Contactin 1: An Important and Emerging Oncogenic Protein Promoting Cancer Progression and Metastasis
Abstract
1. Introduction
2. The Role of CNTN1 in Neuronal Development
3. Relevance of CNTN1 in Tumorigenesis
3.1. Lung Cancer
3.2. Gastric and Upper Gastrointestinal Cancers
3.3. Prostate Cancer
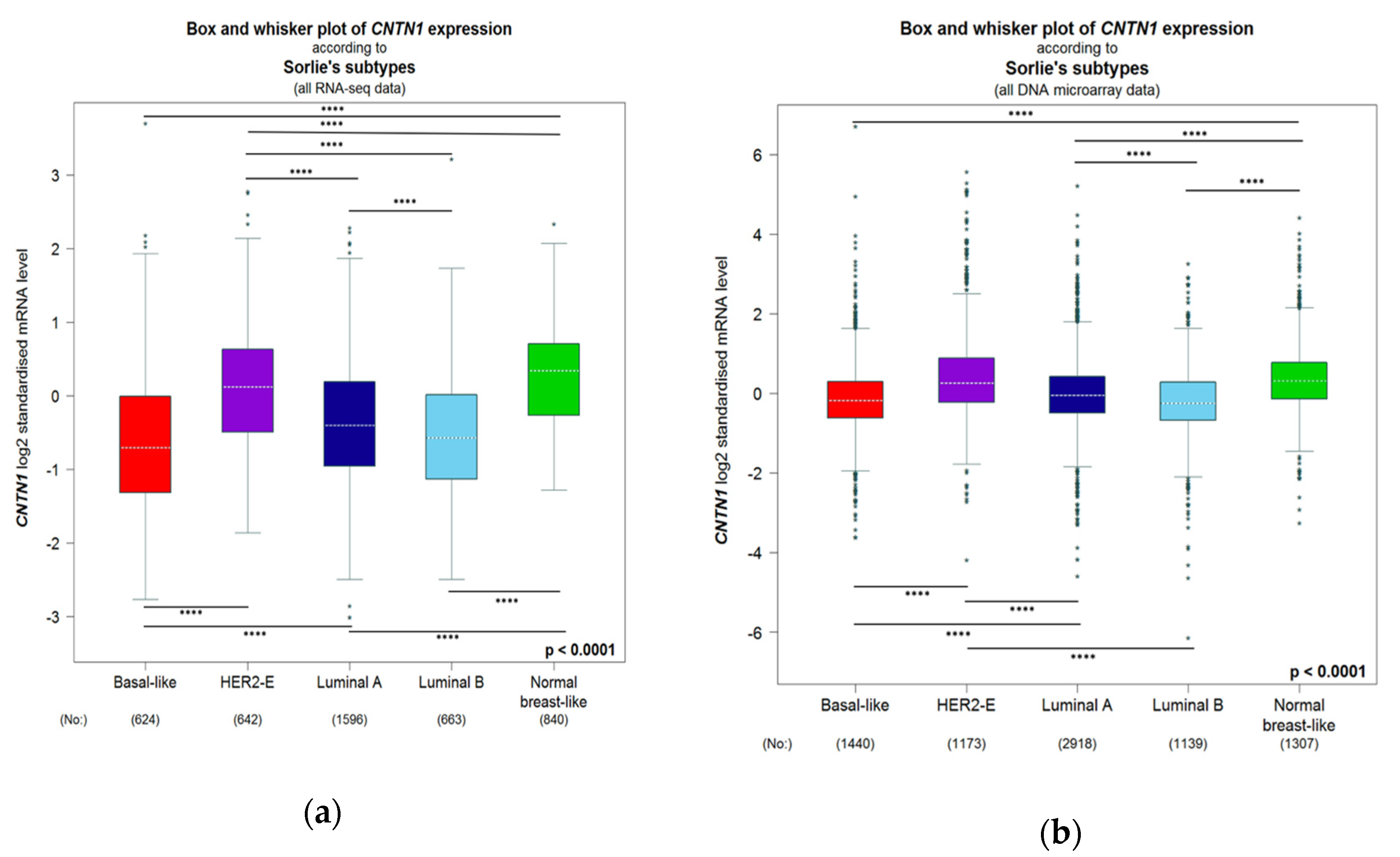
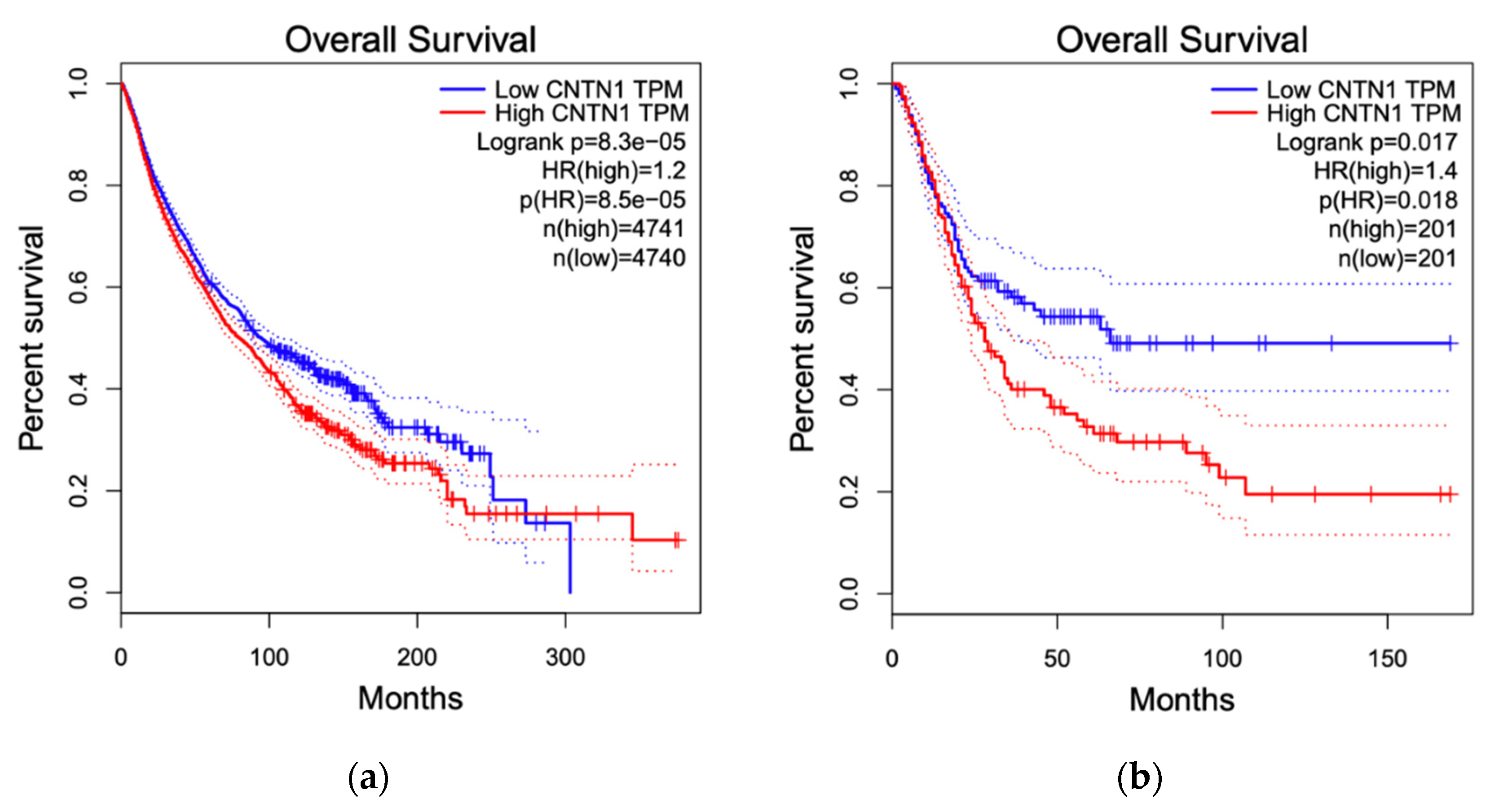
3.4. Breast Cancer
3.5. Other Cancers
4. Perspectives
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Makrilia, N.; Kollias, A.; Syrigos, L.M.K. Cell Adhesion Molecules: Role and clinical significance in cancer. Cancer Investig. 2009, 27, 1023–1037. [Google Scholar] [CrossRef] [PubMed]
- Cavallaro, U.; Christofori, G. Multitasking in tumor progression: Signaling functions of cell adhesion molecules. Ann. N. Y. Acad. Sci. 2004, 1014, 58–66. [Google Scholar] [CrossRef]
- Mehlen, P.; Puisieux, A. Metastasis: A question of life or death. Nat. Rev. Cancer 2006, 6, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Syrigos, K.; Karayiannakis, A.J. Adhesion molecules as targets for the treatment of neoplastic diseases. Curr. Pharm. Des. 2006, 12, 2849–2861. [Google Scholar] [CrossRef]
- Low, M.G. Glycosyl-phosphatidylinositol: A versatile anchor for cell surface proteins 1. FASEB J. 1989, 3, 1600–1608. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.-H.; Yu, J.-W.; Jiang, B.-J. Contactin 1: A potential therapeutic target and biomarker in gastric cancer. World J. Gastroenterol. 2015, 21, 9707–9716. [Google Scholar] [CrossRef]
- Shi, K.; Xu, N.; Yang, C.; Wang, L.; Pan, W.; Zheng, C.; Fan, L. Contactin 1 as a potential biomarker promotes cell proliferation and invasion in thyroid cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 12473–12481. [Google Scholar]
- Zhang, R.; Xie, L.; Liu, C.; Yang, H.; Lin, H.; Zhang, Q.; Tang, W.; Ji, F.; Sun, S. Contactin-1: A promising progression biomarker and therapeutic target of carcinoma. Minerva Med. 2017, 108, 193–195. [Google Scholar]
- Gennarini, G.; Bizzoca, A.; Picocci, S.; Puzzo, D.; Corsi, P.; Furley, A.J. The role of Gpi-anchored axonal glycoproteins in neural development and neurological disorders. Mol. Cell. Neurosci. 2017, 81, 49–63. [Google Scholar] [CrossRef]
- Bizzoca, A.; Corsi, P.; Gennarini, G. The mouse F3/contactin glycoprotein. Cell Adhes. Migr. 2009, 3, 53–63. [Google Scholar] [CrossRef]
- Bizzocaa, A.; Corsia, P.; Polizzia, A.; Pintoa, M.F.; Xenaki, D. F3/Contactin acts as a modulator of neurogenesis during cerebral cortex development. Dev. Biol. 2012, 365, 133–151. [Google Scholar] [CrossRef][Green Version]
- Çolakoğlu, G.; Bergstrom-Tyrberg, U.; Berglund, E.O.; Ranscht, B. Contactin-1 regulates myelination and nodal/paranodal domain organization in the central nervous system. Proc. Natl. Acad. Sci. USA 2014, 111, E394–E403. [Google Scholar] [CrossRef] [PubMed]
- Dityatev, A.; Bukalo, O.; Schachner, M. Modulation of synaptic transmission and plasticity by cell adhesion and repulsion molecules. Neuron Glia Boil. 2008, 4, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.-D.; Ang, B.-T.; Karsak, M.; Hu, W.-P.; Cui, X.-Y.; Duka, T.; Takeda, Y.; Chia, W.; Sankar, N.; Ng, Y.-K.; et al. F3/contactin acts as a functional ligand for notch during oligodendrocyte maturation. Cell 2003, 115, 163–175. [Google Scholar] [CrossRef]
- Berglund, E.O.; Murai, K.K.; Fredette, B.; Sekerková, G.; Marturano, B.; Weber, L.; Mugnaini, E.; Ranscht, B. Ataxia and abnormal cerebellar microorganization in mice with ablated contactin gene expression. Neuron 1999, 24, 739–750. [Google Scholar] [CrossRef]
- Shimoda, Y.; Watanabe, K. Contactins: Emerging key roles in the development and function of the nervous system. Cell Adh. Migr. 2009, 3, 64–70. [Google Scholar] [CrossRef]
- Bouyain, S.; Watkins, D.J. The protein tyrosine phosphatases PTPRZ and PTPRG bind to distinct members of the contactin family of neural recognition molecules. Proc. Natl. Acad. Sci. USA 2010, 107, 2443–2448. [Google Scholar] [CrossRef]
- Peles, E.; Nativ, M.; Campbell, P.L.; Sakurai, T.; Martinez, R.; Lev, S.; O Clary, D.; Schilling, J.; Barnea, G.; Plowman, G.D.; et al. The carbonic anhydrase domain of receptor tyrosine phosphatase beta is a functional ligand for the axonal cell recognition molecule contactin. Cell 1995, 82, 251–260. [Google Scholar] [CrossRef]
- Sakurai, T.; Lustig, M.; Nativ, M.; Hemperly, J.J.; Schlessinger, J.; Peles, E.; Grumet, M. Induction of neurite outgrowth through contactin and nr-CAM by extracellular regions of glial receptor tyrosine phosphatase β. J. Cell Boil. 1997, 136, 907–918. [Google Scholar] [CrossRef]
- Parent, A.-S.; Mungenast, A.E.; Lomniczi, A.; Sandau, U.S.; Peles, E.; Bosch, M.A.; Rønnekleiv, O.K.; Ojeda, S.R. A contactin–receptor-like protein tyrosine phosphatase β complex mediates adhesive communication between astroglial cells and gonadotrophin-releasing hormone neurones. J. Neuroendocr. 2007, 19, 847–859. [Google Scholar] [CrossRef]
- Lamprianou, S.; Chatzopoulou, E.; Thomas, J.-L.; Bouyain, S.; Harroch, S. A complex between contactin-1 and the protein tyrosine phosphatase PTPRZ controls the development of oligodendrocyte precursor cells. Proc. Natl. Acad. Sci. USA 2011, 108, 17498–17503. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.-D.; Ma, Q.-H.; Gennarini, G.; Xiao, Z.-C. Cross-talk between F3/contactin and notch at axoglial interface: A role in oligodendrocyte development. Dev. Neurosci. 2006, 28, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Umemori, H.; Satot, S.; Yagi, T.; Aizawal, S.; Yamamoto, T. Initial events of myelination involve Fyn tyrosine kinase signalling. Nature 1994, 367, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T. The role of nr. CAM in neural development and disorders—Beyond a simple glue in the brain. Mol. Cell. Neurosci. 2012, 49, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Pesheva, P.; Gennarini, G.; Goridis, C.; Schachner, M. The F3/11 cell adhesion molecule mediates the repulsion of neurons by the extracellular matrix glycoprotein J1-160/180. Neuron 1993, 10, 69–82. [Google Scholar] [CrossRef]
