A Systematic Review of Inverse Agonism at Adrenoceptor Subtypes
Abstract
:1. Introduction
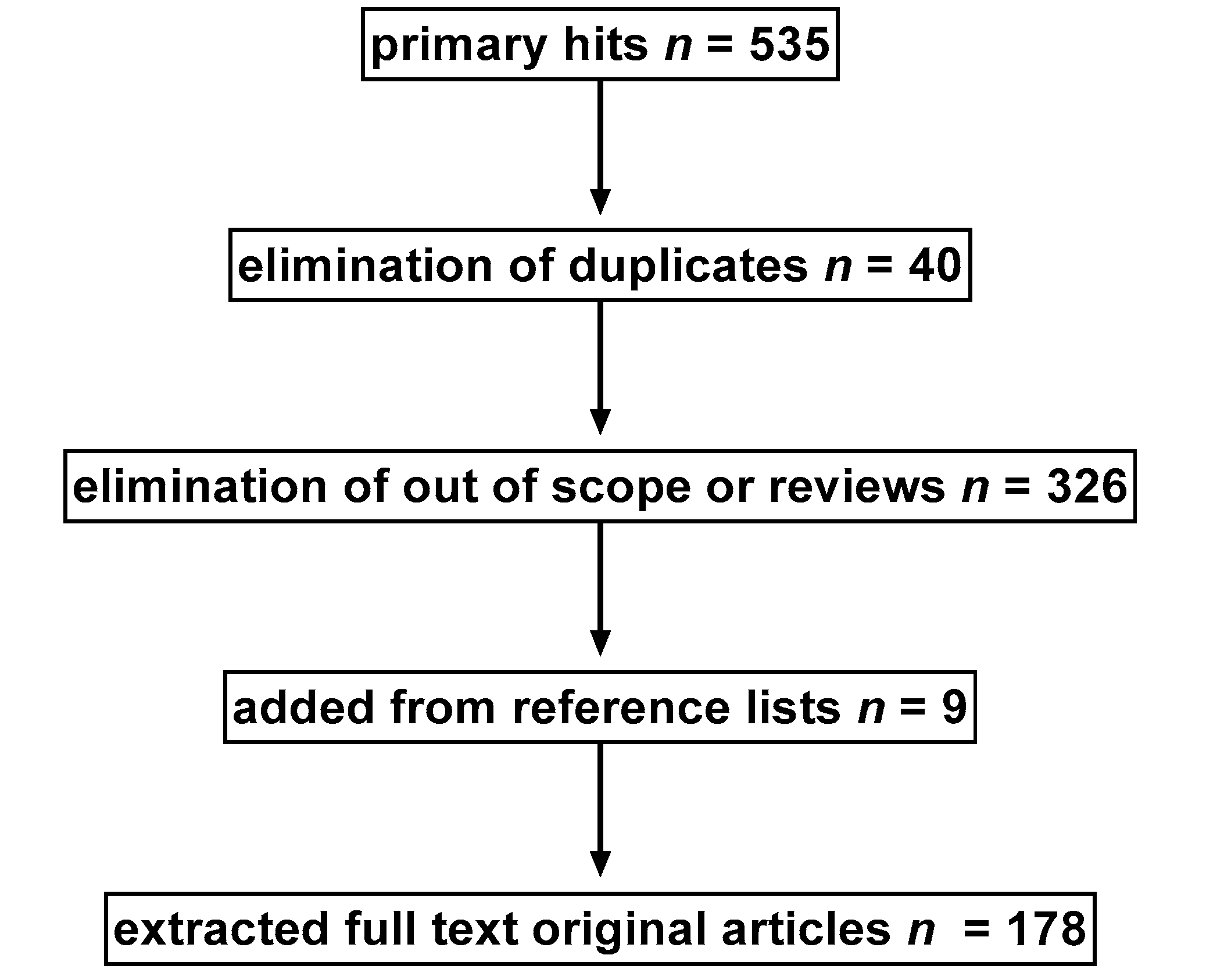
2. Search Strategy
3. Methodological Aspects of Studying IA
4. Compounds Exhibiting IA at AR Subtypes in Cellular Models
4.1. α1-AR
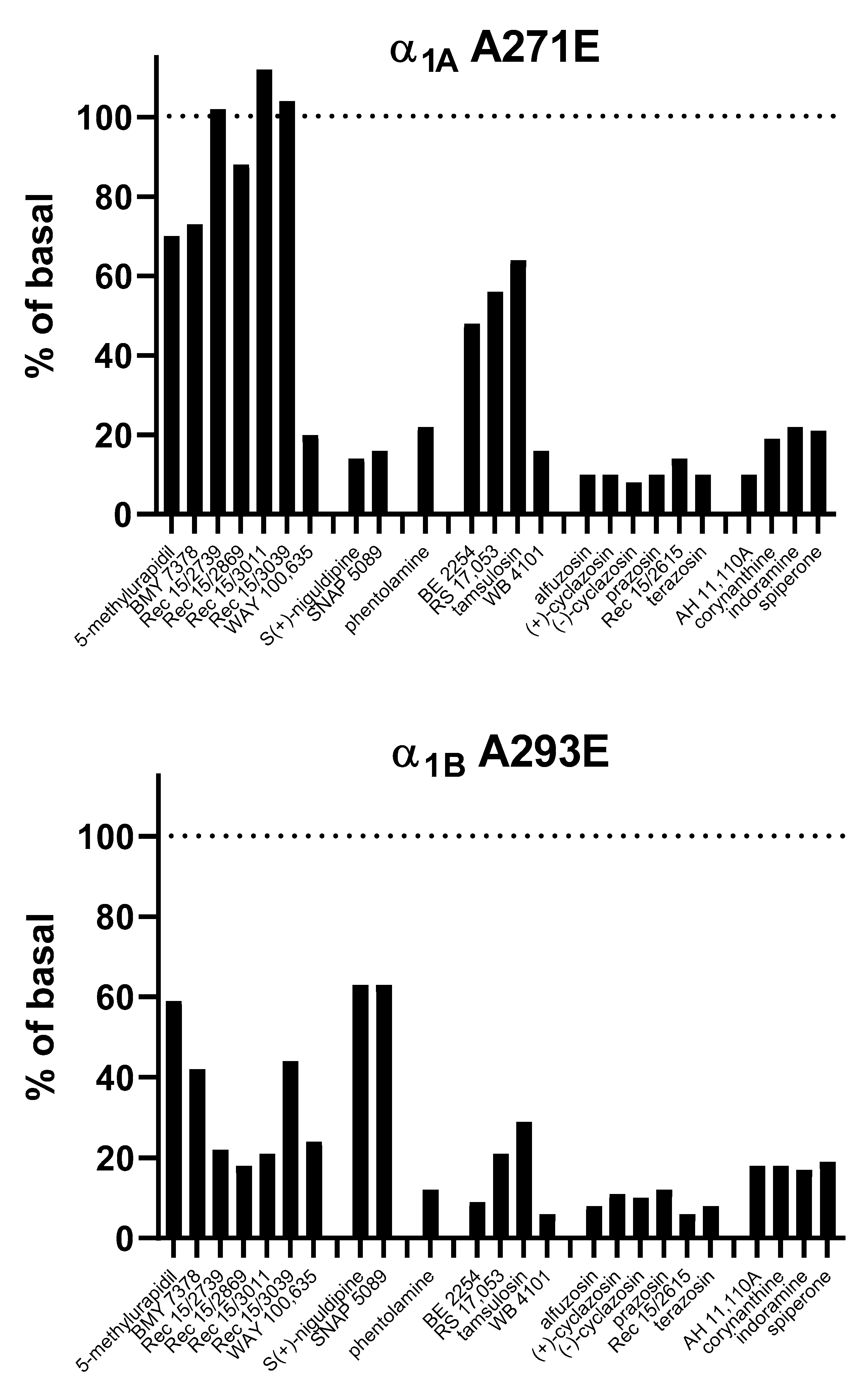
4.1.1. α1A-AR
4.1.2. α1B-AR
4.1.3. α1D-AR
4.2. α2-AR
4.2.1. α2A-AR
4.2.2. α2B-AR
4.2.3. α2C-AR
4.3. β-AR
4.3.1. β1-AR
4.3.2. β2-AR
5. Effects of Compounds with IA Data for Tissue and In Vivo Function
5.1. α1-AR
5.2. α2-AR
5.3. β-AR
5.3.1. Heart
5.3.2. Lung
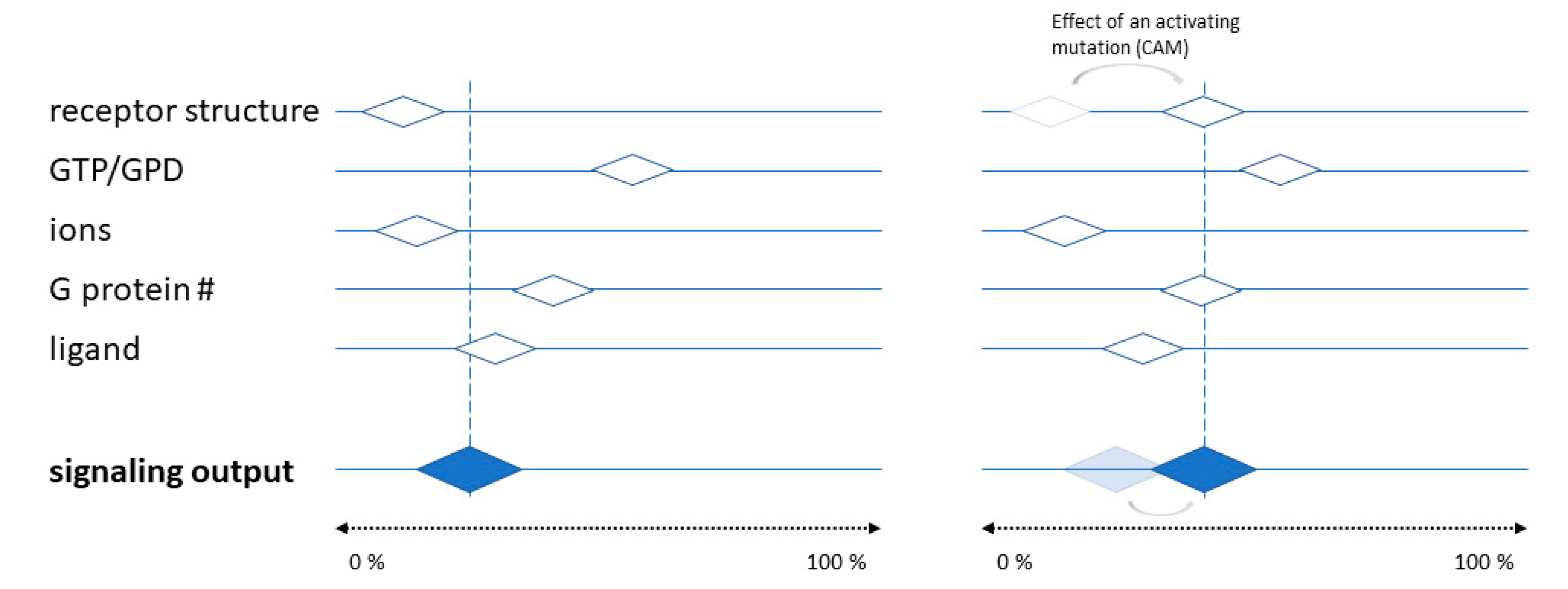
6. Molecular Mechanisms and Structural Basis of Inverse Agonism at AR
7. Conclusions and Implications for Drug Development
Author Contributions
Funding
Conflicts of Interest
References
- Bylund, D.B.; Eikenberg, D.C.; Hieble, J.P.; Langer, S.Z.; Lefkowitz, R.J.; Minneman, K.P.; Molinoff, P.B.; Ruffolo, R.R., Jr.; Trendelenburg, U. International Union of Pharmacology Nomenclature of Adrenoceptors. Pharmacol. Rev. 1994, 46, 121–136. [Google Scholar] [PubMed]
- Hieble, J.P.; Bylund, D.B.; Clarke, D.E.; Eikenburg, D.C.; Langer, S.Z.; Lefkowitz, R.J.; Minneman, K.P.; Ruffolo, R.R., Jr. International Union of Pharmacology X. Recommendation for nomenclature of α1-adrenoceptors: Consensus update. Pharmacol. Rev. 1995, 47, 267–270. [Google Scholar] [PubMed]
- Michel, M.C.; Kenny, B.A.; Schwinn, D.A. Classification of α1-adrenoceptor subtypes. Naunyn Schmiedeberg’s Arch. Pharmacol. 1995, 352, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cotecchia, S.; Exum, S.; Caron, M.G.; Lefkowitz, R.J. Regions of the α1-adrenergic receptor involved in coupling to phosphatidylinositol hydrolysis and enhanced sensitivity of biological function. Proc. Natl. Acad. Sci. USA 1990, 87, 2896–2900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, T.; Cotecchia, S. Historical review: Negative efficacy and the constitutive activity of G-protein-coupled receptors. Trends Pharmacol. Sci. 2005, 26, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Schütz, W.; Freissmuth, M. Reverse intrinsic activity of antagonists on G protein-coupled receptors. Trends Pharmacol. Sci. 1992, 13, 376–380. [Google Scholar] [CrossRef]
- Milligan, G.; Bond, R.A.; Lee, M. Inverse agonism: Pharmacological curiosity or potential therapeutic strategy? Trends Pharmacol. Sci. 1995, 16, 10–13. [Google Scholar] [CrossRef]
- de Ligt, R.A.F.; Kourounakis, A.P.; Ijzerman, A.P. Inverse agonism at G protein-coupled receptors: (patho)physiological relevance and implications for drug discovery. Br. J. Pharmacol. 2000, 130, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Michel, M.C.; Foster, C.; Brunner, H.R.; Liu, L. A systematic comparison of the properties of clinically used angiotensin II type 1 receptor antagonists. Pharmacol. Rev. 2013, 65, 809–848. [Google Scholar] [CrossRef] [Green Version]
- Nelson, C.P.; Nahorski, S.R.; Chaliss, R.A.J. Constitutive activity and inverse agonism at the M2 muscarinic acetylcholine receptor. J. Pharmacol. Exp. Ther. 2006, 316, 279–288. [Google Scholar] [CrossRef]
- Monczor, F.; Fernandez, N.; Fitzsimons, C.P.; Shayo, C.; Davio, C. Antihistaminergics and inverse agonism: Potential therapeutic applications. Eur. J. Pharmacol. 2013, 715, 26–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strange, P.G. Antipsychotic drug action: Antagonism, inverse agonism or partial agonism. Trends Pharmacol. Sci. 2008, 29, 314–321. [Google Scholar] [CrossRef]
- Kenakin, T. Efficacy as a vector: The relative prevalence and paucity of inverse agonism. Mol. Pharmacol. 2004, 65, 2–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galandrin, S.; Oligny-Longpré, G.; Bonin, H.; Ogawa, K.; Galés, C.; Bouvier, M. Conformational rearrangements and signaling cascades involved in ligand-biased mitogen-activated protein kinase signaling through the ß1-adrenergic receptor. Mol. Pharmacol. 2008, 74, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.G.; Hall, I.P.; Hill, S.J. Agonist and inverse agonist actions of ß-blockers at the human ß2-adrenoceptor provide evidence for agonist-directed signaling. Mol. Pharmacol. 2003, 64, 1357–1369. [Google Scholar] [CrossRef]
- Zhu, J.; Taniguchi, T.; Takauji, R.; Suzuki, F.; Tanaka, T.; Muramatsu, I. Inverse agonism and neutral antagonism at a constitutively active alpha-1a adrenoceptor. Br. J. Pharmacol. 2000, 131, 546–552. [Google Scholar]
- Carrillo, J.J.; Stevens, P.A.; Milligan, G. Measurement of agonist-dependent and -independent signal initiation of α1b-adrenoceptor mutants by direct analysis of guanine nucleotide exchange on the G protein Gα11. J. Pharmacol. Exp. Ther. 2002, 302, 1080–1088. [Google Scholar] [CrossRef]
- Pauwels, P.J.; Tardif, S.; Wurch, T.; Colpaert, C.F. Facilitation of constitutive α2A-adrenoceptor activity by both single amino acid mutation (Thr373Lys) and Gαo protein coexpression: Evidence for inverse agonism. J. Pharmacol. Exp. Ther. 2000, 292, 654–663. [Google Scholar]
- Rossier, O.; Abuin, L.; Fanelli, F.; Leonardi, A.; Cotecchia, S. Inverse agonism and neutral antagonism at α1a- and α1b-adrenergic receptor subtypes. Mol. Pharmacol. 1999, 56, 858–866. [Google Scholar] [CrossRef]
- Ammer, H.; Schulz, R. Chronic morphine treatment increases stimulatory beta-2 adrenoceptor signaling in A431 cells stably expressing the Mu opioid receptor. J. Pharmacol. Exp. Ther. 1997, 280, 512–520. [Google Scholar]
