1. Introduction
Kabuki syndrome (KS) is characterized by distinct facial dysmorphism, growth retardation, psychomotor developmental delay and a wide spectrum of other manifestations affecting various body systems. Its genetic etiology has been proven approximately a decade ago [
1]. Since then, KS has been increasingly observed in the family medicine and pediatrics setting. This paper reviews the current clinical knowledge, provides molecular and scientific links and sheds light on the treatment as well as quality of life of Kabuki syndrome individuals.
KS was first described in the early 1980s in Japan. It was named after the characteristic facial features which resemble the makeup of actors in the Kabuki theater in Tokyo. Initially, this syndrome was thought to be specific to the East Asian race; however, new data do not demonstrate clear prevalence of KS in any ethnic population. Total prevalence is estimated at 1 in 32 thousand [
1,
2]. The clinical diagnosis of KS often requires long-term monitoring because the phenotype changes over time—characteristic dysmorphism and other cardinal features tend to appear after several years of life. Prenatal, neonatal and family history is often unremarkable. Facial dysmorphism presence may be enough to raise suspicion of KS, after which genetic diagnostics must be performed to ultimately confirm KS. Usually, this is performed using Next-Generation Sequencing or Sanger Sequencing (more commonly in the past) [
3]. These techniques aim to identify
KMT2D or
KDM6A pathogenic/likely pathogenic variants (the genes responsible for KS). Alternatively, Whole-Exome Sequencing can be used [
4]. Even so, in some cases no genetic etiology is found in patients whose clinical picture is consistent with KS. In such patients, either an atypical clinical picture is concluded, or a different syndrome is diagnosed. Such cases are described in other parts of this review.
International consensus diagnostic criteria for KS were established in 2019. These aimed to facilitate the process of diagnosing KS. It was concluded that a definite diagnosis of KS can be made in patients of any age with a history of infantile hypotonia, developmental delay and at least one of the major criteria:
A pathogenic or a likely pathogenic variant in KMT2D or KDM6A.
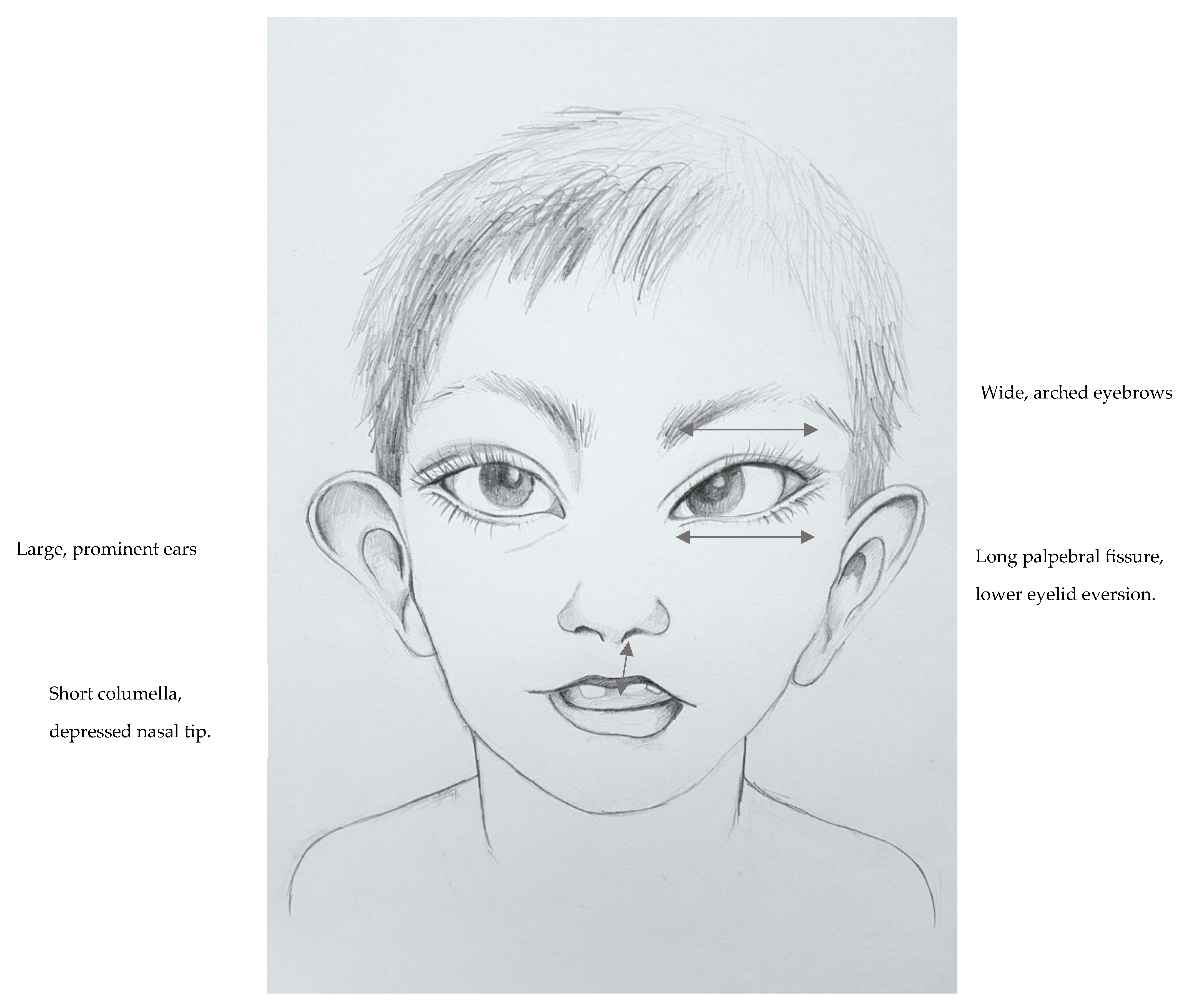
Typical dysmorphic features including long palpebral features, eversion of the lower eyelid and two more of:
- a
Arched and broad eyebrows, with notching or sparseness
- b
Short columella and depressed nasal tip
- c
Large, prominent or cupped ears
- d
Persistent fingertip pads
A list of supportive clinical features was agreed upon, including short stature, microcephaly, cleft palate, lip pits, hearing loss, congenital heart defects, feeding difficulties and immunological disorders. A probable diagnosis can be made in a patient with a history of infantile hypotonia, developmental delay and at least three of the supportive clinical features, and a possible diagnosis can be made in a patient with two of the supportive clinical features. These criteria may need to be expanded in the future; however, the authors argue that their use should already be implemented in order to make the KS diagnostic process universal [
5].
In the next chapters, we analyze the clinical characteristics of KS with regard to growth and endocrinology, neurology, cardiology, gastroenterology, laryngology, ophthalmology, oncology, nephrology and immunology. We also examine the effect of the KS molecular background on the clinical picture.
2. Molecular Genetics
In 2010, Ng et al. performed the sequencing of exomes of 10 unrelated patients with Kabuki syndrome (KS) diagnosed clinically based on a specific scoring system. They demonstrated that
KMT2D variants underlie Kabuki syndrome [
3].
