Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
2.2. Parasites and Sera
2.3. Cloning and Expression of Di-CPI and Di-TIM
2.4. Molecular Characterisation of Di-CPI
2.5. Preparation of Polyclonal Antibody Against rDi-CPI
2.6. Western Blotting and Immunochemical Analysis
2.7. Cell Isolation and Culturing
2.8. Proliferation Assay
2.9. Cytokine Analysis
2.10. Statistical Analysis
2.11. Ethics Statement
3. Results
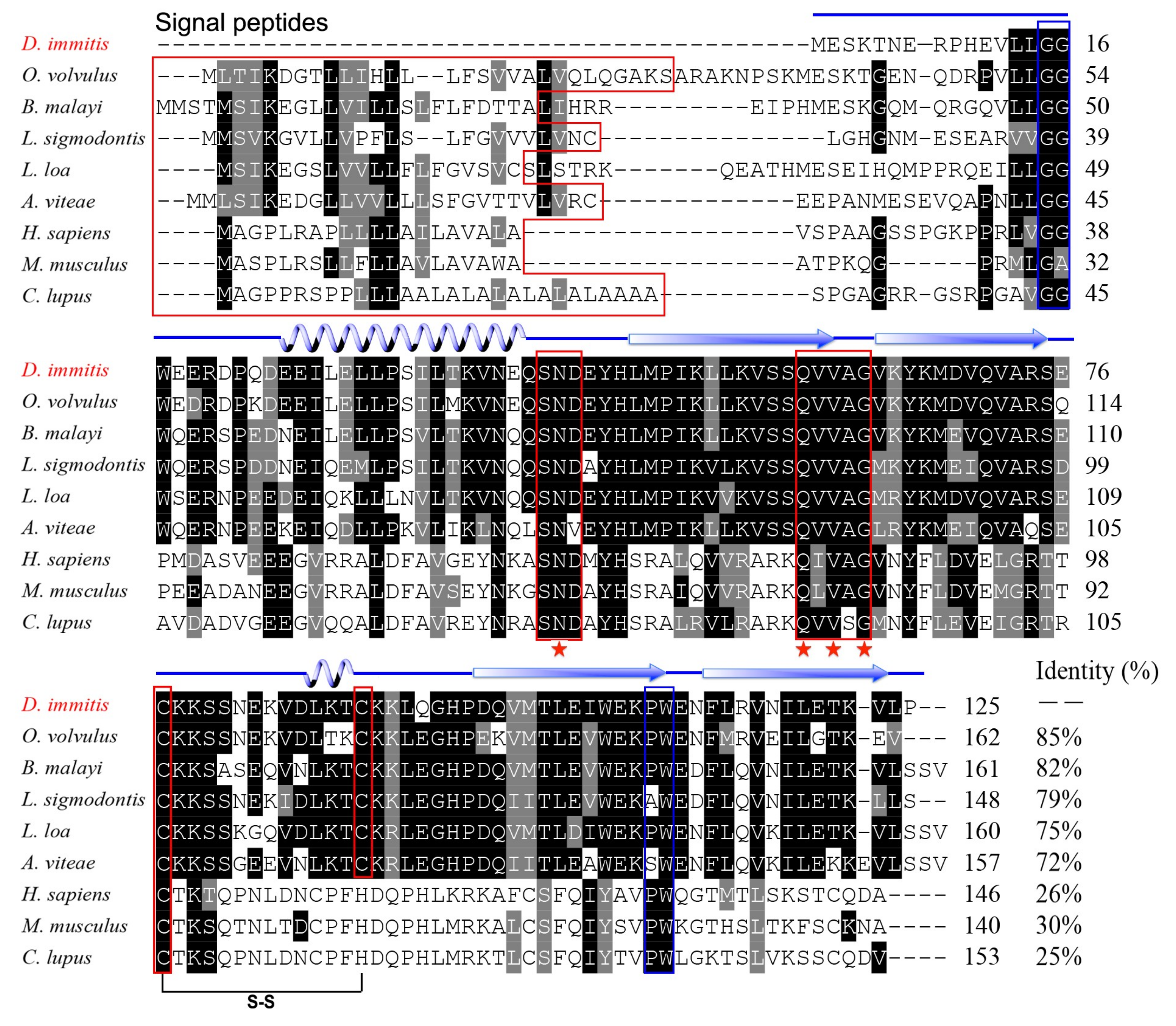
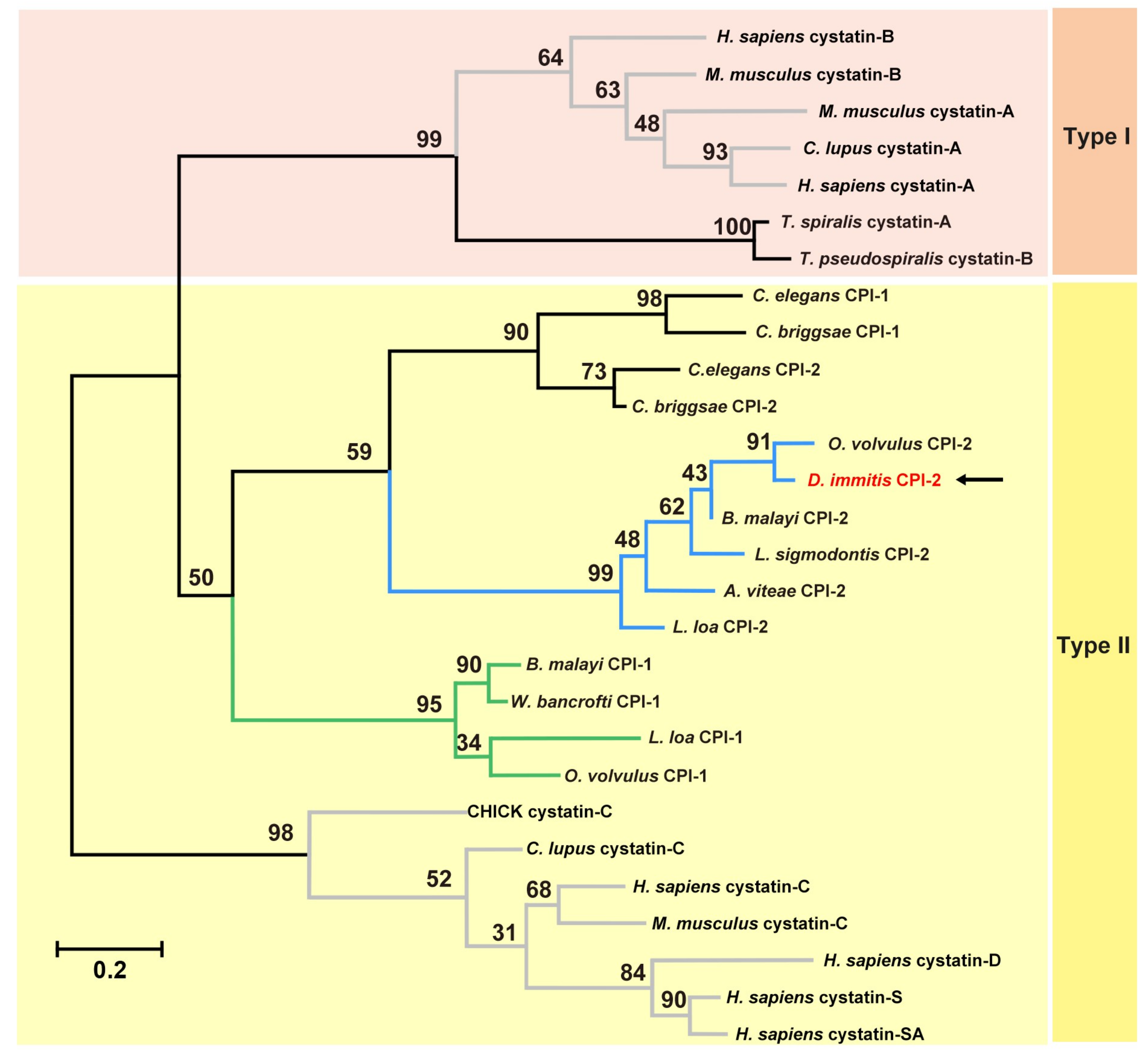
3.1. Molecular Characterisation of Di-CPI
3.2. Expression and Characterisation of Di-CPI
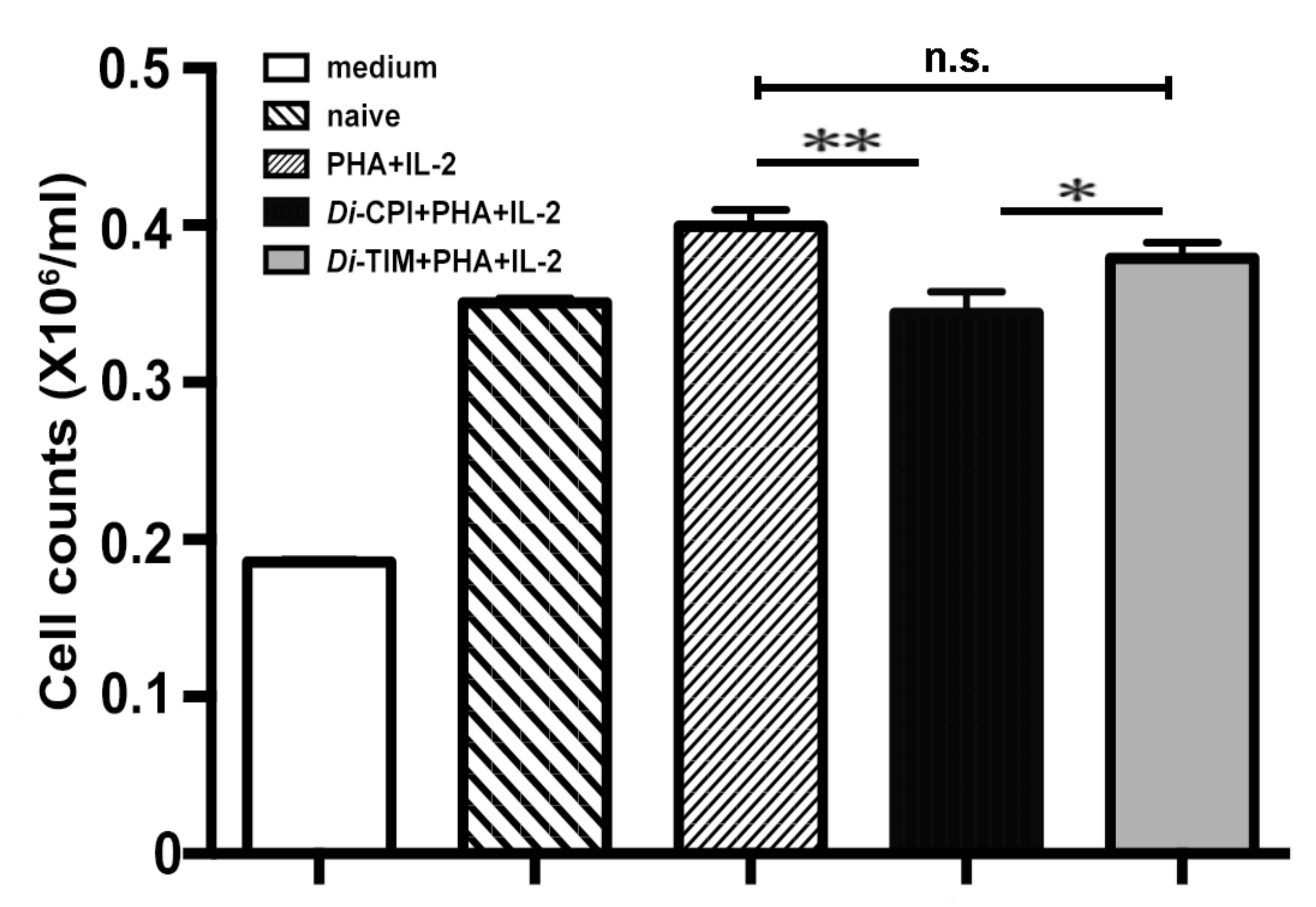
3.3. Inhibition of the Proliferation of Canine PBMCs by rDi-CPI
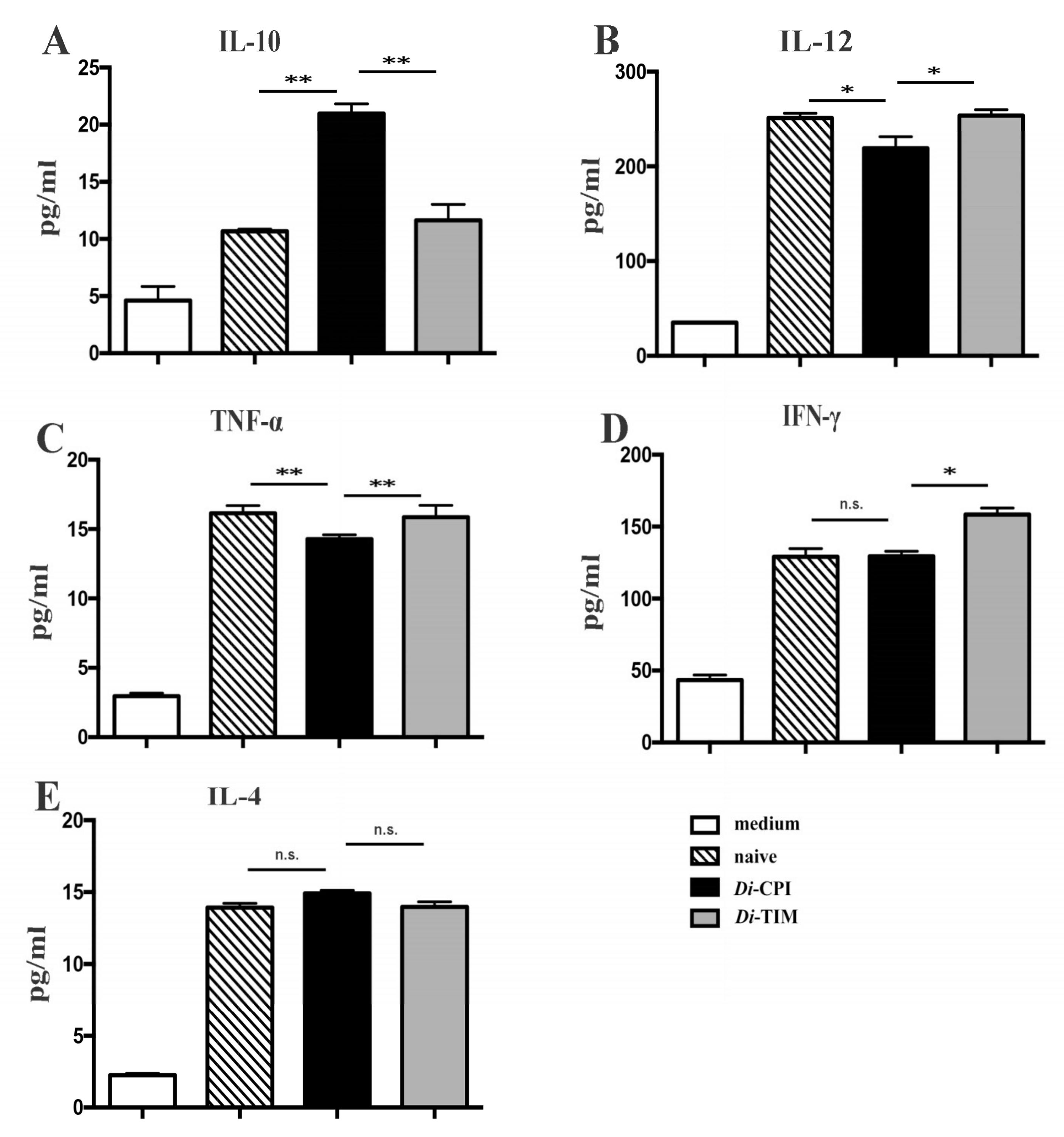
3.4. Cytokine Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Simon, F.; Siles-Lucas, M.; Morchon, R.; Gonzalez-Miguel, J.; Mellado, I.; Carreton, E.; Montoya-Alonso, J.A. Human and animal dirofilariasis: The emergence of a zoonotic mosaic. Clin. Microbiol Rev. 2012, 25, 507–544. [Google Scholar] [CrossRef] [PubMed]
- Bockova, E.; Iglodyova, A.; Kocisova, A. Potential mosquito (Diptera:Culicidae) vector of Dirofilaria repens and Dirofilaria immitis in urban areas of Eastern Slovakia. Parasitol Res. 2015, 114, 4487–4492. [Google Scholar] [CrossRef] [PubMed]
- Dillon, R.A. Influence of number of parasites and exercise on the severity of heartworm disease in dogs. Proc. Heartworm Symp. 1995, 113, 95. [Google Scholar]
- Kitagawa, H.; Ishihara, K.; Sasaki, Y. Canine dirofilarial hemoglobinuria: Changes in right heart hemodynamics and heartworm migration from pulmonary artery towards right atrium following β 1-blocker administration. J. Vet. Med. Sci 1987, 49, 1081–1086. [Google Scholar] [CrossRef]
- Simón, F.; Lopez-Belmonte, J.; Marcos-Atxutegi, C.; Morchón García, R.; R Martín-Pacho, J. What is happening outside North America regarding human Dirofilariasis? Vet. Parasitol 2005, 133, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Theis, J.H. Public health aspects of dirofilariasis in the United States. Vet. Parasitol 2005, 133, 157–180. [Google Scholar] [CrossRef] [PubMed]
