Coupling between Sequence-Mediated Nucleosome Organization and Genome Evolution
Abstract
1. Introduction
2. How Is Nucleosome Positioning Encoded in the DNA Sequence?
2.1. DNA Sequence Does Influence Nucleosome Positioning
2.2. Sequence Motifs with 10 Base Pair Periodicity as Nucleosome Positioning Signals
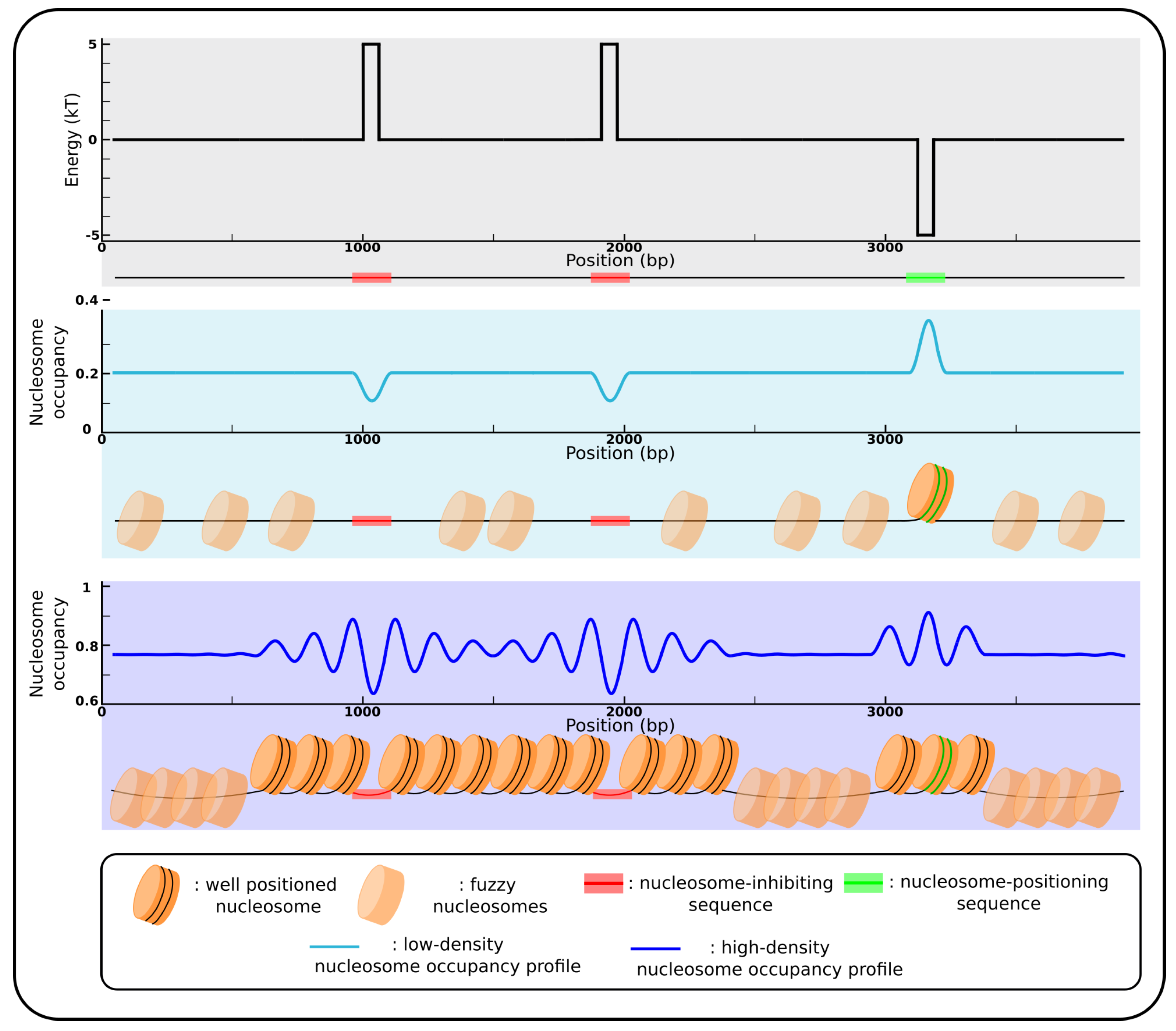
2.3. Sequence-Encoded Nucleosome Depleted Regions and Statistical Positioning
2.4. Predicting Nucleosomal Positioning from Sequences
3. Nucleosome Positioning during Evolution
3.1. Nucleosome Position as a Darwinian Feature
3.2. Nucleosome Positioning and the Evolution of Gene Regulation
3.3. Is Chromatin Organization Selected Genome-Wide?
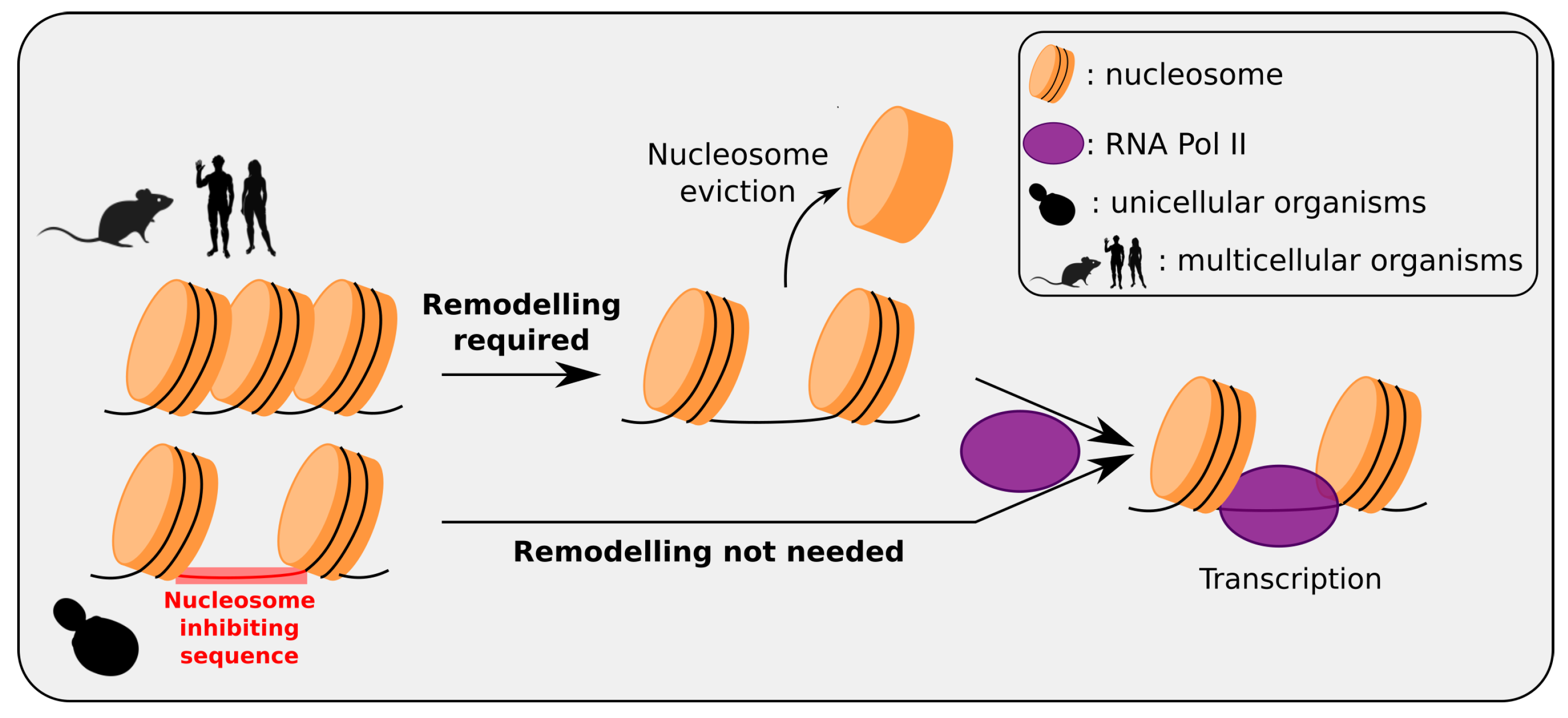
3.4. Are Transposable Elements Involved in Chromatin Organization?
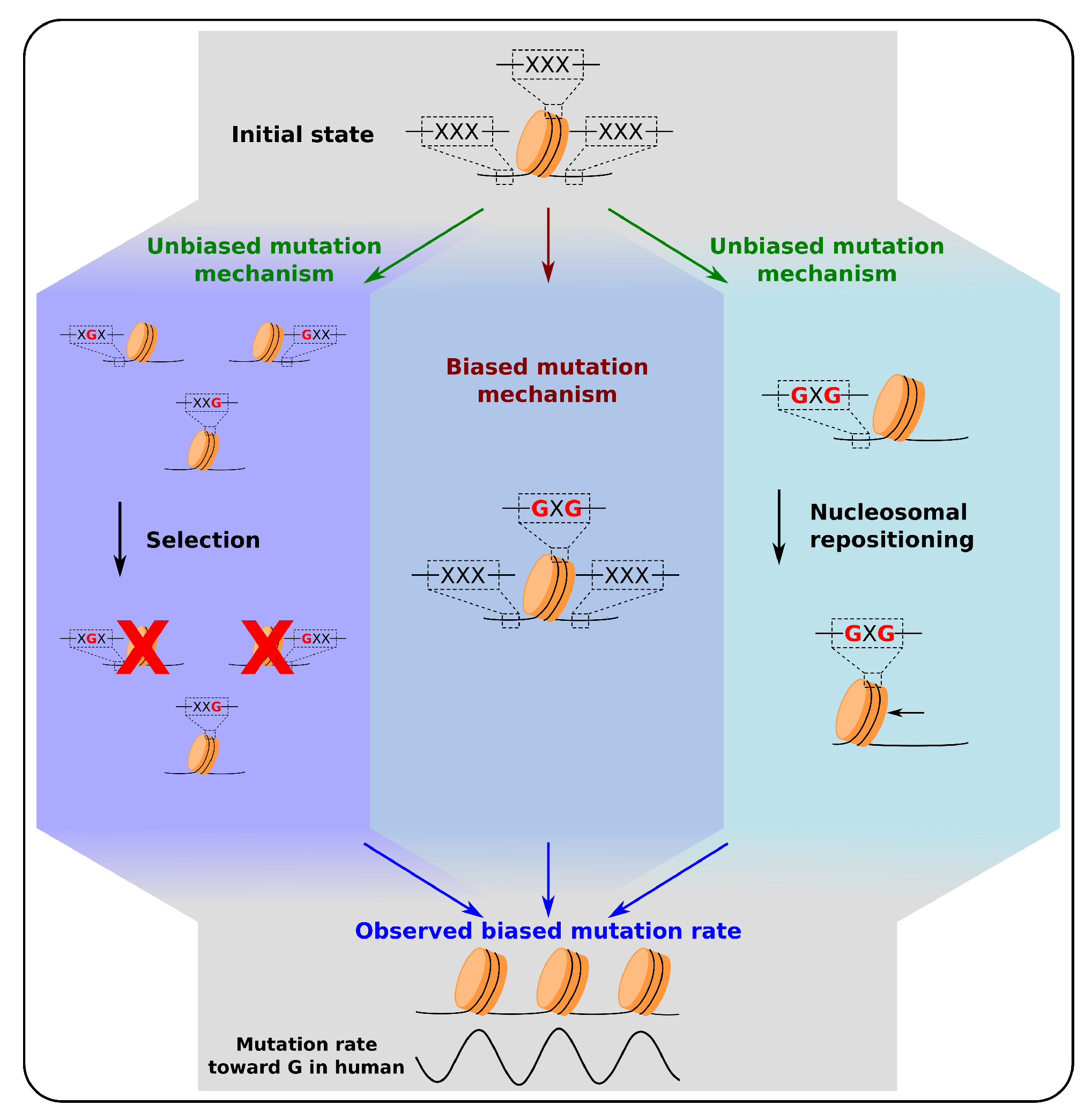
4. Feedback of Nucleosomal Positioning on Mutational Patterns
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BER | Base excision repair |
| bp | Base pair |
| CPD | Cyclobutane pyrimidine dimer |
| MNase | Micrococcal nuclease |
| NAR | Nucleosome attracting region |
| NER | Nucleotide excision repair |
| NDR | Nucleosome depleted region |
| NIEB | Nucleosome-inhibiting energy barrier |
| NRL | Nucleosome repeat length |
| PTM | Post-translational modification |
| SNV | Single nucleotide variation |
| TE | Transposable element |
| TFBS | Transcription factor binding site |
| TSS | Transcription start site |
References
- Segal, E.; Widom, J. What controls nucleosome positions? Trends Genet. 2009, 25, 335–343. [Google Scholar] [CrossRef]
- Kornberg, R.D.; Lorch, Y. Twenty-five years of the nucleosome, fundamental particle of the eukaryote chromosome. Cell 1999, 98, 285–294. [Google Scholar] [CrossRef]
- Finch, J.T.; Lutter, L.C.; Rhodes, D.; Brown, R.S.; Rushton, B.; Levitt, M.; Klug, A. Structure of nucleosome core particles of chromatin. Nature 1977, 269, 29–36. [Google Scholar] [CrossRef] [PubMed]
- McGinty, R.K.; Tan, S. Nucleosome Structure and Function. Chem. Rev. 2015, 115, 2255–2273. [Google Scholar] [CrossRef] [PubMed]
- Lantermann, A.B.; Straub, T.; Strålfors, A.; Yuan, G.C.; Ekwall, K.; Korber, P. Schizosaccharomyces pombe genome-wide nucleosome mapping reveals positioning mechanisms distinct from those of Saccharomyces cerevisiae. Nat. Struct. Mol. Biol. 2010, 17, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Athey, B.D.; Smith, M.F.; Rankert, D.A.; Williams, S.P.; Langmore, J.P. The diameters of frozen-hydrated chromatin fibers increase with DNA linker length: Evidence in support of variable diameter models for chromatin. J. Cell Biol. 1990, 111, 795–806. [Google Scholar] [CrossRef]
- Kurumizaka, H.; Kujirai, T.; Takizawa, Y. Contributions of Histone Variants in Nucleosome Structure and Function. J. Mol. Biol. 2021, 433, 166678. [Google Scholar] [CrossRef]
- Szenker, E.; Ray-Gallet, D.; Almouzni, G. The double face of the histone variant H3.3. Cell Res. 2011, 21, 421–434. [Google Scholar] [CrossRef]
- Correll, S.J.; Schubert, M.H.; Grigoryev, S.A. Short nucleosome repeats impose rotational modulations on chromatin fibre folding. EMBO J. 2012, 31, 2416–2426. [Google Scholar] [CrossRef]
- Valouev, A.; Johnson, S.M.; Boyd, S.D.; Smith, C.L.; Fire, A.Z.; Sidow, A. Determinants of nucleosome organization in primary human cells. Nature 2011, 474, 516–520. [Google Scholar] [CrossRef]
- Pisano, S.; Galati, A.; Cacchione, S. Telomeric nucleosomes: Forgotten players at chromosome ends. Cell. Mol. Life Sci. 2008, 65, 3553–3563. [Google Scholar] [CrossRef] [PubMed]
- Tommerup, H.; Dousmanis, A.; de Lange, T. Unusual chromatin in human telomeres. Mol. Cell. Biol. 1994, 14, 5777–5785. [Google Scholar] [CrossRef] [PubMed]
- Makarov, V.L.; Lejnine, S.; Bedoyan, J.; Langmore, J.P. Nucleosomal organization of telomere-specific chromatin in rat. Cell 1993, 73, 775–787. [Google Scholar] [CrossRef]
