Bayes Factor-Based Regulatory Gene Network Analysis of Genome-Wide Association Study of Economic Traits in a Purebred Swine Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Genotypes and Phenotypes
2.2. Measurement of Economic Traits
2.3. Response Variable
2.4. Bayesian Method for Genome Wide Association Study
2.5. Identification of Significant Window Regions and Single Nucleotide Polymorphism Markers
2.6. Association Weight Matrix Construction
2.7. Network Analysis Using Transcription Factor and Target Gene Information
3. Results and Discussion
3.1. Genome-Wide Association Study Using Single Nucleotide Polymorphisms Markers with Illumina PorcineSNP60
3.2. Growth-Related Traits
3.3. Production-Related Traits
3.4. Reproduction-Related Traits
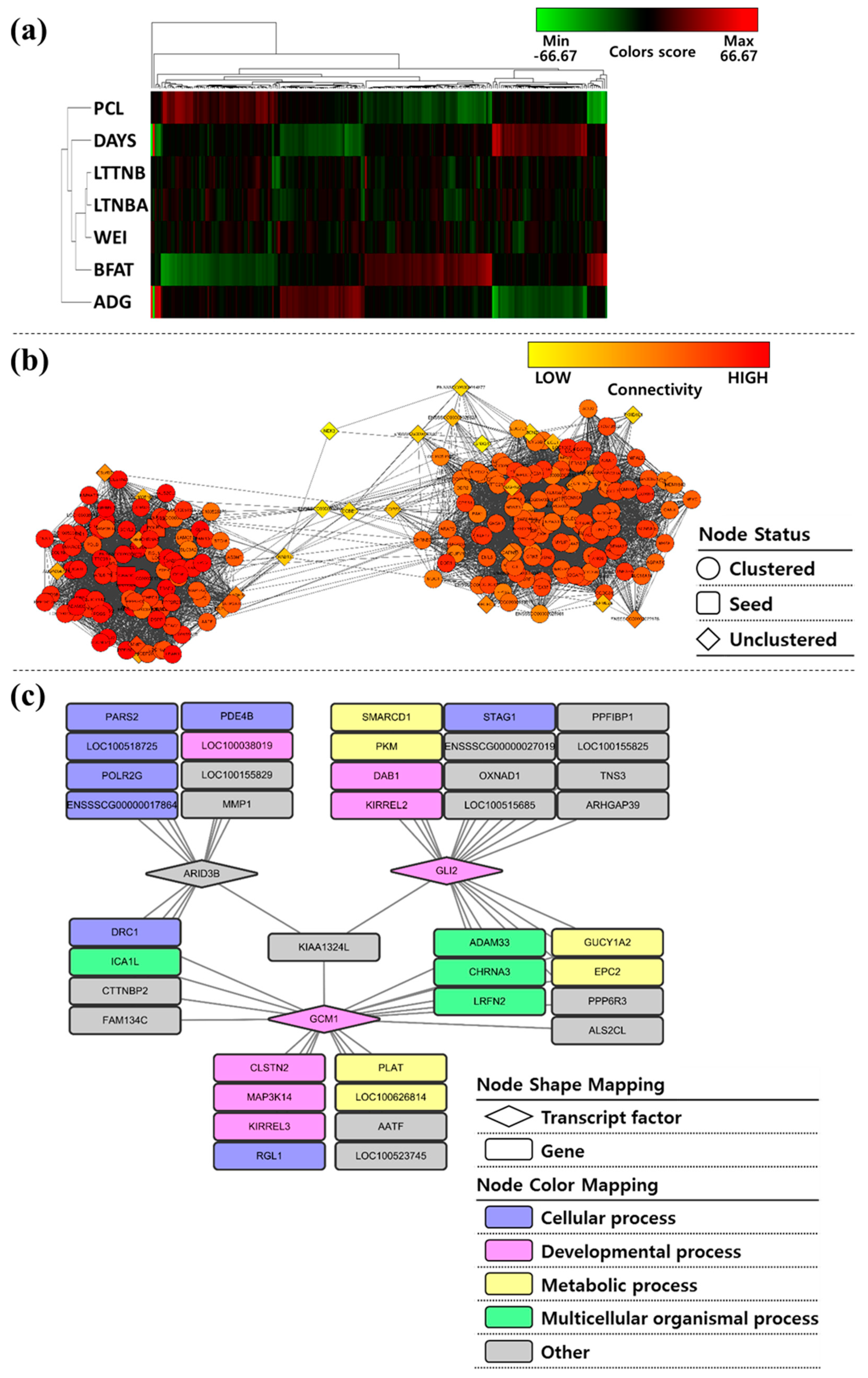
3.5. Co-Association Network Based on Association Weight Matrix
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Andersson, L. Genome-wide association analysis in domestic animals: a powerful approach for genetic dissection of trait loci. Genetica 2009, 136, 341–349. [Google Scholar] [CrossRef]
- Li, J.; Das, K.; Fu, G.; Li, R.; Wu, R. The Bayesian lasso for genome-wide association studies. Bioinformatics 2011, 27, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Meuwissen, T.H.E.; Hayes, B.J.; Goddard, M.E. Prediction of Total Genetic Value Using Genome-Wide Dense Marker Maps. Genetics 2001, 157, 1819. [Google Scholar]
- Dashab, G.R.; Kadri, N.K.; Shariati, M.M.; Sahana, G. Comparison of linear mixed model analysis and genealogy-based haplotype clustering with a Bayesian approach for association mapping in a pedigreed population. BMC Proc 2012, 6 (Suppl. S2), S4. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Xu, S. Mapping viability loci using molecular markers. Heredity (Edinb) 2003, 90, 459–467. [Google Scholar] [CrossRef]
- Fortes, M.R.; Reverter, A.; Zhang, Y.; Collis, E.; Nagaraj, S.H.; Jonsson, N.N.; Prayaga, K.C.; Barris, W.; Hawken, R.J. Association weight matrix for the genetic dissection of puberty in beef cattle. Proc. Natl. Acad. Sci. USA 2010, 107, 13642–13647. [Google Scholar] [CrossRef] [PubMed]
- Ramayo-Caldas, Y.; Fortes, M.R.; Hudson, N.J.; Porto-Neto, L.R.; Bolormaa, S.; Barendse, W.; Kelly, M.; Moore, S.S.; Goddard, M.E.; Lehnert, S.A.; et al. A marker-derived gene network reveals the regulatory role of PPARGC1A, HNF4G, and FOXP3 in intramuscular fat deposition of beef cattle. J. Anim. Sci. 2014, 92, 2832–2845. [Google Scholar] [CrossRef]