KMT2D is also known as MLL2 (the latter name is considered outdated) [
6]. Two years later, Lederer et al. described three patients with deletions encompassing the
KDM6A gene on the X-chromosome. They claimed that
KDM6A, which normally escapes inactivation, would be more likely expressed from an active X chromosome, so that the deletion of
KDM6A would manifest itself in the affected individuals [
7]. Miyake et al. then demonstrated that the point variants in
KDM6A also cause KS. They examined 32 patients with KS diagnosed clinically who were negative for
KMT2D variants. Two patients had nonsense
KDM6A variants while one had a three base-pair deletion in
KDM6A [
8]. UTY is a homologue of
KDM6A on the Y-chromosome. In the past, UTY was thought to have lost its demethylase activity and to be non-functional; however, knockout-mice models suggest that the gene is expressed in a cell. Female mice with homozygous
KDM6A deletion do not survive, but hemizygous males do, as this is thought to be due to the UTY gene function. This suggests that UTY does play a role similar to
KDM6A in metabolism and development [
9].
The
KMT2D protein is a histone (H3) lysine methyltransferase protein, while
KDM6A acts as a histone demethylase. Both proteins have complementary functions.
KMT2D is responsible for cell-type specific gene expression during differentiation. It functions to trimethylate histone-3-lysine-4, opening the chromatin and activating homeobox and NESTIN genes during cell differentiation.
KDM6A demethylates histone-3-lysine-27, closing the chromatin. Both genes affect the opening of chromatin and promote gene expression. Dosage sensitivity may be important in Kabuki syndrome. The switch between open and closed chromatin affects the access of transcriptional machinery to target genes, which may lead to the development of the disease [
10]. Pathogenic/likely pathogenic variants seen in KS tend to reduce
KMT2D’s catalytic activity. This in turn leads to an increase in methylated histone levels, which decreases homeobox gene expression, thus affecting global cell differentiation [
11]. Building on this microbiological knowledge, Lee et al. demonstrated that a deletion in
KMT2D decreases mediator and polymerase levels, which leads to defects in cell-type specific gene expression and cell differentiation.
KMT2D was identified as a mono and di-methyltransferase, which is essential for enhancer activation during cell differentiation [
6]. Drug-induced balance-restoration that promotes open-chromatin (for example, with histone-deacetylase inhibitors) may be considered as a novel KS treatment. Drug administration in mice normalized structural and functional deficits in dentate gyrus function after H3K4 trimethylation [
10].
In humans, the
KMT2D gene is located on chromosome 12. It is over 19 kilobase pairs in length and contains 54 exons.
KMT2D codes for the enhancer histone-3-lysine-4 methyltransferase protein, one of six KMT proteins working as part of a chromatin modifier protein complex. Enhancers aid in gene expression regulation. They are often bound to transcription factors.
KMT2D selectively binds to specific regions during various cell differentiation stages, activating gene expression depending on the differentiation stage [
12]. A recently published Xenopus frog model provides evidence that
KMT2D loss-of-function variants inhibit neural crest development, suggesting that KS be considered a neurocristopathy. The expression of
KMT2D in the neural crest during pre-migratory as well as migratory stages was found.
KMT2D may be required for neural crest cell differentiation and consequent migration [
13]. Cell-autonomous proliferation and cell cycle defects along with early neural stem cell progenitor maturation in an in vitro and mouse model were confirmed.
KMT2D-caused transcription suppression led to the activity of hypoxia-response pathways. This proves that the loss of
KMT2D function suppresses oxygen-responsive gene programs crucial for neural progenitor maintenance, ultimately leading to precocious neuronal differentiation [
14]. A recent zebrafish model aimed to analyze the role of
KMT2D and
KDM6A in the development of tissue abnormalities, including craniofacial, heart and brain deformities. Variants in both
KMT2D and
KDM6A lead to improper cell differentiation, ultimately causing a characteristic dysmorphism and developmental delay [
15]. A fluorescence in situ hybridization and Whole Genome Sequencing study was performed, analyzing a female KS patient with a known
KDM6A variant and a phenotype including hypotonia, developmental delay, short stature, microcephaly, seizures, facial dysmorphism and cleft palate.
KDM6A expression is significantly reduced in neural crest cells, leading to delayed neural development. The dosage sensitivity of
KDM6A is associated with characteristic Kabuki syndrome dysmorphism.
KMT2D and
KDM6A proteins regulate the differentiation of mesenchymal cell lineages: myoblasts and osteoblasts. Mouse models have shown that lysine demethylase plays a crucial role in regulating mesenchymal lineage cell differentiation. Loss-of-function variants in lysine demethylase genes cause improper development of cells in this lineage and, presumably, several clinical neurological abnormalities that have been seen in KS patients. The anomalies present in KS children were also observed in mice with a loss-of-function
KMT2D variant and brain development disorders: hypotonia, enhanced activity (corresponding to hyperactivity in KS children), a decreased auricular reflex (corresponding to hearing impairment) and motor coordination dysfunction [
16].
KMT and lysine demethylase proteins’ specific role in the regulation of myoblast differentiation was also recently examined using the
C2C12 myoblast model system.
KDM1A is the only one crucial for myogenic differentiation, while
KDM6A (among others) is required for osteoblast differentiation.
KDM1A, via histone-demethylation, represses the regulator of osteoblast differentiation, thus promoting myogenesis.
KMT2D, however, is also required for osteoblast differentiation by the demethylation of a different receptor. These mechanisms of histone modification (lysine methylation) act as a signal, thus impacting the fate, rate and process of cell differentiation [
17].
The effect of the deletion in the
KMT2D gene of the region encoding for methyltransferase (thus leading to the impairment of its function) resulted in impaired histone acetylation and methylation, eventually leading to a deficiency of the dentate gyrus granule cell layer and to reduced neurogenesis and hippocampal memory defects. A deficiency in postnatal neurogenesis directly results in the
KMT2D defect as the base for intellectual disability in KS [
10].
KMT2D variant demonstrated sensory and psychomotor impairments, including hypotonia and impaired reflexes (suggesting impaired neurological function) and a reduction in the auricular reflex (suggesting hearing impairment) in mouse mutants. Increased general activity with decreased motor coordination, increased rearing and grooming and increased distance traveled and speed were observed. This study builds on Bjornsson et al.’s findings by presenting the
KMT2D variant phenotype as more complex. This mouse model may shed light onto the wide range of psychomotor and behavioral impairments associated with KS in humans [
16].
Missense
KMT2D variant s likely cause reduced histone methylation due to impaired WRAD protein complex formation [
18]. They may result in a novel multiple-malformation syndrome [
19]. Notable clinical differences may be observed: no patient with such variant s presented with intellectual disability, clefting, renal abnormalities or seizures, while six patients presented with hypothyroidism. Missense variants may cause a disturbance in the
KMT2D secondary structure through increased alpha-helical structure (coiled protein shape leading to dysfunction).
3. Growth and Endocrinology
Growth pattern in the pediatric population is dependent on several factors. These can be divided into intrinsic factors and external factors. Intrinsic factors (influenced by genetic factors) largely result from constitutive and familial growth or the presence of major congenital anomalies. External factors are shaped by prenatal care, labor mode, adaptation period, hormonal characteristics, feeding and nutrition during the first year of life and later into adolescence. On top of that, syndromic diagnosis by itself seems to carry important contributions to the growth pattern. Early observations of physical characteristics in Kabuki syndrome by Niikawa et al. suggested that birth weight and length were normal with ensuing postnatal growth retardation [
20].