- Muyldermans, S.; De Jonge, J.; Wyns, L.; Travers, A.A. Differential association of linker histones H1 and H5 with telomeric nucleosomes in chicken erythrocytes. Nucleic Acids Res. 1994, 22, 5635–5639. [Google Scholar] [CrossRef][Green Version]
- Smilenov, L.B.; Dhar, S.; Pandita, T.K. Altered telomere nuclear matrix interactions and nucleosomal periodicity in ataxia telangiectasia cells before and after ionizing radiation treatment. Mol. Cell. Biol. 1999, 19, 6963–6971. [Google Scholar] [CrossRef]
- Lejnine, S.; Makarov, V.L.; Langmore, J.P. Conserved nucleoprotein structure at the ends of vertebrate and invertebrate chromosomes. Proc. Natl. Acad. Sci. USA 1995, 92, 2393–2397. [Google Scholar] [CrossRef]
- Fajkus, J.; Kovarík, A.; Královics, R.; Bezdĕk, M. Organization of telomeric and subtelomeric chromatin in the higher plant Nicotiana tabacum. Mol. Gen. Genet. 1995, 247, 633–638. [Google Scholar] [CrossRef]
- Sýkorová, E.; Fajkus, J.; Ito, M.; Fukui, K. Transition between two forms of heterochromatin at plant subtelomeres. Chromosome Res. 2001, 9, 309–323. [Google Scholar] [CrossRef] [PubMed]
- Vershinin, A.V.; Heslop-Harrison, J.S. Comparative analysis of the nucleosomal structure of rye, wheat and their relatives. Plant Mol. Biol. 1998, 36, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Rando, O.J.; Ahmad, K. Rules and regulation in the primary structure of chromatin. Curr. Opin. Cell Biol. 2007, 19, 250–256. [Google Scholar] [CrossRef]
- Struhl, K.; Segal, E. Determinants of nucleosome positioning. Nat. Struct. Mol. Biol. 2013, 20, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Tillo, D.; Hughes, T.R. G+C content dominates intrinsic nucleosome occupancy. BMC Bioinform. 2009, 10, 442. [Google Scholar] [CrossRef] [PubMed]
- Field, Y.; Fondufe-Mittendorf, Y.; Moore, I.K.; Mieczkowski, P.; Kaplan, N.; Lubling, Y.; Lieb, J.D.; Widom, J.; Segal, E. Gene expression divergence in yeast is coupled to evolution of DNA-encoded nucleosome organization. Nat. Genet. 2009, 41, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Tsankov, A.M.; Thompson, D.A.; Socha, A.; Regev, A.; Rando, O.J. The role of nucleosome positioning in the evolution of gene regulation. PLoS Biol. 2010, 8, e1000414. [Google Scholar] [CrossRef]
- Chen, J.; Li, E.; Lai, J. The coupled effect of nucleosome organization on gene transcription level and transcriptional plasticity. Nucleus 2017, 8, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, E.; Zhang, X.; Dong, X.; Lei, L.; Song, W.; Zhao, H.; Lai, J. Genome-wide Nucleosome Occupancy and Organization Modulates the Plasticity of Gene Transcriptional Status in Maize. Mol. Plant 2017, 10, 962–974. [Google Scholar] [CrossRef]
- Prendergast, J.G.D.; Semple, C.A.M. Widespread signatures of recent selection linked to nucleosome positioning in the human lineage. Genome Res. 2011, 21, 1777–1787. [Google Scholar] [CrossRef][Green Version]
- Drillon, G.; Audit, B.; Argoul, F.; Arneodo, A. Evidence of selection for an accessible nucleosomal array in human. BMC Genom. 2016, 17, 526. [Google Scholar] [CrossRef]
- Tompitak, M.; Vaillant, C.; Schiessel, H. Genomes of Multicellular Organisms Have Evolved to Attract Nucleosomes to Promoter Regions. Biophys. J. 2017, 112, 505–511. [Google Scholar] [CrossRef]
- Warnecke, T.; Becker, E.A.; Facciotti, M.T.; Nislow, C.; Lehner, B. Conserved substitution patterns around nucleosome footprints in eukaryotes and Archaea derive from frequent nucleosome repositioning through evolution. PLoS Comput. Biol. 2013, 9, e1003373. [Google Scholar] [CrossRef]
- Gonzalez-Perez, A.; Sabarinathan, R.; Lopez-Bigas, N. Local Determinants of the Mutational Landscape of the Human Genome. Cell 2019, 177, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Widlund, H.R.; Cao, H.; Simonsson, S.; Magnusson, E.; Simonsson, T.; Nielsen, P.E.; Kahn, J.D.; Crothers, D.M.; Kubista, M. Identification and characterization of genomic nucleosome-positioning sequences. J. Mol. Biol. 1997, 267, 807–817. [Google Scholar] [CrossRef] [PubMed]
