Nuclear Receptors as Autophagy-Based Antimicrobial Therapeutics
Abstract
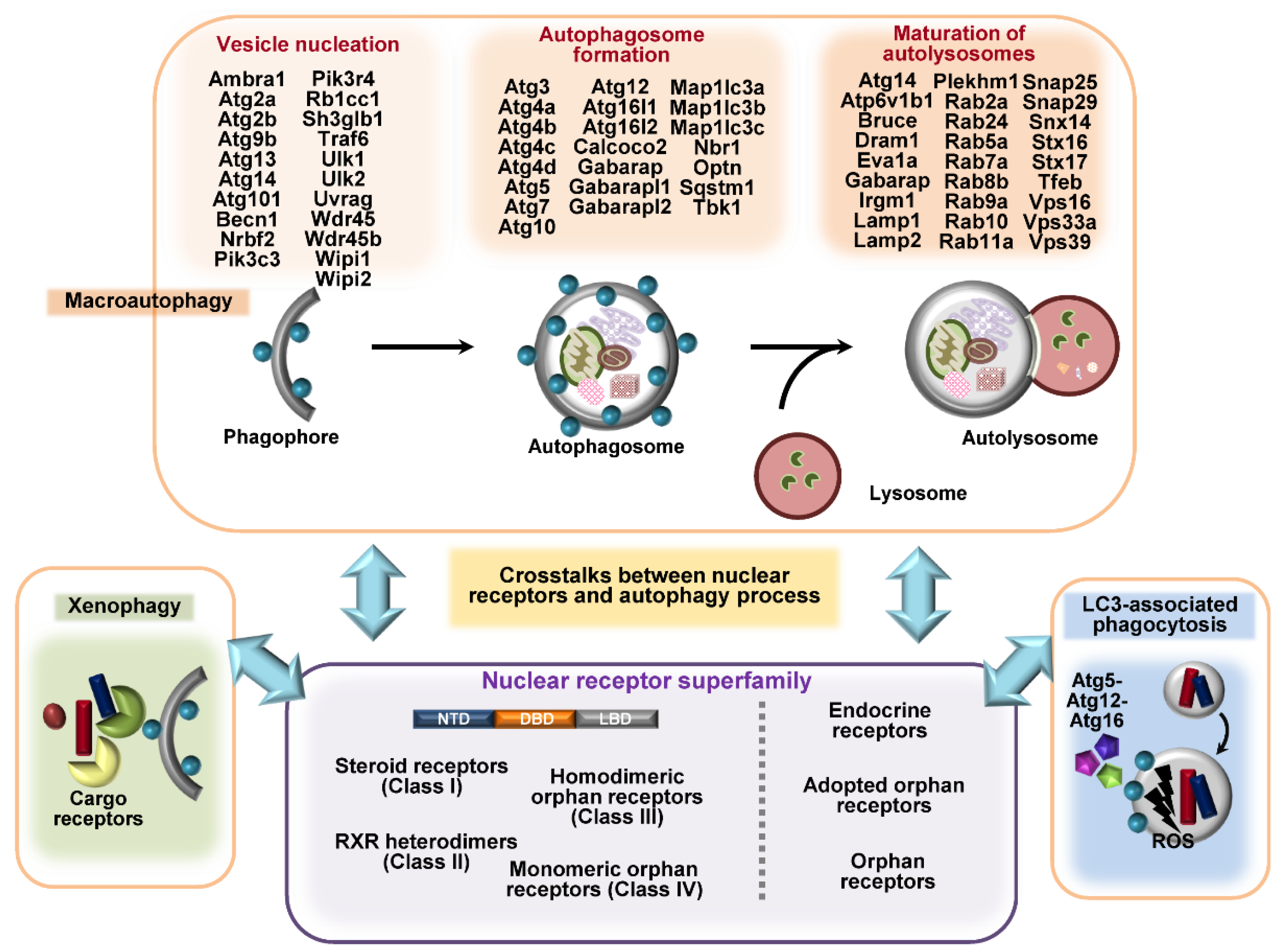
:1. Introduction
2. Overview of Autophagy and Autophagy-Related Genes
3. Overview of Nuclear Receptors
4. Vitamin D Receptor in Autophagy-Mediated Defense against Infection
5. Estrogen Receptors
6. Estrogen-Related Receptors
7. Peroxisome Proliferator-Activated Receptors
7.1. Peroxisome Proliferator-Activated Receptor-α
7.2. PPARβ/δ and PPARγ
8. Other Nuclear Receptors Potentially Linking Autophagy and Host Defenses
8.1. REV-ERBα and REV-ERBβ
8.2. Retinoic Acid Receptor-α (RARα; RARA; NR1B1), -β (RARβ; RARB; NR1B2), and -γ (RARγ; RARG; NR1B3)
8.3. Retinoic Acid-Related Orphan-α (RORα; RORA; NR1F1), -β (RORβ; RORB; NR1F2), and -γ (RORγ; RORC; NR1F3)
8.4. Farnesoid X Receptors-α (FXR-α)
8.5. Liver X Receptor (LXR)-α (LXRα; NR1H3) and -β (LXRβ; NR1H2)
8.6. Thyroid Hormone Receptors-α (TRα; THRA; NR1A1) and -β (TRβ; THRB; NR1A2)
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Huang, J.; Brumell, J.H. Autophagy in immunity against intracellular bacteria. Curr. Top. Microbiol. Immunol. 2009, 335, 189–215. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, M.D.S.; Ribeiro, R.M.; Travassos, L.H. Autophagy and Its Interaction With Intracellular Bacterial Pathogens. Front. Immunol. 2018, 9, 935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campoy, E.; Colombo, M.I. Autophagy in intracellular bacterial infection. Biochim. Biophys. Acta 2009, 1793, 1465–1477. [Google Scholar] [CrossRef] [Green Version]
- Yuk, J.M.; Yoshimori, T.; Jo, E.K. Autophagy and bacterial infectious diseases. Exp. Mol. Med. 2012, 44, 99–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahnazari, S.; Brumell, J.H. Mechanisms and consequences of bacterial targeting by the autophagy pathway. Curr. Opin. Microbiol. 2011, 14, 68–75. [Google Scholar] [CrossRef]
- Huang, J.; Brumell, J.H. Bacteria-autophagy interplay: A battle for survival. Nat. Rev. Microbiol. 2014, 12, 101–114. [Google Scholar] [CrossRef]
- Winchell, C.G.; Steele, S.; Kawula, T.; Voth, D.E. Dining in: Intracellular bacterial pathogen interplay with autophagy. Curr. Opin. Microbiol. 2016, 29, 9–14. [Google Scholar] [CrossRef] [Green Version]
- Ogawa, M.; Nakagawa, I.; Yoshikawa, Y.; Hain, T.; Chakraborty, T.; Sasakawa, C. Streptococcus-, Shigella-, and Listeria-induced autophagy. Methods Enzymol. 2009, 452, 363–381. [Google Scholar] [CrossRef]
- Ogawa, M.; Mimuro, H.; Yoshikawa, Y.; Ashida, H.; Sasakawa, C. Manipulation of autophagy by bacteria for their own benefit. Microbiol. Immunol. 2011, 55, 459–471. [Google Scholar] [CrossRef]
- Lerena, M.C.; Vazquez, C.L.; Colombo, M.I. Bacterial pathogens and the autophagic response. Cell Microbiol. 2010, 12, 10–18. [Google Scholar] [CrossRef]
- Ogawa, M.; Sasakawa, C. Bacterial evasion of the autophagic defense system. Curr. Opin. Microbiol. 2006, 9, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Radomski, N.; Rebbig, A.; Leonhardt, R.M.; Knittler, M.R. Xenophagic pathways and their bacterial subversion in cellular self-defense—pialphanutaualpha rhoepsiloniota—everything is in flux. Int. J. Med. Microbiol. 2018, 308, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.W.; Li, F. Bacterial interaction with host autophagy. Virulence 2019, 10, 352–362. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Qin, Y.; Chen, M. Viral strategies for triggering and manipulating mitophagy. Autophagy 2018, 14, 1665–1673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porter, B.A.; Ortiz, M.A.; Bratslavsky, G.; Kotula, L. Structure and Function of the Nuclear Receptor Superfamily and Current Targeted Therapies of Prostate Cancer. Cancers (Basel) 2019, 11, 1852. [Google Scholar] [CrossRef] [Green Version]
- Meinsohn, M.C.; Smith, O.E.; Bertolin, K.; Murphy, B.D. The Orphan Nuclear Receptors Steroidogenic Factor-1 and Liver Receptor Homolog-1: Structure, Regulation, and Essential Roles in Mammalian Reproduction. Physiol. Rev. 2019, 99, 1249–1279. [Google Scholar] [CrossRef] [PubMed]
- Wnuk, A.; Kajta, M. Steroid and Xenobiotic Receptor Signalling in Apoptosis and Autophagy of the Nervous System. Int. J. Mol. Sci. 2017, 18, 2394. [Google Scholar] [CrossRef] [Green Version]
- Fiorucci, S.; Biagioli, M.; Zampella, A.; Distrutti, E. Bile Acids Activated Receptors Regulate Innate Immunity. Front. Immunol. 2018, 9, 1853. [Google Scholar] [CrossRef] [Green Version]
- Bakke, D.; Sun, J. Ancient Nuclear Receptor VDR With New Functions: Microbiome and Inflammation. Inflamm. Bowel Dis. 2018, 24, 1149–1154. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.S.; Silwal, P.; Kim, S.Y.; Yoshimori, T.; Jo, E.K. Autophagy-activating strategies to promote innate defense against mycobacteria. Exp. Mol. Med. 2019, 51, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Xiang, J.; Liu, X.; Ren, J.; Chen, K.; Wang, H.L.; Miao, Y.Y.; Qi, M.M. How does estrogen work on autophagy? Autophagy 2019, 15, 197–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.M.; Wagner, M.; Xiao, R.; Kim, K.H.; Feng, D.; Lazar, M.A.; Moore, D.D. Nutrient-sensing nuclear receptors coordinate autophagy. Nature 2014, 516, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Yang, C.S.; Lee, H.M.; Kim, J.K.; Kim, Y.S.; Kim, Y.R.; Kim, J.S.; Kim, T.S.; Yuk, J.M.; Dufour, C.R.; et al. ESRRA (estrogen-related receptor alpha) is a key coordinator of transcriptional and post-translational activation of autophagy to promote innate host defense. Autophagy 2018, 14, 152–168. [Google Scholar] [CrossRef] [PubMed]
- Veras Ribeiro Filho, H.; Tambones, I.L.; Mariano Goncalves Dias, M.; Bernardi Videira, N.; Bruder, M.; Amorim Amato, A.; Migliorini Figueira, A.C. Modulation of nuclear receptor function: Targeting the protein-DNA interface. Mol. Cell Endocrinol. 2019, 484, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Neavin, D.R.; Liu, D.; Ray, B.; Weinshilboum, R.M. The Role of the Aryl Hydrocarbon Receptor (AHR) in Immune and Inflammatory Diseases. Int. J. Mol. Sci. 2018, 19, 3851. [Google Scholar] [CrossRef] [Green Version]
- Scheschowitsch, K.; Leite, J.A.; Assreuy, J. New Insights in Glucocorticoid Receptor Signaling-More Than Just a Ligand-Binding Receptor. Front. Endocrinol. (Lausanne) 2017, 8, 16. [Google Scholar] [CrossRef]
- Tripathi, M.; Yen, P.M.; Singh, B.K. Estrogen-Related Receptor Alpha: An Under-Appreciated Potential Target for the Treatment of Metabolic Diseases. Int. J. Mol. Sci. 2020, 21, 1645. [Google Scholar] [CrossRef] [Green Version]
- Levine, B.; Kroemer, G. Autophagy in the pathogenesis of disease. Cell 2008, 132, 27–42. [Google Scholar] [CrossRef] [Green Version]
- Deretic, V.; Levine, B. Autophagy, immunity, and microbial adaptations. Cell Host Microbe 2009, 5, 527–549. [Google Scholar] [CrossRef] [Green Version]
- Deretic, V.; Saitoh, T.; Akira, S. Autophagy in infection, inflammation and immunity. Nat. Rev. Immunol. 2013, 13, 722–737. [Google Scholar] [CrossRef]
- Keller, M.D.; Torres, V.J.; Cadwell, K. Autophagy and microbial pathogenesis. Cell Death Differ. 2020, 27, 872–886. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, N.; Yoshimori, T.; Ohsumi, Y. The role of Atg proteins in autophagosome formation. Annu. Rev. Cell Dev. Biol. 2011, 27, 107–132. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, N. The ATG conjugation systems in autophagy. Curr. Opin. Cell Biol. 2020, 63, 1–10. [Google Scholar] [CrossRef]
- Levine, B.; Kroemer, G. Biological Functions of Autophagy Genes: A Disease Perspective. Cell 2019, 176, 11–42. [Google Scholar] [CrossRef] [Green Version]
