Evaluating Serum Heat Shock Protein Levels as Novel Biomarkers for Atrial Fibrillation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Popultion
2.2. HSP Measurement in Serum Samples
2.3. Statistical Analysis
3. Results
3.1. Study Population
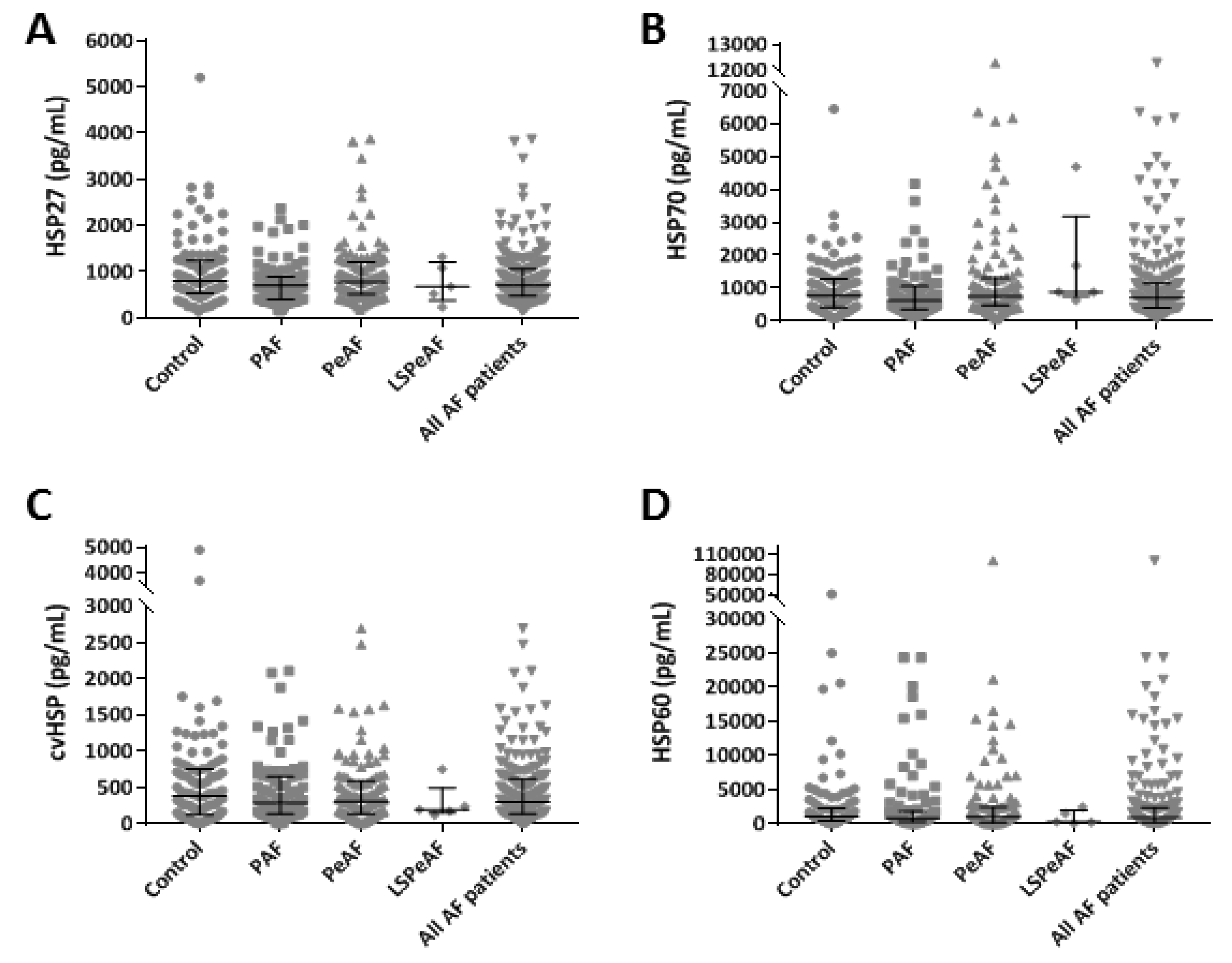
3.2. Baseline HSP Levels Related to Clinical Stage of AF
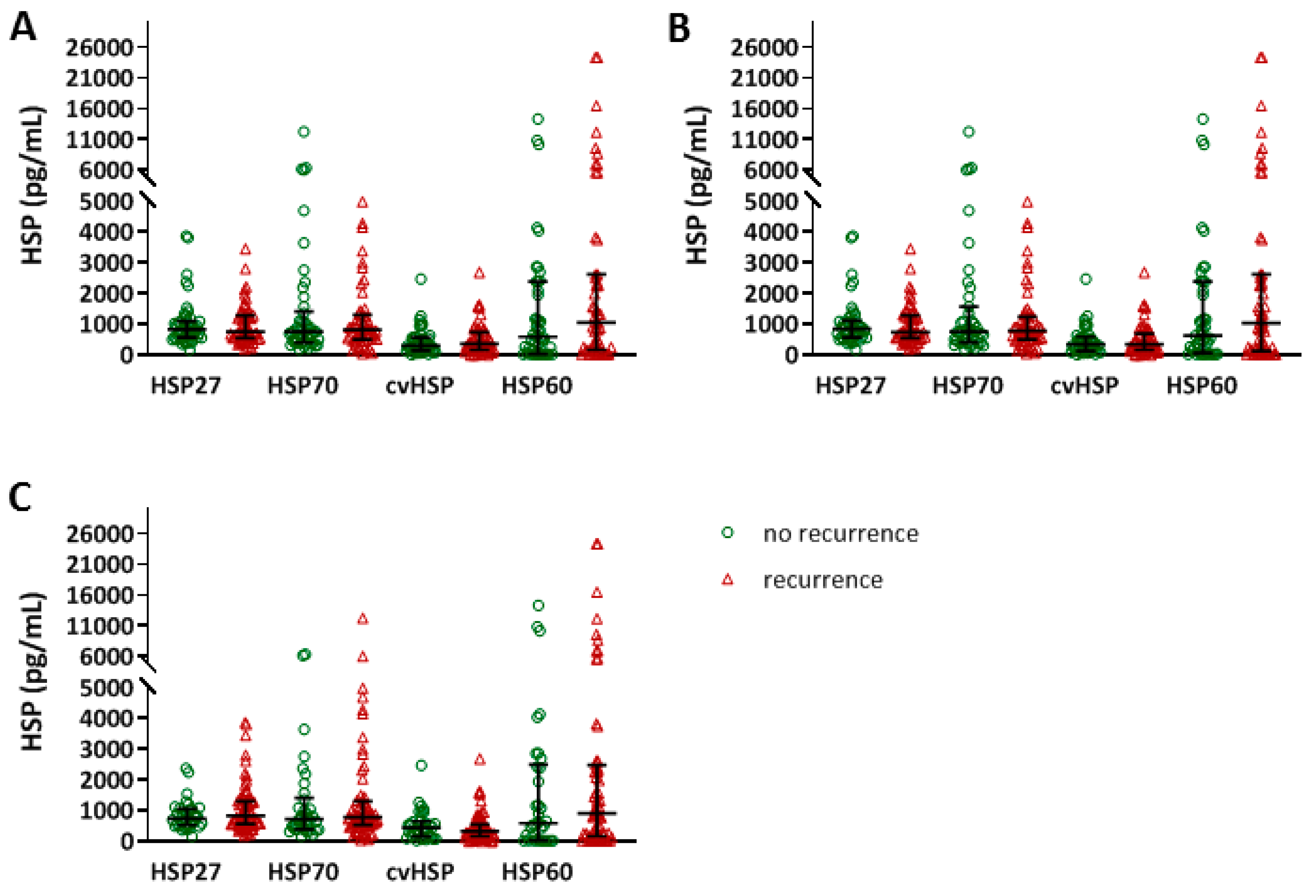
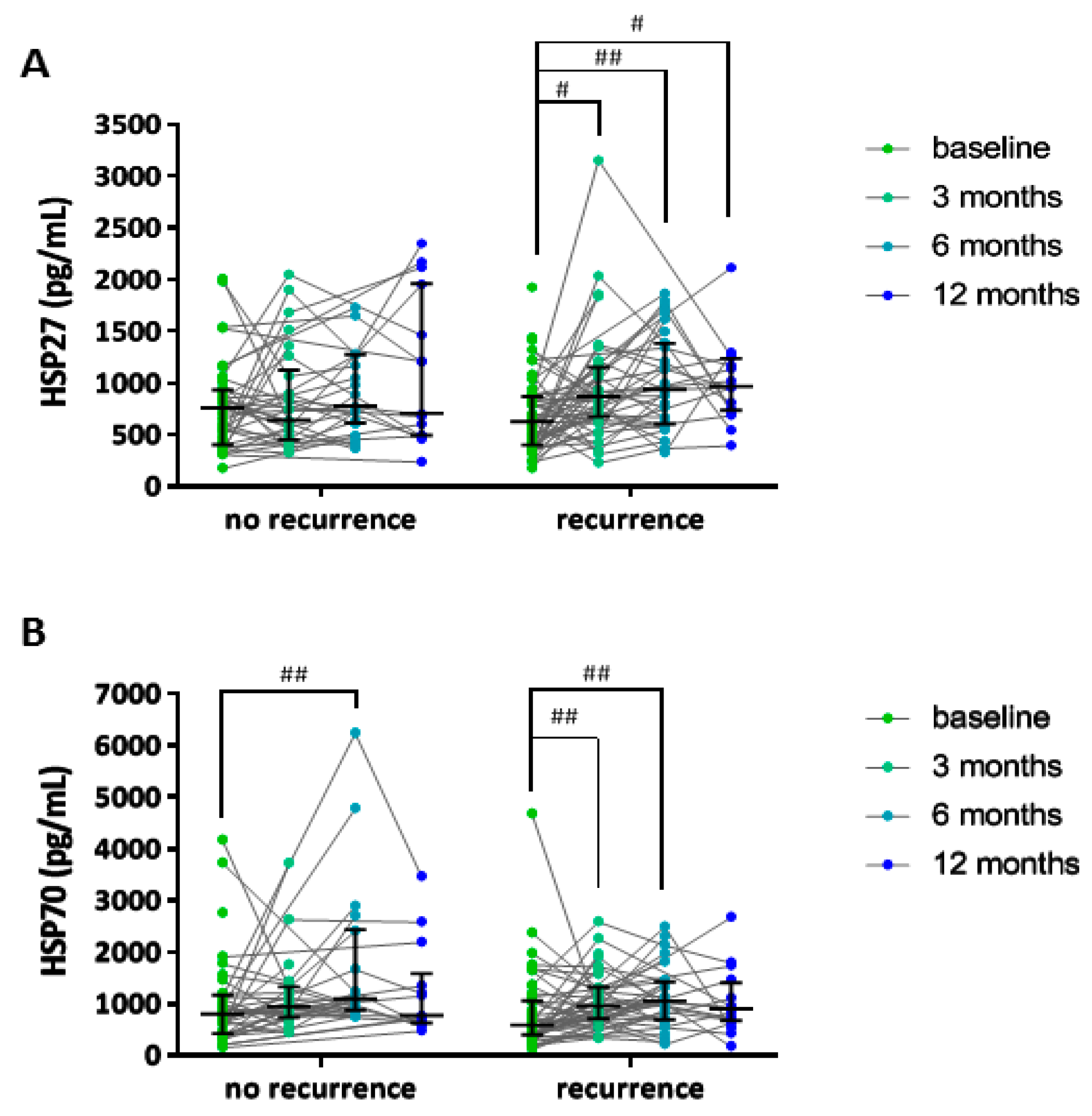
3.3. Relation between Baseline HSP Levels and AF Recurrence
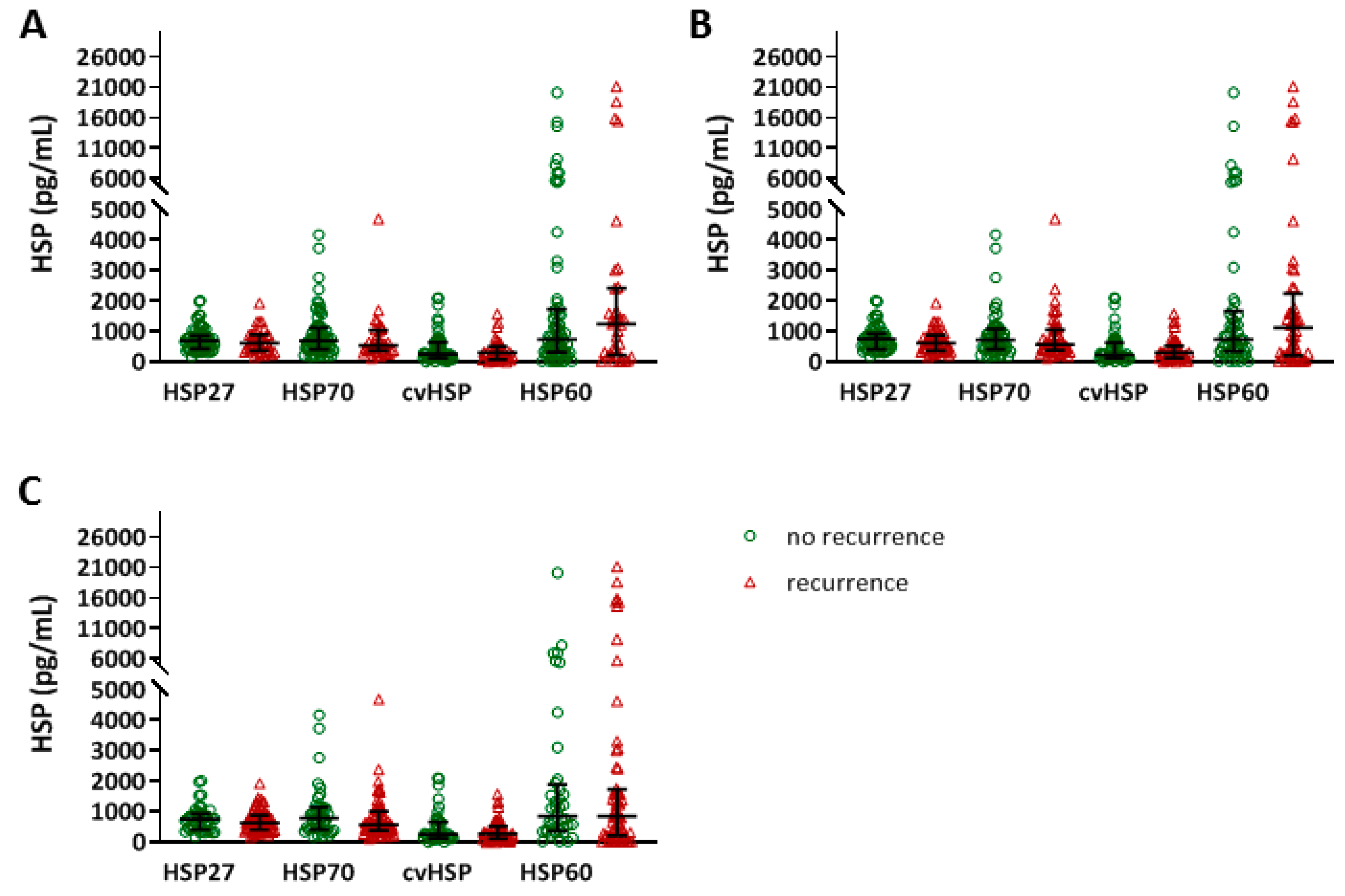
3.4. Sensitivity Analysis Non-Random Dropout
4. Discussion
4.1. Heat Shock Proteins Are Not Biomarkers to Differentiate the Stage of Atrial Fibrillation
4.2. Limitations and Future Directions: HSP in Relation to Degree of Electropathology
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; De Ferranti, S.; Després, J.-P.; Fullerton, H.J.; Howard, V.J.; et al. Executive Summary: Heart Disease and Stroke Statistics—2015 Update: A report from the American Heart Association. Circulation 2015, 131, 434–441. [Google Scholar] [CrossRef]
- Heijman, J.; Guichard, J.-B.; Dobrev, D.; Nattel, S. Translational Challenges in Atrial Fibrillation. Circ. Res. 2018, 122, 752–773. [Google Scholar] [CrossRef] [PubMed]
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Castellà, M.; Diener, H.-C.; Heidbuchel, H.; Hendriks, J.; et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace 2016, 18, 1609–1678. [Google Scholar] [CrossRef] [PubMed]
- Turakhia, M.P.; Ullal, A.J.; Hoang, D.D.; Than, C.T.; Miller, J.D.; Friday, K.J.; Perez, M.V.; Freeman, J.V.; Wang, P.J.; Heidenreich, P.A. Feasibility of Extended Ambulatory Electrocardiogram Monitoring to Identify Silent Atrial Fibrillation in High-risk Patients: The Screening Study for Undiagnosed Atrial Fibrillation (STUDY-AF). Clin. Cardiol. 2015, 38, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.G.; Deyell, M.W.; Verma, A.; Macle, L.; Champagne, J.; Leong-Sit, P.; Novak, P.; Badra-Verdu, M.; Sapp, J.; Khairy, P.; et al. Association of Atrial Fibrillation Episode Duration with Arrhythmia Recurrence Following Ablation. JAMA Netw. Open 2020, 3, e208748. [Google Scholar] [CrossRef]