- Simón, F.; Kramer, L.H.; Román, A.; Blasini, W.; Morchón, R.; Marcos-Atxutegi, C.; Grandi, G.; Genchi, C. Immunopathology of Dirofilaria immitis infection. Vet. Res. Commun 2007, 31, 161–171. [Google Scholar] [CrossRef]
- McCall, J.W.; Genchi, C.; Kramer, L.H.; Guerrero, J.; Venco, L.J.A.i.p. Heartworm disease in animals and humans. Adv. Parasit 2008, 66, 193–285. [Google Scholar]
- Kadipasaoglu, A.K.; Bilge, F.H. Partial characterization of the adsorbed protein layer on Dirofilaria immitis (Nematoda) cuticle. Parasitol Res. 1989, 75, 554–558. [Google Scholar] [CrossRef]
- Bilge, F.H.; Kadipasaoglu, K.A.; Mccormick, C.M.; Baier, R.E. Surface characterization of the cuticle of Dirofilaria immitis. J. Biomed. Mater. Res. A 1989, 23, 1027–1047. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, S.; Lucius, R. Modulation of host immune responses by nematode cystatins. Int J. Parasitol 2003, 33, 1291–1302. [Google Scholar] [CrossRef]
- Turk, V.; Stoka, V.; Turk, D. Cystatins: Biochemical and structural properties, and medical relevance. Front. Biosci 2008, 13, 5780–5786. [Google Scholar] [CrossRef]
- Cancela, M.; Corvo, I.; da Salva, E.; Teichmann, A.; Roche, L.; Diaz, A.; Tort, J.F.; Ferreira, H.B.; Zaha, A. Functional characterization of single-domain cystatin-like cysteine proteinase inhibitors expressed by the trematode Fasciola hepatica. Parasitology 2017, 144, 1695–1707. [Google Scholar] [CrossRef]
- Nata; Kopitarjerala, A. The Role of Cysteine Proteinases and their Inhibitors in the Host-Pathogen Cross Talk. Curr Protein Pept Sc 2012, 13, 767–775. [Google Scholar]