- Pauwels, P.J.; Rauly, I.; Wurch, T. Dissimilar pharmacological responses by a new series of imidazoline derivatives at precoupled and ligand-activated α2A-adrenoceptor states: Evidence for effector pathway-dependent differential antagonism. J. Pharmacol. Exp. Ther. 2003, 305, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Wade, S.M.; Lan, K.L.; Moore, D.J.; Neubig, R.R. Inverse agonist activity at the α2A-adrenergic receptor. Mol. Pharmacol. 2001, 59, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.W.; Cotecchia, S.; Milligan, G. Up-regulation of the levels of expression and function of a constitutively active mutant of the hamster α1B-adrenoceptor by ligands that act as inverse agonists. Biochem. J. 1997, 325, 733–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lei, B.; Morris, D.P.; Smith, M.P.; Schwinn, D.A. Lipid rafts constrain basal α1A-adrenergic receptor signaling by maintaining receptor in an inactive conformation. Cell. Signal. 2009, 21, 1532–1539. [Google Scholar] [CrossRef] [PubMed]
- MacEwan, J.D.; Milligan, G. Inverse agonist-induced up-regulation of the human ß2-adrenoceptor in transfected neuroblastoma x glioma hybrid cells. Mol. Pharmacol. 1996, 50, 1479–1486. [Google Scholar] [PubMed]
- Peng, H.; Bond, R.A.; Knoll, B.J. The effects of acute and chronic nadolol treatment on ß2AR signaling in HEK293 cells. Naunyn Schmiedeberg’s Arch. Pharmacol. 2011, 383, 209–216. [Google Scholar] [CrossRef]
- Ramsay, D.; Bevan, N.; Rees, S.; Milligan, G. Detection of receptor ligands by monitoring selective stabilization of a Renilla luciferase-tagged, constitutively active mutant, G-protein-coupled receptor. Br. J. Pharmacol. 2001, 133, 315–323. [Google Scholar] [CrossRef] [Green Version]
- Samama, P.; Pei, G.; Costa, T.; Cotecchia, S.; Lefkowitz, R.J. Negative antagonists promote an inactive conformation of the ß2-adrenergic receptor. Mol. Pharmacol. 1994, 45, 390–394. [Google Scholar]
- Nobles, M.; Benians, A.; Tinker, A. Heterotrimeric G proteins precouple with G protein-coupled receptors in living cells. Proc. Natl. Acad. Sci. USA 2005, 102, 18706–18711. [Google Scholar] [CrossRef] [Green Version]
- Rochais, F.; Vilardaga, J.P.; Nikolaev, V.O.; Bünemann, M.; Lohse, M.J.; Engelhardt, S. Real-time optical recording of ß1-adrenergic receptor activation reveals supersensitivity of the Arg389 variant to carvedilol. J. Clin. Investig. 2007, 117, 229–235. [Google Scholar] [CrossRef] [Green Version]
- Malik, R.U.; Ritt, M.; DeVree, B.T.; Neubig, R.R.; Sunahara, R.K.; Sivaramakrishnan, S. Detection of G protein-selective G protein-coupled receptor (GPCR) conformations in live cells. J. Biol. Chem. 2013, 288, 17167–17178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seifert, R.; Wenzel-Seifert, K.; Lee, T.W.; Gether, U.; Sanders-Bush, E.; Kobilka, B.K. Different effects of Gsα splice variants on ß2-adrenoreceptor-mediated signaling. The ß2-adrenoceptor coupled to the long splice variant of Gsα has properties of a constitutively active receptor. J. Biol. Chem. 1998, 273, 5109–55116. [Google Scholar] [CrossRef] [PubMed]
- Hothersall, J.D.; Black, J.; Caddick, S.; Vinter, J.G.; Tinker, A.; Baker, J.R. The design, synthesis and pharmacological characterization of novel β2-adrenoceptor antagonists. Br. J. Pharmacol. 2011, 164, 317–331. [Google Scholar] [CrossRef] [Green Version]
- McCune, D.F.; Edelmann, S.E.; Olges, J.R.; Post, G.R.; Waldrop, B.A.; Waugh, D.J.J.; Perez, D.M.; Piascik, M.T. Regulation of cellular localization and signaling properties of the α1B- and α1D-adrenoceptors by agonists and inverse agonists. Mol. Pharmacol. 2000, 57, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Sainz, A.J.; Torres-Padilla, M.E. Modulation of basal intracellular calcium by inverse agonists and phorbol myristate acetate in rat-1 fibroblasts stably expressing α1d-adrenoceptors. FEBS Lett. 1999, 443, 277–281. [Google Scholar] [CrossRef] [Green Version]
- Mijares, A.; Lebesgue, D.; Argibay, J.; Hoebeke, J. Anti-peptide antibodies sensitive to the ‘active’ state of the β2adrenergic receptor. FEBS Lett. 1996, 399, 188–191. [Google Scholar] [CrossRef] [Green Version]
- Duffy, S.M.; Cruse, G.; Lawley, W.J.; Bradding, P. β2-Adrenoceptor regulation of the K+ channel iKCa1 in human mast cells. FASEB J. 2005, 19, 1006–1008. [Google Scholar] [CrossRef]
- Saeki, S.; Kunitomo, H.; Narita, Y.; Mimura, H.; Nishi, T.; Sasaki, K. A reporter assay for G-protein-coupled receptors using a B-cell line suitable for stable episomal expression. Anal. Biochem. 2010, 400, 163–172. [Google Scholar] [CrossRef]
- Noguera, M.A.; Ivorra, M.D.; D’Ocon, P. Functional evidence of inverse agonism in vascular smooth muscle. Br. J. Pharmacol. 1996, 119, 158–164. [Google Scholar] [CrossRef] [Green Version]
- Ragnarsson, L.; Wang, C.-I.A.; Andersson, Å.; Fajarningsih, D.; Monks, T.; Brust, A.; Rosengren, K.J.; Lewis, R.J. Conopeptide ρ-TIA defines a new allosteric site on the extracellular surface of the α1B-adrenoceptor. J. Biol. Chem. 2013, 288, 1814–1827. [Google Scholar] [CrossRef] [Green Version]
