Comprehensive Transcriptomic Analysis Identifies ST8SIA1 as a Survival-Related Sialyltransferase Gene in Breast Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. RNA Sequencing Analysis
2.3. Statistical Analysis
2.4. Gene Set Enrichment Analysis (GSEA) and Gene Ontology (GO)
3. Results
3.1. Characteristics of the Study Subjects from TCGA-BRCA
3.2. The Difference in RNA-Seq Expression of Sialyltransferase Genes
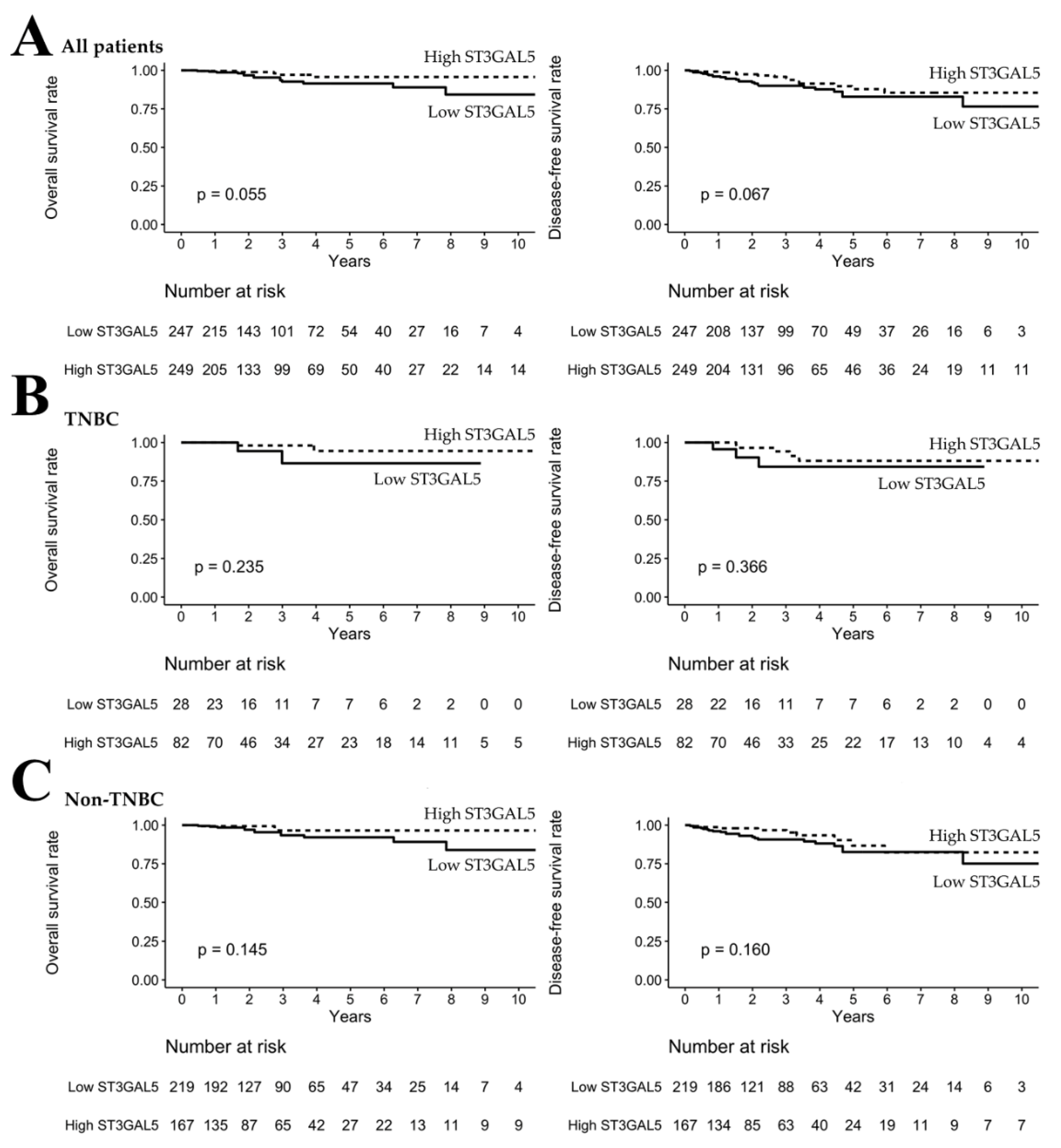
3.3. Distinction of OS by Sialyltransferase Gene in Breast Cancer
3.4. High ST8SIA1 Expression Was Correlated with Poor OS/DFS in TNBC Patients
3.5. Cox Proportional Hazard Regression Model of ST8SIA1
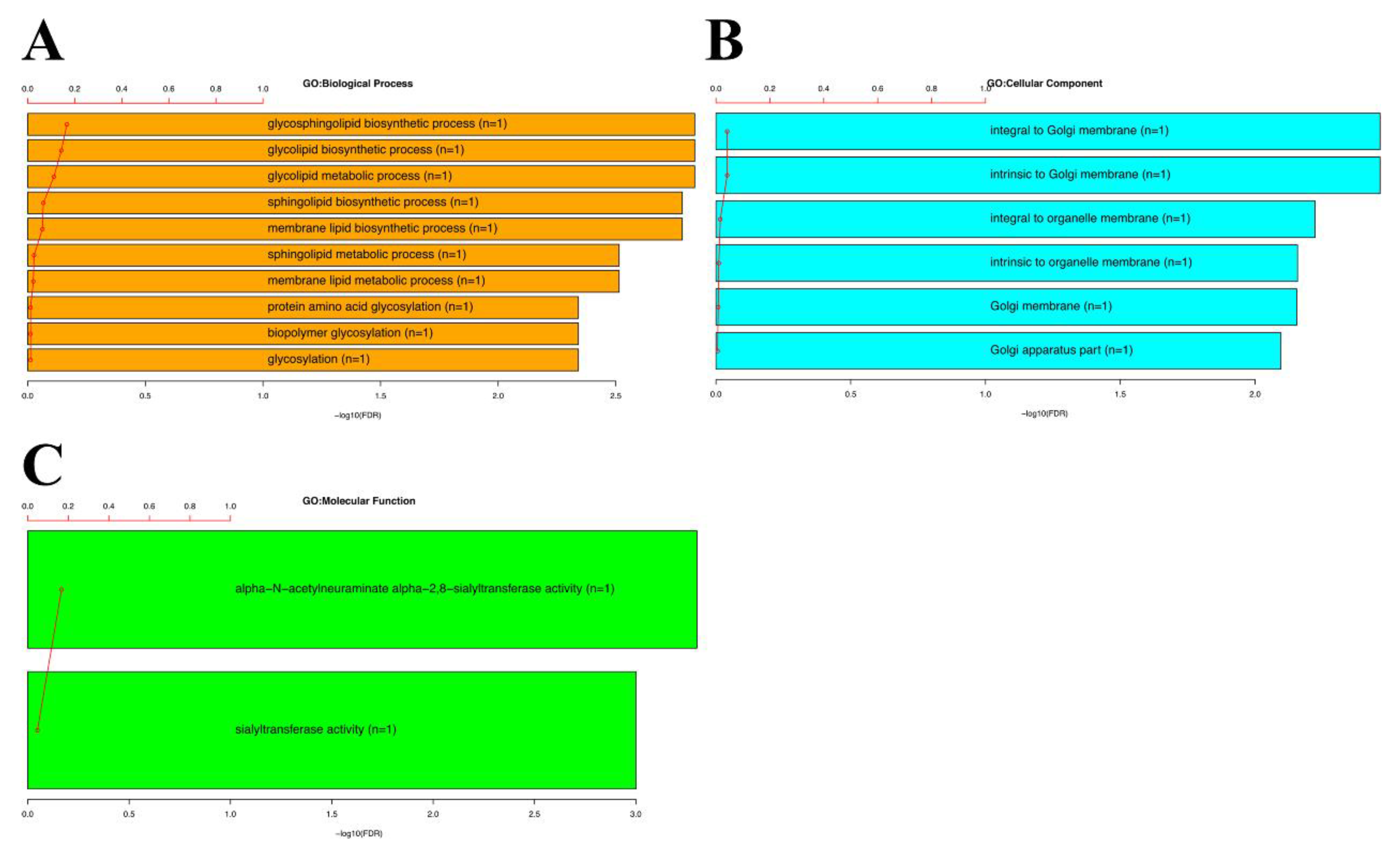
3.6. Gene Ontology Analysis (GO) of Candidate ST8SIA1 Gene
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fuster, M.M.; Esko, J.D. The sweet and sour of cancer: Glycans as novel therapeutic targets. Nat. Rev. Cancer 2005, 5, 526–542. [Google Scholar] [CrossRef] [PubMed]
- Schauer, R. Achievements and challenges of sialic acid research. Glycoconj. J. 2000, 17, 485–499. [Google Scholar] [CrossRef] [PubMed]
- Varki, A. Glycan-based interactions involving vertebrate sialic-acid-recognizing proteins. Nat. Cell Biol. 2007, 446, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Ashwell, G.; Morell, A.G. The Role of Surface Carbohydrates in the Hepatic Recognition and Transport of Circulating Glycoproteins. Adv. Enzymol. Relat. Areas Mol. Biol. 2006, 41, 99–128. [Google Scholar] [CrossRef]
- Bratosin, D.; Mazurier, J.; Tissier, J.; Estaquier, J.; Huart, J.; Ameisen, J.; Aminoff, D.; Montreuil, J. Cellular and molecular mechanisms of senescent erythrocyte phagocytosis by macrophages: A review. Biochimie 1998, 80, 173–195. [Google Scholar] [CrossRef]
- Garnham, R.; Scott, E.; Livermore, K.E.; Munkley, J. ST6GAL1: A key player in cancer (Review). Oncol. Lett. 2019, 18, 983–989. [Google Scholar] [CrossRef]
- Seales, E.C.; Shaikh, F.M.; Woodard-Grice, A.V.; Aggarwal, P.; McBrayer, A.C.; Hennessy, K.M.; Bellis, S.L. A Protein Kinase C/Ras/ERK Signaling Pathway Activates Myeloid Fibronectin Receptors by Altering β1 Integrin Sialylation. J. Biol. Chem. 2005, 280, 37610–37615. [Google Scholar] [CrossRef]
