Snorkeling Strategy: Tolerance to Flooding in Rice and Potential Application for Weed Management
Abstract
1. Introduction
2. Rice Flooded Strategy
| Timing of Flooding | Tolerant Genotypes | Main Traits Associated with Tolerance | Main Genes * | References |
|---|---|---|---|---|
| Germination and early growth | KHAO HLAN ON KHAIYAN F291 F274-2A 8391 8753 ITJ03 AV04 | Early development of root and coleoptile underwater; Green shoots appear earlier; Rapidly growing coleoptiles | RAmy3D ** ADH1 ADH2 SUS1 PDC SNORKEL1 SNORKEL2 OsTPP7 | [3,5,6,13,20,25,26,27,28] |
| Vegetative | FR13A KURKARUPPAN GODA HEENATI THAVALU IR64-SUB1 SWARNA-SUB1 SAVITRI-SUB1 CIHERANG | Reduced elongation; Slow carbohydrate consumption during submergence; Underwater photosynthesis; Chlorophyll retention underwater; Fast recovery | SUB1A-1 | [8,9,21,23,24,29,30,31,32] |
| IRRI119 IRRI154 JALMAGNA BAISBISH RAYADA 16-3 SUDU GRIES TIL BAJAL FULKARI TUNG LU 3 | Fast internode elongation with rising water; Sufficient leaf area above water; Photosynthesis underwater; Kneeling ability when the water recedes; Large fertile panicles; | SNORKEL1 SNORKEL2 ADH1 ADH2 SUS1 | [20,33,34,35,36] |
3. Mechanisms of Tolerance to Flooding in Rice and Weedy Species
3.1. Tolerance to Flooding during Germination
3.2. Tolerance to Flooding during Seedling Emergence
3.3. Tolerance to Flooding during the Vegetative Stage
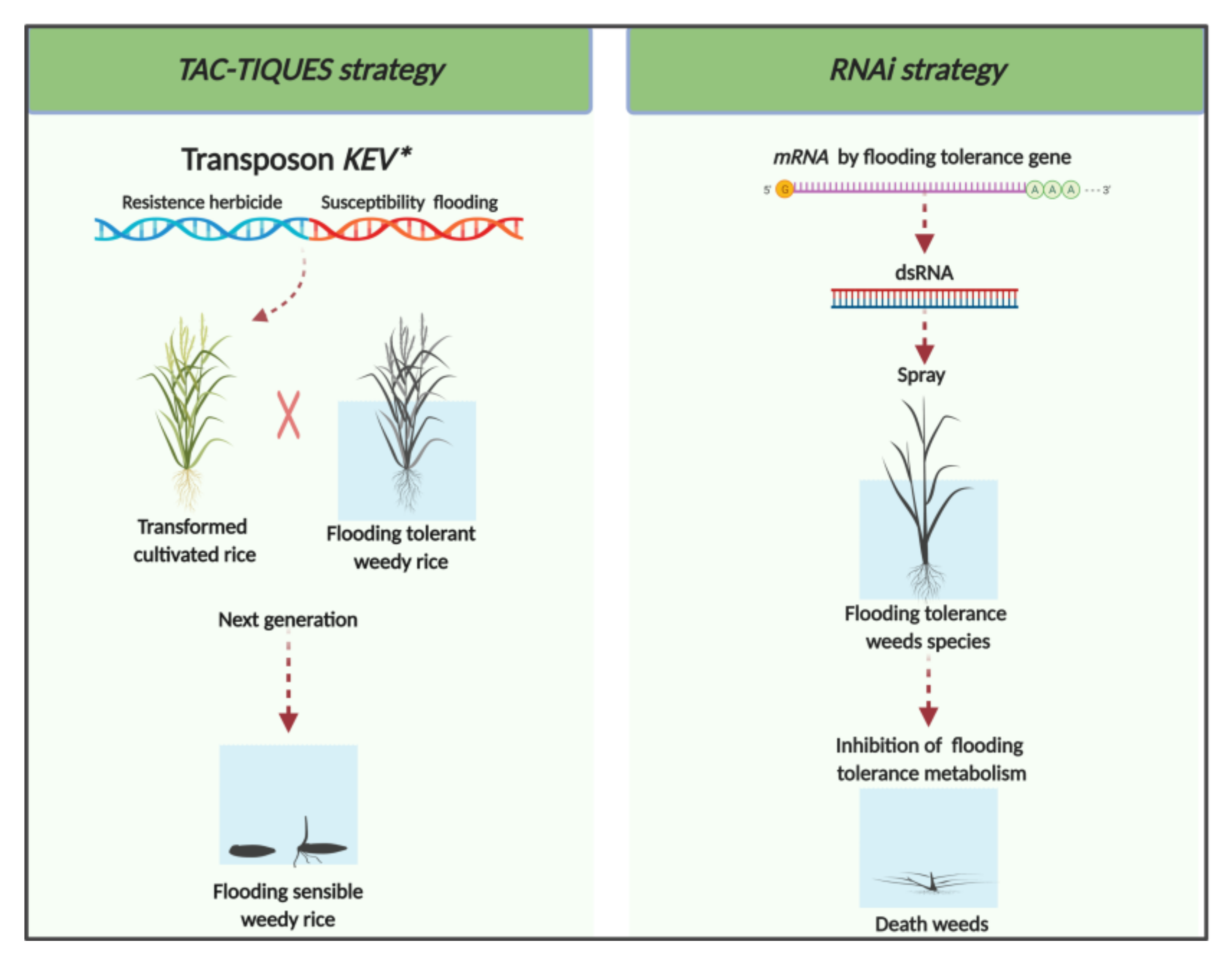
4. Biotechnological Approaches for Managing Flood-Tolerant Weeds
4.1. TAC-TIQUES Strategy
4.2. RNAi Strategy
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Colmer, T.D.; Cox, M.C.H.; Voesenek, L.A.C.J. Root Aeration in Rice (Oryza sativa): Evaluation of Oxygen, Carbon Dioxide, and Ethylene as Possible Regulators of Root Acclimatizations. New Phytol. 2006, 170, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Loreti, E.; van Veen, H.; Perata, P. Plant Responses to Flooding Stress. Curr. Opin. Plant Biol. 2016, 33, 64–71. [Google Scholar] [CrossRef]
- Hsu, S.K.; Tung, C.W. RNA-Seq Analysis of Diverse Rice Genotypes to Identify the Genes Controlling Coleoptile Growth during Submerged Germination. Front. Plant Sci. 2017, 8, 1–15. [Google Scholar] [CrossRef]
- Wu, C.; Mozzoni, L.A.; Moseley, D.; Hummer, W.; Ye, H.; Chen, P.; Shannon, G.; Nguyen, H. Genome-Wide Association Mapping of Flooding Tolerance in Soybean. Mol. Breed. 2020, 40, 1–14. [Google Scholar] [CrossRef]