- Lowary, P.T.; Widom, J. New DNA sequence rules for high affinity binding to histone octamer and sequence-directed nucleosome positioning. J. Mol. Biol. 1998, 276, 19–42. [Google Scholar] [CrossRef]
- Thåström, A.; Bingham, L.M.; Widom, J. Nucleosomal locations of dominant DNA sequence motifs for histone-DNA interactions and nucleosome positioning. J. Mol. Biol. 2004, 338, 695–709. [Google Scholar] [CrossRef] [PubMed]
- Gencheva, M.; Boa, S.; Fraser, R.; Simmen, M.W.; A Whitelaw, C.B.; Allan, J. In Vitro and in Vivo nucleosome positioning on the ovine beta-lactoglobulin gene are related. J. Mol. Biol. 2006, 361, 216–230. [Google Scholar] [CrossRef]
- Parmar, J.J.; Marko, J.F.; Padinhateeri, R. Nucleosome positioning and kinetics near transcription-start-site barriers are controlled by interplay between active remodeling and DNA sequence. Nucleic Acids Res. 2014, 42, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Sexton, B.S.; Avey, D.; Druliner, B.R.; Fincher, J.A.; Vera, D.L.; Grau, D.J.; Borowsky, M.L.; Gupta, S.; Girimurugan, S.B.; Chicken, E.; et al. The spring-loaded genome: Nucleosome redistributions are widespread, transient, and DNA-directed. Genome Res. 2014, 24, 251–259. [Google Scholar] [CrossRef][Green Version]
- Vavouri, T.; Lehner, B. Chromatin organization in sperm may be the major functional consequence of base composition variation in the human genome. PLoS Genet 2011, 7, e1002036. [Google Scholar] [CrossRef]
- Zhang, Z.; Wippo, C.J.; Wal, M.; Ward, E.; Korber, P.; Pugh, B.F. A packing mechanism for nucleosome organization reconstituted across a eukaryotic genome. Science 2011, 332, 977–980. [Google Scholar] [CrossRef]
- Sulkowski, E.; Laskowski, M. Mechanism of action of micrococcal nuclease on deoxyribonucleic acid. J. Biol. Chem. 1962, 237, 2620–2625. [Google Scholar] [CrossRef]
- Axel, R. Cleavage of DNA in nuclei and chromatin with staphylococcal nuclease. Biochemistry 1975, 14, 2921–2925. [Google Scholar] [CrossRef]
- Pajoro, A.; Muiño, J.M.; Angenent, G.C.; Kaufmann, K. Profiling Nucleosome Occupancy by MNase-seq: Experimental Protocol and Computational Analysis. Methods Mol. Biol. 2018, 1675, 167–181. [Google Scholar] [CrossRef]
- Yuan, G.C.; Liu, Y.J.; Dion, M.F.; Slack, M.D.; Wu, L.F.; Altschuler, S.J.; Rando, O.J. Genome-scale identification of nucleosome positions in S. cerevisiae. Science 2005, 309, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Schones, D.E.; Cui, K.; Cuddapah, S.; Roh, T.Y.; Barski, A.; Wang, Z.; Wei, G.; Zhao, K. Dynamic regulation of nucleosome positioning in the human genome. Cell 2008, 132, 887–898. [Google Scholar] [CrossRef]
- Gaffney, D.J.; McVicker, G.; Pai, A.A.; Fondufe-Mittendorf, Y.N.; Lewellen, N.; Michelini, K.; Widom, J.; Gilad, Y.; Pritchard, J.K. Controls of nucleosome positioning in the human genome. PLoS Genet. 2012, 8, e1003036. [Google Scholar] [CrossRef] [PubMed]
- Lai, B.; Gao, W.; Cui, K.; Xie, W.; Tang, Q.; Jin, W.; Hu, G.; Ni, B.; Zhao, K. Principles of nucleosome organization revealed by single-cell micrococcal nuclease sequencing. Nature 2018, 562, 281–285. [Google Scholar] [CrossRef]
- Oberbeckmann, E.; Wolff, M.; Krietenstein, N.; Heron, M.; Ellins, J.L.; Schmid, A.; Krebs, S.; Blum, H.; Gerland, U.; Korber, P. Absolute nucleosome occupancy map for the Saccharomyces cerevisiae genome. Genome Res. 2019, 29, 1996–2009. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Tillo, D.; Bray, N.; Morse, R.H.; Davis, R.W.; Hughes, T.R.; Nislow, C. A high-resolution atlas of nucleosome occupancy in yeast. Nat. Genet. 2007, 39, 1235–1244. [Google Scholar] [CrossRef]