- Klotz, C.; Ziegler, T.; Figueiredo, A.S.; Rausch, S.; Hepworth, M.R.; Obsivac, N.; Sers, C.; Lang, R.; Hammerstein, P.; Lucius, R.; et al. A helminth immunomodulator exploits host signaling events to regulate cytokine production in macrophages. Plos Pathog 2011, 7, e1001248. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, G.; Li, Z.; Chen, Y.; Liu, Y.; Liu, B.; Zhong, S. Modulation of dendritic cell function and immune response by cysteine protease inhibitor from murine nematode parasite Heligmosomoides polygyrus. Immunology 2013, 138, 370–381. [Google Scholar] [CrossRef] [PubMed]
- Schuijs, M.J.; Hartmann, S.; Selkirk, M.E.; Roberts, L.B.; Openshaw, P.J.; Schnoeller, C. The Helminth-Derived Immunomodulator AvCystatin Reduces Virus Enhanced Inflammation by Induction of Regulatory IL-10+ T Cells. PLoS ONE 2016, 11, e0161885. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.; Manoury, B.; Balic, A.; Watts, C.; Maizels, R.M. Bm-CPI-2, a cystatin from Brugia malayi nematode parasites, differs from Caenorhabditis elegans cystatins in a specific site mediating inhibition of the antigen-processing enzyme AEP. Mol. Biochem. Parasitol 2005, 139, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Manoury, B.; Gregory, W.F.; Maizels, R.M.; Watts, C. Bm-CPI-2, a cystatin homolog secreted by the filarial parasite Brugia malayi, inhibits class II MHC-restricted antigen processing. Curr. Biol. 2001, 11, 447–451. [Google Scholar] [CrossRef]
- Schnoeller, C.; Rausch, S.; Pillai, S.; Avagyan, A.; Wittig, B.M.; Loddenkemper, C.; Hamann, A.; Hamelmann, E.; Lucius, R.; Hartmann, S. A Helminth Immunomodulator Reduces Allergic and Inflammatory Responses by Induction of IL-10-Producing Macrophages. J. Immunol. 2008, 180, 4265–4272. [Google Scholar] [CrossRef] [PubMed]
- Danilowicz-Luebert, E.; Steinfelder, S.; Kuhl, A.A.; Drozdenko, G.; Lucius, R.; Worm, M.; Hamelmann, E.; Hartmann, S. A nematode immunomodulator suppresses grass pollen-specific allergic responses by controlling excessive Th2 inflammation. Int. J. Parasitol 2013, 43, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Whelan, R.A.; Rausch, S.; Ebner, F.; Günzel, D.; Richter, J.F.; Hering, N.A.; Schulzke, J.-D.; Kühl, A.A.; Keles, A.; Janczyk, P.; et al. A Transgenic Probiotic Secreting a Parasite Immunomodulator for Site-Directed Treatment of Gut Inflammation. Mol. Ther. 2014, 22, 1730–1740. [Google Scholar] [CrossRef]
