Transglutaminase 3: The Involvement in Epithelial Differentiation and Cancer
Abstract
:1. Introduction
2. Contribution of TGM3 and Other TGMs to Skin Morphogenesis
2.1. Epidermal Cornification
2.2. Hair Keratinization
2.3. TGM3 Affinity to Substrates and Suggested Functional Compensation by Other TGMs
2.4. Mouse Models for Studying the Role of TGM3 in Skin Morphogenesis
3. Contribution of TGM3 to Pathology
3.1. Structural Role of TGM3 in Disease: Uncombable Hair Syndrome
3.2. Functional Role of TGM3 in Disease: TGM3 as a Tumor Marker
3.3. The Role of TGM3 in Development of Dermatitis Herpetiformis, Atopic Dermatitis and Oral Lichen Planus
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TGM | Transglutaminase |
| KIFs | Keratin intermidiate filaments |
| ORS | Outer root sheath |
| IRS | Inner root sheath |
| CCE | Cornified cell envelope |
| UHS | Uncombable hair syndrome |
| EMT | Epithelial-to-mesenchymal transition |
| BCC | Basal cell carcinoma |
| HCC | Hepatocellular carcinoma |
| DH | Dermatitis herpetiformis |
| AD | Atopic dermatitis |
References
- Greenberg, C.S.; Birckbichler, P.J.; Rice, R.H. Transglutaminases: Multifunctional cross-linking enzymes that stabilize tissues. FASEB J. 1991, 5, 3071–3077. [Google Scholar] [CrossRef]
- Lorand, L.; Graham, R.M. Transglutaminases: Crosslinking enzymes with pleiotropic functions. Nat. Rev. Mol. Cell Biol. 2003, 4, 140–156. [Google Scholar] [CrossRef]
- Lorand, L.; Conrad, S.M. Transglutaminases. Mol. Cell. Biochem. 1984, 58, 9–35. [Google Scholar] [CrossRef] [PubMed]
- Nemes, Z.; Marekov, L.N.; Fésüs, L.; Steinert, P.M. A novel function for transglutaminase 1: Attachment of long-chain omega-hydroxyceramides to involucrin by ester bond formation. Proc. Natl. Acad. Sci. USA 1999, 96, 8402–8407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasegawa, G.; Suwa, M.; Ichikawa, Y.; Ohtsuka, T.; Kumagai, S.; Kikuchi, M.; Sato, Y.; Saito, Y. A novel function of tissue-type transglutaminase: Protein disulphide isomerase. Biochem. J. 2003, 373, 793–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iismaa, S.E.; Mearns, B.M.; Lorand, L.; Graham, R.M. Transglutaminases and disease: Lessons from genetically engineered mouse models and inherited disorders. Physiol. Rev. 2009, 89, 991–1023. [Google Scholar] [CrossRef] [Green Version]
- Eckert, R.L.; Kaartinen, M.T.; Nurminskaya, M.; Belkin, A.M.; Colak, G.; Johnson, G.V.; Mehta, K. Transglutaminase regulation of cell function. Physiol. Rev. 2014, 94, 383–417. [Google Scholar] [CrossRef] [Green Version]
- Klöck, C.; Khosla, C. Regulation of the activities of the mammalian transglutaminase family of enzymes. Protein Sci. 2012, 21, 1781–1791. [Google Scholar] [CrossRef] [Green Version]
- Rezza, A.; Wang, Z.; Sennett, R.; Qiao, W.; Wang, D.; Heitman, N.; Mok, K.W.; Clavel, C.; Yi, R.; Zandstra, P.; et al. Signaling Networks among Stem Cell Precursors, Transit-Amplifying Progenitors, and their Niche in Developing Hair Follicles. Cell Rep. 2016, 14, 3001–3018. [Google Scholar] [CrossRef] [Green Version]
- MGI:95402. Available online: http://www.informatics.jax.org/marker/MGI:95402 (accessed on 5 May 2020).
- Dubbink, H.J.; de Waal, L.; van Haperen, R.; Verkaik, N.S.; Trapman, J.; Romijn, J.C. The human prostate-specific transglutaminase gene (TGM4): Genomic organization, tissue-specific expression, and promoter characterization. Genomics 1998, 51, 434–444. [Google Scholar] [CrossRef]
- Grenard, P.; Bates, M.K.; Aeschlimann, D. Evolution of transglutaminase genes: Identification of a transglutaminase gene cluster on human chromosome 15q15. Structure of the gene encoding transglutaminase X and a novel gene family member, transglutaminase Z. J. Biol. Chem. 2001, 276, 33066–33078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ichinose, A.; Bottenus, R.E.; Davie, E.W. Structure of transglutaminases. J. Biol. Chem. 1990, 265, 13411–13414. [Google Scholar]
- Orosz, Z.Z.; Bárdos, H.; Shemirani, A.H.; Beke Debreceni, I.; Lassila, R.; Riikonen, A.S.; Kremer Hovinga, J.A.; Seiler, T.G.; van Dorland, H.A.; Schroeder, V.; et al. Cellular Factor XIII, a Transglutaminase in Human Corneal Keratocytes. Int. J. Mol. Sci. 2019, 20, 5963. [Google Scholar] [CrossRef] [Green Version]
- Satchwell, T.J.; Shoemark, D.K.; Sessions, R.B.; Toye, A.M. Protein 4.2: A complex linker. Blood Cells Mol. Dis. 2009, 42, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Matsuki, M.; Yamashita, F.; Ishida-Yamamoto, A.; Yamada, K.; Kinoshita, C.; Fushiki, S.; Ueda, E.; Morishima, Y.; Tabata, K.; Yasuno, H.; et al. Defective stratum corneum and early neonatal death in mice lacking the gene for transglutaminase 1 (keratinocyte transglutaminase). Proc. Natl. Acad. Sci. USA 1998, 95, 1044–1049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nanda, N.; Iismaa, S.E.; Owens, W.A.; Husain, A.; Mackay, F.; Graham, R.M. Targeted inactivation of Gh/tissue transglutaminase II. J. Biol. Chem. 2001, 276, 20673–20678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.J.; Lee, K.B.; Son, Y.H.; Shin, J.; Lee, J.H.; Kim, H.J.; Hong, A.Y.; Bae, H.W.; Kwon, M.A.; Lee, W.J.; et al. Transglutaminase 2 mediates UV-induced skin inflammation by enhancing inflammatory cytokine production. Cell Death. Dis. 2017, 8, e3148. [Google Scholar] [CrossRef] [PubMed]
