Genetic Predisposition to Alzheimer’s Disease Is Associated with Enlargement of Perivascular Spaces in Centrum Semiovale Region
Abstract
1. Introduction
2. Material and Methods
2.1. Participants
2.2. Standard Protocol Approvals, Registrations, and Patient Consents
2.3. Genotyping
2.4. Image Acquisition and Rating of ePVS
2.5. Statistical Analysis
3. Results
3.1. Sample Descriptive
3.2. APOE and ePVS
3.3. BIN1-rs744373 and ePVS
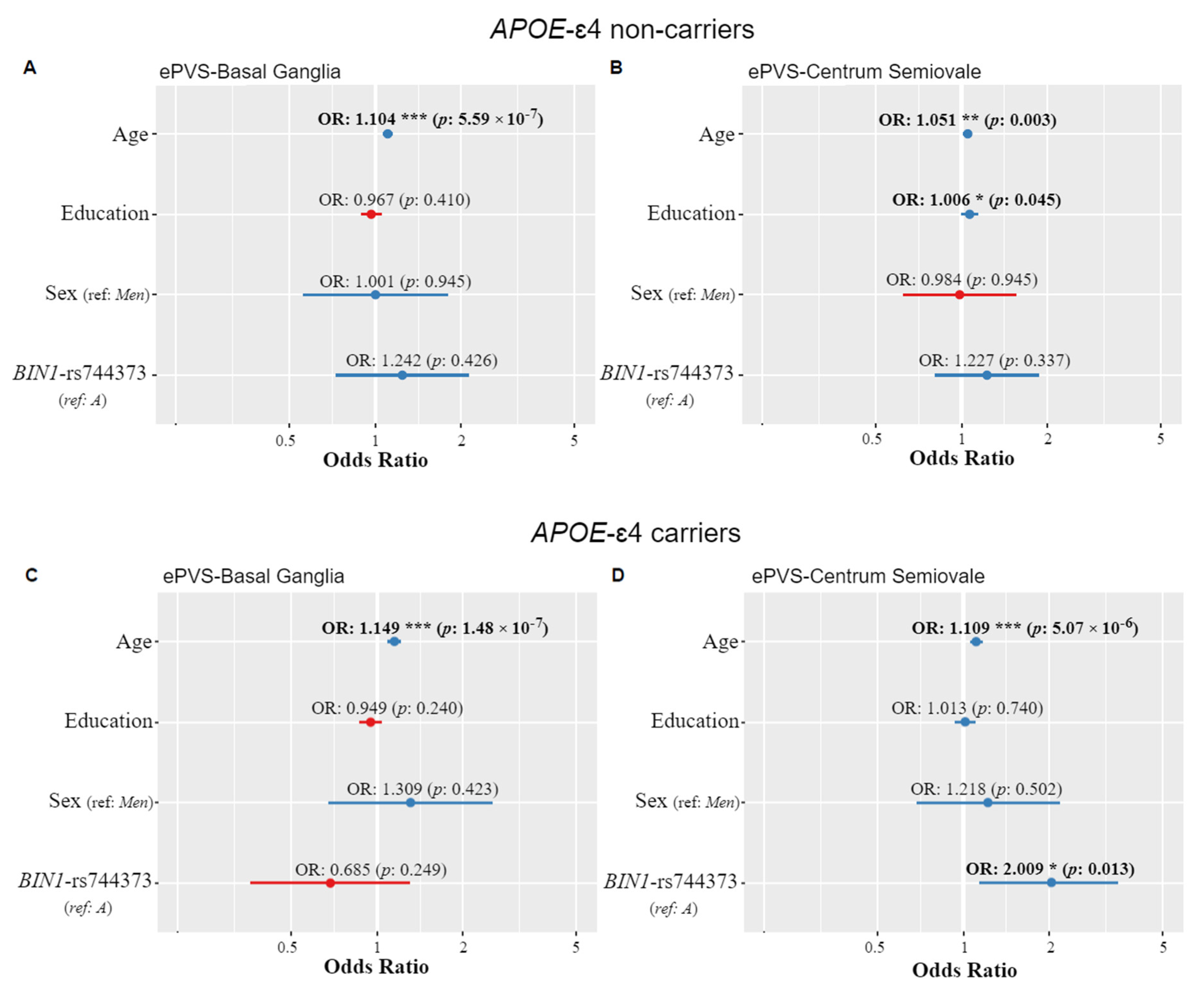
3.4. Age-Dependent Effects
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wardlaw, J.M.; Benveniste, H.; Nedergaard, M.; Zlokovic, B.V.; Mestre, H.; Lee, H.; Doubal, F.N.; Brown, R.; Ramirez, J.; MacIntosh, B.J.; et al. Perivascular spaces in the brain: Anatomy, physiology and pathology. Nat. Rev. Neurol. 2020, 16, 137–153. [Google Scholar] [CrossRef] [PubMed]
- Zong, X.; Lian, C.; Jimenez, J.; Yamashita, K.; Shen, D.; Lin, W. Morphology of perivascular spaces and enclosed blood vessels in young to middle-aged healthy adults at 7T: Dependences on age, brain region, and breathing gas. NeuroImage 2020, 218, 116978. [Google Scholar] [CrossRef] [PubMed]
- Kelsey, R. Perivascular spaces are associated with cognition. Nat. Rev. Neurol. 2019, 15, 246–247. [Google Scholar] [CrossRef] [PubMed]
- Wuerfel, J.; Haertle, M.; Waiczies, H.; Tysiak, E.; Bechmann, I.; Wernecke, K.D.; Zipp, F.; Paul, F. Perivascular spaces—MRI marker of inflammatory activity in the brain? Brain 2008, 131, 2332–2340. [Google Scholar] [CrossRef]
- Brown, R.; Benveniste, H.; E Black, S.; Charpak, S.; Dichgans, M.; Joutel, A.; Nedergaard, M.; Smith, K.J.; Zlokovic, B.V.; Wardlaw, J.M. Understanding the role of the perivascular space in cerebral small vessel disease. Cardiovasc. Res. 2018, 114, 1462–1473. [Google Scholar] [CrossRef]
- Chan, S.T.; Mercaldo, N.D.; Ravina, B.; Hersch, S.M.; Rosas, H.D. Association of dilated perivascular spaces and disease severity in patients with Huntington’s disease. Neurology 2021, 96, e890–e894. [Google Scholar] [CrossRef]
- Ding, J.; Sigurðsson, S.; Jónsson, P.V.; Eiriksdottir, G.; Charidimou, A.; Lopez, O.L.; A Van Buchem, M.; Guðnason, V.; Launer, L.J. Large Perivascular Spaces Visible on Magnetic Resonance Imaging, Cerebral Small Vessel Disease Progression, and Risk of Dementia. JAMA Neurol. 2017, 74, 1105–1112. [Google Scholar] [CrossRef]
