Differences in Ovarian and Other Cancers Risks by Population and BRCA Mutation Location
Abstract
1. Introduction
2. Differences of the Prevalence of Germline BRCA Mutation in Ovarian Cancer Patients
2.1. Prevalence of Germline BRCA1/2 Mutation in Ovarian Cancer Patients
2.2. Histological Subtypes in BRCA-Related Ovarian Cancer
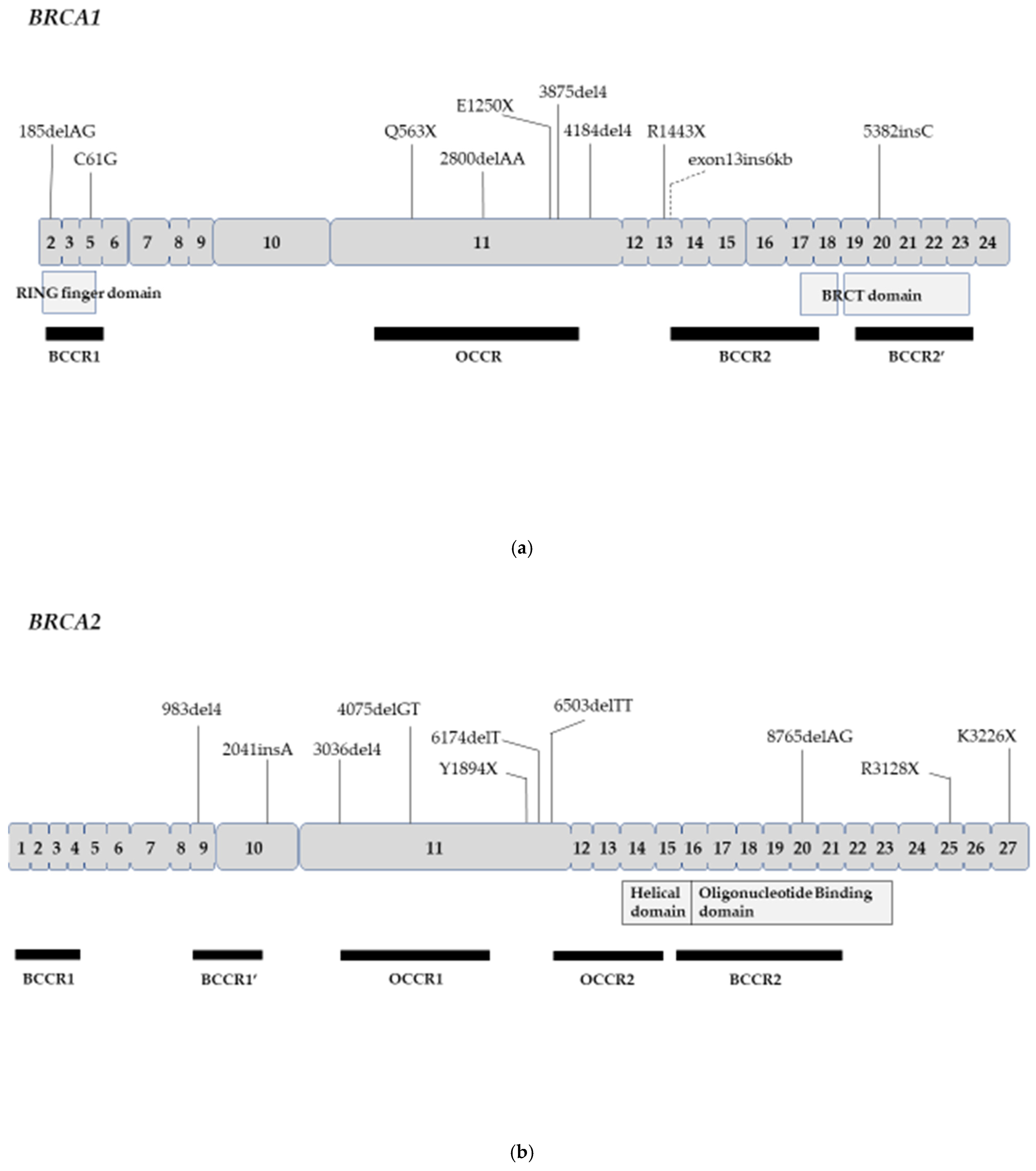
3. Association of Breast/Ovarian Cancer Risk and BRCA Mutation Location
4. Differences of other BRCA-Related Cancers Risks by BRCA1/2 Mutation
4.1. Contralateral Breast Cancer Risk
4.2. Male Breast Cancer
4.3. Prostate Cancer
4.4. Pancreatic Cancer
4.5. Melanoma
4.6. Endometrial Cancer
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Miki, Y.; Swensen, J.; Shattuck-Eidens, D.; Futreal, P.A.; Harshman, K.; Tavtigian, S.; Liu, Q.; Cochran, C.; Bennett, L.M.; Ding, W.; et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 1994, 266, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Wooster, R.; Neuhausen, S.L.; Mangion, J.; Quirk, Y.; Ford, D.; Collins, N.; Nguyen, K.; Seal, S.; Tran, T.; Averill, D.; et al. Localization of a breast cancer susceptibility gene, BRCA2, to chromosome 13q12-13. Science 1994, 265, 2088–2090. [Google Scholar] [CrossRef] [PubMed]
- Dullens, B.; de Putter, R.; Lambertini, M.; Toss, A.; Han, S.; Van Nieuwenhuysen, E.; Van Gorp, T.; Vanderstichele, A.; Van Ongeval, C.; Keupers, M.; et al. Cancer Surveillance in Healthy Carriers of Germline Pathogenic Variants in BRCA1/2: A Review of Secondary Prevention Guidelines. J. Oncol. 2020, 2020, 9873954. [Google Scholar] [CrossRef] [PubMed]
- Cipak, L.; Watanabe, N.; Bessho, T. The role of BRCA2 in replication-coupled DNA interstrand cross-link repair In Vitro. Nat. Struct. Mol. Biol. 2006, 13, 729–733. [Google Scholar] [CrossRef]
- Hu, C.; Hart, S.N.; Gnanaolivu, R.; Huang, H.; Lee, K.Y.; Na, J.; Gao, C.; Lilyquist, J.; Yadav, S.; Boddicker, N.J.; et al. A Population-Based Study of Genes Previously Implicated in Breast Cancer. N. Engl. J. Med. 2021, 384, 440–451. [Google Scholar] [CrossRef]
- Foulkes, W.D. Inherited susceptibility to common cancers. N. Engl. J. Med. 2008, 359, 2143–2153. [Google Scholar] [CrossRef]
- Metcalfe, K.A.; Poll, A.; Royer, R.; Llacuachaqui, M.; Tulman, A.; Sun, P.; Narod, S.A. Screening for founder mutations in BRCA1 and BRCA2 in unselected Jewish women. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 387–391. [Google Scholar] [CrossRef]
- Sekine, M.; Nagata, H.; Tsuji, S.; Hirai, Y.; Fujimoto, S.; Hatae, M.; Kobayashi, I.; Fujii, T.; Nagata, I.; Ushijima, K.; et al. Mutational analysis of BRCA1 and BRCA2 and clinicopathologic analysis of ovarian cancer in 82 ovarian cancer families: Two common founder mutations of BRCA1 in Japanese population. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2001, 7, 3144–3150. [Google Scholar]
- Sekine, M.; Nagata, H.; Tsuji, S.; Hirai, Y.; Fujimoto, S.; Hatae, M.; Kobayashi, I.; Fujii, T.; Nagata, I.; Ushijima, K.; et al. Localization of a novel susceptibility gene for familial ovarian cancer to chromosome 3p22-p25. Hum. Mol. Genet. 2001, 10, 1421–1429. [Google Scholar] [CrossRef]
- Genetics, A.C. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 103: Hereditary breast and ovarian cancer syndrome. Obstet. Gynecol. 2009, 113, 957–966. [Google Scholar] [CrossRef]
- Csokay, B.; Udvarhelyi, N.; Sulyok, Z.; Besznyak, I.; Ramus, S.; Ponder, B.; Olah, E. High frequency of germ-line BRCA2 mutations among Hungarian male breast cancer patients without family history. Cancer Res. 1999, 59, 995–998. [Google Scholar]
- Ji, J.; Hemminki, K. Familial risk for histology-specific bone cancers: An updated study in Sweden. Eur. J. Cancer 2006, 42, 2343–2349. [Google Scholar] [CrossRef] [PubMed]