- Behrends, C.; Sowa, M.E.; Gygi, S.P.; Harper, J.W. Network organization of the human autophagy system. Nature 2010, 466, 68–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, R.; Zeh, H.J.; Lotze, M.T.; Tang, D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ. 2011, 18, 571–580. [Google Scholar] [CrossRef]
- Codogno, P.; Mehrpour, M.; Proikas-Cezanne, T. Canonical and non-canonical autophagy: Variations on a common theme of self-eating? Nat. Rev. Mol. Cell Biol. 2011, 13, 7–12. [Google Scholar] [CrossRef]
- Heckmann, B.L.; Boada-Romero, E.; Cunha, L.D.; Magne, J.; Green, D.R. LC3-Associated Phagocytosis and Inflammation. J. Mol. Biol. 2017, 429, 3561–3576. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, J.; Fan, W.; Wong, K.N.; Ding, X.; Chen, S.; Zhong, Q. The RUN domain of rubicon is important for hVps34 binding, lipid kinase inhibition, and autophagy suppression. J. Biol. Chem. 2011, 286, 185–191. [Google Scholar] [CrossRef] [Green Version]
- Dikic, I.; Elazar, Z. Mechanism and medical implications of mammalian autophagy. Nat. Rev. Mol. Cell Biol. 2018, 19, 349–364. [Google Scholar] [CrossRef]
- Sanchez-Martin, P.; Komatsu, M. Physiological Stress Response by Selective Autophagy. J. Mol. Biol. 2020, 432, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Martin, P.; Komatsu, M. p62/SQSTM1—steering the cell through health and disease. J. Cell Sci. 2018, 131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, S.J.; Zhang, X.; Rodriguez-Velez, A.; Evans, T.D.; Razani, B. p62/SQSTM1 and Selective Autophagy in Cardiometabolic Diseases. Antioxid. Redox Signal. 2019, 31, 458–471. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.; Cunha, L.D.; Park, S.; Yang, M.; Lu, Q.; Orchard, R.; Li, Q.Z.; Yan, M.; Janke, L.; Guy, C.; et al. Noncanonical autophagy inhibits the autoinflammatory, lupus-like response to dying cells. Nature 2016, 533, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Wesselborg, S.; Stork, B. Autophagy signal transduction by ATG proteins: From hierarchies to networks. Cell Mol. Life Sci. 2015, 72, 4721–4757. [Google Scholar] [CrossRef] [Green Version]
- Maruzs, T.; Simon-Vecsei, Z.; Kiss, V.; Csizmadia, T.; Juhasz, G. On the Fly: Recent Progress on Autophagy and Aging in Drosophila. Front. Cell Dev. Biol. 2019, 7, 140. [Google Scholar] [CrossRef]
- Kuo, C.J.; Hansen, M.; Troemel, E. Autophagy and innate immunity: Insights from invertebrate model organisms. Autophagy 2018, 14, 233–242. [Google Scholar] [CrossRef] [Green Version]
- Jin, M.; Klionsky, D.J. Regulation of autophagy: Modulation of the size and number of autophagosomes. FEBS Lett. 2014, 588, 2457–2463. [Google Scholar] [CrossRef] [Green Version]
- Feng, Y.; Yao, Z.; Klionsky, D.J. How to control self-digestion: Transcriptional, post-transcriptional, and post-translational regulation of autophagy. Trends Cell Biol. 2015, 25, 354–363. [Google Scholar] [CrossRef] [Green Version]
- Feng, Y.; He, D.; Yao, Z.; Klionsky, D.J. The machinery of macroautophagy. Cell Res. 2014, 24, 24–41. [Google Scholar] [CrossRef] [Green Version]
- McEwan, D.G.; Dikic, I. The Three Musketeers of Autophagy: Phosphorylation, ubiquitylation and acetylation. Trends Cell Biol. 2011, 21, 195–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banreti, A.; Sass, M.; Graba, Y. The emerging role of acetylation in the regulation of autophagy. Autophagy 2013, 9, 819–829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamai, A.; Codogno, P. New targets for acetylation in autophagy. Sci. Signal. 2012, 5, pe29. [Google Scholar] [CrossRef]
- Xie, Y.; Kang, R.; Sun, X.; Zhong, M.; Huang, J.; Klionsky, D.J.; Tang, D. Posttranslational modification of autophagy-related proteins in macroautophagy. Autophagy 2015, 11, 28–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Malta, C.; Cinque, L.; Settembre, C. Transcriptional Regulation of Autophagy: Mechanisms and Diseases. Front. Cell Dev. Biol. 2019, 7, 114. [Google Scholar] [CrossRef] [PubMed]
- Jo, E.K.; Yuk, J.M.; Shin, D.M.; Sasakawa, C. Roles of autophagy in elimination of intracellular bacterial pathogens. Front. Immunol. 2013, 4, 97. [Google Scholar] [CrossRef] [Green Version]
- Campoy, E.; Colombo, M.I. Autophagy subversion by bacteria. Curr. Top. Microbiol. Immunol. 2009, 335, 227–250. [Google Scholar] [CrossRef]
- Paul, P.; Munz, C. Autophagy and Mammalian Viruses: Roles in Immune Response, Viral Replication, and Beyond. Adv. Virus Res. 2016, 95, 149–195. [Google Scholar] [CrossRef] [Green Version]
- Choi, Y.; Bowman, J.W.; Jung, J.U. Autophagy during viral infection—A double-edged sword. Nat. Rev. Microbiol. 2018, 16, 341–354. [Google Scholar] [CrossRef]
- Kudchodkar, S.B.; Levine, B. Viruses and autophagy. Rev. Med. Virol. 2009, 19, 359–378. [Google Scholar] [CrossRef]
- Mangelsdorf, D.J.; Thummel, C.; Beato, M.; Herrlich, P.; Schutz, G.; Umesono, K.; Blumberg, B.; Kastner, P.; Mark, M.; Chambon, P.; et al. The nuclear receptor superfamily: The second decade. Cell 1995, 83, 835–839. [Google Scholar] [CrossRef] [Green Version]
- Umesono, K.; Evans, R.M. Determinants of target gene specificity for steroid/thyroid hormone receptors. Cell 1989, 57, 1139–1146. [Google Scholar] [CrossRef]
- Huang, P.; Chandra, V.; Rastinejad, F. Structural overview of the nuclear receptor superfamily: Insights into physiology and therapeutics. Annu. Rev. Physiol. 2010, 72, 247–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jimenez-Sousa, M.A.; Martinez, I.; Medrano, L.M.; Fernandez-Rodriguez, A.; Resino, S. Vitamin D in Human Immunodeficiency Virus Infection: Influence on Immunity and Disease. Front. Immunol. 2018, 9, 458. [Google Scholar] [CrossRef] [PubMed]
- Zdrenghea, M.T.; Makrinioti, H.; Bagacean, C.; Bush, A.; Johnston, S.L.; Stanciu, L.A. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev. Med. Virol. 2017, 27. [Google Scholar] [CrossRef]
- Wu, S.; Sun, J. Vitamin D, vitamin D receptor, and macroautophagy in inflammation and infection. Discov. Med. 2011, 11, 325–335. [Google Scholar]
- Paik, S.; Kim, J.K.; Chung, C.; Jo, E.K. Autophagy: A new strategy for host-directed therapy of tuberculosis. Virulence 2019, 10, 448–459. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.K.; Jeong, J.H.; Lee, J.M.; Kim, K.S.; Park, S.H.; Kim, Y.D.; Koh, M.; Shin, M.; Jung, Y.S.; Kim, H.S.; et al. Inverse agonist of estrogen-related receptor gamma controls Salmonella typhimurium infection by modulating host iron homeostasis. Nat. Med. 2014, 20, 419–424. [Google Scholar] [CrossRef]
- Korf, H.; Vander Beken, S.; Romano, M.; Steffensen, K.R.; Stijlemans, B.; Gustafsson, J.A.; Grooten, J.; Huygen, K. Liver X receptors contribute to the protective immune response against Mycobacterium tuberculosis in mice. J. Clin. Investig. 2009, 119, 1626–1637. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.S.; Kim, J.K.; Hanh, B.T.B.; Kim, S.Y.; Kim, H.J.; Kim, Y.J.; Jeon, S.M.; Park, C.R.; Oh, G.T.; Park, J.W.; et al. The Peroxisome Proliferator-Activated Receptor alpha-Agonist Gemfibrozil Promotes Defense Against Mycobacterium abscessus Infections. Cells 2020, 9, 648. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.S.; Jin, Y.B.; Kim, Y.S.; Kim, S.; Kim, J.K.; Lee, H.M.; Suh, H.W.; Choe, J.H.; Kim, Y.J.; Koo, B.S.; et al. SIRT3 promotes antimycobacterial defenses by coordinating mitochondrial and autophagic functions. Autophagy 2019, 15, 1356–1375. [Google Scholar] [CrossRef]