- Weller, R.O.; Boche, D.; Nicoll, J.A.R. Microvasculature changes and cerebral amyloid angiopathy in Alzheimer’s disease and their potential impact on therapy. Acta Neuropathol. 2009, 118, 87–102. [Google Scholar] [CrossRef]
- Boespflug, E.L.; Simon, M.J.; Leonard, E.; Grafe, M.; Woltjer, R.; Silbert, L.C.; Kaye, J.A.; Iliff, J.J. Targeted Assessment of Enlargement of the Perivascular Space in Alzheimer’s Disease and Vascular Dementia Subtypes Implicates Astroglial Involvement Specific to Alzheimer’s Disease. J. Alzheimer’s Dis. 2018, 66, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Smeijer, D.; Ikram, M.K.; Hilal, S. Enlarged Perivascular Spaces and Dementia: A Systematic Review. J. Alzheimer’s Dis. 2019, 72, 247–256. [Google Scholar] [CrossRef]
- Shams, S.; Martola, J.; Charidimou, A.; Larvie, M.; Granberg, T.; Shams, M.; Kristoffersen-Wiberg, M.; Wahlund, L.-O. Topography and determinants of magnetic resonance imaging (MRI)-visible perivascular spaces in a large memory clinic cohort. J. Am. Heart Assoc. 2017, 6, e006279. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, G.; Kim, H.J.; Fox, Z.; Jäger, H.R.; Wilson, D.; Charidimou, A.; Na, H.K.; Na, D.L.; Seo, S.W.; Werring, D.J. MRI-visible perivascular space location is associated with Alzheimer’s disease independently of amyloid burden. Brain 2017, 140, 1107–1116. [Google Scholar] [CrossRef]
- Liu, C.-C.; Kanekiyo, T.; Xu, H.; Bu, G. Apolipoprotein E and Alzheimer disease: Risk, mechanisms and therapy. Nat. Rev. Neurol. 2013, 9, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Das, S.; Hyman, B.T. APOE and Alzheimer’s disease: Advances in genetics, pathophysiology, and therapeutic approaches. Lancet Neurol. 2021, 20, 68–80. [Google Scholar] [CrossRef]
- Montagne, A.; Nation, D.A.; Sagare, A.P.; Barisano, G.; Sweeney, M.D.; Chakhoyan, A.; Pachicano, M.; Joe, E.; Nelson, A.R.; D’Orazio, L.M.; et al. APOE4 leads to blood–brain barrier dysfunction predicting cognitive decline. Nature 2020, 581, 71–76. [Google Scholar] [CrossRef]