- Chen, Y.A.; Lu, I.L.; Tsai, J.W. Contactin-1/F3 regulates neuronal migration and morphogenesis through modulating rhoa activity. Front Mol. Neurosci. 2018, 11, 422. [Google Scholar] [CrossRef]
- Shimazaki, K.; Hosoya, H.; Takeda, Y.; Kobayashi, S.; Watanabe, K. Age-related decline of F3/contactin in rat hippocampus. Neurosci. Lett. 1998, 245, 117–120. [Google Scholar] [CrossRef]
- Compton, A.G.; Albrecht, D.E.; Seto, J.; Cooper, S.T.; Ilkovski, B.; Jones, K.J.; Challis, D.; Mowat, D.; Ranscht, B.; Bahlo, M.; et al. Mutations in Contactin-1, a Neural adhesion and neuromuscular junction protein, cause a familial form of lethal congenital myopathy. Am. J. Hum. Genet. 2008, 83, 714–724. [Google Scholar] [CrossRef]
- Falk, J.; Bonnon, C.A.; Girault, J.; Faivre-Sarrailh, C. F3/contactin, a neuronal cell adhesion molecule implicated in axogenesis and myelination. Boil. Cell 2002, 94, 327–334. [Google Scholar] [CrossRef]
- Prag, S.A.; Lepekhin, E.; Kolkova, K.; Hartmann-Petersen, R.; Kawa, A.; Walmod, P.S.; Belman, V.; Gallagher, H.C.; Berezin, V.; Bock, E.; et al. NCAM regulates cell motility. J. Cell Sci. 2002, 115, 283–292. [Google Scholar]
- Lehembre, F.; Yilmaz, M.; Wicki, A.; Schomber, T.; Strittmatter, K.; Ziegler, D.; Kren, A.; Went, P.; Derksen, P.W.B.; Berns, A.; et al. NCAM-induced focal adhesion assembly: A functional switch upon loss of E-cadherin. EMBO J. 2008, 27, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Blaheta, R.A.; Hundemer, M.; Mayer, G.; Vogel, J.-U.; Kornhuber, B.; Činátl, J.; Markus, B.H.; Driever, P.H.; Cinatl, J., Jr. Expression Level of Neural Cell Adhesion Molecule (NCAM) Inversely Correlates with the Ability of Neuroblastoma Cells to Adhere to Endothelium In Vitro. Cell Commun. Adhes. 2002, 9, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Blaheta, R.A.; Daher, F.H.; Michaelis, M.; Hasenberg, C.; Weich, E.M.; Jonas, D.; Kotchetkov, R.; Doerr, H.W.; Cinatl, J. Chemoresistance induces enhanced adhesion and transendothelial penetration of neuroblastoma cells by down-regulating NCAM surface expression. BMC Cancer 2006, 6, 294. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Santón, A.; Bellas, C. Neural cell adhesion molecule expression in plasma cells in bone marrow biopsies and aspirates allows discrimination between multiple myeloma, monoclonal gammopathy of uncertain significance and polyclonal plasmacytosis. Histopathology 2004, 44, 375–380. [Google Scholar] [CrossRef]
- Bataille, R.; Jégo, G.; Robillard, N.; Barillé-Nion, S.; Harousseau, J.-L.; Moreau, P.; Amiot, M.; Pellat-Deceunynck, C. The phenotype of normal, reactive and malignant plasma cells. Identification of “many and multiple myelomas” and of new targets for myeloma therapy. Haematologica 2006, 91, 1234–1240. [Google Scholar]
- Raspadori, D.; Damiani, D.; Lenoci, M.; Rondelli, D.; Testoni, N.; Nardi, G.; Sestigiani, C.; Mariotti, C.; Birtolo, S.; Tozzi, M.; et al. CD56 antigenic expression in acute myeloid leukemia identifies patients with poor clinical prognosis. Leukemia 2001, 15, 1161–1164. [Google Scholar] [CrossRef]
- Yang, D.-H.; Lee, J.-J.; Mun, Y.-C.; Shin, H.-J.; Kim, Y.-K.; Cho, S.-H.; Chung, I.-J.; Seong, C.-M.; Kim, H.-J. Predictable prognostic factor of CD56 expression in patients with acute myeloid leukemia with t(8:21) after high dose cytarabine or allogeneic hematopoietic stem cell transplantation. Am. J. Hematol. 2007, 82, 1–5. [Google Scholar] [CrossRef]
- Kameda, K.; Shimada, H.; Ishikawa, T.; Takimoto, A.; Momiyama, N.; Hasegawa, S.; Misuta, K.; Nakano, A.; Nagashima, Y.; Ichikawa, Y. Expression of highly polysialylated neural cell adhesion molecule in pancreatic cancer neural invasive lesion. Cancer Lett. 1999, 137, 201–207. [Google Scholar] [CrossRef]
- Tezel, E.; Kawase, Y.; Takeda, S.; Oshima, K.; Nakao, A. Expression of Neural Cell Adhesion Molecule in Pancreatic Cancer. Pancreas 2001, 22, 122–125. [Google Scholar] [CrossRef]
- Naito, Y.; Kinoshita, H.; Okabe, Y.; Kawahara, R.; Sakai, T.; Suga, H.; Arikawa, S.; Oshima, K.; Kojiro, M. CD56 (NCAM) expression in pancreatic carcinoma and the surrounding pancreatic tissue. Kurume Med. J. 2006, 53, 59–62. [Google Scholar] [CrossRef]
- Miyahara, R.; Tanaka, F.; Nakagawa, T.; Matsuoka, K.; Isii, K.; Wada, H. Expression of neural cell adhesion molecules (polysialylated form of neural cell adhesion molecule and L1-cell adhesion molecule) on resected small cell lung cancer specimens: In relation to proliferation state. J. Surg. Oncol. 2001, 77, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Berglund, E.O.; Ranscht, B. Molecular Cloning and in Situ Localization of the Human Contactin Gene (CNTN1) on Chromosome 12q11-q12. Genomics 1994, 21, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Acampora, D.; D’Esposito, M.; Faiella, A.; Pannese, M.; Migliaccio, E.; Morelli, F.; Stornaiuolo, A.; Nigro, V.; Simeone, A.; Boncinelli, E. The human HOX gene family. Nucleic Acids Res. 1989, 17, 10385–10402. [Google Scholar] [CrossRef]
- Sosnoski, D.M.; Emanuel, B.S.; Hawkins, A.L.; Van Tuinen, P.; Ledbetter, D.H.; Nussbaum, R.L.; Kaos, F.T.; Schwartz, E.; Phillips, D.; Bennett, J.S. Chromosomal localization of the genes for the vitronectin and fibronectin receptors alpha subunits and for platelet glycoproteins IIb and IIIa. J. Clin. Investig. 1988, 81, 1993–1998. [Google Scholar] [CrossRef] [PubMed]