- Kim, Y.S.; Lee, H.M.; Kim, J.K.; Yang, C.S.; Kim, T.S.; Jung, M.; Jin, H.S.; Kim, S.; Jang, J.; Oh, G.T.; et al. PPAR-alpha Activation Mediates Innate Host Defense through Induction of TFEB and Lipid Catabolism. J. Immunol. 2017, 198, 3283–3295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Omeragic, A.; Kara-Yacoubian, N.; Kelschenbach, J.; Sahin, C.; Cummins, C.L.; Volsky, D.J.; Bendayan, R. Peroxisome Proliferator-Activated Receptor-gamma agonists exhibit anti-inflammatory and antiviral effects in an EcoHIV mouse model. Sci. Rep. 2019, 9, 9428. [Google Scholar] [CrossRef] [PubMed]
- Leopold Wager, C.M.; Arnett, E.; Schlesinger, L.S. Mycobacterium tuberculosis and macrophage nuclear receptors: What we do and don’t know. Tuberculosis (Edinb) 2019, 116S, S98–S106. [Google Scholar] [CrossRef] [PubMed]
- Leopold Wager, C.M.; Arnett, E.; Schlesinger, L.S. Macrophage nuclear receptors: Emerging key players in infectious diseases. PLoS Pathog. 2019, 15, e1007585. [Google Scholar] [CrossRef] [Green Version]
- Petta, I.; Dejager, L.; Ballegeer, M.; Lievens, S.; Tavernier, J.; De Bosscher, K.; Libert, C. The Interactome of the Glucocorticoid Receptor and Its Influence on the Actions of Glucocorticoids in Combatting Inflammatory and Infectious Diseases. Microbiol. Mol. Biol. Rev. 2016, 80, 495–522. [Google Scholar] [CrossRef] [Green Version]
- Montoya, M.C.; Krysan, D.J. Repurposing Estrogen Receptor Antagonists for the Treatment of Infectious Disease. mBio 2018, 9. [Google Scholar] [CrossRef] [Green Version]
- Medina-Estrada, I.; Alva-Murillo, N.; Lopez-Meza, J.E.; Ochoa-Zarzosa, A. Immunomodulatory Effects of 17beta-Estradiol on Epithelial Cells during Bacterial Infections. J. Immunol. Res. 2018, 2018, 6098961. [Google Scholar] [CrossRef]
- Erickson, S.L.; Alston, L.; Nieves, K.; Chang, T.K.H.; Mani, S.; Flannigan, K.L.; Hirota, S.A. The xenobiotic sensing pregnane X receptor regulates tissue damage and inflammation triggered by C difficile toxins. FASEB J. 2020, 34, 2198–2212. [Google Scholar] [CrossRef] [Green Version]
- Qiu, Z.; Cervantes, J.L.; Cicek, B.B.; Mukherjee, S.; Venkatesh, M.; Maher, L.A.; Salazar, J.C.; Mani, S.; Khanna, K.M. Pregnane X Receptor Regulates Pathogen-Induced Inflammation and Host Defense against an Intracellular Bacterial Infection through Toll-like Receptor 4. Sci. Rep. 2016, 6, 31936. [Google Scholar] [CrossRef] [Green Version]
- Bhagyaraj, E.; Nanduri, R.; Saini, A.; Dkhar, H.K.; Ahuja, N.; Chandra, V.; Mahajan, S.; Kalra, R.; Tiwari, D.; Sharma, C.; et al. Human Xenobiotic Nuclear Receptor PXR Augments Mycobacterium tuberculosis Survival. J. Immunol. 2016, 197, 244–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Curtis, A.M.; Bellet, M.M.; Sassone-Corsi, P.; O’Neill, L.A. Circadian clock proteins and immunity. Immunity 2014, 40, 178–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibbs, J.E.; Blaikley, J.; Beesley, S.; Matthews, L.; Simpson, K.D.; Boyce, S.H.; Farrow, S.N.; Else, K.J.; Singh, D.; Ray, D.W.; et al. The nuclear receptor REV-ERBalpha mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines. Proc. Natl. Acad. Sci. USA 2012, 109, 582–587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levy, G.; Habib, N.; Guzzardi, M.A.; Kitsberg, D.; Bomze, D.; Ezra, E.; Uygun, B.E.; Uygun, K.; Trippler, M.; Schlaak, J.F.; et al. Nuclear receptors control pro-viral and antiviral metabolic responses to hepatitis C virus infection. Nat. Chem. Biol. 2016, 12, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Prince, L.R.; Prosseda, S.D.; Higgins, K.; Carlring, J.; Prestwich, E.C.; Ogryzko, N.V.; Rahman, A.; Basran, A.; Falciani, F.; Taylor, P.; et al. NR4A orphan nuclear receptor family members, NR4A2 and NR4A3, regulate neutrophil number and survival. Blood 2017, 130, 1014–1025. [Google Scholar] [CrossRef]
- Mazaira, G.I.; Zgajnar, N.R.; Lotufo, C.M.; Daneri-Becerra, C.; Sivils, J.C.; Soto, O.B.; Cox, M.B.; Galigniana, M.D. The Nuclear Receptor Field: A Historical Overview and Future Challenges. Nucl. Receptor Res. 2018, 5. [Google Scholar] [CrossRef]
- Heldin, C.H.; Lu, B.; Evans, R.; Gutkind, J.S. Signals and Receptors. Cold Spring Harb. Perspect. Biol. 2016, 8, a005900. [Google Scholar] [CrossRef] [Green Version]
- Avior, Y.; Bomze, D.; Ramon, O.; Nahmias, Y. Flavonoids as dietary regulators of nuclear receptor activity. Food Funct. 2013, 4, 831–844. [Google Scholar] [CrossRef] [Green Version]
- Sever, R.; Glass, C.K. Signaling by nuclear receptors. Cold Spring Harb. Perspect. Biol. 2013, 5, a016709. [Google Scholar] [CrossRef] [Green Version]
- Ouyang, Q.; Zhang, K.; Lin, D.; Feng, C.G.; Cai, Y.; Chen, X. Bazedoxifene Suppresses Intracellular Mycobacterium tuberculosis Growth by Enhancing Autophagy. mSphere 2020, 5. [Google Scholar] [CrossRef] [Green Version]
- Jang, W.S.; Kim, S.; Podder, B.; Jyoti, M.A.; Nam, K.W.; Lee, B.E.; Song, H.Y. Anti-Mycobacterial Activity of Tamoxifen Against Drug-Resistant and Intra-Macrophage Mycobacterium tuberculosis. J. Microbiol. Biotechnol. 2015, 25, 946–950. [Google Scholar] [CrossRef] [PubMed]
- Weikum, E.R.; Liu, X.; Ortlund, E.A. The nuclear receptor superfamily: A structural perspective. Protein Sci. 2018, 27, 1876–1892. [Google Scholar] [CrossRef]
- Collingwood, T.N.; Urnov, F.D.; Wolffe, A.P. Nuclear receptors: Coactivators, corepressors and chromatin remodeling in the control of transcription. J. Mol. Endocrinol. 1999, 23, 255–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haussler, M.R.; Whitfield, G.K.; Haussler, C.A.; Hsieh, J.C.; Thompson, P.D.; Selznick, S.H.; Dominguez, C.E.; Jurutka, P.W. The nuclear vitamin D receptor: Biological and molecular regulatory properties revealed. J. Bone Miner. Res. 1998, 13, 325–349. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.; Silwal, P.; Kim, I.; Modlin, R.L.; Jo, E.K. Vitamin D-Cathelicidin Axis: At the Crossroads between Protective Immunity and Pathological Inflammation during Infection. Immune Netw. 2020, 20, e12. [Google Scholar] [CrossRef]
- Christofides, A.; Konstantinidou, E.; Jani, C.; Boussiotis, V.A. The role of Peroxisome Proliferator-Activated Receptors (PPAR) in immune responses. Metabolism 2020. [Google Scholar] [CrossRef]
- Wagner, K.D.; Wagner, N. Peroxisome proliferator-activated receptor beta/delta (PPARbeta/delta) acts as regulator of metabolism linked to multiple cellular functions. Pharmacol. Ther. 2010, 125, 423–435. [Google Scholar] [CrossRef]
- Glaria, E.; Letelier, N.A.; Valledor, A.F. Integrating the roles of liver X receptors in inflammation and infection: Mechanisms and outcomes. Curr. Opin. Pharmacol. 2020, 53, 55–65. [Google Scholar] [CrossRef]
- Glass, C.K.; Rosenfeld, M.G. The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev. 2000, 14, 121–141. [Google Scholar]
- Tremblay, A.M.; Wilson, B.J.; Yang, X.J.; Giguere, V. Phosphorylation-dependent sumoylation regulates estrogen-related receptor-alpha and -gamma transcriptional activity through a synergy control motif. Mol. Endocrinol. 2008, 22, 570–584. [Google Scholar] [CrossRef]
- Massafra, V.; van Mil, S.W.C. Farnesoid X receptor: A "homeostat" for hepatic nutrient metabolism. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.K.; Sinha, R.A.; Ohba, K.; Yen, P.M. Role of thyroid hormone in hepatic gene regulation, chromatin remodeling, and autophagy. Mol. Cell Endocrinol. 2017, 458, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Basu, J.; Shin, D.M.; Jo, E.K. Mycobacterial signaling through toll-like receptors. Front. Cell Infect. Microbiol. 2012, 2, 145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, S.T.; Litonjua, A.A. Vitamin D in Host Defense: Implications for Future Research. Am. J. Respir. Cell Mol. Biol. 2017, 56, 692–693. [Google Scholar] [CrossRef] [PubMed]
- Del Pinto, R.; Ferri, C.; Cominelli, F. Vitamin D Axis in Inflammatory Bowel Diseases: Role, Current Uses and Future Perspectives. Int. J. Mol. Sci. 2017, 18, 2360. [Google Scholar] [CrossRef] [Green Version]
