianhua, X.; Zheyao, G.; Jian, Z. Association of Gly82Ser polymorphism of RAGE gene with diabetic retinopathy in Han people with type 2 diabetes of Wuxi region. Chin. J. Exp. Ophthalmol. 2016, 34, 910–914. [Google Scholar]
- Kang, P.; Tian, C.; Jia, C. Association of RAGE gene polymorphisms with type 2 diabetes mellitus, diabetic retinopathy and diabetic nephropathy. Gene 2012, 500, 1–9. [Google Scholar] [CrossRef]
- Niu, W.; Qi, Y.; Wu, Z.; Liu, Y.; Zhu, D.; Jin, W. A meta-analysis of receptor for advanced glycation end products gene: Four well-evaluated polymorphisms with diabetes mellitus. Mol. Cell. Endocrinol. 2012, 358, 9–17. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, L.; Wang, L.; Liao, Y.; Wu, Z.; Ye, F.; Xu, S.; Yi, L. Association of 1704G/T and G82S polymorphisms in the receptor for advanced glycation end products gene with diabetic retinopathy in Chinese population. J. Endocrinol. Investig. 2009, 32, 258–262. [Google Scholar] [CrossRef]
- Yoshioka, K.; Yoshida, T.; Takakura, Y.; Umekawa, T.; Kogure, A.; Toda, H.; Yoshikawa, T. Relation between polymorphisms G1704T and G82S of rage gene and diabetic retinopathy in Japanese type 2 diabetic patients. Intern. Med. 2005, 44, 417–421. [Google Scholar] [CrossRef]
- Yang, L.; Wu, Q.; Li, Y.; Fan, X.; Hao, Y.; Sun, H.; Cui, Y.; Han, L. Association of the receptor for advanced glycation end products gene polymorphisms and circulating RAGE levels with diabetic retinopathy in the Chinese population. J. Diabetes Res. 2013, 2013, 264579. [Google Scholar] [CrossRef]
- Holmes, D.I.; Zachary, I. The vascular endothelial growth factor (VEGF) family: Angiogenic factors in health and disease. Genome Biol. 2005, 6, 1–10. [Google Scholar] [CrossRef][Green Version]
- Zhou, Z.; Ju, H.; Sun, M.; Chen, H. Serum Vascular Endothelial Growth Factor Levels Correlate with Severity of Retinopathy in Diabetic Patients: A Systematic Review and Meta-Analysis. Dis. Markers 2019, 2019, 9401628. [Google Scholar] [CrossRef]
- Morello, C.M. Etiology and natural history of diabetic retinopathy: An overview. Am. J. Health Syst. Pharm. 2007, 64, S3–S7. [Google Scholar] [CrossRef] [PubMed]
- Stevens, A.; Soden, J.; Brenchley, P.E.; Ralph, S.; Ray, D.W. Haplotype analysis of the polymorphic human vascular endothelial growth factor gene promoter. Cancer Res. 2003, 63, 812–816. [Google Scholar]
- Khan, S.Z.; Ajmal, N.; Shaikh, R. Diabetic Retinopathy and Vascular Endothelial Growth Factor Gene Insertion/Deletion Polymorphism. Can. J. Diabetes 2020, 44, 287–291. [Google Scholar] [CrossRef]
- Hu, L.; Gong, C.; Chen, X.; Zhou, H.; Yan, J.; Hong, W. Associations between Vascular Endothelial Growth Factor Gene Polymorphisms and Different Types of Diabetic Retinopathy Susceptibility: A Systematic Review and Meta-Analysis. J. Diabetes Res. 2021, 2021, 7059139. [Google Scholar] [CrossRef]
- Xie, X.-J.; Yang, Y.-M.; Jiang, J.-K.; Lu, Y.-Q. Association between the vascular endothelial growth factor single nucleotide polymorphisms and diabetic retinopathy risk: A meta-analysis. J. Diabetes 2017, 9, 738–753. [Google Scholar] [CrossRef]