- Ahn, S.; Kahsai, A.W.; Pani, B.; Wang, Q.-T.; Zhao, S.; Wall, A.L.; Strachan, R.T.; Staus, D.P.; Wingler, L.M.; Sun, L.D.; et al. Allosteric “beta-blocker” isolated from a DNA-encoded small molecule library. Proc. Natl. Acad. Sci. USA 2017, 114, 1708–1713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hopkinson, H.E.; Latif, M.L.; Hill, S.J. Non-competitive antagonism of ß2-agonist-mediated cyclic AMP accumulation by ICI 118,551 in BC3H1 cells endogenously expressing constitutively active ß2-adrenoceptors. Br. J. Pharmacol. 2000, 131, 124–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jansson, C.C.; Kukkonen, J.P.; Näsman, J.; Huifang, G.; Wurster, S.; Virtanen, R.; Savola, J.M.; Cockcroft, V.; Akerman, K.E.O. Protean agonism at α2A-adrenoceptors. Mol. Pharmacol. 1998, 53, 963–968. [Google Scholar] [PubMed]
- Peter, J.-C.; Eftekhari, P.; Billiald, P.; Wallukat, G.; Hoebeke, J. scFv single chain antibody variable fragment as inverse agonist of the β2-adrenergic receptor. J. Biol. Chem. 2003, 278, 36740–36747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scheer, A.; Fanelli, F.; Costa, T.; de Benedetti, P.G.; Cotecchia, S. Constitutitvely active mutants of the α1B-adrenergic receptor, Role of highly conserved polar amino acids in receptor activation. EMBO J. 1996, 15, 3566–3578. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Olli-Lähdesmäki, T.; Kallio, J.; Scheinin, M. α2B-Adrenoceptor levels govern agonist and inverse agonist responses in PC12 cells. Biochem. Biophys. Res. Commun. 2003, 308, 12–18. [Google Scholar] [CrossRef]
- Engelhardt, S.; Grimmer, Y.; Fan, F.H.; Lohse, M.J. Constitutive activity of the human ß1-adrenergic receptor in ß1-receptor transgenic mice. Mol. Pharmacol. 2001, 60, 712–717. [Google Scholar]
- Chidiac, P.; Hebert, T.E.; Valiquette, M.; Dennis, M.; Bouvier, M. Inverse agonist activity of ß-adrenergic antagonists. Mol. Pharmacol. 1994, 45, 490–499. [Google Scholar]
- Small, K.M.; Forbes, S.L.; Rahman, F.F.; Liggett, S.B. Fusion of β2-adrenergic receptor to Gαs in mammalian cells: Identification of a specific signal transduction species not characteristic of constitutive activation or precoupling. Biochemistry 2000, 39, 2815–2821. [Google Scholar] [CrossRef]
- Wurch, T.; Colpaert, F.C.; Pauwels, P.J. G-protein activation by putative antagonists at mutant Thr373Lys α2A adrenergic receptors. Br. J. Pharmacol. 1999, 126, 939–948. [Google Scholar] [CrossRef] [Green Version]
- Stevens, P.A.; Milligan, G. Efficacy of inverse agonists in cells overexpressing a constitutively active ß2-adrenoceptor and type II adenylyl cyclase. Br. J. Pharmacol. 1998, 123, 335–343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seifert, R. Monovalent anions differentially modulate coupling of the β2-adrenoceptor to Gsα splice variants. J. Pharmacol. Exp. Ther. 2001, 298, 840–847. [Google Scholar] [PubMed]
- Ammer, H.; Schulz, R. Opioid tolerance/dependence in neuroblastoma×glioma (NG108-15) hybrid cells is associated with a reduction in spontaneous stimulatory receptor activity. FEBS Lett. 2000, 485, 157–162. [Google Scholar] [CrossRef] [Green Version]
- Rathz, D.A.; Gregory, K.N.; Fang, Y.; Brown, K.M.; Liggett, S.B. Hierarchy of polymorphic variation and desensitization permutations relative to β1- and β2-adrenergic receptor signaling. J. Biol. Chem. 2003, 278, 10784–10789. [Google Scholar] [CrossRef] [Green Version]
- Levin, M.C.; Marullo, S.; Muntaner, O.; Andersson, B.; Magnusson, Y. The myocardium-protective Gly-49 variant of the ß1-adrenergic receptor exhibits constitutive activity and increased desensitization and down-regulation. J. Biol. Chem. 2002, 277, 30429–30435. [Google Scholar] [CrossRef] [Green Version]
- Seifert, R.; Wenzel-Seifert, K. Constitutive activity of G-protein-coupled receptors: Cause of disease and common property of wild-type receptors. Naunyn Schmiedeberg’s Arch. Pharmacol. 2002, 366, 381–416. [Google Scholar] [CrossRef]
- Hein, P.; Goepel, M.; Cotecchia, S.; Michel, M.C. A quantitative analysis of antagonism and inverse agonism at wild-type and constitutively active hamster α1B-adrenoceptors. Naunyn Schmiedeberg’s Arch. Pharmacol. 2001, 363, 34–39. [Google Scholar] [CrossRef]
- Varma, D.R.; Shen, H.; Deng, X.F.; Peri, K.G.; Chemtob, S.; Mulay, S. Inverse agonist activities of ß-adrenoceptor antagonists in rat myocardium. Br. J. Pharmacol. 1999, 127, 895–902. [Google Scholar] [CrossRef] [Green Version]
- Neubig, R.R.; Spedding, M.; Kenakin, T.; Christopoulos, A. International Union of Pharmacology Committee on Receptor Nomenclature and Drug Classification. XXXVIII. Update on terms and symbols in quantitative pharmacology. Pharmacol. Rev. 2003, 55, 597–606. [Google Scholar] [CrossRef] [Green Version]
- Ozkan, M.H.; Uma, S. β-Adrenergic receptor blocker ICI 118,551 selectively increases intermediate-conductance calcium-activated potassium channel (IKCa)-mediated relaxations in rat main mesenteric artery. Basic Clin. Pharmacol. Toxicol. 2018, 122, 570–576. [Google Scholar] [CrossRef] [Green Version]
- Cotecchia, S. Constitutive activity and inverse agonism at the α1adrenoceptors. Biochem. Pharmacol. 2007, 73, 1076–1083. [Google Scholar] [CrossRef] [PubMed]
