Regeneration Potential of Jellyfish: Cellular Mechanisms and Molecular Insights
Abstract
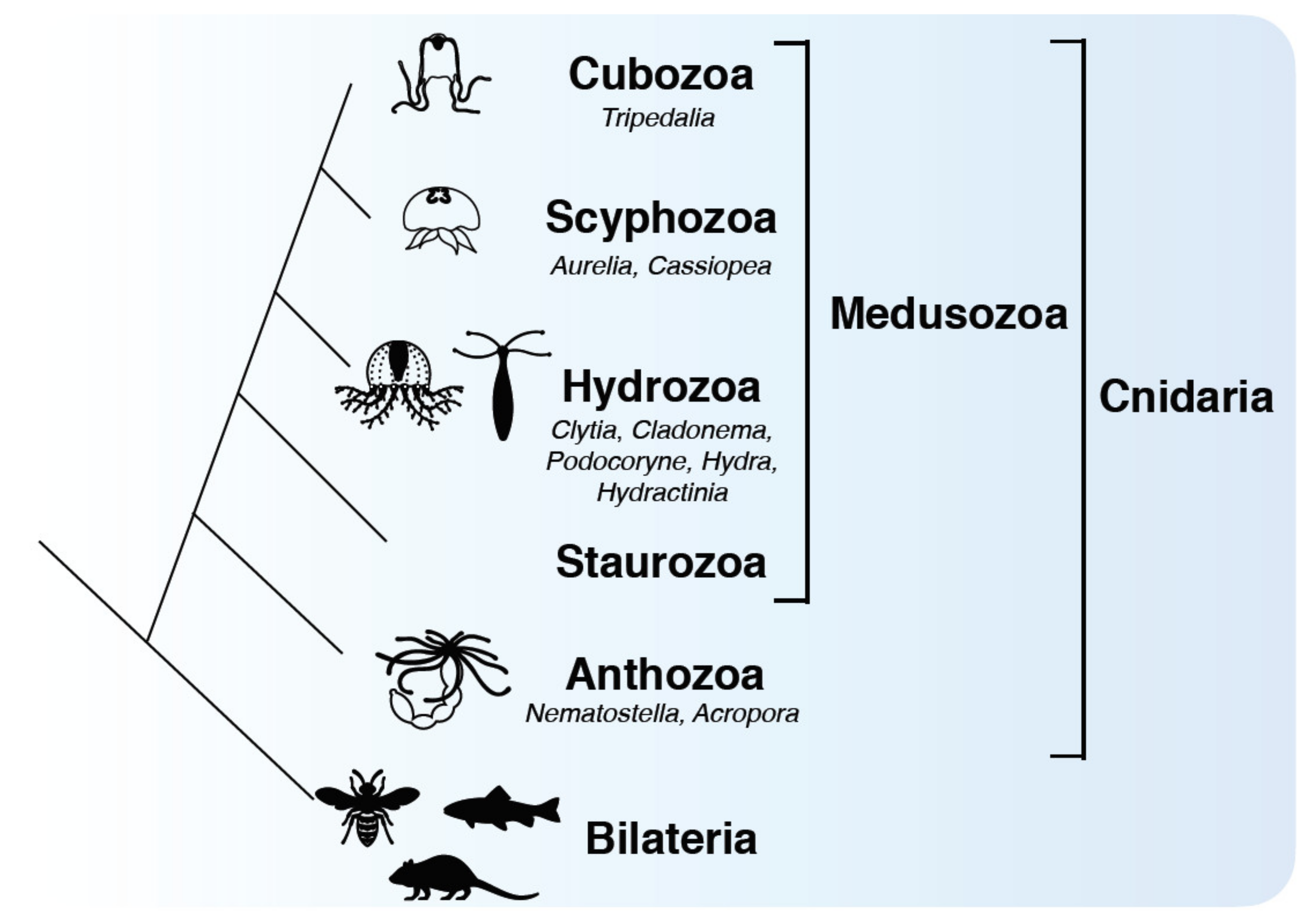
1. Introduction
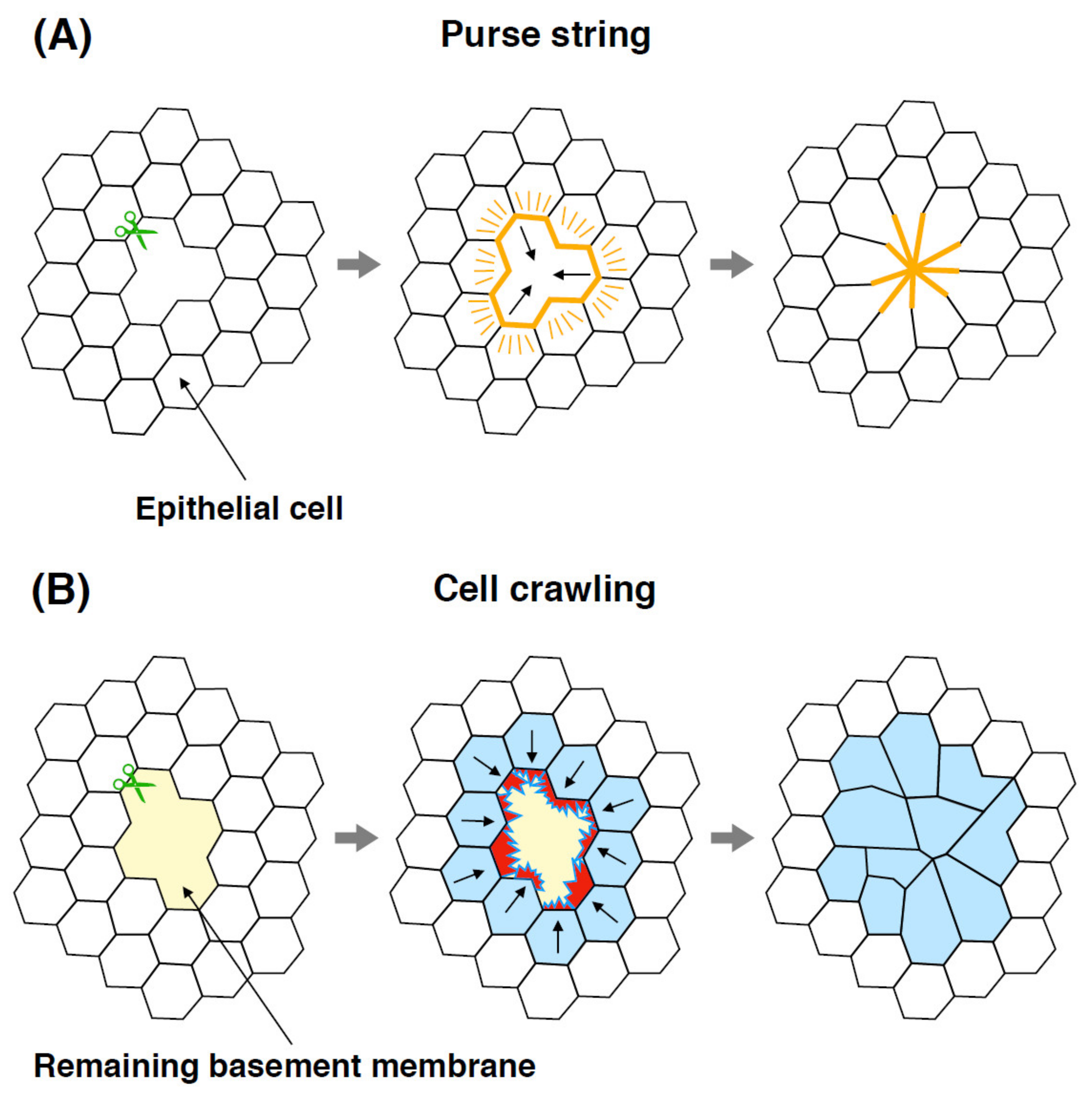
2. Wound Healing
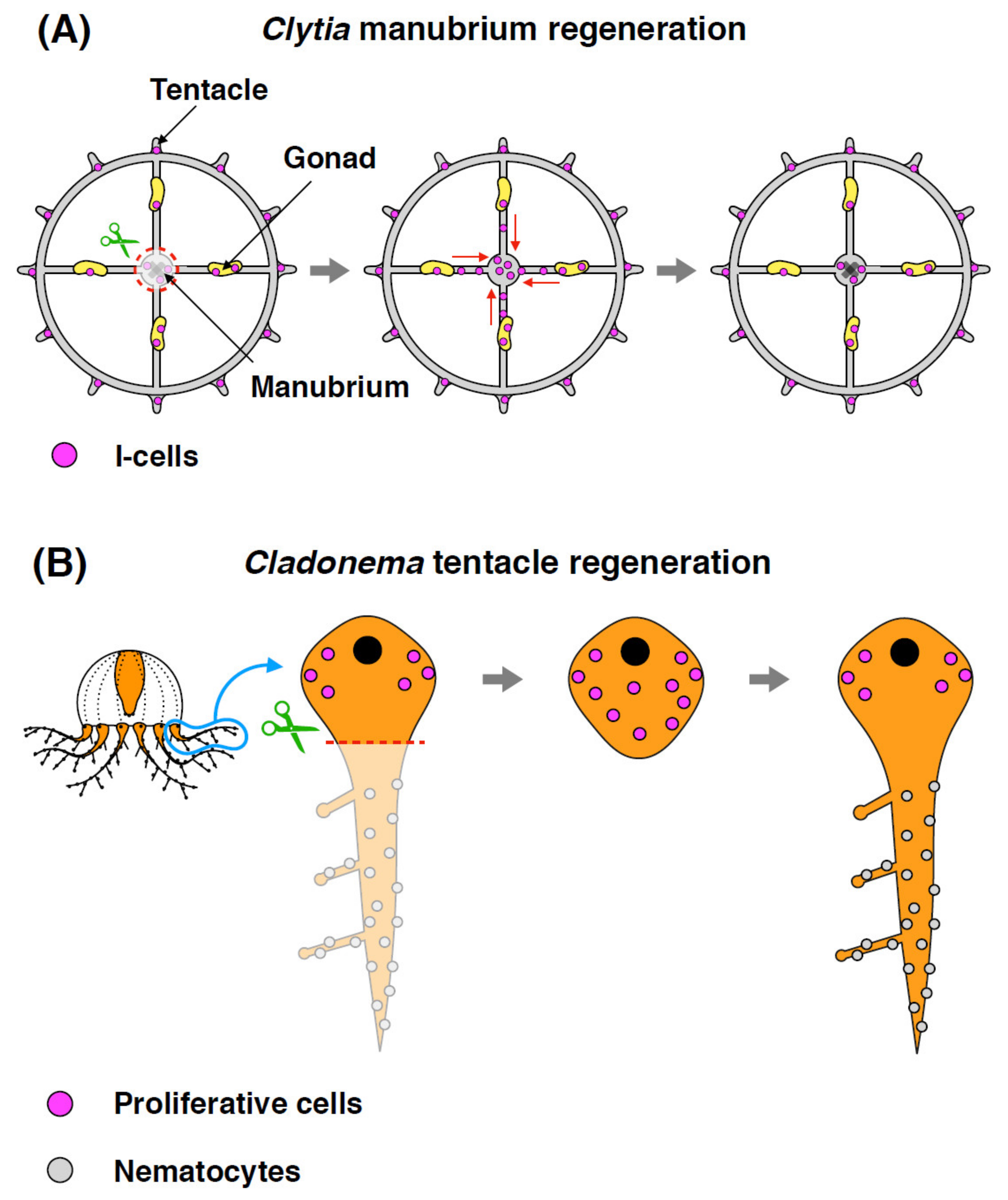
3. Stem/Progenitor Cell Proliferation
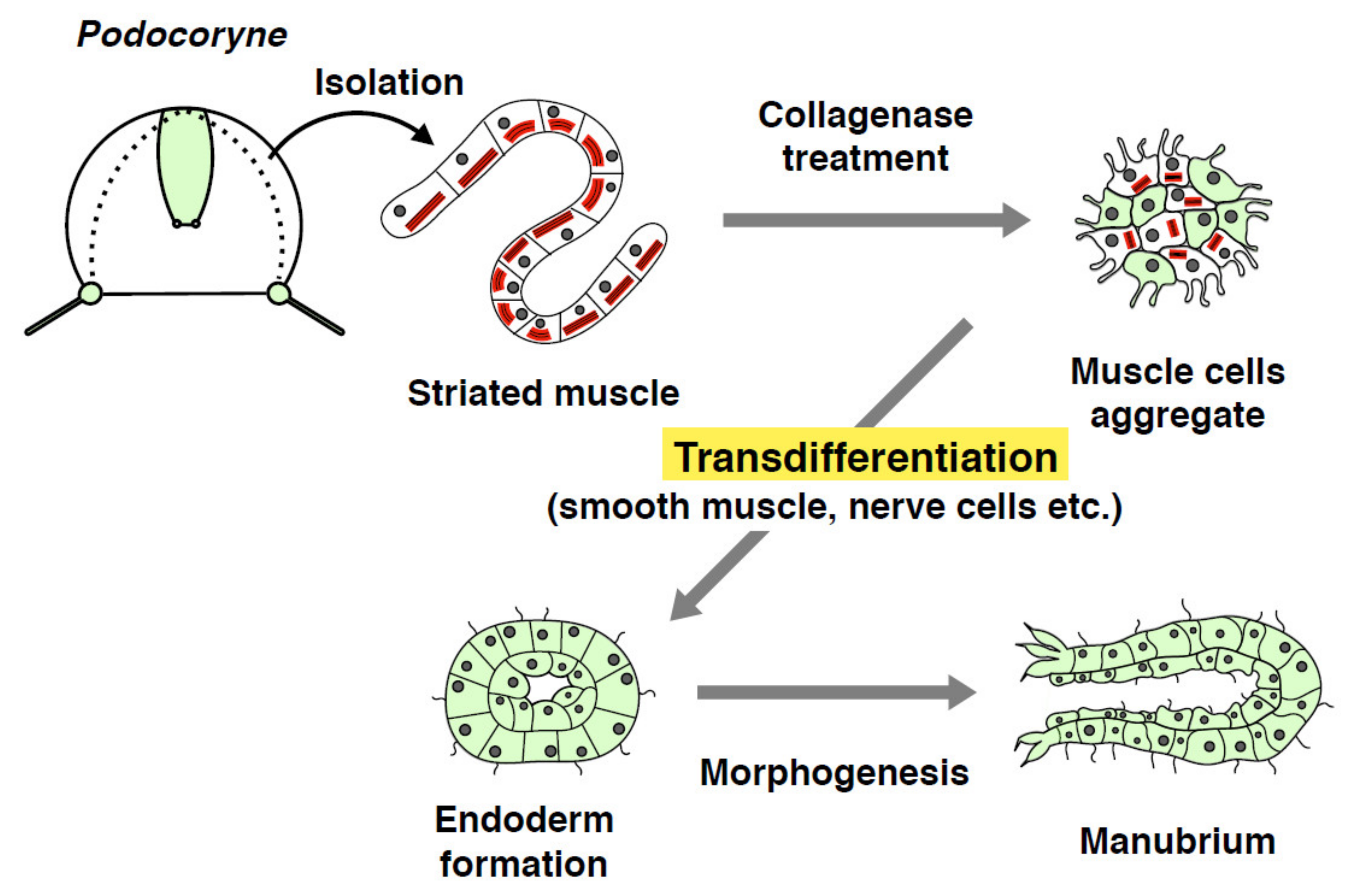
4. Transdifferentiation and Dedifferentiation
5. Patterning
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Technau, U.; Steele, R.E. Evolutionary crossroads in developmental biology: Cnidaria. Development 2011, 138, 1447–1458. [Google Scholar] [CrossRef] [PubMed]
- Kayal, E.; Roure, B.; Philippe, H.; Collins, A.G.; Lavrov, D.V. Cnidarian phylogenetic relationships as revealed by mitogenomics. BMC Evol. Biol. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Leclère, L.; Röttinger, E. Diversity of cnidarian muscles: Function, anatomy, development and regeneration. Front. Cell Dev. Biol. 2017, 4. [Google Scholar] [CrossRef]
- Martindale, M.Q. Investigating the origins of triploblasty: ‘Mesodermal’ gene expression in a diploblastic animal, the sea anemone Nematostella vectensis (phylum, Cnidaria; class, Anthozoa). Development 2004, 131, 2463–2474. [Google Scholar] [CrossRef]
- Galliot, B.; Schmid, V. Cnidarians as a model system for understanding evolution and regeneration. Int. J. Dev. Biol. 2002, 46, 39–48. [Google Scholar] [PubMed]
- Miles, J.G.; Battista, N.A. Naut your everyday jellyfish model: Exploring how tentacles and oral arms impact locomotion. Fluids 2019, 4, 169. [Google Scholar] [CrossRef]
- Solé, M.; Lenoir, M.; Fontuño, J.M.; Durfort, M.; Van Der Schaar, M.; André, M. Evidence of cnidarians sensitivity to sound after exposure to low frequency noise underwater sources. Sci. Rep. 2016, 6, 37979. [Google Scholar] [CrossRef]
- Weber, C. Structure, histochemistry, ontogenetic development, and regeneration of the ocellus of Cladonema radiatum dujardin (cnidaria, hydrozoa, anthomedusae). J. Morphol. 1981, 167, 313–331. [Google Scholar] [CrossRef]
- Stamatis, S.-A.; Worsaae, K.; Garm, A. Regeneration of the rhopalium and the rhopalial nervous system in the box jellyfish Tripedalia Cystophora. Biol. Bull. 2018, 234, 22–36. [Google Scholar] [CrossRef]
- Sinigaglia, C.; Peron, S.; Eichelbrenner, J.; Chevalier, S.; Steger, J.; Barreau, C.; Houliston, E.; Leclère, L. Pattern regulation in a regenerating jellyfish. ELife 2020, 9, e54868. [Google Scholar] [CrossRef]
- Fujita, S.; Kuranaga, E.; Nakajima, Y. Cell proliferation controls body size growth, tentacle morphogenesis, and regeneration in hydrozoan jellyfish Cladonema pacificum. PeerJ 2019, 7, e7579. [Google Scholar] [CrossRef] [PubMed]
- Schmid, V.; Tardent, P. The reconstitutional performances of the Leptomedusa Campanularia jonstoni. Mar. Biol. 1971, 8, 99–104. [Google Scholar] [CrossRef]
- Hargitt, C.W. Experimental studies upon hydromedusae. Biol. Bull. 1899, 1, 35–51. [Google Scholar] [CrossRef]
- Hargitt, G.T. Regeneration in hydromedusae. Arch. Entwickl. Org. 1903, 17, 64–91. [Google Scholar] [CrossRef][Green Version]
- Morgan, T.H. Regeneration in the hydromedusa, gonionemus vertens. Am. Nat. 1899, 33, 939–951. [Google Scholar] [CrossRef]
