Marfan Syndrome Caused by Disruption of the FBN1 Gene due to A Reciprocal Chromosome Translocation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Panel Diagnostics
2.3. Conventional Cytogenetic Analysis
2.4. Microarray Based Molecular Cytogenomic Analysis
2.5. Molecular Cytogenetic Analysis
2.6. Optical Genome Mapping and Sequencing
3. Results
3.1. Clinical Report
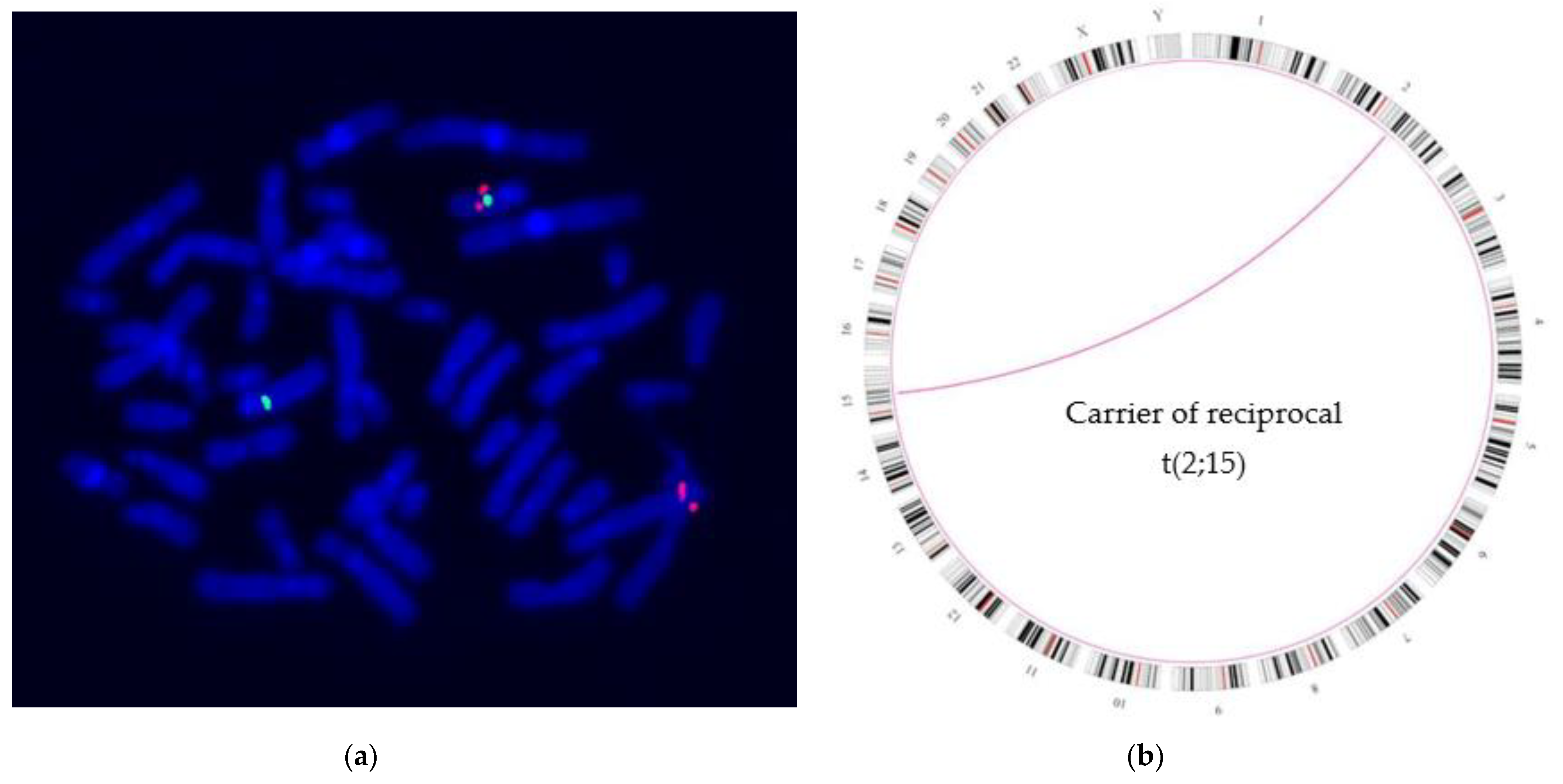
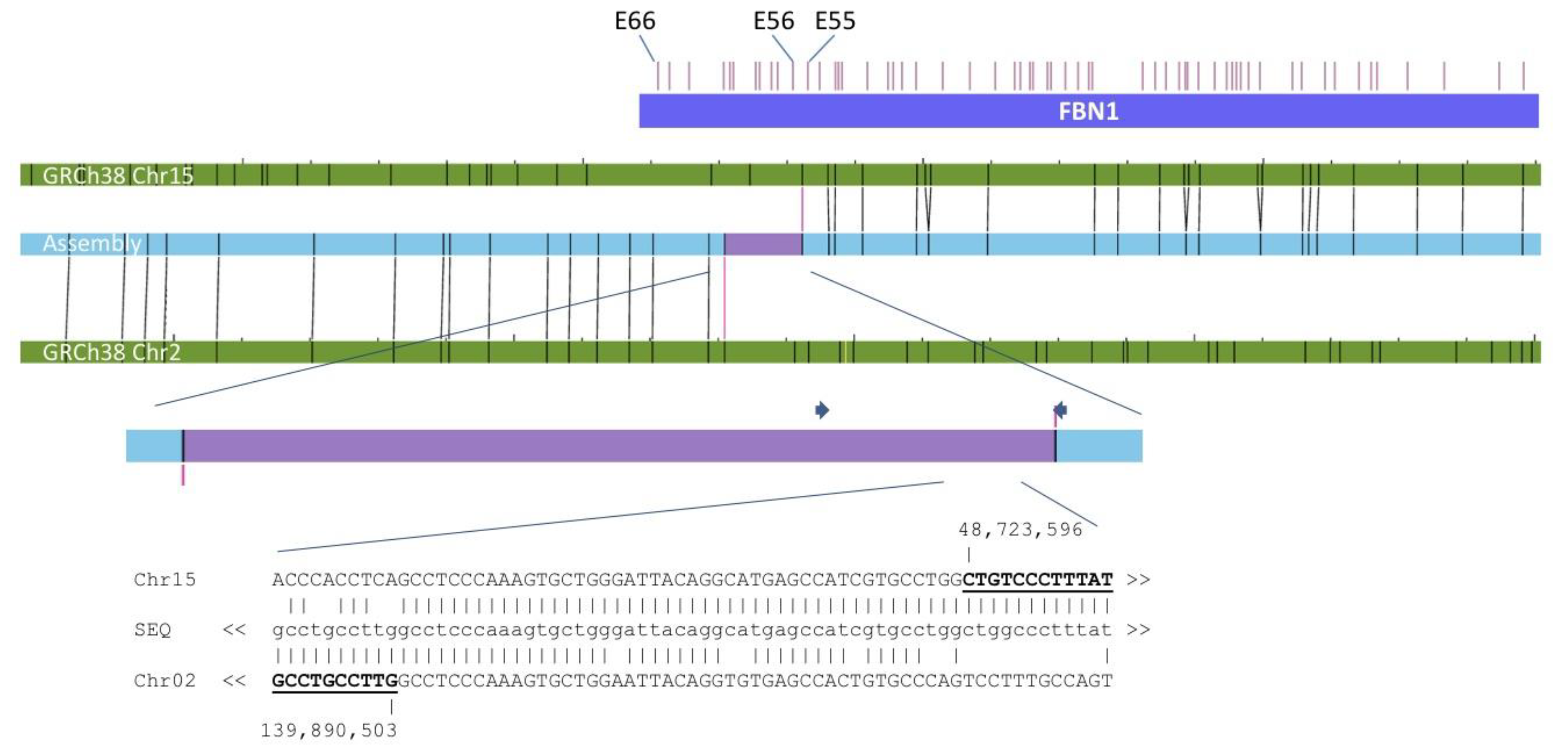
3.2. Cytogenetic, Cytogenomic, and Molecular Genetic Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dietz, H.C.; Cutting, C.R.; Pyeritz, R.E.; Maslen, C.L.; Sakai, L.Y.; Corson, G.M.; Puffenberger, E.G.; Hamosh, A.; Nanthakumar, E.J.; Curristin, S.M.; et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature 1991, 352, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.; Singh, M.; Eckersley, A.; Cain, S.A.; Sherratt, M.J.; Baldock, C. Fibrillin microfibrils and elastic fibre proteins: Functional interactions and extracellular regulation of growth factors. Semin. Cell Dev. Biol. 2019, 89, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Loeys, B.L.; Dietz, H.C.; Braverman, A.C.; Callewaert, B.L.; Backer, J.D.; Devereux, R.B.; Hilhorst-Hofstee, Y.; Jondeau, G.; Faivre, L.; Milewicz, D.M.; et al. The revised Ghent nosology for the Marfan syndrome. J. Med. Genet. 2010, 47, 476–485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- HGMD®Professional March 2021. Available online: https://my.qiagendigitalinsights.com/bbp/view/hgmd/pro/all.php (accessed on 28 September 2021).
- Franken, R.; Teixido-Tura, G.; Brion, M.; Forteza, A.; Rodriguez-Palomares, J.; Gutierrez, L.; Dorado, D.G.; Pals, G.; Mulder, B.J.M.; Evangelista, A. Relationship between fibrillin-1 genotype and severity of cardiovascular involvement in Marfan syndrome. Heart 2017, 103, 1795–1799. