Asymmetric Introgressive Hybridization Among Louisiana Iris Species
Abstract
:1. Introduction
2. Louisiana Irises and asymmetric introgression in natural hybrid zones
| Adult Plant Genotypes | Seed Genotypes | |||||
|---|---|---|---|---|---|---|
| bb | bf | ff | bb | bf | ff | |
| Iris brevicaulis-like | 17 | 20 | 0 | 29 | 89 | 9 |
| Iris fulva-like | 0 | 0 | 37 | 0 | 2 | 42 |
3. Louisiana Irises and asymmetric introgression: experimental hybrid populations
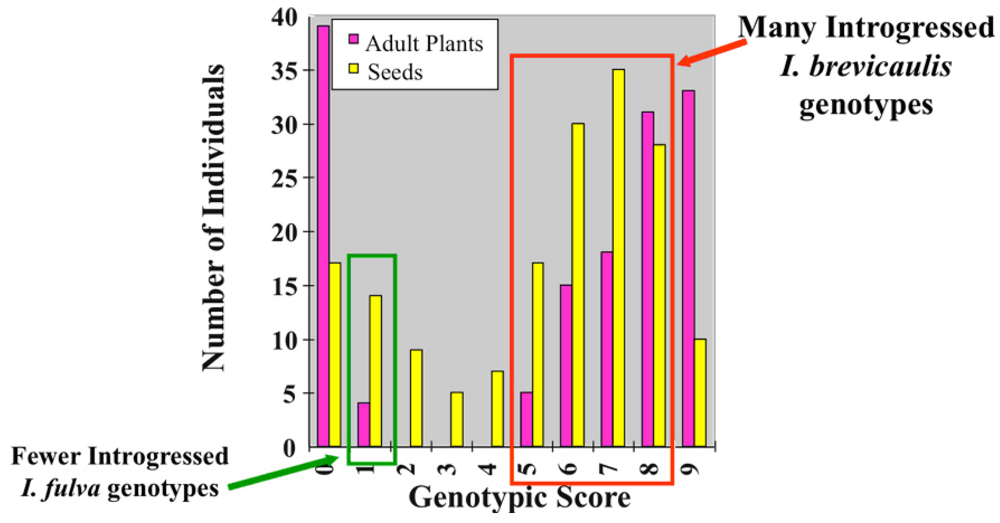
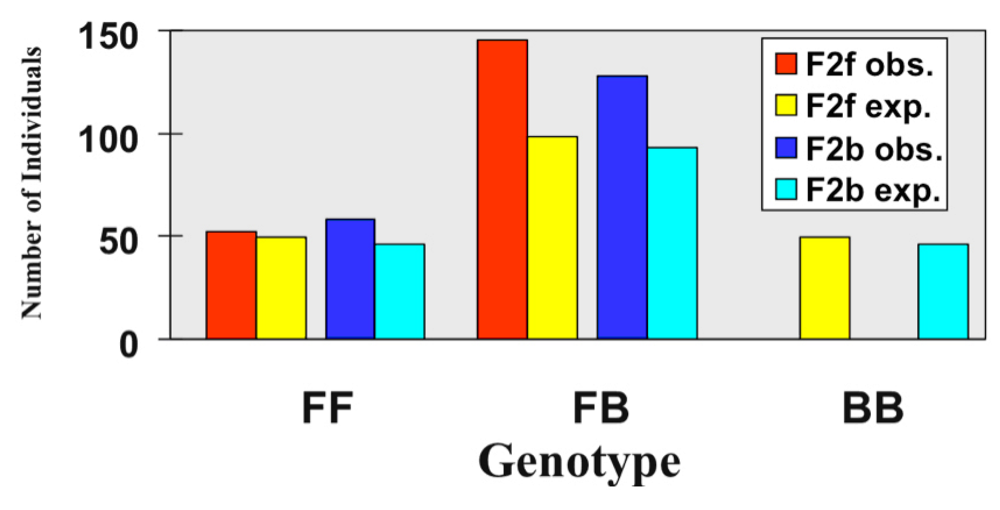
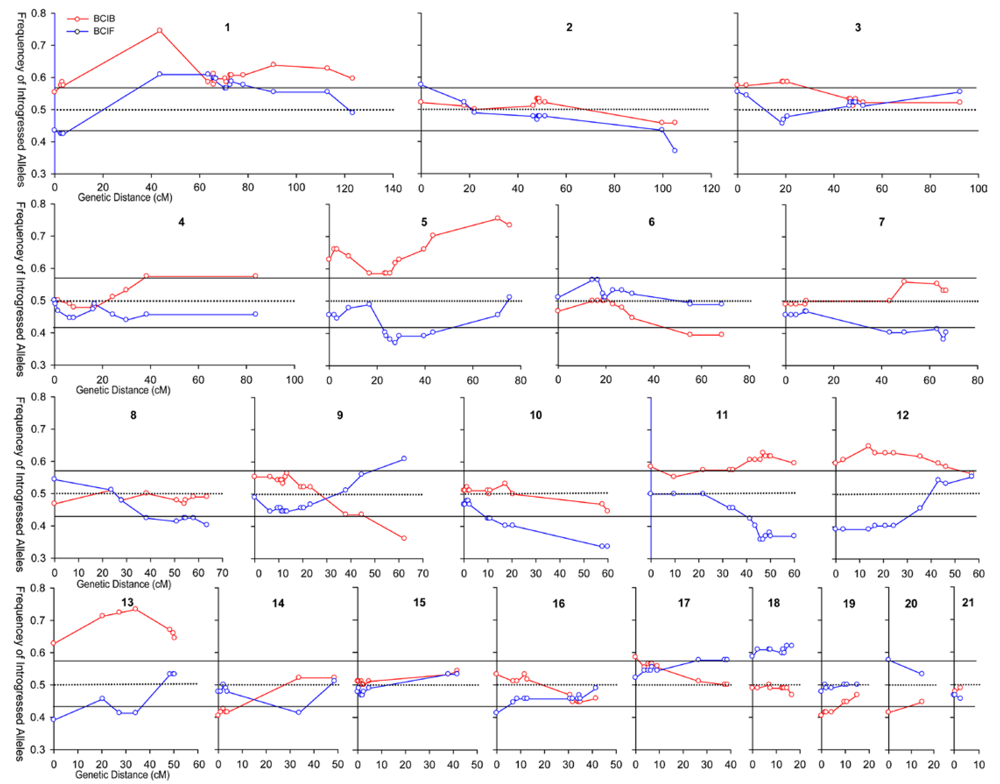
3.1. I. fulva x I. brevicaulis
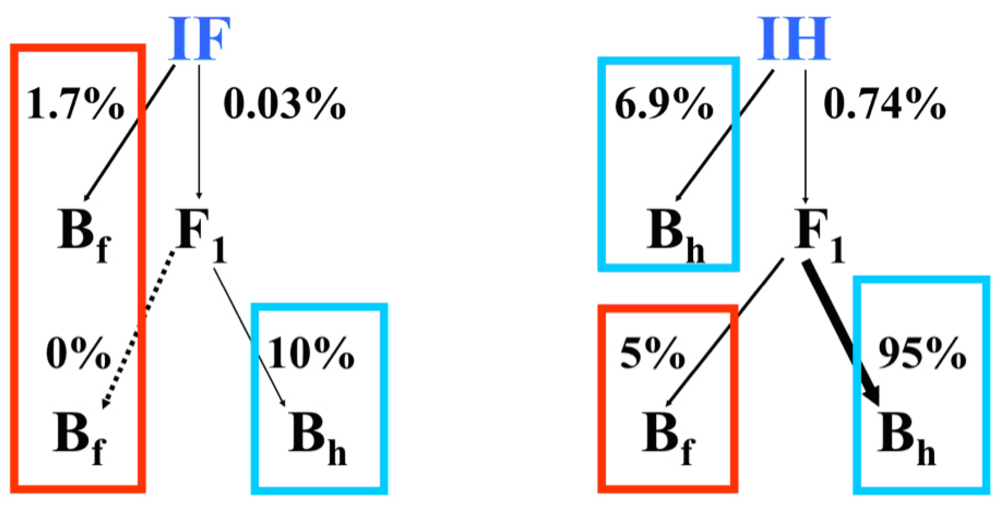
3.2. I. fulva x I. hexagona

4. The causes of asymmetric introgression in Louisiana Irises: Prezygotic reproductive isolation
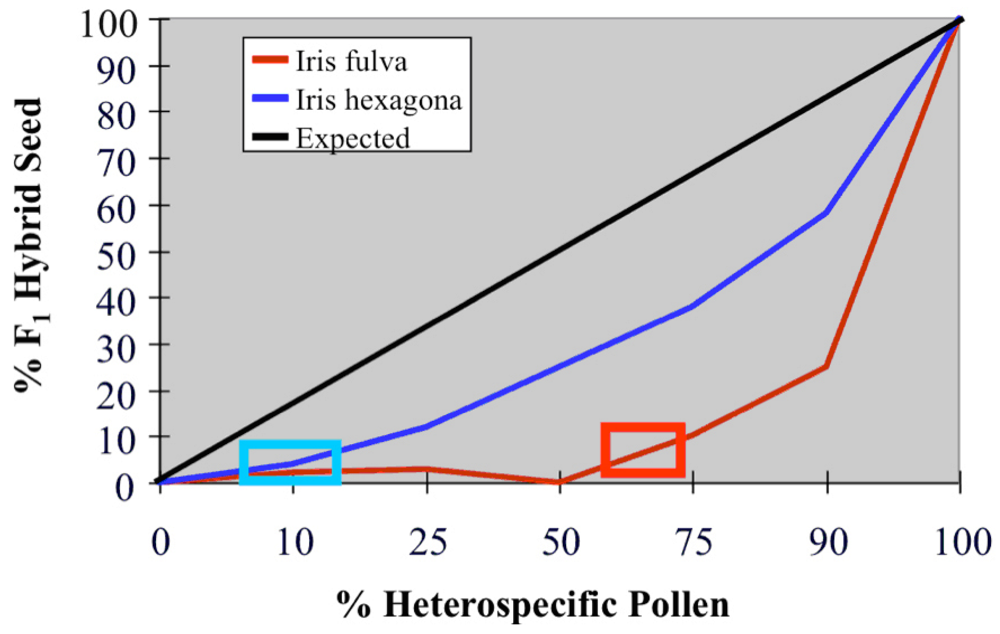
5. The causes of asymmetric introgression in Louisiana Irises: Postzygotic reproductive isolation
5.1. Selection at early life history stages
5.2. Selection at later life history stages
| Class | Alive | Dead | % Survival |
|---|---|---|---|
| I. brevicaulis | 0 | 13 | 0 |
| Introgressed I. brevicaulis | 23 | 393 | 0.055 |
| Introgressed I. fulva | 33 | 325 | 0.092 |
| I. fulva | 3 | 8 | 0.273 |
6. Asymmetric introgression and adaptive trait transfer

7. Conclusions
Acknowledgements
References and Notes
- Anderson, E.; Hubricht, L. Hybridization in Tradescantia. III. The evidence for introgressive hybridization. Am. J. Bot. 1938, 25, 396–402. [Google Scholar] [CrossRef]
- Anderson, E. Introgressive Hybridization; John Wiley and Sons, Inc.: New York, NY, USA, 1949. [Google Scholar]
- Lewontin, R.C.; Birch, L.C. Hybridization as a source of variation for adaptation to new environments. Evolution 1966, 20, 315–336. [Google Scholar] [CrossRef]
- Grant, V. Plant Speciation; Columbia University Press: New York, NY, USA, 1981. [Google Scholar]