KS patients commonly present with failure to thrive during infancy, feeding problems and reflux, with 65–74% of patients even requiring nasogastric tube insertion or gastrostomy. Conversely, during late childhood these children commonly present with obesity. Awareness and intervention by a dietician should be considered [
2].
Postnatal growth retardation was present in 50–70% of patients described in ensuing studies. Prenatal growth retardation was observed in a minority of analyzed cases and postnatal growth retardation was observed in the absolute majority, regardless of ethnic origins. [
21]. In a review of 300 patients, birth weight and length were generally normal, with significant growth delay starting as early as the first year of life. Short stature was present in 75% of patients. This is consistent with previous reports of mean heights of over two standard deviations below average. Growth hormone deficiencies are present in a few cases [
22].
KS children are generally born at term and with a normal birth weight. In one study, not one individual had a birth weight under the 3rd centile. Failure to thrive and feeding problems are observed in two thirds of patients. During the fifth year of life, progression to obesity is prevalent in up to 57% of patients [
23].
Up to 70% of KS patients present with short stature. This has been suggested to be directly caused by the
KMT2D variant [
4]. Genotype-phenotype correlations revealed a significantly higher frequency of short stature in patients harboring a
KMT2D variant than in those without it (
p-value < 0.0001). Patients with a mosaic
KMT2D variant less frequently presented with short stature. Growth Hormone -deficiency is the most common endocrinological finding in KS patients. Endocrinological evaluation and even the possibility of rh-GH-therapy for KS-patients may be beneficial [
4].
Indications for rh-GH-therapy in pediatric patients include confirmed GH-deficiency, Chronic Kidney Disease, Turner syndrome, Intrauterine growth restriction/Small for gestational age infants who fail to catch up to normal growth centiles, Prader-Willi syndrome, idiopathic short stature, SHOX gene haploinsufficiency and Noonan syndrome (USA) [
24]. Genetic syndromes without GH-deficiency (as in many cases of KS) can be treated with rh-GH-therapy as a trial.
The largest study to date by Schott et al. in 2017 analyzed the effect of growth hormone treatment on linear height and body proportions in KS patients. The result was a statistically significant linear height increase after one year’s worth of rh-GH-therapy. Children who received GH-treatment at a younger age tended to display greater height increases than those who started at an older age. No significant difference was described between the
KMT2D versus the
KDM6A group. Additionally, throughout the treatment course, patients’ body proportions were not affected [
25].
Normative growth curves especially designed for children with KS based on the largest cohorts to date were recently created. They reaffirmed observations by Schott et al. in 2017 that
KMT2D pathogenic variants showed growth patterns roughly two standard deviations below the norm, enabling growth monitoring using growth charts. It is crucial not to miss a separate non-syndromic etiology of disrupted growth (e.g., GH-deficiency, which can be observed in 2–22% of KS individuals). The limitation is that it is difficult to predict whether final KS patient growth rate will catch up to that of children not affected by KS. The normal distribution of Body mass index BMI in KS patient groups is much wider than in the general population, which suggests the increased presence of excess weight or even obesity in KS patients [
26]. This obesity has been observed starting at the age of 4. This is consistent with findings in other studies [
23,
27].
There is support in the literature for evidence of a link between molecular genetics mechanisms and growth pattern in KS. First, a balanced interplay between gonadal and adrenal steroid hormones and GH is essential for the pubertal growth spurt. Patients with a deficiency in any of these hormones experience a deficient growth spurt.
During puberty, estradiol promotes the epiphyseal fusion essential for bone growth and thus directly affects adult height in both sexes. Ligand-dependent estrogen-receptor-α acts as a mediator in estrogen-sensitive tissues. Molecularly,
KMT2D is essential for a ligand-dependent estrogen-receptor-α activation [
28]. More details are in the
Molecular Genetics section.
Decreased growth associated with decreased serum levels of Insulin-like growth factor-1 has been described in mouse models. The GH to IGF-I axis is highly active during puberty. The effect of estrogen on this axis has been theorized to happen through the direct action of testosterone/androgen receptors, or through the indirect action of estrogens on estrogen-receptor-α. This suggests a biological mechanism for a significantly diminished pubertal growth spurt in KS. Finally, testosterone stimulates GH-secretion in puberty and throughout adulthood. This effect depends on the level of aromatization of testosterone within estrogen [
29]. Perhaps a
KMT2D variant causes a decrease in the GH/IGF-I axis activity, and therefore a diminished growth spurt and short stature.
KS mice phenotypes are comprised of skeletal growth retardation and decreased length and weight as a cause of shortened long bones [
30]. These findings are also seen in KS patients [
31]. The
KMT2D and
KDM6A gene variant influences endochondral ossification at long bone growth plates by disrupting the estrogen receptors in developing skeletal tissues. Sex-based differences are not only dependent on hindered estrogen receptor activity as a result of gene variant. SOX9 has been linked to the pathogenesis of the KS phenotype by describing disrupted endochondral bone formation. In theory,
KMT2D allows SOX9 to become uninhibited, thus activating chondrocyte differentiation (SOX9 being a well-described chondrocyte-differentiation mediator). Variants in
KMT2D prevent this process from taking place. SOX9 has also been described as involved in inhibiting osteoblast differentiation. Abnormal SOX9 expression (for instance, as a result of
KMT2D variant) prevents the differentiation of chondrocytes into osteoblasts, ultimately causing too many chondrocytes and too few osteoblasts in the skeletal system. This probably causes improper growth, specifically growth retardation in KS (shortening of long bones and ineffective bone formation) [
30].
A wide variety of hormonal abnormalities can occur as a result of KS. Kabuki syndrome has been recently associated with hyperinsulinism and thus to transient neonatal or infantile hypoglycemia. Persistent hypoglycemia, however, is uncommon. KS patients with a
KDM6A variant have an increased risk of hyperinsulinism (and thus hypoglycemic episodes) than those with
KMT2D pathogenic variants [
32].
KDM6A demethylation of the H3K27 protein may lead to the deregulation of β-cell development [
33].
KDM6A codes for a demethylase that acts on H3K27me3/me2 and induces a steady-state in proliferating cells [
34]. Most actively these domains increase in number within endocrine pancreatic cells, which leads to disorders of pancreatic β-cell development.
It is hypothesized that KS is associated with excess gonadotropin secretion, which causes premature thelarche in 41% of patients [
2]. One patient exhibited delayed thelarche (constitutional delay of puberty) along with other endocrine symptoms (obesity, congenital hypothyroidism and GH-deficiency). This may be rooted in estrogen-receptor downregulation as a result of KS. The majority of patients present with short stature as a direct result of GH deficiency [
2]. Additionally, KS has also been linked to combined pituitary hormone deficiency, rooted in the deletion of polyalanine tracts. One Japanese male KS patient with combined pituitary hormone deficiency was reported. Genetic tests identified a novel hemizygous 21-base pair deletion in this patient, resulting in the loss of seven alanine residues from the polyalanine tracts in the SOX3 gene. This suggests that deletions in the polyalanine tracts can cause hypopituitarism in these patients [
35].