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Li, L.; Fu, Y.; Wang, X.; Sun, H.; Han, L.; Wu, Z.; Liu, Y.; Zhu, J.; Sun, L.; et al. Increased frequency of FBN1 frameshift and nonsense mutations in Marfan syndrome patients with aortic dissection. Mol. Genet. Genomic Med. 2020, 8, e1041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hilhorst-Hofstee, Y.; Hamel, B.C.J.; Verheij, J.B.G.M.; Rijlaarsdam, M.E.B.; Mancini, G.M.S.; Cobben, J.M.; Giroth, C.; Ruivenkamp, C.A.L.; Hansson, K.B.M.; Timmermans, J.; et al. The clinical spectrum of complete FBN1 allele deletions. Eur. J. Hum. Genet. 2011, 19, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Colovati, M.E. Marfan syndrome with a complex chromosomal rearrangement including deletion of the FBN1 gene. Mol. Cytogenet. 2012, 5, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tiecke, F.; Katzke, S.; Booms, P.; Robinson, P.N.; Neumann, L.; Godfrey, M.; Mathews, K.R.; Scheuner, M.; Hinkel, G.K.; Brenner, R.E.; et al. Classic, atypically severe and neonatal Marfan syndrome: Twelve mutations and genotype–phenotype correlations in FBN1 exons 24–40. Eur. J. Hum. Genet. 2001, 9, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Faivre, L.; Colland-Beroud, G.; Loeys, B.L.; Child, A.; Binquet, C.; Gautier, E.; Callewaert, B.; Arbustini, E.; Mayer, K.; Arslan-Kirchner, M.; et al. Effect of Mutation Type and Location on Clinical Outcome in 1,013 Probands with Marfan Syndrome or Related Phenotypes and FBN1 Mutations: An International Study. Am. J. Hum. Genet. 2007, 81, 454–466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baudhuin, L.M.; Kotzer, K.E.; Lagerstedt, S.A. Decreased frequency of FBN1 missense variants in Ghent criteria-positive Marfan syndrome and characterization of novel FBN1 variants. J. Hum. Genet. 