Attempts at the Characterization of In-Cell Biophysical Processes Non-Invasively—Quantitative NMR Diffusometry of a Model Cellular System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample of a Model Cellular System
2.2. System
2.3. Experiments
2.4. Models for Diffusion in Cellular System
2.5. Time-Dependent Diffusion Coefficient (TDDC)
2.6. Simulations
2.7. Permeability
3. Results
3.1. Relaxation Times
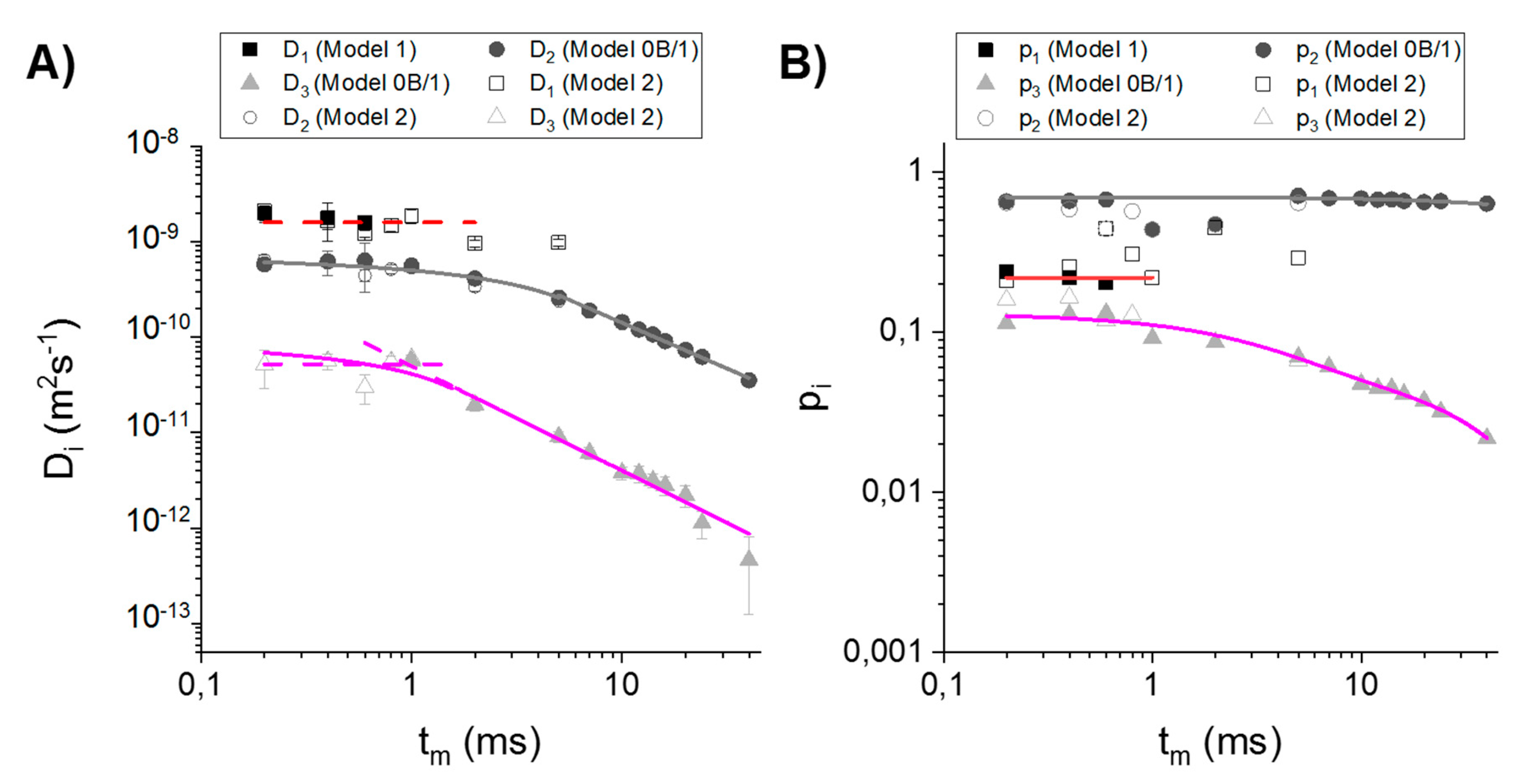
3.2. Choosing the Appropriate Diffusion Model
3.3. Relating Compartments with Cellular Structures
3.4. Simulation of the Diffusion Behavior in Cells
3.5. Extraction of Compartmental Characteristics from TDDCs
4. Discussion
4.1. Violation of the tm ≫ τ Condition
4.2. Comparison of the Diffusion Models
4.3. Characterization of the Compartments Based on TDDCs
4.4. Characterization of the Sample’s Microgeometry
4.5. Diffusive Permeabilities
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Setter, T.L.; Flannigan, B.A. Water deficit inhibits cell division and expression of transcripts involved in cell proliferation and endoreduplication in maize endosperm. J. Exp. Bot. 2001, 52, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
- Ames 3rd, A.; Nesbett, F.B. Pathophysiology of Ischemic Cell Death: II. Changes in Plasma Membrane Permeability and Cell Volume. Stroke 1982, 14, 227–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reuss, L. Water Transport Across Cell Membranes. In eLS; John Wiley Sons Ltd.: Hoboken, NJ, USA, 2012; p. 8. [Google Scholar] [CrossRef]
- Latour, L.L.; Mitra, P.P.; Kleinberg, R.L.; Sotak, C.H. Time-Dependent Diffusion Coefficient of Fluids in Porous Media as a Probe of Surface-to-Volume Ratio. J. Magn. Reson. 1993, 101, 342–346. [Google Scholar] [CrossRef]
- Åslund, I.; Nowacka, A.; Nilsson, M.; Topgaard, D. Filter-exchange PGSE NMR determination of cell membrane permeability. J. Magn. Reson. 2009, 200, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Åslund, I.; Topgaard, D. Determination of the self-diffusion coefficient of intracellular water using PGSE NMR with variable gradient pulse length. J. Magn. Reson. 2009, 201, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.; Hong, Y.; Kang, K.; Volkov, V.I.; Skirda, V.; Lee, C.J.; Lee, C. Water self-diffusion in Chlorella sp. studied by pulse field gradient NMR. Magn. Reson. Imaging 2003, 21, 1009–1017. [Google Scholar] [CrossRef]
- Schoberth, S.M.; Ba, N.; Kra, R. Pulsed High-Field Gradient in Vivo NMR Spectroscopy to Measure Diffusional Water Permeability in Corynebacterium glutamicum. Anal. Biochem. 2000, 279, 100–105. [Google Scholar] [CrossRef]
- Tanner, J.E.; Stejskal, E.O. Restricted Self-Diffusion of Protons in Colloidal Systems by the Pulsed-Gradient, Spin-Echo Method. J. Chem. Phys. 1968, 49, 1768–1777. [Google Scholar] [CrossRef]