- Mavrich, T.N.; Jiang, C.; Ioshikhes, I.P.; Li, X.; Venters, B.J.; Zanton, S.J.; Tomsho, L.P.; Qi, J.; Glaser, R.; Schuster, S.C.; et al. Nucleosome organization in the Drosophila genome. Nature 2008, 453, 358–362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, W.; Jiang, J. Genome-Wide Nucleosome Occupancy and Positioning and Their Impact on Gene Expression and Evolution in Plants. Plant Physiol. 2015, 168, 1406–1416. [Google Scholar] [CrossRef] [PubMed]
- Teif, V.B.; Vainshtein, Y.; Caudron-Herger, M.; Mallm, J.P.; Marth, C.; Höfer, T.; Rippe, K. Genome-wide nucleosome positioning during embryonic stem cell development. Nat. Struct. Mol. Biol. 2012, 19, 1185–1192. [Google Scholar] [CrossRef]
- Jeffers, T.E.; Lieb, J.D. Nucleosome fragility is associated with future transcriptional response to developmental cues and stress in C. elegans. Genome Res. 2017, 27, 75–86. [Google Scholar] [CrossRef]
- Kaplan, N.; Moore, I.K.; Fondufe-Mittendorf, Y.; Gossett, A.J.; Tillo, D.; Field, Y.; LeProust, E.M.; Hughes, T.R.; Lieb, J.D.; Widom, J.; et al. The DNA-encoded nucleosome organization of a eukaryotic genome. Nature 2009, 458, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Teif, V.B. Nucleosome positioning: Resources and tools online. Brief. Bioinform. 2016, 17, 745–757. [Google Scholar] [CrossRef] [PubMed]
- Chevereau, G.; Arnéodo, A.; Vaillant, C. Influence of the genomic sequence on the primary structure of chromatin. Front. Life Sci. 2011, 5, 29–68. [Google Scholar] [CrossRef]
- Segal, E.; Fondufe-Mittendorf, Y.; Chen, L.; Thåström, A.; Field, Y.; Moore, I.K.; Wang, J.P.Z.; Widom, J. A genomic code for nucleosome positioning. Nature 2006, 442, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Vaillant, C.; Audit, B.; Arneodo, A. Experiments confirm the influence of genome long-range correlations on nucleosome positioning. Phys. Rev. Lett. 2007, 99, 218103. [Google Scholar] [CrossRef]
- Field, Y.; Kaplan, N.; Fondufe-Mittendorf, Y.; Moore, I.K.; Sharon, E.; Lubling, Y.; Widom, J.; Segal, E. Distinct Modes of Regulation by Chromatin Encoded through Nucleosome Positioning Signals. PLoS Comput. Biol. 2008, 4. [Google Scholar] [CrossRef]
- Miele, V.; Vaillant, C.; d’Aubenton Carafa, Y.; Thermes, C.; Grange, T. DNA physical properties determine nucleosome occupancy from yeast to fly. Nucleic Acids Res. 2008, 36, 3746–3756. [Google Scholar] [CrossRef]
- Scipioni, A.; De Santis, P. Predicting nucleosome positioning in genomes: Physical and bioinformatic approaches. Biophys. Chem. 2011, 155, 53–64. [Google Scholar] [CrossRef]
- Kaplan, N.; Hughes, T.R.; Lieb, J.D.; Widom, J.; Segal, E. Contribution of histone sequence preferences to nucleosome organization: Proposed definitions and methodology. Genome Biol. 2010, 11, 140. [Google Scholar] [CrossRef][Green Version]
- Trifonov, E.N.; Sussman, J.L. The pitch of chromatin DNA is reflected in its nucleotide sequence. Proc. Natl. Acad. Sci. USA 1980, 77, 3816–3820. [Google Scholar] [CrossRef] [PubMed]
- Trifonov, E.N. Sequence-dependent deformational anisotropy of chromatin DNA. Nucleic Acids Res. 1980, 8, 4041–4053. [Google Scholar] [CrossRef]
- Levene, S.D.; Crothers, D.M. A computer graphics study of sequence-directed bending in DNA. J. Biomol. Struct. Dyn. 1983, 1, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, D. Nucleosome cores reconstituted from poly (dA-dT) and the octamer of histones. Nucleic Acids Res. 1979, 6, 1805–1816. [Google Scholar] [CrossRef][Green Version]
- Simpson, R.T.; Künzler, P. Cromatin and core particles formed from the inner histones and synthetic polydeoxyribonucleotides of defined sequence. Nucleic Acids Res. 1979, 6, 1387–1415. [Google Scholar] [CrossRef]
- Drew, H.R.; Travers, A.A. DNA bending and its relation to nucleosome positioning. J. Mol. Biol. 1985, 186, 773–790. [Google Scholar] [CrossRef]
- Satchwell, S.C.; Drew, H.R.; Travers, A.A. Sequence periodicities in chicken nucleosome core DNA. J. Mol. Biol. 1986, 191, 659–675. [Google Scholar] [CrossRef]