- Haroon, Z.A.; Hettasch, J.M.; Lai, T.S.; Dewhirst, M.W.; Greenbergб, C.S. Tissue transglutaminase is expressed, active, and directly involved in rat dermal wound healing and angiogenesis. FASEB J. 1999, 13, 1787–1795. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Griffin, M. TG2, a novel extracellular protein with multiple functions. Amino Acids 2012, 42, 939–949. [Google Scholar] [CrossRef]
- Szondy, Z.; Korponay-Szabó, I.; Király, R.; Sarang, Z.; Tsay, G.J. Transglutaminase 2 in human diseases. Biomedicine (Taipei) 2017, 7, 15. [Google Scholar] [CrossRef] [Green Version]
- Beninati, S.; Piacentini, M.; Bergamini, C.M. Transglutaminase 2, a double face enzyme. Amino Acids 2017, 49, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Candi, E.; Oddi, S.; Paradisi, A.; Terrinoni, A.; Ranalli, M.; Teofoli, P.; Citro, G.; Scarpato, S.; Puddu, P.; Melino, G. Expression of transglutaminase 5 in normal and pathologic human epidermis. J. Investig. Dermatol. 2002, 119, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.T.; Tang, B.S.; Lan, W.; Song, N.N.; Huang, Y.; Zhang, L.; Guan, W.J.; Shi, Y.T.; Shen, L.; Jiang, H.; et al. Distribution of transglutaminase 6 in the central nervous system of adult mice. Anat. Rec. (Hoboken) 2013, 296, 1576–1587. [Google Scholar] [CrossRef] [PubMed]
- Thomas, H.; Beck, K.; Adamczyk, M.; Aeschlimann, P.; Langley, M.; Oita, R.C.; Thiebach, L.; Hils, M.; Aeschlimann, D. Transglutaminase 6: A protein associated with central nervous system development and motor function. Amino Acids 2013, 44, 161–177. [Google Scholar] [CrossRef] [Green Version]
- Steinert, P.M.; Parry, D.A.; Marekov, L.N. Trichohyalin mechanically strengthens the hair follicle: Multiple cross-bridging roles in the inner root sheath. J. Biol. Chem. 2003, 278, 41409–41419. [Google Scholar] [CrossRef] [PubMed]
- Thibaut, S.; Candi, E.; Pietroni, V.; Melino, G.; Schmidt, R.; Bernard, B.A. Transglutaminase 5 expression in human hair follicle. J. Investig. Dermatol. 2005, 125, 581–585. [Google Scholar] [CrossRef]
- Candi, E.; Schmidt, R.; Melino, G. The cornified envelope: A model of cell death in the skin. Nat. Rev. Mol. Cell Biol. 2005, 6, 328–340. [Google Scholar] [CrossRef]
- Ogawa, H.; Goldsmith, L.A. Human epidermal transglutaminase. Preparation and properties. J. Biol. Chem. 1976, 251, 7281–7288. [Google Scholar]
- Buxman, M.M.; Wuepper, K.D. Isolation, purification and characterization of bovine epidermal transglutaminase. Biochim. Biophys. Acta 1976, 452, 356–369. [Google Scholar] [CrossRef]
- Hitomi, K.; Horio, Y.; Ikura, K.; Yamanishi, K.; Maki, M. Analysis of epidermal-type transglutaminase (TGase 3) expression in mouse tissues and cell lines. Int. J. Biochem. Cell Biol. 2001, 33, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhi, H.Y.; Ding, F.; Luo, A.P.; Liu, Z.H. Transglutaminase 3 expression in C57BL/6J mouse embryo epidermis and the correlation with its differentiation. Cell Res. 2005, 15, 105–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.Y.; Grant, P.; Lee, J.H.; Pant, H.C.; Steinert, P.M. Differential expression of multiple transglutaminases in human brain. Increased expression and cross-linking by transglutaminases 1 and 2 in Alzheimer’s disease. Biol. Chem. 1999, 274, 30715–30721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, X.; Wang, R.; Jiao, J.; Li, S.; Yu, J.; Yin, Z.; Zhou, L.; Gong, Z. Transglutaminase 3 contributes to malignant transformation of oral leukoplakia to cancer. Int. J. Biochem. Cell Biol. 2018, 104, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Cao, W.; Wang, X.; Zhang, J.; Lv, Z.; Qin, X.; Wu, Y.; Chen, W. TGM3, a candidate tumor suppressor gene, contributes to human head and neck cancer. Mol. Cancer 2013, 12, 151. [Google Scholar] [CrossRef] [Green Version]
- Feng, Y.; Ji, D.; Huang, Y.; Ji, B.; Zhang, Y.; Li, J.; Peng, W.; Zhang, C.; Zhang, D.; Sun, Y. TGM3 functions as a tumor suppressor by repressing epithelial-to-mesenchymal transition and the PI3K/AKT signaling pathway in colorectal cancer. Oncol. Rep. 2020, 43, 864–876. [Google Scholar] [CrossRef] [Green Version]
- Marukian, N.V.; Hu, R.H.; Craiglow, B.G.; Milstone, L.M.; Zhou, J.; Theos, A.; Kaymakcalan, H.; Akkaya, D.A.; Uitto, J.J.; Vahidnezhad, H.; et al. Expanding the Genotypic Spectrum of Bathing Suit Ichthyosis. JAMA Dermatol. 2017, 153, 537–543. [Google Scholar] [CrossRef]