5. Cardiovascular Issues in Kabuki Syndrome
Cardiological manifestations are cardinal features of Kabuki syndrome. The three most commonly observed cardiac defects in KS patients are atrial septal defect, (ASD), ventricular septal defect (VSD) and coarctation of aorta (CoA) [
2]. An analysis of specific congenital heart disease (CHD) manifestations in a 60 patient cohort was conducted. CHD was diagnosed in 58% of them, mostly in males. While analyzing for specific CHD, significant male predominance was observed in CoA cases, while no sex-related predominance was observed in VSD patients [
68]. Rarer cardiac defects include aortic stenosis, pulmonary stenosis, tetralogy of Fallot (ToF), double-outlet right ventricle, complete transposition and Ebstein’s anomaly [
69].
Up to 80% of KS patients with
KMT2D variants present with CHD [
70]. CoA is often described in the literature in the context of KS. Juxtaductal CoA is the most common subtype observed in KS patients, sometimes considered a cardinal feature of KS [
71]. CoA may be present with or without [
69] other heart defects such as anomalous left pulmonary artery [
72] or left ventricular diverticulum [
73]. Taking the prevalence of different CHD types into account, a predominance of left-sided obstructive lesions can be observed (nearly half of all patients), e.g., Shone complex type of anomalies [
70]. Together, aortic coarctation and septal defects are the most common CHD in patients with
KMT2D variants (nearly half of patients). Of septal defects, VSD and ASD-II are predominant. Bicuspid aortic valve has been reported in 1/8 of patients. Mitral stenosis, hypoplastic left heart syndrome, conotruncal defects, cardiomegaly, tetralogy of Fallot and other rare syndromes have been reported in under 5% of patients [
70].
KMT2D’s role in cardiogenesis has been demonstrated in mice.
KMT2D deletions lead to decreased histone-3-lysine-4 methyltransferase expression at enhancers and promoters of cardiogenesis. This results in the downregulation of ion transport and cell cycle genes. Moreover,
KMT2D-bound regions within cardiomyocytes have been defined [
74]. Variants cause decreased gene expression in
KMT2D knockdown mice hearts [
74]. Disorders of ion transport, hypoxia-reoxygenation and cell cycle regulation were observed. While analyzing the development of congenital cardiac abnormalities, a single functional copy of
KMT2D may be enough for normal heart function. A deletion of both copies leads to general disruption of myocardial development [
74]. On the contrary, a newborn with a de novo
KMT2D heterozygous frameshift deletion presented with VSD as well as a myriad of other cardinal KS features. Good general condition and prognosis was seen, and surgical correction of cardiological abnormalities was performed. This highlights the importance of a case-by-case surgical treatment approach to KS patients as well as genotype-phenotype-driven diagnostics [
75]. Knockdown
Xenopus frogs develop hypoplastic hearts with defective chamber development. Cardiomyocyte differentiation is severely affected by a loss of
KMT2D function.
KMT2D is required for the development of primary and secondary heart fields [
76]. A link between the Notch pathway and
KMT2D during endothelium and endocardium development has been suggested in zebrafish. Notch pathway inhibition leads to physiological cardiovascular development. Extrapolating on this finding,
KMT2D may play a role in regulating vasculogenesis and angiogenesis (connective tissue development) [
77]. Yet,
KMT2D’s function as a cardioregulatory gene in humans is not yet fully understood, nor is its role in the development of specific congenital cardiac diseases [
74,
76,
77]. Fewer than half of patients with
KDM6A variants present with CHD, and those who do show mostly right-sided lesions [
70]. Mouse models presented with abnormal atrial and/or ventricular development and myocardial wall bulging defects in heterozygous as well as homozygous subjects [
15,
70].
Surgery is commonly indicated to treat congenital cardiac anomalies in KS patients. Usually, KS patients tolerate the surgical correction of CoA well, and cardiological prognosis is positive [
71,
73,
78]. Similarly, ASD may be repaired using minimally invasive robotic techniques, further improving KS patient postoperative quality of life [
79]. Indications for surgical intervention depend on the general condition and prognosis of the child. No special considerations based on
KMT2D variants have been suggested [
75].
Underlying connective tissue diseases may ultimately lead to cardiac defects in KS patients. Connective tissue developmental disorder in the context of KS has been proposed for decades, mostly while describing hip, knee and shoulder joint hypermobility in patients with
KMT2D variants [
2,
80]. Digilio et al. discuss clinical overlaps between KS male patients and Turner syndrome patients since CoA is the most common congenital heart defect present in Turner syndrome [
68]. Perhaps a similar pathomechanism leads to the development of CoA in KS patients. KS patients may be predisposed to express connective tissue abnormalities. The management of these anomalies is conservative because, when compared with anticoagulation therapy, no difference in effectivity has been demonstrated [
80]. Great vessel pathologies, including aortic and/or main pulmonary artery aneurysms [
81] and double aortic arch [
82], may arise from connective tissue disease associated with KS. These are usually successfully managed with aortoplasty [
81].
Systemic vascular abnormalities in the context of KS have been recently reported. Gatto et al. describe a case of a KS patient who suffered a transient ischemic stroke due to occlusion of the right internal carotid artery. Despite the fact that cardiac and great vessel anomalies are common (as we demonstrate above), some rarer systemic vascular anomalies have been reported too, affecting among others small vessels of the brain [
83]. It is important to consider vascular anomalies as risk factors for ischemic disease as well as stroke [
83].
Besides conotruncal, great vessel or connective tissue anomalies, an association of left-sided heart anomalies with KS has been theorized. The presence of normal X chromosomes can prevent the development of left-sided anomalies. Left-heart anomalies are of multifactorial origin [
84]. KS patients with hypoplastic left heart syndrome (HLHS) tend to present with more severe cardiac symptoms than those with shunt anomalies. These may include failure to thrive and feeding difficulties. Prognosis is poorer than that of patients with shunt lesions. Patients typically undergo palliative surgery aimed only at improving quality of life, since the underlying disease cannot be corrected (unless cardiac transplantation is considered) [
78,
85]. Left-heart anomalies may shorten the lifespan of KS individuals.
One rare case of a child with KS, who died of untreated arrhythmia at the age of 11 months, was reported. The arrhythmia resulted from a cardiac conduction abnormality causing episodes of bradycardia unresponsive to pacing, which eventually led to asystole and death. On top of that, the child suffered from severe immunodeficiency. Autopsy revealed coronary sinus dilatation, tricuspid valve dysplasia, thickened chordae tendinae and right ventricular hypertrophy. Conduction abnormalities in KS children have not been extensively investigated [
86].
Most reports do not describe severe symptomatic heart disease in KS patients. In fact, some KS patients have remained cardiologically asymptomatic, or their symptoms have been masked by the underlying disease (such as in the case of congenital heart defects described by Digilio et al. [
68]). Thus, when considering the quality of life of KS patients, it is important to emphasize early recognition and treatment of cardiological abnormalities. Echocardiography should be performed in all the patients at the time of diagnosis in order to enable early recognition of structural cardiac anomalies [
70]. In patients with
KMT2D variants, attention should be paid to the detection of left-sided obstructive lesions, while in patients with
KDM6A variants, right-sided lesions should be the subject of attention [
70]. Individuals with aortic anomalies should be monitored at least annually for aortic dilatation [
82]. All patients with structural cardiac anomalies should be referred to a cardiologist [
70]. If treatable, appropriate therapy and recovery from cardiac disease will enable effective treatment directed at other ailments associated with KS in order to improve quality of life.