- Johnson, S.M.; Tan, F.J.; McCullough, H.L.; Riordan, D.P.; Fire, A.Z. Flexibility and constraint in the nucleosome core landscape of Caenorhabditis elegans chromatin. Genome Res. 2006, 16, 1505–1516. [Google Scholar] [CrossRef]
- Albert, I.; Mavrich, T.N.; Tomsho, L.P.; Qi, J.; Zanton, S.J.; Schuster, S.C.; Pugh, B.F. Translational and rotational settings of H2A.Z nucleosomes across the Saccharomyces cerevisiae genome. Nature 2007, 446, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Raveh-Sadka, T.; Levo, M.; Shabi, U.; Shany, B.; Keren, L.; Lotan-Pompan, M.; Zeevi, D.; Sharon, E.; Weinberger, A.; Segal, E. Manipulating nucleosome disfavoring sequences allows fine-tune regulation of gene expression in yeast. Nat. Genet. 2012, 44, 743–750. [Google Scholar] [CrossRef]
- Fedor, M.J.; Lue, N.F.; Kornberg, R.D. Statistical positioning of nucleosomes by specific protein-binding to an upstream activating sequence in yeast. J. Mol. Biol. 1988, 204, 109–127. [Google Scholar] [CrossRef]
- Kornberg, R.D.; Stryer, L. Statistical distributions of nucleosomes: Nonrandom locations by a stochastic mechanism. Nucleic Acids Res. 1988, 16, 6677–6690. [Google Scholar] [CrossRef]
- Iyer, V.R. Nucleosome positioning: Bringing order to the eukaryotic genome. Trends Cell Biol. 2012, 22, 250–256. [Google Scholar] [CrossRef]
- Milani, P.; Chevereau, G.; Vaillant, C.; Audit, B.; Haftek-Terreau, Z.; Marilley, M.; Bouvet, P.; Argoul, F.; Arneodo, A. Nucleosome positioning by genomic excluding-energy barriers. Proc. Natl. Acad. Sci. USA 2009, 106, 22257–22262. [Google Scholar] [CrossRef] [PubMed]
- Drillon, G.; Audit, B.; Argoul, F.; Arneodo, A. Ubiquitous human ‘master’ origins of replication are encoded in the DNA sequence via a local enrichment in nucleosome excluding energy barriers. J. Phys. Condens. Matter 2015, 27, 064102. [Google Scholar] [CrossRef] [PubMed]
- Tompitak, M.; Barkema, G.T.; Schiessel, H. Benchmarking and refining probability-based models for nucleosome-DNA interaction. BMC Bioinform. 2017, 18, 157. [Google Scholar] [CrossRef] [PubMed]
- Nishida, H. Nucleosome Positioning. ISRN Mol. Biol. 2012, 2012, 245706. [Google Scholar] [CrossRef]
- Chevereau, G.; Palmeira, L.; Thermes, C.; Arneodo, A.; Vaillant, C. Thermodynamics of intragenic nucleosome ordering. Phys. Rev. Lett. 2009, 103, 188103. [Google Scholar] [CrossRef]
- Vaillant, C.; Palmeira, L.; Chevereau, G.; Audit, B.; d’Aubenton Carafa, Y.; Thermes, C.; Arneodo, A. A novel strategy of transcription regulation by intragenic nucleosome ordering. Genome Res. 2010, 20, 59–67. [Google Scholar] [CrossRef]
- Brogaard, K.; Xi, L.; Wang, J.P.; Widom, J. A map of nucleosome positions in yeast at base-pair resolution. Nature 2012, 486, 496–501. [Google Scholar] [CrossRef]
- Shivaswamy, S.; Bhinge, A.; Zhao, Y.; Jones, S.; Hirst, M.; Iyer, V.R. Dynamic remodeling of individual nucleosomes across a eukaryotic genome in response to transcriptional perturbation. PLoS Biol. 2008, 6, e65. [Google Scholar] [CrossRef]
- Bochkis, I.M.; Przybylski, D.; Chen, J.; Regev, A. Changes in nucleosome occupancy associated with metabolic alterations in aged mammalian liver. Cell Rep. 2014, 9, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Chen, K.; Xia, Z.; Chavez, M.; Pal, S.; Seol, J.H.; Chen, C.C.; Li, W.; Tyler, J.K. Nucleosome loss leads to global transcriptional up-regulation and genomic instability during yeast aging. Genes Dev. 2014, 28, 396–408. [Google Scholar] [CrossRef]
- Sexton, B.S.; Druliner, B.R.; Vera, D.L.; Avey, D.; Zhu, F.; Dennis, J.H. Hierarchical regulation of the genome: Global changes in nucleosome organization potentiate genome response. Oncotarget 2016, 7, 6460–6475. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Weghorn, D.; Lässig, M. Fitness landscape for nucleosome positioning. Proc. Natl. Acad. Sci. USA 2013, 110, 10988–10993. [Google Scholar] [CrossRef] [PubMed]