- Hu, W.; Zhang, L.; Li, M.X.; Shen, J.; Liu, X.D.; Xiao, Z.G.; Wu, D.L.; Ho, I.H.T.; Wu, J.C.Y.; Cheung, C.K.Y.; et al. Vitamin D3 activates the autolysosomal degradation function against Helicobacter pylori through the PDIA3 receptor in gastric epithelial cells. Autophagy 2019, 15, 707–725. [Google Scholar] [CrossRef] [Green Version]
- Yuk, J.M.; Shin, D.M.; Lee, H.M.; Yang, C.S.; Jin, H.S.; Kim, K.K.; Lee, Z.W.; Lee, S.H.; Kim, J.M.; Jo, E.K. Vitamin D3 induces autophagy in human monocytes/macrophages via cathelicidin. Cell Host Microbe 2009, 6, 231–243. [Google Scholar] [CrossRef] [Green Version]
- Jo, E.K. Innate immunity to mycobacteria: Vitamin D and autophagy. Cell Microbiol. 2010, 12, 1026–1035. [Google Scholar] [CrossRef]
- Shin, D.M.; Yuk, J.M.; Lee, H.M.; Lee, S.H.; Son, J.W.; Harding, C.V.; Kim, J.M.; Modlin, R.L.; Jo, E.K. Mycobacterial lipoprotein activates autophagy via TLR2/1/CD14 and a functional vitamin D receptor signalling. Cell Microbiol. 2010, 12, 1648–1665. [Google Scholar] [CrossRef] [Green Version]
- Sato, E.; Imafuku, S.; Ishii, K.; Itoh, R.; Chou, B.; Soejima, T.; Nakayama, J.; Hiromatsu, K. Vitamin D-dependent cathelicidin inhibits Mycobacterium marinum infection in human monocytic cells. J. Dermatol. Sci. 2013, 70, 166–172. [Google Scholar] [CrossRef]
- Campbell, G.R.; Spector, S.A. Hormonally active vitamin D3 (1alpha,25-dihydroxycholecalciferol) triggers autophagy in human macrophages that inhibits HIV-1 infection. J. Biol. Chem. 2011, 286, 18890–18902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campbell, G.R.; Spector, S.A. Vitamin D inhibits human immunodeficiency virus type 1 and Mycobacterium tuberculosis infection in macrophages through the induction of autophagy. PLoS Pathog. 2012, 8, e1002689. [Google Scholar] [CrossRef] [Green Version]
- Campbell, G.R.; Spector, S.A. Toll-like receptor 8 ligands activate a vitamin D mediated autophagic response that inhibits human immunodeficiency virus type 1. PLoS Pathog. 2012, 8, e1003017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, M.; Tang, X.; Rekha, R.S.; Muvva, S.; Brighenti, S.; Agerberth, B.; Haeggstrom, J.Z. Prostaglandin E2 suppresses hCAP18/LL-37 expression in human macrophages via EP2/EP4: Implications for treatment of Mycobacterium tuberculosis infection. FASEB J. 2018, 32, 2827–2840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wahyunitisari, M.R.; Mertaniasih, N.M.; Amin, M.; Artama, W.T.; Koendhori, E.B. Vitamin D, cell death pathways, and tuberculosis. Int. J. Mycobacteriol 2017, 6, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Rekha, R.S.; Mily, A.; Sultana, T.; Haq, A.; Ahmed, S.; Mostafa Kamal, S.M.; van Schadewijk, A.; Hiemstra, P.S.; Gudmundsson, G.H.; Agerberth, B.; et al. Immune responses in the treatment of drug-sensitive pulmonary tuberculosis with phenylbutyrate and vitamin D3 as host directed therapy. BMC Infect. Dis. 2018, 18, 303. [Google Scholar] [CrossRef] [PubMed]
- Mily, A.; Rekha, R.S.; Kamal, S.M.; Arifuzzaman, A.S.; Rahim, Z.; Khan, L.; Haq, M.A.; Zaman, K.; Bergman, P.; Brighenti, S.; et al. Significant Effects of Oral Phenylbutyrate and Vitamin D3 Adjunctive Therapy in Pulmonary Tuberculosis: A Randomized Controlled Trial. PLoS ONE 2015, 10, e0138340. [Google Scholar] [CrossRef]
- Bekele, A.; Gebreselassie, N.; Ashenafi, S.; Kassa, E.; Aseffa, G.; Amogne, W.; Getachew, M.; Aseffa, A.; Worku, A.; Raqib, R.; et al. Daily adjunctive therapy with vitamin D3 and phenylbutyrate supports clinical recovery from pulmonary tuberculosis: A randomized controlled trial in Ethiopia. J. Intern. Med. 2018, 284, 292–306. [Google Scholar] [CrossRef]
- Wallis, R.S.; Zumla, A. Vitamin D as Adjunctive Host-Directed Therapy in Tuberculosis: A Systematic Review. Open Forum Infect. Dis. 2016, 3, ofw151. [Google Scholar] [CrossRef] [Green Version]
- Kearns, M.D.; Alvarez, J.A.; Seidel, N.; Tangpricha, V. Impact of vitamin D on infectious disease. Am. J. Med. Sci. 2015, 349, 245–262. [Google Scholar] [CrossRef] [Green Version]
- Fabri, M.; Stenger, S.; Shin, D.M.; Yuk, J.M.; Liu, P.T.; Realegeno, S.; Lee, H.M.; Krutzik, S.R.; Schenk, M.; Sieling, P.A.; et al. Vitamin D is required for IFN-gamma-mediated antimicrobial activity of human macrophages. Sci. Transl. Med. 2011, 3, 104ra102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klug-Micu, G.M.; Stenger, S.; Sommer, A.; Liu, P.T.; Krutzik, S.R.; Modlin, R.L.; Fabri, M. CD40 ligand and interferon-gamma induce an antimicrobial response against Mycobacterium tuberculosis in human monocytes. Immunology 2013, 139, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Gough, M.E.; Graviss, E.A.; May, E.E. The dynamic immunomodulatory effects of vitamin D3 during Mycobacterium infection. Innate Immun. 2017, 23, 506–523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, R.; Yang, E.; Shen, L.; Modlin, R.L.; Shen, H.; Chen, Z.W. IL-12+IL-18 Cosignaling in Human Macrophages and Lung Epithelial Cells Activates Cathelicidin and Autophagy, Inhibiting Intracellular Mycobacterial Growth. J. Immunol. 2018, 200, 2405–2417. [Google Scholar] [CrossRef] [Green Version]
- Godbole, N.M.; Sinha, R.A.; Tiwari, S.; Pawar, S.D.; Dhole, T.N. Analysis of influenza virus-induced perturbation in autophagic flux and its modulation during Vitamin D3 mediated anti-apoptotic signaling. Virus Res. 2020, 282, 197936. [Google Scholar] [CrossRef]
- Lu, R.; Shang, M.; Zhang, Y.G.; Jiao, Y.; Xia, Y.; Garrett, S.; Bakke, D.; Bauerl, C.; Martinez, G.P.; Kim, C.H.; et al. Lactic Acid Bacteria Isolated From Korean Kimchi Activate the Vitamin D Receptor-autophagy Signaling Pathways. Inflamm. Bowel. Dis. 2020, 26, 1199–1211. [Google Scholar] [CrossRef]
- Dai, J.; Liang, Y.; Li, H.; Zhou, W.; Wang, B.; Gong, A.; Zhang, R. Vitamin D enhances resistance to aspergillus fumigatus in mice via inhibition of excessive autophagy. Am. J. Transl Res. 2018, 10, 381–391. [Google Scholar]
- Tian, G.; Liang, X.; Chen, D.; Mao, X.; Yu, J.; Zheng, P.; He, J.; Huang, Z.; Yu, B. Vitamin D3 supplementation alleviates rotavirus infection in pigs and IPEC-J2 cells via regulating the autophagy signaling pathway. J. Steroid Biochem. Mol. Biol 2016, 163, 157–163. [Google Scholar] [CrossRef]
- Wei, Y.; Huang, J. Role of estrogen and its receptors mediated-autophagy in cell fate and human diseases. J. Steroid Biochem. Mol. Biol. 2019, 191, 105380. [Google Scholar] [CrossRef]
- Lipovka, Y.; Konhilas, J.P. The complex nature of oestrogen signalling in breast cancer: Enemy or ally? Biosci. Rep. 2016, 36. [Google Scholar] [CrossRef] [Green Version]
- Fan, D.; Liu, S.Y.; van Hasselt, C.A.; Vlantis, A.C.; Ng, E.K.; Zhang, H.; Dong, Y.; Ng, S.K.; Chu, R.; Chan, A.B.; et al. Estrogen receptor alpha induces prosurvival autophagy in papillary thyroid cancer via stimulating reactive oxygen species and extracellular signal regulated kinases. J. Clin. Endocrinol. Metab. 2015, 100, E561–E571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Selyunin, A.S.; Hutchens, S.; McHardy, S.F.; Mukhopadhyay, S. Tamoxifen blocks retrograde trafficking of Shiga toxin 1 and 2 and protects against lethal toxicosis. Life Sci. Alliance 2019, 2. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, A.J.; Drozda, A.A.; Blader, I.J. Drug Repurposing Screening Identifies Novel Compounds That Effectively Inhibit Toxoplasma gondii Growth. mSphere 2016, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, M.T.; Lee, Y.M.; Shen, H.H.; Cheng, P.Y.; Huang, Y.C.; Lin, Y.J.; Huang, Y.Y.; Lam, K.K. Activation of autophagy is involved in the protective effect of 17beta-oestradiol on endotoxaemia-induced multiple organ dysfunction in ovariectomized rats. J. Cell Mol. Med. 2017, 21, 3705–3717. [Google Scholar] [CrossRef] [PubMed]