- Ismail, A.M.; Johnson, D.E.; Ella, E.S.; Vergara, G.V.; Baltazar, A.M. Adaptation to Flooding during Emergence and Seedling Growth in Rice and Weeds, and Implications for Crop Establishment. AoB Plants 2012, 2012, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Estioko, L.P.; Miro, B.; Baltazar, A.M.; Merca, F.E.; Ismail, A.M.; Johnson, D.E. Differences in Responses to Flooding by Germinating Seeds of Two Contrasting Rice Cultivars and Two Species of Economically Important Grass Weeds. AoB Plants 2014, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Peralta Ogorek, L.; Striker, G.G.; Mollard, F.P.O. Echinochloa Crus-Galli Seed Physiological Dormancy and Germination Responses to Hypoxic Floodwaters. Plant Biol. 2019, 21, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Mackill, D.J.; Ismail, A.; Singh, U.; Labios, R.; Paris, T.R. Development and Rapid Adoption of Submergence-Tolerant (Sub1) Rice Varieties. Adv. Agron. 2012, 115, 299–352. [Google Scholar]
- Kato, Y.; Collard, B.C.Y.; Septiningsih, E.M.; Ismail, A.M. Increasing Flooding Tolerance in Rice: Combining Tolerance of Submergence and of Stagnant Flooding. Ann. Bot. 2019, 124, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Kar, G.; Sahoo, N.; Kumar, A. Deep-Water Rice Production as Influenced by Time and Depth of Flooding on the East Coast of India. Arch. Agron. Soil Sci. 2012, 58, 573–592. [Google Scholar] [CrossRef]
- Rao, A.N.; Johnson, D.E.; Sivaprasad, B.; Ladha, J.K.; Mortimer, A.M. Weed Management in Direct-Seeded Rice. Adv. Agron. 2007, 93, 153–255. [Google Scholar] [CrossRef]
- Chauhan, B.S. Weedy Rice (Oryza sativa) II. Response of Weedy Rice to Seed Burial and Flooding Depth. Weed Sci. 2012, 60, 385–388. [Google Scholar] [CrossRef]
- Kaspary, T.E.; Cutti, L.; Rafaeli, R.S.; Delattore, C.A.; Merotto, A. Genes Related to Flooding Tolerance during Germination and Early Growth of Weedy Rice. Weed Res. 2020, in press. [Google Scholar]
- Gressel, J.; Levy, A.A. Use of Multicopy Transposons Bearing Unfitness Genes in Weed Control: Four Example Scenarios. Plant Physiol. 2014, 166, 1221–1231. [Google Scholar] [CrossRef][Green Version]
- Shaner, D.L.; Beckie, H.J. The Future for Weed Control and Technology. Pest Manag. Sci. 2014, 70, 1329–1339. [Google Scholar] [CrossRef]
- Wu, C.S.; Kuo, W.T.; Chang, C.Y.; Kuo, J.Y.; Tsai, Y.T.; Yu, S.M.; Wu, H.T.; Chen, P.W. The Modified Rice AAmy8 Promoter Confers High-Level Foreign Gene Expression in a Novel Hypoxia-Inducible Expression System in Transgenic Rice Seedlings. Plant Mol. Biol. 2014, 85, 147–161. [Google Scholar] [CrossRef]
- Andersson, M.; Vicente, M.; Andersson, M.S.; de Vicente, M.C. Gene Flow between Crops and Their Wild Relatives. Genet. Resour. Crop Evol. 2010, 58, 619. [Google Scholar] [CrossRef][Green Version]
- Ellstrand, N.C.; Meirmans, P.; Rong, J.; Bartsch, D.; Ghosh, A.; de Jong, T.J.; Haccou, P.; Lu, B.-R.; Snow, A.A.; Neal Stewart, C.; et al. Introgression of Crop Alleles into Wild or Weedy Populations. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 325–345. [Google Scholar] [CrossRef]