6. Gastrointestinal (GI) Issues in Kabuki Syndrome
Kabuki syndrome patients frequently present with poor feeding and nutrition, which leads to poor growth. They often have sensory issues that interfere with eating, rendering them more sensitive than other children. Textures, smells and temperatures of food may cause these children to present with aversions. Due to poorly coordinated suck and swallow reflexes, a nasogastric (NG) feeding tube or even a gastrostomy may be required [
87,
88]. Reflux tends to be more common and more severe in the KS population [
87]. Severe reflux can present with recurrent aspiration, eventually culminating in oxygen-dependency, chronic lung disease and death due to complications of neuromuscular and pulmonary involvement [
87]. At later ages, feeding therapy should be recommended for parents in order to educate these children to an encouraging mealtime experience and to improve these children’s eating habits. Proper feeding and nutrition is crucial for quality of life improvement [
87,
88].
Gastrointestinal (GI) anatomical dysmorphism has been described in single KS cases too, especially in females. It has even been suggested that KS in females is a risk factor for anorectal malformations [
89]. KS individuals may present with intestinal malrotation, anal atresia, anovestibular fistula or anterior anus. Lower GI anomalies are more likely in KS patients and generally have a positive prognosis, given that the management of these abnormalities in KS patients is not different from that of nonsyndromic patients [
89]. KS should be initially suspected clinically in female patients with characteristic dysmorphism and anorectal malformations. Pediatric surgeons should be aware of the potential for serious cardiac defects in these patients [
89]. These suggestions are, however, only based on two cases. Nevertheless, they can positively impact these patients’ quality of life thanks to early recognition. KS children may be at increased risk for pancreatic ductogenesis abnormalities, leading to pancreatitis. The specific cause of abnormal pancreatic duct morphology and pancreatitis is unclear, but may result from cytochrome C deficiency and/or abnormal enzyme production [
90]. There may be an increased risk of esophageal or gastric polyp development—a phenomenon occurring in 0.1% of pediatric patients. These polyps are usually symptomatic at onset, causing heartburn, nausea and vomiting. Treatment should be individualized and may include proton pump inhibitors and, in case of lack of improvement, endoscopic polypectomy to relieve symptoms [
91].
Hepatobiliary abnormalities, biliary atresia, hepatic fibrosis and sclerosing cholangitis have been documented in 2–21% of KS patients [
92]. Very little information is available regarding long-term follow-up of these patients’ liver disease. Liver transplantation in young adulthood may be the only way to overcome underlying liver disease, though severe progression may be preventable thanks to early recognition. Sclerosing cholangitis and dyslipidemia may be successfully treated with ursodiol and cholecystyramine [
92]. The specific etiology, pathophysiology and hyperbilirubinemia clinical course in these patients in the context of KS remains unclear, because even though liver disease is described in KS, there appears to be no common factor between cases [
92]. Liver disease may have an immunological basis, especially considering that KS patients often suffer from other autoimmune diseases. In such cases, based on findings by Suskind et al., ursodiol in combination with cholecystyramine should be considered in KS patients who suffer from cholestatic disease. When possible, using these drugs significantly improves quality of life and may, in theory, delay or even cancel liver transplantation. Primary liver tumors (namely hepatocellular carcinoma), though rare in children, may be at risk for development in KS patients due to the underlying condition.
KMT2D has been found to be somatically mutated in hepatocellular carcinoma (HCC) and has also been confirmed as a hepatitis B virus integration site. Variants in
KMT2D may result in abnormal enhancer regulation, which leads to changes in transcription, DNA breaks and tumor development. This also regulates hepatic metabolism and coactivates PPARy2, which leads to increased bile acid levels, which have been linked to hepatic tumor development (unspecific marker). Whether or not KS should be considered a risk factor for hepatocellular carcinoma remains unknown [
93]. The association between KS and autoimmune diseases has been described in the literature (see
Immunology section) [
94]. In one case, an increased risk of Crohn’s disease development was demonstrated [
95].
8. Orodental Symptomatology
Oral anomalies are present in over 60% of KS cases. Documented orodental pathology cases include congenital tooth absence, malocclusion, high-arched palate, abnormal dentition, widely spaced teeth, hypodontia, conical incisors, screwdriver-shaped incisors, delayed tooth eruption and ectopic upper molars [
113,
115,
116]. The role of
KMT2D and
KDM6A genes in tooth development has been recently theorized. Currently, the maxillomandibular relationship, tooth size and dental arch forms are being studied. The expression of
KMT2D and
KDM6A in human tooth buds at 7–12 Hbd was recently demonstrated. Gene expression was observed in the dental epithelium of primary incisors, canines and molars at the bud and cap stages of tooth development. This suggests that loss-of-function
KMT2D or
KDM6A variants may underlie the observed orodental anomalies [
113]. The function of
KMT2D and
KDM6A as epigenetic modulators of several biologic processes was recently discussed. Loss of their function leads to a variety of manifestations, in this context namely hypodontia, widely spaced teeth, absence of teeth, enamel hypoplasia, dental agenesis and caries [
116]. Cephalometric analysis can give insight to craniofacial structure and development. Using cephalometry, they found that severe maxillary recession and midfacial hypoplasia are common among KS patients. This is practical for the clinical diagnosis of KS [
115]. Cephalometric follow up of one patient showed the improvement of maxillary and mandibular growth in their patient. During this period, proper dental care, tooth extractions and fluoride treatments were ordered [
114]. Tooth buds for lateral upper and lower incisors as well as molars may be absent [
110]. Incisors may have large pulp chambers, may be affected by external root resorption or root canal division, tooth retention, retrognathia of the upper jaw, clefting, or lower lip fistula [
110]. KS cases with taurodontism (a condition in which the molar body and pulp chamber is enlarged at the expense of the roots, leading to apical displacement of the tooth furcation) were described [
110,
117]. These dental findings may help support the clinical diagnosis of KS [
117]. Tooth extractions and fluoride treatment seem to be effective treatment methods. Missing permanent teeth, sometimes up to seven in total, can also be confirmed in KS patients. This is most commonly true of maxillary and mandibular incisors; however, a missing maxillary canine can also be seen [
119]. Nursing bottle syndrome likely caused screwdriver-shaped incisors and high-arched maxilla manifested poor oral hygiene and early childhood caries in one KS boy, which highlights the importance of dental health in the improvement of life quality. KS patients may be more prone to root resorption than healthy patients due to their organic disease. Thus, their orthodontic state must be monitored [
120].
The treatment of orodental symptoms includes orthodontic treatment to correct bite function in addition to esthetics. Extraction and implantation procedures are often used to correct disorders of tooth development and increase patient quality of life. Orthodontic treatment can also be aimed at correcting arch length discrepancy. Due to feeding difficulties and an increased risk of nursing bottle syndrome, dental and orthodontic care must be integrated with child speech therapy in order to maintain long-term effects of orthodontic treatment. This aims at quality of life improvement [
101,
118,
120].