- Latour, L.L. Time-dependent diffusion of water in a biological model system. Proc. Natl. Acad. Sci. USA 1994, 91, 1229–1233. [Google Scholar] [CrossRef] [Green Version]
- Suh, K.; Hong, Y.; Skirda, V.D.; Volkov, V.I. Water self-diffusion behavior in yeast cells studied by pulsed field gradient NMR. Biophys. Chem. 2003, 104, 121–130. [Google Scholar] [CrossRef]
- Fischer, E.; Kimmich, R. Constant time steady gradient NMR diffusometry using the secondary stimulated echo. J. Magn. Reson. 2004, 166, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Rata, D.G.; Casanova, F.; Perlo, J.; Demco, D.E.; Blümich, B. Self-diffusion measurements by a mobile single-sided NMR sensor with improved magnetic field gradient. J. Magn. Reson. 2006, 180, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Stanisz, G.J.; Szafer, A.; Wright, G.A.; Henkelman, R.M. An Analytical Model of Restricted Diffusion in Bovine Optic Nerve. Magn. Reson. Med. 1997, 37, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Blümich, B.; Haber-Pohlmeier, S.; Zia, W. Compact NMR, 1st ed.; Walter deGruyter GmbH: Berlin, Germany, 2014; ISBN 9783110266283. [Google Scholar]
- Wei, D.; Jacobs, S.; Modla, S.; Zhang, S.; Young, C.L.; Cirino, R.; Czymmek, K. High-resolution three-dimensional reconstruction of a whole yeast cell using focused-ion beam scanning electron microscopy. Biotechniques 2012, 54, 41–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitra, P.P.; Sen, M.P.N.; Schwartz, L.M.; Doussal, P. Le Diffusion Propagator as a Probe of the Structure of Porous Media. Phys. Rev. Lett. 1992, 68, 3555–3558. [Google Scholar] [CrossRef]
- Obata, T.; Kershaw, J.; Tachibana, Y.; Miyauch, T.; Abe, Y.; Shibata, S.; Kawaguchi, H.; Ikoma, Y.; Takuwa, H.; Aoki, I. Comparison of diffusion-weighted MRI and anti-Stokes Raman scattering (CARS) measurements of the inter-compartmental exchange- time of water in expression- controlled aquaporin-4 cells. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Novak, I.L.; Kraikivski, P.; Slepchenko, B.M. Diffusion in Cytoplasm: Effects of Excluded Volume Due to Internal Membranes and Cytoskeletal Structures. Biophys. J. 2009, 97, 758–767. [Google Scholar] [CrossRef] [Green Version]
- Jorgensen, P.; Edgington, N.P.; Schneider, B.L.; Rupes, I.; Tyers, M.; Futcher, B. The Size of the Nucleus Increases as Yeast Cells Grow. Mol. Biol. Cell 2007, 18, 3523–3532. [Google Scholar] [CrossRef] [Green Version]
- Oeffinger, M.; Zenklusen, D. To the pore and through the pore: A story of mRNA export kinetics. Biochim. Biophys. Acta 2012, 1819, 494–506. [Google Scholar] [CrossRef] [Green Version]
- Misteli, T. Physiological importance of RNA and protein mobility in the cell nucleus. Histochem. Cell Biol. 2008, 129, 5–11. [Google Scholar] [CrossRef] [Green Version]