- Mikaelsdottir, E.K.; Valgeirsdottir, S.; Eyfjord, J.E.; Rafnar, T. The Icelandic founder mutation BRCA2 999del5: Analysis of expression. Breast Cancer Res. 2004, 6, R284–R290. [Google Scholar] [CrossRef]
- Petrij-Bosch, A.; Peelen, T.; van Vliet, M.; van Eijk, R.; Olmer, R.; Drüsedau, M.; Hogervorst, F.B.; Hageman, S.; Arts, P.J.; Ligtenberg, M.J.; et al. BRCA1 genomic deletions are major founder mutations in Dutch breast cancer patients. Nat. Genet. 1997, 17, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Tonin, P.N.; Mes-Masson, A.M.; Futreal, P.A.; Morgan, K.; Mahon, M.; Foulkes, W.D.; Cole, D.E.; Provencher, D.; Ghadirian, P.; Narod, S.A. Founder BRCA1 and BRCA2 mutations in French Canadian breast and ovarian cancer families. Am. J. Hum. Genet. 1998, 63, 1341–1351. [Google Scholar] [CrossRef]
- Díez, O.; Osorio, A.; Durán, M.; Martinez-Ferrandis, J.I.; de la Hoya, M.; Salazar, R.; Vega, A.; Campos, B.; Rodríguez-López, R.; Velasco, E.; et al. Analysis of BRCA1 and BRCA2 genes in Spanish breast/ovarian cancer patients: A high proportion of mutations unique to Spain and evidence of founder effects. Hum. Mutat. 2003, 22, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Tung, N.; Battelli, C.; Allen, B.; Kaldate, R.; Bhatnagar, S.; Bowles, K.; Timms, K.; Garber, J.E.; Herold, C.; Ellisen, L.; et al. Frequency of mutations in individuals with breast cancer referred for BRCA1 and BRCA2 testing using next-generation sequencing with a 25-gene panel. Cancer 2015, 121, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Buys, S.S.; Sandbach, J.F.; Gammon, A.; Patel, G.; Kidd, J.; Brown, K.L.; Sharma, L.; Saam, J.; Lancaster, J.; Daly, M.B. A study of over 35,000 women with breast cancer tested with a 25-gene panel of hereditary cancer genes. Cancer 2017, 123, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.J.; Obeid, E.; Daly, M.B. Multigene Panels to Evaluate Hereditary Cancer Risk: Reckless or Relevant? J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 4186–4187. [Google Scholar] [CrossRef]
- Reid, B.M.; Permuth, J.B.; Sellers, T.A. Epidemiology of ovarian cancer: A review. Cancer Biol. Med. 2017, 14, 9–32. [Google Scholar] [CrossRef]
- La Vecchia, C. Ovarian cancer: Epidemiology and risk factors. Eur. J. Cancer Prev. Off. J. Eur. Cancer Prev. Organ. 2017, 26, 55–62. [Google Scholar] [CrossRef]
- Modan, B.; Hartge, P.; Hirsh-Yechezkel, G.; Chetrit, A.; Lubin, F.; Beller, U.; Ben-Baruch, G.; Fishman, A.; Menczer, J.; Struewing, J.P.; et al. Parity, oral contraceptives, and the risk of ovarian cancer among carriers and noncarriers of a BRCA1 or BRCA2 mutation. N. Engl. J. Med. 2001, 345, 235–240. [Google Scholar] [CrossRef]
- Goldgar, D.E.; Easton, D.F.; Cannon-Albright, L.A.; Skolnick, M.H. Systematic population-based assessment of cancer risk in first-degree relatives of cancer probands. J. Natl. Cancer Inst. 1994, 86, 1600–1608. [Google Scholar] [CrossRef] [PubMed]
- Ebell, M.H. Estimating the risk of ovarian cancer. Am. Fam. Physician 2009, 80, 632–634. [Google Scholar] [PubMed]
- Heintz, A.P.; Hacker, N.F.; Lagasse, L.D. Epidemiology and etiology of ovarian cancer: A review. Obstet. Gynecol. 1985, 66, 127–135. [Google Scholar]
- Daly, M.B.; Pal, T.; Berry, M.P.; Buys, S.S.; Dickson, P.; Domchek, S.M.; Elkhanany, A.; Friedman, S.; Goggins, M.; Hutton, M.L.; et al. Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2021, 19, 77–102. [Google Scholar] [CrossRef]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.A.; Mooij, T.M.; Roos-Blom, M.J.; Jervis, S.; van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed]
- Shanmughapriya, S.; Nachiappan, V.; Natarajaseenivasan, K. BRCA1 and BRCA2 mutations in the ovarian cancer population across race and ethnicity: Special reference to Asia. Oncology 2013, 84, 226–232. [Google Scholar] [CrossRef]
