Primary Chicken and Duck Endothelial Cells Display a Differential Response to Infection with Highly Pathogenic Avian Influenza Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Influenza Virus
2.3. Viral Infection
2.4. FITC-Labelled Acetylated Low-Density Lipoprotein (FITC-AcLDL) Uptake
2.5. RNA Extraction, cDNA Synthesis and Reverse-Transcriptase Polymerase Chain Reaction (RT-PCR)
2.6. Flow Cytometry for Chicken CD45
2.7. Tube Formation Assay
2.8. RNA Seq
2.9. Immunofluorescence
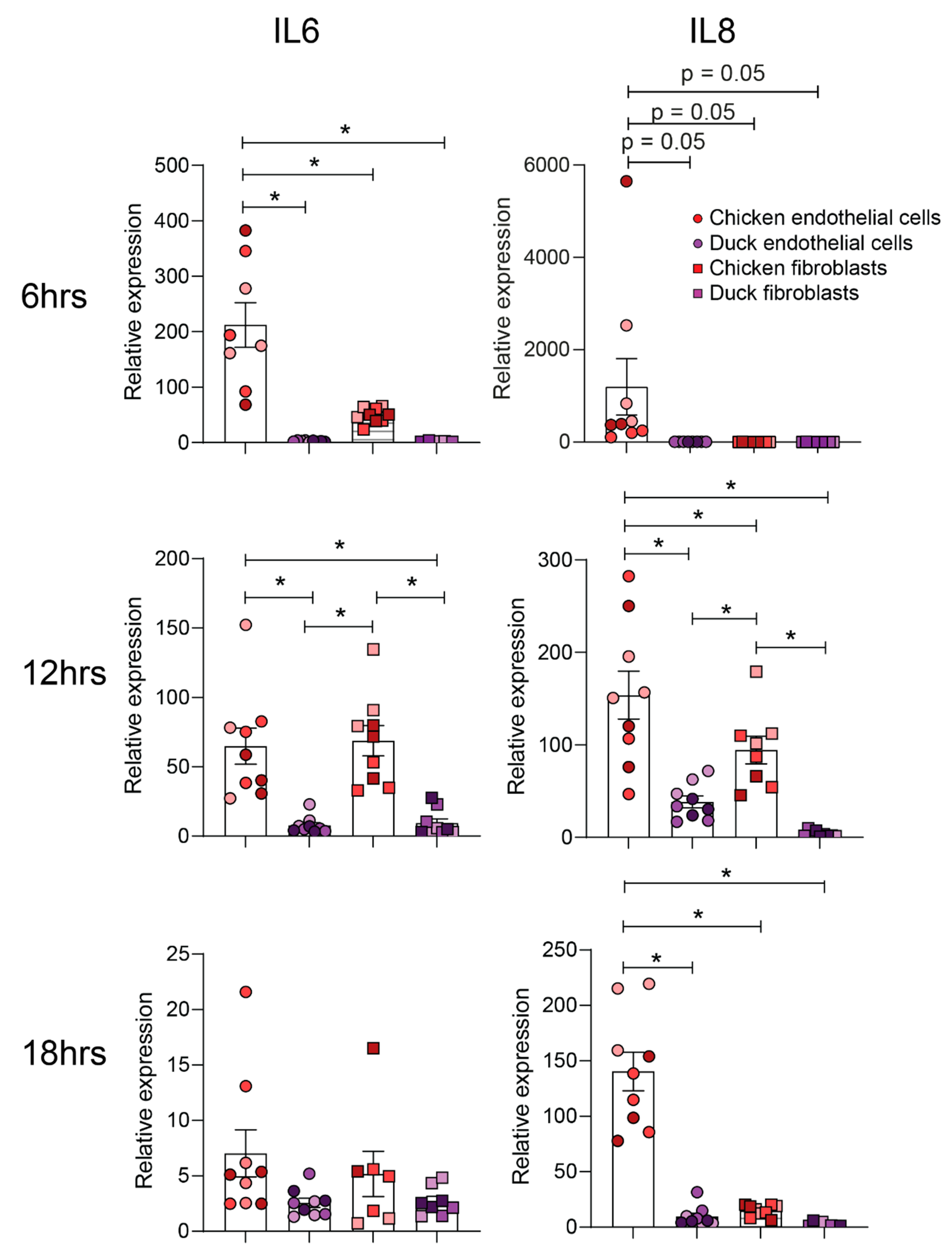
2.10. PolyI:C Stimulation and Real-Time Quantitative PCR (RT-qPCR)
2.11. Statistical Analysis
3. Results
3.1. Chicken and Duck Endothelial Cells Can Be Cultured In Vitro
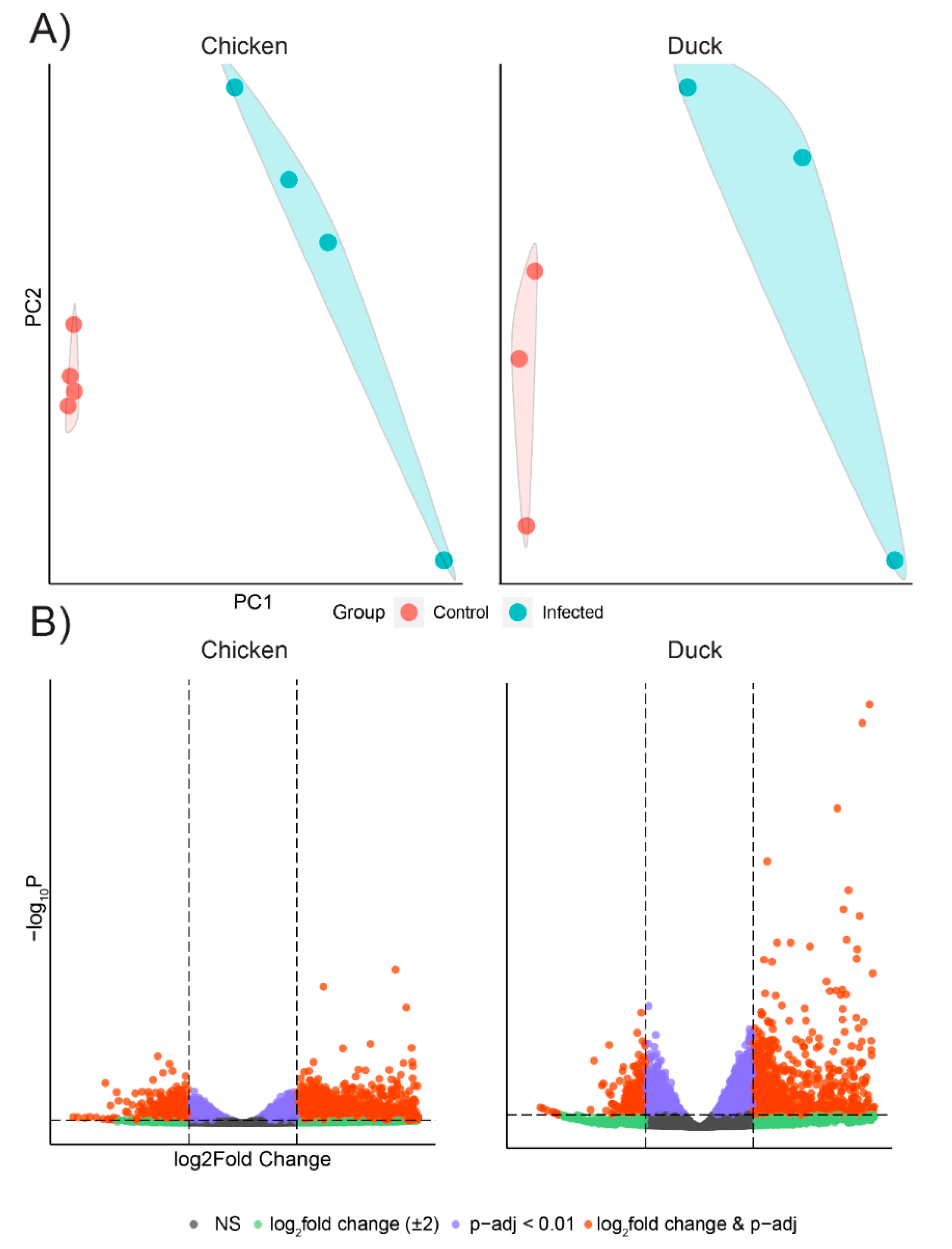
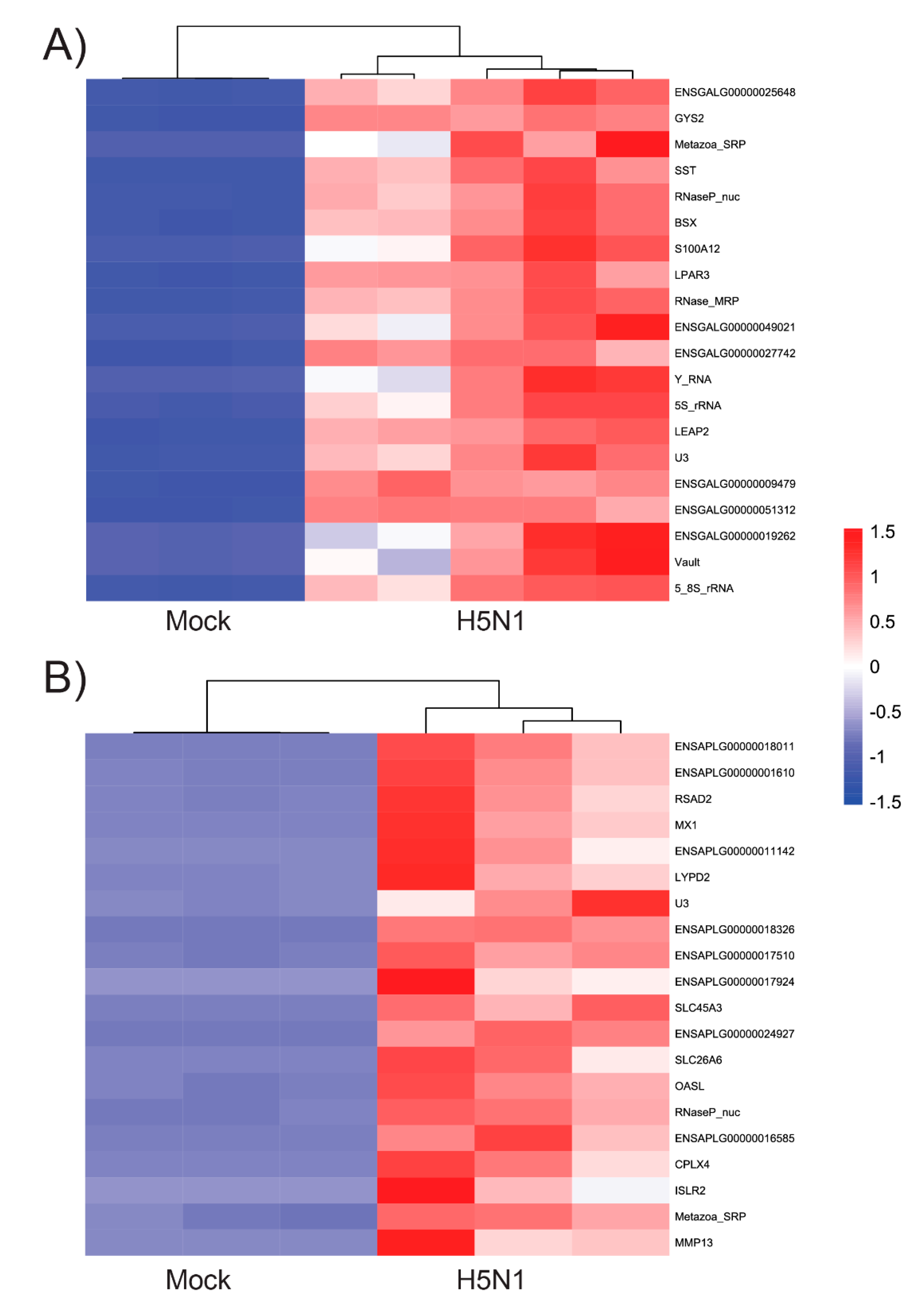
3.2. Chicken and Duck Endothelial Cells Have a Different Transcriptional Response to HPAIV Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Short, K.; Kroeze, E.J.V.; Reperant, L.A.; Richard, M.; Kuiken, T. Influenza virus and endothelial cells: A species specific relationship. Front. Microbiol. 2014, 5, 653. [Google Scholar] [CrossRef]
- Short, K.R.; Richard, M.; Verhagen, J.H.; van Riel, D.; Schrauwen, E.J.; van den Brand, J.M.; Mänz, B.; Bodewes, R.; Herfst, S. One health, multiple challenges: The interspecies transmission of influenza A virus. One Health 2015, 1, 1–13. [Google Scholar] [CrossRef]