- Arheden, K.; Mandahl, N.; Heim, S.; Mitelman, F. In situ hybridization localizes the human type II alpha 1 collagen gene (COL2A1) to 12q13. Hereditas 2008, 110, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Kraus, M.H.; Issing, W.; Miki, T.; Popescu, N.C.; Aaronson, S.A. Isolation and characterization of ERBB3, a third member of the ERBB/epidermal growth factor receptor family: Evidence for overexpression in a subset of human mammary tumors. Proc. Natl. Acad. Sci. USA 1989, 86, 9193–9197. [Google Scholar] [CrossRef]
- Arheden, K.; Mandahl, N.; Strömbeck, B.; Isaksson, M.; Mitelman, F. Chromosome localization of the human oncogene INT1 to 12q13 by in situ hybridization. Cytogenet Cell Genet. 1988, 47, 86–87. [Google Scholar] [CrossRef]
- Desgrosellier, S.J.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef]
- Stetten, G.; Sroka, B.; Corson, V.L.; Opitz, J.M.; Reynolds, J.F.; Norbury-Glaser, M. Fragile site in chromosome 12 in a patient with two miscarriages. Am. J. Med Genet. 1988, 31, 521–525. [Google Scholar] [CrossRef]
- Mitelman, F. Catalog of Chromosome Abberrations in Cancer, 4th ed.; Wiley-Liss: New York, NY, USA, 1991. [Google Scholar]
- Reifenberger, G.; Reifenberger, J.; Ichimura, K.; Collins, V.P. Amplification at 12q13–14 in human malignant gliomas is frequently accompanied by loss of heterozygosity at loci proximal and distal to the amplification site. Cancer Res. 1995, 55, 731–734. [Google Scholar]
- Warr, T.J.; Ward, S.; Burrows, J.; Harding, B.; Wilkins, P.; Harkness, W.; Hayward, R.; Darling, J.; Thomas, D. Identification of extensive genomic loss and gain by comparative genomic hybridisation in malignant astrocytoma in children and young adults. Genes Chromosom. Cancer 2001, 31, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Su, J.-L.; Yang, P.-C.; Shih, J.-Y.; Yang, C.-Y.; Wei, L.-H.; Hsieh, C.-Y.; Chou, C.-H.; Jeng, Y.-M.; Wang, M.-Y.; Chang, K.-J.; et al. The VEGF-C/Flt-4 axis promotes invasion and metastasis of cancer cells. Cancer Cell 2006, 9, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Shigetomi, S.; Imanishi, Y.; Shibata, K.; Sakai, N.; Sakamoto, K.; Fujii, R.; Habu, N.; Otsuka, K.; Sato, Y.; Watanabe, Y.; et al. VEGF-C/Flt-4 axis in tumor cells contributes to the progression of oral squamous cell carcinoma via upregulating VEGF-C itself and contactin-1 in an autocrine manner. Am. J. Cancer Res. 2018, 8, 2046–2063. [Google Scholar] [PubMed]
- Wang, J.; Huang, Y.; Zhang, J.; Wei, Y.; Mahoud, S.; Bakheet, A.M.H.; Tang, J. Pathway-related molecules of VEGFC/D-VEGFR3/NRP2 axis in tumor lymphangiogenesis and lymphatic metastasis. Clin. Chim. Acta 2016, 461, 165–171. [Google Scholar] [CrossRef]
- Hung, Y.-H.; Hung, W.-C. 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) enhances invasiveness of lung cancer cells by up-regulating contactin-1 via the α7 nicotinic acetylcholine receptor/ERK signaling pathway. Chem. Interact. 2009, 179, 154–159. [Google Scholar] [CrossRef]
- Xu, S.; Lam, S.-K.; Cheng, P.N.-M.; Ho, J. Contactin 1 modulates pegylated arginase resistance in small cell lung cancer through induction of epithelial-mesenchymal transition. Sci. Rep. 2019, 9, 12030. [Google Scholar] [CrossRef]
- Yan, J.; Wong, N.; Hung, C.L.; Chen, W.X.-Y.; Tang, D. Contactin-1 reduces e-cadherin expression via activating akt in lung cancer. PLoS ONE 2013, 8, e65463. [Google Scholar] [CrossRef]
- Zhang, R.; Sun, S.; Ji, F.; Liu, C.; Lin, H.; Xie, L.; Yang, H.; Tang, W.; Zhou, Y.; Xu, J.; et al. CNTN-1 enhances chemoresistance in human lung adenocarcinoma through induction of epithelial-mesenchymal transition by targeting the PI3K/Akt pathway. Cell. Physiol. Biochem. 2017, 43, 465–480. [Google Scholar] [CrossRef]
- Yu, J.-W.; Wu, S.-H.; Lu, R.-Q.; Wu, J.-G.; Ni, X.-C.; Zhou, G.-C.; Jiang, H.-G.; Zheng, L.-H.; Li, X.-Q.; Du, G.-Y.; et al. Expression and significances of contactin-1 in human gastric cancer. Gastroenterol. Res. Pr. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Zhang, D.; Zhou, S.; Liu, B. Identification and validation of an individualized emt-related prognostic risk score formula in gastric adenocarcinoma patients. BioMed Res. Int. 2020, 2020, 7082408. [Google Scholar] [CrossRef]
- Xu, S.; Lam, S.-K.; Cheng, P.N.-M.; Ho, J. Recombinant human arginase induces apoptosis through oxidative stress and cell cycle arrest in small cell lung cancer. Cancer Sci. 2018, 109, 3471–3482. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Yang, X.; Zhao, T.; Du, H.; Wang, T.; Zhong, S.; Yang, B.; Li, H. Upregulation of contactin-1 expression promotes prostate cancer progression. Oncol. Lett. 2019, 19, 1611–1618. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Ojo, D.; Kapoor, A.; Lin, X.; Pinthus, J.H.; Aziz, T.; Bismar, T.A.; Wei, F.; Wong, N.; De Melo, J.; et al. Neural cell adhesion protein CNTN1 promotes the metastatic progression of prostate cancer. Cancer Res. 2016, 76, 1603–1614. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-C.; Zhao, J.; Hu, C.-E.; Gan, J.; Zhang, W.; Huang, G. Comprehensive analysis of vascular endothelial growth factor-c related factors in stomach cancer. Asian Pac. J. Cancer Prev. 2014, 15, 1925–1929. [Google Scholar] [CrossRef][Green Version]
- Liu, P.; Zhou, J.; Zhu, H.; Xie, L.; Wang, F.; Liu, B.; Shen, W.; Ye, W.; Xiang, B.; Zhu, X.; et al. VEGF-C promotes the development of esophageal cancer via regulating CNTN-1 expression. Cytokine 2011, 55, 8–17. [Google Scholar] [CrossRef]
- Tsai, K.-H.; Hsien, H.-H.; Chen, L.-M.; Ting, W.-J.; Yang, Y.-S.; Kuo, C.-H.; Tsai, C.-H.; Tsai, F.J.; Tsai, H.J.; Huang, E.-J. Rhubarb inhibits hepatocellular carcinoma cell metastasis via GSK-3-β activation to enhance protein degradation and attenuate nuclear translocation of β-catenin. Food Chem. 2013, 138, 278–285. [Google Scholar] [CrossRef]