- Kim, J.; Basak, J.M.; Holtzman, D.M. The Role of Apolipoprotein E in Alzheimer’s Disease. Neuron 2009, 63, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Jiaerken, Y.; Yu, X.; Huang, P.; Qiu, T.; Jia, Y.; Li, K.; Xu, X.; Shen, Z.; Guan, X.; et al. Associations between APOE genotype and cerebral small-vessel disease: A longitudinal study. Oncotarget 2017, 8, 44477–44489. [Google Scholar] [CrossRef]
- Bertram, L.; McQueen, M.B.; Mullin, K.; Blacker, D.; E Tanzi, R. Systematic meta-analyses of Alzheimer disease genetic association studies: The AlzGene database. Nat. Genet. 2007, 39, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.-S.; Yu, J.-T.; Tan, L. Bridging integrator 1 (BIN1): Form, function, and Alzheimer’s disease. Trends Mol. Med. 2013, 19, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Franzmeier, N.; Rubinski, A.; Neitzel, J.; Ewers, M.; The Alzheimer’s Disease Neuroimaging Initiative (ADNI). The BIN1 rs744373 SNP is associated with increased tau-PET levels and impaired memory. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Crotti, A.; Sait, H.R.; McAvoy, K.M.; Estrada, K.; Ergun, A.; Szak, S.; Marsh, G.; Jandreski, L.; Peterson, M.; Reynolds, T.L.; et al. BIN1 favors the spreading of Tau via extracellular vesicles. Sci. Rep. 2019, 9, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.C.; Ibrahim-Verbaas, C.A.; Harold, D.; Naj, A.C.; Sims, R.; Bellenguez, C.; DeStafano, A.L.; Bis, J.C.; Beecham, G.W.; Grenier-Boley, B.; et al. Meta-Analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat. Genet. 2013, 45, 1452–1458. [Google Scholar] [CrossRef]
- Jansen, I.E.; Savage, J.E.; Watanabe, K.; Bryois, J.; Williams, D.M.; Steinberg, S.; Sealock, J.; Karlsson, I.K.; Hägg, S.; Athanasiu, L.; et al. Genome-wide meta-analysis identifies new loci and functional pathways influencing Alzheimer’s disease risk. Nat. Genet. 2019, 51, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.; Harold, D.; Williams, J. Genetic evidence for the involvement of lipid metabolism in Alzheimer’s disease. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2010, 1801, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Villegas-Llerena, C.; Phillips, A.; Garcia-Reitboeck, P.; Hardy, J.; Pocock, J.M. Microglial genes regulating neuroinflammation in the progression of Alzheimer’s disease. Curr. Opin. Neurobiol. 2016, 36, 74–81. [Google Scholar] [CrossRef]