- Liu, Q.; Ma, H.; Sun, X.; Liu, B.; Xiao, Y.; Pan, S.; Zhou, H.; Dong, W.; Jia, L. The regulatory ZFAS1/miR-150/ST6GAL1 crosstalk modulates sialylation of EGFR via PI3K/Akt pathway in T-cell acute lymphoblastic leukemia. J. Exp. Clin. Cancer Res. 2019, 38, 199. [Google Scholar] [CrossRef]
- Chang, T.-C.; Chin, Y.-T.; Nana, A.W.; Wang, S.-H.; Liao, Y.-M.; Chen, Y.-R.; Shih, Y.-J.; Changou, C.A.; Yang, Y.-C.S.; Wang, K.; et al. Enhancement by Nano-Diamino-Tetrac of Antiproliferative Action of Gefitinib on Colorectal Cancer Cells: Mediation by EGFR Sialylation and PI3K Activation. Horm. Cancer 2018, 9, 420–432. [Google Scholar] [CrossRef]
- Swindall, A.F.; Bellis, S.L. Sialylation of the Fas Death Receptor by ST6Gal-I Provides Protection against Fas-mediated Apoptosis in Colon Carcinoma Cells. J. Biol. Chem. 2011, 286, 22982–22990. [Google Scholar] [CrossRef]
- Chiang, C.-H.; Wang, C.-H.; Chang, H.-C.; More, S.V.; Li, W.-S.; Hung, W.-C. A novel sialyltransferase inhibitor AL10 suppresses invasion and metastasis of lung cancer cells by inhibiting integrin-mediated signaling. J. Cell. Physiol. 2010, 223, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Ding, J. Sialylation is involved in cell fate decision during development, reprogramming and cancer progression. Protein Cell 2019, 10, 550–565. [Google Scholar] [CrossRef] [PubMed]
- Chiodelli, P.; Urbinati, C.; Paiardi, G.; Monti, E.; Rusnati, M. Sialic acid as a target for the development of novel antiangiogenic strategies. Futur. Med. Chem. 2018, 10, 2835–2854. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yang, G.; Guan, F. Biological Functions and Analytical Strategies of Sialic Acids in Tumor. Cells 2020, 9, 273. [Google Scholar] [CrossRef]
- Ip, C.; Patel, J.; Dao, T.L. Serum Sialyltransferase and 5′-Nucleotidase as Reliable Biomarkers in Women with Breast Cancer. J. Natl. Cancer Inst. 1980, 65, 529–534. [Google Scholar] [CrossRef]
- Su, M.-L.; Chang, T.-M.; Chiang, C.-P.; Chang, H.-C.; Hou, M.-F.; Li, W.-S.; Hung, W.-C. Inhibition of Chemokine (C-C Motif) Receptor 7 Sialylation Suppresses CCL19-Stimulated Proliferation, Invasion and Anti-Anoikis. PLoS ONE 2014, 9, e98823. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, R.; Zhong, G.; Chen, X.; Cheng, Y.; Li, W.; Yang, Y. ST6GAL2 Downregulation Inhibits Cell Adhesion and Invasion and is Associated with Improved Patient Survival in Breast Cancer. OncoTargets Ther. 2020, 13, 903–914. [Google Scholar] [CrossRef]