- Coronado, S.; Barrios, L.; Zakzuk, J.; Regino, R.; Ahumada, V.; Franco, L.; Ocampo, Y.; Caraballo, L. A recombinant cystatin from Ascaris lumbricoides attenuates inflammation of DSS-induced colitis. Parasite Immunol. 2017, 39, e12425. [Google Scholar] [CrossRef]
- Schönemeyer, A.; Lucius, R.; Sonnenburg, B.; Brattig, N.; Sabat, R.; Schilling, K.; Bradley, J.; Hartmann, S. Modulation of Human T Cell Responses and Macrophage Functions by Onchocystatin, a Secreted Protein of the Filarial Nematode Onchocerca volvulus. J. Immunol 2001, 167, 3207–3215. [Google Scholar] [CrossRef]
- Hartmann, S.; Kyewski, B.; Sonnenburg, B.; Lucius, R. A filarial cysteine protease inhibitor down-regulates T cell proliferation and enhances interleukin-10 production. Eur. J. Immunol 1997, 27, 2253–2260. [Google Scholar] [CrossRef]
- Khatri, V.; Amdare, N.; Tarnekar, A.; Goswami, K.; Reddy, M.V.R. Brugia malayi cystatin therapeutically ameliorates dextran sulfate sodium-induced colitis in mice. J. Digest Dis. 2016, 16, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Won, J.S.; Kyoung, C.M.; Kyung, P.M.; Kang, S.A.; Na, B.K.; Cheol, A.S.; Dong-Hee, K.; Yu, H.S. Parasitic Helminth Cystatin Inhibits DSS-Induced Intestinal Inflammation Via IL-10+F4/80+ Macrophage Recruitment. Korean J. Parasitol 2011, 49, 245–254. [Google Scholar]
- 28 Schierack, P.; Lucius, R.; Sonnenburg, B.; Schilling, K.; Hartmann, S. Parasite-Specific Immunomodulatory Functions of Filarial Cystatin. Infect. Immun. 2003, 71, 2422–2429. [Google Scholar] [CrossRef]
- Xie, Y.; Zhou, X.; Chen, L.; Zhang, Z.; Wang, C.; Gu, X.; Wang, T.; Peng, X.; Yang, G. Cloning and characterization of a novel sigma-like glutathione S-transferase from the giant panda parasitic nematode, Baylisascaris schroederi. Parasite Vector 2015, 8, 44. [Google Scholar] [CrossRef]