- Molinuevo, J.L.; Gramunt, N.; Gispert, J.D.; Fauria, K.; Esteller, M.; Minguillon, C.; Sánchez-Benavides, G.; Huesa, G.; Morán, S.; Dal-Ré, R.; et al. The ALFA project: A research platform to identify early pathophysiological features of Alzheimer’s disease. Alzheimer’s Dementia: Transl. Res. Clin. Interv. 2016, 2, 82–92. [Google Scholar] [CrossRef]
- Blauwendraat, C.; Faghri, F.; Pihlstrom, L.; Geiger, J.T.; Elbaz, A.; Lesage, S.; Corvol, J.-C.; May, P.; Nicolas, A.; Abramzon, Y.; et al. NeuroChip, an updated version of the NeuroX genotyping platform to rapidly screen for variants associated with neurological diseases. Neurobiol. Aging 2017, 57, 247.e9–247.e13. [Google Scholar] [CrossRef]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef]
- Vilor-Tejedor, N.; for the ALFA Study; Operto, G.; Evans, T.E.; Falcon, C.; Crous-Bou, M.; Minguillón, C.; Cacciaglia, R.; Milà-Alomà, M.; Grau-Rivera, O.; et al. Effect of BDNF Val66Met on hippocampal subfields volumes and compensatory interaction with APOE-ε4 in middle-age cognitively unimpaired individuals from the ALFA study. Brain Struct. Funct. 2020, 225, 2331–2345. [Google Scholar] [CrossRef]
- Radmanesh, F.; (Adni), F.T.A.D.N.I.; Devan, W.J.; Anderson, C.D.; Rosand, J.; Falcone, G.J. Accuracy of imputation to infer unobserved APOE epsilon alleles in genome-wide genotyping data. Eur. J. Hum. Genet. 2014, 22, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- A Heier, L.; Bauer, C.J.; Schwartz, L.; Zimmerman, R.D.; Morgello, S.; Deck, M.D. Large Virchow-Robin spaces: MR-clinical correlation. Am. J. Neuroradiol. 1989, 10, 929–936. [Google Scholar]
- MacLullich, A.M.J.; Wardlaw, J.M.; Ferguson, K.J.; Starr, J.M.; Seckl, J.R.; Deary, I.J. Enlarged perivascular spaces are associated with cognitive function in healthy elderly men. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1519–1523. [Google Scholar] [CrossRef] [PubMed]