- Karawita, A.C.; Tong, M.Z.; Short, K.R. A delicate balancing act: Immunity and immunopathology in human H7N9 influenza virus infections. Curr. Opin. Infect. Dis. 2019, 32, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Schat, K.A.; Bingham, J.; Butler, J.M.; Chen, L.-M.; Lowther, S.; Crowley, T.M.; Moore, R.J.; Donis, R.O.; Lowenthal, J.W. Role of Position 627 of PB2 and the Multi-basic Cleavage Site of the Hemagglutinin in the Virulence of H5N1 Avian Influenza Virus in Chickens and Ducks. PLoS ONE 2012, 7, e30960. [Google Scholar] [CrossRef] [PubMed]
- Karpala, A.J.; Bingham, J.; Schat, K.A.; Chen, L.M.; Donis, R.O.; Lowenthal, J.W.; Bean, A.D.G. Highly pathogenic (H5N1) avian influenza induces an inflammatory T helper type 1 cytokine response in the chicken. J. Interf. Cytokine Res. 2011, 31, 393–400. [Google Scholar] [CrossRef]
- Brown, J.D.; Stallknecht, D.E.; Beck, J.R.; Suarez, D.L.; Swayne, D.E. Susceptibility of North American ducks and gulls to H5N1 highly pathogenic avian influenza viruses. Emerg. Infect. Dis. 2006, 12, 1663. [Google Scholar] [CrossRef] [PubMed]
- Lion, A.; Richard, M.; Esnault, E.; Kut, E.; Soubieux, D.; Guillory, V.; Germond, M.; Blondeau, C.; Guabiraba, R.; Short, K.; et al. Productive replication of avian influenza viruses in chicken endothelial cells is determined by hemagglutinin cleavability and is related to innate immune escape. Virology 2018, 513, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L.; Choi, G.; Kuiken, T.; Quéré, P.; Trapp, S.; Short, K.R.; Richard, M. The culture of primary duck endothelial cells for the study of avian influenza. BMC Microbiol. 2018, 18, 138. [Google Scholar] [CrossRef]
- Docherty, D.E.; Slota, P.G. Use of muscovy duck embryo fibroblasts for the isolation of viruses from wild birds. J. Tissue Cult. Methods 1988, 11, 165–170. [Google Scholar] [CrossRef]