- Yan, F.; Lan, J.; Zhang, Z.; Rong, H.; Wu, X.; Yang, D.; Zhang, R.; Zheng, W.; Nie, H.; Yue, X. Novel Insights into the Transcriptome of Dirofilaria immitis. PLoS ONE 2012, 7, e41639. [Google Scholar]
- Ma, L.; Blackshields, G.; Np, B.; Chenna, R.; Pa, M.G.; Mcwilliam, H.; Valentin, F.; Im, W.; Wilm, A.; Lopez, R. ClustalW and ClustalX version 2. Curr. Protoc. Bioinform. 2007, 23, 2947–2948. [Google Scholar]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Xu, J.; Huang, X.; He, M.; Ren, Y.; Shen, N.; Li, C.; He, R.; Xie, Y.; Gu, X.; Jing, B. Identification of a novel PYP-1 gene in Sarcoptes scabiei and its potential as a serodiagnostic candidate by indirect-ELISA. Parasitology 2017, 145, 752–761. [Google Scholar] [CrossRef]
- Gregory, W.F.; Maizels, R.M. Cystatins from filarial parasites: Evolution, adaptation and function in the host-parasite relationship. Int. J. Biochem. Cell Biol. 2008, 40, 1389–1398. [Google Scholar] [CrossRef]
- Lustigman, S.; Brotman, B.; Huima, T.; Prince, A.M. Characterization of an Onchocerca volvulus cDNA clone encoding a genus specific antigen present in infective larvae and adult worms. Mol. Biochem. Parasitol. 1991, 45, 65–75. [Google Scholar] [CrossRef]
- Lustigman, S.; Brotman, B.; Huima, T.; Prince, A.M.; Mckerrow, J.H. Molecular cloning and characterization of onchocystatin, a cysteine proteinase inhibitor of Onchocerca volvulus. J. Biol. Chem. 1992, 267, 17339–17346. [Google Scholar]
- Hashmi, S.; Zhang, J.; Oksov, Y.; Ji, Q.; Lustigman, S. The Caenorhabditis elegans CPI-2a cystatin-like inhibitor has an essential regulatory role during oogenesis and fertilization. J. Biol. Chem. 2006, 281, 28415–28429. [Google Scholar] [CrossRef]
- Alvarez-Fernandez, M.; Barrett, A.J.; Gerhartz, B.; Dando, P.M.; Ni, J.; Abrahamson, M. Inhibition of mammalian legumain by some cystatins is due to a novel second reactive site. J. Biol. Chem. 1999, 274, 19195–19203. [Google Scholar] [CrossRef]