- Li, G.-Y.; Huang, M.; Pan, T.-T.; Jia, W.-D. Expression and prognostic significance of contactin 1 in human hepatocellular carcinoma. OncoTargets Ther. 2016, 9, 387–394. [Google Scholar] [CrossRef]
- Eckerich, C.; Zapf, S.; Ulbricht, U.; Müller, S.; Fillbrandt, R.; Westphal, M.; Lamszus, K. Contactin is expressed in human astrocytic gliomas and mediates repulsive effects. Glia 2005, 53, 1–12. [Google Scholar] [CrossRef]
- Liu, P.; Chen, S.; Wu, W.; Liu, B.; Shen, W.; Wang, F.; He, X.; Zhang, S. Contactin-1 (CNTN-1) Overexpression is Correlated with Advanced Clinical Stage and Lymph Node Metastasis in Oesophageal Squamous Cell Carcinomas. Jpn. J. Clin. Oncol. 2012, 42, 612–618. [Google Scholar] [CrossRef]
- Wu, H.-M.; Cao, W.; Ye, D.; Ren, G.-X.; Wu, Y.-N.; Guo, W. Contactin 1 (CNTN1) expression associates with regional lymph node metastasis and is a novel predictor of prognosis in patients with oral squamous cell carcinoma. Mol. Med. Rep. 2012, 6, 265–270. [Google Scholar] [CrossRef]
- Zhang, R.; Yao, W.; Qian, P.; Li, Y.; Jiang, C.; Ao, Z.; Qian, G.; Wang, C.; Wu, G.; Li, J.; et al. Increased sensitivity of human lung adenocarcinoma cells to cisplatin associated with downregulated contactin-1. Biomed. Pharmacother. 2015, 71, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Zhang, Y.; Li, C.; Xu, P.; Gao, Y.; Xu, Y. CNTN-1 promotes docetaxel resistance and epithelial-to-mesenchymal transition via the PI3K/Akt signaling pathway in prostate cancer. Arch. Med. Sci. 2020. [Google Scholar] [CrossRef]
- Kaushal, V.; Mukunyadzi, P.; A Dennis, R.; Siegel, E.R.; E Johnson, D.; Kohli, M. Stage-specific characterization of the vascular endothelial growth factor axis in prostate cancer: Expression of lymphangiogenic markers is associated with advanced-stage disease. Clin. Cancer Res. 2005, 11, 584–593. [Google Scholar] [PubMed]
- Li, Q.; Dong, X.; Gu, W.; Qiu, X.; Wang, E. Clinical significance of co-expression of VEGF-C and VEGFR-3 in non-small cell lung cancer. Chin. Med J. 2003, 116, 727–730. [Google Scholar] [PubMed]
- Witte, D.; Thomas, A.; Ali, N.; Carlson, N.; Younes, M. Expression of the vascular endothelial growth factor receptor-3 (VEGFR-3) and its ligand VEGF-C in human colorectal adenocarcinoma. Anticancer. Res. 2002, 22, 1463–1466. [Google Scholar]
- Neuchrist, C.; Erovic, B.M.; Handisurya, A.; Fischer, M.B.; Steiner, G.E.; Hollemann, D.; Gedlicka, C.; Saaristo, A.; Burian, M. Vascular endothelial growth factor C and vascular endothelial growth factor receptor 3 expression in squamous cell carcinomas of the head and neck. Head Neck 2003, 25, 464–474. [Google Scholar] [CrossRef]
- Van Trappen, P.O.; Steele, D.; Lowe, D.G.; Baithun, S.; Beasley, N.; Thiele, W.; Weich, H.; Krishnan, J.; Shepherd, J.H.; Pepper, M.S.; et al. Expression of vascular endothelial growth factor (VEGF)-C and VEGF-D, and their receptor VEGFR-3, during different stages of cervical carcinogenesis. J. Pathol. 2003, 201, 544–554. [Google Scholar] [CrossRef]
- Zhang, Y.; Weinberg, R.A. Epithelial-to-mesenchymal transition in cancer: Complexity and opportunities. Front. Med. 2018, 12, 361–373. [Google Scholar] [CrossRef]
- Ksiazkiewicz, M.; Markiewicz, A.; Zaczek, A.J. Epithelial-mesenchymal transition: A hallmark in metastasis formation linking circulating tumor cells and cancer stem cells. Pathobiolohy 2012, 79, 195–208. [Google Scholar] [CrossRef]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial–mesenchymal transition. Nat. Rev. Mol. Cell Boil. 2014, 15, 178–196. [Google Scholar] [CrossRef]
- Molina, J.R.; Agarwal, N.K.; Morales, F.C.; Hayashi, Y.; Aldape, K.D.; Cote, G.; Georgescu, M.-M. PTEN, NHERF1 and PHLPP form a tumor suppressor network that is disabled in glioblastoma. Oncogene 2011, 31, 1264–1274. [Google Scholar] [CrossRef] [PubMed]
- Batlle, E.; Clevers, H. Cancer stem cells revisited. Nat. Med. 2017, 23, 1124–1134. [Google Scholar] [CrossRef] [PubMed]
- Chae, Y.C.; Kim, J.H. Cancer stem cell metabolism: Target for cancer therapy. BMB Rep. 2018, 51, 319–326. [Google Scholar] [CrossRef]
- Eun, K.; Ham, S.W.; Kim, H. Cancer stem cell heterogeneity: Origin and new perspectives on CSC targeting. BMB Rep. 2017, 50, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Colombel, M.; Eaton, C.L.; Hamdy, F.; Ricci, E.; Van Der Pluijm, G.; Cecchini, M.; Mege-Lechevallier, F.; Clézardin, P.; Thalmann, G. Increased expression of putative cancer stem cell markers in primary prostate cancer is associated with progression of bone metastases. Prostate 2011, 72, 713–720. [Google Scholar] [CrossRef]
- Wang, Z.A.; Mitrofanova, A.; Bergren, S.K.; Abate-Shen, C.; Cardiff, R.D.; Califano, A.; Shen, M.M. Lineage analysis of basal epithelial cells reveals their unexpected plasticity and supports a cell-of-origin model for prostate cancer heterogeneity. Nature 2013, 15, 274–283. [Google Scholar] [CrossRef]
- Chen, N.; He, S.; Geng, J.; Song, Z.-J.; Han, P.-H.; Qin, J.; Zhao, Z.; Song, Y.-C.; Wang, H.-X.; Dang, C.-X. Overexpression of Contactin 1 promotes growth, migration and invasion in Hs578T breast cancer cells. BMC Cell Boil. 2018, 19, 5. [Google Scholar] [CrossRef]
- Jézéquel, P.; Campone, M.; Gouraud, W.; Guérin-Charbonnel, C.; Leux, C.; Ricolleau, G.; Campion, L. bc-GenExMiner: An easy-to-use online platform for gene prognostic analyses in breast cancer. Breast Cancer Res. Treat. 2011, 131, 765–775. [Google Scholar] [CrossRef]
- Jézéquel, P.; Frénel, J.-S.; Campion, L.; Guérin-Charbonnel, C.; Gouraud, W.; Ricolleau, G.; Campone, M. bc-GenExMiner 3.0: New mining module computes breast cancer gene expression correlation analyses. Database 2013, 2013, bas060. [Google Scholar] [CrossRef]
- Breast Cancer Gene-Expression Miner v4.5. 22 June 2020. Available online: http://bcgenex.ico.unicancer.fr (accessed on 15 June 2020).