- Hernandez, R.; Brown, D.T. Growth and maintenance of chick embryo fibroblasts (CEF). Curr. Protoc. Microbiol. 2010, 17, A.4I.1–A.4I.8. [Google Scholar] [CrossRef]
- Short, K.R.; Diavatopoulos, D.A.; Reading, P.C.; Brown, L.E.; Rogers, K.L.; Strugnell, R.A.; Wijburg, O.L. Using Bioluminescent Imaging to Investigate Synergism between Streptococcus pneumoniae and Influenza A Virus in Infant Mice. J. Vis. Exp. 2011, e2357. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data; Babraham Bioinformatics, Babraham Institute: Cambridge, UK, 2010. [Google Scholar]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq: A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 1–21. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S.; Furumichi, M.; Tanabe, M.; Hirakawa, M. KEGG for representation and analysis of molecular networks involving diseases and drugs. Nucleic Acids Res. 2010, 38, D355–D360. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Friedman, M.S.; Shedden, K.; Hankenson, K.D.; Woolf, P.J. GAGE: Generally applicable gene set enrichment for pathway analysis. BMC Bioinform. 2009, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Brouwer, C. Pathview: An R/Bioconductor package for pathway-based data integration and visualization. Bioinformatics 2013, 29, 1830–1831. [Google Scholar] [CrossRef] [PubMed]
- Bingham, J.; Green, D.J.; Lowther, S.; Klippel, J.; Burggraaf, S.; Anderson, D.E.; Wibawa, H.; Hoa, D.M.; Long, N.T.; Vu, P.P.; et al. Infection studies with two highly pathogenic avian influenza strains (Vietnamese and Indonesian) in Pekin ducks (Anas platyrhynchos), with particular reference to clinical disease, tissue tropism and viral shedding. Avian Pathol. 