- Nguyen, K.; Yan, Y.; Yuan, B.; Dasgupta, A.; Sun, J.C.; Mu, H.; Do, K.-A.; Ueno, N.T.; Andreeff, M.; Battula, V.L. ST8SIA1 Regulates Tumor Growth and Metastasis in TNBC by Activating the FAK–AKT–mTOR Signaling Pathway. Mol. Cancer Ther. 2018, 17, 2689–2701. [Google Scholar] [CrossRef] [PubMed]
- Julien, S.; Videira, P.A.; Delannoy, P. Sialyl-Tn in Cancer: (How) Did We Miss the Target? Biomology 2012, 2, 435–466. [Google Scholar] [CrossRef]
- Murugaesu, N.; Iravani, M.; Van Weverwijk, A.; Ivetic, A.; Johnson, D.A.; Antonopoulos, A.; Fearns, A.; Jamal-Hanjani, M.; Sims, D.; Fenwick, K.; et al. An In Vivo Functional Screen Identifies ST6GalNAc2 Sialyltransferase as a Breast Cancer Metastasis Suppressor. Cancer Discov. 2014, 4, 304–317. [Google Scholar] [CrossRef]
- Saeui, C.T.; Nairn, A.V.; Galizzi, M.; Douville, C.; Gowda, P.; Park, M.; Dharmarha, V.; Shah, S.R.; Clarke, A.; Austin, M.; et al. Integration of genetic and metabolic features related to sialic acid metabolism distinguishes human breast cell subtypes. PLoS ONE 2018, 13, e0195812. [Google Scholar] [CrossRef]
- Saeui, C.T.; Cho, K.-C.; Dharmarha, V.; Nairn, A.V.; Galizzi, M.; Shah, S.R.; Gowda, P.; Park, M.; Austin, M.; Clarke, A.; et al. Cell Line-, Protein-, and Sialoglycosite-Specific Control of Flux-Based Sialylation in Human Breast Cells: Implications for Cancer Progression. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Drake, P.M.; Schilling, B.; Niles, R.K.; Prakobphol, A.; Li, B.; Jung, K.; Cho, W.; Braten, M.; Inerowicz, H.D.; Williams, K.; et al. Lectin Chromatography/Mass Spectrometry Discovery Workflow Identifies Putative Biomarkers of Aggressive Breast Cancers. J. Proteome Res. 2012, 11, 2508–2520. [Google Scholar] [CrossRef]
- Da Huang, W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Todeschini, A.R.; Hakomori, S.-I. Functional role of glycosphingolipids and gangliosides in control of cell adhesion, motility, and growth, through glycosynaptic microdomains. Biochim. Biophys. Acta 2008, 1780, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Grouxdegroote, S.; Rodríguez-Walker, M.; Dewald, J.H.; Daniotti, J.L.; Delannoy, P. Gangliosides in Cancer Cell Signaling. Prog. Mol. Biol. Transl. Sci. 2018, 156, 197–227. [Google Scholar] [CrossRef]