- Raghunath, M.; Hennies, H.C.; Ahvazi, B.; Vogel, M.; Reis, A.; Steinert, P.M.; Traupe, H. Self-healing collodion baby: A dynamic phenotype explained by a particular transglutaminase-1 mutation. J. Investig. Dermatol. 2003, 120, 224–228. [Google Scholar] [CrossRef] [Green Version]
- Kuramoto, N.; Takizawa, T.; Takizawa, T.; Matsuki, M.; Morioka, H.; Robinson, J.M.; Yamanishi, K. Development of ichthyosiform skin compensates for defective permeability barrier function in mice lacking transglutaminase 1. J. Clin. Investig. 2002, 109, 243–250. [Google Scholar] [CrossRef]
- Laiho, E.; Ignatius, J.; Mikkola, H.; Yee, V.C.; Teller, D.C.; Niemi, K.M.; Saarialho-Kere, U.; Kere, J.; Palotie, A. Transglutaminase 1 mutations in autosomal recessive congenital ichthyosis: Private and recurrent mutations in an isolated population. Am. J. Hum. Genet. 1997, 61, 529–538. [Google Scholar] [CrossRef] [Green Version]
- Inada, R.; Matsuki, M.; Yamada, K.; Morishima, Y.; Shen, S.C.; Kuramoto, N.; Yasuno, H.; Takahashi, K.; Miyachi, Y.; Yamanishi, K. Facilitated wound healing by activation of the Transglutaminase 1 gene. Am. J. Pathol. 2000, 157, 1875–1882. [Google Scholar] [CrossRef] [Green Version]
- Basmanav, F.B.Ü.; Cau, L.; Tafazzoli, A.; Méchin, M.C.; Wolf, S.; Romano, M.T.; Valentin, F.; Wiegmann, H.; Huchenq, A.; Kandil, R.; et al. Mutations in three genes encoding proteins involved in hair shaft formation cause uncombable hair syndrome. Am. J. Hum. Genet. 2016, 99, 1292–1304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sárdy, M.; Kárpáti, S.; Merkl, B.; Paulsson, M.; Smyth, N. Epidermal transglutaminase (TGase 3) is the autoantigen of dermatitis herpetiformis. J. Exp. Med. 2002, 195, 747–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hitomi, K.; Presland, R.B.; Nakayama, T.; Fleckman, P.; Dale, B.A.; Maki, M. Analysis of epidermal-type transglutaminase (transglutaminase 3) in human stratified epithelia and cultured keratinocytes using monoclonal antibodies. J. Dermatol. Sci. 2003, 32, 95–103. [Google Scholar] [CrossRef]
- Cassidy, A.J.; van Steensel, M.A.; Steijlen, P.M.; van Geel, M.; van der Velden, J.; Morley, S.M.; Terrinoni, A.; Melino, G.; Candi, E.; McLean, W.H. A homozygous missense mutation in TGM5 abolishes epidermal transglutaminase 5 activity and causes acral peeling skin syndrome. Am. J. Hum. Genet. 2005, 77, 909–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eckert, R.L.; Yaffe, M.B.; Crish, J.F.; Murthy, S.; Rorke, E.A.; Welter, J.F. Involucrin–structure and role in envelope assembly. J. Investig. Dermatol. 1993, 100, 613–617. [Google Scholar] [CrossRef] [Green Version]
- Nemes, Z.; Steinert, P.M. Bricks and mortar of the epidermal barrier. Exp. Mol. Med. 1999, 31, 5–19. [Google Scholar] [CrossRef]
- Schmuth, M.; Gruber, R.; Elias, P.M.; Williams, M.L. Ichthyosis update: Towards a function-driven model of pathogenesis of the disorders of cornification and the role of corneocyte proteins in these disorders. Adv. Dermatol. 2007, 23, 231–256. [Google Scholar] [CrossRef] [Green Version]
- Candi, E.; Knight, R.A.; Panatta, E.; Smirnov, A.; Melino, G. Cornification of the skin: A non-apoptotic cell death mechanism. In eLS; John Wiley & Sons, Ltd.: Chichester, UK, 2016. [Google Scholar] [CrossRef]
- Candi, E.; Tarcsa, E.; Digiovanna, J.J.; Compton, J.G.; Elias, P.M.; Marekov, L.N.; Steinert, P.M. A highly conserved lysine residue on the head domain of type II keratins is essential for the attachment of keratin intermediate filaments to the cornified cell envelope through isopeptide crosslinking by transglutaminases. Proc. Natl. Acad. Sci. USA 1998, 95, 2067–2072. [Google Scholar] [CrossRef] [Green Version]
- Simon, M.; Haftek, M.; Sebbag, M.; Montézin, M.; Girbal-Neuhauser, E.; Schmitt, D.; Serre, G. Evidence that filaggrin is a component of cornified cell envelopes in human plantar epidermis. Biochem. J. 1996, 317, 173–177. [Google Scholar] [CrossRef] [Green Version]
- Candi, E.; Tarcsa, E.; Idler, W.W.; Kartasova, T.; Marekov, L.N.; Steinert, P.M. Transglutaminase cross-linking properties of the small proline-rich 1 family of cornified cell envelope proteins. Integration with loricrin. J. Biol. Chem. 1999, 274, 7226–7237. [Google Scholar] [CrossRef] [Green Version]
- Eckert, R.L.; Sturniolo, M.T.; Broome, A.M.; Ruse, M.; Rorke, E.A. Transglutaminases in epidermis. Prog. Exp. Tumor Res. 2005, 38, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.G.; Gorman, J.J.; Park, S.C.; Chung, S.I.; Steinert, P.M. The deduced sequence of the novel protransglutaminase E (TGase3) of human and mouse. J. Biol. Chem. 1993, 268, 12682–12690. [Google Scholar] [PubMed]