- Puig-Oliveras, A.; Ballester, M.; Corominas, J.; Revilla, M.; Estellé, J.; Fernández, A.I.; Ramayo-Caldas, Y.; Folch, J.M. A Co-Association Network Analysis of the Genetic Determination of Pig Conformation, Growth and Fatness. PLoS ONE 2014, 9, e114862. [Google Scholar] [CrossRef]
- Duarte, D.A.S.; Fortes, M.R.S.; Duarte, M.d.S.; Guimarães, S.E.F.; Verardo, L.L.; Veroneze, R.; Ribeiro, A.M.F.; Lopes, P.S.; de Resende, M.D.V.; Fonseca e Silva, F. Genome-wide association studies, meta-analyses and derived gene network for meat quality and carcass traits in pigs. Anim. Prod. Sci. 2018, 58, 1100–1108. [Google Scholar] [CrossRef]
- Kang, J.-H.; Lee, E.-A.; Hong, K.-C.; Kim, J.-M. Regulatory gene network from a genome-wide association study for sow lifetime productivity traits. Anim. Genet. 2018, 49, 254–258. [Google Scholar] [CrossRef]
- Aguilar, I.; Misztal, I.; Tsuruta, S.; Legarra, A.; Wang, H. PREGSF90–POSTGSF90: Computational tools for the implementation of single-step genomic selection and genome-wide association with ungenotyped individuals in BLUPF90 programs. In Proceedings of the 10th World Congress of Genetics Applied to Livestock Production, Vancouver, BC, Canada, 17–22 August 2014. [Google Scholar]
- Sargolzaei, M.; Chesnais, J.P.; Schenkel, F.S. A new approach for efficient genotype imputation using information from relatives. BMC Genomics 2014, 15, 478. [Google Scholar] [CrossRef]
- Choi, J.G.; Cho, C.I.; Choi, I.S.; Lee, S.S.; Choi, T.J.; Cho, K.H.; Park, B.H.; Choy, Y.H. Genetic parameter estimation in seedstock Swine population for growth performances. Asian-Australas. J. Anim. Sci. 2013, 26, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Choy, Y.H.; Mahboob, A.; Cho, C.I.; Choi, J.G.; Choi, I.S.; Choi, T.J.; Cho, K.H.; Park, B.H. Genetic Parameters of Pre-adjusted Body Weight Growth and Ultrasound Measures of Body Tissue Development in Three Seedstock Pig Breed Populations in Korea. Asian-Australas. J. Anim. Sci. 2015, 28, 1696. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gilmour, A.; Gogel, B.; Cullis, B.; Welham, S.; Thompson, R. ASReml user guide release 4.1 structural specification. Hemel hempstead: VSN international ltd, 2015. [Google Scholar]
- Garrick, D.J.; Taylor, J.F.; Fernando, R.L. Deregressing estimated breeding values and weighting information for genomic regression analyses. Genet. Sel. Evol. 2009, 41, 55. [Google Scholar] [CrossRef] [PubMed]