- Norquist, B.M.; Harrell, M.I.; Brady, M.F.; Walsh, T.; Lee, M.K.; Gulsuner, S.; Bernards, S.S.; Casadei, S.; Yi, Q.; Burger, R.A.; et al. Inherited Mutations in Women With Ovarian Carcinoma. JAMA Oncol. 2016, 2, 482–490. [Google Scholar] [CrossRef]
- Risch, H.A.; McLaughlin, J.R.; Cole, D.E.; Rosen, B.; Bradley, L.; Kwan, E.; Jack, E.; Vesprini, D.J.; Kuperstein, G.; Abrahamson, J.L.; et al. Prevalence and penetrance of germline BRCA1 and BRCA2 mutations in a population series of 649 women with ovarian cancer. Am. J. Hum. Genet. 2001, 68, 700–710. [Google Scholar] [CrossRef]
- Sarantaus, L.; Vahteristo, P.; Bloom, E.; Tamminen, A.; Unkila-Kallio, L.; Butzow, R.; Nevanlinna, H. BRCA1 and BRCA2 mutations among 233 unselected Finnish ovarian carcinoma patients. Eur. J. Hum. Genet. 2001, 9, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Malander, S.; Ridderheim, M.; Måsbäck, A.; Loman, N.; Kristoffersson, U.; Olsson, H.; Nilbert, M.; Borg, A. One in 10 ovarian cancer patients carry germ line BRCA1 or BRCA2 mutations: Results of a prospective study in Southern Sweden. Eur. J. Cancer 2004, 40, 422–428. [Google Scholar] [CrossRef]
- Soegaard, M.; Kjaer, S.K.; Cox, M.; Wozniak, E.; Høgdall, E.; Høgdall, C.; Blaakaer, J.; Jacobs, I.J.; Gayther, S.A.; Ramus, S.J. BRCA1 and BRCA2 mutation prevalence and clinical characteristics of a population-based series of ovarian cancer cases from Denmark. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2008, 14, 3761–3767. [Google Scholar] [CrossRef] [PubMed]
- Harter, P.; Hauke, J.; Heitz, F.; Reuss, A.; Kommoss, S.; Marmé, F.; Heimbach, A.; Prieske, K.; Richters, L.; Burges, A.; et al. Prevalence of deleterious germline variants in risk genes including BRCA1/2 in consecutive ovarian cancer patients (AGO-TR-1). PLoS ONE 2017, 12, e0186043. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, T.; Aoki, D.; Hattori, K.; Jinushi, M.; Kigawa, J.; Takeshima, N.; Tsuda, H.; Watanabe, Y.; Yoshihara, K.; Sugiyama, T. The first Japanese nationwide multicenter study of BRCA mutation testing in ovarian cancer: CHARacterizing the cross-sectionaL approach to Ovarian cancer geneTic TEsting of BRCA (CHARLOTTE). Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2019, 29, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Xie, R.; Zhi, Q.; Deng, Y.; Wu, Y.; Li, W.; Yang, L.; Jiao, Z.; Luo, J.; Zi, Y.; et al. BRCA germline mutations in an unselected nationwide cohort of Chinese patients with ovarian cancer and healthy controls. Gynecol. Oncol. 2018, 151, 145–152. [Google Scholar] [CrossRef]
- Ha, H.I.; Ryu, J.S.; Shim, H.; Kong, S.Y.; Lim, M.C. Reclassification of BRCA1 and BRCA2 variants found in ovarian epithelial, fallopian tube, and primary peritoneal cancers. J. Gynecol. Oncol. 2020, 31, e83. [Google Scholar] [CrossRef]
- Alsop, K.; Fereday, S.; Meldrum, C.; deFazio, A.; Emmanuel, C.; George, J.; Dobrovic, A.; Birrer, M.J.; Webb, P.M.; Stewart, C.; et al. BRCA mutation frequency and patterns of treatment response in BRCA mutation-positive women with ovarian cancer: A report from the Australian Ovarian Cancer Study Group. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2012, 30, 2654–2663. [Google Scholar] [CrossRef]
- Rafnar, T.; Benediktsdottir, K.R.; Eldon, B.J.; Gestsson, T.; Saemundsson, H.; Olafsson, K.; Salvarsdottir, A.; Steingrimsson, E.; Thorlacius, S. BRCA2, but not BRCA1, mutations account for familial ovarian cancer in Iceland: A population-based study. Eur. J. Cancer 2004, 40, 2788–2793. [Google Scholar] [CrossRef]
- Majdak, E.J.; De Bock, G.H.; Brozek, I.; Perkowska, M.; Ochman, K.; Debniak, J.; Milczek, T.; Cornelisse, C.J.; Jassem, J.; Emerich, J.; et al. Prevalence and clinical correlations of BRCA1/BRCA2 unclassified variant carriers among unselected primary ovarian cancer cases-preliminary report. Eur. J. Cancer 2005, 41, 143–150. [Google Scholar] [CrossRef]