- Arnold, M.L. Natural hybridization as an evolutionary process. Annu. Rev. Ecol. Syst. 1992, 23, 237–261. [Google Scholar] [CrossRef]
- Arnold, M.L. Natural Hybridization and Evolution; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Arnold, M.L. Evolution Through Genetic Exchange; Oxford University Press: Oxford, UK, 2006. [Google Scholar]
- Rieseberg, L.H. Hybrid origins of plant species. Annu. Rev. Ecol. Syst. 1997, 28, 359–389. [Google Scholar] [CrossRef]
- Wirtz, P. Mother species–father species: unidirectional hybridization in animals with female choice. Anim. Behav. 1999, 58, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Excoffier, L.; Foll, M.; Petit, R. Genetic consequences of range expansions. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 481–501. [Google Scholar] [CrossRef]
- Shaw, D.D.; Coates, D.J.; Arnold, M.L.; Wilkinson, P. Temporal variation in the chromosomal structure of a hybrid zone and its relationship to karyotypic repatterning. Heredity 1985, 55, 293–306. [Google Scholar] [CrossRef]
- Martin, N.H.; Willis, J.H. Ecological divergence associated with mating system causes nearly complete reproductive isolation between sympatric Mimulus species. Evolution 2007, 61, 68–82. [Google Scholar] [CrossRef] [PubMed]
- Viosca Jr., P. The irises of southeastern Louisiana – a taxonomic and ecological interpretation. Bull. Am. Iris Soc. 1935, 57, 3–56. [Google Scholar]
- Riley, H.P. A character analysis of colonies of Iris fulva, Iris hexagona var. giganticaerulea and natural hybrids. Am. J. Bot. 1938, 25, 727–738. [Google Scholar] [CrossRef]
- Randolph, L.F.; Nelson, I.S.; Plaisted, R.L. Negative evidence of introgression affecting the stability of Louisiana Iris species. Cornell Uni. Ag. Exp. Stat. Mem. 1967, 398, 1–56. [Google Scholar]