- Saatchi, M.; Schnabel, R.D.; Rolf, M.M.; Taylor, J.F.; Garrick, D.J. Accuracy of direct genomic breeding values for nationally evaluated traits in US Limousin and Simmental beef cattle. Genet. Sel. Evol. 2012, 44, 38. [Google Scholar] [CrossRef]
- Garrick, D.J.; Fernando, R.L. Implementing a QTL detection study (GWAS) using genomic prediction methodology. In Genome-Wide Association Studies and Genomic Prediction; Humana Press: Totowa, NJ, USA, 2013; pp. 275–298. [Google Scholar]
- Sun, X.; Habier, D.; Fernando, R.L.; Garrick, D.J.; Dekkers, J.C. Genomic breeding value prediction and QTL mapping of QTLMAS2010 data using Bayesian methods. In Proceedings of the BMC Proceedings, Poznan, Poland, 17–18 May 2010. [Google Scholar]
- Fan, B.; Onteru, S.K.; Du, Z.-Q.; Garrick, D.J.; Stalder, K.J.; Rothschild, M.F. Genome-wide association study identifies loci for body composition and structural soundness traits in pigs. PLoS ONE 2011, 6, e14726. [Google Scholar] [CrossRef]
- Yi, N.; Yandell, B.S.; Churchill, G.A.; Allison, D.B.; Eisen, E.J.; Pomp, D. Bayesian model selection for genome-wide epistatic quantitative trait loci analysis. Genetics 2005, 170, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Kass, R.E.; Raftery, A.E. Bayes factors. J. Am. Stat. Assoc. 1995, 90, 773–795. [Google Scholar] [CrossRef]
- Watson-Haigh, N.S.; Kadarmideen, H.N.; Reverter, A. PCIT: an R package for weighted gene co-expression networks based on partial correlation and information theory approaches. Bioinformatics 2010, 26, 411–413. [Google Scholar] [CrossRef]
- Caraux, G.; Pinloche, S. PermutMatrix: a graphical environment to arrange gene expression profiles in optimal linear order. Bioinformatics 2005, 21, 1280–1281. [Google Scholar] [CrossRef]
- Bader, G.D.; Hogue, C.W. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinformatics 2003, 4, 2. [Google Scholar] [CrossRef]
- Hu, H.; Miao, Y.-R.; Jia, L.-H.; Yu, Q.-Y.; Zhang, Q.; Guo, A.-Y. AnimalTFDB 3.0: a comprehensive resource for annotation and prediction of animal transcription factors. Nucleic Acids Res. 2018, 47, D33–D38. [Google Scholar] [CrossRef]
- CIS-BP. Available online: http://cisbp.ccbr.utoronto.ca/ (accessed on 15 April 2015).
- Khan, A.; Fornes, O.; Stigliani, A.; Gheorghe, M.; Castro-Mondragon, J.A.; van der Lee, R.; Bessy, A.; Cheneby, J.; Kulkarni, S.R.; Tan, G. JASPAR 2018: update of the open-access database of transcription factor binding profiles and its web framework. Nucleic Acids Res. 2017, 46, D260–D266. [Google Scholar] [CrossRef]
- Ensembl ASIA. Available online: http://asia.ensembl.org/biomart/martview (accessed on 15 April 2019).