- January, C.T.; Wann, L.S.; Alpert, J.S.; Calkins, H.; Cigarroa, J.E.; ClevelandJr, J.C.; Conti, J.B.; Ellinor, P.T.; Ezekowitz, M.D.; Field, M.E.; et al. 2014 AHA/ACC/HRS Guideline for the Management of Patients with Atrial Fibrillation. Circulation 2014, 130, e1–e76. [Google Scholar] [CrossRef]
- Dudink, E.A.; Weijs, B.; Tull, S.; Luermans, J.G.; Fabritz, L.; Chua, W.; Rienstra, M.; Van Gelder, I.C.; Schotten, U.; Kirchhof, P.; et al. The Biomarkers NT-proBNP and CA-125 are Elevated in Patients with Idiopathic Atrial Fibrillation. J. Atr. Fibrillation 2018, 11, 2058. [Google Scholar] [CrossRef]
- Chua, W.; Purmah, Y.; Cardoso, V.R.; Gkoutos, G.; Tull, S.P.; Neculau, G.; Thomas, M.R.; Kotecha, D.; Lip, G.Y.H.; Kirchhof, P.; et al. Data-driven discovery and validation of circulating blood-based biomarkers associated with prevalent atrial fibrillation. Eur. Hear. J. 2019, 40, 1268–1276. [Google Scholar] [CrossRef] [Green Version]
- Hui, T.H.; McClelland, R.L.; Allison, M.A.; Rodriguez, C.J.; Kronmal, R.A.; Heckbert, S.R.; Michos, E.D.; Barter, P.J.; Rye, K.-A.; Ong, K.L. The relationship of circulating fibroblast growth factor 21 levels with incident atrial fibrillation: The Multi-Ethnic Study of Atherosclerosis. Atherosclerosis 2018, 269, 86–91. [Google Scholar] [CrossRef]
- Zhu, K.; Hung, J.; Divitini, M.; Murray, K.; Lim, E.M.; John, A.S.; Walsh, J.P.; Knuiman, M.W. High-sensitivity cardiac troponin I and risk of incident atrial fibrillation hospitalisation in an Australian community-based cohort: The Busselton health study. Clin. Biochem. 2018, 58, 20–25. [Google Scholar] [CrossRef]