Cleft palate is a feature observed in 33–50% of KS cases. Pediatric follow-up is crucial in KS patients with clefting. Usually the first symptoms of cleft palate are speech development disorders and nasal leakage of milk during early infancy (submucous cleft palate). They mention, however, that mental retardation often complicates the accurate correct diagnosis of cleft palate because the above symptoms may be attributed to it. Therefore, they suggest that cleft palates are more frequently present than reported. They highlight the importance of routine specialist examination for clefting [
112]. KS children with cleft palate suffered from velopharyngeal insufficiency and ended up requiring two-flat palatoplasty procedures. These interventions ultimately improved speech development and quality of life [
111]. Another KS children cohort with cleft palate also had drooping lower lips, which is a symptom that may aid in the clinical diagnosis of KS [
121]. A KS girl whose parents are first cousins presented with multiple congenital abnormalities, including anorectal malformation—specifically imperforate anus with recto-vestibular fistula—diaphragmatic defect, lower lip pits, hypopigmentation, hypogammaglobulinemia as well as cleft palate. The severe concomitantly present multiple congenital abnormalities are likely to worsen prognosis. No other KS patient was reported with such severe anomalies [
122].
Lower lip pits are another feature that has been reported in KS, albeit more rarely than other cardinal features. Two KS patients were initially diagnosed with Van der Woude syndrome (a condition that affects facial development—lower lip pits are a cardinal feature of said syndrome, often with some combination of cleft lip and palate). These patients presented with lower lip pits and cleft palate and were clinically diagnosed with Van der Woude syndrome in early infancy. Upon follow-up during the second to fourth years of life, it was consequently noticed that they exhibited clinical features that do not concur with Van der Woude syndrome: facial dysmorphism—widened palpebral fissures, eyelid eversion and prominent ear; chronic otitis media leading to hearing impairment; velopharyngeal insufficiency and delayed psychomotor development. These patients’ clinical picture resembled KS, which was later confirmed by genetic testing. These cases emphasize the need for the careful evaluation of patients with lip pits and raise awareness for differential diagnosis [
99].
9. Ophthalmologic Issues in Kabuki Syndrome
Extraocular features have been defined as cardinal clinical diagnostic signs of Kabuki syndrome. They are present in nearly all KS patients [
123] and include: long palpebral fissures, lower palpebral eversion, arched eyebrows, epicanthus, ptosis, and a Marcus Gunn pupil (a type of relative afferent pupil defect that causes a pathologically decreased pupillary light reflex). These and other cardinal KS dysmorphisms are presented in
Figure 1. Another frequent feature occurring in over a half of KS patients is nocturnal lagophthalmos (sleeping with open eyes). It can predispose patients to infectious complications due to dry eye [
123,
124]. Ocular signs comprise amblyopia (“lazy eye”), refractive error, strabismus, nystagmus, microphthalmia, retinal or disc coloboma, and optic disc anomalies [
123,
125,
126]. Strabismus may take the form of alternate convergence as well as esotropia, including large-angle congenital esotropia [
126,
127]. Caruncular lipoma, bilateral blepharitis (most likely as a complication of nocturnal lagophthalmos) and bilateral inferior corneal pannus (fibrovascular granulation tissue) have been described in single patients [
124]. Ocular abnormalities often affect visual function. The prevalence of amblyopia and impaired acuity may be underestimated because many patients with intellectual disability do not undergo visual acuity tests. Vision disorders tend to worsen development; therefore, careful ophthalmologic evaluation at as young an age as possible is critical for each patient in order to correct treatable visual impairments [
123].
Coloboma is frequently reported in KS patients. It is sometimes associated with congenital heart disease and ear and renal defects, leading to the misdiagnosis of CHARGE syndrome before the presentation of typical facial dysmorphisms. It may affect the iris, choroid, retina or optic nerve. Coloboma derives from incomplete embryonic fissure closure in utero. Normally, the embryonic fissure develops from an invagination of the optic vesicle and leaves a gap that lets the hyaloid artery supply the inner eye, a process crucial for normal development. Iris or ciliary body colobomas result from incomplete anterior closure, choroid, retina or optic nerve colobomas result from incomplete posterior closure, whereas lens colobomas may be caused by incomplete closure of any segment. Eyelid colobomas are of a different embryologic origin and arise at a later stage of development. These are generally unrelated to disorders of globe development [
128]. CHARGE syndrome may phenotypically resemble Kabuki syndrome [
129]. It is genotypically different from KS (variants in
CHD7—a gene coding for a helicase) [
130]. Infrequent features that are shared between KS and CHARGE include microphthalmia, coloboma, anal atresia and panhypopituitarism. This similarity may be explained by the modifier genes effect [
130].
A wide array of rare ophthalmologic findings is present in almost all Kabuki syndrome individuals. Currently, it is not possible to directly associate these features with KS, yet a recommendation is widely supported that all KS patients should undergo ophthalmological assessment at the earliest age possible [
125]. Abnormal corneal development in utero has been demonstrated to lead to congenital corneal staphyloma—a severe corneal defect that causes a forward projection between the eyelids. The anomaly is now linked with
KMT2D variant [
131]. Bilateral congenital corneal opacity as an early-onset ocular KS manifestation has also been described. This is a rare congenital loss of corneal tissue transparency originating between the 6 and 16th week of gestation. In the same patient, bilateral corneal transplantations were performed in order to prevent deprivation amblyopia [
132,
133]. Agenesis of the lacrimal punctae was found as an incidental finding in a 29-year-old patient with
KMT2D variant [
134]. A KS patient with typical periorbital dysmorphism (lower eyelid eversion, epicanthus inversus, depressed nasal root, high-arched eyebrows, prominent ears and prominent eyelashes) underwent a simultaneous surgical repair of medial and lateral canthus. The deformity was characterized by raphe dysplasia in the orbicularis oculi muscle, which was corrected satisfactorily by lateral tarsorrhaphy—a partial surgical closure of the lateral part of the eyelids. In this case, surgical correction was recommended to prevent the drying of the cornea and conjunctiva, besides esthetic considerations. The surgical correction of canthus deformity should be considered in KS patients in whom corneal dryness may affect eye function [
135].
Microphthalmia was described in several KS patients. In one neonate, extreme microphthalmia with an anomaly of globe development, hyperplastic primary vitreous and hypoplastic optic chiasm were all present [
136].
KMT2D variant was later confirmed in this patient.
KMT2D proteins are active as part of a multi-subunit complex (see
Molecular Genetics chapter) and have been known to interact with
CHD7 and
CHD8 proteins through the WAR complex. Additionally,
KMT2D has been known to interact with the
PAX-interacting protein 1 (
PAXIP-1) as well as other transcription factors encoded by
PAX2. These factors have a high rate of expression in the developing eye, which may also explain microphthalmia and eye developmental disorder in utero [
136]. Ocular muscle dysfunction was recently reported. The overaction of inferior oblique muscles together with weakness of the superior oblique muscles associated with a V-shape motion was observed in four patients, and the underestimation of this phenomenon in KS patients has been put forward.
A summary of other rare ophthalmologic findings that were reported in under 10 KS patients in the literature is presented in
Table 3 below. Many of these ophthalmologic findings tended to affect the cornea.
10. Oncological Issues in Kabuki Syndrome
KMT2D is expressed in most cells and tissues. Its pathogenic variant results in the interruption of histone methylation related to gene expression, thus affecting normal growth and development as well as many other processes described in other chapters. Its role in development, metabolism, cell differentiation and tumor suppression has been recently described [
145]. Defective histone demethylation, as a result of a somatic variant, may dysregulate gene expression and predispose to cancer.