- Grant, C.E.; Bailey, T.L.; Noble, W.S. FIMO: scanning for occurrences of a given motif. Bioinformatics 2011, 27, 1017–1018. [Google Scholar] [CrossRef]
- Touzet, H.; Varre, J.S. Efficient and accurate P-value computation for Position Weight Matrices. Algorithms Mol. Biol. 2007, 2, 15. [Google Scholar] [CrossRef]
- Mi, H.; Huang, X.; Muruganujan, A.; Tang, H.; Mills, C.; Kang, D.; Thomas, P.D. PANTHER version 11: expanded annotation data from Gene Ontology and Reactome pathways, and data analysis tool enhancements. Nucleic Acids Res. 2016, 45, D183–D189. [Google Scholar] [CrossRef]
- Van Laere, A.S.; Nguyen, M.; Braunschweig, M.; Nezer, C.; Collette, C.; Moreau, L.; Archibald, A.L.; Haley, C.S.; Buys, N.; Tally, M.; et al. A regulatory mutation in IGF2 causes a major QTL effect on muscle growth in the pig. Nature 2003, 425, 832–836. [Google Scholar] [CrossRef]
- Zhu, P.R.; Wu, Q.Y.; Yu, M.M.; Zhang, M.C.; Ni, M.X.; Liu, S.M.; Jiang, W.J.; Zhang, J.; Li, W.W.; Cao, J.; et al. [Nucleotide polymorphism rs4880 of the SOD2 gene and the risk of male infertility]. Zhonghua nan ke xue (Natl. J. Androl.) 2017, 23, 137–141. [Google Scholar]
- Hernandez, S.; Finlayson, H.; Ashworth, C.; Haley, C.; Archibald, A. A genome-wide linkage analysis for reproductive traits in F 2 Large White × Meishan cross gilts. Anim. Genet. 2014, 45, 191–197. [Google Scholar] [CrossRef]
- Wood, J.J.; Boyne, J.R.; Paulus, C.; Jackson, B.R.; Nevels, M.M.; Whitehouse, A.; Hughes, D.J. ARID3B: a Novel Regulator of the Kaposi’s Sarcoma-Associated Herpesvirus Lytic Cycle. J. Virol. 2016, 90, 9543–9555. [Google Scholar] [CrossRef]
- Casanova, J.C.; Uribe, V.; Badia-Careaga, C.; Giovinazzo, G.; Torres, M.; Sanz-Ezquerro, J.J. Apical ectodermal ridge morphogenesis in limb development is controlled by Arid3b-mediated regulation of cell movements. Development 2011, 138, 1195–1205. [Google Scholar] [CrossRef]
- Wirschell, M.; Olbrich, H.; Werner, C.; Tritschler, D.; Bower, R.; Sale, W.S.; Loges, N.T.; Pennekamp, P.; Lindberg, S.; Stenram, U.; et al. The nexin-dynein regulatory complex subunit DRC1 is essential for motile cilia function in algae and humans. Nat. Genet. 2013, 45, 262–268. [Google Scholar] [CrossRef]
- Meka, H.; Werner, F.; Cordell, S.C.; Onesti, S.; Brick, P. Crystal structure and RNA binding of the Rpb4/Rpb7 subunits of human RNA polymerase II. Nucleic Acids Res. 2005, 33, 6435–6444. [Google Scholar] [CrossRef]
- Sofou, K.; Kollberg, G.; Holmstrom, M.; Davila, M.; Darin, N.; Gustafsson, C.M.; Holme, E.; Oldfors, A.; Tulinius, M.; Asin-Cayuela, J. Whole exome sequencing reveals mutations in NARS2 and PARS2, encoding the mitochondrial asparaginyl-tRNA synthetase and prolyl-tRNA synthetase, in patients with Alpers syndrome. Mol. Genet. Genom. Med. 2015, 3, 59–68. [Google Scholar] [CrossRef]
- Yamada, K.; Ogawa, H.; Honda, S.; Harada, N.; Okazaki, T. A GCM motif protein is involved in placenta-specific expression of human aromatase gene. J. Biol. Chem. 1999, 274, 32279–32286. [Google Scholar] [CrossRef]
- Lee, R.T.; Zhao, Z.; Ingham, P.W. Hedgehog signalling. Development 2016, 143, 367–372. [Google Scholar] [CrossRef]