- Yan, H.; Li, L.; Liu, P.; Jiang, X.; Wang, L.; Fang, J.; Lin, Z.; Wang, F.; Su, J.; Lu, B. Reduced weed seed shattering by silencing a cultivated rice gene: Strategic mitigation for escaped transgenes. Transgenic Res. 2017, 26, 465–475. [Google Scholar] [CrossRef]
- Hattori, Y.; Nagai, K.; Furukawa, S.; Song, X.J.; Kawano, R.; Sakakibara, H.; Wu, J.; Matsumoto, T.; Yoshimura, A.; Kitano, H.; et al. The Ethylene Response Factors SNORKEL1 and SNORKEL2 Allow Rice to Adapt to Deep Water. Nature 2009, 460, 1026–1030. [Google Scholar] [CrossRef]
- Bailey-Serres, J.; Fukao, T.; Gibbs, D.J.; Holdsworth, M.J.; Lee, S.C.; Licausi, F.; Perata, P.; Voesenek, L.A.C.J.; van Dongen, J.T. Making Sense of Low Oxygen Sensing. Trends Plant Sci. 2012, 17, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Voesenek, L.A.C.J.; Bailey-Serres, J. Flood Adaptive Traits and Processes: An Overview. New Phytol. 2015, 206, 57–73. [Google Scholar] [CrossRef] [PubMed]
- Fukao, T.; Xu, K.; Ronald, P.C.; Bailey-Serres, J. A Variable Cluster of Ethylene Response Factor-Like Genes Regulates Metabolic and Developmental Acclimation Responses to Submergence in Rice. Plant Cell 2006, 18, 2021–2034. [Google Scholar] [CrossRef] [PubMed]
- Sasidharan, R.; Voesenek, L.A.C.J. Ethylene-Mediated Acclimations to Flooding Stress. Plant Physiol. 2015, 169, 3–12. [Google Scholar] [CrossRef]
- Miro, B.; Ismail, A.M. Tolerance of Anaerobic Conditions Caused by Flooding during Germination and Early Growth in Rice (Oryza sativa L.). Front. Plant Sci. 2013, 4, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.M.; Ella, E.S.; Vergara, G.V.; Mackill, D.J. Mechanisms Associated with Tolerance to Flooding during Germination and Early Seedling Growth in Rice (Oryza sativa). Ann. Bot. 2009, 103, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Qi Ye, X.; Meng, J.L.; Zeng, B.; Wu, M. Improved Flooding Tolerance and Carbohydrate Status of Flood-Tolerant Plant Arundinella Anomala at Lower Water Temperature. PLoS ONE 2018, 13, 1–12. [Google Scholar] [CrossRef]
- Kretzschmar, T.; Pelayo, M.A.F.; Trijatmiko, K.R.; Gabunada, L.F.M.; Alam, R.; Jimenez, R.; Mendioro, M.S.; Slamet-Loedin, I.H.; Sreenivasulu, N.; Bailey-Serres, J.; et al. A Trehalose-6-Phosphate Phosphatase Enhances Anaerobic Germination Tolerance in Rice. Nat. Plants 2015, 1, 1–5. [Google Scholar] [CrossRef]
- Xu, K.; Xu, X.; Fukao, T.; Canlas, P.; Maghirang-Rodriguez, R.; Heuer, S.; Ismail, A.M.; Bailey-Serres, J.; Ronald, P.C.; Mackill, D.J. Sub1A is an Ethylene-Response-Factor-like Gene That Confers Submergence Tolerance to Rice. Nature 2006, 442, 705–708. [Google Scholar] [CrossRef]