- Gupta, S.; Rajappa, S.; Advani, S.; Agarwal, A.; Aggarwal, S.; Goswami, C.; Palanki, S.D.; Arya, D.; Patil, S.; Kodagali, R. Prevalence of BRCA1 and BRCA2 Mutations Among Patients With Ovarian, Primary Peritoneal, and Fallopian Tube Cancer in India: A Multicenter Cross-Sectional Study. JCO Glob. Oncol. 2021, 7, 849–861. [Google Scholar] [CrossRef] [PubMed]
- Yazici, H.; Glendon, G.; Yazici, H.; Burnie, S.J.; Saip, P.; Buyru, F.; Bengisu, E.; Andrulis, I.L.; Dalay, N.; Ozcelik, H. BRCA1 and BRCA2 mutations in Turkish familial and non-familial ovarian cancer patients: A high incidence of mutations in non-familial cases. Hum. Mutat. 2002, 20, 28–34. [Google Scholar] [CrossRef]
- Liede, A.; Malik, I.A.; Aziz, Z.; Rios Pd Pde, L.; Kwan, E.; Narod, S.A. Contribution of BRCA1 and BRCA2 mutations to breast and ovarian cancer in Pakistan. Am. J. Hum. Genet. 2002, 71, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, A.O.; Llacuachaqui, M.; Pardo, G.G.; Royer, R.; Larson, G.; Weitzel, J.N.; Narod, S.A. BRCA1 and BRCA2 mutations among ovarian cancer patients from Colombia. Gynecol. Oncol. 2012, 124, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Kamura, T.; Kigawa, J.; Terakawa, N.; Kikuchi, Y.; Kita, T.; Suzuki, M.; Sato, I.; Taguchi, K. Clinical characteristics of clear cell carcinoma of the ovary: A distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer 2000, 88, 2584–2589. [Google Scholar] [CrossRef]
- Konstantinopoulos, P.A.; Brady, W.E.; Farley, J.; Armstrong, A.; Uyar, D.S.; Gershenson, D.M. Phase II study of single-agent cabozantinib in patients with recurrent clear cell ovarian, primary peritoneal or fallopian tube cancer (NRG-GY001). Gynecol. Oncol. 2018, 150, 9–13. [Google Scholar] [CrossRef]
- Yen, C.F.; Kim, M.R.; Lee, C.L. Epidemiologic Factors Associated with Endometriosis in East Asia. Gynecol. Minim. Invasive Ther. 2019, 8, 4–11. [Google Scholar] [CrossRef]
- Zhu, C.; Zhu, J.; Qian, L.; Liu, H.; Shen, Z.; Wu, D.; Zhao, W.; Xiao, W.; Zhou, Y. Clinical characteristics and prognosis of ovarian clear cell carcinoma: A 10-year retrospective study. BMC Cancer 2021, 21, 322. [Google Scholar] [CrossRef]
- Gerlinger, C.; Faustmann, T.; Hassall, J.J.; Seitz, C. Treatment of endometriosis in different ethnic populations: A meta-analysis of two clinical trials. BMC Women Health 2012, 12, 9. [Google Scholar] [CrossRef]
- Rebbeck, T.R.; Mitra, N.; Wan, F.; Sinilnikova, O.M.; Healey, S.; McGuffog, L.; Mazoyer, S.; Chenevix-Trench, G.; Easton, D.F.; Antoniou, A.C.; et al. Association of type and location of BRCA1 and BRCA2 mutations with risk of breast and ovarian cancer. JAMA 2015, 313, 1347–1361. [Google Scholar] [CrossRef]
- Gayther, S.A.; Warren, W.; Mazoyer, S.; Russell, P.A.; Harrington, P.A.; Chiano, M.; Seal, S.; Hamoudi, R.; van Rensburg, E.J.; Dunning, A.M.; et al. Germline mutations of the BRCA1 gene in breast and ovarian cancer families provide evidence for a genotype-phenotype correlation. Nat. Genet. 1995, 11, 428–433. [Google Scholar] [CrossRef]
- Thompson, D.; Easton, D. Variation in BRCA1 cancer risks by mutation position. Cancer Epidemiol. Biomark. Prev. 2002, 11, 329–336. [Google Scholar]
- Gayther, S.A.; Mangion, J.; Russell, P.; Seal, S.; Barfoot, R.; Ponder, B.A.; Stratton, M.R.; Easton, D. Variation of risks of breast and ovarian cancer associated with different germline mutations of the BRCA2 gene. Nat. Genet. 1997, 15, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Solsky, I.; Chen, J.; Rebbeck, T.R. Precision prophylaxis: Identifying the optimal timing for risk-reducing salpingo-oophorectomy based on type of BRCA1 and BRCA2 cluster region mutations. Gynecol. Oncol. 2020, 156, 363–376. [Google Scholar] [CrossRef]
- Breast Cancer Information Core. Available online: https://research.nhgri.nih.gov/projects/bic/Member/index.shtml (accessed on 18 June 2021).