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; Van De Rijn, M.; Jeffrey, S.S.; A Rees, C.; Pollack, J.R.; Ross, U.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Sørlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar]
- Haakensen, V.D.; Lingjærde, O.C.; Lüders, T.; Riis, M.L.; Prat, A.; Troester, M.A.; Holmen, M.M.; Frantzen, J.O.; Romundstad, L.; Navjord, D.; et al. Gene expression profiles of breast biopsies from healthy women identify a group with claudin-low features. BMC Med Genom. 2011, 4, 77. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Oprea-Ilies, G.M.; Krishnamurti, U. New developments in breast cancer and their impact on daily practice in pathology. Arch. Pathol. Lab. Med. 2017, 141, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Yersal, Ö.; Barutca, S. Biological subtypes of breast cancer: Prognostic and therapeutic implications. World J. Clin. Oncol. 2014, 5, 412–424. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Esposito, M.; Kang, Y. Bone metastasis and the metastatic niche. J. Mol. Med. 2015, 93, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Guise, T.A.; Kang, Y. The Biology of Bone Metastasis. Cold Spring Harb. Perspect. Med. 2018, 8, a031252. [Google Scholar] [CrossRef]
- Bagnall, P. Diagnosis and treatment of prostate cancer. Nurs. Times 2014, 110, 12–15. [Google Scholar]
- Schweppe, R.E.; Kerege, A.A.; Sharma, V.; Poczobutt, J.M.; Gutierrez-Hartmann, A.; Grzywa, R.L.; Haugen, B.R. Distinct genetic alterations in the mitogen-activated protein kinase pathway dictate sensitivity of thyroid cancer cells to mitogen-activated protein kinase kinase 1/2 inhibition. Thyroid 2009, 19, 825–835. [Google Scholar] [CrossRef]
- Grubbs, E.G.; Ng, P.K.-S.; Bui, J.; Busaidy, N.L.; Chen, K.; Lee, J.E.; Lu, X.; Lu, H.; Meric-Bernstam, F.; Mills, G.B.; et al. RET fusion as a novel driver of medullary thyroid carcinoma. J. Clin. Endocrinol. Metab. 2014, 100, 788–793. [Google Scholar] [CrossRef]
- Jeong, W.-J.; Mo, J.H.; Park, M.W.; Choi, I.J.; An, S.-Y.; Jeon, E.-H.; Ahn, S.-H. Sunitinib inhibits papillary thyroid carcinoma with RET/PTC rearrangement but not BRAF mutation. Cancer Boil. Ther. 2011, 12, 458–465. [Google Scholar] [CrossRef]
- Shiozaki, A.; Shen-Tu, G.; Bai, X.; Iitaka, D.; De Falco, V.; Santoro, M.; Keshavjee, S.; Liu, M. XB130 mediates cancer cell proliferation and survival through multiple signaling events downstream of akt. PLoS ONE 2012, 7, e43646. [Google Scholar] [CrossRef] [PubMed]
- Łuczkowska, K.; Rogińska, D.; Litwińska, Z.; Paczkowska, E.; Schmidt, C.A.; Machaliński, B. Molecular Mechanisms of Bortezomib Action: Novel Evidence for the miRNA–mRNA Interaction Involvement. Int. J. Mol. Sci. 2020, 21, 350. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Zhao, R.; Ni, H.; Zhao, K.; He, Y.; Fang, S.; Chen, Q.-L. Molecule mechanisms of Ganoderma lucidum treated hepatocellular carcinoma based on the transcriptional profiles and miRNA-target network. Biomed. Pharmacother. 2020, 125, 110028. [Google Scholar] [CrossRef]
- Mauerer, A.; Roesch, A.; Hafner, C.; Stempfl, T.; Wild, P.; Meyer, S.; Landthaler, M.; Vogt, T. Identification of new genes associated with melanoma. Exp. Dermatol. 2011, 20, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Pinnix, C.C.; Lee, J.T.; Liu, Z.-J.; McDaid, R.; Balint, K.; Beverly, L.J.; Brafford, P.A.; Xiao, M.; Himes, B.; Zabierowski, S.E.; et al. Active Notch1 confers a transformed phenotype to primary human melanocytes. Cancer Res. 2009, 69, 5312–5320. [Google Scholar] [CrossRef]