- Winkel, A.; Colmer, T.D.; Ismail, A.M.; Pedersen, O. Internal Aeration of Paddy Field Rice (Oryza sativa) during Complete Submergence—Importance of Light and Floodwater O2. New Phytol. 2013, 197, 1193–1203. [Google Scholar] [CrossRef]
- Korinsak, S.; Siangliw, M.; Kotcharerk, J.; Jairin, J.; Siangliw, J.L.; Jongdee, B.; Pantuwan, G.; Sidthiwong, N.; Toojinda, T. Improvement of the Submergence Tolerance and the Brown Planthopper Resistance of the Thai Jasmine Rice Cultivar KDML105 by Pyramiding Sub1 and Qbph12. F. Crop. Res. 2016, 188, 105–112. [Google Scholar] [CrossRef]
- Banayo, N.P.M.; Mabesa-Telosa, R.C.; Singh, S.; Kato, Y. Improved Early Season Management of SUB1 Rice Varieties Enhances Post-Submergence Recovery and Yield. Exp. Agric. 2019, 55, 105–116. [Google Scholar] [CrossRef]
- Hattori, Y.; Miura, K.; Asano, K.; Yamamoto, E.; Mori, H.; Kitano, H.; Matsuoka, M.; Ashikari, M. A Major QTL Confers Rapid Internode Elongation in Response to Water Rise in Deepwater Rice. Breed. Sci. 2007, 57, 305–314. [Google Scholar] [CrossRef]
- Iftekharuddaula, K.M.; Amin, A.; Shalahuddin, A.K.M.; Halder, T.; Yasmeen, R.; Hossain, M.A.; Aditya, T.L.; Ali, M.A.; Kabir, M.S. Current Scenarios, Progress and Prospects of Developing Technologies for Flood-Tolerant Rice in Bangladesh; Elsevier: New York, NY, USA, 2019. [Google Scholar] [CrossRef]
- Singh, S.; Mackill, D.J.; Ismail, A.M. Tolerance of Longer-Term Partial Stagnant Flooding Is Independent of the SUB1 Locus in Rice. Field Crop. Res. 2011, 121, 311–323. [Google Scholar] [CrossRef]
- Yang, S.Y.; Wu, Y.S.; Chen, C.T.; Lai, M.H.; Yen, H.M.; Yang, C.Y. Physiological and Molecular Responses of Seedlings of an Upland Rice (‘Tung Lu 3’) to Total Submergence Compared to Those of a Submergence-Tolerant Lowland Rice (‘FR13A’). Rice 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, B.S.; Johnson, D.E. Ecological Studies on Cyperus Difformis, Cyperus Iria and Fimbristylis Miliacea: Three Troublesome Annual Sedge Weeds of Rice. Ann. Appl. Biol. 2009, 155, 103–112. [Google Scholar] [CrossRef]
- Peña-Fronteras, J.T.; Villalobos, M.C.; Baltazar, A.M.; Merca, F.E.; Ismail, A.M.; Johnson, D.E. Adaptation to Flooding in Upland and Lowland Ecotypes of Cyperus rotundus, a Troublesome Sedge Weed of Rice: Tuber Morphology and Carbohydrate Metabolism. Ann. Bot. 2009, 103, 295–302. [Google Scholar] [CrossRef]
- Liu, B.; Jiang, M.; Tong, S.; Zhang, W.; Wu, H.; Liu, Y.; Lu, X. Differential Flooding Impacts on Echinochloa caudata and Scirpus planiculmis: Implications for Weed Control in Wetlands. Wetlands 2016, 36, 979–984. [Google Scholar] [CrossRef]