- Kopitar-Jerala, N. The role of cystatins in cells of the immune system. FEBS Lett. 2006, 580, 6295–6301. [Google Scholar] [CrossRef]
- Pfaff, A.W.; Schulz-Key, H.; Soboslay, P.T.; Taylor, D.W.; Maclennan, K.; Hoffmann, W.H. Litomosoides sigmodontis cystatin acts as an immunomodulator during experimental filariasis. Int. J. Parasitol. 2002, 32, 171–178. [Google Scholar] [CrossRef]
- Geary, J.; Satti, M.; Moreno, Y.; Madrill, N.; Whitten, D.; Headley, S.A.; Agnew, D.; Geary, T.; Mackenzie, C.J.P. First analysis of the secretome of the canine heartworm, Dirofilaria immitis. Parasite Vector 2012, 5, 140. [Google Scholar] [CrossRef]
- Twu, O.; de Miguel, N.; Lustig, G.; Stevens, G.C.; Vashisht, A.A.; Wohlschlegel, J.A.; Johnson, P.J.J.P.p. Trichomonas vaginalis exosomes deliver cargo to host cells and mediate host: Parasite interactions. PLoS Pathog. 2013, 9, e1003482. [Google Scholar] [CrossRef]
- Marcilla, A.; Trelis, M.; Cortés, A.; Sotillo, J.; Cantalapiedra, F.; Minguez, M.T.; Valero, M.L.; Del Pino, M.M.S.; Muñoz-Antoli, C.; Toledo, R.J.P.o. Extracellular vesicles from parasitic helminths contain specific excretory/secretory proteins and are internalized in intestinal host cells. PLoS ONE 2012, 7, e45974. [Google Scholar] [CrossRef]
- Bernal, D.; Trelis, M.; Montaner, S.; Cantalapiedra, F.; Galiano, A.; Hackenberg, M.; Marcilla, A.J.J.o.p. Surface analysis of Dicrocoelium dendriticum. The molecular characterization of exosomes reveals the presence of miRNAs. J. Proteomics 2014, 105, 232–241. [Google Scholar] [CrossRef]
- Silverman, J.M.; Clos, J.; de’Oliveira, C.C.; Shirvani, O.; Fang, Y.; Wang, C.; Foster, L.J.; Reiner, N.E.J.J.C.S. An exosome-based secretion pathway is responsible for protein export from Leishmania and communication with macrophages. J. Cell Sci. 2010, 123, 842–852. [Google Scholar] [CrossRef]