- Al-Kateb, H.; Mirea, L.; Xie, X.; Sun, L.; Liu, M.; Chen, H.; Bull, S.B.; Boright, A.P.; Paterson, A.D. Multiple variants in vascular endothelial growth factor (VEGFA) are risk factors for time to severe retinopathy in type 1 diabetes: The DCCT/EDIC genetics study. Diabetes 2007, 56, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Abhary, S.; Burdon, K.P.; Gupta, A.; Lake, S.; Selva, D.; Petrovsky, N.; Craig, J.E. Common sequence variation in the VEGFA gene predicts risk of diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2009, 50, 5552–5558. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Paterson, A.D.; Roshandel, D.; Raza, A.; Ajmal, M.; Waheed, N.K.; Azam, M.; Qamar, R. Association of IGF1 and VEGFA polymorphisms with diabetic retinopathy in Pakistani population. Acta Diabetol. 2020, 57, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Sajovic, J.; Cilenšek, I.; Mankoč, S.; Tajnšek, Š.; Kunej, T.; Petrovič, D.; Petrovič, M.G. Vascular endothelial growth factor (VEGF)-related polymorphisms rs10738760 and rs6921438 are not risk factors for proliferative diabetic retinopathy (PDR) in patients with type 2 diabetes mellitus (T2DM). Bosn. J. Basic Med. Sci. 2019, 19, 94. [Google Scholar] [CrossRef] [PubMed]
- Terzić, R.; Cilenšek, I.; Pleskovič, R.Z.; Mankoč, S.; Milutinović, A. Vascular endothelial growth factor (VEGF)-related single nucleotide polymorphisms rs10738760 and rs6921438 are not associated with diabetic retinopathy (DR) in Slovenian patients with type 2 diabetes mellitus (T2DM). Bosn. J. Basic Med. Sci. 2017, 17, 328. [Google Scholar] [CrossRef][Green Version]
- Gonzalez-Salinas, R.; Garcia-Gutierrez, M.C.; Garcia-Aguirre, G.; Morales-Canton, V.; Velez-Montoya, R.; Soberon-Ventura, V.R.; Gonzalez, V.; Lechuga, R.; Garcia-Solis, P.; Garcia-Gutierrez, D.G. Evaluation of VEGF gene polymorphisms and proliferative diabetic retinopathy in Mexican population. Int. J. Ophthalmol. 2017, 10, 135. [Google Scholar]
- Amer, A.K.; Khalaf, N.A.; Aboelmakarem, S.H.; Elsobky, M.S.; Abdelrasoul, M.R.; Abdelazeem, A.A.; Noweir, S.R.; Refaat, S.; Moemen, L.A.; Mohammed, S.A.; et al. Vascular endothelial growth factor +405G/C polymorphism as a predictor of diabetic retinopathy. Bull. Natl. Res. Cent. 2020, 44, 54. [Google Scholar] [CrossRef][Green Version]
- Hussin, H.Y.; Hussein, W.; Ibrahim, S.; Kambal, S.; Almhdi, S.; Abdrabo, A.E. Association of Vascular Endothelial Growth Factor +405G/C Polymorphism with Diabetic Retinopathy among Sudanese Patients. Am. J. Biomed. Sci. Res. 2021, 11, 437–443. [Google Scholar] [CrossRef]
- Chun, M.-Y.; Hwang, H.-S.; Cho, H.-Y.; Chun, H.-J.; Woo, J.-T.; Lee, K.-W.; Nam, M.-S.; Baik, S.-H.; Kim, Y.-S.; Park, Y. Association of vascular endothelial growth factor polymorphisms with nonproliferative and proliferative diabetic retinopathy. J. Clin. Endocrinol. Metab 2010, 95, 3547–3551. [Google Scholar] [CrossRef]
- Yang, X.; Deng, Y.; Gu, H.; Ren, X.; Li, N.; Lim, A.; Snellingen, T.; Liu, X.; Wang, N.; Liu, N. Candidate gene association study for diabetic retinopathy in Chinese patients with type 2 diabetes. Mol. Vis. 2014, 20, 200–214. [Google Scholar]
- Nakamura, S.; Iwasaki, N.; Funatsu, H.; Kitano, S.; Iwamoto, Y. Impact of variants in the VEGF gene on progression of proliferative diabetic retinopathy. Graefes Arch. Clin. Exp. Ophthalmol. 2009, 247, 21–26. [Google Scholar] [CrossRef]