- Kubota, Y.; Alonso, Á.; Heckbert, S.R.; Norby, F.L.; Folsom, A.R. Homocysteine and Incident Atrial Fibrillation: The Atherosclerosis Risk in Communities Study and the Multi-Ethnic Study of Atherosclerosis. Hear. Lung Circ. 2019, 28, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Csecsei, P.; Várnai, R.; Nagy, L.; Kéki, S.; Molnár, T.; Illés, Z.; Farkas, N.; Szapáry, L. L-arginine pathway metabolites can discriminate paroxysmal from permanent atrial fibrillation in acute ischemic stroke. Ideggyogy. Szle. 2019, 72, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, A.E.; Vatasescu, R.-G.; Stanciu, M.M.; Serdarevic, N.; Dorobantu, M. The role of pro-fibrotic biomarkers in paroxysmal and persistent atrial fibrillation. Cytokine 2018, 103, 63–68. [Google Scholar] [CrossRef]
- Henning, R.H.; Brundel, B.J.J.M. Proteostasis in cardiac health and disease. Nat. Rev. Cardiol. 2017, 14, 637–653. [Google Scholar] [CrossRef]
- Brundel, B.J.J.M.; Shiroshita-Takeshita, A.; Qi, X.; Yeh, Y.-H.; Chartier, D.; Van Gelder, I.C.; Henning, R.H.; Kampinga, H.H.; Nattel, S. Induction of Heat Shock Response Protects the Heart Against Atrial Fibrillation. Circ. Res. 2006, 99, 1394–1402. [Google Scholar] [CrossRef] [Green Version]
- Wiersma, M.; Meijering, R.A.M.; Qi, X.; Zhang, D.; Liu, T.; Hoogstra-Berends, F.; Sibon, O.C.M.; Henning, R.H.; Nattel, S.; Brundel, B.J.J.M. Endoplasmic Reticulum Stress Is Associated with Autophagy and Cardiomyocyte Remodeling in Experimental and Human Atrial Fibrillation. J. Am. Hear. Assoc. 2017, 6. [Google Scholar] [CrossRef] [Green Version]
- Baler, R.; Dahl, G.; Voellmy, R. Activation of human heat shock genes is accompanied by oligomerization, modification, and rapid translocation of heat shock transcription factor HSF1. Mol. Cell. Boil. 1993, 13, 2486–2496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brundel, B.J.J.M.; Henning, R.H.; Ke, L.; Van Gelder, I.C.; Crijns, H.J.; Kampinga, H.H. Heat shock protein upregulation protects against pacing-induced myolysis in HL-1 atrial myocytes and in human atrial fibrillation. J. Mol. Cell. Cardiol. 2006, 41, 555–562. [Google Scholar] [CrossRef]