- Schmid, V.; Alder, H. Isolated, mononucleated, striated muscle can undergo pluripotent transdifferentiation and form a complex regenerate. Cell 1984, 38, 801–809. [Google Scholar] [CrossRef]
- Piraino, S.; De Vito, D.; Schmich, J.; Bouillon, J.; Boero, F. Reverse development in Cnidaria. Can. J. Zool. 2004, 82, 1748–1754. [Google Scholar] [CrossRef]
- Piraino, S.; Boero, F.; Aeschbach, B.; Schmid, V. Reversing the life cycle: Medusae transforming into polyps and cell transdifferentiation in Turritopsis nutricula (Cnidaria, Hydrozoa). Biol. Bull. 1996, 190, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Ceilley, R. Chronic wound healing: A review of current management and treatments. Adv. Ther. 2017, 34, 599–610. [Google Scholar] [CrossRef]
- Girault, A.; Brochiero, E. Evidence of K+ channel function in epithelial cell migration, proliferation, and repair. Am. J. Physiol. Cell Physiol. 2014, 306, C307–C319. [Google Scholar] [CrossRef]
- Martin, P. Parallels between tissue repair and embryo morphogenesis. Development 2004, 131, 3021–3034. [Google Scholar] [CrossRef]
- Kamran, Z.; Zellner, K.; Kyriazes, H.; Kraus, C.M.; Reynier, J.-B.; Malamy, J.E. In Vivo imaging of epithelial wound healing in the cnidarian Clytia hemisphaerica demonstrates early evolution of purse string and cell crawling closure mechanisms. BMC Dev. Biol. 2017, 17, 17. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.J.; Grigoriev, N.G.; Spencer, A.N. Wound healing in jellyfish striated muscle involves rapid switching between two modes of cell motility and a change in the source of regulatory calcium. Dev. Biol. 2000, 225, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Anon, E.; Serra-Picamal, X.; Hersen, P.; Gauthier, N.C.; Sheetz, M.P.; Trepat, X.; Ladoux, B. Cell crawling mediates collective cell migration to close undamaged epithelial gaps. Proc. Natl. Acad. Sci. USA 2012, 109, 10891–10896. [Google Scholar] [CrossRef] [PubMed]
- Ravasio, A.; Cheddadi, I.; Chen, T.; Pereira, T.; Ong, H.T.; Bertocchi, C.; Brugues, A.; Jacinto, A.; Kabla, A.J.; Toyama, Y.; et al. Gap geometry dictates epithelial closure efficiency. Nat. Commun. 2015, 6, 7683. [Google Scholar] [CrossRef]
- Farooqui, R.; Fenteany, G. Multiple rows of cells behind an epithelial wound edge extend cryptic lamellipodia to collectively drive cell-sheet movement. J. Cell Sci. 2005, 118, 51–63. [Google Scholar] [CrossRef]
- Klarlund, J.K. Dual modes of motility at the leading edge of migrating epithelial cell sheets. Proc. Natl. Acad. Sci. USA 2012, 109, 15799–15804. [Google Scholar] [CrossRef]
- Lesch, C.; Jo, J.; Wu, Y.; Fish, G.S.; Galko, M.J. A targeted UAS-RNAi screen in Drosophila larvae identifies wound closure genes regulating distinct cellular processes. Genetics 2010, 186, 943–957. [Google Scholar] [CrossRef]
- Tsai, C.-R.; Anderson, A.E.; Burra, S.; Jo, J.; Galko, M.J. Yorkie regulates epidermal wound healing in Drosophila larvae independently of cell proliferation and apoptosis. Dev. Biol. 2017, 427, 61–71. [Google Scholar] [CrossRef]
- DuBuc, T.Q.; Traylor-Knowles, N.; Martindale, M.Q. Initiating a regenerative response; cellular and molecular features of wound healing in the cnidarian Nematostella vectensis. BMC Biol. 2014, 12, 24. [Google Scholar] [CrossRef]