- Das, S.; Teoh, S.L. Notch signalling pathways and their importance in the treatment of cancers. Curr. Drug Targets 2017, 18, 1. [Google Scholar]
- Takebe, N.; Miele, L.; Harris, P.J.; Jeong, W.; Bando, H.; Kahn, M.; Yang, S.X.; Ivy, S.P. Targeting Notch, Hedgehog, and Wnt pathways in cancer stem cells: Clinical update. Nat. Rev. Clin. Oncol. 2015, 12, 445–464. [Google Scholar] [CrossRef]
- Tang, Z.; Li, C.; Kang, B.; Gao, G.; Li, C.; Zhang, Z. GEPIA: A web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017, 45, W98–W102. [Google Scholar] [CrossRef]
- Tratar, U.L.; Horvat, S.; Cemazar, M. Transgenic mouse models in cancer research. Front. Oncol. 2018, 8, 268. [Google Scholar] [CrossRef] [PubMed]
- Nord, H.; Hartmann, C.; Andersson, R.; Menzel, U.; Pfeifer, S.; Piotrowski, A.; Bogdan, A.; Kloc, W.; Sandgren, J.; Olofsson, T.; et al. Characterization of novel and complex genomic aberrations in glioblastoma using a 32K BAC array. Neuro-Oncology 2009, 11, 803–818. [Google Scholar] [CrossRef]
- Kok-Sin, T.; Mokhtar, N.M.; Hassan, N.Z.A.; Sagap, I.; Rose, I.M.; Harun, R.; Jamal, R. Identification of diagnostic markers in colorectal cancer via integrative epigenomics and genomics data. Oncol. Rep. 2015, 34, 22–32. [Google Scholar] [CrossRef]
- Luo, L.; Tao, W.; He, T.; Li, L.-Y. Prediction of potential prognostic biomarkers in metastatic prostate cancer based on a competing endogenous RNA regulatory network. Res. Sq. 2020. preprint. [Google Scholar]
- Cury, S.S.; De Moraes, D.; Freire, P.P.; De Oliveira, G.; Marques, D.V.P.; Fernandez, G.J.; Dal-Pai-Silva, M.; Hasimoto, É.N.; Reis, P.; Rogatto, S.R.; et al. Tumor transcriptome reveals high expression of IL-8 in non-small cell lung cancer patients with low pectoralis muscle area and reduced survival. Cancers 2019, 11, 1251. [Google Scholar] [CrossRef] [PubMed]
- Harjunpää, H.; Asens, M.L.; Guenther, C.; Fagerholm, S.C. Cell adhesion molecules and their roles and regulation in the immune and tumor microenvironment. Front. Immunol. 2019, 10, 1078. [Google Scholar] [CrossRef]
- O’Day, S.; on the behalf of the CNTO 95 Investigators; Pavlick, A.; Loquai, C.; Lawson, D.; Gutzmer, R.; Richards, J.; Schadendorf, D.; Schadendorf, D.; Thompson, J.A.; et al. A randomised, phase II study of intetumumab, an anti-αv-integrin mAb, alone and with dacarbazine in stage IV melanoma. Br. J. Cancer 2011, 105, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Le Moulec, S.; Gimmi, C.; Bruns, R.; Straub, J.; Miller, K. Differential effect on bone lesions of targeting integrins: Randomized phase II trial of abituzumab in patients with metastatic castration-resistant prostate cancer. Clin. Cancer Res. 2016, 22, 3192–3200. [Google Scholar] [CrossRef]
- Kwan, B.H.; Zhu, E.F.; Tzeng, A.; Sugito, H.R.; Eltahir, A.; Ma, B.; Delaney, M.K.; Murphy, P.A.; Kauke, M.J.; Angelini, A.; et al. Integrin-targeted cancer immunotherapy elicits protective adaptive immune responses. J. Exp. Med. 2017, 214, 1679–1690. [Google Scholar] [CrossRef]