- Kotter, S.; Unger, A.; Hamdani, N.; Lang, P.; Vorgerd, M.; Nagel-Steger, L.; Linke, W.A. Human myocytes are protected from titin aggregation-induced stiffening by small heat shock proteins. J. Cell Biol. 2014, 204, 187–202. [Google Scholar] [CrossRef]
- Ghosh, J.G.; Houck, S.A.; Clark, J.I. Interactive Domains in the Molecular Chaperone Human αB Crystallin Modulate Microtubule Assembly and Disassembly. PLoS ONE 2007, 2, e498. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Li, J.; Van Marion, D.M.; Zhang, D.; Brundel, B.J.J.M. Heat shock protein inducer GGA*-59 reverses contractile and structural remodeling via restoration of the microtubule network in experimental Atrial Fibrillation. J. Mol. Cell. Cardiol. 2019, 134, 86–97. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.-F.; Yeh, H.-I.; Tsao, H.-M.; Tai, C.-T.; Lin, Y.-J.; Chang, S.-L.; Lo, L.-W.; Tuan, T.-C.; Suenari, K.; Li, C.-H.; et al. Electrophysiological Correlation and Prognostic Impact of Heat Shock Protein 27 in Atrial Fibrillation. Circ. Arrhythmia Electrophysiol. 2012, 5, 334–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lanters, E.A.; Van Marion, D.M.S.; Kik, C.; Steen, H.; Bogers, A.J.; Allessie, M.A.; Brundel, B.; De Groot, N.M.S. HALT & REVERSE: Hsf1 activators lower cardiomyocyt damage; towards a novel approach to REVERSE atrial fibrillation. J. Transl. Med. 2015, 13, 347. [Google Scholar] [CrossRef] [Green Version]
- Hijazi, Z.; Aulin, J.; Andersson, U.; Alexander, J.H.; Gersh, B.; Granger, C.B.; Hanna, M.; Horowitz, J.D.; Hylek, E.M.; Lopes, R.D.; et al. Biomarkers of inflammation and risk of cardiovascular events in anticoagulated patients with atrial fibrillation. Heart 2016, 102, 508–517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellinor, P.T.; Low, A.F.; Patton, K.K.; Shea, M.A.; Macrae, C.A. Discordant atrial natriuretic peptide and brain natriuretic peptide levels in lone atrial fibrillation. J. Am. Coll. Cardiol. 2005, 45, 82–86. [Google Scholar] [CrossRef] [Green Version]