- Tursch, A.; Bartsch, N.; Holstein, T.W. MAPK signaling links the injury response to Wnt-regulated patterning in Hydra regeneration. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zakrzewski, W.; Dobrzyński, M.; Szymonowicz, M.; Rybak, Z. Stem cells: Past, present, and future. Stem Cell Res. Ther. 2019, 10, 68. [Google Scholar] [CrossRef]
- Gold, D.A.; Jacobs, D.K. Stem Cell Dynamics in Cnidaria: Are there unifying principles? Dev. Genes Evol. 2013, 223, 53–66. [Google Scholar] [CrossRef]
- Mochizuki, K.; Sano, H.; Kobayashi, S.; Nishimiya-Fujisawa, C.; Fujisawa, T. Expression and evolutionary conservation of nanos-related genes in Hydra. Dev. Genes Evol. 2000, 210, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, K.; Nishimiya-Fujisawa, C.; Fujisawa, T. Universal occurrence of the vasa-related genes among metazoans and their germline expression in Hydra. Dev. Genes Evol. 2001, 211, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Juliano, C.E.; Reich, A.; Liu, N.; Gotzfried, J.; Zhong, M.; Uman, S.; Reenan, R.A.; Wessel, G.M.; Steele, R.E.; Lin, H. PIWI proteins and PIWI-interacting RNAs function in Hydra somatic stem cells. Proc. Natl. Acad. Sci. USA 2014, 111, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Lim, R.S.M.; Anand, A.; Nishimiya-Fujisawa, C.; Kobayashi, S.; Kai, T. Analysis of Hydra PIWI proteins and piRNAs uncover early evolutionary origins of the piRNA pathway. Dev. Biol. 2014, 386, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Rebscher, N.; Volk, C.; Teo, R.; Plickert, G. The germ plasm component vasa allows tracing of the interstitial stem cells in the cnidarian Hydractinia echinata. Dev. Dyn. 2008, 237, 1736–1745. [Google Scholar] [CrossRef] [PubMed]
- Kanska, J.; Frank, U. New roles for Nanos in neural cell fate determination revealed by studies in a cnidarian. J. Cell Sci. 2013, 126, 3192–3203. [Google Scholar] [CrossRef] [PubMed]
- McMahon, E. Characterization of PIWI+ Stem Cells in Hydractinia. Ph.D. Thesis, NUI Galway, Galway, Ireland, 2017. Available online: https://aran.library.nuigalway.ie/handle/10379/7174 (accessed on 23 February 2018).
- Plickert, G.; Frank, U.; Müller, W.A. Hydractinia, a pioneering model for stem cell biology and reprogramming somatic cells to pluripotency. Int. J. Dev. Biol. 2012, 56, 519–534. [Google Scholar] [CrossRef]
- Chera, S.; Ghila, L.; Dobretz, K.; Wenger, Y.; Bauer, C.; Buzgariu, W.; Martinou, J.-C.; Galliot, B. Apoptotic cells provide an unexpected source of Wnt3 signaling to drive Hydra head regeneration. Dev. Cell 2009, 17, 279–289. [Google Scholar] [CrossRef]
- Passamaneck, Y.J.; Martindale, M.Q. Cell proliferation is necessary for the regeneration of oral structures in the anthozoan cnidarian Nematostella vectensis. BMC Dev. Biol. 2012, 12, 34. [Google Scholar] [CrossRef]
- Bradshaw, B.; Thompson, K.; Frank, U. Distinct mechanisms underlie oral vs. aboral regeneration in the cnidarian Hydractinia echinata. ELife 2015, 4, e05506. [Google Scholar] [CrossRef] [PubMed]
- Leclère, L.; Jager, M.; Barreau, C.; Chang, P.; Le Guyader, H.; Manuel, M.; Houliston, E. Maternally localized germ plasm mRNAs and germ cell/stem cell formation in the cnidarian Clytia. Dev. Biol. 2012, 364, 236–248. [Google Scholar] [CrossRef] [PubMed]