- Doubal, F.N.; MacLullich, A.M.; Ferguson, K.J.; Dennis, M.S.; Wardlaw, J.M. Enlarged Perivascular Spaces on MRI Are a Feature of Cerebral Small Vessel Disease. Stroke 2010, 41, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Potter, G.M.; Chappell, F.M.; Morris, Z.; Wardlaw, J.M. Cerebral Perivascular Spaces Visible on Magnetic Resonance Imaging: Development of a Qualitative Rating Scale and its Observer Reliability. Cerebrovasc. Dis. 2015, 39, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.C.; Tzourio, C.; Soumaré, A.; Mazoyer, B.; Dufouil, C.; Chabriat, H. Severity of dilated virchow-robin spaces is associated with age, blood pressure, and MRI markers of small vessel disease: A population-based study. Stroke 2010, 41, 2483–2490. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-F.; Yu, J.-T.; Tan, L. Bridging integrator 1 (BIN1) genotypes Induce Alzheimer’s disease—Related Brain atrophy, abnormal glucose and Aβ metabolisms in ADNI cohort (P2.159). Neurology 2015, 84 (Suppl. S14). [Google Scholar]
- Calafate, S.; Flavin, W.; Verstreken, P.; Moechars, D. Loss of Bin1 Promotes the Propagation of Tau Pathology. Cell Rep. 2016, 17, 931–940. [Google Scholar] [CrossRef]
- Cook, C.; Kang, S.S.; Carlomagno, Y.; Lin, W.-L.; Yue, M.; Kurti, A.; Shinohara, M.; Jansen-West, K.; A Perkerson, E.; Castanedes-Casey, M.; et al. Tau deposition drives neuropathological, inflammatory and behavioral abnormalities independently of neuronal loss in a novel mouse model. Hum. Mol. Genet. 2015, 24, 6198–6212. [Google Scholar] [CrossRef]
- Charidimou, A.; Gang, Q.; Werring, D.J. Sporadic cerebral amyloid angiopathy revisited: Recent insights into pathophysiology and clinical spectrum. J. Neurol. Neurosurg. Psychiatry 2011, 83, 124–137. [Google Scholar] [CrossRef]
- Martinez-Ramirez, S.; Pontes-Neto, O.M.; Dumas, A.P.; Auriel, E.; Halpin, A.; Quimby, M.; Gurol, M.E.; Greenberg, S.M.; Viswanathan, A. Topography of dilated perivascular spaces in subjects from a memory clinic cohort. Neurology 2013, 80, 1551–1556. [Google Scholar] [CrossRef]
- Patankar, T.F.; Mitra, D.; Varma, A.; Snowden, J.; Neary, D.; Jackson, A. Dilatation of the Virchow-Robin space is a sensitive indicator of cerebral microvascular disease: Study in elderly patients with dementia. Am. J. Neuroradiol. 2005, 26, 1512–1520. [Google Scholar] [PubMed]
- Cacciaglia, R.; Molinuevo, J.L.; Falcón, C.; Brugulat-Serrat, A.; Sánchez-Benavides, G.; Gramunt, N.; Esteller, M.; Morán, S.; Minguillón, C.; Fauria, K.; et al. Effects of APOE-ε4 allele load on brain morphology in a cohort of middle-aged healthy individuals with enriched genetic risk for Alzheimer’s disease. Alzheimer’s Dement. 2018, 14, 902–912. [Google Scholar] [CrossRef]