- Rocha, M.; Licausi, F.; Araújo, W.L.; Nunes-Nesi, A.; Sodek, L.; Fernie, A.R.; van Dongen, J.T. Glycolysis and the Tricarboxylic Acid Cycle Are Linked by Alanine Aminotransferase during Hypoxia Induced by Waterlogging of Lotus japonicus. Plant. Physiol. 2010, 152, 1501–1513. [Google Scholar] [CrossRef]
- He, D.; Zhang, H.; Yang, P. The Mitochondrion-Located Protein OsB12D1 Enhances Flooding Tolerance during Seed Germination and Early Seedling Growth in Rice. Int. J. Mol. Sci. 2014, 15, 13461–13481. [Google Scholar] [CrossRef]
- Lasanthi-Kudahettige, R.; Magneschi, L.; Loreti, E.; Gonzali, S.; Licausi, F.; Novi, G.; Beretta, O.; Vitulli, F.; Alpi, A.; Perata, P. Transcript Profiling of the Anoxic Rice Coleoptile. Plant. Physiol. 2007, 144, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Sadiq, I.; Fanucchi, F.; Paparelli, E.; Alpi, E.; Bachi, A.; Alpi, A.; Perata, P. Proteomic Identification of Differentially Expressed Proteins in the Anoxic Rice Coleoptile. J. Plant. Physiol. 2011, 168, 2234–2243. [Google Scholar] [CrossRef] [PubMed]
- Shiono, K.; Ogawa, S.; Yamazaki, S.; Isoda, H.; Fujimura, T.; Nakazono, M.; Colmer, T.D. Contrasting Dynamics of Radial O2-Loss Barrier Induction and Aerenchyma Formation in Rice Roots of Two Lengths. Ann. Bot. 2011, 107, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Watanabe, K.; Fukazawa, A.; Mori, H.; Abe, F.; Kawaguchi, K.; Oyanagi, A.; Nakazono, M. Ethylene and Reactive Oxygen Species Are Involved in Root Aerenchyma Formation and Adaptation of Wheat Seedlings to Oxygen-Deficient Conditions. J. Exp. Bot. 2014, 65, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.E. Aerenchyma Formation. New Phytol. 2004, 161, 35–49. [Google Scholar] [CrossRef]
- Dordas, C. Nonsymbiotic Hemoglobins and Stress Tolerance in Plants. Plant. Sci. 2009, 176, 433–440. [Google Scholar] [CrossRef]
- Herzog, M.; Pedersen, O. Partial versus Complete Submergence: Snorkelling Aids Root Aeration in Rumex palustris but Not in R. acetosa. Plant. Cell Environ. 2014, 37, 2381–2390. [Google Scholar] [CrossRef]
- Van Veen, H.; Mustroph, A.; Barding, G.A.; Vergeer-van Eijk, M.; Welschen-Evertman, R.A.M.; Pedersen, O.; Visser, E.J.W.; Larive, C.K.; Pierik, R.; Bailey-Serres, J.; et al. Two Rumex Species from Contrasting Hydrological Niches Regulate Flooding Tolerance through Distinct Mechanisms. Plant. Cell 2013, 25, 4691–4707. [Google Scholar] [CrossRef]
- Tsai, K.J.; Chou, S.J.; Shih, M.C. Ethylene Plays an Essential Role in the Recovery of Arabidopsis during Post-Anaerobiosis Reoxygenation. Plant. Cell Environ. 2014, 37, 2391–2405. [Google Scholar] [CrossRef]
- Pfeifer, T.A.; Grigliatti, T.A. Minireview Future Perspectives on Insect Pest Management: Engineering. J. Invertebr. Pathol. 2011, 119, 109–119. [Google Scholar]