- Pan, Y.B.; Gong, Y.; Ruan, H.F.; Pan, L.Y.; Wu, X.K.; Tang, C.; Wang, C.J.; Zhu, H.B.; Zhang, Z.M.; Tang, L.F.; et al. Sonic hedgehog through Gli2 and Gli3 is required for the proper development of placental labyrinth. Cell Death Amp Dis. 2015, 6, e1653. [Google Scholar] [CrossRef]

| Trait 1 | N | Mean | SD | Min. | Max. | |||
|---|---|---|---|---|---|---|---|---|
| BFAT (mm) | 39,406 | 13.76 | 2.96 | 7.60 | 23.20 | 2.93 | 6.99 | 0.36 |
| ADG (g) | 39,516 | 609.30 | 74.48 | 449.00 | 952.00 | 0.12 | 0.35 | 0.42 |
| DAYS (days) | 39,221 | 149.3 | 14.4 | 112.00 | 188.00 | 0.44 | 1.26 | 0.35 |
| PCL (%) | 39,508 | 58.14 | 2.97 | 49.60 | 65.80 | 3.48 | 8.33 | 0.42 |
| LTTNB | 39,518 | 11.80 | 2.95 | 2 | 25 | 0.76 | 7.20 | 0.11 |
| LTNBA | 39,518 | 10.65 | 2.70 | 1 | 24 | 0.73 | 7.07 | 0.10 |
| WEI | 12,975 | 4.88 | 1.40 | 1 | 15 | 0.15 | 1.86 | 0.08 |
| Trait 1 | SSC_Mb 2 | GV% | Informative SNP | rs Number | Position (Mb) | Effect | BF 3 | Region Annotation | Gene Annotation |
|---|---|---|---|---|---|---|---|---|---|
| BFAT | 2_162 | 3.51 | ASGA0084103 | rs81341288 | 162.15 | 0.092 | 57.2 | intronic | COX8H, IFITM2, IFITM3 |
| ASGA0085784 | rs81328276 | 162.3 | 0.085 | 49.58 | intronic | IFITM2, IFITM3 | |||
| 2_76 | 2.87 | MARC0048160 | rs81239450 | 76.09 | −0.076 | 41.77 | intergenic | GNA11(dist = 3121), THOP1(dist = 278492) | |
| MARC0030590 | rs81224732 | 76.32 | −0.056 | 28 | intergenic | GNA11(dist = 227056), THOP1(dist = 54557) | |||
| 2_0 | 2.68 | ASGA0097367 | rs81317307 | 0.37 | 0.132 | 141.12 | intergenic | IRF7(dist = 63158), PHLDA2(dist = 62536) | |
| ASGA0098481 | rs81318741 | 0.92 | 0.039 | 34.24 | intergenic | NAP1L4(dist = 452192), FADD (dist = 644399) | |||
| 15_132 | 2.31 | INRA0050241 | rs339585634 | 132.56 | 0.126 | 244.75 | intergenic | LOC100738836(dist = 879831), ARPC2(dist = 791438) | |
| 5_65 | 2.15 | ALGA0114229 | rs81343150 | 65.63 | 0.146 | 304.09 | intergenic | MFAP5(dist = 49502), CD163L1(dist = 347959) | |
| 14_142 | 1.13 | ALGA0082467 | rs80835167 | 142.22 | −0.084 | 126.1 | intergenic | MCMBP (dist = 953163), FGFR2(dist = 277982 | |
| 8_11 | 1.08 | MARC0034108 | rs81227701 | 11.32 | 0.04 | 40.69 | intergenic | CD38(dist = 542438), QDPR (dist = 1008577) | |
| 14_4 | 0.89 | ALGA0074404 | rs80792287 | 4.23 | 0.078 | 136.21 | intergenic | SYK (dist = 1355883), LPL (dist = 230942) | |
| 16_79 | 0.93 | ALGA0091967 | rs81462835 | 79.94 | −0.077 | 133.5 | intergenic | TNIP1(dist = 1618306) | |
| ADG | 17_17 | 1.88 | INRA0052808 | rs342665431 | 17.55 | 0.03 | 298.75 | intergenic | BMP2(dist = 135846), HAO1(dist = 1265868) |
| 5_93 | 1.4 | DRGA0006163 | rs345168974 | 93.83 | 0.025 | 209.22 | intergenic | SOCS2(dist = 113304), BTG1(dist = 1259303) | |
| 10_28 | 1.1 | ALGA0057938 | rs81422478 | 28.94 | −0.02 | 89.32 | intergenic | TNNI1(dist = 728658), ADIPOR1(dist = 328094) | |
| 1_177 | 1.03 | ALGA0006599 | rs80799429 | 177.01 | 0.015 | 54.25 | intergenic | SERPINB10(dist = 1651991), RNF152(dist = 58137) | |
| 2_2 | 0.89 | M1GA0002244 | rs81362590 | 28.34 | −0.017 | 103.16 | intronic | CPT1A | |
| DAYS | 17_17 | 2.22 | INRA0052808 | rs342665431 | 17.55 | −0.065 | 764.87 | intergenic | BMP2(dist = 135846), HAO1(dist = 1265868) |