KMT2D has been defined as one of the most commonly mutated genes in a number of cancers such as gastric cancer, lymphoma and medulloblastoma [
145]. The lung-specific loss of
KMT2D promotes lung tumorigenesis and upregulates tumorigenic processes such as glycolysis in a recent mouse model [
146]. The oncogenic potential of
KMT2D loss-of-function variant has also been demonstrated; however, the rarity of such variants makes it difficult to assess their pathogenicity exactly [
147]. Using recombination and nuclease-mediated gene editing, it was proven that a
KMT2D loss-of-function variant causes the proliferation of neoplastic cells [
148]. This pathomechanism is likely responsible for medulloblastoma formation [
149]. Additionally,
KMT2D deficiency attenuates cancer cell migration, promoting tumor growth [
148]. The study also confirmed previously published findings which showed that
KMT2D is required for effective H3K4 methylation [
148].
KMT2D and
KDM6A belong to the same beta-globin and estrogen-receptor regulator multiprotein complex (called ASCOM) and interact with each other [
145,
150]. Recently,
KDM6A has been shown to interact with CBP transcription-activator protein in a breast cancer cell-line
MCF-7 in a
Drosophila model. Clinically, this variant has been found in many tumor types, including multiple myeloma, esophageal squamous cell carcinoma, renal cell carcinoma, glioblastoma, and breast, urinary tract, pancreatic and colorectal cancers.
KDM6A has been demonstrated as a tumor suppressor that leads to slow cell growth [
151,
152,
153]. Several mouse models analyzed
KDM6A in the context of urinary tract, pancreatic and lung tumors and acute myeloid leukemia [
151]. Conversely,
KDM6A has a pro-oncogenic role. The loss of
KDM6A in human breast cancer cells causes a decrease in estrogen-induced cell proliferation in vitro. This is, however, based on one recent study and conclusions as for in vitro pro-oncogenic trends of
KDM6A are yet to be determined [
151].
The above described role of somatic
KMT2D variants, i.e., restricted to cancer tissue, is well known. However, the significance of germline (present in all tissues) pathogenic variants in this gene in KS patients is yet to be determined. KS patients may present with a wide range of tumors. The new research on growth hormone therapy (see
Growth chapter) suggests that it may worsen tumor burden [
145]. Cancer risks, on an individual basis, should be established in KS patients in whom growth hormone therapy is considered.
In a 3-year-old KS patient in whom Wilms tumor was diagnosed, no negative impact on lifespan was demonstrated and the tumor was successfully managed according to procedures used in non-KS patients [
154]. Neuroblastoma, the most common extracranial solid tumor in infancy, was described in clinically diagnosed KS patients. This patient’s prognosis was assessed to not be different from sporadic cases, but constitutional molecular defects associated with KS may play a role in oncogenesis [
155,
156]. The tumor spectrum was expanded to include: a low-grade fibromyxoid sarcoma in a clinically diagnosed KS patient—the mass was successfully resected [
157]; and a case of aggressive desmoid fibromatosis, successfully treated surgically [
158].
Hepatoblastoma was described in one 6-year-old clinically diagnosed KS patient. Prognosis was assessed to be favorable and this patient was treated with chemotherapy (which led to mild hematotoxicity). Later, a right lobe hepatectomy was performed, followed by adjuvant chemotherapy, successfully leading to complete remission [
155]. Hepatocellular carcinoma was reported in another KS patient, a 15-year-old female with a
KMT2D variant. She underwent donor liver transplantation. On biopsy, besides characteristics of hepatocellular carcinoma, the explanted liver exhibited hepatic adenomas with a low Ki-67 proliferation index [
93]. The association between genetic syndromes and liver cancer has been suggested [
93,
159].
KMT2D has been shown to be mutated in hepatocellular carcinoma cases, and has even been suggested to be associated with earlier recurrence of disease, greater microvascular invasion and a more aggressive phenotype [
93,
160]. It has also been suggested to disrupt the control of bile acid production in mutant mice [
93,
161]. Despite the cases above presenting a benign phenotype, the potential development of malignant liver cancer must be considered in KS patients.
Pilomatricoma—a benign hair cell tumor characterized by lobules of epithelial cells—has been associated with several genetic syndromes (Turner, Gardner, Rubinstein-Taybi, Sotos, Trisomy 9, myotonic dystrophy, spina bifida, sarcoidosis, gliomatosis cerebri) [
162,
163,
164]. The only known tumor that has been confirmed in pilomatricoma is caused by a somatic variant in
CTNNB1. KS may predispose patients to pilomatricoma formation because
CTNNB1 plays a role in the
WNT pathway, which is regulated by
KMT2D [
162,
163]. Surgical excision is indicated [
164]. Another dermatologic lesion, Becker nevus, was also reported in KS [
165].
Giant cell fibroblastoma, a soft-tissue malignancy that is especially rare in childhood, was reported in a 12-year-old female patient with a
KMT2D variant. She presented with a recurring neck mass despite excision and received a favorable prognosis. The tumor was successfully excised [
166].
Spinal ependymoma was diagnosed in a 23-year-old KS patient. Ependymoma is a tumor originating from the ependymal lining of the ventricular system. Most cases are located intracranially; however, spinal presentation is less common. The patient presented with hypoesthesia of the feet and lumbar pain. Laminotomy tumor resection was performed and no neurological complications were observed. Ki-67 index was low upon biopsy. This case sheds light on atypical symptom presentation in KS patients—even though ependymoma does not typically present in the spine, spinal abnormalities are typical in KS patients [
167].
Several KS patients with hematological malignancies were reported. Hodgkin Lymphoma was reported in a patient with a severe multiorgan phenotype and
KMT2D germline variant. He presented with clubfeet, hirsutism, hypotonia, and feeding problems during infanthood; chronic otitis media, obesity and failure to thrive during late childhood; and confirmed Immunoglobulin IgA and IgG deficiencyType 1 diabetes mellitus1 and pancreatic failure in young adulthood. He required pancreatic transplant thrice, after which he developed Ebstein-Barr virus-positive Hodgkin Lymphoma and underwent chemotherapy, achieving complete remission.
KMT2D is one of the most frequently mutated genes in follicular lymphomas as well as in diffuse large B-cell lymphomas. By the loss of H3K4 methylation at enhancer DNA regions, truncating variants of the
KMT2D protein lead to the loss of enzymatic activity and haploinsufficiency, causing malignant transformation [
168,
169]. A clinically diagnosed KS patient with IgA and IgG deficiency developed acute lymphocytic leukemia. She had a history of recurrent infections, indicating an immunodeficiency with susceptibility to cancer [
170]. Several cases of KS patients with Burkitt lymphoma were also reported [
171,
172,
173,
174]. In the most recently reported case, genetic testing was available to reveal a
KMT2D variant causing KS as well as a translocation t (8;14) causing Burkitt lymphoma in an EBV-positive KS patient. KS patients may thus be at increased risk for hematological cancers. Decreased histone regulation may cause DNA damage due to
KMT2D variant, and that may increase the risk for other permanent DNA damage to occur, in this case, t (8;14) translocation [
174].