- Buck, A.H.; Coakley, G.; Simbari, F.; McSorley, H.J.; Quintana, J.F.; Le Bihan, T.; Kumar, S.; Abreu-Goodger, C.; Lear, M.; Harcus, Y.J.N.c. Exosomes secreted by nematode parasites transfer small RNAs to mammalian cells and modulate innate immunity. Nat. Commun. 2014, 5, 5488. [Google Scholar] [CrossRef]
- Zamanian, M.; Fraser, L.M.; Agbedanu, P.N.; Harischandra, H.; Moorhead, A.R.; Day, T.A.; Bartholomay, L.C.; Kimber, M.J.J.P.n.t.d. Release of small RNA-containing exosome-like vesicles from the human filarial parasite Brugia malayi. PLoS Neglected Trop D 2015, 9, e0004069. [Google Scholar] [CrossRef]
- Johnstone, I.L.; Barry, J.D. Temporal reiteration of a precise gene expression pattern during nematode development. Embo J. 1996, 15, 3633–3639. [Google Scholar] [CrossRef]
- Marcos-Atxutegi, C.; Kramer, L.H.; Fernandez, I.; Simoncini, L.; Genchi, M.; Prieto, G.; Simón, F. Th1 response in BALB/c mice immunized with Dirofilaria immitis soluble antigens: A possible role for Wolbachia? Vet. Parasitol. 2003, 112, 117–130. [Google Scholar] [CrossRef]
- Morchón, R.; López-Belmonte, J.; Bazzocchi, C.; Grandi, G.; Kramer, L.; Simón, F.J.V.i. Dogs with patent Dirofilaria immitis infection have higher expression of circulating IL-4, IL-10 and iNOS mRNA than those with occult infection. Vet. Immunol. Immuno. 2007, 115, 184–188. [Google Scholar] [CrossRef]
- Osborne, J.; Devaney, E. Interleukin-10 and Antigen-Presenting Cells Actively Suppress Th1 Cells in BALB/c Mice Infected with the Filarial Parasite Brugia pahangi. Infect. Immunol. 1999, 67, 1599–1605. [Google Scholar]
- Mahanty, S.; Mollis, S.N.; Ravichandran, M.; Abrams, J.S.; Kumaraswami, V.; Jayaraman, K.; Ottesen, E.A.; Nutman, T.B. High Levels of Spontaneous and Parasite Antigen-Driven Interieukin-10 Production Are Associated with Antigen-Specific Hyporesponsiveness in Human Lymphatic Filariasis. J. Infect. Dis. 1996, 173, 769–772. [Google Scholar] [CrossRef]