- Nusse, R. Wnt signaling and stem cell control. Cell Res. 2008, 18, 5. [Google Scholar] [CrossRef] [PubMed]
- Miki, T.; Yasuda, S.; Kahn, M. Wnt/β-catenin signaling in embryonic stem cell self-renewal and somatic cell reprogramming. Stem. Cell Rev. Rep. 2011, 7, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Polesskaya, A.; Seale, P.; Rudnicki, M.A. Wnt signaling induces the myogenic specification of resident CD45+ adult stem cells during muscle regeneration. Cell 2003, 113, 841–852. [Google Scholar] [CrossRef]
- Brock, C.K.; Wallin, S.T.; Ruiz, O.E.; Samms, K.M.; Mandal, A.; Sumner, E.A.; Eisenhoffer, G.T. Stem cell proliferation is induced by apoptotic bodies from dying cells during epithelial tissue maintenance. Nat. Commun. 2019, 10, 1044. [Google Scholar] [CrossRef]
- Vergara, M.N.; Tsissios, G.; Del Rio-Tsonis, K. Lens regeneration: A historical perspective. Int. J. Dev. Biol. 2018, 62, 351–361. [Google Scholar] [CrossRef]
- Jopling, C.; Sleep, E.; Raya, M.; Martí, M.; Raya, A.; Belmonte, J.C.I. Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 2010, 464, 606–609. [Google Scholar] [CrossRef]
- Siebert, S.; Anton-Erxleben, F.; Bosch, T.C.G. Cell type complexity in the basal metazoan Hydra is maintained by both stem cell based mechanisms and transdifferentiation. Dev. Biol. 2008, 313, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Schmid, V. Structural alterations in cultivated striated muscle cells from anthomedusae (Hydrozoa): A metaplasiaic event. Exp. Cell Res. 1974, 86, 193–198. [Google Scholar] [CrossRef]
- Schmid, V. The transformational potential of striated muscle in hydromedusae. Dev. Biol. 1976, 49, 508–517. [Google Scholar] [CrossRef]
- Schmid, V.; Wydler, M.; Alder, H. Transdifferentiation and regeneration in vitro. Dev. Biol. 1982, 92, 476–488. [Google Scholar] [CrossRef]
- Schmid, V.; Alder, H.; Plickert, G.; Weber, C. Transdifferentiation from striated muscle of medusae in vitro. Cell Differ. Dev. 1988, 25, 137–146. [Google Scholar] [CrossRef]
- Schmid, V. Striated muscle: Influence of an acellular layer on the maintenance of muscle differentiation in anthomedusa. Dev. Biol. 1978, 64, 48–59. [Google Scholar] [CrossRef]
- Alder, H.; Schmid, V. Cell cycles and in vitro transdifferentiation and regeneration of isolated, striated muscle of jellyfish. Dev. Biol. 1987, 124, 358–369. [Google Scholar] [CrossRef]
- Seipel, K.; Yanze, N.; Schmid, V. The germ line and somatic stem cell gene Cniwi in the jellyfish podocoryne carnea. Int. J. Dev. Biol. 2004, 48, 1–7. [Google Scholar] [CrossRef]
- Odelberg, S.J.; Kollhoff, A.; Keating, M.T. Dedifferentiation of mammalian myotubes induced by msx1. Cell 2000, 103, 1099–1109. [Google Scholar] [CrossRef]
- Galle, S.; Yanze, N.; Seipel, K. The homeobox gene Msx in development and transdifferentiation of jellyfish striated muscle. Int. J. Dev. Biol. 2005, 49, 961–967. [Google Scholar] [CrossRef]
- Seipel, K.; Yanze, N.; Schmid, V. Developmental and evolutionary aspects of the basic helix–loop–helix transcription factors Atonal-like 1 and Achaete-scute homolog 2 in the jellyfish. Dev. Biol. 2004, 269, 331–345. [Google Scholar] [CrossRef]