- García-Pérez, A.; López-Beltrán, E.; Klüner, P.; Luque, J.; Ballesteros, P.; Cerdán, S. Molecular crowding and viscosity as determinants of translational diffusion of metabolites in subcellular organelles. Arch. Biochem. Biophys. 1999, 362, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Politz, J.C.; Browne, E.S.; Wolf, D.E.; Pederson, T. Intranuclear diffusion and hybridization state of oligonucleotides measured by fluorescence correlation spectroscopy in living cells. Proc. Natl. Acad. Sci. USA 1998, 95, 6043–6048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milani, M.; Batani, D.; Bortolotto, F.; Botto, C.; Baroni, G.; Cozzi, S.; Costato, M.; Pozzi, A.; Salsi, F.; Allott, R.; et al. Differential Two Colour X-ray Radiobiology of Membrane/Cytoplasm Yeast Cells; TMR Large-Scale Facilities Access Programme; Rutherford Appleton Laboratory: Milan, Italy, 1998. [Google Scholar]
- Schneiter, R.; Brügger, B.; Sandhoff, R.; Zellnig, G.; Leber, A.; Lampl, M.; Athenstaedt, K.; Hrastnik, C.; Eder, S.; Daum, G.; et al. Electrospray ionization tandem mass spectrometry (ESI-MS/MS) analysis of the lipid molecular species composition of yeast subcellular membranes reveals acyl chain-based sorting/remodeling of distinct molecular species en route to the plasma membrane. J. Cell Biol. 1999, 146, 741–754. [Google Scholar] [CrossRef] [PubMed]
- Karakatsanis, P.; Bayerl, T.M. Diffusion measurements in oriented phospholipid bilayers by 1NMR in a static fringe field gradient. Phys. Rev. E 1996, 54, 1785–1790. [Google Scholar] [CrossRef]
- Srinorakutara, T. Determination of yeast cell wall thickness and cell diameter using new methods. J. Ferment. Bioeng. 1998, 86, 253–260. [Google Scholar] [CrossRef]
- Kramer, E.; Frazer, N.; Baskin, T. Measurement of diffusion within the cell wall in living roots of Arabidopsis thaliana. J. Exp. Bot. 2007, 58, 3005–3015. [Google Scholar] [CrossRef] [Green Version]
- Partikian, A.; Ölveczky, B.; Swaminathan, R.; Li, Y.; Verkman, A.S. Rapid Diffusion of Green Fluorescent Protein in the Mitochondrial Matrix. J. Cell Biol. 1998, 140, 821–829. [Google Scholar] [CrossRef]
- Jiang, H.; Song, C.; Chen, C.-C.; Xu, R.; Raines, K.S.; Fahimian, B.P.; Lu, C.-H.; Lee, T.-K.; Nakashima, A.; Urano, J.; et al. Quantitative 3D imaging of whole, unstained cells by using X-ray diffraction microscopy. Proc. Natl. Acad. Sci. USA 2010, 107, 11234–11239. [Google Scholar] [CrossRef] [Green Version]
- Visser, W.; van Spronsen, E.; Nanninga, N.; Pronk, J.; Gijs Kuenen, J.; van Dijken, J. Effects of growth conditions on mitochondrial morphology in Saccharomyces cerevisiae. Antonie Leeuwenhoek 1995, 67, 243–253. [Google Scholar] [CrossRef]
- López-Beltrán, E.A.; Maté, M.J.; Cerdán, S. Dynamics and Environment of Mitochondrial Water as Detected by 1H NMR. J. Biol. Chem. 1996, 271, 10648–10653. [Google Scholar] [CrossRef] [Green Version]
- Michaillat, L.; Baars, T.; Mayer, A. Cell-free reconstitution of vacuole membrane fragmentation reveals regulation of vacuole size and number by TORC. Mol. Biol. Cell 2012, 23, 881–895. [Google Scholar]