- Yoshihara, K.; Enomoto, T.; Aoki, D.; Watanabe, Y.; Kigawa, J.; Takeshima, N.; Inomata, H.; Hattori, K.; Jinushi, M.; Tsuda, H.; et al. Association of gBRCA1/2 mutation locations with ovarian cancer risk in Japanese patients from the CHARLOTTE study. Cancer Sci. 2020, 111, 3350–3358. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, F.C.; Goncalves, S.; Mele, P.G.; Liria, N.C.; Sganga, L.; Diaz Perez, I.; Podesta, E.J.; Solano, A.R. BRCA1 and BRCA2 mutations and clinical interpretation in 398 ovarian cancer patients: Comparison with breast cancer variants in a similar population. Hum. Genom. 2018, 12, 39. [Google Scholar] [CrossRef]
- Teixeira, N.; Mourits, M.J.; Vos, J.R.; Kolk, D.M.; Jansen, L.; Oosterwijk, J.C.; Bock, G.H. Ovarian cancer in BRCA1/2 mutation carriers: The impact of mutation position and family history on the cancer risk. Maturitas 2015, 82, 197–202. [Google Scholar] [CrossRef]
- Felix, G.E.S.; Zheng, Y.; Olopade, O.I. Mutations in context: Implications of BRCA testing in diverse populations. Fam. Cancer 2018, 17, 471–483. [Google Scholar] [CrossRef]
- Satagopan, J.M.; Boyd, J.; Kauff, N.D.; Robson, M.; Scheuer, L.; Narod, S.; Offit, K. Ovarian cancer risk in Ashkenazi Jewish carriers of BRCA1 and BRCA2 mutations. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2002, 8, 3776–3781. [Google Scholar]
- Menkiszak, J.; Chudecka-Głaz, A.; Gronwald, J.; Cymbaluk-Płoska, A.; Celewicz, A.; Świniarska, M.; Wężowska, M.; Bedner, R.; Zielińska, D.; Tarnowska, P.; et al. Prophylactic salpingo-oophorectomy in BRCA1 mutation carriers and postoperative incidence of peritoneal and breast cancers. J. Ovarian Res. 2016, 9, 11. [Google Scholar] [CrossRef]
- Lubinski, J.; Phelan, C.M.; Ghadirian, P.; Lynch, H.T.; Garber, J.; Weber, B.; Tung, N.; Horsman, D.; Isaacs, C.; Monteiro, A.N.; et al. Cancer variation associated with the position of the mutation in the BRCA2 gene. Fam. Cancer 2004, 3, 1–10. [Google Scholar] [CrossRef]
- Antoniou, A.C.; Pharoah, P.D.; Narod, S.; Risch, H.A.; Eyfjord, J.E.; Hopper, J.L.; Olsson, H.; Johannsson, O.; Borg, A.; Pasini, B.; et al. Breast and ovarian cancer risks to carriers of the BRCA1 5382insC and 185delAG and BRCA2 6174delT mutations: A combined analysis of 22 population based studies. J. Med. Genet. 2005, 42, 602–603. [Google Scholar] [CrossRef]
- Meeks, H.D.; Song, H.; Michailidou, K.; Bolla, M.K.; Dennis, J.; Wang, Q.; Barrowdale, D.; Frost, D.; McGuffog, L.; Ellis, S.; et al. BRCA2 Polymorphic Stop Codon K3326X and the Risk of Breast, Prostate, and Ovarian Cancers. J. Natl. Cancer Inst. 2016, 108. [Google Scholar] [CrossRef]
- Yoshida, R.; Watanabe, C.; Yokoyama, S.; Inuzuka, M.; Yotsumoto, J.; Arai, M.; Nakamura, S. Analysis of clinical characteristics of breast cancer patients with the Japanese founder mutation BRCA1 L63X. Oncotarget 2019, 10, 3276–3284. [Google Scholar] [CrossRef]
- Sekine, M.; Nishino, K.; Enomoto, T. BRCA Genetic Test and Risk-Reducing Salpingo-Oophorectomy for Hereditary Breast and Ovarian Cancer: State-of-the-Art. Cancers 2021, 13, 2562. [Google Scholar] [CrossRef]
- Sekine, M.; Enomoto, T. Precision medicine for hereditary tumors in gynecologic malignancies. J. Obstet. Gynaecol. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- van den Broek, A.J.; van ’t Veer, L.J.; Hooning, M.J.; Cornelissen, S.; Broeks, A.; Rutgers, E.J.; Smit, V.T.; Cornelisse, C.J.; van Beek, M.; Janssen-Heijnen, M.L.; et al. Impact of Age at Primary Breast Cancer on Contralateral Breast Cancer Risk in BRCA1/2 Mutation Carriers. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2016, 34, 409–418. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Metcalfe, K.; Gershman, S.; Lynch, H.T.; Ghadirian, P.; Tung, N.; Kim-Sing, C.; Olopade, O.I.; Domchek, S.; McLennan, J.; Eisen, A.; et al. Predictors of contralateral breast cancer in BRCA1 and BRCA2 mutation carriers. Br. J. Cancer 2011, 104, 1384–1392. [Google Scholar] [CrossRef] [PubMed]