- Cheng, T.; Hitomi, K.; van Vlijmen-Willems, I.M.; de Jongh, G.J.; Yamamoto, K.; Nishi, K.; Watts, C.; Reinheckel, T.; Schalkwijk, J.; Zeeuwen, P.L. Cystatin M/E is a high affinity inhibitor of cathepsin V and cathepsin L by a reactive site that is distinct from the legumain-binding site. A novel clue for the role of cystatin M/E in epidermal cornification. J. Biol. Chem. 2006, 281, 15893–15899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.C.; Lewis, M.S.; Gorman, J.J.; Park, S.C.; Girard, J.E.; Folk, J.E.; Chung, S.I. Protransglutaminase E from guinea pig skin. Isolation and partial characterization. J. Biol. Chem. 1990, 265, 21971–21978. [Google Scholar]
- Hitomi, K.; Kanehiro, S.; Ikura, K.; Maki, M. Characterization of recombinant mouse epidermal-type transglutaminase (TGase 3): Regulation of its activity by proteolysis and guanine nucleotides. J. Biochem. 1999, 125, 1048–1054. [Google Scholar] [CrossRef]
- Teshima, H.; Kato, M.; Tatsukawa, H.; Hitomi, K. Analysis of the expression of transglutaminases in the reconstructed human epidermis using a three-dimensional cell culture. Anal. Biochem. 2020, 603, 113606. [Google Scholar] [CrossRef]
- Kalinin, A.E.; Kajava, A.V.; Steinert, P.M. Epithelial barrier function: Assembly and structural features of the cornified cell envelope. Bioessays 2002, 24, 789–800. [Google Scholar] [CrossRef]
- Ahvazi, B.; Kim, H.C.; Kee, S.H.; Nemes, Z.; Steinert, P.M. Three-dimensional structure of the human transglutaminase 3 enzyme: Binding of calcium ions changes structure for activation. EMBO J. 2002, 21, 2055–2067. [Google Scholar] [CrossRef]
- Akiyama, M.; Smith, L.T.; Yoneda, K.; Holbrook, K.A.; Shimizu, H. Transglutaminase and major cornified cell envelope precursor proteins, loricrin, small proline-rich proteins 1 and 2, and involucrin are coordinately expressed in the sites defined to form hair canal in developing human hair follicle. Exp. Dermatol. 1999, 8, 313–314. [Google Scholar]
- Commo, S.; Bernard, B.A. Immunohistochemical analysis of tissue remodelling during the anagen-catagen transition of the human hair follicle. Br. J. Dermatol. 1997, 137, 31–38. [Google Scholar] [CrossRef]
- Hashimoto, K. The structure of human hair. Clin. Dermatol. 1988, 6, 7–21. [Google Scholar] [CrossRef]
- Harland, D.P. Introduction to Hair Development. Adv. Exp. Med. Biol. 2018, 1054, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Oshima, H.; Rochat, A.; Kedzia, C.; Kobayashi, K.; Barrandon, Y. Morphogenesis and renewal of hair follicles from adult multipotent stem cells. Cell 2001, 104, 233–245. [Google Scholar] [CrossRef] [Green Version]
- Hardy, M.H. The secret life of the hair follicle. Trends Genet. 1992, 8, 55–61. [Google Scholar] [CrossRef]
- Fuchs, E. Scratching the surface of skin development. Nature 2007, 445, 834–842. [Google Scholar] [CrossRef] [Green Version]
- Schneider, M.R.; Schmidt-Ullrich, R.; Paus, R. The hair follicle as a dynamic miniorgan. Curr. Biol. 2009, 19, 132–142. [Google Scholar] [CrossRef] [Green Version]
- Joost, S.; Annusver, K.; Jacob, T.; Sun, X.; Dalessandri, T.; Sivan, U.; Sequeira, I.; Sandberg, R.; Kasper, M. The Molecular Anatomy of Mouse Skin during Hair Growth and Rest. Cell Stem Cell 2020, 26, 441–457.e7. [Google Scholar] [CrossRef] [Green Version]
- Joshi, R.S. The Inner Root Sheath and the Men Associated with it Eponymically. Int. J. Trichol. 2011, 3, 57–62. [Google Scholar] [CrossRef] [Green Version]
- Priestley, G.C. Histological studies of the skin follicle types of the rat with special reference to the structure of the Huxley layer. J. Anat. 1967, 101, 491–504. [Google Scholar]
- Tarcsa, E.; Marekov, L.N.; Andreoli, J.; Idler, W.W.; Candi, E.; Chung, S.I.; Steinert, P.M. The fate of trichohyalin. Sequential post-translational modifications by peptidyl-arginine deiminase and transglutaminases. J. Biol. Chem. 1997, 272, 27893–27901. [Google Scholar] [CrossRef] [Green Version]
- Kanno, T.; Kawada, A.; Yamanouchi, J.; Yosida-Noro, C.; Yoshiki, A.; Shiraiwa, M.; Kusakabe, M.; Manabe, M.; Tezuka, T.; Takahara, H. Human peptidylarginine deiminase type III: Molecular cloning and nucleotide sequence of the cDNA, properties of the recombinant enzyme, and immunohistochemical localization in human skin. J. Investig. Dermatol. 2000, 115, 813–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méchin, M.C.; Takahara, H.; Simon, M. Deimination and Peptidylarginine Deiminases in Skin Physiology and Diseases. Int. J. Mol. Sci. 2020, 21, 566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langbein, L.; Yoshida, H.; Praetzel-Wunder, S.; Parry, D.A.; Schweizer, J. The keratins of the human beard hair medulla: The riddle in the middle. J. Investig. Dermatol. 