- Ruckhäberle, E.; Rody, A.; Engels, K.; Gaetje, R.; Von Minckwitz, G.; Schiffmann, S.; Grösch, S.; Geisslinger, G.; Holtrich, U.; Karn, T.; et al. Microarray analysis of altered sphingolipid metabolism reveals prognostic significance of sphingosine kinase 1 in breast cancer. Breast Cancer Res. Treat. 2008, 112, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Ruckhaeberle, E.; Karn, T.; Rody, A.; Hanker, L.; Gätje, R.; Metzler, D.; Holtrich, U.; Kaufmann, M. Gene expression of ceramide kinase, galactosyl ceramide synthase and ganglioside GD3 synthase is associated with prognosis in breast cancer. J. Cancer Res. Clin. Oncol. 2009, 135, 1005–1013. [Google Scholar] [CrossRef] [PubMed]
- Cazet, A.; Lefebvre, J.; Adriaenssens, E.; Julien, S.; Bobowski, M.; Grigoriadis, A.; Tutt, A.; Tulasne, D.; Le Bourhis, X.; Delannoy, P. GD3 Synthase Expression Enhances Proliferation and Tumor Growth of MDA-MB-231 Breast Cancer Cells through c-Met Activation. Mol. Cancer Res. 2010, 8, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Li, Z.; Wang, H.; Cai, F.; Wang, L. ST8SIA1 inhibition sensitizes triple negative breast cancer to chemotherapy via suppressing Wnt/beta-catenin and FAK/Akt/mTOR. Clin. Transl. Oncol. 2020. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Y.; Wu, L.; Sun, X.-L. Sialyltransferase inhibition and recent advances. Biochim. Biophys. Acta 2016, 1864, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Szabo, R.; Skropeta, D. Advancement of Sialyltransferase Inhibitors: Therapeutic Challenges and Opportunities. Med. Res. Rev. 2017, 37, 219–270. [Google Scholar] [CrossRef] [PubMed]
- Shamon, L.A.; Pezzuto, J.M.; Graves, J.M.; Mehta, R.R.; Wangcharoentrakul, S.; Sangsuwan, R.; Chaichana, S.; Tuchinda, P.; Cleason, P.; Reutrakul, V. Evaluation of the mutagenic, cytotoxic, and antitumor potential of triptolide, a highly oxygenated diterpene isolated from Tripterygium wilfordii. Cancer Lett. 1997, 112, 113–117. [Google Scholar] [CrossRef]