Table 4 describes rarely reported tumors in KS patients.
11. Immunological Issues in Kabuki Syndrome
KS patients present with numerous immunopathological manifestations.
KMT2D and
KDM6A encode, among others, proteins acting in the COMPASS complex, which serves as a chromatin opener. Loss of function of
KMT2D and
KDM6A epigenetically leads to defective B-lymphocyte terminal differentiation and thus hypogammaglobulinemia and reduced memory B-cell numbers [
174]. B-cell lineage development deficiencies were demonstrated in a recent mouse model. Mice with a
KMT2D variant had reduced serum IgA and elevated IgM. The bone marrow, spleen and intestine of
KMT2D-mutated mice contained fewer IgA-secreting cells. There were more B-lineage cells in the Peyer patches; however, there were fewer Peyer patches overall. This was also found in vivo.
KMT2D epigenetically controls the gene
ITGB7, which encodes for an adhesion protein that mediates intestinal homing (the process by which white blood cells target both inflamed and non-inflamed regions of the gut to provide an immune response).
KMT2D insufficiency has deleterious effects on B-cell differentiation, explaining this mechanism of immunodeficiency in KS patients [
175].
KMT2D takes part in chromosomal translocations that create chimeric proteins. Translocation partners exist in a super elongation complex, which is required for
Hox gene expression in white blood cells as well as in leukemic cells (potentially increasing the risk of leukemogenesis, see
Oncology chapter) [
150]. The COMPASS complex epigenetically regulates FOXP3, which affects the differentiation of naïve CD4+ T-cells into T-regulatory cells. Reduced T-cell tolerance also explains the increased risk of autoimmune pathology observed in KS patients.
Considering the genotype, the prevalence in KS patients of immune deficiency or autoimmune manifestations does not differ by gene type (whether
KMT2D or
KDM6A). An increased risk of immune thrombocytopenic purpura development with missense variants than with truncating variants has been suggested [
176]. The immunological phenotype observed in KS patients has been recently described in a large cohort and includes (in order of prevalence) infection susceptibility (especially to acute otitis media—see
Otolaryngology chapter), hypogammaglobulinemia, increased risk of autoimmune disease, increased risk of immune thrombocytopenic purpura, vitiligo, autoimmune hemolytic anemia, thyroiditis and other rare autoimmune cases [
176]. The prevalence of immune thrombocytopenic purpura usually decreases with age in healthy children; conversely, its prevalence slightly increases in KS patients [
176]. KS patients have an increased risk of developing refractory immune thrombocytopenic purpura, for which Rituximab seems to be an effective treatment method [
177]. IVIG and steroid treatment of immune thrombocytopenic purpura and autoimmune hemolytic anemia seems to be equally effective as in healthy children [
178,
179].
KS patients are more likely to develop Common Variable Immunodeficiency, which is known to increase the risk of autoimmune disease development, especially immune thrombocytopenic purpura and autoimmune hemolytic anemia [
180]. The KS immunological phenotype has been compared to that of common variable immunodeficiency, which is characterized by recurrent bacterial infections, hypogammaglobulinemia and impaired antibody responses as well as an increased prevalence of inflammatory and autoimmune disorders [
181,
182,
183]. There is heterogeneity of immune pathology in KS [
182]. Decreased or even undetectable IgA levels are observed in over 80% of KS patients. Low IgG levels are seen in nearly half of KS patients [
174,
184]. It is recommended that each KS patient be regularly tested as blood counts, IgA levels and urinalysis may present early clues for immune abnormalities. Patients with recurrent otitis and low levels of serum Ig should be consulted by an immunologist (see
Otolaryngology chapter). The routine administration of IVIG has not been proven to be effective in every KS patient. IVIG should only be considered in case of low total serum IgA [
182,
183].
MTOR inhibitors, as a more targeted treatment for immunodeficiencies associated with KS, are a subject of recent research, and singular case studies present the remission of cytopenia (see Table 6) [
184].
Table 5 and
Table 6 present singular KS cases of autoimmune and immunodeficiency manifestations, respectively, reported in the literature. These cases suggest a broadened KS immunological phenotype.
12. Nephrological Issues in Kabuki Syndrome
Up to 40% of KS patients present with urinary system abnormalities. Of these, about half present with renal malformations and half with urinary tract anomalies. The concomitant presentation of renal and urinary tract abnormalities is rare. Renal malformations and diseases include, in order of prevalence, horseshoe kidney, renal hypodysplasia, renal ectopy and renal duplication and renal insufficiency (single reported cases). Urinary tract malformations include, in order of prevalence, hydronephrosis and ureteral duplication. No phenotype correlation between KS and vesicoureteral reflux or ureteropelvic junction obstruction seems to be prevalent [
189]. Considering genotype-phenotype correlation, it is suggested that
KMT2D variant increases the risk of renal manifestations, but not of urinary tract anomalies [
1,
189,
190]. The etiology of urinary system abnormalities in the context of KS has been suggested to be rooted in the
KMT2D epigenetic role in the regulation of renal development. Dysfunctional
KMT2D may ultimately lead to renal dysplasia. However, the specific role of
KMT2D in renal development has not been established [
190].
Several KS patients with severe renal insufficiency have been reported. Some of these patients also presented with renal dysplasia. One KS patient presented with severe renal insufficiency during the first month of life due to dysplastic kidneys. She had a positive family history of Polycystic Kidney Disease. Poor prognosis was concluded [
189]. Another KS patient presented with terminal renal insufficiency due to bilateral renal dysplasia at the age of 6 years. She underwent successful transplantation and prognosis was good [
191]. Another KS patient with a nonfunctional dysplastic kidney underwent successful transplantation at the age of 14 [
192]. One KS patient did not undergo renal transplantation in time and died at the age of 5 years due to renal failure and concomitant pulmonary hypertension [
26]. Renal hypoplasia characteristics can be seen upon renal ultrasonography and may be associated with increased risk of renal insufficiency. This suggests that regular ultrasonographic assessment, blood pressure measurement, glomerular filtration rate evaluation and proteinuria screening are necessary as preventative measures in KS patients. If worsening kidney function is suspected, nephrotoxic drugs and high-protein diet should be avoided. These patients should be promptly referred to a pediatric nephrologist as they can often be treated with good prognosis [
189].
Kabuki syndrome has often been compared to Au Kline syndrome while considering kidney diseases. Au Kline Syndrome is caused by a variant in
HNRNPK, which codes for a ribonucleoprotein. The variant causes a multiorgan development disorder among other effects, ultimately leading to chronic kidney disease and kidney failure. Renal anomalies leading to chronic kidney disease are not unheard of in KS patients too. Renal ultrasound, kidney function evaluation and, when appropriate, early dialysis are recommended in both Au Kline Syndrome and KS patients too [
193].
Generally, the treatment of renal anomalies in KS patients is no different than in patients without KS [
189]. Several successful renal outcomes of kidney transplantation in KS patients have been reported in the literature [
192,
194]. Considering the progressive nature of the urinary system malformations, renal transplantation may be a realistic treatment option for KS patients (see
Table 7).
Rare urinary system manifestations reported in the literature are presented in
Table 7 below. These cases often present with concomitant clinical findings, namely congenital heart defects and ear disease (recurrent acute otitis media and/or ear malformation).