- Reber-Muller, S.; Streitwolf-Engel, R.; Yanze, N.; Schmid, V.; Stierwald, M.; Erb, M.; Seipel, K. BMP2/4 and BMP5-8 in jellyfish development and transdifferentiation. Int. J. Dev. Biol. 2006, 50, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Cowles, M.W.; Brown, D.D.R.; Nisperos, S.V.; Stanley, B.N.; Pearson, B.J.; Zayas, R.M. Genome-wide analysis of the BHLH gene family in planarians identifies factors required for adult neurogenesis and neuronal regeneration. Development 2013, 140, 4691–4702. [Google Scholar] [CrossRef] [PubMed]
- Wagner, I.; Wang, H.; Weissert, P.M.; Straube, W.L.; Shevchenko, A.; Gentzel, M.; Brito, G.; Tazaki, A.; Oliveira, C.; Sugiura, T.; et al. Serum proteases potentiate BMP-induced cell cycle re-entry of dedifferentiating muscle cells during newt limb regeneration. Dev. Cell 2017, 40, 608–617.e6. [Google Scholar] [CrossRef] [PubMed]
- Haensel, D.; Dai, X. Epithelial-to-mesenchymal transition in cutaneous wound healing: Where we are and where we are heading: EMT in cutaneous wound healing. Dev. Dyn. 2018, 247, 473–480. [Google Scholar] [CrossRef]
- Pellettieri, J. Regenerative tissue remodeling in planarians—The mysteries of morphallaxis. Semin. Cell Dev. Biol. 2019, 87, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Vogg, M.C.; Galliot, B.; Tsiairis, C.D. Model systems for regeneration: Hydra. Development 2019, 146. [Google Scholar] [CrossRef]
- Abrams, M.J.; Basinger, T.; Yuan, W.; Guo, C.-L.; Goentoro, L. Self-repairing symmetry in jellyfish through mechanically driven reorganization. Proc. Natl. Acad. Sci. USA 2015, 112, E3365–E3373. [Google Scholar] [CrossRef]
- Hobmayer, B.; Rentzsch, F.; Kuhn, K.; Happel, C.M.; Von Laue, C.C.; Snyder, P.; Rothbächer, U.; Holstein, T.W. WNT signalling molecules act in axis formation in the diploblastic metazoan Hydra. Nat. 2000, 407, 186–189. [Google Scholar] [CrossRef]
- Gufler, S.; Artes, B.; Bielen, H.; Krainer, I.; Eder, M.-K.; Falschlunger, J.; Bollmann, A.; Ostermann, T.; Valovka, T.; Hartl, M.; et al. β-catenin acts in a position-independent regeneration response in the simple eumetazoan Hydra. Dev. Biol. 2018, 433, 310–323. [Google Scholar] [CrossRef]
- Livshits, A.; Shani-Zerbib, L.; Maroudas-Sacks, Y.; Braun, E.; Keren, K. Structural inheritance of the actin cytoskeletal organization determines the body axis in regenerating Hydra. Cell Rep. 2017, 18, 1410–1421. [Google Scholar] [CrossRef]
- Petersen, C.P.; Reddien, P.W. Wnt signaling and the polarity of the primary body axis. Cell 2009, 139, 1056–1068. [Google Scholar] [CrossRef] [PubMed]
- Bier, E.; De Robertis, E.M. BMP gradients: A paradigm for morphogen-mediated developmental patterning. Science 2015, 348, aaa5838. [Google Scholar] [CrossRef] [PubMed]
- Yazawa, S.; Umesono, Y.; Hayashi, T.; Tarui, H.; Agata, K. Planarian Hedgehog/Patched establishes anterior–posterior polarity by regulating Wnt signaling. Proc. Natl. Acad. Sci. USA 2009, 106, 22329–22334. [Google Scholar] [CrossRef]
- Hueber, S.D.; Weiller, G.F.; Djordjevic, M.A.; Frickey, T. Improving HOX protein classification across the major model organisms. PLoS ONE 2010, 5, e10820. [Google Scholar] [CrossRef] [PubMed]
- Lengfeld, T.; Watanabe, H.; Simakov, O.; Lindgens, D.; Gee, L.; Law, L.; Schmidt, H.A.; Özbek, S.; Bode, H.; Holstein, T.W. Multiple Wnts are involved in Hydra organizer formation and regeneration. Dev. Biol. 2009, 330, 186–199. [Google Scholar] [CrossRef]