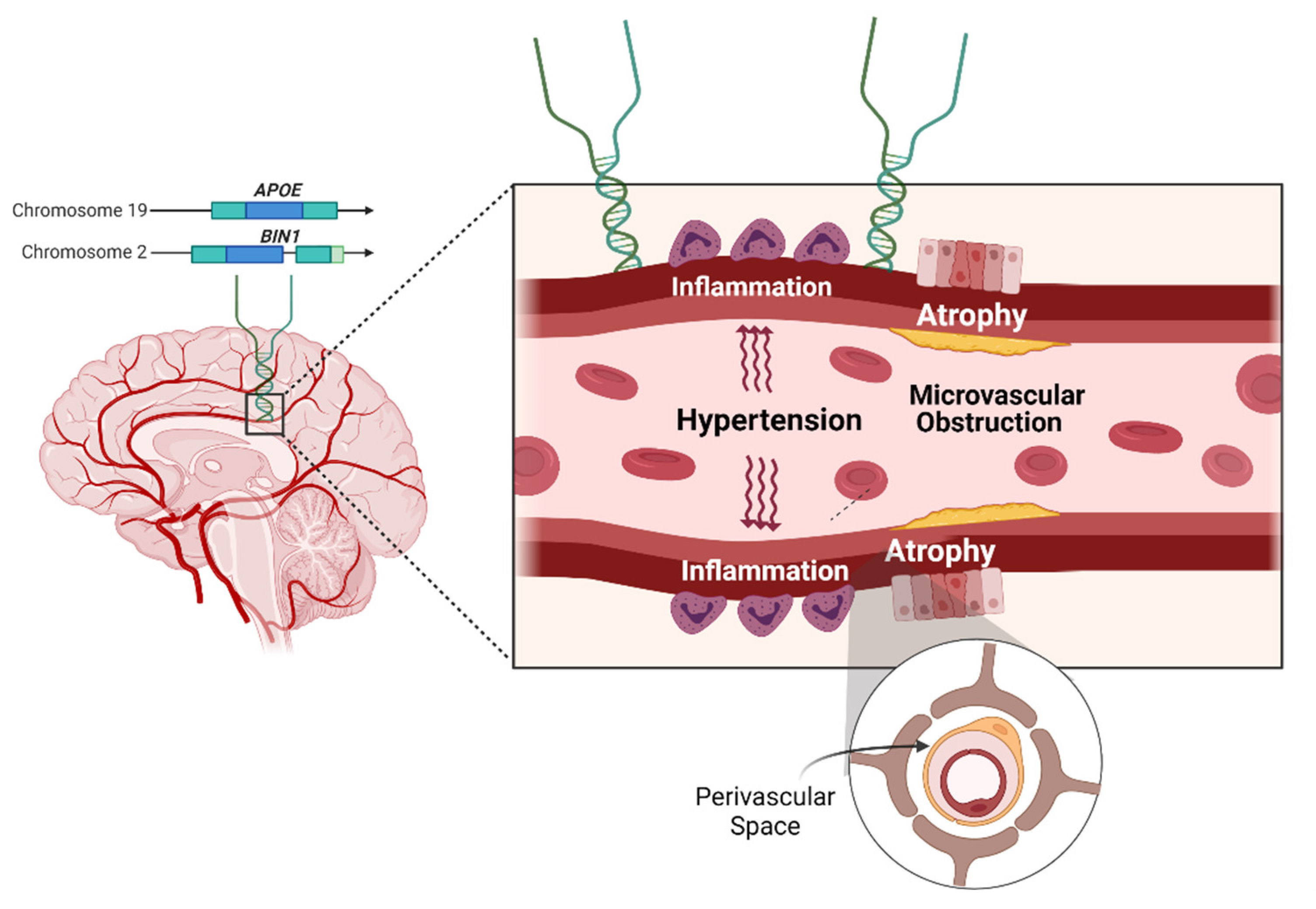
| Non-Severe BG Rating (n = 550) | Severe BG Rating (n = 130) | Total (n = 680) | p (χ2, F) | |
|---|---|---|---|---|
| Age (m ± SD; years) | 58.99 (±6.5) | 64.02 (±5.81) | 59.95 (±6.67) | 0.120 |
| Sex (female), n (%) | 374 (68%) | 86 (66%) | 460 (67%) | 0.763 |
| Education (m ± SD; years) | 13.58 (±3.42) | 13 (±3.41) | 13.47 (±3.42) | 0.987 |
| APOE-ε4 carriers, n (%) | 221 (40%) | 59 (45%) | 280 (41%) | 0.324 |
| Number of APOE-ε4 alleles, n (%) | 0:329 (60%); 1:192 (35%); 2:29 (5%) | 0:71 (55%); 1:47 (36%); 2:12 (9%) | 0:400 (59%); 1:239 (35%); 2:41 (6%) | 0.195 |
| APOE-ε4 isoforms, n (%) | 23:28 (5%); 33:297 (54%); 34:180 (33%); 44:29 (5%) | 23:7 (5%); 33:60 (46%); 34:46 (35%); 44:12 (9%) | 23:35 (5%); 33:357 (52%); 34:226 (33%); 44:41 (6%) | 0.068 |
| BIN1-rs744373 G allele carriers n (%) | 280 (51%) | 65 (50%) | 345 (51%) | 0.929 |
| Non-Severe CS Rating (n = 227) | Severe CS Rating (n = 453) | Total (n = 680) | p (χ2, F) | |
|---|---|---|---|---|
| Age (m ± SD; years) | 58.06 (±5.75) | 60.9 (±6.9) | 59.95 (±6.67) | 0.002 |
| Sex (female), n (%) | 155 (68%) | 305 (67%) | 460 (67%) | 0.87 |
| Education (m ± SD; years) | 13.21 (±3.47) | 13.6 (±3.39) | 13.47 (±3.42) | 0.663 |
| APOE-ε4 carriers, n (%) | 87 (38%) | 193 (43%) | 280 (41%) | 0.323 |
| Number of APOE-ε4 alleles, n (%) | 0:140 (62%); 1:77 (34%); 2:10 (4%) | 0:260 (57%); 1:162 (36%); 2:31 (7%) | 0:400 (59%); 1:239 (35%); 2:41 (6%) | 0.348 |
| APOE genotypes, n (%) | 23:12 (5%); 33:126 (55%); 34:73 (32%); 44:10 (4%) | 23:23 (5%); 33:231 (51%); 34:153 (34%); 44:31 (7%) | 23:35 (5%); 33:357 (52%); 34:226 (33%); 44:41 (6%) | 0.776 |
| BIN1-rs744373 G allele carriers n (%) | 130 (57%) | 215 (47%) | 345 (51%) | 0.019 |
| Basal Ganglia | Centrum Semiovale | |||
|---|---|---|---|---|
| OR (IC 95%) | p-Value ₸ | OR (IC 95%) | p-Value ₸ | |
| Age (years) | 1.121 [1.087;1.155] | <0.001 | 1.071 [1.043;1.099] | <0.001 |
| Sex | ||||
| Male | Ref. | Ref. | Ref. | Ref. |
| Female | 1.17 [0.721;1.626] | 0.437 | 1.044 [0.743;1.474] | 0.743 |
| APOE genotypes | ||||
| ε3ε3 | Ref. | Ref. | Ref. | Ref. |