2010, 130, 55–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thibaut, S.; Cavusoglu, N.; de Becker, E.; Zerbib, F.; Bednarczyk, A.; Schaeffer, C.; van Dorsselaer, A.; Bernard, B.A. Transglutaminase-3 enzyme: A putative actor in human hair shaft scaffolding? J. Investig. Dermatol. 2009, 129, 449–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamada, Y.; Takama, H.; Kitamura, T.; Ikeya, T.; Yokochi, T. Expression of transglutaminase I in human anagen hair follicles. Acta Derm. Venereol. 1995, 75, 190–192. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Parry, D.A.; Jones, L.N.; Idler, W.W.; Marekov, L.N.; Steinert, P.M. In vitro assembly and structure of trichocyte keratin intermediate filaments: A novel role for stabilization by disulfide bonding. J. Cell Biol. 2000, 151, 1459–1468. [Google Scholar] [CrossRef]
- Akiyama, M.; Matsuo, I.; Shimizu, H. Formation of cornified cell envelope in human hair follicle development. Br. J. Dermatol. 2002, 146, 968–976. [Google Scholar] [CrossRef]
- Tarcsa, E.; Candi, E.; Kartasova, T.; Idler, W.W.; Marekov, L.N.; Steinert, P.M. Structural and transglutaminase substrate properties of the small proline-rich 2 family of cornified cell envelope proteins. J. Biol. Chem. 1998, 273, 23297–23303. [Google Scholar] [CrossRef] [Green Version]
- Park, G.T.; Lim, S.E.; Jang, S.I.; Morasso, M.I. Suprabasin, a novel epidermal differentiation marker and potential cornified envelope precursor. J. Biol. Chem. 2002, 277, 45195–45202. [Google Scholar] [CrossRef] [Green Version]
- Fischer, J.; Koblyakova, Y.; Latendorf, T.; Wu, Z.; Meyer-Hoffert, U. Cross-linking of SPINK6 by transglutaminases protects from epidermal proteases. J. Investig. Dermatol. 2013, 133, 1170–1177. [Google Scholar] [CrossRef] [Green Version]
- Henry, J.; Hsu, C.Y.; Haftek, M.; Nachat, R.; de Koning, H.D.; Gardinal-Galera, I.; Hitomi, K.; Balica, S.; Jean-Decoster, C.; Schmitt, A.M.; et al. Hornerin is a component of the epidermal cornified cell envelopes. FASEB J. 2011, 25, 1567–1576. [Google Scholar] [CrossRef] [PubMed]
- Candi, E.; Oddi, S.; Terrinoni, A.; Paradisi, A.; Ranalli, M.; Finazzi-Agró, A.; Melino, G. Transglutaminase 5 cross-links loricrin, involucrin, and small proline-rich proteins in vitro. J. Biol. Chem. 2001, 276, 35014–35023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deasey, S.; Shanmugasundaram, S.; Nurminskaya, M. Tissue-specific responses to loss of transglutaminase 2. Amino Acids 2013, 44, 179–187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- John, S.; Thiebach, L.; Frie, C.; Mokkapati, S.; Bechtel, M.; Nischt, R.; Rosser-Davies, S.; Paulsson, M.; Smyth, N. Epidermal transglutaminase (TGase 3) is required for proper hair development, but not the formation of the epidermal barrier. PLoS ONE 2012, 7, e34252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bognar, P.; Nemeth, I.; Mayer, B.; Haluszka, D.; Wikonkal, N.; Ostorhazi, E.; John, S.; Paulsson, M.; Smyth, N.; Paszto, M.I.; et al. Reduced inflammatory threshold indicates skin barrier defect in transglutaminase 3 knockout mice. J. Investig. Dermatol. 2014, 134, 105–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frezza, V.; Terrinoni, A.; Pitolli, C.; Mauriello, A.; Melino, G.; Candi, E. Transglutaminase 3 protects against photodamage. J. Investig. Dermatol. 2017, 137, 1590–1594. [Google Scholar] [CrossRef] [PubMed]
- Piro, M.C.; Ventura, A.; Smirnov, A.; Saggini, A.; Lena, A.M.; Mauriello, A.; Bianchi, L.; Melino, G.; Candi, E. Transglutaminase 3 Reduces the Severity of Psoriasis in Imiquimod-Treated Mouse Skin. Int. J. Mol. Sci. 2020, 21, 1566. [Google Scholar] [CrossRef] [Green Version]
- Hertwig, P. Neue mutationen und koppelungsgruppen bei der hausmaus. Z. Indukt. Abstamm. Vererbungsl. 1942, 80, 220–246. [Google Scholar] [CrossRef]
- Koniukhov, B.V.; Kupriianov, S.D. The mutant gene wellhaarig disturbs the differentiation of hair follicle cells in the mouse. Ontogenez 1990, 21, 56–62. (In Russian) [Google Scholar]
- Brennan, B.M.; Huynh, M.T.; Rabah, M.A.; Shaw, H.E.; Bisaillon, J.J.; Radden, L.A., 2nd; Nguyen, T.V.; King, T.R. The mouse wellhaarig (we) mutations result from defects in epidermal-type transglutaminase 3 (Tgm3). Mol. Genet. Metab. 2015, 116, 187–191. [Google Scholar] [CrossRef] [Green Version]
- Bryson, W.G.; Harland, D.P.; Caldwell, J.P.; Vernon, J.A.; Walls, R.J.; Woods, J.L.; Nagase, S.; Itou, T.; Koike, K. Cortical Cell Types and Intermediate Filament Arrangements Correlate with Fiber Curvature in Japanese Human Hair. J. Struct. Biol. 2009, 166, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Cloete, E.; Khumalo, N.P.; Ngoepe, M.N. The what, why and how of curly hair: A review. Proc. Math. Phys. Eng. Sci. 2019, 475, 20190516. [Google Scholar] [CrossRef] [PubMed]