2009, 38, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Burggraaf, S.; Karpala, A.J.; Bingham, J.; Lowther, S.; Selleck, P.; Kimpton, W.; Bean, A.D.G. H5N1 infection causes rapid mortality and high cytokine levels in chickens compared to ducks. Virus Res. 2014, 185, 23–31. [Google Scholar] [CrossRef]
- Kuchipudi, S.V.; Tellabati, M.; Sebastian, S.; Londt, B.Z.; Jansen, C.; Vervelde, L.; Brookes, S.M.; Brown, I.H.; Dunham, S.P.; Chang, K.-C. Highly pathogenic avian influenza virus infection in chickens but not ducks is associated with elevated host immune and pro-inflammatory responses. Vet. Res. 2014, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, J.B.; Vervelde, L.; Post, J.; Rebel, J.M.J. Differences in highly pathogenic avian influenza viral pathogenesis and associated early inflammatory response in chickens and ducks. Avian Pathol. 2013, 42, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Teijaro, J.R.; Walsh, K.B.; Cahalan, S.; Fremgen, D.M.; Roberts, E.; Scott, F.; Martinborough, E.; Peach, R.; Oldstone, M.B.A.; Rosen, H. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell 2011, 146, 980–991. [Google Scholar] [CrossRef] [PubMed]
| Upregulated in Duck (Total Number of Genes) | Upregulated in Chicken (Total Number of Genes) | Downregulated in Duck (Total Number of Genes) | Downregulated in Chicken (Total Number of Genes) | |
|---|---|---|---|---|