- Arnold, M.L.; Buckner, C.M.; Robinson, J.J. Pollen mediated introgression and hybrid speciation in Louisiana irises. Proc. N. Acad. Sci. USA 1991, 88, 1398–1402. [Google Scholar] [CrossRef]
- Arnold, M.L.; Robinson, J.J.; Buckner, C.M.; Bennett, B.D. Pollen dispersal and interspecific gene flow in Louisiana irises. Heredity 1992, 68, 399–404. [Google Scholar] [CrossRef]
- Cruzan, M.B.; Arnold, M.L. Ecological and genetic associations in an Iris hybrid zone. Evolution 1993, 47, 1432–1445. [Google Scholar] [CrossRef]
- Johnston, J.A.; Wesselingh, R.A.; Bouck, A.C.; Donovan, L.A.; Arnold, M.L. Intimately linked or hardly speaking? The relationship between genotypic variation and environmental gradients in a Louisiana Iris hybrid population. Mol. Ecol. 2001, 10, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Cruzan, M.B.; Arnold, M.L. Assortative mating and natural selection in an Iris hybrid zone. Evolution 1994, 48, 1946–1958. [Google Scholar] [CrossRef]
- Arnold, M.L. Iris nelsonii: origin and genetic composition of a homoploid hybrid species. Am. J. Bot. 1993, 80, 577–583. [Google Scholar] [CrossRef]
- Arnold, M.L.; Hodges, S.A. Are natural hybrids fit or unfit relative to their parents? TREE 1995, 10, 67–71. [Google Scholar] [PubMed]
- Burke, J.M.; Voss, T.J.; Arnold, M.L. Genetic interactions and natural selection in Louisiana Iris hybrids. Evolution 1998, 52, 1304–1310. [Google Scholar] [CrossRef]
- Tang, S.; Okashah, R.A.; Knapp, S.J.; Arnold, M.L.; Martin, N.H. Reproductive isolation in Louisiana irises: transmission ratio distortion. BMC Plant Biol. 2010, in press. [Google Scholar]
- Arnold, M.L.; Bennett, B.D.; Zimmer, E.A. Natural hybridization between Iris fulva and I. hexagona: pattern of ribosomal DNA variation. Evolution 1990, 44, 1512–1521. [Google Scholar] [CrossRef]