- Bleda, S.; De Haro, J.; Varela, C.; Esparza, L.; Ferruelo, A.; Acin, F. Vascular endothelial growth factor polymorphisms are involved in the late vascular complications in Type II diabetic patients. Diabetes Vasc. Dis. Res. 2011, 9, 68–74. [Google Scholar] [CrossRef]
- Abdel Fattah, R.; Eltanamly, R.; Nabih, M.; Kamal, M. Vascular endothelial growth factor gene polymorphism is not associated with diabetic retinopathy in Egyptian Patients. Middle East. Afr. J. Ophthalmol. 2016, 23, 75–78. [Google Scholar]
- Fan, X.; Wu, Q.; Li, Y.; Hao, Y.; Ning, N.; Kang, Z.; Cui, Y.; Liu, R.; Han, L. Association of polymorphisms in the vascular endothelial growth factor gene and its serum levels with diabetic retinopathy in Chinese patients with type 2 diabetes: A cross-sectional study. Chin. Med. J. 2014, 127, 651–657. [Google Scholar]
- Han, L.; Zhang, L.; Xing, W.; Zhuo, R.; Lin, X.; Hao, Y.; Wu, Q.; Zhao, J. The associations between VEGF gene polymorphisms and diabetic retinopathy susceptibility: A meta-analysis of 11 case-control studies. J. Diabetes Res. 2014, 2014, 805801. [Google Scholar] [CrossRef]
- Dorner, G.T.; Garhofer, G.; Kiss, B.; Polska, E.; Polak, K.; Riva, C.E.; Schmetterer, L. Nitric oxide regulates retinal vascular tone in humans. Am. J. Physiol. Heart Circ. Physiol. 2003, 285, H631–H636. [Google Scholar] [CrossRef]
- Chen, K.; Pittman, R.N.; Popel, A.S. Nitric oxide in the vasculature: Where does it come from and where does it go? A quantitative perspective. Antioxid Redox Signal 2008, 10, 1185–1198. [Google Scholar] [CrossRef]
- Sessa, W.C. eNOS at a glance. J. Cell Sci. 2004, 117, 2427–2429. [Google Scholar] [CrossRef]
- Albrecht, E.W.; Stegeman, C.A.; Heeringa, P.; Henning, R.H.; van Goor, H. Protective role of endothelial nitric oxide synthase. J. Pathol. A J. Pathol. Soc. Great Br. Irel. 2003, 199, 8–17. [Google Scholar] [CrossRef]
- Gericke, A.; Wolff, I.; Musayeva, A.; Zadeh, J.K.; Manicam, C.; Pfeiffer, N.; Li, H.; Xia, N. Retinal arteriole reactivity in mice lacking the endothelial nitric oxide synthase (eNOS) gene. Exp. Eye Res. 2019, 181, 150–156. [Google Scholar] [CrossRef]
- Li, Q.; Verma, A.; Han, P.-Y.; Nakagawa, T.; Johnson, R.J.; Grant, M.B.; Campbell-Thompson, M.; Jarajapu, Y.P.; Lei, B.; Hauswirth, W.W. Diabetic eNOS-knockout mice develop accelerated retinopathy. Investig. Ophthalmol. Vis. Sci. 2010, 51, 5240–5246. [Google Scholar] [CrossRef]
- McAuley, A.K.; Sanfilippo, P.G.; Hewitt, A.W.; Liang, H.; Lamoureux, E.; Wang, J.J.; Connell, P.P. Vitreous biomarkers in diabetic retinopathy: A systematic review and meta-analysis. J. Diabetes Complicat. 2014, 28, 419–425. [Google Scholar] [CrossRef]
- Mihoubi, E.; Bouldjennet, F.; Raache, R.; Amroun, H.; Azzouz, M.; Benazouz, N.; Touil-Boukoffa, C.; Attal, N. T-786C endothelial nitric oxide gene polymorphism and type 1 diabetic retinopathy in the Algerian population. J. Fr. d’Ophtalmol. 2019, 42, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Cheema, B.S.; Sharma, R.; Bhansali, A.; Khullar, M. Endothelial nitric oxide synthase gene polymorphism and Type 2 diabetic retinopathy among Asian Indians. Acta Diabetol. 2012, 49, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Li, T.; Zheng, B.; Zheng, Z. Nitric oxide synthase 3 (NOS3) 4b/a, T-786C and G894T polymorphisms in association with diabetic retinopathy susceptibility: A meta-analysis. Ophthalmic Genet. 2012, 33, 200–207. [Google Scholar] [CrossRef]