- Phillips, P.A.; Dudeja, V.; McCarroll, J.A.; Borja-Cacho, D.; Dawra, R.K.; Grizzle, W.E.; Vickers, S.M.; Saluja, A.K. Triptolide Induces Pancreatic Cancer Cell Death via Inhibition of Heat Shock Protein. Cancer Res. 2007, 67, 9407–9416. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.-Y.; Kim, S.-J.; Kim, C.-H.; Son, S.-W.; Kim, K.-S.; Lee, J.-H.; Do, S.-I.; Lee, Y.-C. Triptolide downregulates human GD3 synthase (hST8Sia I) gene expression in SK-MEL-2 human melanoma cells. Exp. Mol. Med. 2010, 42, 849–855. [Google Scholar] [CrossRef]


| Variables | Total, n = 496 | TNBC, n = 110 | Non-TNBC, n = 386 | p |
|---|---|---|---|---|
| Age (years) | 56.7 ± 13.0 | 55.8 ± 12.4 | 56.9 ± 13.1 | 0.390 |
| Ethnicity | 0.546 | |||
| Hispanic or Latino | 13 (2.6%) | 3 (2.7%) | 10 (2.6%) | |
| Not Hispanic or Latino | 383 (77.2%) | 89 (80.9%) | 294 (76.2%) | |
| Not reported | 100 (20.2%) | 18 (16.4%) | 82 (21.2%) | |
| Pathological stage | 0.013 | |||
| Stage I | 95 (19.2%) | 18 (16.4%) | 77 (19.9%) | |
| Stage II | 310 (62.5%) | 81 (73.6%) | 229 (59.3%) | |
| Stage III | 91 (18.3%) | 11 (10.0%) | 80 (20.7%) | |
| Lymph node invasion | 234 (47.2%) | 40 (36.4%) | 194 (50.3%) | <0.001 |
| Treatment | ||||
| Radiation | 274 (55.2%) | 64 (58.2%) | 210 (54.4%) | 0.552 |
| Pharmaceutical | 410 (82.7%) | 90 (81.8%) | 320 (82.9%) | 0.903 |
| All-cause mortality (OS) | 21 (4.2%) | 4 (3.6%) | 17 (4.4%) | 0.933 |
| Disease-progressed (PFI) | 41 (8.3%) | 8 (7.3%) | 33 (8.5%) | 0.816 |
| Genes | Total | TNBC | Non-TNBC | p |
|---|---|---|---|---|
| ST3GAL2 | 2.79 ± 0.58 | 2.84 ± 0.66 | 2.78 ± 0.56 | 0.406 |
| ST3GAL3 | 1.10 ± 0.30 | 1.12 ± 0.33 | 1.10 ± 0.29 | 0.638 |
| ST3GAL5 | 3.67 ± 0.33 | 3.86 ± 0.35 | 3.62 ± 0.30 | <0.001 |
| ST6GAL1 | 0.02 ± 0.10 | 0.02 ± 0.06 | 0.03 ± 0.11 | 0.402 |
| ST6GALNAC3 | 1.93 ± 0.20 | 1.95 ± 0.26 | 1.92 ± 0.18 | 0.177 |
| ST6GALNAC4 | 4.05 ± 0.44 | 3.66 ± 0.47 | 4.16 ± 0.36 | <0.001 |
| ST8SIA1 | 0.98 ± 0.86 | 0.75 ± 0.76 | 1.04 ± 0.87 | <0.001 |
| ST8SIA3 | 0.16 ± 0.37 | 0.11 ± 0.27 | 0.17 ± 0.39 | 0.053 |
| Target Genes | OS | |||
| Optimal Cutoff Point | AUC | Sensitivity | Specificity | |
| ST3GAL5 | ≤3.63 | 0.62 | 0.51 | 0.71 |
| ST6GALNAC4 | ≥4.01 | 0.54 | 0.71 | 0.44 |
| ST8SIA1 | ≥1.31 | 0.68 | 0.71 | 0.73 |