- Stanasila, L.; Perez, J.-B.; Vogel, H.; Cotecchia, S. Oligomerization of the α1a- and α1b-adrenergic receptor subtypes: Potential implications for internalization. J. Biol. Chem. 2003, 278, 40239–40251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stevens, P.A.; Bevan, N.; Rees, S.; Milligan, G. Resolution of inverse agonist-induced up-regulation from constitutive activitiy mutants of the α1b-adrenoceptor. Mol. Pharmacol. 2000, 58, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.P.; Price, R.R.; Smith, M.P.; Lei, B.; Schwinn, D.A. Cellular trafficking of human α1a-adrenergic receptors is continuous and primarily agonist-independent. Mol. Pharmacol. 2004, 66, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Kolarovszki-Sipiczki, Z.; Gáspár, R.; Ducza, E.; Páldy, E.; Benyhe, S.; Borsodi, A.; Falkay, G. Effect of α1-adrenoceptor subtype-selective inverse agonists on non-pregnant and late-pregnant cervical resistance in vitro in the rat. Clin. Exp. Pharmacol. Physiol. 2007, 34, 42–47. [Google Scholar] [CrossRef]
- Pauwels, P.J.; Tardif, S. Enhanced stability of wild-type and constitutively active α2A-adrenoceptors by ligands with agonist, silent and inverse agonist properties. Naunyn Schmiedeberg’s Arch. Pharmacol. 2002, 166, 134–141. [Google Scholar] [CrossRef]
- Ge, H.; Scheinin, M.; Kallio, J. Constitutive precoupling to Gi and increased agonist potency in the α2B-adrenoceptor. Biochem. Biophys. Res. Commun. 2003, 306, 959–965. [Google Scholar] [CrossRef]
- Vilardaga, J.-P.; Steinmeyer, R.; Harms, G.S.; Lohse, M.J. Molecular basis of inverse agonism in a G protein–coupled receptor. Nat. Chem. Biol. 2005, 1, 25–28. [Google Scholar] [CrossRef]
- Tian, W.N.; Duzic, E.; Lanier, S.M.; Deth, R.C. Determinants of α2-sdrenergic receptor activation of G proteins: Evidence for a precoupled receptor/G protein state. Mol. Pharmacol. 1994, 45, 524–531. [Google Scholar]
- Tian, W.-N.; Miller, D.D.; Deth, R.C. Bidirectional allosteric effects of agonists and GTP at α2A/D-adrenoceptors. J. Pharmacol. Exp. Ther. 2000, 292, 664–671. [Google Scholar]
- Zhu, Q.; Qi, L.J.; Shi, A.; Abou-Samra, A.; Deth, R.C. Protein kinase C regulates α2A/D-adrenoceptor constitutive activity. Pharmacology 2004, 71, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Polanco, M.J.; López-Giménez, J.F.; González-Martín, C.; Alguacil, L.F. Yohimbine does not affect opioid receptor activation but prevents adenylate cyclase regulation by morphine in NG108-15 cells. Life Sci. 2011, 89, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Cayla, C.; Schaak, S.; Roquelaine, C.; Gales, C.; Quinchon, F.; Paris, H. Homologous regulation of the α2C-adrenoceptor subtype in human hepatocarcinoma, HepG2. Br. J. Pharmacol. 1999, 126, 69–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murrin, L.C.; Gerety, M.E.; Happe, H.K.; Bylund, D.B. Inverse agonism at α2-adrenoceptors in native tissue. Eur. J. Pharmacol. 2000, 398, 185–191. [Google Scholar] [CrossRef]
- Erdbrügger, W.; Raulf, M.; Otto, T.; Michel, M.C. Does [3H]2-methoxy-idazoxan (RX 821002) detect more alpha-2-adrenoceptor agonist high-affinity sites than [3H]rauwolscine? A comparison of nine tissues and cell lines. J. Pharmacol. Exp. Ther. 1995, 273, 1287–1294. [Google Scholar] [PubMed]
- Michel, M.C.; Brass, L.F.; Williams, A.; Bokoch, G.M.; LaMorte, V.J.; Motulsky, H.J. α2-Adrenergic receptor stimulation mobilizes intracellular Ca2+ in human erythroleukemia cells. J. Biol. Chem. 1989, 264, 4986–4991. [Google Scholar]
- Dixon, R.A.F.; Kobilka, B.K.; Strader, D.J.; Benovic, J.L.; Dohlman, H.G.; Frielle, T.; Bolanowski, M.A.; Bennett, C.D.; Rands, E.; Diehl, R.E.; et al. Cloning of the gene and cDNA for mammalian ß-adrenergic receptor and homology with rhodopsin. Nature 1986, 321, 75–79. [Google Scholar] [CrossRef]
- Sato, T.; Baker, J.; Warne, T.; Brown, G.A.; Leslie, A.G.W.; Congreve, M.; Tate, C.G. Pharmacological analysis and structure determination of 7-methylcyanopindolol–bound β1-adrenergic receptor. Mol. Pharmacol. 2015, 88, 1024–1034. [Google Scholar] [CrossRef] [Green Version]
- Warne, T.; Serrano-Vega, M.J.; Baker, J.G.; Moukhametzianov, R.; Edwards, P.C.; Henderson, R.; Leslie, A.G.W.; Tate, C.G.; Schertler, G.F.X. Structure of a ß1-adrenergic G-protein-coupled receptor. Nature 2008, 454, 486–491. [Google Scholar] [CrossRef] [Green Version]
- Delos Santos, N.M.; Gardner, L.A.; White, S.W.; Bahouth, S.W. Characterization of the residues in Helix 8 of the Human β1-adrenergic receptor that are involved in coupling the receptor to G proteins. J. Biol. Chem. 2006, 281, 12896–12907. [Google Scholar] [CrossRef] [Green Version]
- Janssens, K.; Boussemaere, M.; Wagner, S.; Kopka, K.; Denef, C. β1-Adrenoceptors in Rat Anterior Pituitary May Be Constitutively Active. Inverse Agonism of CGP 20712A on Basal 3′,5′-Cyclic Adenosine 5′-Monophosphate Levels. Endocrinology 2008, 149, 2391–2402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chidiac, P.; Nouet, S.; Bouvier, M. Agonist-induced modulation of inverse agonist efficacy at the ß2-adrenergic receptor. Mol. Pharmacol. 1996, 50, 662–669. [Google Scholar] [PubMed]
- Nakahara, T.; Maruko, T.; Sakamoto, K.; Ishii, K. Influence of receptor number on the cAMP response to forskolin in Chinese hamster ovary cells transfected with human b2-adrenoceptor. Biol. Pharmacol. Bull. 2004, 27, 239–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaya, A.I.; Uǧur, Ö.; Öner, S.S.; Bastepe, M.; Onaran, H.O. Coupling of β2-adrenoceptors to XLαs and Gαs: A new insight into ligand-induced G protein activation. J. Pharmacol. Exp. Ther. 2009, 329, 350–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLean, A.J.; Bevan, N.; Rees, S.; Milligan, G. Visualizing differences in ligand regulation of wild-type and constitutively active mutant ß2-adrenoceptor-green fluorescent protein fusion proteins. Mol. Pharmacol. 1999, 56, 1182–1191. [Google Scholar] [CrossRef] [PubMed]