- Giguere, V. Transcriptional control of energy homeostasis by the estrogen-related receptors. Endocr. Rev. 2008, 29, 677–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huss, J.M.; Garbacz, W.G.; Xie, W. Constitutive activities of estrogen-related receptors: Transcriptional regulation of metabolism by the ERR pathways in health and disease. Biochim. Biophys. Acta 2015, 1852, 1912–1927. [Google Scholar] [CrossRef] [Green Version]
- Audet-Walsh, E.; Giguere, V. The multiple universes of estrogen-related receptor alpha and gamma in metabolic control and related diseases. Acta Pharmacol. Sin. 2015, 36, 51–61. [Google Scholar] [CrossRef]
- Singh, B.K.; Sinha, R.A.; Tripathi, M.; Mendoza, A.; Ohba, K.; Sy, J.A.C.; Xie, S.Y.; Zhou, J.; Ho, J.P.; Chang, C.Y.; et al. Thyroid hormone receptor and ERRalpha coordinately regulate mitochondrial fission, mitophagy, biogenesis, and function. Sci. Signal. 2018, 11. [Google Scholar] [CrossRef] [Green Version]
- Suresh, S.N.; Chavalmane, A.K.; Pillai, M.; Ammanathan, V.; Vidyadhara, D.J.; Yarreiphang, H.; Rai, S.; Paul, A.; Clement, J.P.; Alladi, P.A.; et al. Modulation of Autophagy by a Small Molecule Inverse Agonist of ERRalpha Is Neuroprotective. Front. Mol. Neurosci. 2018, 11, 109. [Google Scholar] [CrossRef] [Green Version]
- Sonoda, J.; Laganiere, J.; Mehl, I.R.; Barish, G.D.; Chong, L.W.; Li, X.; Scheffler, I.E.; Mock, D.C.; Bataille, A.R.; Robert, F.; et al. Nuclear receptor ERR alpha and coactivator PGC-1 beta are effectors of IFN-gamma-induced host defense. Genes Dev. 2007, 21, 1909–1920. [Google Scholar] [CrossRef] [Green Version]
- Hwang, J.; Purdy, J.G.; Wu, K.; Rabinowitz, J.D.; Shenk, T. Estrogen-related receptor alpha is required for efficient human cytomegalovirus replication. Proc. Natl. Acad. Sci. USA 2014, 111, E5706–E5715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, X.; Ma, S.; Tian, Y.; Wei, C.; Zhu, Y.; Li, F.; Zhang, P.; Wang, P.; Zhang, Y.; Zhong, H. ERRalpha negatively regulates type I interferon induction by inhibiting TBK1-IRF3 interaction. PLoS Pathog. 2017, 13, e1006347. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.Q.; Lee, J.N.; Son, M.; Lim, J.Y.; Dutta, R.K.; Maharjan, Y.; Kwak, S.; Oh, G.T.; Byun, K.; Choe, S.K.; et al. Ciliogenesis is reciprocally regulated by PPARA and NR1H4/FXR through controlling autophagy in vitro and in vivo. Autophagy 2018, 14, 1011–1027. [Google Scholar] [CrossRef] [PubMed]
- Jiao, M.; Ren, F.; Zhou, L.; Zhang, X.; Zhang, L.; Wen, T.; Wei, L.; Wang, X.; Shi, H.; Bai, L.; et al. Peroxisome proliferator-activated receptor alpha activation attenuates the inflammatory response to protect the liver from acute failure by promoting the autophagy pathway. Cell Death Dis. 2014, 5, e1397. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Chen, X.; Qu, N.; Zhang, B.; Xia, C. Chondroprotection of PPARalpha activation by WY14643 via autophagy involving Akt and ERK in LPS-treated mouse chondrocytes and osteoarthritis model. J. Cell Mol. Med. 2019, 23, 2782–2793. [Google Scholar] [CrossRef] [Green Version]
- Saito, T.; Kuma, A.; Sugiura, Y.; Ichimura, Y.; Obata, M.; Kitamura, H.; Okuda, S.; Lee, H.C.; Ikeda, K.; Kanegae, Y.; et al. Autophagy regulates lipid metabolism through selective turnover of NCoR1. Nat. Commun. 2019, 10, 1567. [Google Scholar] [CrossRef] [Green Version]
- Visvikis, O.; Ihuegbu, N.; Labed, S.A.; Luhachack, L.G.; Alves, A.F.; Wollenberg, A.C.; Stuart, L.M.; Stormo, G.D.; Irazoqui, J.E. Innate host defense requires TFEB-mediated transcription of cytoprotective and antimicrobial genes. Immunity 2014, 40, 896–909. [Google Scholar] [CrossRef] [Green Version]
- Chandra, S.; Roy, A.; Patel, D.R.; Pahan, K. PPARalpha Between Aspirin and Plaque Clearance. J. Alzheimers Dis. 2019, 71, 389–397. [Google Scholar] [CrossRef]
- Chandra, S.; Roy, A.; Jana, M.; Pahan, K. Cinnamic acid activates PPARalpha to stimulate Lysosomal biogenesis and lower Amyloid plaque pathology in an Alzheimer’s disease mouse model. Neurobiol. Dis. 2019, 124, 379–395. [Google Scholar] [CrossRef]
- Chandra, S.; Jana, M.; Pahan, K. Aspirin Induces Lysosomal Biogenesis and Attenuates Amyloid Plaque Pathology in a Mouse Model of Alzheimer’s Disease via PPARalpha. J. Neurosci. 2018, 38, 6682–6699. [Google Scholar] [CrossRef]
- Ghosh, A.; Jana, M.; Modi, K.; Gonzalez, F.J.; Sims, K.B.; Berry-Kravis, E.; Pahan, K. Activation of peroxisome proliferator-activated receptor alpha induces lysosomal biogenesis in brain cells: Implications for lysosomal storage disorders. J. Biol. Chem. 2015, 290, 10309–10324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, V.; Makhdoomi, M.; Singh, L.; Kumar, P.; Khan, N.; Singh, S.; Verma, H.N.; Luthra, K.; Sarkar, S.; Kumar, D. Trehalose limits opportunistic mycobacterial survival during HIV co-infection by reversing HIV-mediated autophagy block. Autophagy 2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rusmini, P.; Cortese, K.; Crippa, V.; Cristofani, R.; Cicardi, M.E.; Ferrari, V.; Vezzoli, G.; Tedesco, B.; Meroni, M.; Messi, E.; et al. Trehalose induces autophagy via lysosomal-mediated TFEB activation in models of motoneuron degeneration. Autophagy 2019, 15, 631–651. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.H.; Jasuja, R.; Kumar, R. Trehalose induces functionally active conformation in the intrinsically disordered N-terminal domain of glucocorticoid receptor. J. Biomol. Struct Dyn. 2017, 35, 2248–2256. [Google Scholar] [CrossRef]
- Palomer, X.; Capdevila-Busquets, E.; Botteri, G.; Salvado, L.; Barroso, E.; Davidson, M.M.; Michalik, L.; Wahli, W.; Vazquez-Carrera, M. PPARbeta/delta attenuates palmitate-induced endoplasmic reticulum stress and induces autophagic markers in human cardiac cells. Int. J. Cardiol. 2014, 174, 110–118. [Google Scholar] [CrossRef]
- Busch, D.; Kapoor, A.; Rademann, P.; Hildebrand, F.; Bahrami, S.; Thiemermann, C.; Osuchowski, M.F. Delayed activation of PPAR-beta/delta improves long-term survival in mouse sepsis: Effects on organ inflammation and coagulation. Innate Immun. 2018, 24, 262–273. [Google Scholar] [CrossRef] [Green Version]
- Kapoor, A.; Shintani, Y.; Collino, M.; Osuchowski, M.F.; Busch, D.; Patel, N.S.; Sepodes, B.; Castiglia, S.; Fantozzi, R.; Bishop-Bailey, D.; et al. Protective role of peroxisome proliferator-activated receptor-beta/delta in septic shock. Am. J. Respir. Crit. Care Med. 2010, 182, 1506–1515. [Google Scholar] [CrossRef]
- Kim, T.H.; Kim, H.K.; Hwang, E.S. Novel anti-adipogenic activity of anti-malarial amodiaquine through suppression of PPARgamma activity. Arch. Pharm. Res. 2017, 40, 1336–1343. [Google Scholar] [CrossRef]
- Penas, F.N.; Carta, D.; Cevey, A.C.; Rada, M.J.; Pieralisi, A.V.; Ferlin, M.G.; Sales, M.E.; Mirkin, G.A.; Goren, N.B. Pyridinecarboxylic Acid Derivative Stimulates Pro-Angiogenic Mediators by PI3K/AKT/mTOR and Inhibits Reactive Nitrogen and Oxygen Species and NF-kappaB Activation Through a PPARgamma-Dependent Pathway in T. cruzi-Infected Macrophages. Front. Immunol. 2019, 10, 2955. [Google Scholar] [CrossRef]
- Omeragic, A.; Saikali, M.F.; Currier, S.; Volsky, D.J.; Cummins, C.L.; Bendayan, R. Selective peroxisome proliferator-activated receptor-gamma modulator, INT131 exhibits anti-inflammatory effects in an EcoHIV mouse model. FASEB J. 2020, 34, 1996–2010. [Google Scholar] [CrossRef] [Green Version]