| DFS | ||||
| Optimal Cutoff Point | AUC | Sensitivity | Specificity | |
| ST3GAL5 | ≤3.63 | 0.56 | 0.52 | 0.63 |
| ST6GALNAC4 | ≥4.01 | 0.54 | 0.71 | 0.44 |
| ST8SIA1 | ≥1.31 | 0.56 | 0.41 | 0.72 |
| Target Genes | OS | |||
| Optimal Cutoff Point | AUC | Sensitivity | Specificity | |
| TNBC ST3GAL5 | ≤3.63 | 0.64 | 0.75 | 0.50 |
| ST6GALNAC4 | ≥4.01 | 0.67 | 0.22 | 0.75 |
| ST8SIA1 | ≥1.31 | 0.78 | 0.75 | 0.85 |
| Non-TNBC ST3GAL5 | ≤3.63 | 0.61 | 0.44 | 0.76 |
| ST6GALNAC4 | ≥4.01 | 0.55 | 0.82 | 0.34 |
| ST8SIA1 | ≥1.31 | 0.68 | 0.71 | 0.69 |
| DFS | ||||
| Optimal Cutoff Point | AUC | Sensitivity | Specificity | |
| TNBC ST3GAL5 | ≤3.63 | 0.52 | 0.63 | 0.25 |
| ST6GALNAC4 | ≥4.01 | 0.50 | 0.22 | 0.75 |
| ST8SIA1 | ≥1.31 | 0.74 | 0.50 | 0.85 |
| Non-TNBC ST3GAL5 | ≤3.63 | 0.58 | 0.45 | 0.70 |
| ST6GALNAC4 | ≥4.01 | 0.54 | 0.82 | 0.34 |
| ST8SIA1 | ≥1.31 | 0.48 | 0.32 | 0.61 |
| Variables | Comparison | Univariate | Multivariate | ||
|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | ||
| ST8SIA1 | High vs. low | 8.57 (2.84, 25.9) | <0.001 | 9.95 (3.25, 30.5) | <0.001 |
| Diagnosis age | ≥50 vs. <50 | 2.88 (0.94, 8.89) | 0.065 | 3.49 (1.09, 11.1) | 0.035 |
| Subtypes | TNBC vs. non-TNBC | 0.77 (0.26, 2.29) | 0.6 | 1.13 (0.36, 3.58) | 0.8 |
| Pathological stage | Stage II vs I | 5.93 (0.78, 45.0) | 0.085 | 14.4 (1.55, 134) | 0.019 |
| Stage III vs I | 5.46 (0.61, 49.3) | 0.13 | 20.4 (1.47, 283) | 0.025 | |
| Lymph node invasion | Positive vs. negative | 1.45 (0.61, 3.48) | 0.4 | 0.84 (0.30, 2.36) | 0.7 |
| Radiation | Yes vs. no | 0.35 (0.13, 0.99) | 0.047 | 0.22 (0.07, 0.69) | 0.009 |
| Pharmaceutical | Yes vs. no | 0.64 (0.27, 1.51) | 0.3 | 0.74 (0.30, 1.81) | 0.5 |
| Cox proportional hazard regression of overall survival (ST8SIA1) in TNBC patients (n = 110). | |||||
| ST8SIA1 | High vs. low | 11.9 (1.23, 114.00) | 0.032 | 9.45 (0.78, 115.00) | 0.078 |
| Diagnosis age | ≥50 vs. <50 | 0.75 (0.10, 5.30) | 0.77 | 0.52 (0.07, 4.05) | 0.5 |
| Pathological stage | Stage III vs I | Omitted † | - | Omitted † | - |
| Stage II vs I | Omitted † | - | Omitted † | - | |
| Lymph node invasion | Positive vs. negative | 5.66 (0.59, 54.50) | 0.134 | 2.02 (0.13, 32.50) | 0.6 |