- Qian-Qian, Y.; Yong, Y.; Jing, Z.; Dong-Hong, F.; Tian-Hua, X.; Li, Y.; Lan, L.; Jia, C.; Zhe-Yao, G. Association between a 27-bp variable number of tandem repeat polymorphism in intron 4 of the eNOS gene and risk for diabetic retinopathy Type 2 diabetes mellitus: A meta-analysis. Curr. Eye Res. 2014, 39, 1052–1058. [Google Scholar] [CrossRef] [PubMed]
- Cilenšek, I.; Mankoč, S.; Globočnik Petrovič, M.; Petrovič, D. The 4a/4a genotype of the VNTR polymorphism for endothelial nitric oxide synthase (eNOS) gene predicts risk for proliferative diabetic retinopathy in Slovenian patients (Caucasians) with type 2 diabetes mellitus. Mol. Biol. Rep. 2012, 39, 7061–7067. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.-J.; Chen, R.; Ren, H.-Z.; Guo, X.; Guo, J.; Chen, L.-M. Association between eNOS 4b/a polymorphism and the risk of diabetic retinopathy in type 2 diabetes mellitus: A meta-analysis. J. Diabetes Res. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- She, C.; Yang, X.; Gu, H.; Deng, Y.; Xu, J.; Ma, K.; Liu, N. The association of variable number of tandem repeats polymorphism in the endothelial nitric oxide synthase gene and diabetic retinopathy. Zhonghua Yan Ke Za Zhi 2015, 51, 338–343. [Google Scholar] [PubMed]
- Momeni, A.; Chaleshtori, M.H.; Saadatmand, S.; Kheiri, S. Correlation of Endothelial Nitric Oxide Synthase Gene Polymorphism (GG, TT and GT Genotype) with Proteinuria and Retinopathy in Type 2 Diabetic Patients. J. Clin. Diagn Res. 2016, 10, Oc32–Oc35. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, E.L.; Phipps, J.A.; Ward, M.M.; Vessey, K.A.; Wilkinson-Berka, J.L. The renin–angiotensin system in retinal health and disease: Its influence on neurons, glia and the vasculature. Prog. Retin. Eye Res. 2010, 29, 284–311. [Google Scholar] [CrossRef]
- Li, Y.; Yan, Z.; Chaudhry, K.; Kazlauskas, A. The Renin-Angiotensin-Aldosterone System (RAAS) Is One of the Effectors by Which Vascular Endothelial Growth Factor (VEGF)/Anti-VEGF Controls the Endothelial Cell Barrier. Am. J. Pathol. 2020, 190, 1971–1981. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wang, F.; Zhang, Y.; Zhao, S.H.; Zhao, W.J.; Yan, S.L.; Wang, Y.G. Effects of RAS inhibitors on diabetic retinopathy: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2015, 3, 263–274. [Google Scholar] [CrossRef]
- Sjølie, A.K.; Klein, R.; Porta, M.; Orchard, T.; Fuller, J.; Parving, H.H.; Bilous, R.; Chaturvedi, N. Effect of candesartan on progression and regression of retinopathy in type 2 diabetes (DIRECT-Protect 2): A randomised placebo-controlled trial. Lancet 2008, 372, 1385–1393. [Google Scholar] [CrossRef]
- Mauer, M.; Zinman, B.; Gardiner, R.; Suissa, S.; Sinaiko, A.; Strand, T.; Drummond, K.; Donnelly, S.; Goodyer, P.; Gubler, M.C.; et al. Renal and retinal effects of enalapril and losartan in type 1 diabetes. N. Engl. J. Med. 2009, 361, 40–51. [Google Scholar] [CrossRef]
- Neroev, V.V.; Chesnokova, N.B.; Pavlenko, T.A.; Okhotsimskaya, T.D.; Beznos, O.V.; Fadeeva, V.A.; Struchkova, S.V. Changes in the concentration of angiotensin II, angiotensin-transforming enzyme and matrix metaloproteinase-9 in tear fluid and serum in patients with diabetic retinopathy. Ophthalmology 2020, 17, 771–778. [Google Scholar]