| ε2ε3 | 1.256 [0.481;2.879] | 0.620 | 1.039 [0.506;2.239] | 0.918 |
| ε3ε4 | 1.265 [0.822;1.937] | 0.282 | 1.142 [0.803;1.631] | 0.460 |
| ε4ε4 | 2.057 [0.956;4.193] | 0.064 | 1.672 [0.816;3.721] | 0.165 |
| APOE-ε4 | ||||
| non-carriers | Ref. | Ref. | Ref. | Ref. |
| carriers | 1.19 [0.839;1.818] | 0.379 | 1.194 [0.862;1.658] | 0.445 |
| APOE-ε4 genotypes | ||||
| 0 alleles | Ref. | Ref. | Ref. | Ref. |
| 1 allele | 1.135 [0.750;1.706] | 0.546 | 1.132 [0.806;1.596] | 0.475 |
| 2 alleles | 1.927 [0.902;3.893] | 0.088 | 1.651 [0.809;3.663] | 0.174 |
| BIN1-rs744373 (dominant model) | ||||
| AA genotype | Ref. | Ref. | Ref. | Ref. |
| AG + GG genotype | 1.037 [0.707;1.522] | 0.848 | 1.481 [1.075;2.049] | 0.022 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciampa, I.; Operto, G.; Falcon, C.; Minguillon, C.; Castro de Moura, M.; Piñeyro, D.; Esteller, M.; Molinuevo, J.L.; Guigó, R.; Navarro, A.; et al. Genetic Predisposition to Alzheimer’s Disease Is Associated with Enlargement of Perivascular Spaces in Centrum Semiovale Region. Genes 2021, 12, 825. https://doi.org/10.3390/genes12060825
Ciampa I, Operto G, Falcon C, Minguillon C, Castro de Moura M, Piñeyro D, Esteller M, Molinuevo JL, Guigó R, Navarro A, et al. Genetic Predisposition to Alzheimer’s Disease Is Associated with Enlargement of Perivascular Spaces in Centrum Semiovale Region. Genes. 2021; 12(6):825. https://doi.org/10.3390/genes12060825
Chicago/Turabian StyleCiampa, Iacopo, Grégory Operto, Carles Falcon, Carolina Minguillon, Manuel Castro de Moura, David Piñeyro, Manel Esteller, Jose Luis Molinuevo, Roderic Guigó, Arcadi Navarro, and et al. 2021. "Genetic Predisposition to Alzheimer’s Disease Is Associated with Enlargement of Perivascular Spaces in Centrum Semiovale Region" Genes 12, no. 6: 825. https://doi.org/10.3390/genes12060825
APA StyleCiampa, I., Operto, G., Falcon, C., Minguillon, C., Castro de Moura, M., Piñeyro, D., Esteller, M., Molinuevo, J. L., Guigó, R., Navarro, A., Gispert, J. D., Vilor-Tejedor, N., & for the ALFA Study. (2021). Genetic Predisposition to Alzheimer’s Disease Is Associated with Enlargement of Perivascular Spaces in Centrum Semiovale Region. Genes, 12(6), 825. https://doi.org/10.3390/genes12060825