- Rentzsch, F.; Guder, C.; Vocke, D.; Hobmayer, B.; Holstein, T.W. An ancient chordin-like gene in organizer formation of Hydra. Proc. Natl. Acad. Sci. USA 2007, 104, 3249–3254. [Google Scholar] [CrossRef]
- Genikhovich, G.; Fried, P.; Prünster, M.M.; Schinko, J.B.; Gilles, A.F.; Fredman, D.; Meier, K.; Iber, D.; Technau, U. Axis patterning by BMPs: Cnidarian network reveals evolutionary constraints. Cell Rep. 2015, 10, 1646–1654. [Google Scholar] [CrossRef]
- Chen, C.-Y.; McKinney, S.A.; Ellington, L.R.; Gibson, M.C. Hedgehog signaling is required for endomesodermal patterning and germ cell development in the sea anemone Nematostella vectensis. ELife 2020, 9, e54573. [Google Scholar] [CrossRef]
- He, S.; Del Viso, F.; Chen, C.-Y.; Ikmi, A.; Kroesen, A.E.; Gibson, M.C. An axial HOX code controls tissue segmentation and body patterning in Nematostella vectensis. Science 2018, 361, 1377–1380. [Google Scholar] [CrossRef]
- Sinigaglia, C.; Averof, M. The multifaceted role of nerves in animal regeneration. Curr. Opin. Genet. Dev. 2019, 57, 98–105. [Google Scholar] [CrossRef]
- Lucas, C.H. Reproduction and life history strategies of the common jellyfish, Aurelia aurita, in relation to its ambient environment. Hydrobiologia 2001, 451, 229–246. [Google Scholar] [CrossRef]
- Wang, G.; Zhen, Y.; Yu, Z.; Shi, Y.; Zhao, Q.; Wang, J.; Mi, T. The physiological and molecular response of Aurelia sp.1 under hypoxia. Sci. Rep. 2017, 7, 1558. [Google Scholar] [CrossRef]
- Fuchs, B.; Wang, W.; Graspeuntner, S.; Li, Y.; Insua, S.; Herbst, E.-M.; Dirksen, P.; Böhm, A.-M.; Hemmrich, G.; Sommer, F.; et al. Regulation of polyp-to-jellyfish transition in Aurelia aurita. Curr. Biol. 2014, 24, 263–273. [Google Scholar] [CrossRef]
- Brekhman, V.; Malik, A.; Haas, B.; Sher, N.; Lotan, T. Transcriptome profiling of the dynamic life cycle of the scypohozoan jellyfish Aurelia aurita. BMC Genom. 2015, 16, 74. [Google Scholar] [CrossRef]
- Gold, D.A.; Katsuki, T.; Li, Y.; Yan, X.; Regulski, M.; Ibberson, D.; Holstein, T.; Steele, R.E.; Jacobs, D.K.; Greenspan, R.J. The genome of the jellyfish Aurelia and the evolution of animal complexity. Nat. Ecol. Evol. 2019, 3, 96–104. [Google Scholar] [CrossRef]
- Laurie-Lesh, G.E.; Corriel, R. Scyphistoma regeneration from isolated tentacles in Aurelia aurita. J. Mar. Biol. Ass. 1973, 53, 885–894. [Google Scholar] [CrossRef]
- Lesh-Laurie, G.E.; Hujer, A.; Suchy, P. Polyp regeneration from isolated tentacles of Aurelia scyphistomae: A role for gating mechanisms and cell division. Hydrobiologia 1991, 216, 91–97. [Google Scholar] [CrossRef]
- Ames, C.L.; Klompen, A.M.L.; Badhiwala, K.; Muffett, K.; Reft, A.J.; Kumar, M.; Janssen, J.D.; Schultzhaus, J.N.; Field, L.D.; Muroski, M.E.; et al. Cassiosomes are stinging-cell structures in the mucus of the upside-down jellyfish Cassiopea xamachana. Commun. Biol. 2020, 3, 67. [Google Scholar] [CrossRef]
- Ohdera, A.; Ames, C.L.; Dikow, R.B.; Kayal, E.; Chiodin, M.; Busby, B.; La, S.; Pirro, S.; Collins, A.G.; Medina, M.; et al. Box, stalked, and upside-down? Draft genomes from diverse jellyfish (Cnidaria, Acraspeda) lineages: Alatina Alata (Cubozoa), Calvadosia cruxmelitensis (Staurozoa), and Cassiopea xamachana (Scyphozoa). GigaScience 2019, 8. [Google Scholar] [CrossRef]