2015, 60, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, P.; Milleron, O.; Hanna, N.; Ropers, J.; Ould Ouali, N.; Affoune, A.; Langeois, M.; Eliahou, L.; Arnoult, F.; Renard, P.; et al. Clinical relevance of genotype–phenotype correla-tions beyond vascular events in a cohort study of 1500 Marfan syndrome patients with FBN1 pathogenic variants. Genet. Med. 2021, 23, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.N.; Booms, P.; Katzke, S.; Ladewig, M.; Neumann, L.; Palz, M.; Pregla, R.; Tiecke, F.; Rosenberg, T. Mutations of FBN1 and genotype-phenotype correlations in Marfan syndrome and related fibrillinopathies. Hum. Mutat. 2002, 20, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Schluth-Bolard, C.; Diguet, F.; Chatron, N.; Rollat-Farnier, P.-A.; Bardel, C.; Afenjar, A.; Amblard, F.; Amiel, J.; Blesson, S.; Callier, P.; et al. Whole genome paired-end sequencing elucidates functional and phenotypic consequences of balanced chromosomal rearrangement in patients with developmental disorders. J. Med. Genet. 2019, 56, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Sismani, C.; Kitsiou-Tzeli, S.; Ioannides, M.; Christodoulou, C.; Anastasiadou, V.; Stylianidou, G.; Papadopoulou, E.; Kanavakis, E.; Kosmaidou-Aravidou, Z.; Patsalis, P.C. Cryptic genomic imbalances in patients with de novo or familial apparently balanced translocations and abnormal phenotype. Mol. Cytogenet. 2008, 1, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Vree, P.J.; Simon, M.E.H.; Vn Dooren, M.F.; Stoevelaar, G.H.T.; Hilkmann, J.T.W.; Rongen, M.A.; Huijbregts, G.C.M.; Verkerk, A.J.M.H.; Poddighe, P.J. Application of molecular cytogenetic techniques to clarify apparently balanced complex chromosomal rearrangements in two patients with an abnormal phenotype: Case report. Mol. Cytogenet. 