- Mavaddat, N.; Peock, S.; Frost, D.; Ellis, S.; Platte, R.; Fineberg, E.; Evans, D.G.; Izatt, L.; Eeles, R.A.; Adlard, J.; et al. Cancer risks for BRCA1 and BRCA2 mutation carriers: Results from prospective analysis of EMBRACE. J. Natl. Cancer Inst. 2013, 105, 812–822. [Google Scholar] [CrossRef] [PubMed]
- Petrucelli, N.; Daly, M.B.; Pal, T. BRCA1- and BRCA2-Associated Hereditary Breast and Ovarian Cancer. In GeneReviews(®); Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Mirzaa, G., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993; Copyright © 1993-2021, University of Washington, Seattle. GeneReviews is a registered trademark of the University of Washington, Seattle. All rights reserved.: Seattle (WA), 1993. [Google Scholar]
- Tai, Y.C.; Domchek, S.; Parmigiani, G.; Chen, S. Breast cancer risk among male BRCA1 and BRCA2 mutation carriers. J. Natl. Cancer Inst. 2007, 99, 1811–1814. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.G.; Susnerwala, I.; Dawson, J.; Woodward, E.; Maher, E.R.; Lalloo, F. Risk of breast cancer in male BRCA2 carriers. J. Med. Genet. 2010, 47, 710–711. [Google Scholar] [CrossRef]
- Struewing, J.P.; Coriaty, Z.M.; Ron, E.; Livoff, A.; Konichezky, M.; Cohen, P.; Resnick, M.B.; Lifzchiz-Mercerl, B.; Lew, S.; Iscovich, J. Founder BRCA1/2 mutations among male patients with breast cancer in Israel. Am. J. Hum. Genet. 1999, 65, 1800–1802. [Google Scholar] [CrossRef]
- Leongamornlert, D.; Mahmud, N.; Tymrakiewicz, M.; Saunders, E.; Dadaev, T.; Castro, E.; Goh, C.; Govindasami, K.; Guy, M.; O’Brien, L.; et al. Germline BRCA1 mutations increase prostate cancer risk. Br. J. Cancer 2012, 106, 1697–1701. [Google Scholar] [CrossRef]
- Kote-Jarai, Z.; Leongamornlert, D.; Saunders, E.; Tymrakiewicz, M.; Castro, E.; Mahmud, N.; Guy, M.; Edwards, S.; O’Brien, L.; Sawyer, E.; et al. BRCA2 is a moderate penetrance gene contributing to young-onset prostate cancer: Implications for genetic testing in prostate cancer patients. Br. J. Cancer 2011, 105, 1230–1234. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.; Alkhushaym, N.; Fallatah, S.; Althagafi, A.; Aljadeed, R.; Alsowaida, Y.; Jeter, J.; Martin, J.R.; Babiker, H.M.; McBride, A.; et al. The association of BRCA1 and BRCA2 mutations with prostate cancer risk, frequency, and mortality: A meta-analysis. Prostate 2019, 79, 880–895. [Google Scholar] [CrossRef]
- Gallagher, D.J.; Gaudet, M.M.; Pal, P.; Kirchhoff, T.; Balistreri, L.; Vora, K.; Bhatia, J.; Stadler, Z.; Fine, S.W.; Reuter, V.; et al. Germline BRCA mutations denote a clinicopathologic subset of prostate cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2010, 16, 2115–2121. [Google Scholar] [CrossRef] [PubMed]
- Thorne, H.; Willems, A.J.; Niedermayr, E.; Hoh, I.M.; Li, J.; Clouston, D.; Mitchell, G.; Fox, S.; Hopper, J.L.; Bolton, D. Decreased prostate cancer-specific survival of men with BRCA2 mutations from multiple breast cancer families. Cancer Prev. Res. 2011, 4, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Leão, R.R.N.; Price, A.J.; James Hamilton, R. Germline BRCA mutation in male carriers-ripe for precision oncology? Prostate Cancer Prostatic Dis. 2018, 21, 48–56. [Google Scholar] [CrossRef]
- Agalliu, I.; Gern, R.; Leanza, S.; Burk, R.D. Associations of high-grade prostate cancer with BRCA1 and BRCA2 founder mutations. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2009, 15, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.; Easton, D.F. Cancer Incidence in BRCA1 mutation carriers. J. Natl. Cancer Inst. 2002, 94, 1358–1365. [Google Scholar] [CrossRef]
- Risch, H.A.; McLaughlin, J.R.; Cole, D.E.; Rosen, B.; Bradley, L.; Fan, I.; Tang, J.; Li, S.; Zhang, S.; Shaw, P.A.; et al. Population BRCA1 and BRCA2 mutation frequencies and cancer penetrances: A kin-cohort study in Ontario, Canada. J. Natl. Cancer Inst. 2006, 98, 1694–1706. [Google Scholar] [CrossRef] [PubMed]