- Chan, Y.-H.M.; Marshall, W.F. Organelle Size Scaling of the Budding Yeast Vacuole Is Tuned by Membrane Trafficking Rates. Biophys. J. 2014, 106, 1986–1996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darrah, P.R.; Tlalka, M.; Ashford, A.; Watkinson, S.C.; Fricker, M.D. The Vacuole System Is a Significant Intracellular Pathway for Longitudinal Solute Transport in Basidiomycete Fungi. Eukaryot. Cell 2006, 5, 1111–1125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Dusschoten, D.; de Jager, P.A.; Van As, H. Extracting Diffusion Constants from Echo-Time-Dependent PFG NMR Data Using Relaxation-Time Information. J. Magn. Reson. Ser. A 1995, 116, 22–28. [Google Scholar] [CrossRef]
- Zhang, Y.; Poirier-quinot, M.; Springer, C.S.; Balschi, J.A. Active Trans-Plasma Membrane Water Cycling in Yeast Is Revealed by NMR. Biophys. J. 2011, 101, 2833–2842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfeuffer, J.; Provencher, S.W.; Gruetter, R. Water diffusion in rat brain in vivo as detected at very large b values is multicompartmental. Magn. Reson. Mater. Physics, Biol. Med. 1999, 8, 98–108. [Google Scholar] [CrossRef]
- Kärger, J. NMR Self-Diffusion studies in heterogeneous systems. Adv. Colloid Interface Sci. 1985, 23, 129–148. [Google Scholar] [CrossRef]
- Williamson, N.H.; Ravin, R.; Benjamini, D.; Merkle, H.; Falgairolle, M.; Donovan, M.J.O.; Ide, D.; Cai, T.X.; Ghorashi, N.S.; Bai, R.; et al. Magnetic resonance measurements of cellular and sub-cellular membrane structures in live and fixed neural tissue. eLife 2019, 8, e51101. [Google Scholar] [CrossRef]
- Conway, E.J.; Downey, M. An Outer Metabolic Region of the Yeast Cell. Biochem. J. 1950, 47, 347–355. [Google Scholar] [CrossRef]
- Puchkov, E.O.; Wiley, J. Brownian motion of polyphosphate complexes in yeast vacuoles: Characterization by fluorescence microscopy. Yeast 2010, 27, 309–315. [Google Scholar] [CrossRef]
- Wente, S.R.; Rout, M.P. The Nuclear Pore Complex and NuclearTransport. Cold Spring Harb. Perspect. Biol. 2010, 2, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Miao, L.; Schulten, K. Probing a Structural Model of the Nuclear Pore Complex Channel through Molecular Dynamics. Biophys. J. 2010, 98, 1658–1667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paine, P.L.; Moore, L.C.; Horowitz, S.B. Nuclear envelope permeability. Nature 1975, 254, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Fraser, P.A. Diffusional and Osmotic Permeability to Water. In Physiology and Pharmacology of the Blood-Brain Barrier; Bradbury, M.W.B., Ed.; Springer-Verlag: Berlin/Heidelberg, Germany, 1992; pp. 53–64. ISBN 978-3-642-76896-5. [Google Scholar]