- Nath, R.D.; Bedbrook, C.N.; Abrams, M.J.; Basinger, T.; Bois, J.S.; Prober, D.A.; Sternberg, P.W.; Gradinaru, V.; Goentoro, L. The jellyfish Cassiopea exhibits a sleep-like state. Curr. Biol. 2017, 27, 2984–2990.e3. [Google Scholar] [CrossRef]
- Verde, E.; McCloskey, L. Production, respiration, and photophysiology of the mangrove jellyfish Cassiopea xamachana symbiotic with zooxanthellae: Effect of jellyfish size and season. Mar. Ecol. Prog. Ser. 1998, 168, 147–162. [Google Scholar] [CrossRef]
- Neumann, R. Polyp morphogenesis in a scyphozoan: Evidence for a head inhibitor from the presumptive foot end in vegetative buds of Cassiopeia andromeda. Wilhelm Roux Archiv. Dev. Biol. 1977, 183, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Cary, L.R. The influence of the marginal sense organs on metabolic activity in Cassiopea xamachana bigelow. Proc. Natl. Acad. Sci. USA 1916, 2, 709–712. [Google Scholar] [CrossRef]
- Stockard, C. Studies of tissue growth. Arch. Entwickl. Org. 1910, 29, 15–32. [Google Scholar] [CrossRef]
- Gamero-Mora, E.; Halbauer, R.; Bartsch, V.; Stampar, S.N.; Morandini, A.C. Regenerative capacity of the upside-down jellyfish Cassiopea xamachana. Zool. Stud. 2019, 58. [Google Scholar] [CrossRef]
- Garm, A.; O’Connor, M.; Parkefelt, L.; Nilsson, D. Visually guided obstacle avoidance in the box jellyfish Tripedalia cystophora and Chiropsella bronzie. J. Exp. Biol. 2007, 210, 3616–3623. [Google Scholar] [CrossRef]
- Coates, M.M.; Garm, A.; Theobald, J.C.; Thompson, S.H.; Nilsson, D.-E. The spectral sensitivity of the lens eyes of a box jellyfish, Tripedalia cystophora (conant). J. Exp. Biol. 2006, 209, 3758–3765. [Google Scholar] [CrossRef]
- Petie, R.; Garm, A.; Nilsson, D.-E. Visual control of steering in the box jellyfish Tripedalia cystophora. J. Exp. Biol. 2011, 214, 2809–2815. [Google Scholar] [CrossRef]
- Petie, R.; Garm, A.; Nilsson, D.-E. Contrast and rate of light intensity decrease control directional swimming in the box jellyfish Tripedalia cystophora (Cnidaria, Cubomedusae). Hydrobiologia 2013, 703, 69–77. [Google Scholar] [CrossRef][Green Version]
- Piatigorsky, J.; Kozmik, Z. Cubozoan jellyfish: An evo/devo model for eyes and other sensory systems. Int. J. Dev. Biol. 2004, 48, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Koenig, K.M.; Gross, J.M. Evolution and development of complex eyes: A celebration of diversity. Development 2020, 147, dev182923. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.K.D.; Koch, T.L.; Hauser, F.; Garm, A.; Grimmelikhuijzen, C.J.P. De novo transcriptome assembly of the cubomedusa Tripedalia cystophora, including the analysis of a set of genes involved in peptidergic neurotransmission. BMC Genom. 2019, 20, 175. [Google Scholar] [CrossRef] [PubMed]
- Houliston, E.; Momose, T.; Manuel, M. Clytia hemisphaerica: A jellyfish cousin joins the laboratory. Trends Genet. 2010, 26, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Lechable, M.; Jan, A.; Duchene, A.; Uveira, J.; Weissbourd, B.; Gissat, L.; Collet, S.; Gilletta, L.; Chevalier, S.; Leclère, L.; et al. An improved whole life cycle culture protocol for the Hydrozoan genetic model Clytia hemisphaerica. Biol. Open 2020, 9. [Google Scholar] [CrossRef]