- MacEwan, D.J.; Milligan, G. Up-regulation of a constitutively active form of the ß2-adrenoceptor by sustained treatment with inverse agonists but not antagonists. FEBS Lett. 1996, 339, 108–112. [Google Scholar] [CrossRef] [Green Version]
- Scarselli, M.; Annibale, P.; Radenovic, A. Cell type-specific β2-adrenergic receptor clusters identified using photoactivated localization microscopy are not lipid raft related, but depend on actin cytoskeleton integrity. J. Biol. Chem. 2012, 287, 16768–16780. [Google Scholar] [CrossRef] [Green Version]
- Azzi, M.; Pineyro, G.; Pontier, S.; Parent, S.; Ansanay, H.; Bouvier, M. Allosteric effects of G protein overexpression on the binding of ß-adrenergic ligands with distinct inverse efficacies. Mol. Pharmacol. 2001, 60, 999–1007. [Google Scholar] [CrossRef]
- Chakir, K.; Xiang, Y.; Yang, D.; Zhang, S.J.; Cheng, H.; Kobilka, B.K.; Xiao, R.P. The third intracellular loop and the carboxyl terminus of ß2-adrenergic receptor confer spontaneous activity of the receptor. Mol. Pharmacol. 2003, 64, 1048–1058. [Google Scholar] [CrossRef] [Green Version]
- Gregg, C.J.; Steppan, J.; Gonzalez, D.R.; Champion, H.C.; Phan, A.C.; Nyhan, D.; Shoukas, A.A.; Hare, J.M.; Barouch, L.A.; Berkowitz, D.E. β2-Adrenergic receptor-coupled phosphoinositide 3-kinase constrains cAMP-dependent increases in cardiac inotropy through phosphodiesterase 4 activation. Anesth. Analg. 2010, 111, 870–877. [Google Scholar] [CrossRef]
- Rosenbaum, D.M.; Zhang, C.; Lyons, J.A.; Holl, R.; Aragao, D.; Arlow, D.H.; Rasmussen, S.G.F.; Choi, H.J.; DeVree, B.T.; Sunahara, R.K.; et al. Structure and function of an irreversible agonist-ß2-adrenoceptor complex. Nature 2011, 469, 236–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boychuk, C.R.; Bateman, R.J.; Philbin, K.E.; Mendelowitz, D. α1-Adrenergic receptors facilitate inhibitory neurotransmission to cardiac vagal neurons in the nucleus ambiguus. Neuroscience 2011, 193, 154–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rank, M.M.; Murray, K.C.; Stephens, M.J.; D'Amico, J.; Gorassini, M.A.; Bennett, D.J. Adrenergic receptors modulate motoneuron excitability, sensory synaptic transmission and muscle spasms after chronic spinal cord injury. J. Neurophysiol. 2011, 105, 410–422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manoharan, A.; Morrison, A.E.; Lipworth, B.J. Effects of the inverse alpha-agonist doxazosin in allergic rhinitis. Clin. Exp. Allergy 2016, 46, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Micucci, M.; Chiarini, A.; Budriesi, R. Neutral/negative α1-AR antagonists and calcium channel blockers at comparison in functional tests on guinea-pig smooth muscle and myocardium. Pharmacol. Rep. 2019, 71, 128–132. [Google Scholar] [CrossRef]
- Doze, V.A.; Handel, E.M.; Jensen, K.A.; Darsie, B.; Luger, E.J.; Haselton, J.R.; Talbot, J.N.; Rorabaugh, B.R. α1A- and α1B-adrenergic receptors differentially modulate antidepressant-like behavior in the mouse. Brain Res. 2009, 1285, 148–157. [Google Scholar] [CrossRef] [Green Version]
- Han, X.; Liu, Y.; Kam, W.R.; Sullivan, D.A. Effect of brimonidine, an α2 adrenergic agonist, on human meibomian gland epithelial cells. Exp. Eye Res. 2018, 170, 20–28. [Google Scholar] [CrossRef]
- Bruzzone, A.; Perez, C.; Castillo, L.F.; Sarappa, M.G.; Rojas, P.; Lanari, C.; Lüthy, I.A. α2-Adrenoceptor action on cell proliferation and mammary tumour growth in mice. Br. J. Pharmacol. 2008, 155, 494–504. [Google Scholar] [CrossRef] [Green Version]
- Janhunen, S.K.; van der Zwaal, E.M.; la Fleur, S.E.; Adan, R.A.H. Inverse agonism at α2A adrenoceptors augments the hypophagic effect of sibutramine in rats. Obesity 2011, 19, 1979–1986. [Google Scholar] [CrossRef]
- Janhunen, S.K.; la Fleur, S.E.; Adan, R.A.H. Blocking alpha2A adrenoceptors, but not dopamine receptors, augments bupropion-induced hypophagia in rats. Obesity 2013, 21, E700–E708. [Google Scholar] [CrossRef]
- Akaishi, Y.; Hattori, Y.; Kanno, M.; Sakuma, I.; Kitabatake, A. Agonist-independent tonic inhibitory influence of Gi on adenylate cyclase activity in rabbit ventricular myocardium and its removal by pertussis toxin: A role of empty receptor-mediated Gi activation. J. Mol. Cell. Cardiol. 1997, 29, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Liggett, S.B.; Mialet-Perez, J.; Thaneemit-Chen, S.; Weber, S.A.; Greene, S.M.; Hodne, D.; Nelson, B.; Morrison, J.; Domanski, M.J.; Wagoner, L.E.; et al. A polymorphism within a conserved β1-adrenergic receptor motif alters cardiac function and β-blocker response in human heart failure. Proc. Natl. Acad. Sci. USA 2006, 103, 11288–11293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.Y.; Yang, D.; Zhu, W.Z.; Zhang, S.J.; Wang, D.J.; Rohrer, D.K.; Devic, E.; Kobilka, B.; Lakatta, E.G.; Cheng, H.; et al. Spontaneous activation of ß2- but not ß1-adrenoceptors expressed in cardiac myocytes from ß1ß2 double knockout mice. Mol. Pharmacol. 2000, 58, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Sun, H.; Koch, W.J.; Rau, T.; Eschenhagen, T.; Ravens, U.; Heubach, J.F.; Adamson, D.L.; Harding, S.E. Specific b2AR blocker ICI 118,551 actively decreases contraction through a Gi-coupled form of the b2AR in myocytes from failing human heart. Circulation 2002, 105, 2497–2503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bond, R.A.; Leff, P.; Johnson, T.D.; Milano, C.A.; Rockman, H.A.; McMinn, T.R.; Apparsundaram, S.; Hyek, M.F.; Kenakin, T.P.; Allen, L.F.; et al. Physiological effects of inverse agonists in transgenic mice with myocardial overexpression of the ß2-adrenoceptor. Nature 1995, 374, 272–276. [Google Scholar] [CrossRef] [Green Version]