- Eslami-Mossallam, B.; Schram, R.D.; Tompitak, M.; van Noort, J.; Schiessel, H. Multiplexing Genetic and Nucleosome Positioning Codes: A Computational Approach. PLoS ONE 2016, 11, e0156905. [Google Scholar] [CrossRef] [PubMed]
- Mieczkowski, J.; Cook, A.; Bowman, S.K.; Mueller, B.; Alver, B.H.; Kundu, S.; Deaton, A.M.; Urban, J.A.; Larschan, E.; Park, P.J.; et al. MNase titration reveals differences between nucleosome occupancy and chromatin accessibility. Nat. Commun. 2016, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chereji, R.V.; Bryson, T.D.; Henikoff, S. Quantitative MNase-seq accurately maps nucleosome occupancy levels. Genome Biol. 2019, 20, 198. [Google Scholar] [CrossRef]
- Arneodo, A.; Drillon, G.; Argoul, F.; Audit, B. 2-The Role of Nucleosome Positioning in Genome Function and Evolution. In Nuclear Architecture and Dynamics; Translational Epigenetics; Lavelle, C., Victor, J.M., Eds.; Academic Press: Boston, MA, USA, 2018; Volume 2, pp. 41–79. [Google Scholar] [CrossRef]
- Saxonov, S.; Berg, P.; Brutlag, D.L. A genome-wide analysis of CpG dinucleotides in the human genome distinguishes two distinct classes of promoters. Proc. Natl. Acad. Sci. USA 2006, 103, 1412–1417. [Google Scholar] [CrossRef]
- Ramirez-Carrozzi, V.R.; Braas, D.; Bhatt, D.M.; Cheng, C.S.; Hong, C.; Doty, K.R.; Black, J.C.; Hoffmann, A.; Carey, M.; Smale, S.T. A unifying model for the selective regulation of inducible transcription by CpG islands and nucleosome remodeling. Cell 2009, 138, 114–128. [Google Scholar] [CrossRef]
- Deaton, A.M.; Bird, A. CpG islands and the regulation of transcription. Genes Dev. 2011, 25, 1010–1022. [Google Scholar] [CrossRef]
- Sandman, K.; Reeve, J.N. Structure and functional relationships of archaeal and eukaryal histones and nucleosomes. Arch. Microbiol. 2000, 173, 165–169. [Google Scholar] [CrossRef]
- Brunet, F.G.; Audit, B.; Drillon, G.; Argoul, F.; Volff, J.N.; Arneodo, A. Evidence for DNA Sequence Encoding of an Accessible Nucleosomal Array across Vertebrates. Biophys. J. 2018. [Google Scholar] [CrossRef]
- Durbin, R.M.; Altshuler, D.; Durbin, R.M.; Abecasis, G.R.; Bentley, D.R.; Chakravarti, A.; Clark, A.G.; Collins, F.S.; De La Vega, F.M.; Donnelly, P.; et al. A map of human genome variation from population-scale sequencing. Nature 2010, 467, 1061–1073. [Google Scholar] [CrossRef]
- Biémont, C.; Vieira, C. Junk DNA as an evolutionary force. Nature 2006, 443, 521–524. [Google Scholar] [CrossRef]
- Bourque, G.; Burns, K.H.; Gehring, M.; Gorbunova, V.; Seluanov, A.; Hammell, M.; Imbeault, M.; Izsvák, Z.; Levin, H.L.; Macfarlan, T.S.; et al. Ten things you should know about transposable elements. Genome Biol. 2018, 19, 199. [Google Scholar] [CrossRef]
- Pace, J.K.; Feschotte, C. The evolutionary history of human DNA transposons: Evidence for intense activity in the primate lineage. Genome Res. 2007, 17, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Chalopin, D.; Naville, M.; Plard, F.; Galiana, D.; Volff, J.N. Comparative Analysis of Transposable Elements Highlights Mobilome Diversity and Evolution in Vertebrates. Genome Biol. Evol. 2015, 7, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Wicker, T.; Sabot, F.; Hua-Van, A.; Bennetzen, J.L.; Capy, P.; Chalhoub, B.; Flavell, A.; Leroy, P.; Morgante, M.; Panaud, O.; et al. A unified classification system for eukaryotic transposable elements. Nat. Rev. Genet. 2007, 8, 973–982. [Google Scholar] [CrossRef] [PubMed]
- Etchegaray, E.; Naville, M.; Volff, J.N.; Haftek-Terreau, Z. Transposable element-derived sequences in vertebrate development. Mob. DNA 2021, 12, 1. [Google Scholar] [CrossRef]