- Peres, L.D.P.; Da Luz, F.A.C.; Pultz, B.D.A.; Brigido, P.C.; De Araujo, R.A.; Goulart, L.R.; Silva, M. Peptide vaccines in breast cancer: The immunological basis for clinical response. Biotechnol. Adv. 2015, 33, 1868–1877. [Google Scholar] [CrossRef]
- Dettling, S.; Stamova, S.; Warta, R.; Schnölzer, M.; Rapp, C.; Rathinasamy, A.; Reuss, D.E.; Pocha, K.; Roesch, S.; Jungk, C.; et al. Identification of CRKII, CFL1, CNTN1, NME2, and TKT as novel and frequent T-cell targets in human IDH-mutant Gglioma. Clin. Cancer Res. 2018, 24, 2951–2962. [Google Scholar] [CrossRef]
- Mei, W.; Lin, X.; Kapoor, A.; Gu, Y.; Zhao, K.; Tang, D. The contributions of prostate cancer stem cells in prostate cancer initiation and metastasis. Cancers 2019, 11, 434. [Google Scholar] [CrossRef]
- Nie, D.; Boral, D. Cancer stem cells and niche mircoenvironments. Front. Biosci. 2012, 4, 2502–2514. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Pestell, T.G.; Lisanti, M.P.; Pestell, R.G. Cancer stem cells. Int. J. Biochem. Cell Biol. 2012, 44, 2144–2151. [Google Scholar] [CrossRef] [PubMed]
- Barcellos-Hoff, M.H.; Lyden, D.; Wang, T.C. The evolution of the cancer niche during multistage carcinogenesis. Nat. Rev. Cancer 2013, 13, 511–518. [Google Scholar] [CrossRef] [PubMed]
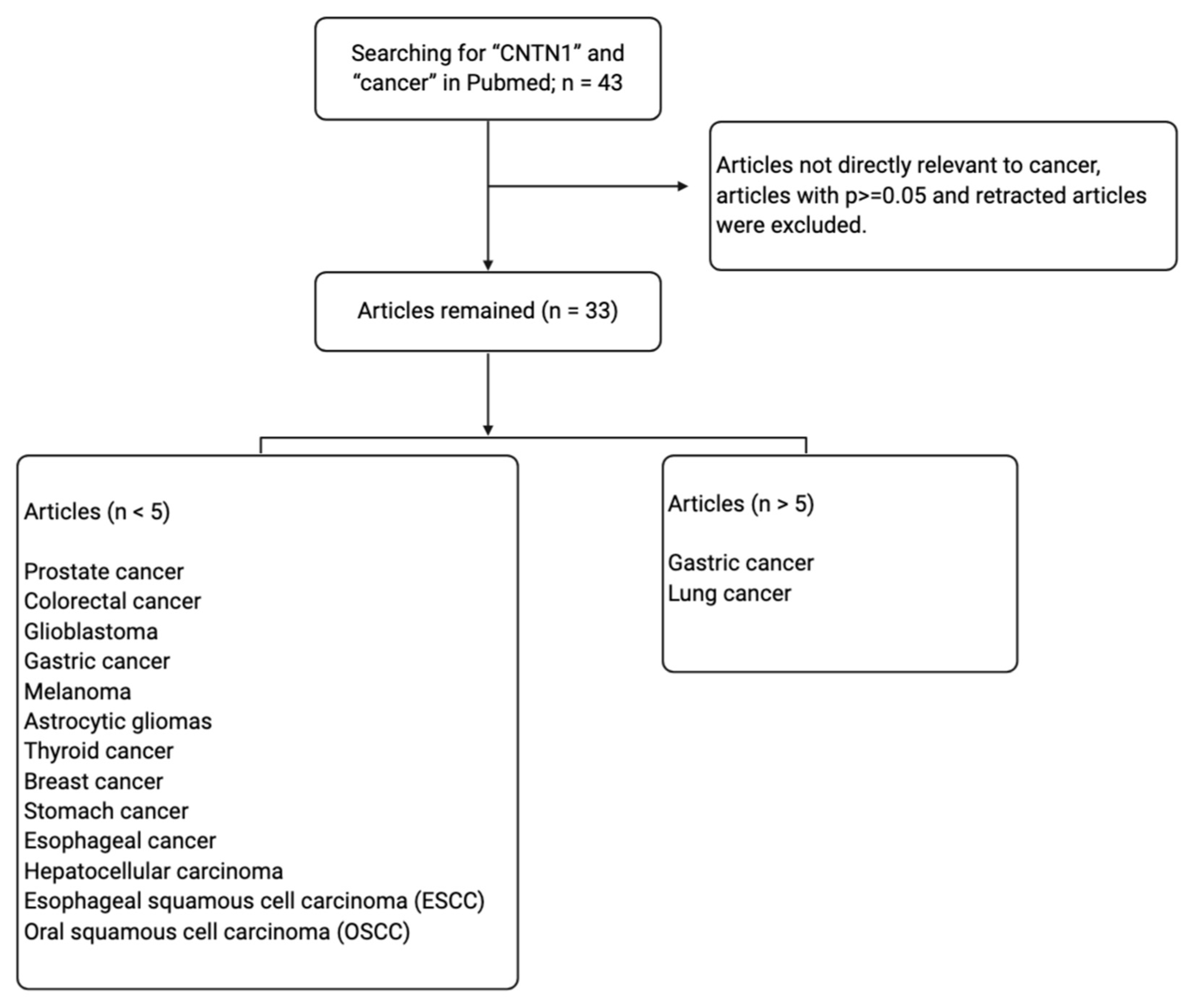
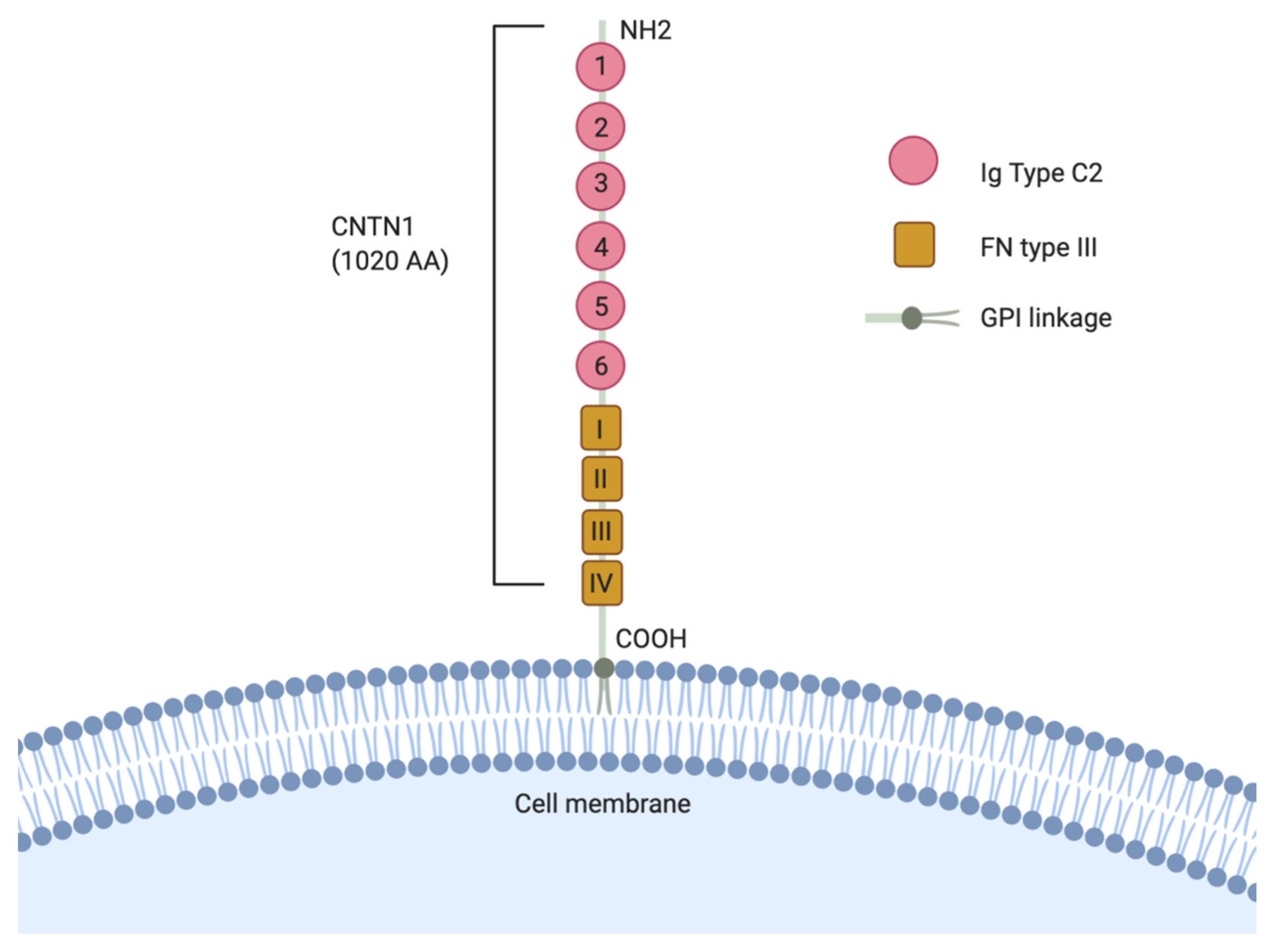
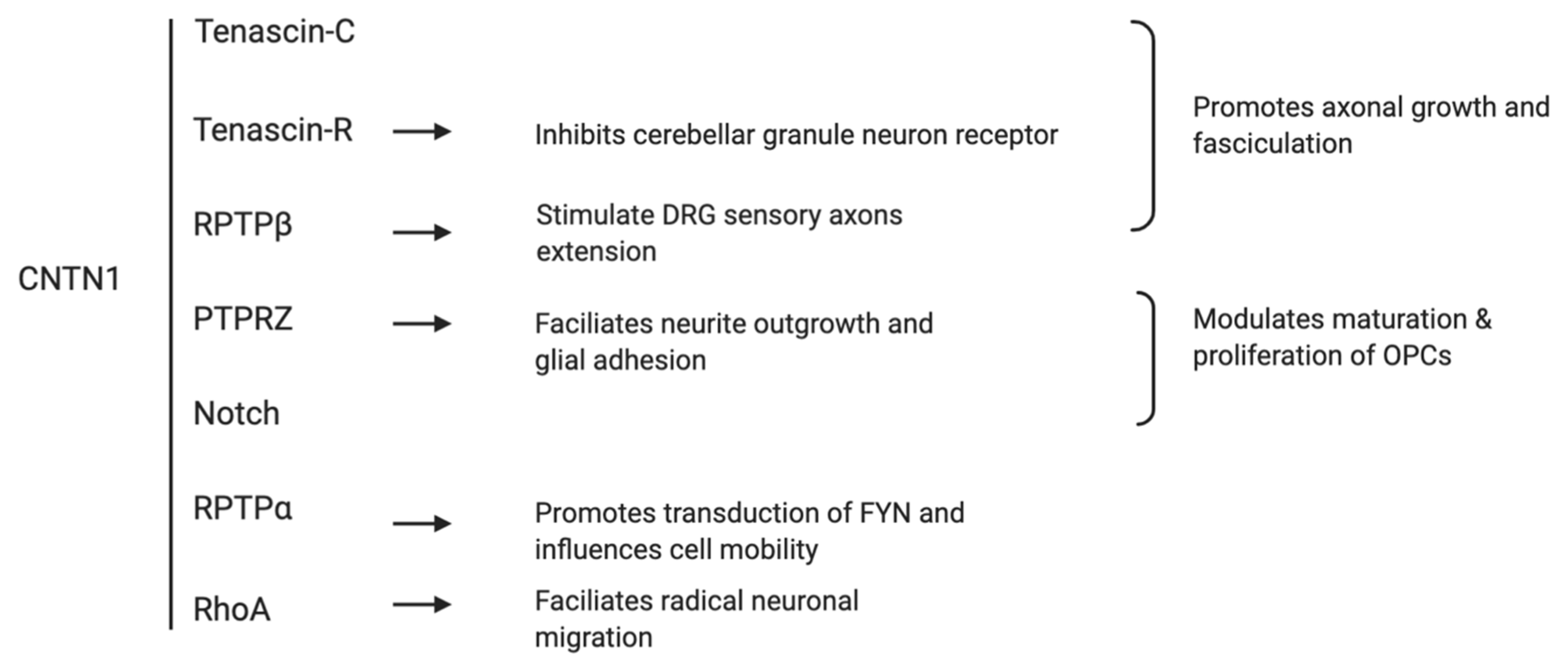
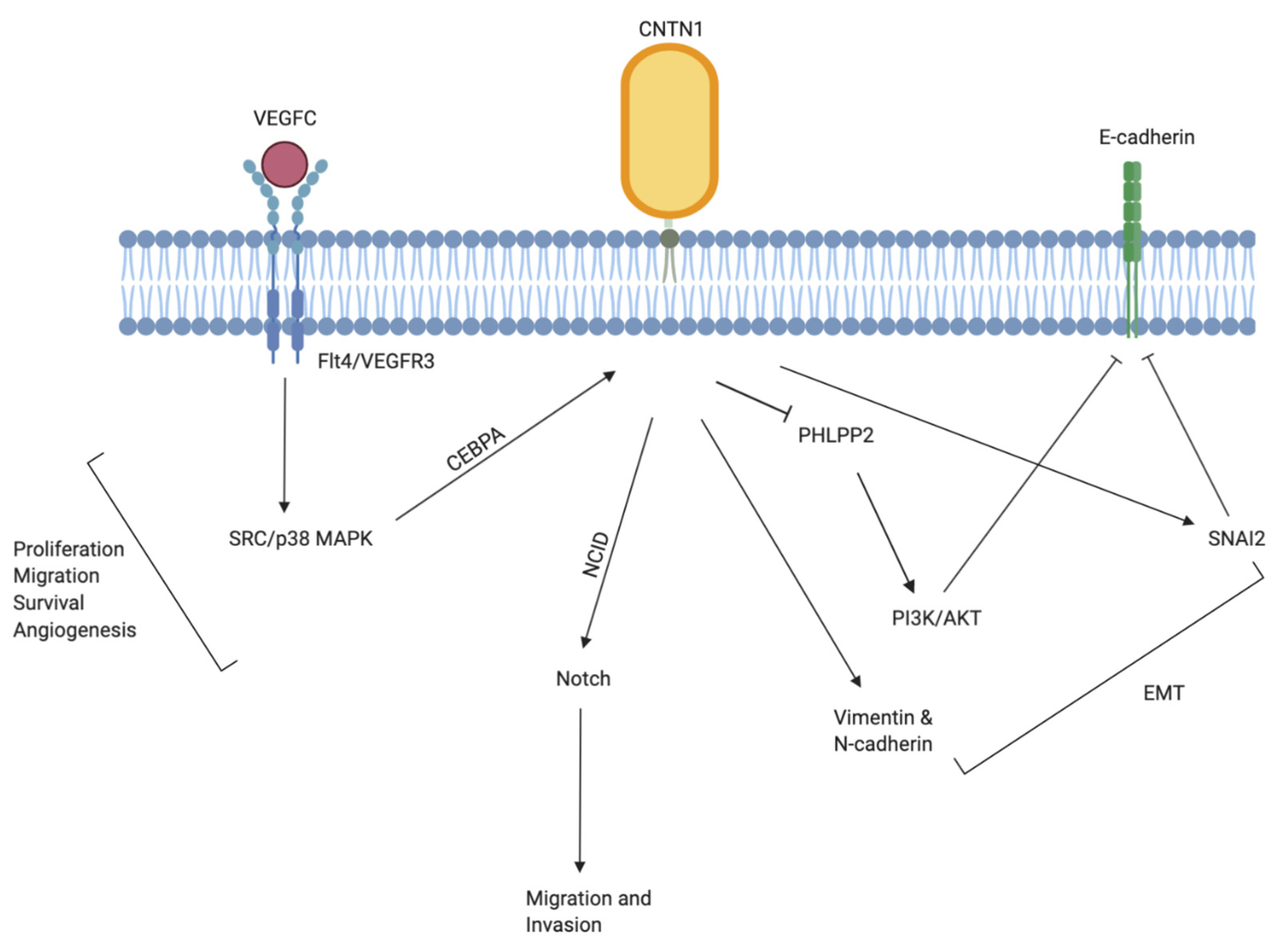

| Tumor Type | CNTN1 Expression in Cancer and Major Findings | References |
|---|---|---|
| Lung cancer |
| [58] |
| [59] | |
| [56] | |
| [57] | |
| [53] | |
| [72] | |
| Gastric adenocarcinoma (GAC) |
| [61] |
| [60] | |
| Oesophageal squamous cell carcinoma (ESCC) |
| [70] |
| [66] | |
| Oral squamous cell carcinoma (OSCC) |
| [54] |
| [71] | |
| Hepatocellular carcinoma (HCC) |
| [68] |
| Astrocytic Gliomas |
| [69] |
| Breast cancer |
| [62] |
| Thyroid cancer |
| [7] |
| Stomach cancer |
| [65] |
| Prostate cancer (PC) |
| [64] |
| [63] | |
| [73] |
| Go ID | Term | p-Value | %Target List | Associated Genes |
|---|---|---|---|---|
| All patients | ||||
| GO:0030198 | extracellular matrix organization | −5 | 14.63 | CCDC80, COL8A2, ECM2, FBN1, ITGA11, LOX |
| GO:0007155 | cell adhesion | −5 | 17.07 | CCN4, CDH11, CNTN1, COL12A1, ITGA11, OMD, SPON1 |
| Basal-like | ||||
| GO:0030198 | extracellular matrix organization | −15 | 14.08 | ADAM12, APBB2, CCDC80, COL3A1, COL6A3, CRISPLD2, DCN, ECM2, FBLN1, FBN1, JAM2, JAM3, LAMA2, MATN3, MMP16, MMP2, NDNF, POSTN, SMOC2, VCAN |
| GO:0001501 | skeletal system development | −8 | 7.75 | CDH11, CHRD, COL3A1, EVC, EBN1, IGF2, MATN3, MMP16, PTH1R, TLL1, VCAN |
| GO:0007155 | cell adhesion | −8 | 12.68 | ADAM12, CDH11, CNTN1, COL6A3, EDIL3, FAP, FEZ1, JCAD, LAMA2, NUAK1, OMD, PCDH7, POSTN, SEMA5A, SPOCK1, SVEP1, THBS2, VCAN |
| Her2-E | ||||
| GO:0030198 | extracellular matrix organization | −19 | 21.74 | ADAM12, COL1A1, COL1A2, COL3A1, COL5A1, COL5A2, COL8A1, COL8A2, CRISPLD2, DCN, DDR2, ECM2, FBN1, JAM3, LOX, LUM, MMP2, RECK, SPARC, VCAN |
| GO:0001501 | skeletal system development | −7 | 8.7 | CDH11, COL1A2, COL1A2, COL3A1, COL5A2, EVC, FBN1, VCAN |
| GO:0007155 | cell adhesion | −7 | 15.22 | ADAM12, CCN4, CDH11, CNTN1, COL5A1, COL8A1, DDR2, EDIL3, FAP, OMD, PCDH7, SPON1, THBS2, VCAN |
| Luminal A | ||||