- Baba, M.; Osumi, M. Transmission and Scanning Electron Microscopic Examination of Intracellular Organelles in Freeze-Su bstituted Kloeckera and Saccharomyces cerevisiae Yeast Cells. J. Electron Microsc. Tech. 1987, 5, 249–261. [Google Scholar] [CrossRef]
- Cory, D.G.; Garroway, A.N. Measurement of Translational Displacement Probabilities by NMR: An Indicator of Compartmentation. Megnetic Reson. Med. 1990, 14, 435–444. [Google Scholar] [CrossRef]
- Avilova, I.A.; Vasil, S.G.; Rimareva, L.V.; Serba, E.M.; Volkova, L.D.; Volkov, V.I. Water Metabolism in Cells of Saccharomyces Cerevisiae of Races Y-3137 and Y-3327, According to Pulsed-Field Gradient NMR Data. Russ. J. Phys. Chem. A 2015, 89, 710–714. [Google Scholar] [CrossRef]




| Cell Structure | Size (μm) | Ns | Vt (μm3) | f | D0 (×10−9 m2/s) | f p02 |
|---|---|---|---|---|---|---|
| Whole cell | ~3 | 1 | 113 | -- | -- | -- |
| Intracellular space (whole cell without CW) | 2.82–2.92 | 1 | 93.9–104 | 1 | ~0.5–0.7 [5,6,9,19] | 0.65 |
| Nucleus | 1 [20,21] | 1 | 4.19 | 0.04 | ~0.01–0.1 [22], 0.23 (erythrocyte) [23], 0.04 (oligonucleotides) [24] | 0.026–0.029 |
| Cell Wall (CW) and cell membrane (combined) | 0.0792–0.180 [25] | 1 | 8.72–19.2 | 0.08–0.204 | ~0.03 (weighted mean of CW and cell membrane) | 0.054–0.133 |
| Cell membrane | 0.0092 [26] | 1 | 0.922–0.989 | 0.00989–0.00922 | 0.44 (water between lipid bilayer), <0.0006 (lipids) [27] | 0.0062–0.0063 |
| Cell Wall (CW) | 0.070–0.1708 [28] | 1 | 7.73–18.2 | 0.07–0.194 | 0.032 ± 0.014 (Carboxyfloresceine in Thale cress) [29] | 0.048–0.126 |
| Mitochondrion | 0.25 [30,31] | 2.3 [32] | 0.151 | 0.0014–0.0016 | 0.58 (liver mitochondrion) [23], ~0.01–0.1 Dbulk [33] | 0.00104 |
| Vacuole | 1 [34] | 2.7 [35] | 11.3 | 0.11–0.12 | 0.34 (Besidiomycete fungi at 20 °C) [36], 1.7 (apples) [37] | 0.0704–0.0783 |
| i | D0i(×10−9 m2s−1) | Si/Vi (μm−1) | P0i | τi (s) | Ri (μm) | Pd (μm/s) |
|---|---|---|---|---|---|---|
| 1 | 1.64 ± 0.15 | – | 0.2188 ± 0.0075 | 0.201 ± 0.039 | – | – |
| 2 | 0.692 ± 0.060 | 1.28 ± 0.22 | 0.6985 ± 0.0068 | 0.390 ± 0.056 | 2.252 ± 0.053 | 1.93 ± 0.10 |
| 3 | 0.095 ± 0.011 | 7.22 ± 0.28 | 0.070 ± 0.021 | 3.3 ± 2.0 | 0.277 ± 0.048 | 2.38 ± 0.66 |
| 0.060 ± 0.028 | 0.039 ± 0.028 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazur, W.; Krzyżak, A.T. Attempts at the Characterization of In-Cell Biophysical Processes Non-Invasively—Quantitative NMR Diffusometry of a Model Cellular System. Cells 2020, 9, 2124. https://doi.org/10.3390/cells9092124
Mazur W, Krzyżak AT. Attempts at the Characterization of In-Cell Biophysical Processes Non-Invasively—Quantitative NMR Diffusometry of a Model Cellular System. Cells. 2020; 9(9):2124. https://doi.org/10.3390/cells9092124
Chicago/Turabian StyleMazur, Weronika, and Artur T. Krzyżak. 2020. "Attempts at the Characterization of In-Cell Biophysical Processes Non-Invasively—Quantitative NMR Diffusometry of a Model Cellular System" Cells 9, no. 9: 2124. https://doi.org/10.3390/cells9092124