- Patton, K.K.; Ellinor, P.T.; Heckbert, S.R.; Christenson, R.H.; Defilippi, C.; Gottdiener, J.S.; Kronmal, R.A. N-Terminal Pro-B-Type Natriuretic Peptide Is a Major Predictor of the Development of Atrial Fibrillation. Circulation 2009, 120, 1768–1774. [Google Scholar] [CrossRef] [Green Version]
- Takanori, Y.; Yamaguchi, T.; Fukui, A.; Node, K. Bipolar Voltage Mapping for the Evaluation of Atrial Substrate: Can We Overcome the Challenge of Directionality? J. Atr. Fibrillation 2019, 11, 2116. [Google Scholar] [CrossRef]
- Kornej, J.; Reinhardt, C.; Kosiuk, J.; Arya, A.; Hindricks, G.; Adams, V.; Husser, D.; Bollmann, A. Response of circulating heat shock protein 70 and anti-heat shock protein 70 antibodies to catheter ablation of atrial fibrillation. J. Transl. Med. 2013, 11, 49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandal, K.; Torsney, E.; Poloniecki, J.; Camm, A.J.; Xu, Q.; Jahangiri, M. Association of High Intracellular, but Not Serum, Heat Shock Protein 70 with Postoperative Atrial Fibrillation. Ann. Thorac. Surg. 2005, 79, 865–871. [Google Scholar] [CrossRef]
- Afzal, A.R.; Mandal, K.; Nyamweya, S.; Foteinos, G.; Poloniecki, J.; Camm, A.J.; Jahangiri, M.; Xu, Q. Association of Met439Thr Substitution in Heat Shock Protein 70 Gene with Postoperative Atrial Fibrillation and Serum HSP70 Protein Levels. Cardiology 2008, 110, 45–52. [Google Scholar] [CrossRef]
- Maan, A.; Jorgensen, N.W.; Mansour, M.; Dudley, S., Jr.; Jenny, N.S.; Defilippi, C.; Szklo, M.; Alonso, A.; Refaat, M.M.; Ruskin, J.; et al. Association between Heat Shock Protein-60 and Development of Atrial Fibrillation: Results from the Multi-Ethnic Study of Atherosclerosis (MESA). Pacing Clin. Electrophysiol. 2016, 39, 1373–1378. [Google Scholar] [CrossRef]