- Rigat, B.; Hubert, C.; Alhenc-Gelas, F.; Cambien, F.; Corvol, P.; Soubrier, F. An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J. Clin. Investig. 1990, 86, 1343–1346. [Google Scholar] [CrossRef]
- Li, N.; Yang, X.F.; Gu, H.; Deng, Y.; Xu, J.; Ma, K.; Liu, N.P. Relationship of angiotensin converting enzyme gene polymorphism with diabetic retinopathy. Zhonghua Yan Ke Za Zhi 2013, 49, 52–57. [Google Scholar]
- Pirozzi, F.F.; Belini Junior, E.; Okumura, J.V.; Salvarani, M.; Bonini-Domingos, C.R.; Ruiz, M.A. The relationship between of ACE I/D and the MTHFR C677T polymorphisms in the pathophysiology of type 2 diabetes mellitus in a population of Brazilian obese patients. Arch. Endocrinol. Metab. 2018, 62, 21–26. [Google Scholar] [CrossRef]
- Qiao, Y.C.; Wang, M.; Pan, Y.H.; Zhang, X.X.; Tian, F.; Chen, Y.L.; Zhao, H.L. The relationship between ACE/AGT gene polymorphisms and the risk of diabetic retinopathy in Chinese patients with type 2 diabetes. J. Renin Angiotensin Aldosterone Syst. 2018, 19, 1470320317752955. [Google Scholar] [CrossRef]
- Wiwanitkit, V. Angiotensin-converting enzyme gene polymorphism is correlated to diabetic retinopathy: A meta-analysis. J. Diabetes Complicat. 2008, 22, 144–146. [Google Scholar] [CrossRef]
- Reid, G.; Lois, N. Erythropoietin in diabetic retinopathy. Vis. Res. 2017, 139, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Davidović, S.; Babić, N.; Jovanović, S.; Barišić, S.; Grković, D.; Miljković, A. Serum erythropoietin concentration and its correlation with stage of diabetic retinopathy. BMC Ophthalmol. 2019, 19, 227. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Liu, N. Meta-analysis of association between EPO gene polymorphisms and diabetic retinopathy. Ophthalmol. China 2020, 29, 454–457. [Google Scholar]
- Li, H.; Xu, H.; Li, Y.; Zhao, D.; Ma, B. Associations between erythropoietin polymorphisms and risk of diabetic microvascular complications. Oncotarget 2017, 8, 112675–112684. [Google Scholar] [CrossRef] [PubMed]
- Pulst, S.M. Genetic linkage analysis. Arch. Neurol. 1999, 56, 667–672. [Google Scholar] [CrossRef]
- Ott, J.; Wang, J.; Leal, S.M. Genetic linkage analysis in the age of whole-genome sequencing. Nat. Rev. Genet. 2015, 16, 275–284. [Google Scholar] [CrossRef]
- Hallman, D.M.; Boerwinkle, E.; Gonzalez, V.H.; Klein, B.E.; Klein, R.; Hanis, C.L. A genome-wide linkage scan for diabetic retinopathy susceptibility genes in Mexican Americans with type 2 diabetes from Starr County, Texas. Diabetes 2007, 56, 1167–1173. [Google Scholar] [CrossRef][Green Version]
- Norrgard, K. Genetic Variation and Disease: GWAS. Nat. Educ. 2008, 1, 87. [Google Scholar]
- Genome-Wide Association Studies Fact Sheet. Available online: https://www.genome.gov/about-genomics/fact-sheets/Genome-Wide-Association-Studies-Fact-Sheet (accessed on 30 June 2021).
- Haines, J.L.; Hauser, M.A.; Schmidt, S.; Scott, W.K.; Olson, L.M.; Gallins, P.; Spencer, K.L.; Kwan, S.Y.; Noureddine, M.; Gilbert, J.R. Complement factor H variant increases the risk of age-related macular degeneration. Science 2005, 308, 419–421. [Google Scholar]
- Tony Burdett, E.H.D.W.S.E.-E.B.I.N. GWAS Catalog. Available online: https://www.ebi.ac.uk/gwas/search?query=diabetic%20retinopathy (accessed on 30 June 2021).