2009, 2, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahajpal, N.S.; Barseghyan, H.; Kolhe, R.; Hastie, A.; Chaubey, A. Optical Genome Mapping as a Next-Generation Cytogenomic Tool for Detection of Structural and Copy Number Variations for Prenatal Genomic Analyses. Genes 2021, 12, 398. [Google Scholar] [CrossRef] [PubMed]
- Benarroch, L.; Aubart, M.; Gross, M.-S.; Arnaud, P.; Hanna, N.; Jondeau, G.; Boileau, C. Reference Expression Profile of Three FBN1 Transcript Isoforms and Their Association with Clinical Variability in Marfan Syndrome. Genes 2019, 10, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schnause, A.C.; Komlosi, K.; Herr, B.; Neesen, J.; Dremsek, P.; Schwarz, T.; Tzschach, A.; Jägle, S.; Lausch, E.; Fischer, J.; et al. Marfan Syndrome Caused by Disruption of the FBN1 Gene due to A Reciprocal Chromosome Translocation. Genes 2021, 12, 1836. https://doi.org/10.3390/genes12111836
Schnause AC, Komlosi K, Herr B, Neesen J, Dremsek P, Schwarz T, Tzschach A, Jägle S, Lausch E, Fischer J, et al. Marfan Syndrome Caused by Disruption of the FBN1 Gene due to A Reciprocal Chromosome Translocation. Genes. 2021; 12(11):1836. https://doi.org/10.3390/genes12111836
Chicago/Turabian StyleSchnause, Anna Clara, Katalin Komlosi, Barbara Herr, Jürgen Neesen, Paul Dremsek, Thomas Schwarz, Andreas Tzschach, Sabine Jägle, Ekkehart Lausch, Judith Fischer, and et al. 2021. "Marfan Syndrome Caused by Disruption of the FBN1 Gene due to A Reciprocal Chromosome Translocation" Genes 12, no. 11: 1836. https://doi.org/10.3390/genes12111836
APA StyleSchnause, A. C., Komlosi, K., Herr, B., Neesen, J., Dremsek, P., Schwarz, T., Tzschach, A., Jägle, S., Lausch, E., Fischer, J., & Gläser, B. (2021). Marfan Syndrome Caused by Disruption of the FBN1 Gene due to A Reciprocal Chromosome Translocation. Genes, 12(11), 1836. https://doi.org/10.3390/genes12111836