- Gong, H.; Adamson, D.L.; Ranu, H.K.; Koch, W.J.; Heubach, J.F.; Ravens, U.; Zolk, O.; Harding, S.E. The effect of Gi-protein inactivation on basal, and ß1- and ß2AR-stimulated contraction of myocytes from transgenic mice overexpressing the ß2-adrenoceptor. Br. J. Pharmacol. 2000, 131, 594–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graf, E.M.; Heubach, J.F.; Ravens, U. The hyperpolarization-activated current If in ventricular myocytes of non-transgenic and ß2-adrenoceptor overexpressing mice. Naunyn Schmiedeberg’s Arch. Pharmacol. 2001, 364, 131–139. [Google Scholar] [CrossRef]
- Heubach, J.F.; Graf, E.M.; Molenaar, P.; Jäger, A.; Schröder, F.; Herzig, S.; Harding, S.E.; Ravens, U. Murine ventricular L-type Ca2+ current is enhanced by zinterol via ß1-adrenoceptors, and is reduced in TG4 mice overexpressing the human ß2-adrenoceptor. Br. J. Pharmacol. 2001, 133, 73–82. [Google Scholar] [CrossRef] [Green Version]
- Heubach, J.F.; Blaschke, M.; Harding, S.E.; Ravens, U.; Kaumann, A.J. Cardiostimulant and cardiodepressant effects through overexpressed human ß2-adrenoceptors in murine heart: Regional differences and functional role of ß1-adrenoceptors. Naunyn Schmiedeberg’s Arch. Pharmacol. 2003, 367, 380–390. [Google Scholar] [CrossRef]
- Liu, X.; Callaerts-Vegh, Z.; Evans, K.L.; Bond, R.A. Chronic infusion of ß-adrenoceptor antagonist and inverse agonists decreases elevated protein kinase A activity in transgenic mice with cardiac-specific overexpression of human ß2-adrenoceptor. J. Cardiovasc. Pharmacol. 2002, 40, 448–455. [Google Scholar] [CrossRef]
- Nagaraja, S.; Iyer, S.; Liu, X.; Eichberg, J.; Bond, R.A. Treatment with inverse agonists enhances baseline atrial contractility in transgenic mice with chronic beta2-adrenoceptor activation. Br. J. Pharmacol. 1999, 127, 1099–1104. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.-J.; Cheng, H.; Zhou, Y.-Y.; Wang, D.-J.; Zhu, W.; Ziman, B.; Spurgoen, H.; Lefkowitz, R.J.; Lakatta, E.G.; Koch, W.J.; et al. Inhibition of apontaneous β2-adrenergic activation rescues β1-adrenergic contractile response in cardiomyocytes overexpressing β2-adrenoceptor. J. Biol. Chem. 2000, 275, 21773–21779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.-Y.; Song, L.-S.; Lakatta, E.G.; Xiao, R.-P.; Cheng, H. Constitutive β2-adrenergic signalling enhances sarcoplasmic reticulum Ca2+ cycling to augment contraction in mouse heart. J. Physiol. 1999, 521, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.Y.; Cheng, H.; Song, L.S.; Wang, D.; Lakatta, E.G.; Xio, R.P. Spontaneous ß2-adrenergic signaling fails to modulate L-type Ca2+ current in mouse ventricular myocytes. Mol. Pharmacol. 1999, 56, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Callaerts-Vegh, Z.; Evans, K.L.J.; Shipley, G.L.; Davies, P.J.A.; Cuba, D.L.; Gurji, H.A.; Giles, H.; Bond, R.A. Effects of different beta adrenoceptor ligands in mice with permanent occlusion of the left anterior descending coronary artery. Br. J. Pharmacol. 2003, 138, 1505–1516. [Google Scholar] [CrossRef] [Green Version]
- Escobar, A.L.; Fernández-Gómez, R.; Peter, J.-C.; Mobini, R.; Hoebeke, J.; Mijares, A. IgGs and Mabs against the β2-adrenoreceptor block A-V conduction in mouse hearts: A possible role in the pathogenesis of ventricular arrhythmias. J. Mol. Cell. Cardiol. 2006, 40, 829–837. [Google Scholar] [CrossRef]
- Maack, C.; Cremers, B.; Flesch, M.; Höper, A.; Südkamp, M.; Böhm, M. Different intrinsic activities of bucindolol, carvedilol and metoprolol in human failing myocardium. Br. J. Pharmacol. 2000, 130, 1131–1139. [Google Scholar] [CrossRef]
- Maack, C.; Tyroller, S.; Schnabel, P.; Cremers, B.; Dabew, E.; Südkamp, M.; Böhm, M. Characterization of ß1-selectivity, adrenoceptor-Gs-protein interaction and inverse agonism of nebivolol in human myocardium. Br. J. Pharmacol. 2001, 132, 1817–1826. [Google Scholar] [CrossRef] [Green Version]
- Rozier, K.; Bondarenko, V.E. Mathematical modeling physiological effects of the overexpression of β2-adrenoceptors in mouse ventricular myocytes. Am. J. Physiol. Heart Circ. Physiol. 2018, 314, H643–H658. [Google Scholar] [CrossRef]
- Varma, D.R. Ligand-independent negative chronotropic responses of rat and mouse right atria to beta-adrenoceptor antagonists. Can. J. Physiol. Pharmacol. 1999, 77, 943–949. [Google Scholar] [CrossRef]
- Lin, R.; Peng, H.; Nguyen, L.P.; Dudekula, N.B.; Shardonofsky, F.; Knoll, B.J.; Parra, S.; Bond, R.A. Changes in β2-adrenoceptor and other signaling proteins produced by chronic administration of ‘β-blockers’ in a murine asthma model. Pulm. Pharmacol. Ther. 2008, 21, 115–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, L.P.; Lin, R.; Parra, S.; Omoluabi, O.; Hanania, N.A.; Tuvim, M.J.; Knoll, B.J.; Dickey, B.F.; Bond, R.A. ß2-Adrenoceptor signaling is required for the development of an asthma phenotype in a murine model. Proc. Natl. Acad. Sci. USA 2009, 106, 2435–2440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, L.P.; Singh, B.; Okulate, A.A.; Alfaro, V.Y.; Tuvim, M.J.; Dickey, B.F.; Bond, R.A. Complementary anti-inflammatory effects of a ß-blocker and a corticosteroid in an asthma model. Naunyn Schmiedeberg’s Arch. Pharmacol. 2012, 385, 203–210. [Google Scholar] [CrossRef] [PubMed]