- Ban, N.; Yamada, Y.; Someya, Y.; Ihara, Y.; Adachi, T.; Kubota, A.; Watanabe, R.; Kuroe, A.; Inada, A.; Miyawaki, K. Activating transcription factor-2 is a positive regulator in CaM kinase IV-induced human insulin gene expression. Diabetes 2000, 49, 1142–1148. [Google Scholar] [CrossRef]
- Simó-Servat, O.; Hernández, C.; Simó, R. Genetics in diabetic retinopathy: Current concepts and new insights. Curr. Genom. 2013, 14, 289–299. [Google Scholar] [CrossRef]
- Forrest, I.S.; Chaudhary, K.; Paranjpe, I.; Vy, H.M.T.; Marquez-Luna, C.; Rocheleau, G.; Saha, A.; Chan, L.; Van Vleck, T.; Loos, R.J. Genome-wide polygenic risk score for retinopathy of type 2 diabetes. Hum. Mol. Genet. 2021, 30, 952–960. [Google Scholar] [CrossRef]
| Candidate Gene | Gene Location | Online Mendelian Inheritance in Man (OMIM) Entry | Polymorphisms | Effect on DR | References |
|---|---|---|---|---|---|
| Aldose Reductase (AKR1B1) | 7q33 | 103880 | rs759853 | Protection from DR in T1D patients | [29] |
| Z-2 Z Z+2 | Risk for DR in T1 and T2D Protection from DR in T2D | [26] | |||
| rs9640883 | Duration of diabetes T1D and T2D | [30] | |||
| Receptor for Advanced Glycation End Product (AGER) | 6p21.32 | 600214 | rs1800624 | Risk of DR T1D | [31] |
| rs2070600 | Risk of DR in T2D | [32] | |||
| Vascular Endothelial Growth Factor (VEGFA) | 6p21.1 | 192240 | rs3025039 rs3025021, rs13207351 rs2146323 rs2010963 rs25648, rs833061 rs2010963 | Risk of DR in T2D Risk of DR in T2D Risk of DR in T2D Risk of DR in T2D Risk of PDR in T2D | [33] |
| Endothelial Nitric Oxide Synthase (NOS3) | 7q36.1 | 163729 | rs869109213 rs2070744 | Risk of DR in T2D | [34] |
| Angiotensin-I Converting Enzyme (ACE) | 17q23.3 | 106180 | rs1799752 rs4343 | Risk of DR in T2D Risk of DR in T2D | [35,36] |
| Erythropoietin (EPO) | 7q22.1 | 133170 | rs551238 rs1617640 rs507392 | Risk of DR | [37,38,39] |
| Calcium channel voltage dependent beta-2 sub unit (CACNB2) | 10p12.33-p12.31 | 600003 | rs202152674 rs137886839 | Increased risk of PDR | [40] |
| Intergenic locus in between AKT3 and ZNF238 | 1:24401312 | rs476141 | Increased risk of DR | [41] | |
| Caclium/Calmodulin-Dependent Protein Kinase IV (CAMK4) | 5q22.1 | 114080 | rs2300782 | Increased risk of DR | [42] |
| Formin 1 (FMN1) | 15q13.3 | 136535 | rs10519765 | Increased risk of DR | [42] |
| Growth factor receptor bound-2 (GRB2) | 17q25.1 | 108355 | rs9896052 | Sight threatening DR | [43] |
| Valosin -containing protein like (NVL) | 1q42.11 | 602426 | rs142293996 | Increased risk of DR | [44] |
| STT3 Oligosaccharyltransferase Complex Catalytic Subunit B (STT3B) | 3p23 | 608605 | rs12630354 | Increased risk of DR | [45] |
| Paralemmin-2 (PALM2AKAP2) | 9q31.3 | 604582 | rs140508424 | Increased risk of DR | [45] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhatwadekar, A.D.; Shughoury, A.; Belamkar, A.; Ciulla, T.A. Genetics of Diabetic Retinopathy, a Leading Cause of Irreversible Blindness in the Industrialized World. Genes 2021, 12, 1200. https://doi.org/10.3390/genes12081200
Bhatwadekar AD, Shughoury A, Belamkar A, Ciulla TA. Genetics of Diabetic Retinopathy, a Leading Cause of Irreversible Blindness in the Industrialized World. Genes. 2021; 12(8):1200. https://doi.org/10.3390/genes12081200
Chicago/Turabian StyleBhatwadekar, Ashay D., Aumer Shughoury, Ameya Belamkar, and Thomas A. Ciulla. 2021. "Genetics of Diabetic Retinopathy, a Leading Cause of Irreversible Blindness in the Industrialized World" Genes 12, no. 8: 1200. https://doi.org/10.3390/genes12081200
APA StyleBhatwadekar, A. D., Shughoury, A., Belamkar, A., & Ciulla, T. A. (2021). Genetics of Diabetic Retinopathy, a Leading Cause of Irreversible Blindness in the Industrialized World. Genes, 12(8), 1200. https://doi.org/10.3390/genes12081200