| Radiation | Yes vs. no | 1.28 (0.13, 12.30) | 0.831 | 1.2 (0.09, 16.00) | 0.9 |
| Pharmaceutical | Yes vs. no | Omitted † | - | Omitted † | - |
| † Omitted as imbalanced of sample distribution. | |||||
| Cox proportional hazard regression of overall survival (ST8SIA1) in non-TNBC patients (n = 386). | |||||
| ST8SIA1 | High vs. low | 8.01 (2.26, 28.40) | 0.001 | 11.30 (3.04, 41.90) | <0.001 |
| Diagnosis age | ≥50 vs.<50 | 4.96 (1.09, 22.60) | 0.038 | 6.69 (1.42, 31.50) | 0.016 |
| Pathological stage | Stage III vs I | 4.35 (0.56, 33.80) | 0.16 | 15.50 (1.56, 154.00) | 0.019 |
| Stage II vs I | 4.12 (0.45, 37.40) | 0.209 | 31.20 (1.99, 491.00) | 0.014 | |
| Lymph node invasion | Positive vs. negative | 0.93 (0.35, 2.53) | 0.894 | 0.55 (0.16, 1.93) | 0.4 |
| Radiation | Yes vs. no | 0.28 (0.09, 0.81) | 0.019 | 0.16 (0.05, 0.56) | 0.004 |
| Pharmaceutical | Yes vs. no | 0.52 (0.20, 1.35) | 0.176 | 0.55 (0.19, 1.57) | 0.3 |
| Variables | Comparison | Univariate | Multivariate | ||
|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | ||
| ST8SIA1 | High vs. low | 1.82 (0.97, 3.42) | 0.063 | 1.82 (0.96, 3.45) | 0.068 |
| Diagnosis age | ≥50 vs. <50 | 1.25 (0.65, 2.39) | 0.5 | 1.37 (0.71, 2.66) | 0.3 |
| Subtypes | TNBC vs. non-TNBC | 0.75 (0.34, 1.62) | 0.5 | 0.85 (0.38, 1.90) | 0.7 |
| Pathological stage | Stage II vs I | 2.12 (0.81, 5.54) | 0.13 | 2.57 (0.89, 7.39) | 0.081 |
| Stage III vs I | 2.64 (0.88, 7.92) | 0.083 | 3.32 (0.86, 12.9) | 0.083 | |
| Lymph node invasion | Positive vs. negative | 1.29 (0.70, 2.40) | 0.4 | 0.85 (0.40, 1.82) | 0.7 |
| Radiation | Yes vs. no | 0.96 (0.38, 2.47) | 0.939 | 0.81 (0.31, 2.14) | 0.7 |
| Pharmaceutical | Yes vs. no | 1.03 (0.54, 1.96) | 0.921 | 1.05 (0.54, 2.04) | 0.9 |
| Cox proportional hazard regression of disease-free survival (ST8SIA1) in TNBC patients (n = 110). | |||||
| ST8SIA1 | High vs. low | 4.30 (1.07, 17.30) | 0.04 | 5.13 (1.03, 25.50) | 0.046 |
| Diagnosis age | ≥50 vs. <50 | 1.20 (0.29, 5.03) | 0.804 | 0.98 (0.23, 4.21) | >0.9 |
| Pathological stage | Stage III vs I | 2.06 (0.25, 17.20) | 0.506 | 1.51 (0.13, 17.70) | 0.7 |
| Stage II vs I | 3.70 (0.23, 60.00) | 0.357 | 3.30 (0.10, 111.00) | 0.5 | |
| Lymph node invasion | Positive vs. negative | 3.07 (0.73, 12.90) | 0.125 | 1.44 (0.22, 9.27) | 0.7 |