- Cao, H.; Xue, L.; Xu, X.; Wu, Y.; Zhu, J.; Chen, L.; Chen, D.; Chen, Y. Heat shock proteins in stabilization of spontaneously restored sinus rhythm in permanent atrial fibrillation patients after mitral valve surgery. Cell Stress Chaperones 2011, 16, 517–528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oc, M.; Ucar, H.I.; Pinar, A.; Akbulut, B.; Oc, B.; Akinci, S.B.; Akyon, Y.; Kanbak, M.; Boke, E.; Dogan, R. Heat shock protein 60 antibody. A new marker for subsequent atrial fibrillation development. Saudi Med. J. 2007, 28, 844–847. [Google Scholar] [PubMed]
- Kardys, I.; Rifai, N.; Meilhac, O.; Michel, J.-B.; Martín-Ventura, J.-L.; Buring, J.; Libby, P.; Ridker, P.M. Plasma Concentration of Heat Shock Protein 27 and Risk of Cardiovascular Disease: A Prospective, Nested Case-Control Study. Clin. Chem. 2008, 54, 139–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Groot, N.M.S.; Houben, R.P.; Smeets, J.L.; Boersma, E.; Schotten, U.; Schalij, M.J.; Crijns, H.; Allessie, M.A. Electropathological Substrate of Longstanding Persistent Atrial Fibrillation in Patients With Structural Heart Disease. Circulation 2010, 122, 1674–1682. [Google Scholar] [CrossRef] [Green Version]
- Mouws, E.M.J.P.; Van Der Does, L.J.; Kik, C.; Lanters, E.A.; Teuwen, C.P.; Knops, P.; Bogers, A.J.; De Groot, N.M.S. Impact of the arrhythmogenic potential of long lines of conduction slowing at the pulmonary vein area. Heart Rhythm. 2019, 16, 511–519. [Google Scholar] [CrossRef] [Green Version]
- Fakuade, F.E.; Steckmeister, V.; Seibertz, F.; Gronwald, J.; Kestel, S.; Menzel, J.; Pronto, J.R.D.; Taha, K.; Haghighi, F.; Kensah, G.; et al. Altered atrial cytosolic calcium handling contributes to the development of postoperative atrial fibrillation. Cardiovasc. Res. 2020, 162. [Google Scholar] [CrossRef]
| Control | PAF | PeAF | LSPeAF | All AF Patients | |
|---|---|---|---|---|---|
| n (%) | 98 (33) | 86 (29) | 108 (36.4) | 5 (1.7) | 199 (67) |
| Group, n (%) | |||||
| Control | 98 (100) | - | - | - | - |
| Electro cardioversion (ECV) | - | 12 (14) | 83 (76.9) | 3 (60) | 98 (49) |