- Ziegler, T.; Rausch, S.; Steinfelder, S.; Klotz, C.; Hepworth, M.R.; Kühl, A.A.; Burda, P.-C.; Lucius, R.; Hartmann, S. A Novel Regulatory Macrophage Induced by a Helminth Molecule Instructs IL-10 in CD4+ T Cells and Protects against Mucosal Inflammation. J. Immunol. 2015, 194, 1555–1564. [Google Scholar] [CrossRef]
- Maizels, R.M.; Gomez-Escobar, N.; Gregory, W.F.; Murray, J.; Zang, X. Immune evasion genes from filarial nematodes. INT J. Parasitol. 2001, 31, 889–898. [Google Scholar] [CrossRef]
- Maizels, R.M.; Blaxter, M.L.; Scott, A.L. Immunological genomics of Brugia malayi: Filarial genes implicated in immune evasion and protective immunity. Para. Immunol. 2001, 23, 327–344. [Google Scholar] [CrossRef]
- Vray, B.; Hartmann, S.; Hoebeke, J. Immunomodulatory properties of cystatins. Cell Mol. Life Sci. 2002, 59, 1503–1512. [Google Scholar] [CrossRef]
- Xiao, Y.; Ju, L.; Yuan, Y.; Wei, C.; Man, S.; Ximei, Z.; Zhongkai, W.J.P.R. Cloning, expression and characterisation of a type II cystatin from Schistosoma japonicum, which could regulate macrophage activation. Parasitol. Res. 2014, 113, 3985–3992. [Google Scholar]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, X.; Xu, J.; Song, H.; Liu, Y.; Wu, M.; Zhang, H.; Jing, B.; Lai, W.; Gu, X.; Xie, Y.; et al. Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion. Genes 2019, 10, 300. https://doi.org/10.3390/genes10040300
Dong X, Xu J, Song H, Liu Y, Wu M, Zhang H, Jing B, Lai W, Gu X, Xie Y, et al. Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion. Genes. 2019; 10(4):300. https://doi.org/10.3390/genes10040300
Chicago/Turabian StyleDong, Xiaowei, Jing Xu, Hongyu Song, Yuchen Liu, Maodi Wu, Haojie Zhang, Bo Jing, Weimin Lai, Xiaobin Gu, Yue Xie, and et al. 2019. "Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion" Genes 10, no. 4: 300. https://doi.org/10.3390/genes10040300
APA StyleDong, X., Xu, J., Song, H., Liu, Y., Wu, M., Zhang, H., Jing, B., Lai, W., Gu, X., Xie, Y., Peng, X., & Yang, G. (2019). Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion. Genes, 10(4), 300. https://doi.org/10.3390/genes10040300