| Radiation | Yes vs. no | 3.29 (0.40, 26.80) | 0.266 | 3.31 (0.37, 29.90) | 0.3 |
| Pharmaceutical | Yes vs. no | Omitted † | - | Omitted † | |
| † Omitted as imbalanced of sample distribution. | |||||
| Cox proportional hazard regression of disease-free survival (ST8SIA1) in non-TNBC patients (n = 386). | |||||
| ST8SIA1 | High vs. low | 1.45 (0.71, 2.94) | 0.308 | 1.45 (0.71, 2.98) | 0.3 |
| Diagnosis age | ≥50 vs. <50 | 1.19 (0.57, 2.47) | 0.64 | 1.29 (0.61, 2.72) | 0.5 |
| Pathological stage | Stage III vs I | 2.10 (0.71, 6.17) | 0.179 | 3.05 (0.95, 9.77) | 0.06 |
| Stage II vs I | 2.43 (0.73, 8.11) | 0.15 | 4.76 (1.05, 21.40) | 0.042 | |
| Lymph node invasion | Positive vs. negative | 0.95 (0.47, 1.90) | 0.878 | 0.59 (0.24, 1.42) | 0.2 |
| Radiation | Yes vs. no | 0.81 (0.31, 2.12) | 0.674 | 0.67 (0.24, 1.81) | 0.4 |
| Pharmaceutical | Yes vs. no | 0.88 (0.44, 1.76) | 0.712 | 0.91 (0.44, 1.87) | 0.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kan, J.-Y.; Moi, S.-H.; Hung, W.-C.; Hou, M.-F.; Chen, F.-M.; Shih, S.-L.; Shiau, J.-P.; Li, C.-L.; Chiang, C.-P. Comprehensive Transcriptomic Analysis Identifies ST8SIA1 as a Survival-Related Sialyltransferase Gene in Breast Cancer. Genes 2020, 11, 1436. https://doi.org/10.3390/genes11121436
Kan J-Y, Moi S-H, Hung W-C, Hou M-F, Chen F-M, Shih S-L, Shiau J-P, Li C-L, Chiang C-P. Comprehensive Transcriptomic Analysis Identifies ST8SIA1 as a Survival-Related Sialyltransferase Gene in Breast Cancer. Genes. 2020; 11(12):1436. https://doi.org/10.3390/genes11121436
Chicago/Turabian StyleKan, Jung-Yu, Sin-Hua Moi, Wen-Chun Hung, Ming-Feng Hou, Fang-Ming Chen, Shen-Liang Shih, Jun-Ping Shiau, Chung-Liang Li, and Chih-Po Chiang. 2020. "Comprehensive Transcriptomic Analysis Identifies ST8SIA1 as a Survival-Related Sialyltransferase Gene in Breast Cancer" Genes 11, no. 12: 1436. https://doi.org/10.3390/genes11121436
APA StyleKan, J.-Y., Moi, S.-H., Hung, W.-C., Hou, M.-F., Chen, F.-M., Shih, S.-L., Shiau, J.-P., Li, C.-L., & Chiang, C.-P. (2020). Comprehensive Transcriptomic Analysis Identifies ST8SIA1 as a Survival-Related Sialyltransferase Gene in Breast Cancer. Genes, 11(12), 1436. https://doi.org/10.3390/genes11121436