- Raghuram, S.; Stayrook, K.R.; Huang, P.; Rogers, P.M.; Nosie, A.K.; McClure, D.B.; Burris, L.L.; Khorasanizadeh, S.; Burris, T.P.; Rastinejad, F. Identification of heme as the ligand for the orphan nuclear receptors REV-ERBalpha and REV-ERBbeta. Nat. Struct. Mol. Biol. 2007, 14, 1207–1213. [Google Scholar] [CrossRef] [Green Version]
- Pastore, N.; Vainshtein, A.; Herz, N.J.; Huynh, T.; Brunetti, L.; Klisch, T.J.; Mutarelli, M.; Annunziata, P.; Kinouchi, K.; Brunetti-Pierri, N.; et al. Nutrient-sensitive transcription factors TFEB and TFE3 couple autophagy and metabolism to the peripheral clock. EMBO J. 2019, 38. [Google Scholar] [CrossRef] [PubMed]
- Sulli, G.; Rommel, A.; Wang, X.; Kolar, M.J.; Puca, F.; Saghatelian, A.; Plikus, M.V.; Verma, I.M.; Panda, S. Pharmacological activation of REV-ERBs is lethal in cancer and oncogene-induced senescence. Nature 2018, 553, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Zhang, F.; Ye, Q.; Wang, H. The circadian clock regulates autophagy directly through the nuclear hormone receptor Nr1d1/Rev-erbalpha and indirectly via Cebpb/(C/ebpbeta) in zebrafish. Autophagy 2016, 12, 1292–1309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandra, V.; Bhagyaraj, E.; Nanduri, R.; Ahuja, N.; Gupta, P. NR1D1 ameliorates Mycobacterium tuberculosis clearance through regulation of autophagy. Autophagy 2015, 11, 1987–1997. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Mei, C.; Ercolani, L.; Parodi, C.; Veronesi, M.; Lo Vecchio, C.; Bottegoni, G.; Torrente, E.; Scarpelli, R.; Marotta, R.; Ruffili, R.; et al. Dual inhibition of REV-ERBbeta and autophagy as a novel pharmacological approach to induce cytotoxicity in cancer cells. Oncogene 2015, 34, 2597–2608. [Google Scholar] [CrossRef]
- Woldt, E.; Sebti, Y.; Solt, L.A.; Duhem, C.; Lancel, S.; Eeckhoute, J.; Hesselink, M.K.; Paquet, C.; Delhaye, S.; Shin, Y.; et al. Rev-erb-alpha modulates skeletal muscle oxidative capacity by regulating mitochondrial biogenesis and autophagy. Nat. Med. 2013, 19, 1039–1046. [Google Scholar] [CrossRef]
- Costantini, C.; Renga, G.; Sellitto, F.; Borghi, M.; Stincardini, C.; Pariano, M.; Zelante, T.; Chiarotti, F.; Bartoli, A.; Mosci, P.; et al. Microbes in the Era of Circadian Medicine. Front. Cell Infect. Microbiol. 2020, 10, 30. [Google Scholar] [CrossRef]
- Tognini, P.; Thaiss, C.A.; Elinav, E.; Sassone-Corsi, P. Circadian Coordination of Antimicrobial Responses. Cell Host Microbe 2017, 22, 185–192. [Google Scholar] [CrossRef]
- Man, K.; Loudon, A.; Chawla, A. Immunity around the clock. Science 2016, 354, 999–1003. [Google Scholar] [CrossRef] [Green Version]
- Isakson, P.; Bjoras, M.; Boe, S.O.; Simonsen, A. Autophagy contributes to therapy-induced degradation of the PML/RARA oncoprotein. Blood 2010, 116, 2324–2331. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.W.; Chen, Z.H.; Zhang, X.J.; Han, B.W.; Lin, K.Y.; Li, X.J.; Wei, P.P.; Zhang, H.; Li, Y.; Chen, Y.Q. MIR125B1 represses the degradation of the PML-RARA oncoprotein by an autophagy-lysosomal pathway in acute promyelocytic leukemia. Autophagy 2014, 10, 1726–1737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eriksen, A.B.; Torgersen, M.L.; Holm, K.L.; Abrahamsen, G.; Spurkland, A.; Moskaug, J.O.; Simonsen, A.; Blomhoff, H.K. Retinoic acid-induced IgG production in TLR-activated human primary B cells involves ULK1-mediated autophagy. Autophagy 2015, 11, 460–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Z.H.; Wang, W.T.; Huang, W.; Fang, K.; Sun, Y.M.; Liu, S.R.; Luo, X.Q.; Chen, Y.Q. The lncRNA HOTAIRM1 regulates the degradation of PML-RARA oncoprotein and myeloid cell differentiation by enhancing the autophagy pathway. Cell Death Differ. 2017, 24, 212–224. [Google Scholar] [CrossRef]
- Brigger, D.; Schlafli, A.M.; Garattini, E.; Tschan, M.P. Activation of RARalpha induces autophagy in SKBR3 breast cancer cells and depletion of key autophagy genes enhances ATRA toxicity. Cell Death Dis. 2015, 6, e1861. [Google Scholar] [CrossRef] [Green Version]
- Marchwicka, A.; Cebrat, M.; Laszkiewicz, A.; Sniezewski, L.; Brown, G.; Marcinkowska, E. Regulation of vitamin D receptor expression by retinoic acid receptor alpha in acute myeloid leukemia cells. J. Steroid Biochem. Mol. Biol. 2016, 159, 121–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, J.S.; De Veaux, N.; Rives, A.W.; Lahaye, X.; Lucas, S.Y.; Perot, B.P.; Luka, M.; Garcia-Paredes, V.; Amon, L.M.; Watters, A.; et al. A Comprehensive Map of the Monocyte-Derived Dendritic Cell Transcriptional Network Engaged upon Innate Sensing of HIV. Cell Rep. 2020, 30, 914–931 e919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gutierrez, J.; Garcia-Villa, E.; Ocadiz-Delgado, R.; Cortes-Malagon, E.M.; Vazquez, J.; Roman-Rosales, A.; Alvarez-Rios, E.; Celik, H.; Romano, M.C.; Uren, A.; et al. Human papillomavirus type 16 E7 oncoprotein upregulates the retinoic acid receptor-beta expression in cervical cancer cell lines and K14E7 transgenic mice. Mol. Cell Biochem. 2015, 408, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, Q.; Zhou, Z.; Liu, M.; Chen, Y.; Li, J.; Xu, L.; Guo, J.; Li, Q.; Yang, J.; et al. Retinoic acid receptor beta, a potential therapeutic target in the inhibition of adenovirus replication. Antiviral Res. 2018, 152, 84–93. [Google Scholar] [CrossRef]
- Fan, J.; Lv, Z.; Yang, G.; Liao, T.T.; Xu, J.; Wu, F.; Huang, Q.; Guo, M.; Hu, G.; Zhou, M.; et al. Retinoic Acid Receptor-Related Orphan Receptors: Critical Roles in Tumorigenesis. Front. Immunol. 2018, 9, 1187. [Google Scholar] [CrossRef]
- Jetten, A.M. Retinoid-related orphan receptors (RORs): Critical roles in development, immunity, circadian rhythm, and cellular metabolism. Nucl. Recept. Signal. 2009, 7, e003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friesenhagen, J.; Viemann, D.; Borgeling, Y.; Schmolke, M.; Spiekermann, C.; Kirschnek, S.; Ludwig, S.; Roth, J. Highly pathogenic influenza viruses inhibit inflammatory response in monocytes via activation of rar-related orphan receptor RORalpha. J. Innate Immun. 2013, 5, 505–518. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Zhao, Y.; Xu, L.; Gao, L.; Su, Y.; Lin, N.; Pu, J. The nuclear melatonin receptor RORalpha is a novel endogenous defender against myocardial ischemia/reperfusion injury. J. Pineal Res. 2016, 60, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xu, L.; Ding, S.; Lin, N.; Ji, Q.; Gao, L.; Su, Y.; He, B.; Pu, J. Novel protective role of the circadian nuclear receptor retinoic acid-related orphan receptor-alpha in diabetic cardiomyopathy. J. Pineal Res. 2017, 62. [Google Scholar] [CrossRef]
- Mirza-Aghazadeh-Attari, M.; Mohammadzadeh, A.; Adib, A.; Darband, S.G.; Sadighparvar, S.; Mihanfar, A.; Majidinia, M.; Yousefi, B. Melatonin-mediated regulation of autophagy: Making sense of double-edged sword in cancer. J. Cell Physiol. 2019, 234, 17011–17022. [Google Scholar] [CrossRef]
- Boga, J.A.; Caballero, B.; Potes, Y.; Perez-Martinez, Z.; Reiter, R.J.; Vega-Naredo, I.; Coto-Montes, A. Therapeutic potential of melatonin related to its role as an autophagy regulator: A review. J. Pineal Res. 2019, 66, e12534. [Google Scholar] [CrossRef] [Green Version]
- Brazao, V.; Santello, F.H.; Colato, R.P.; do Prado, J.C., Jr. T. cruzi infection among aged rats: Melatonin as a promising therapeutic molecule. Exp. Gerontol. 2020, 135, 110922. [Google Scholar] [CrossRef]
- Daryani, A.; Montazeri, M.; Pagheh, A.S.; Sharif, M.; Sarvi, S.; Hosseinzadeh, A.; Reiter, R.J.; Hadighi, R.; Joghataei, M.T.; Ghaznavi, H.; et al. The potential use of melatonin to treat protozoan parasitic infections: A review. Biomed. Pharmacother. 2018, 97, 948–957. [Google Scholar] [CrossRef]