- Momose, T.; De Cian, A.; Shiba, K.; Inaba, K.; Giovannangeli, C.; Concordet, J.-P. High doses of CRISPR/Cas9 ribonucleoprotein efficiently induce gene knockout with low mosaicism in the hydrozoan Clytia hemisphaerica through microhomology-mediated deletion. Sci. Rep. 2018, 8, 11734. [Google Scholar] [CrossRef]
- Leclère, L.; Horin, C.; Chevalier, S.; Lapébie, P.; Dru, P.; Peron, S.; Jager, M.; Condamine, T.; Pottin, K.; Romano, S.; et al. The genome of the jellyfish Clytia hemisphaerica and the evolution of the cnidarian life-cycle. Nat. Ecol. Evol. 2019, 3, 801–810. [Google Scholar] [CrossRef]
- Chari, T.; Weissbourd, B.; Gehring, J.; Ferraioli, A.; Leclère, L.; Herl, M.; Gao, F.; Chevalier, S.; Copley, R.R.; Houliston, E.; et al. Whole animal multiplexed single-cell RNA-seq reveals plasticity of Clytia medusa cell types. bioRxiv 2021. [Google Scholar] [CrossRef]
- Weissbourd, B. Functional modules within a distributed neural network control feeding in a model medusa. bioRxiv 2021. [Google Scholar] [CrossRef]
- Fujiki, A. Branching pattern and morphogenesis of medusa tentacles in the jellyfish Cladonema pacificum (Hydrozoa, CNIDARIA). Zool. Lett. 2019, 5, 13. [Google Scholar] [CrossRef]
- Suga, H.; Tschopp, P.; Graziussi, D.F.; Stierwald, M.; Schmid, V.; Gehring, W.J. Flexibly deployed pax genes in eye development at the early evolution of animals demonstrated by studies on a hydrozoan jellyfish. Proc. Natl. Acad. Sci. USA 2010, 107, 14263–14268. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, R.; Kondoh, E.; Itoh, J. Spatiotemporal characteristics and mechanisms of intracellular Ca2+ increases at fertilization in eggs of jellyfish (Phylum Cnidaria, Class Hydrozoa). Dev. Biol. 2005, 279, 291–307. [Google Scholar] [CrossRef]
- Takeda, N.; Kon, Y.; Artigas, G.Q.; Lapébie, P.; Barreau, C.; Koizumi, O.; Kishimoto, T.; Tachibana, K.; Houliston, E.; Deguchi, R. Identification of jellyfish neuropeptides that act directly as oocyte maturation-inducing hormones. Development 2018, 145. [Google Scholar] [CrossRef] [PubMed]
- Kakinuma, Y. On the differentiation of the isolated medusa bud of the hydrozoans, Cladonema uchidai and Cladonema sp. Bull. Mar. Biol. Stn. Asamushi 1969, 13, 169–172. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujita, S.; Kuranaga, E.; Nakajima, Y.-i. Regeneration Potential of Jellyfish: Cellular Mechanisms and Molecular Insights. Genes 2021, 12, 758. https://doi.org/10.3390/genes12050758
Fujita S, Kuranaga E, Nakajima Y-i. Regeneration Potential of Jellyfish: Cellular Mechanisms and Molecular Insights. Genes. 2021; 12(5):758. https://doi.org/10.3390/genes12050758
Chicago/Turabian StyleFujita, Sosuke, Erina Kuranaga, and Yu-ichiro Nakajima. 2021. "Regeneration Potential of Jellyfish: Cellular Mechanisms and Molecular Insights" Genes 12, no. 5: 758. https://doi.org/10.3390/genes12050758
APA StyleFujita, S., Kuranaga, E., & Nakajima, Y.-i. (2021). Regeneration Potential of Jellyfish: Cellular Mechanisms and Molecular Insights. Genes, 12(5), 758. https://doi.org/10.3390/genes12050758