- Liu, C.; Li, J.; Gao, J.; Shen, Z.; Lu, B.R.; Lin, C. A Built-in Mechanism to Mitigate the Spread of Insect-Resistance and Herbicide-Tolerance Transgenes into Weedy Rice Populations. PLoS ONE 2012, 7, 1–7. [Google Scholar] [CrossRef]
- Goulart, I.C.G.R.; Menezes, V.G.; Bortoly, E.D.; Kupas, V.; Merotto, A. Detecting Gene Flow from Als-Resistant Hybrid and Inbred Rice To Weedy Rice Using Single Plant Pollen Donors. Exp. Agric. 2016, 52, 237–250. [Google Scholar] [CrossRef]
- Shivrain, V.K.; Burgos, N.R.; Sales, M.A.; Mauromoustakos, A.; Gealy, D.R.; Smith, K.L.; Black, H.L.; Jia, M. Factors Affecting the Outcrossing Rate between ClearfieldTM Rice and Red Rice (Oryza sativa). Weed Sci. 2009, 57, 394–403. [Google Scholar] [CrossRef]
- San Miguel, K.; Scott, J.G. The next Generation of Insecticides: DsRNA is Stable as a Foliar-Applied Insecticide. Pest. Manag. Sci. 2016, 72, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Sherman, J.H.; Munyikwa, T.; Chan, S.Y.; Petrick, J.S.; Witwer, K.W.; Choudhuri, S. RNAi Technologies in Agricultural Biotechnology: The Toxicology Forum 40th Annual Summer Meeting. Regul. Toxicol. Pharmacol. 2015, 73, 671–680. [Google Scholar] [CrossRef]
- Mitter, N.; Worrall, E.A.; Robinson, K.E.; Li, P.; Jain, R.G.; Taochy, C.; Fletcher, S.J.; Carroll, B.J.; Lu, G.Q.; Xu, Z.P. Clay Nanosheets for Topical Delivery of RNAi for Sustained Protection against Plant Viruses. Nat. Plants 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Robert, D.S.; Ivashuta, S.; Liu, H.; Wang, D.; Feng, P.C.C.; Kouranov, A.Y.; Andersen, S.E. Method for Controlling Herbicide-Resistant Plants. U.S. Patent 9121022B2, 1 September 2015. [Google Scholar]
- Goldsbrough, P. Is RNA Interference the next Big Thing in Crop Protection? In Proceedings of the 28th Annual Integrated Crop Management Conference, Ames, IA, USA, 1 December 2018. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaspary, T.E.; Roma-Burgos, N.; Merotto, A., Jr. Snorkeling Strategy: Tolerance to Flooding in Rice and Potential Application for Weed Management. Genes 2020, 11, 975. https://doi.org/10.3390/genes11090975
Kaspary TE, Roma-Burgos N, Merotto A Jr. Snorkeling Strategy: Tolerance to Flooding in Rice and Potential Application for Weed Management. Genes. 2020; 11(9):975. https://doi.org/10.3390/genes11090975
Chicago/Turabian StyleKaspary, Tiago Edu, Nilda Roma-Burgos, and Aldo Merotto, Jr. 2020. "Snorkeling Strategy: Tolerance to Flooding in Rice and Potential Application for Weed Management" Genes 11, no. 9: 975. https://doi.org/10.3390/genes11090975
APA StyleKaspary, T. E., Roma-Burgos, N., & Merotto, A., Jr. (2020). Snorkeling Strategy: Tolerance to Flooding in Rice and Potential Application for Weed Management. Genes, 11(9), 975. https://doi.org/10.3390/genes11090975