- Rippa, A.; Terskikh, V.; Nesterova, A.; Vasiliev, A.; Vorotelyak, E. Hair follicle morphogenesis and epidermal homeostasis in we/we wal/wal mice with postnatal alopecia. Histochem. Cell Biol. 2015, 143, 481–496. [Google Scholar] [CrossRef] [PubMed]
- Koniukhov, B.V.; Malinina, N.A.; Martynov, M.I. The we gene is a modifier of the wal gene in mice. Genetika 2004, 40, 968–974. (In Russian) [Google Scholar] [CrossRef] [PubMed]
- Hicks, J.; Metry, D.W.; Barrish, J.; Levy, M. Uncombable hair (cheveux incoiffables, pili trianguli et canaliculi) syndrome: Brief review and role of scanning electron microscopy in diagnosis. Ultrastruct. Pathol. 2001, 25, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Hebert, A.A.; Charrow, J.; Esterly, N.B.; Fretzin, D.F. Uncombable hair (pili trianguli et canaliculi): Evidence for dominant inheritance with complete penetrance based on scanning electron microscopy. Am. J. Med. Genet. 1987, 28, 185–193. [Google Scholar] [CrossRef]
- Mallon, E.; Dawber, R.P.; De Berker, D.; Ferguson, D.J. Cheveux incoiffables--diagnostic, clinical and hair microscopic findings, and pathogenic studies. Br. J. Dermatol. 1994, 131, 608–614. [Google Scholar] [CrossRef]
- Rogers, M. Hair shaft abnormalities: Part II. Australas. J. Dermatol. 1996, 37, 1–11. [Google Scholar] [CrossRef]
- Garty, B.; Metzker, A.; Mimouni, M.; Varsano, I. Uncombable hair: A condition with autosomal dominant inheritance. Arch. Dis. Child. 1982, 57, 710–712. [Google Scholar] [CrossRef]
- Shelley, W.B.; Shelley, E.D. Uncombable hair syndrome: Observations on response to biotin and occurrence in siblings with ectodermal dysplasia. J. Am. Acad. Dermatol. 1985, 13, 97–102. [Google Scholar] [CrossRef]
- Fritz, T.M.; Trüeb, R.M. Uncombable hair syndrome with angelshaped phalango-epiphyseal dysplasia. Pediatr. Dermatol. 2000, 17, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Whiting, D.A. Hair shaft defects. In Disorders of Hair Growth: Diagnosis and Treatment; Olsen, E.A., Ed.; McGrawHill: Barcelona, Spain, 2003; pp. 123–175. [Google Scholar]
- Vickers, C.; Oberlin, D.; Shwayder, T.A. A girl with loose anagen hair syndrome and concurrent uncombable hair syndrome. JAAD Case Rep. 2020, 6, 92–95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méchin, M.C.; Sebbag, M.; Arnaud, J.; Nachat, R.; Foulquier, C.; Adoue, V.; Coudane, F.; Duplan, H.; Schmitt, A.M.; Chavanas, S.; et al. Update on peptidylarginine deiminases and deimination in skin physiology and severe human diseases. Int. J. Cosmet. Sci. 2007, 29, 147–168. [Google Scholar] [CrossRef] [PubMed]
- Szpak, M.; Mezzavilla, M.; Ayub, Q.; Chen, Y.; Xue, Y.; Tyler-Smith, C. FineMAV: Prioritizing candidate genetic variants driving local adaptations in human populations. Genome Biol. 2018, 19, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, G.; Zhao, Z.; Fu, W.; Sun, X.; Xu, Z.; Sun, K. Study on the loss of heterozygosity and expression of transglutaminase 3 gene in laryngeal carcinoma. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 2002, 19, 120–123. [Google Scholar] [PubMed]
- Li, W.; Zhang, Z.; Zhao, W.; Han, N. Transglutaminase 3 protein modulates human esophageal cancer cell growth by targeting the NF-κB signaling pathway. Oncol. Rep. 2016, 36, 1723–1730. [Google Scholar] [CrossRef]
- Kim, H.J.; Hawke, N.; Baldwin, A.S. NF-kappaB and IKK as therapeutic targets in cancer. Cell Death Differ. 2006, 13, 738–747. [Google Scholar] [CrossRef] [Green Version]
- Perkins, N.D. The diverse and complex roles of NF-kappaB subunits in cancer. Nat. Rev. Cancer 2012, 12, 121–132. [Google Scholar] [CrossRef]
- Méndez, E.; Fan, W.; Choi, P.; Agoff, S.N.; Whipple, M.; Farwell, D.G.; Futran, N.D.; Weymuller, E.A., Jr.; Zhao, L.P.; Chen, C. Tumor-specific genetic expression profile of metastatic oral squamous cell carcinoma. Head Neck. 2007, 29, 803–814. [Google Scholar] [CrossRef]
- Negishi, A.; Masuda, M.; Ono, M.; Honda, K.; Shitashige, M.; Satow, R.; Sakuma, T.; Kuwabara, H.; Nakanishi, Y.; Kanai, Y.; et al. Quantitative proteomics using formalin-fixed paraffin-embedded tissues of oral squamous cell carcinoma. Cancer Sci. 2009, 100, 1605–1611. [Google Scholar] [CrossRef]
- Nayak, S.; Bhatt, M.L.B.; Goel, M.M.; Gupta, S.; Mahdi, A.A.; Mishra, A.; Mehrotra, D. Tissue and serum expression of TGM-3 may be prognostic marker in patients of oral squamous cell carcinoma undergoing chemo-radiotherapy. PLoS ONE 2018, 13, e0199665. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, Y.; Wan, J.; Liu, Z. Expression of TGM3 protein and its significance in laryngeal carcinoma. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 2012, 26, 101–103. [Google Scholar] [PubMed]
- Chen, B.S.; Wang, M.R.; Xu, X.; Cai, Y.; Xu, Z.X.; Han, Y.L.; Wu, M. Transglutaminase-3, an esophageal cancer-related gene. Int. J. Cancer 2000, 88, 862–865. [Google Scholar] [CrossRef]