| Pulmonary vein isolation (PVI) | - | 74 (86) | 25 (23.1) | 2 (40) | 101 (51) |
| Age (years), mean ± SD | 48.2 ± 15.3 | 61.3 ± 9.5 *** | 60.8 ± 10.6 *** | 57.5 ± 9 | 60.9 ± 10.1 *** |
| Gender, male, n (%) | 51 (52) | 64 (74.4) ** | 81 (75) ** | 4 (80) | 149 (74.9) *** |
| BMI (kg/m2), mean ± SD | 25.1 ± 3.7 | 27.2 ± 3.8 * | 28.8 ± 5.4 *** | 30.4 ± 7.4 | 28.2 ± 4.9 *** |
| Hypertension, yes, n (%) | 23 (23.5) | 43 (50) *** | 51 (47.2) ** | 3 (60) | 97 (48.7) *** |
| Diabetes mellitus, yes, n (%) | 5 (5.1) | 10 (11.6) | 15 (13.9) | 1 (20) | 26 (13.1) * |
| Dyslipidemia, yes, n (%) | 16 (16.3) | 25 (29.1) | 33 (30.6) | 3 (60) | 61 (30.7) ** |
| Thyroid disease, yes, n (%) | 2 (2) | 4 (4.7) | 8 (7.4) | 1 (20) | 13 (6.5) |
| Left ventricular function (LVF), n (%) | */### | ||||
| Normal | 61 (79.2) | 73 (84.9) | 60 (58.3) | 3 (60) | 136 (70.1) |
| Mild impairment | 10 (13) | 9 (10.5) | 29 (28.2) | 2 (40) | 40 (20.6) |
| Moderate impairment | 3 (3.9) | 3 (3.5) | 10 (9.7) | 0 (0) | 13 (6.7) |
| Severe impairment | 3 (3.9) | 1 (1.2) | 4 (3.9) | 0 (0) | 5 (2.9) |
| Missing † | 21 | 0 | 5 | 0 | 5 |
| Left atrial volume index (mL/m2),median [IQR] | 27.9 [21.2–39.7] | 38.6 [29.3–48.4] * | 47 [35.7–60.5] **/## | 43.1 [25.9–73.6] | 41.1 [31.8–54] ** |
| Drugs, yes, n (%) | |||||
| Drugs total | 52 (53.6) | 84 (97.7) *** | 104 (96.3) *** | 5 (100) | 193 (97) *** |
| ACE. ARB. AT2 antagonist | 26 (26.8) | 40 (46.5) * | 48 (44.9) * | 3 (60) | 91 (46) ** |
| Statin | 17 (17.5) | 32 (37.2) * | 37 (34.3) * | 4 (80) * | 73 (36.7) *** |
| Antiarrhythmic drugs (AAD) total ‡ | 43 (44.3) | 79 (91.9) *** | 103 (95.4) *** | 5 (100) | 187 (94) *** |
| Class I AAD | 5 (5.2) | 31 (36.0) *** | 14 (13) ### | 1 (20) | 46 (23.1) *** |
| Class II AAD | 31 (32) | 36 (41.9) | 55 (50.9) * | 1 (20) | 92 (46.2) * |
| Class III AAD | 6 (6.2) | 42 (48.8) | 55 (50.9) | 2 (40) | 99 (49.7) *** |
| Class IV AAD | 3 (3.1) | 4 (4.7) | 7 (6.5) | 5 (100) | 11 (5.5) |
| Digoxin | 0 (0) | 6 (7) * | 18 (16.7) *** | 1 (20) | 25 (12.6) *** |