| Chemokines | CCL21, CCL4, CCL41, CCL4L2, CCL5, CCL28, CCR8, CCR9 (8) | CCL19, CCL21, CCL4, CCL41, CCL42, CCL17, CCL5, CCR8, CCR9, CCR4, CCR5 (11) | CCR11, CCR5, CXCL13, CXCR7 (4) | CCR7 (1) |
| The class I helical cytokines | IL2RB, IL2RG, IL7R, IL21R, IL5RA, CSF2RA, GHR, PRLR, TPO, MPL, CSF3, IL6ST, IL12RB2, IL23R, IL13RA, LIFR, (16) | IL2RA, IL2RB, IL2RG, IL9R, IL21R, CSF2RB, CSF2RA, IL13RA2, PRL, PRLR, GHR, TPO, CSF3, IL6, IL11, IL12, IL6ST, IL12, IL12RB2, IL35, CLCF1, CNTF, (22) | IL9R, IL15RA, CSF2RB, IL13RA2, CSFR, LEPR, IL6R, IL6, OSMR (9) | IL4R, IL13RA1, GH1, GH2, CSF3R, IL6R (6) |
| The class II helical cytokines | IL10RB, IL20RA, IL20, IL28A, IL28B (5) | IL10, IL20, IL20RA, IL22, IL10RB (5) | IL22RA1, IFNGR1 (2) | |
| Interferon family | IFNAR2, IFNGR2 (2) | IFNB1, IFNAR2, IFNG, IFNGR1, IFNGR2 (5) | ||
| IL-1-like cytokines | IL1RAP, IL1RL2, IL18R1, IL18RAP, ST2 (5) | IL1B, IL1RAP, IL1R2, IL1R2, IL18R1, IL18, IL18RAP, ST2 (8) | IL1R1, IL1R2, IL18 (3) | IL1R1 (1) |
| IL-17-like cytokines | IL17RA, IL17B, IL17C, IL17D (4) | IL17F, IL17B (2) | IL17RE (1) | |
| Non-classified | IL16, IL34, CSF1R (3) | IL16, CD4, IL34, CSF1, CSF1R (5) | CSF1 (1) | |
| TNF Family | DCR3, FASLG, FAS, VEGI, DR6, EDA, CD30L, CD40LG, CD30, 4-1BB, OX40L, Ox40G, GITRL, GITR, BCMA, BAFF (16) | FASLG, VEG1, DR4, DR5, DR6, EDA, EDAR, NGF, NGFR, RANKL, OPG, CD30, CD40LG, GITR, BAFF, TACI, RELT (17) | TNFR1, TNFR2, XEDAR, CD40, TROY (5) | TNFR2, DCR3, FAS, RANK, CD40 (5) |