- Hu, W.; Deng, C.; Ma, Z.; Wang, D.; Fan, C.; Li, T.; Di, S.; Gong, B.; Reiter, R.J.; Yang, Y. Utilizing melatonin to combat bacterial infections and septic injury. Br. J. Pharmacol. 2017, 174, 754–768. [Google Scholar] [CrossRef]
- Vielma, J.R.; Bonilla, E.; Chacin-Bonilla, L.; Mora, M.; Medina-Leendertz, S.; Bravo, Y. Effects of melatonin on oxidative stress, and resistance to bacterial, parasitic, and viral infections: A review. Acta Trop. 2014, 137, 31–38. [Google Scholar] [CrossRef]
- Anderson, G.; Reiter, R.J. Melatonin: Roles in influenza, Covid-19, and other viral infections. Rev. Med. Virol. 2020, 30, e2109. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.; Lei, H.; He, M.; Gong, R.; Wang, Y.; He, X.; Li, G.; Pang, P.; Li, X.; Yu, S.; et al. Melatonin triggers autophagic cell death by regulating RORC in Hodgkin lymphoma. Biomed. Pharmacother. 2020, 123, 109811. [Google Scholar] [CrossRef] [PubMed]
- Preidis, G.A.; Kim, K.H.; Moore, D.D. Nutrient-sensing nuclear receptors PPARalpha and FXR control liver energy balance. J. Clin. Investig. 2017, 127, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Panzitt, K.; Jungwirth, E.; Krones, E.; Lee, J.M.; Pollheimer, M.; Thallinger, G.G.; Kolb-Lenz, D.; Xiao, R.; Thorell, A.; Trauner, M.; et al. FXR-dependent Rubicon induction impairs autophagy in models of human cholestasis. J. Hepatol. 2020, 72, 1122–1131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khambu, B.; Li, T.; Yan, S.; Yu, C.; Chen, X.; Goheen, M.; Li, Y.; Lin, J.; Cummings, O.W.; Lee, Y.A.; et al. Hepatic Autophagy Deficiency Compromises Farnesoid X Receptor Functionality and Causes Cholestatic Injury. Hepatology 2019, 69, 2196–2213. [Google Scholar] [CrossRef] [Green Version]
- Endo-Umeda, K.; Makishima, M. Liver X Receptors Regulate Cholesterol Metabolism and Immunity in Hepatic Nonparenchymal Cells. Int. J. Mol. Sci. 2019, 20, 5045. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Qiu, D.K.; Ma, X. Liver X receptors bridge hepatic lipid metabolism and inflammation. J. Dig. Dis. 2012, 13, 69–74. [Google Scholar] [CrossRef]
- Zeng, J.; Wu, D.; Hu, H.; Young, J.A.T.; Yan, Z.; Gao, L. Activation of the liver X receptor pathway inhibits HBV replication in primary human hepatocytes. Hepatology 2020. [Google Scholar] [CrossRef]
- Lange, P.T.; Jondle, C.N.; Darrah, E.J.; Johnson, K.E.; Tarakanova, V.L. LXR Alpha Restricts Gammaherpesvirus Reactivation from Latently Infected Peritoneal Cells. J. Virol. 2019, 93. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Wei, Z.; Zhang, Y.; Ma, X.; Chen, Y.; Yu, M.; Ma, C.; Li, X.; Cao, Y.; Liu, J.; et al. Activation of liver X receptor plays a central role in antiviral actions of 25-hydroxycholesterol. J. Lipid Res. 2018, 59, 2287–2296. [Google Scholar] [CrossRef] [Green Version]
- Ahsan, F.; Maertzdorf, J.; Guhlich-Bornhof, U.; Kaufmann, S.H.E.; Moura-Alves, P. IL-36/LXR axis modulates cholesterol metabolism and immune defense to Mycobacterium tuberculosis. Sci. Rep. 2018, 8, 1520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matalonga, J.; Glaria, E.; Bresque, M.; Escande, C.; Carbo, J.M.; Kiefer, K.; Vicente, R.; Leon, T.E.; Beceiro, S.; Pascual-Garcia, M.; et al. The Nuclear Receptor LXR Limits Bacterial Infection of Host Macrophages through a Mechanism that Impacts Cellular NAD Metabolism. Cell Rep. 2017, 18, 1241–1255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silvente-Poirot, S.; Segala, G.; Poirot, M.C.; Poirot, M. Ligand-dependent transcriptional induction of lethal autophagy: A new perspective for cancer treatment. Autophagy 2018, 14, 555–557. [Google Scholar] [CrossRef] [Green Version]
- Segala, G.; David, M.; de Medina, P.; Poirot, M.C.; Serhan, N.; Vergez, F.; Mougel, A.; Saland, E.; Carayon, K.; Leignadier, J.; et al. Dendrogenin A drives LXR to trigger lethal autophagy in cancers. Nat. Commun. 2017, 8, 1903. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.A.; Singh, B.K.; Zhou, J.; Wu, Y.; Farah, B.L.; Ohba, K.; Lesmana, R.; Gooding, J.; Bay, B.H.; Yen, P.M. Thyroid hormone induction of mitochondrial activity is coupled to mitophagy via ROS-AMPK-ULK1 signaling. Autophagy 2015, 11, 1341–1357. [Google Scholar] [CrossRef] [Green Version]
- Cioffi, F.; Senese, R.; Lanni, A.; Goglia, F. Thyroid hormones and mitochondria: With a brief look at derivatives and analogues. Mol. Cell Endocrinol. 2013, 379, 51–61. [Google Scholar] [CrossRef]
- Sinha, R.A.; You, S.H.; Zhou, J.; Siddique, M.M.; Bay, B.H.; Zhu, X.; Privalsky, M.L.; Cheng, S.Y.; Stevens, R.D.; Summers, S.A.; et al. Thyroid hormone stimulates hepatic lipid catabolism via activation of autophagy. J. Clin. Investig. 2012, 122, 2428–2438. [Google Scholar] [CrossRef] [Green Version]
- Tseng, Y.H.; Ke, P.Y.; Liao, C.J.; Wu, S.M.; Chi, H.C.; Tsai, C.Y.; Chen, C.Y.; Lin, Y.H.; Lin, K.H. Chromosome 19 open reading frame 80 is upregulated by thyroid hormone and modulates autophagy and lipid metabolism. Autophagy 2014, 10, 20–31. [Google Scholar] [CrossRef] [Green Version]
- Chi, H.C.; Chen, S.L.; Lin, S.L.; Tsai, C.Y.; Chuang, W.Y.; Lin, Y.H.; Huang, Y.H.; Tsai, M.M.; Yeh, C.T.; Lin, K.H. Thyroid hormone protects hepatocytes from HBx-induced carcinogenesis by enhancing mitochondrial turnover. Oncogene 2017, 36, 5274–5284. [Google Scholar] [CrossRef]
- Tao, S.; Drexler, I. Targeting Autophagy in Innate Immune Cells: Angel or Demon during Infection and Vaccination? Front. Immunol. 2020, 11, 460. [Google Scholar] [CrossRef] [Green Version]
- Felzen, V.; Hiebel, C.; Koziollek-Drechsler, I.; Reissig, S.; Wolfrum, U.; Kogel, D.; Brandts, C.; Behl, C.; Morawe, T. Estrogen receptor alpha regulates non-canonical autophagy that provides stress resistance to neuroblastoma and breast cancer cells and involves BAG3 function. Cell Death Dis. 2015, 6, e1812. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Ligand/Activator | Pathogen/Disease | Study Model | Autophagy | Effects | Mechanism of Action | Ref. |
|---|---|---|---|---|---|---|
| Bacterial/Fungal infections | ||||||
| Vit-D | H. pylori | Human gastric epithelial cell lines, clinical specimens | ↑ | Bacterial eradication | Activation of PDIA3 receptor and nuclear translocation of PDIA3-STAT3 complex to induce autophagosomal degradation independent of VDR | [106] |
| Mtb | Human primary monocytes, MDMs, THP-1, and RAW264.7 cells | ↑ | Antimicrobial | Beclin-1 and Atg-5 activation mediated through hCAP-18/LL-37-dependent MAPK and C/EBPβ activation | [107] | |
| PGE2, human macrophages | ↓ | Intracellular Mtb survival | PGE2 inhibits hCAP18/LL-37 expression and vitamin D-induced cathelicidin and autophagy by dampening expression of VDR | [114] | ||
| Mouse model | ↑ | Antimicrobial | Vit-D supplementation on 2nd-line anti-TB therapy leads to suppression of MMP1 Induction of VDR, CRAMP, LC3B, and caspase-3 | [115] | ||
| IFN-γ, human macrophages | ↑ | Antimicrobial | Vitamin D-dependent autophagy and autophagolysosomal fusion by IFN-γ VDR-dependent induction of cathelicidin and DEFB4 by IFN-γ | [121] | ||
| A. fumigatus | Alveolar macrophages | ↓ | Antimicrobial | Delayed formation of lysosomes against infection Modulation of Dectin-1, ROS, and LC3 expression | [127] | |
| M. marinum | Human monocytes (THP-1) | ↑ | Antibacterial | Induction of endogenous CAMP and its colocalization with autophagolysosome | [110] | |
| Vit-D-sufficient human serum | Mtb | CD40L, IFN-γ, human PBMC | ↑ | Antimicrobial | Induction of CYP27B1, VDR, cathelicidin, and DEFB4 | [122] |
| - | Mtb lipoprotein LpqH | Human primary monocytes | ↑ | Antibacterial | TLR2/1/CD14-mediated (C/EBP)-β-dependent induction of CYP27B1 | [109] |
| PBA+Vit-D | Mtb | PBMCs and MDMs from TB patients | ↑ | Antimicrobial | Increased LC3 expression, decreased XBP1spl mRNA | [116] |