- De Vries, B.; Meurs, H.; Roffel, A.F.; Elzinga, C.R.S.; Hoiting, B.H.; de Vries, M.M.L.; Zaagsma, J. β-Agonist-induced constitutive β2-adrenergic receptor activity in bovine tracheal smooth muscle. Br. J. Pharmacol. 2000, 131, 915–920. [Google Scholar] [CrossRef] [Green Version]
- de Vries, B.; Roffel, A.F.; Zaagsma, J.; Meurs, H. Effect of fenoterol-induced constitutive β2-adrenoceptor activity on contractile receptor function in airway smooth muscle. Eur. J. Pharmacol. 2001, 431, 353–359. [Google Scholar] [CrossRef]
- Peitzman, E.R.; Zaidman, N.A.; Maniak, P.J.; O'Grady, S.M. Carvedilol binding to β2-adrenergic receptors inhibits CFTR-dependent anion secretion in airway epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 310, L50–L58. [Google Scholar] [CrossRef] [Green Version]
- Baramki, D.; Koester, J.; Anderson, A.J.; Borish, L. Modulation of T-cell function by (R)- and (S)-isomers of albuterol: Anti-inflammatory influences of (R)-isomers are negated in the presence of the (S)-isomer. J. Allergy Clin. Immunol. 2002, 109, 449–454. [Google Scholar] [CrossRef]
- Cherezov, V.; Rosenbaum, D.M.; Hanson, M.A.; Rasmussen, S.G.F.; Thian, F.S.; Kobilka, T.S.; Choi, H.J.; Kuhn, P.; Weis, W.I.; Kobilka, B.K.; et al. High-resolution crystal structure of an engineered human ß2-adrenergic G protein-coupled receptor. Science 2007, 318, 1258–1265. [Google Scholar] [CrossRef] [Green Version]
- Rasmussen, S.G.F.; Choi, H.-J.; Rosenbaum, D.M.; Kobilka, T.S.; Thian, F.S.; Edwards, P.C.; Burghammer, M.; Ratnala, V.R.P.; Sanishvili, R.; Fischetti, R.F.; et al. Crystal structure of the human β2 adrenergic G-protein-coupled receptor. Nature 2007, 450, 383–387. [Google Scholar] [CrossRef]
- Vanni, S.; Neri, M.; Tavernelli, I.; Rothlisberger, U. Predicting novel binding modes of agonists to β adrenergic receptors using all-atom molecular dynamics simulations. PLoS Comput. Biol. 2011, 7, e1001053. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, S.; Hall, S.E.; Vaidehi, N. Agonist-induced conformational changes in bovine rhodopsin: Insight into activation of G-protein-coupled receptors. J. Mol. Biol. 2008, 382, 539–555. [Google Scholar] [CrossRef] [PubMed]
- Gether, U.; Lin, S.; Kobilka, B.K. Flurescent labeling of purified ß2 adrenergic receptor. Evidence for ligand-specific conformational changes. J. Biol. Chem. 1995, 270, 28268–28275. J. Biol. Chem. 1995, 270, 28268–28275. [Google Scholar] [PubMed] [Green Version]
- Goetz, A.; Lanig, H.; Gmeiner, P.; Clark, T. Molecular dynamics simulations of the effect of the G-protein and diffusible ligands on the β2-adrenergic receptor. J. Mol. Biol. 2011, 414, 611–623. [Google Scholar] [CrossRef] [PubMed]
- Katritch, V.; Reynolds, K.A.; Cherezov, V.; Hanson, M.A.; Roth, C.B.; Yeager, M.; Abagyan, R. Analysis of full and partial agonists binding to β2-adrenergic receptor suggests a role of transmembrane helix V in agonist-specific conformational changes. J. Mol. Recognit. 2009, 22, 307–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rasmussen, S.G.F.; Choi, H.J.; Fung, J.J.; Pardon, E.; Casarosa, P.; Chae, P.S.; DeVree, B.T.; Rosenbaum, D.M.; Thian, F.S.; Kobilka, T.S. Structure of a nanobody-stabilized active state of the ß2-adrenoceptor. Nature 2011, 469, 175–180. [Google Scholar] [CrossRef] [Green Version]
- Kofuku, Y.; Ueda, T.; Okude, J.; Shiraishi, Y.; Kondo, K.; Maeda, M.; Tsujishita, H.; Shimada, I. Efficacy of the β2-adrenergic receptor is determined by conformational equilibrium in the transmembrane region. Nat. Commun. 2012, 3, 1045. [Google Scholar] [CrossRef] [Green Version]
- Manglik, A.; Kim, T.; Masureel, M.; Altenbach, C.; Yang, Z.; Hilger, D.; Lerch, M.; Kobilka, T.; Thian, F.; Hubbell, W.; et al. Structural insights into the dynamic process of β2-adrenergic receptor signaling. Cell 2015, 161, 1101–1111. [Google Scholar] [CrossRef] [Green Version]
- Kooistra, A.J.; Leurs, R.; de Esch, I.J.P.; de Graaf, C. Structure-based prediction of G-protein-coupled receptor ligand function: A β-adrenoceptor case study. J. Chem. Inf. Modeling 2015, 55, 1045–1061. [Google Scholar] [CrossRef]
- Reynolds, K.A.; Katritch, V.; Abagyan, R. Identifying conformational changes of the b2 adrenoceptor that enable accurate prediction of ligand/receptor interactions and screening for GPCR modulators. J. Comput. Aided Mol. Des. 2009, 23, 273–288. [Google Scholar] [CrossRef] [Green Version]
- Weiss, D.R.; Ahn, S.; Sassano, M.F.; Kleist, A.; Zhu, X.; Strachan, R.; Roth, B.L.; Lefkowitz, R.J.; Shoichet, B.K. Conformation guides molecular efficacy in docking screens of activated β-2 adrenergic G protein coupled receptor. ACS Chem. Biol. 2013, 8, 1018–1026. [Google Scholar] [CrossRef]
- Kolb, P.; Rosenbaum, D.M.; Irwin, J.J.; Fung, J.J.; Kobilka, B.K.; Shoichet, B.K. Structure-based discovery of β2-adrenergic receptor ligands. Proc. Natl. Acad. Sci. USA 2009, 106, 6843–6848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bokoch, M.P.; Zou, Y.; Rasmussen, S.G.F.; Liu, C.W.; Nygaard, R.; Rosenbaum, D.M.; Fung, J.J.; Choi, H.J.; Thian, F.S.; Kobilka, T.S.; et al. Ligand-specific regulation of the extracellular surface of a G-protein-coupled receptor. Nature 2010, 463, 108–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, Q.; Pérez-Sánchez, H.; Zhang, Y.; Shao, Y.; Shi, D.; Liu, H.; Yao, X. Ligand induced change of β2 adrenergic receptor from active to inactive conformation and its implication for the closed/open state of the water channel: Insight from molecular dynamics simulation, free energy calculation and Markov state model analysis. Phys. Chem. Chem. Phys. 2014, 16, 15874–15885. [Google Scholar] [CrossRef] [PubMed]