| TGF-beta family | TGFBR2, ACVRL1, TGFBR2, GDF15, GDF2, ACVR2B, GDF10, GDF11, MSTN, INHBA, INHBB, GDF1, AMH, BMPR1B, BMP6, BMP8 (16) | GDF15, GDF11m INHBA, INHBB, GDF1, ACVR1C, GDF9, AMH, AMHR2, BMP4, BMPR1B, BMP5, BMP6 (13) | TGFB3, TGFBR1, ACVR2, BMPR2, BMP2, BMP4, GDF6, GDF7, BMP15, BMP5, ACVR2A (11) | TGFBR1, TGFBR2, ACVR2A, BMPR2, INHBC, BMPR1A, ACVR2B (7) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, Z.W.M.; Karawita, A.C.; Kern, C.; Zhou, H.; Sinclair, J.E.; Yan, L.; Chew, K.Y.; Lowther, S.; Trinidad, L.; Challagulla, A.; et al. Primary Chicken and Duck Endothelial Cells Display a Differential Response to Infection with Highly Pathogenic Avian Influenza Virus. Genes 2021, 12, 901. https://doi.org/10.3390/genes12060901
Tong ZWM, Karawita AC, Kern C, Zhou H, Sinclair JE, Yan L, Chew KY, Lowther S, Trinidad L, Challagulla A, et al. Primary Chicken and Duck Endothelial Cells Display a Differential Response to Infection with Highly Pathogenic Avian Influenza Virus. Genes. 2021; 12(6):901. https://doi.org/10.3390/genes12060901
Chicago/Turabian StyleTong, Zhen Wei Marcus, Anjana C. Karawita, Colin Kern, Huaijun Zhou, Jane E. Sinclair, Limin Yan, Keng Yih Chew, Sue Lowther, Lee Trinidad, Arjun Challagulla, and et al. 2021. "Primary Chicken and Duck Endothelial Cells Display a Differential Response to Infection with Highly Pathogenic Avian Influenza Virus" Genes 12, no. 6: 901. https://doi.org/10.3390/genes12060901
APA StyleTong, Z. W. M., Karawita, A. C., Kern, C., Zhou, H., Sinclair, J. E., Yan, L., Chew, K. Y., Lowther, S., Trinidad, L., Challagulla, A., Schat, K. A., Baker, M. L., & Short, K. R. (2021). Primary Chicken and Duck Endothelial Cells Display a Differential Response to Infection with Highly Pathogenic Avian Influenza Virus. Genes, 12(6), 901. https://doi.org/10.3390/genes12060901