| GO:0030198 | extracellular matrix organization | −38 | 9.83 | ADAM12, ADAM19, BGN, CCDC80, COL10A1, COL11A1, COL14A1, COL16A1, COL1A1, COL1A2, COL3A1, COL5A1, COL5A2, COL5A3, COL6A1, COL6A2, COL6A3, COL7A1, COL8A1, COL8A2, COMP, CRISPLD2, CDN, DDR2, ECM2, EGFL6, ELN, FBLN1, FBLN2, FBLN5, FBN1, FN1, FOXF2, ITGA11, ITGB1, JAM2, JAM3, LAMA1, LAMA2, LAMA4, LAMB1, LAMC1, LOX, LOXL1, LUM, MATN3, MFAP2, MFAP5, MMP11, MMP13MMP14, MMP16, MMP19, MMP2, MMP27, NID1, NID2, PDGFRA, POSTN, PXDN, RECK, SH3PXD2A, SPARC, VCAN |
| GO:0001501 | skeletal system development | −14 | 11.67 | ALX4, ARSE, BMP1, BMP7, CDH11, COL10A1, COL1A1, COL1A2, COL3A1, COL6A2, COMP, EVC, FBN1, FRZB, GJA5, GLI2, HOXA11, IGF1, IGF2, MATN3, MMP14, MMP16, NKX3–2, PTH1R, RASSF2, SH3PXD2B, SHOX2, TLL1, VCAN |
| GO:0007155 | cell adhesion | −29 | 3.53 | ADAM12, ADAM23, BOC, CCN2, CCN4, CDH11, CERCAM, CNTN1, COL12A1, COL16A1, COL5A1, COL5A3, COL6A1, COL6A2, COL6A3, COL6A6, COL7A1, COL8A1, CXCL12, CYP1B1, DDR2, DPP4, DPT, EGFL6, EMILIN1, ENTPD1, FAP, FAT1, FEZ1, FN1, GAS6, GPNMB, ISLRITGA11, ITGB1, ITGBL1, JCAD, LAMA1, LAMA2, LAMA4, LAMB1, LAMC1, LOXL2, LSAMP, MFAP4, MFGE8, MXRA8, NID2, NRP2, NTM, NUAK1, OMD, PCDH18, PCDH7, PCDHB12, PCDHB18P, PCDHGA12, PCDHGC3, PLXNC1, POSTN, PPFIBP1, PRKCA, PTK7, ROBO1, SEMA5A, SPOCK1, SPON1, SPON2, SRPX, SSPN, TGFB1I1, THBS2, TLN2, TNFAIP6, VCAN, VCL |
| Luminal B | ||||
| GO:0030198 | extracellular matrix organization | −38 | 11.07 | ADAM12, BGN, CCDC80, COL10A1, COL11A1, COL14A1, COL16A1, COL1A1, COL1A2, COL3A1, COL5A1, COL5A2, COL5A3, COL6A1, COL6A2, COL6A3, COL7A1, COL8A1, COL8A2, COMP, CRISPLD2, DCN, DDR2, ECM2, ELN, FBLN1, FBLN2, FBLN5, FBN1, FN1, FOXF2, HSPG2, ITGA11, JAM3, LAMA1, LAMA2, LAMA4, LAMB1, LAMC1, LOX, LOXL1, LUM, MFAP2, MFAP2, MFAP5, MMP11, MMP13, MMP14, MMP19, MMP2, NDNF, NID1, NID2, PDGFRA, POSTN, PXDN, RECK, SH3PXD2A, SPARC, VCAN |
| GO:0001501 | skeletal system development | −17 | 5.44 | ARSE, BMP1, CDH11, COL10A1, COL1A1, COL1A2, COL3A1, COL5A2, COMP, DLX5, EN1, EVC, FBN1, FRZB, GJA5, GLI2, HOXA10, HOXA11, HOXA4, IGF2, MMP14, NKX3–2, PAX1, PRELP, PTH1R, SH3PXD2B, SHOX2, TLL1, VCAN |
| GO:0007155 | cell adhesion | −25 | 12.01 | ADAM12, CCN4, CDH11, CNTN1, COL5A1, COL8A1, DDR2, EDIL3, FAP, OMD, PCDH7, SPON1, THBS2, VCAN |
| Normal-like | ||||
| GO:0030198 | extracellular matrix organization | −5 | 11.48 | COL14A1, COL8A2, DDR2, JAM3, LAMA2, MMP16, RECK |
| GO:0007155 | cell adhesion | −4 | 11.48 | CNTN1, DDR2, LAMA2, PCDH7, SEMA5A, SPON1, SSPN |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, Y.; Li, T.; Kapoor, A.; Major, P.; Tang, D. Contactin 1: An Important and Emerging Oncogenic Protein Promoting Cancer Progression and Metastasis. Genes 2020, 11, 874. https://doi.org/10.3390/genes11080874
Gu Y, Li T, Kapoor A, Major P, Tang D. Contactin 1: An Important and Emerging Oncogenic Protein Promoting Cancer Progression and Metastasis. Genes. 2020; 11(8):874. https://doi.org/10.3390/genes11080874
Chicago/Turabian StyleGu, Yan, Taosha Li, Anil Kapoor, Pierre Major, and Damu Tang. 2020. "Contactin 1: An Important and Emerging Oncogenic Protein Promoting Cancer Progression and Metastasis" Genes 11, no. 8: 874. https://doi.org/10.3390/genes11080874
APA StyleGu, Y., Li, T., Kapoor, A., Major, P., & Tang, D. (2020). Contactin 1: An Important and Emerging Oncogenic Protein Promoting Cancer Progression and Metastasis. Genes, 11(8), 874. https://doi.org/10.3390/genes11080874