- Bai, Q.; Zhang, Y.; Ban, Y.; Liu, H.; Yao, X. Computational study on the different ligands induced conformation change of β2 adrenergic receptor-Gs protein complex. PLoS ONE 2013, 8, e68138. [Google Scholar] [CrossRef] [PubMed]
- Lakkaraju, S.K.; Lemkul, J.A.; Huang, J.; MacKerell, A.D., Jr. DIRECT-ID: An automated method to identify and quantify conformational variations—Application to β2-adrenergic GPCR. J. Comput. Chem. 2016, 37, 416–425. [Google Scholar] [CrossRef] [Green Version]
- West, G.M.; Chien, E.Y.T.; Katritch, V.; Gatchalian, J.; Chalmers, M.J.; Stevens, R.C.; Griffin, P.R. Ligand-dependent perturbation of the conformational ensemble for the GPCR β2 adrenergic receptor revealed by HDX. Structure 2011, 19, 1424–1432. [Google Scholar] [CrossRef] [Green Version]
- Yao, X.J.; Ruiz, G.V.; Whorton, M.R.; Rasmussen, S.G.F.; DeVree, B.T.; Deupi, X.; Sunahara, R.K.; Kobilka, B. The effect of ligand efficacy on the formation and stability of a GPCR-G protein complex. Proc. Natl. Acad. Sci. USA 2009, 106, 9501–9506. [Google Scholar] [CrossRef] [Green Version]
- Granier, S.; Kim, S.; Shafer, A.M.; Ratnala, V.R.P.; Fung, J.J.; Zare, R.N.; Kobilka, B. Structure and conformational changes in the C-terminal domain of the ß2-adrenoceptor. Insights from fluorescence resonance energy transfer studies. J. Biol. Chem. 2007, 282, 13895–13905. [Google Scholar]
- Hebert, T.E.; Moffett, S.; Morello, J.-P.; Loisel, T.P.; Bichet, D.G.; Barret, C.; Bouvier, M. A peptide derived from a β2-adrenergic receptor transmembrane domain Inhibits both receptor dimerization and activation. J. Biol. Chem. 1996, 271, 16384–16392. [Google Scholar] [CrossRef] [Green Version]
- DeVree, B.T.; Mahoney, J.P.; Vélez-Ruiz, G.A.; Rasmussen, S.G.F.; Kuszak, A.J.; Edwald, E.; Fung, J.-J.; Manglik, A.; Masureel, M.; Du, Y.; et al. Allosteric coupling from G protein to the agonist-binding pocket in GPCRs. Nature 2016, 535, 182–186. [Google Scholar] [CrossRef] [Green Version]
- Lachance, M.; Ethier, N.; Wolbring, G.; Schnetkamp, P.P.M.; Hébert, T.E. Stable association of G proteins with β2AR is independent of the state of receptor activation. Cell. Signal. 1999, 11, 523–533. [Google Scholar] [CrossRef]
- Insel, P.A.; Tang, C.M.; Hahntow, I.; Michel, M.C. Impact of GPCRs in clinical medicine: Genetic variants and drug targets. Biochim. Biophys. Acta 2007, 1768, 994–1005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Port, J.D.; Bristow, M.R. ß-Adrenergic receptors, transgenic mice, and pharmacological model systems. Mol. Pharmacol. 2001, 60, 629–631. [Google Scholar] [PubMed]
- Hanania, N.A.; Singh, S.; El-Wali, R.; Flashner, M.; Franklin, A.E.; Garner, W.J.; Dickey, B.F.; Parra, S.; Ruoss, S.; Shardonofsky, F.R.; et al. The safety and effects of the beta-blocker, nadolol, in mild asthma: An open-label pilot study. Pulm. Pharmacol. Ther. 2008, 21, 134–141. [Google Scholar] [CrossRef] [Green Version]
- Thanawala, V.J.; Valdez, D.J.; Joshi, R.; Forkuo, G.S.; Parra, S.; Knoll, B.J.; Bouvier, M.; Leff, P.; Bond, R.A. ß-Blockers have differential effects on the murina asthma phenotype. Br. J. Pharmacol. 2015, 172, 4833–4846. [Google Scholar] [CrossRef]
- Dickey, B.F.; Walker, J.K.L.; Hanania, N.A.; Bond, R.A. β-Adrenoceptor inverse agonists in asthma. Curr. Opin. Pharmacol. 2010, 10, 254–259. [Google Scholar] [CrossRef] [Green Version]
- Penn, R.B. Agonizing over agonism: Should asthmatics turn their β-receptors on or off? Proc. Natl. Acad. Sci. USA 2009, 106, 2095–2096. [Google Scholar] [CrossRef] [Green Version]
- Parra, S.; Bond, R.A. Inverse agonism: From curiosity to accepted dogma, but is it clinically relevant? Curr. Opin. Pharmacol. 2007, 7, 146–150. [Google Scholar] [CrossRef]
- Michel, M.C.; Seifert, R.; Bond, R.A. Dynamic bias and its implications for GPCR drug discovery. Nat. Rev. Drug Discov. 2014, 13, 869–870. [Google Scholar] [CrossRef] [Green Version]
- Michel, M.C.; Charlton, S.J. Biased agonism in drug discovery—Is it too soon to choose a path? Mol. Pharmacol. 2018, 93, 259–265. [Google Scholar] [CrossRef] [Green Version]
- Kenakin, T. Is the quest for signaling bias worth the effort? Mol. Pharmacol. 2018, 93, 266–269. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michel, M.C.; Michel-Reher, M.B.; Hein, P. A Systematic Review of Inverse Agonism at Adrenoceptor Subtypes. Cells 2020, 9, 1923. https://doi.org/10.3390/cells9091923
Michel MC, Michel-Reher MB, Hein P. A Systematic Review of Inverse Agonism at Adrenoceptor Subtypes. Cells. 2020; 9(9):1923. https://doi.org/10.3390/cells9091923
Chicago/Turabian StyleMichel, Martin C., Martina B. Michel-Reher, and Peter Hein. 2020. "A Systematic Review of Inverse Agonism at Adrenoceptor Subtypes" Cells 9, no. 9: 1923. https://doi.org/10.3390/cells9091923
APA StyleMichel, M. C., Michel-Reher, M. B., & Hein, P. (2020). A Systematic Review of Inverse Agonism at Adrenoceptor Subtypes. Cells, 9(9), 1923. https://doi.org/10.3390/cells9091923