- Hu, J.W.; Yang, Z.F.; Li, J.; Hu, B.; Luo, C.B.; Zhu, K.; Dai, Z.; Cai, J.B.; Zhan, H.; Hu, Z.Q.; et al. TGM3 promotes epithelial-mesenchymal transition and hepatocellular carcinogenesis and predicts poor prognosis for patients after curative resection. Dig. Liver Dis. 2019, 52, 668–676. [Google Scholar] [CrossRef] [PubMed]
- Smirnov, A.; Anemona, L.; Montanaro, M.; Mauriello, A.; Annicchiarico-Petruzzelli, M.; Campione, E.; Melino, G.; Candi, E. Transglutaminase 3 is expressed in basal cell carcinoma of the skin. Eur. J. Dermatol. 2019, 29, 477–483. [Google Scholar] [CrossRef] [Green Version]
- Greenburg, G.; Hay, E.D. Epithelia suspended in collagen gels can lose polarity and express characteristics of migrating mesenchymal cells. J. Cell Biol. 1982, 95, 333–339. [Google Scholar] [CrossRef]
- Galvan, J.A.; Astudillo, A.; Vallina, A.; Fonseca, P.J.; Gomez-Izquierdo, L.; Garcia-Carbonero, R.; Gonzalez, M.V. Epithelial-mesenchymal transition markers in the differential diagnosis of gastroenteropancreatic neuroendocrine tumors. Am. J. Clin. Pathol. 2013, 140, 61–72. [Google Scholar] [CrossRef] [Green Version]
- Bakin, A.V.; Tomlinson, A.K.; Bhowmick, N.A.; Moses, H.L.; Arteaga, C.L. Phosphatidylinositol 3-kinase function is required for transforming growth factor beta-mediated epithelial to mesenchymal transition and cell migration. J. Biol. Chem. 2000, 275, 36803–36810. [Google Scholar] [CrossRef] [Green Version]
- Xu, Q.; Ma, J.; Lei, J.; Duan, W.; Sheng, L.; Chen, X.; Hu, A.; Wang, Z.; Wu, Z.; Wu, E.; et al. Alpha-Mangostin suppresses the viability and epithelial-mesenchymal transition of pancreatic cancer cells by downregulating the PI3K/Akt pathway. Biomed. Res. Int. 2014, 2014, 546353. [Google Scholar] [CrossRef]
- Khavari, P.A. Modelling cancer in human skin tissue. Nat. Rev. Cancer 2006, 6, 270–280. [Google Scholar] [CrossRef]
- Owens, B. Melanoma. Nature 2014, 515, S109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peterson, S.C.; Eberl, M.; Vagnozzi, A.N.; Belkadi, A.; Veniaminova, N.A.; Verhaegen, M.E.; Bichakjian, C.K.; Ward, N.L.; Dlugosz, A.A.; Wong, S.Y. Basal cell carcinoma preferentially arises from stem cells within hair follicle and mechanosensory niches. Cell Stem Cell 2015, 16, 400–412. [Google Scholar] [CrossRef] [Green Version]
- Stacey, S.N.; Sulem, P.; Gudbjartsson, D.F.; Jonasdottir, A.; Thorleifsson, G.; Gudjonsson, S.A.; Masson, G.; Gudmundsson, J.; Sigurgeirsson, B.; Benediktsdottir, K.R.; et al. Germline sequence variants in TGM3 and RGS22 confer risk of basal cell carcinoma. Hum. Mol. Genet. 2014, 23, 3045–3053. [Google Scholar] [CrossRef] [PubMed]
- Antiga, E.; Maglie, R.; Quintarelli, L.; Verdelli, A.; Bonciani, D.; Bonciolini, V.; Caproni, M. Dermatitis Herpetiformis: Novel Perspectives. Front. Immunol. 2019, 10, 1290. [Google Scholar] [CrossRef] [PubMed]
- Bolotin, D.; Petronic-Rosic, V. Dermatitis herpetiformis. Part, I. Epidemiology, pathogenesis, and clinical presentation. J. Am. Acad. Dermatol. 2011, 64, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Van der Meer, J.B. Granular deposits of immunoglobulins in the skin of patients with dermatitis herpetiformis. An immunofluorescent study. Br. J. Dermatol. 1969, 81, 493–503. [Google Scholar] [CrossRef]
- Fry, L. Dermatitis herpetiformis. Baillieres Clin. Gastroenterol. 1995, 9, 371–393. [Google Scholar] [CrossRef]
- Salmi, T.T. Dermatitis herpetiformis. Clin. Exp. Dermatol. 2019, 44, 728–731. [Google Scholar] [CrossRef] [Green Version]
- Dieterich, W.; Ehnis, T.; Bauer, M.; Donner, P.; Volta, U.; Riecken, E.O.; Schuppan, D. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat. Med. 1997, 3, 797–801. [Google Scholar] [CrossRef]
- Rewers, M. Epidemiology of celiac disease: What are the prevalence, incidence, and progression of celiac disease? Gastroenterology 2005, 128, S47–S51. [Google Scholar] [CrossRef]
- Lundin, K.E.; Gjertsen, H.A.; Scott, H.; Sollid, L.M.; Thorsby, E. Function of DQ2 and DQ8 as HLA susceptibility molecules in celiac disease. Hum. Immunol. 1994, 41, 24–27. [Google Scholar] [CrossRef]
- Spurkland, A.; Ingvarsson, G.; Falk, E.S.; Knutsen, I.; Sollid, L.M.; Thorsby, E. Dermatitis herpetiformis and celiac disease are both primarily associated with the HLA-DQ (alpha 1*0501, beta 1*02) or the HLA-DQ (alpha 1*03, beta 1*0302) heterodimers. Tissue Antigens 1997, 49, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Kárpáti, S.; Sárdy, M.; Németh, K.; Mayer, B.; Smyth, N.; Paulsson, M.; Traupe, H. Transglutaminases in autoimmune and inherited skin diseases: The phenomena of epitope spreading and functional compensation. Exp. Dermatol. 2018, 27, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Hietikko, M.; Hervonen, K.; Ilus, T.; Salmi, T.; Huhtala, H.; Laurila, K.; Rauhavirta, T.; Reunala, T.; Kaukinen, K.; Lindfors, K. Ex vivo culture of duodenal biopsies from patients with Dermatitis herpetiformis indicates that transglutaminase 3 antibody production occurs in the gut. Acta Derm. Venereol. 2018, 98, 366–372. [Google Scholar] [CrossRef] [Green Version]