- Dechaud, C.; Volff, J.N.; Schartl, M.; Naville, M. Sex and the TEs: Transposable elements in sexual development and function in animals. Mob. DNA 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Payer, L.M.; Burns, K.H. Transposable elements in human genetic disease. Nat. Rev. Genet. 2019, 20, 760–772. [Google Scholar] [CrossRef] [PubMed]
- Deininger, P. Alu elements: Know the SINEs. Genome Biol. 2011, 12, 236. [Google Scholar] [CrossRef]
- Tanaka, Y.; Yamashita, R.; Suzuki, Y.; Nakai, K. Effects of Alu elements on global nucleosome positioning in the human genome. BMC Genom. 2010, 11, 309. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Luscombe, N.M. Nucleosome positioning stability is a modulator of germline mutation rate variation across the human genome. Nat. Commun. 2020, 11, 1363. [Google Scholar] [CrossRef] [PubMed]
- Hara, R.; Mo, J.; Sancar, A. DNA damage in the nucleosome core is refractory to repair by human excision nuclease. Mol. Cell. Biol. 2000, 20, 9173–9181. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, Y.; Smerdon, M.J. The structural location of DNA lesions in nucleosome core particles determines accessibility by base excision repair enzymes. J. Biol. Chem. 2013, 288, 13863–13875. [Google Scholar] [CrossRef]
- Smerdon, M.J.; Conconi, A. Modulation of DNA damage and DNA repair in chromatin. Prog. Nucleic Acid Res. Mol. Biol. 1999, 62, 227–255. [Google Scholar] [CrossRef]
- Pedersen, J.S.; Valen, E.; Velazquez, A.M.V.; Parker, B.J.; Rasmussen, M.; Lindgreen, S.; Lilje, B.; Tobin, D.J.; Kelly, T.K.; Vang, S.; et al. Genome-wide nucleosome map and cytosine methylation levels of an ancient human genome. Genome Res. 2014, 24, 454–466. [Google Scholar] [CrossRef]
- Hanghøj, K.; Seguin-Orlando, A.; Schubert, M.; Madsen, T.; Pedersen, J.S.; Willerslev, E.; Orlando, L. Fast, Accurate and Automatic Ancient Nucleosome and Methylation Maps with epiPALEOMIX. Mol. Biol. Evol. 2016, 33, 3284–3298. [Google Scholar] [CrossRef]
- Chen, X.; Chen, Z.; Chen, H.; Su, Z.; Yang, J.; Lin, F.; Shi, S.; He, X. Nucleosomes suppress spontaneous mutations base-specifically in eukaryotes. Science 2012, 335, 1235–1238. [Google Scholar] [CrossRef] [PubMed]
- Makova, K.D.; Hardison, R.C. The effects of chromatin organization on variation in mutation rates in the genome. Nat. Rev. Genet. 2015, 16, 213–223. [Google Scholar] [CrossRef]
- Wu, J.; McKeague, M.; Sturla, S.J. Nucleotide-Resolution Genome-Wide Mapping of Oxidative DNA Damage by Click-Code-Seq. J. Am. Chem. Soc. 2018, 140, 9783–9787. [Google Scholar] [CrossRef]
- Morganella, S.; Alexandrov, L.B.; Glodzik, D.; Zou, X.; Davies, H.; Staaf, J.; Sieuwerts, A.M.; Brinkman, A.B.; Martin, S.; Ramakrishna, M.; et al. The topography of mutational processes in breast cancer genomes. Nat. Commun. 2016, 7, 11383. [Google Scholar] [CrossRef] [PubMed]
- Nik-Zainal, S.; Morganella, S. Mutational Signatures in Breast Cancer: The Problem at the DNA Level. Clin. Cancer Res. 2017, 23, 2617–2629. [Google Scholar] [CrossRef]
- ICGC/TCGA Pan-Cancer Analysis of Whole Genomes Consortium. Pan-cancer analysis of whole genomes. Nature 2020, 578, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Alexandrov, L.B.; Kim, J.; Haradhvala, N.J.; Huang, M.N.; Tian Ng, A.W.; Wu, Y.; Boot, A.; Covington, K.R.; Gordenin, D.A.; Bergstrom, E.N.; et al. The repertoire of mutational signatures in human cancer. Nature 2020, 578, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Sabarinathan, R.; Mularoni, L.; Deu-Pons, J.; Gonzalez-Perez, A.; López-Bigas, N. Nucleotide excision repair is impaired by binding of transcription factors to DNA. Nature 2016, 532, 264–267. [Google Scholar] [CrossRef]
- Pich, O.; Muiños, F.; Sabarinathan, R.; Reyes-Salazar, I.; Gonzalez-Perez, A.; Lopez-Bigas, N. Somatic and Germline Mutation Periodicity Follow the Orientation of the DNA Minor Groove around Nucleosomes. Cell 2018, 175, 1074–1087. [Google Scholar] [CrossRef]
- Langley, S.A.; Karpen, G.H.; Langley, C.H. Nucleosomes shape DNA polymorphism and divergence. PLoS Genet. 2014, 10, e1004457. [Google Scholar] [CrossRef]