| 5_93 | 1.12 | DRGA0006163 | rs345168974 | 93.83 | −0.032 | 111.06 | intergenic | SOCS2(dist = 113304), BTG1(dist = 1259303) | |
| PCL | 2_162 | 5.87 | ASGA0085784 | rs81328276 | 162.3 | −0.212 | 154 | intronic | IFITM2, IFITM3 |
| ASGA0084103 | rs81341288 | 162.15 | −0.13 | 61.09 | intronic | COX8H, IFITM2, IFITM3 | |||
| 2_0 | 4.48 | ASGA0097367 | rs81317307 | 0.37 | −0.287 | 1268.4 | intergenic | IRF7(dist = 63158), PHLDA2(dist = 62536) | |
| 2_76 | 1.49 | MARC0048160 | rs81239450 | 76.09 | 0.053 | 25.2 | intergenic | GNA11(dist = 3121), THOP1(dist = 278492) | |
| 15_132 | 1.37 | INRA0050241 | rs339585634 | 132.56 | −0.078 | 77.91 | intergenic | LOC100738836(dist = 879831), ARPC2(dist = 791438) | |
| 5_65 | 1.28 | ALGA0114229 | rs81343150 | 65.64 | −0.362 | 214.29 | intergenic | MFAP5(dist = 49502), CD163L1(dist = 347959) | |
| 6_157 | 1.15 | M1GA0009131 | rs81394508 | 157.39 | −0.175 | 221.8 | intergenic | GUCA2B (dist = 903014), MIR30C-1(dist = 85847) | |
| 14_4 | 1.06 | ALGA0074404 | rs80792287 | 4.23 | −0.121 | 281.92 | intergenic | SYK (dist = 1355883), LPL (dist = 230942) | |
| 7_18 | 0.99 | MARC0003814 | rs80894864 | 18.13 | 0.15 | 205.05 | intergenic | ID4(dist = 1948801), PRL (dist = 284338) | |
| 4_42 | 0.86 | INRA0013856 | rs337241703 | 42.84 | 0.131 | 110.57 | intronic | CPQ |
| Trait 1 | SSC_Mb 2 | GV% | Informative SNP | rs Number | Position (Mb) | Effect | BF 3 | Region Annotation | Gene Annotation |
|---|---|---|---|---|---|---|---|---|---|
| LTTNB | 16_78 | 1.45 | ASGA0074339 | rs81462568 | 78.56 | 0.057 | 79.31 | iIntergenic | TNIP1 (dist = 236491) |
| 1_9 | 1.13 | DIAS0003564 | rs80972878 | 9.86 | −0.044 | 53.1 | iIntergenic | SOD2 (dist = 373282), TAGAP (dist = 183449) | |
| 16_44 | 1.1 | MARC0073405 | rs81259195 | 44.88 | 0.031 | 34.12 | intergenic | RGS7BP (dist = 1268524) | |
| ASGA0073217 | rs81459064 | 44.83 | 0.02 | 20.38 | intergenic | RGS7BP (dist = 1315507) | |||
| LTNBA | 1_177 | 1.38 | ASGA0004992 | rs80843328 | 177.74 | 0.034 | 23 | intergenic | RNF152 (dist = 679283), MC4R (dist = 808927) |
| WEI | 12_57 | 1.96 | ASGA0092942 | rs81311789 | 57.41 | −0.024 | 100.2 | intergenic | NTN1 (dist = 349904), GLP2R (dist = 32195) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.; Kang, J.-H.; Kim, J.-M. Bayes Factor-Based Regulatory Gene Network Analysis of Genome-Wide Association Study of Economic Traits in a Purebred Swine Population. Genes 2019, 10, 293. https://doi.org/10.3390/genes10040293
Lee J, Kang J-H, Kim J-M. Bayes Factor-Based Regulatory Gene Network Analysis of Genome-Wide Association Study of Economic Traits in a Purebred Swine Population. Genes. 2019; 10(4):293. https://doi.org/10.3390/genes10040293
Chicago/Turabian StyleLee, Jungjae, Ji-Hoon Kang, and Jun-Mo Kim. 2019. "Bayes Factor-Based Regulatory Gene Network Analysis of Genome-Wide Association Study of Economic Traits in a Purebred Swine Population" Genes 10, no. 4: 293. https://doi.org/10.3390/genes10040293
APA StyleLee, J., Kang, J.-H., & Kim, J.-M. (2019). Bayes Factor-Based Regulatory Gene Network Analysis of Genome-Wide Association Study of Economic Traits in a Purebred Swine Population. Genes, 10(4), 293. https://doi.org/10.3390/genes10040293