- Nason, J.D.; Ellstrand, N.C.; Arnold, M.L. Patterns of hybridization and introgression in populations of oaks, manzanitas and irises. Am. J. Bot. 1992, 79, 101–111. [Google Scholar] [CrossRef]
- Arnold, M.L.; Hamrick, J.L.; Bennett, B.D. Interspecific pollen competition and reproductive isolation in Iris. J. Hered. 1993, 84, 13–16. [Google Scholar]
- Hodges, S.A.; Burke, J.M.; Arnold, M.L. Natural formation of Iris hybrids: experimental evidence on the establishment of hybrid zones. Evolution 1996, 50, 2504–2509. [Google Scholar] [CrossRef]
- Carney, S.E.; Cruzan, M.B.; Arnold, M.L. Reproductive interactions between hybridizing irises: analyses of pollen tube growth and fertilization success. Am. J. Bot. 1994, 81, 1169–1175. [Google Scholar] [CrossRef]
- Emms, S.K.; Hodges, S.A.; Arnold, M.L. Pollen-tube competition, siring success, and consistent asymmetric hybridization in Louisiana Irises. Evolution 1996, 50, 2201–2206. [Google Scholar] [CrossRef]
- Martin, N.H.; Bouck, A. C.; Arnold, M.L. Detecting adaptive trait introgression between Iris fulva and I. brevicaulis in highly selective field conditions. Genetics 2006, 172, 2481–2489. [Google Scholar] [CrossRef] [PubMed]
- Barton, N.H.; Hewitt, G.M. Analysis of hybrid zones. Annu. Rev. Ecol. Syst. 1985, 16, 113–148. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Arnold, M.L.; Tang, S.; Knapp, S.J.; Martin, N.H. Asymmetric Introgressive Hybridization Among Louisiana Iris Species. Genes 2010, 1, 9-22. https://doi.org/10.3390/genes1010009
Arnold ML, Tang S, Knapp SJ, Martin NH. Asymmetric Introgressive Hybridization Among Louisiana Iris Species. Genes. 2010; 1(1):9-22. https://doi.org/10.3390/genes1010009
Chicago/Turabian StyleArnold, Michael L., Shunxue Tang, Steven J. Knapp, and Noland H. Martin. 2010. "Asymmetric Introgressive Hybridization Among Louisiana Iris Species" Genes 1, no. 1: 9-22. https://doi.org/10.3390/genes1010009