- Mocci, E.; Milne, R.L.; Méndez-Villamil, E.Y.; Hopper, J.L.; John, E.M.; Andrulis, I.L.; Chung, W.K.; Daly, M.; Buys, S.S.; Malats, N.; et al. Risk of pancreatic cancer in breast cancer families from the breast cancer family registry. Cancer Epidemiol. Biomark. Prev. 2013, 22, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Ragone, A.; Lubinski, J.; Lynch, H.T.; Moller, P.; Ghadirian, P.; Foulkes, W.D.; Armel, S.; Eisen, A.; Neuhausen, S.L.; et al. The incidence of pancreatic cancer in BRCA1 and BRCA2 mutation carriers. Br. J. Cancer 2012, 107, 2005–2009. [Google Scholar] [CrossRef] [PubMed]
- Rosen, M.N.; Goodwin, R.A.; Vickers, M.M. BRCA mutated pancreatic cancer: A change is coming. World J. Gastroenterol. 2021, 27, 1943–1958. [Google Scholar] [CrossRef] [PubMed]
- Ferrone, C.R.; Levine, D.A.; Tang, L.H.; Allen, P.J.; Jarnagin, W.; Brennan, M.F.; Offit, K.; Robson, M.E. BRCA germline mutations in Jewish patients with pancreatic adenocarcinoma. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2009, 27, 433–438. [Google Scholar] [CrossRef]
- Hearle, N.; Damato, B.E.; Humphreys, J.; Wixey, J.; Green, H.; Stone, J.; Easton, D.F.; Houlston, R.S. Contribution of germline mutations in BRCA2, P16(INK4A), P14(ARF) and P15 to uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2003, 44, 458–462. [Google Scholar] [CrossRef]
- Moran, A.; O’Hara, C.; Khan, S.; Shack, L.; Woodward, E.; Maher, E.R.; Lalloo, F.; Evans, D.G. Risk of cancer other than breast or ovarian in individuals with BRCA1 and BRCA2 mutations. Fam. Cancer 2012, 11, 235–242. [Google Scholar] [CrossRef]
- Goshen, R.; Chu, W.; Elit, L.; Pal, T.; Hakimi, J.; Ackerman, I.; Fyles, A.; Mitchell, M.; Narod, S.A. Is uterine papillary serous adenocarcinoma a manifestation of the hereditary breast-ovarian cancer syndrome? Gynecol. Oncol. 2000, 79, 477–481. [Google Scholar] [CrossRef]
- Beiner, M.E.; Finch, A.; Rosen, B.; Lubinski, J.; Moller, P.; Ghadirian, P.; Lynch, H.T.; Friedman, E.; Sun, P.; Narod, S.A. The risk of endometrial cancer in women with BRCA1 and BRCA2 mutations. A prospective study. Gynecol. Oncol. 2007, 104, 7–10. [Google Scholar] [CrossRef]
- Shu, C.A.; Pike, M.C.; Jotwani, A.R.; Friebel, T.M.; Soslow, R.A.; Levine, D.A.; Nathanson, K.L.; Konner, J.A.; Arnold, A.G.; Bogomolniy, F.; et al. Uterine Cancer After Risk-Reducing Salpingo-oophorectomy Without Hysterectomy in Women With BRCA Mutations. JAMA Oncol. 2016, 2, 1434–1440. [Google Scholar] [CrossRef]
- de Jonge, M.M.; de Kroon, C.D.; Jenner, D.J.; Oosting, J.; de Hullu, J.A.; Mourits, M.J.E.; Garcia, E.B.G.; Ausems, M.; Collée, J.M.; van Engelen, K.; et al. Endometrial Cancer Risk in Women with Germline BRCA1 or BRCA2 Mutations: Multicenter Cohort Study. J. Natl. Cancer Inst. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kauff, N.D.; Domchek, S.M.; Friebel, T.M.; Robson, M.E.; Lee, J.; Garber, J.E.; Isaacs, C.; Evans, D.G.; Lynch, H.; Eeles, R.A.; et al. Risk-reducing salpingo-oophorectomy for the prevention of BRCA1- and BRCA2-associated breast and gynecologic cancer: A multicenter, prospective study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008, 26, 1331–1337. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.L.; Wang, K.; Liu, Q.; Li, J.; Zhang, X.; Li, H.Y. Risk Reduction and Survival Benefit of Risk-Reducing Salpingo-oophorectomy in Hereditary Breast Cancer: Meta-analysis and Systematic Review. Clin. Breast Cancer 2019, 19, e48–e65. [Google Scholar] [CrossRef] [PubMed]
| Country/Population | No. of Cases | BRCA1 | BRCA2 | Ratio |
|---|---|---|---|---|
| BRCA1:BRCA2 | ||||
| Ashkenazi Jews [22] | 840 | 182 (21.7%) | 64 (7.6%) | 2.8:1 |
| USA [29] | 1915 | 182 (8.5%) | 98 (6.3%) | 1.9:1 |
| Canada [30] | 977 | 75 (7.7%) | 54 (5.5%) | 1.4:1 |
| Finland [31] | 233 | 11 (4.7%) | 2 (0.9%) | 5.5:1 |
| Sweden [32] | 161 | 12 (7.5%) | 1 (0.6%) | 12:1 |
| Denmark [33] | 445 | 22 (4.9%) | 4 (0.9%) | 5.5:1 |
| Iceland [39] | 179 | 2 (1.1%) | 10 (5.6%) | 0.5:1 |