- Hietikko, M.; Hervonen, K.; Salmi, T.; Ilus, T.; Zone, J.J.; Kaukinen, K.; Reunala, T.; Lindfors, K. Disappearance of epidermal transglutaminase and IgA deposits from the papillary dermis of dermatitis herpetiformis patients after a long-term gluten-free diet. Br. J. Dermatol. 2018, 178, e198–e201. [Google Scholar] [CrossRef]
- Caproni, M.; Antiga, E.; Melani, L.; Fabbri, P.; Italian Group for Cutaneous Immunopathology. Guidelines for the diagnosis and treatment of dermatitis herpetiformis. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 633–638. [Google Scholar] [CrossRef]
- Su, H.; Luo, Y.; Sun, J.; Liu, X.; Ling, S.; Xu, B.; Zhang, Y.; Liu, J.; Li, W.; Wang, B.; et al. Transglutaminase 3 promotes skin inflammation in atopic dermatitis by activating monocyte-derived dendritic cells via DC-SIGN. J. Investig. Dermatol. 2020, 140, 370–379. [Google Scholar] [CrossRef]
- Shimada, K.; Ochiai, T.; Hasegawa, H. Ectopic transglutaminase 1 and 3 expression accelerating keratinization in oral lichen planus. J. Int. Med. Res. 2018, 46, 4722–4730. [Google Scholar] [CrossRef] [Green Version]
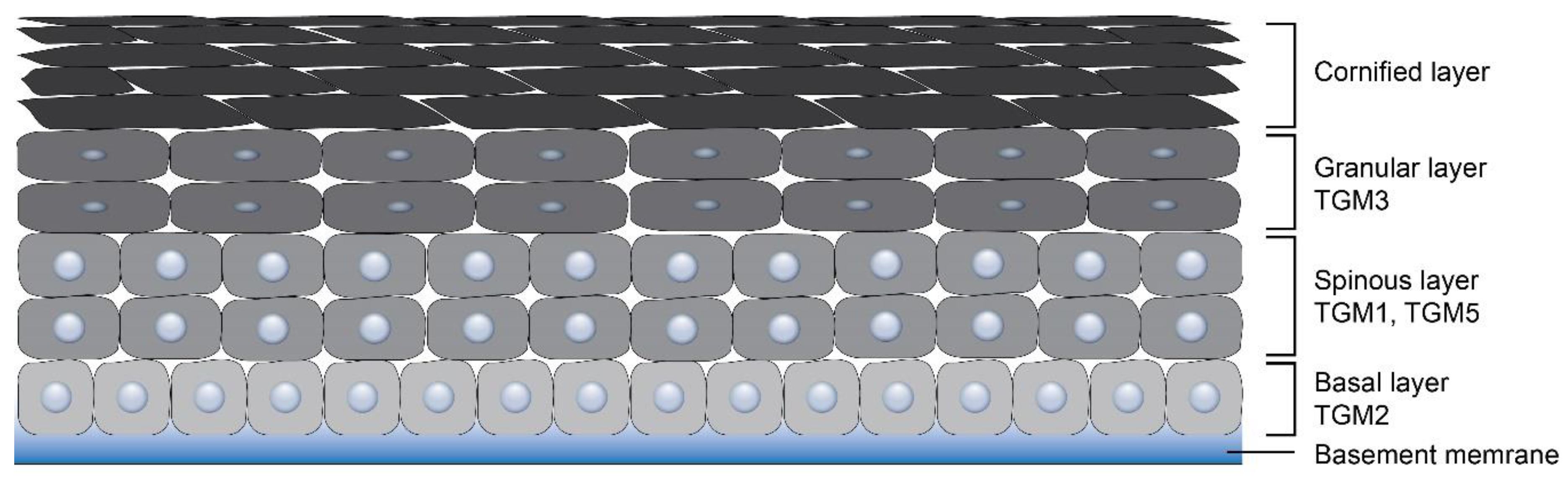
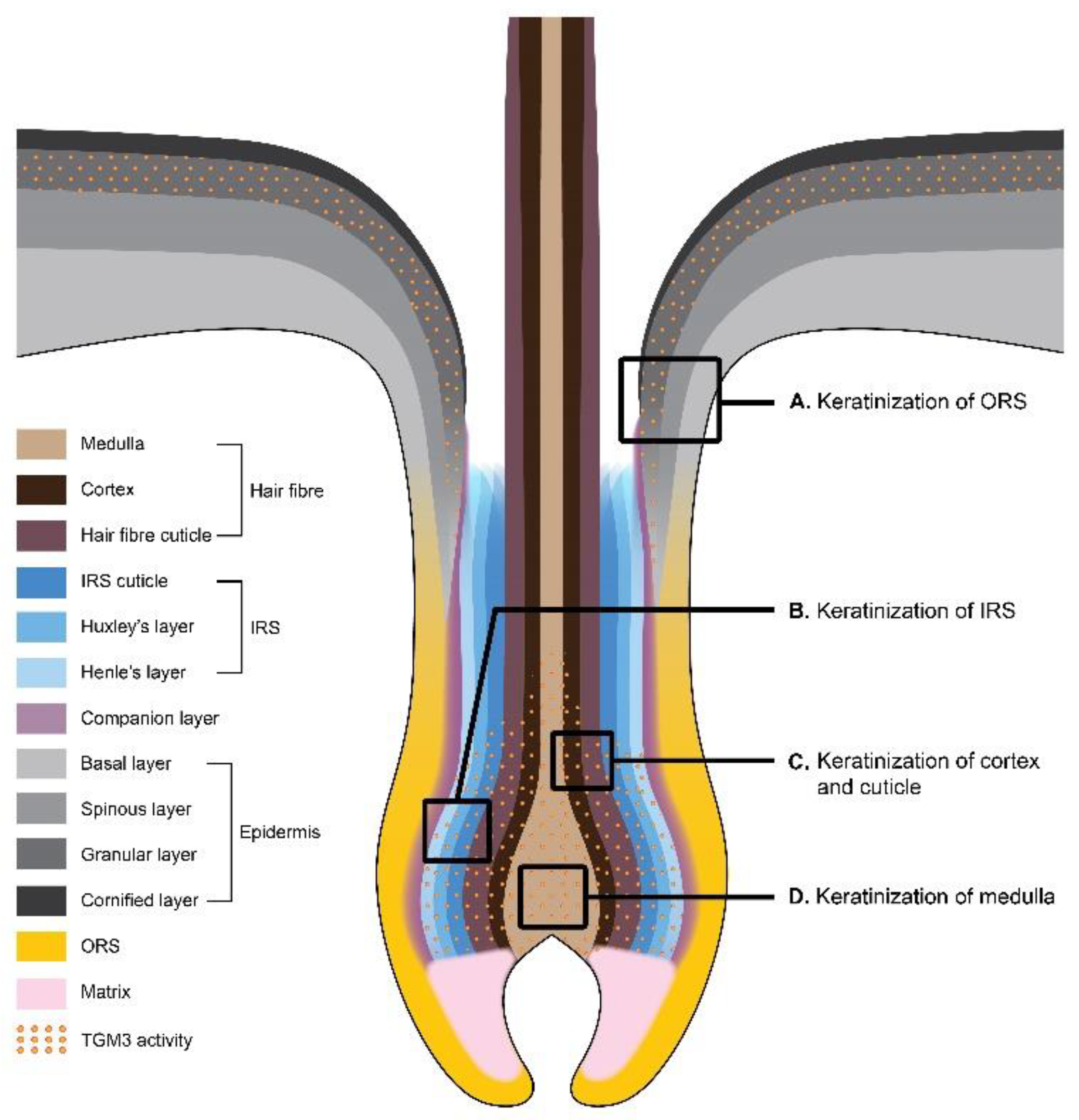
| Type of TGM | Synonyms | Function | Human Skin Diseases | Knockout Mouse Models | References |
|---|---|---|---|---|---|
| TGM1 | Keratinocyte TGM | Cornified cell envelope (CCE) formation, wound healing | Ichthyosis (lamellar ichthyosis, non-bullous congenital ichthyosiform erythroderma) | Defective stratum corneum and early neonatal death | [16,37,38,39,40,41] |
| TGM2 | Tissue TGM | Apoptosis, wound healing, angiogenesis, matrix stabilization, cell differentiation | Associated with various human disorders, including inflammation, cancer, and fibrosis, a mediator of the epidermal inflammatory response to UV irradiation | TGM2 null mice appear normal; upon UV exposure, display decreased skin inflammation compared to that of wild-type mice; Tgm2−/− fibroblasts demonstrate impaired adhesion in culture | [17,18,19,20,21,22] |
| TGM3 | Epidermal TGM | CCE formation, hair fiber stabilization | Uncombable hair syndrome, dermatitis herpetiformis | Impaired hair development | [42,43,44] |
| TGM5 | TGM X | Epidermal differentiation, CCE formation | Skin peeling syndrome, also involved in the hyperkeratosis in ichthyosis and psoriasis patients | No skin defects | [12,45] |
| TGM6 | TGM Y | Late stage CCE formation in the epidermis and the hair follicle | No skin defects | No skin defects | [12] |
| Type of Transglutaminase | Epidermis | Hair Follicle | References |
|---|---|---|---|
| TGM1 | Granular layer | Three layers of IRS. The innermost layer of ORS in the distal part (close to isthmus) of ORS | [27,62] |
| TGM2 | Basal layer | Hair germ and IRS of the bulbous hair peg | [19,79] |
| TGM3 | Upper granular layers | Cortex, medulla, cuticle, IRS, companion layer | [23,27,69] |
| TGM5 | A gradient of concentration from the basal layer to the stratum corneum | All three IRS layers, and residual quantities in the hair cuticle and the hair shaft, outer bulge, hair germ | [23,27,69] |
| Name of Cancer | Type of Epithelium | Type of Regulation | References |
|---|---|---|---|
| Oral carcinoma | Non-keratinizing stratified squamous epithelium | Downregulation | [34,112,113,114] |
| Laryngeal carcinoma | Non-keratinizing stratified squamous epithelium | Downregulation | [115] |
| Esophageal carcinoma | Non-keratinizing stratified squamous epithelium | Downregulation | [109,116] |
| Colorectal carcinoma | Simple columnar epithelium | Downregulation | [36] |
| Hepatocellular carcinoma | Simple cuboidal epithelium | Upregulation | [117] |
| Basal cell carcinoma (skin cancer) | Keratinized stratified squamous epithelium | Upregulation | [118] |
| Squamous cell carcinoma (skin cancer) | Keratinized stratified squamous epithelium | Downregulation | [118] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chermnykh, E.S.; Alpeeva, E.V.; Vorotelyak, E.A. Transglutaminase 3: The Involvement in Epithelial Differentiation and Cancer. Cells 2020, 9, 1996. https://doi.org/10.3390/cells9091996
Chermnykh ES, Alpeeva EV, Vorotelyak EA. Transglutaminase 3: The Involvement in Epithelial Differentiation and Cancer. Cells. 2020; 9(9):1996. https://doi.org/10.3390/cells9091996
Chicago/Turabian StyleChermnykh, Elina S., Elena V. Alpeeva, and Ekaterina A. Vorotelyak. 2020. "Transglutaminase 3: The Involvement in Epithelial Differentiation and Cancer" Cells 9, no. 9: 1996. https://doi.org/10.3390/cells9091996