| Patients | Samples | HSP27 | HSP70 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AF | AF-Free | AF | AF-Free | HR | 95%CI LL | 95%CI UL | p-Value | HR | 95%CI LL | 95%CI UL | p-Value | |
| All events | 59 | 41 | 114 | 99 | 1.32 | 1.71 | 1.54 | <0.001 | model does not converge | |||
| Complete until 3 m | 21 | 54 | 42 | 129 | 1.31 | 0.84 | 2.89 | 0.258 | 0.97 | 0.56 | 1.5 | 0.969 |
| Complete until 6 m | 29 | 36 | 63 | 99 | 2.03 | 1.17 | 3.7 | <0.001 | 1.03 | 0.54 | 1.97 | 0.926 |
| Complete until 12 m | 31 | 13 | 70 | 34 | model does not converge | model does not converge | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marion, D.M.S.v.; Lanters, E.A.H.; Ramos, K.S.; Li, J.; Wiersma, M.; Baks-te Bulte, L.; J. Q. M. Muskens, A.; Boersma, E.; de Groot, N.M.S.; Brundel, B.J.J.M. Evaluating Serum Heat Shock Protein Levels as Novel Biomarkers for Atrial Fibrillation. Cells 2020, 9, 2105. https://doi.org/10.3390/cells9092105
Marion DMSv, Lanters EAH, Ramos KS, Li J, Wiersma M, Baks-te Bulte L, J. Q. M. Muskens A, Boersma E, de Groot NMS, Brundel BJJM. Evaluating Serum Heat Shock Protein Levels as Novel Biomarkers for Atrial Fibrillation. Cells. 2020; 9(9):2105. https://doi.org/10.3390/cells9092105
Chicago/Turabian StyleMarion, Denise M. S. van, Eva A. H. Lanters, Kennedy S. Ramos, Jin Li, Marit Wiersma, Luciënne Baks-te Bulte, Agnes J. Q. M. Muskens, Eric Boersma, Natasja M. S. de Groot, and Bianca J. J. M. Brundel. 2020. "Evaluating Serum Heat Shock Protein Levels as Novel Biomarkers for Atrial Fibrillation" Cells 9, no. 9: 2105. https://doi.org/10.3390/cells9092105
APA StyleMarion, D. M. S. v., Lanters, E. A. H., Ramos, K. S., Li, J., Wiersma, M., Baks-te Bulte, L., J. Q. M. Muskens, A., Boersma, E., de Groot, N. M. S., & Brundel, B. J. J. M. (2020). Evaluating Serum Heat Shock Protein Levels as Novel Biomarkers for Atrial Fibrillation. Cells, 9(9), 2105. https://doi.org/10.3390/cells9092105