| Poland [40] | 309 | 23 (7.4%) | 29 (9.4%) | 0.8:1 |
| Germany [34] | 523 | 81 (15.5%) | 28 (5.4%) | 2.9:1 |
| India [41] | 239 | 37 (15.5%) | 14 (5.9%) | 2.6:1 |
| Turkey [42] | 102 | 10 (9.8%) | 7 (6.9%) | 1.4:1 |
| Pakistan [43] | 120 | 16 (13.3%) | 3 (2.5%) | 5.3:1 |
| Colombia [44] | 100 | 13 (13.0%) | 2 (0.2%) | 6.5:1 |
| Australia [38] | 809 | 70 (8.7%) | 39 (4.8%) | 1.8:1 |
| Japan [35] | 634 | 63 (9.9%) | 30 (4.7%) | 2.1:1 |
| China [36] | 1331 | 228 (17.1%) | 70 (5.3%) | 3.3:1 |
| Korea [37] | 805 | 106 (13.2%) | 51 (6.3%) | 2.1:1 |
| Histological Classification | USA [29] (n = 1699) | Australia [38] (n = 891) | Germany [34] (n = 462) | Japan [35] (n = 609) | China [36] (n = 1044) | Korea [37] (n = 591) |
|---|---|---|---|---|---|---|
| High-grade serous | 16.0% (240/1498) | 16.6% * (118/709) | 23.2% (94/406) | 28.5% (78/274) | 27.2% * (229/843) | 22.3% (95/426) |
| Low-grade serous | 5.7% (4/70) | N/A * | 5.6% (1/18) | 20.0% (1/5) | N/A * | 19.4% (6/31) |
| Endometrioid | 10.9% (7/64) | 8.4% (10/119) | 13.0% (3/23) | 6.7% (8/120) | 10.8% (7/65) | 13.0% (7/54) |
| Clear cell | 6.9% (4/58) | 6.3% (4/63) | 0.0% (0/6) | 2.1% (4/187) | 7.6% (6/79) | 7.3% (4/55) |
| Mucinous | 0.0% (0/9) | N/A | 0.0% (0/9) | 0.0% (0/19) | 7.0% (4/57) | 5.6% (1/18) |
| Seromucinous | N/A | N/A | N/A | 0.0% (0/4) | N/A | 0.0% (0/7) |
| BIC Designation | Number of Entries | Exon | HGVS cDNA | HGVS Protein | Mutation Type | Population |
|---|---|---|---|---|---|---|
| BRCA1 | ||||||
| 185delAG | 2038 | 2 | c.66_67delAG | p.Glu23ValfsTer17 | Frameshift | Ashkenazi Jewish |
| 5382insC | 1093 | 20 | c.5263_5264insC | p.Gln1756ProfsTer74 | Frameshift | Ashkenazi Jewish |
| C61G | 239 | 5 | c.181T>G | p.Cys61Gly | Missense | Europe |
| 4184del4 | 144 | 11 | c.4065_4068delTCAA | p.Asn1355LysfsTer10 | Frameshift | Asia |
| R1443X | 136 | 13 | c.4327C>T | p.Arg1443Ter | Nonsense | Europe |
| 3875del4 | 124 | 11 | c.3756_3759delGTCT | p.Ser1253ArgfsTer10 | Frameshift | Europe |
| exon13ins6kb | 111 | 13 | N/A | N/A | Frameshift | N/A |
| E1250X | 98 | 11 | c.3748G>T | p.Glu1250Ter | Nonsense | Europe/Americas |
| Q563X | 94 | 11 | c.1687C>T | p.Gln563Ter | Nonsense | N/A |
| 2800delAA | 81 | 11 | c.2681_2682delAA | p.Lys894ThrfsTer8 | Frameshift | Europe |
| BRCA2 | ||||||
| 6174delT | 1093 | 11 | c.5946_5946delT | p.Ser1982ArgfsTer22 | Frameshift | Ashkenazi Jewish |
| K3326X | 301 | 27 | c.9976A>T | p.Lys3326Ter | Nonsense | N/A |
| 3036del4 | 111 | 11 | c.2808_2811delACAA | p.Ala938ProfsTer21 | Frameshift | Americas |
| 6503delTT | 95 | 11 | c.6275_6276delTT | p.Leu2092ProfsTer7 | Frameshift | Americas/Europe |
| 8765delAG | 76 | 20 | c.8537_8538delAG | p.Glu2846GlyfsTer22 | Frameshift | Americas/Europe |
| 2041insA | 75 | 10 | c.1813_1814insA | p.Asp605GlufsTer2 | Frameshift | Europe |
| 4075delGT | 64 | 11 | c.3847_3848delGT | p.Val1283LysfsTer2 | Frameshift | N/A |
| Y1894X | 62 | 11 | c.5682C>G | p.Tyr1894Ter | Nonsense | N/A |
| 983del4 | 61 | 9 | c.755_758delACAG | p.Asp252ValfsTer24 | Frameshift | N/A |
| R3128X | 50 | 25 | c.9382C>T | p.Arg3128Ter | Nonsense | Europe |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sekine, M.; Nishino, K.; Enomoto, T. Differences in Ovarian and Other Cancers Risks by Population and BRCA Mutation Location. Genes 2021, 12, 1050. https://doi.org/10.3390/genes12071050
Sekine M, Nishino K, Enomoto T. Differences in Ovarian and Other Cancers Risks by Population and BRCA Mutation Location. Genes. 2021; 12(7):1050. https://doi.org/10.3390/genes12071050
Chicago/Turabian StyleSekine, Masayuki, Koji Nishino, and Takayuki Enomoto. 2021. "Differences in Ovarian and Other Cancers Risks by Population and BRCA Mutation Location" Genes 12, no. 7: 1050. https://doi.org/10.3390/genes12071050
APA StyleSekine, M., Nishino, K., & Enomoto, T. (2021). Differences in Ovarian and Other Cancers Risks by Population and BRCA Mutation Location. Genes, 12(7), 1050. https://doi.org/10.3390/genes12071050