| TB patients | Clinical trial | - | Antimicrobial | Increased LL-37 | [117] | |
| Clinical trial | - | Clinical recovery | Increased serum Vit-D levels after PBA+Vit-D supplementation | [118] | ||
| LAB | S. enterica | HCT116, MEFs cell lines, in vivo mice | ↑ | Anti-inflammatory | Enhanced expression of Beclin-1 and ATG16L1 Increased expression of VDR and cathelicidin | [126] |
| Viral infections | ||||||
| Vit-D | HIV | Human MDMs | ↑ | Inhibition of virus replication | PI3K-, ATG-5-, and Beclin-1-dependent autophagy activation | [111] |
| HIV, Mtb | Human MDMs | ↑ | Inhibition of virus replication and mycobacterial growth | Induced expression of CAMP | [112] | |
| Influenza A | A549 cell lines | ↑ | Antiviral | Restoration of virus-induced inhibition of autophagic flux through Syntaxin-17 and ATP6V0A2 | [125] | |
| Rotavirus | Pigs, IPEC-J2 cells | ↑ | Inhibition of virus infection | Regulation of autophagic maturation and expression of porcine cathelicidin genes | [128] | |
| TLR8 ligands | HIV | Human macrophages | ↑ | Reduced virus replication | Induced expression of CAMP, VDR, and CYP27B1 | [113] |
| NRs | Ligands/Activator | Pathogen/Study Model | Autophagy | Effects | Mechanism of Action | Ref. |
|---|---|---|---|---|---|---|
| ER | Estrogen (E2) | Thyroid cancer patients samples, Nthy-ori 3-1, PTC cell, BCPAP-ERα, MCF-7 cells | ↑ | Tumor cell survival | Generation of ROS, activation of ERK1/2 | [131] |
| Bazedoxifene | Mtb/THP-1 cells | ↑ | Inhibition of intracellular growth of Mtb | Increased ROS and phosphorylation of Akt/mTOR signaling | [90] | |
| ERRα | AICAR | Mtb/BMDMs, RAW264.7, HEK293T cells | ↑ | Antimicrobial host defense | Transcriptional activation of autophagy-related genes, and post transcriptional activation of autophagy through SIRT1 activation | [23] |
| Thyroid hormone | THRB1-HepG2 cells, in vivo mice model | - | Mitophagy induction | Increased ESRRA expression via PPARGC1A Induction of ULK1 mRNA and protein through ESRRA-dependent transcription | [138] | |
| XCT 790 | SH-SY5Y, HeLa cells, in vivo mice | ↑ | Neuroprotective | Regulation of autophagy by ERRα through its localization with autophagosome | [139] |
| NRs | Ligands/Activator | Pathogen | Pathogen/Study Model | Autophagy | Effects | Mechanism of Action | Ref. |
|---|---|---|---|---|---|---|---|
| PPARα | GW7647, Wy14643 | Mtb | BMDMs, in vivo mice | ↑ | Antimicrobial | Increased expression and nuclear translocation of TFEB | [72] |
| Gemfibrozil | M. abscessus | BMDMs, in vivo mice | ↑ | Antimicrobial | Increased nuclear translocation of TFEB | [70] | |
| PPARβ/δ | GW501516 | - | Human cardiac AC16 cells, in vivo mice | ↑ | Inhibition of palmitate induced ER stress | Upregulation of Beclin-1 and LC3II | [155] |
| PPARγ | HP24 | T. cruzi | Peritoneal macrophages, in vivo mice | - | Pro-angiogenic and anti-inflammatory | Induction of pro-angiogenic mediators (eNOS and VEGF-A) through PI3K/Akt/mTOR and PPARγ pathway Inhibition of NF-κB pathway in PPARγ-dependent manner | [159] |
| INT131 | EcoHIV | Primary mouse glial cells, in vivo mice | - | Anti-inflammatory | Inhibition of proinflammatory cytokines | [160] |
| NRs | Ligands/Activator | Pathogen/Study Model | Autophagy | Effects | Mechanism of Action | Ref. |
|---|---|---|---|---|---|---|
| REV-ERBα | GSK4122 | Zebrafish | Rhythmic with circadian clock | Regulation of autophagy rhythms | Direct regulation of NR1D1 and CEBPB by nutritional signals and circadian clock | [164] |
| - | Mouse model, MEF, Hepa1-6 and HEK293 cells | Rhythmic with circadian clock | Regulation of autophagy rhythms | Autophagy activation through TFEB and TFE3; repression by REV-ERBα | [162] | |
| GSK4112 | Mtb/Human macrophages | ↑ | Antimicrobial | Modulation of LAMP1 and TFEB, repression of IL10 | [165] | |
| SR9009, SR9011 | Cancer cell lines, human glioblastoma stem cells, in vivo mice | ↓ | Anticancer | REV-ERB agonist inhibit autophagy (decreased LC3, increased p62, and increased LAMP1) and de novo lipogenesis to induce apoptotic responses | [163] | |
| in vivo mice, C2C12 myoblasts | ↓ | Improved muscle oxidative function | LKB1-AMPK-SIRT1–PPARGC1A signaling pathway | [167] | ||
| RARα | ATRA | HeLa, APL NB4 cells | ↑ | Differentiation of APL cells | Inhibition of mTOR pathway to induce autophagy-dependent PML/RARA degradation | [171] |
| APL patients samples, NB4 cell lines | ↑ | Differentiation of APL cells | MIR125B1 overexpression enhanced PML-RARA expression DRAM2 as a target of MIR125B1 | [172] | ||
| RORα | - | HPAIV (H5N1)/human monocytes, | - | Inhibition of inflammatory responses | H5N1 inhibits NF-κB and activates RORα in monocytes | [182] |
| - | MI/R injury mice model | ↑ or preservation of autophagy function | Protection against MI/R injury | Inhibition of ER stress and mitochondrial apoptosis pathway, restoration of autophagy function, reduced oxidative/nitrative stress | [183] | |
| RORγ | Melatonin | Human HL cell line L428 | ↑ | Cell death | Induction of autophagic cell death by melatonin via increased level of RORC | [192] |
| FXR/PPARα | GW4064/GW7647 | Mouse primary hepatocytes, mouse liver | FXR: ↓, PPAR: ↑ | - | FXR and PPAR compete for binding to common sites in autophagic gene promotors, with opposite transcriptional outputs | [22] |
| GW4064/GW7646, Wy14643 | Human RPE cells, MEFs, HK2, A549 cells | FXR: ↓, PPAR: ↑ | FXR represses and PPAR facilitates cliogenesis | Regulation of expression of autophagic genes, FXR acts in opposite way with PPARA | [143] | |
| FXR | Bile acids | Liver tissue from cholestasis patients, HepG2 cells | ↓ | Autophagy and Rubicon could be novel treatment target for cholestatic liver disease | Prevention of proper fusion of autophagolysosome with lysosomes by bile acids, through FXR-dependent induction of Rubicon | [194] |
| GW4064 | In vivo mice with hepatic deletion of Atg7 or Atg5 with or without Nrf2 codeletion | ↓ | Liver injury | NRF2 activation in autophagy deficiency leading to downregulation of FXR, causing cholestasis | [195] | |
| LXR | T0901317, GW3965, LXR-623 | HBV/primary human hepatocytes, HepaRG cells | - | Anti-HBV effects | Inhibition of cholesterol 7α-hydroxylase 1 (CYP7A1) mRNA levels | [198] |
| DDA | Melanoma and AML cell lines, AML patients samples, in vivo mice | ↑ | Anti-tumor | DDA acting as partial agonist on LRX to increase Nur77, Nor1, and LC3 expression | [204] | |
| TR | T3 | HepG2, Huh7 cells | ↑ | Lipid metabolism | Upregulation of C19orf80 expression, which is involved in lipid metabolism through breakdown of lipid droplets | [208] |
| Mice model of hepatocarcinogenesis, HepG2 cells | ↑ | Inhibition of hepatic DNA damage, inflammation, and carcinogenesis | Induction of hepatic PINK1 expression, which ubiquitinates HBx protein to trigger mitophagy | [209] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silwal, P.; Paik, S.; Jeon, S.M.; Jo, E.-K. Nuclear Receptors as Autophagy-Based Antimicrobial Therapeutics. Cells 2020, 9, 1979. https://doi.org/10.3390/cells9091979
Silwal P, Paik S, Jeon SM, Jo E-K. Nuclear Receptors as Autophagy-Based Antimicrobial Therapeutics. Cells. 2020; 9(9):1979. https://doi.org/10.3390/cells9091979
Chicago/Turabian StyleSilwal, Prashanta, Seungwha Paik, Sang Min Jeon, and Eun-Kyeong Jo. 2020. "Nuclear Receptors as Autophagy-Based Antimicrobial Therapeutics" Cells 9, no. 9: 1979. https://doi.org/10.3390/cells9091979
APA StyleSilwal, P., Paik, S., Jeon, S. M., & Jo, E.-K. (2020). Nuclear Receptors as Autophagy-Based Antimicrobial Therapeutics. Cells, 9(9), 1979. https://doi.org/10.3390/cells9091979