- Mao, P.; Brown, A.J.; Malc, E.P.; Mieczkowski, P.A.; Smerdon, M.J.; Roberts, S.A.; Wyrick, J.J. Genome-wide maps of alkylation damage, repair, and mutagenesis in yeast reveal mechanisms of mutational heterogeneity. Genome Res. 2017, 27, 1674–1684. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Wyrick, J.J. Organization of DNA damage, excision repair, and mutagenesis in chromatin: A genomic perspective. DNA Repair 2019, 81, 102645. [Google Scholar] [CrossRef] [PubMed]
- Gale, J.M.; Nissen, K.A.; Smerdon, M.J. UV-induced formation of pyrimidine dimers in nucleosome core DNA is strongly modulated with a period of 10.3 bases. Proc. Natl. Acad. Sci. USA 1987, 84, 6644–6648. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Wyrick, J.J.; Roberts, S.A.; Smerdon, M.J. UV-Induced DNA Damage and Mutagenesis in Chromatin. Photochem. Photobiol. 2017, 93, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Smerdon, M.J.; Roberts, S.A.; Wyrick, J.J. Chromosomal landscape of UV damage formation and repair at single-nucleotide resolution. Proc. Natl. Acad. Sci. USA 2016, 113, 9057–9062. [Google Scholar] [CrossRef]
- Brown, A.J.; Mao, P.; Smerdon, M.J.; Wyrick, J.J.; Roberts, S.A. Nucleosome positions establish an extended mutation signature in melanoma. PLoS Genet. 2018, 14. [Google Scholar] [CrossRef]
- Hayward, N.K.; Wilmott, J.S.; Waddell, N.; Johansson, P.A.; Field, M.A.; Nones, K.; Patch, A.M.; Kakavand, H.; Alexandrov, L.B.; Burke, H.; et al. Whole-genome landscapes of major melanoma subtypes. Nature 2017, 545, 175–180. [Google Scholar] [CrossRef]
- Pestov, N.A.; Gerasimova, N.S.; Kulaeva, O.I.; Studitsky, V.M. Structure of transcribed chromatin is a sensor of DNA damage. Sci. Adv. 2015, 1, e1500021. [Google Scholar] [CrossRef]
- Adar, S.; Hu, J.; Lieb, J.D.; Sancar, A. Genome-wide kinetics of DNA excision repair in relation to chromatin state and mutagenesis. Proc. Natl. Acad. Sci. USA 2016, 113, E2124–E2133. [Google Scholar] [CrossRef]
- Li, W.; Hu, J.; Adebali, O.; Adar, S.; Yang, Y.; Chiou, Y.Y.; Sancar, A. Human genome-wide repair map of DNA damage caused by the cigarette smoke carcinogen benzo[a]pyrene. Proc. Natl. Acad. Sci. USA 2017, 114, 6752–6757. [Google Scholar] [CrossRef]
- Polak, P.; Karlić, R.; Koren, A.; Thurman, R.; Sandstrom, R.; Lawrence, M.; Reynolds, A.; Rynes, E.; Vlahoviček, K.; Stamatoyannopoulos, J.A.; et al. Cell-of-origin chromatin organization shapes the mutational landscape of cancer. Nature 2015, 518, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Lieb, J.D.; Sancar, A.; Adar, S. Cisplatin DNA damage and repair maps of the human genome at single-nucleotide resolution. Proc. Natl. Acad. Sci. USA 2016, 113, 11507–11512. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Mueller-Planitz, F. Nucleosome Positioning and Spacing: From Mechanism to Function. J. Mol. Biol. 2021, 433, 166847. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbier, J.; Vaillant, C.; Volff, J.-N.; Brunet, F.G.; Audit, B. Coupling between Sequence-Mediated Nucleosome Organization and Genome Evolution. Genes 2021, 12, 851. https://doi.org/10.3390/genes12060851
Barbier J, Vaillant C, Volff J-N, Brunet FG, Audit B. Coupling between Sequence-Mediated Nucleosome Organization and Genome Evolution. Genes. 2021; 12(6):851. https://doi.org/10.3390/genes12060851
Chicago/Turabian StyleBarbier, Jérémy, Cédric Vaillant, Jean-Nicolas Volff, Frédéric G. Brunet, and Benjamin Audit. 2021. "Coupling between Sequence-Mediated Nucleosome Organization and Genome Evolution" Genes 12, no. 6: 851. https://doi.org/10.3390/genes12060851
APA StyleBarbier, J., Vaillant, C., Volff, J.-N., Brunet, F. G., & Audit, B. (2021). Coupling between Sequence-Mediated Nucleosome Organization and Genome Evolution. Genes, 12(6), 851. https://doi.org/10.3390/genes12060851






