1. Introduction
The heart is the first organ to form during mammalian embryogenesis and is critical for the viability of the embryo. Complex communication between cells and tissue is required to ensure the correct specification of cell lineage necessary for embryological heart formation [
1]. Failure of proper cellular differentiation, migration and/or apoptosis results in congenital heart disease (CHD). CHD is a major cause of childhood morbidity and mortality, and remains a substantial challenge even in countries with advanced health care systems. Heart defects are the most common birth defect, occurring in approximately eight per 1000 live births [
2]. Mendelian and chromosomal syndromes account for about 20% of all cases of CHD. The remaining 80% of CHDs are “sporadic”, and have proven intractable to genetic investigation. These sporadic events are most often inherited from unaffected parents, suggesting incomplete penetrance [
3]. Variable penetrance can be explained, at least in part, by differences in the genetic buffering capacity between individuals [
4,
5].
Tetralogy of Fallot (TOF) is one of the more common severe forms of CHD with an incidence estimated at five to seven per 10,000 live births, thus representing 5%–7% of all congenital heart lesions. TOF is characterized by a misalignment of the conal septum leading to a rightward deviation of the aorta. This results in a large ventricular septal defect (VSD) and varying degrees of right ventricular outflow tract narrowing. TOF generally requires surgical repair in the first year of life.
MicroRNAs are important posttranscriptional inhibitors of gene expression resulting from the degradation of target mRNA or inhibition of translation. A powerful aspect of microRNA function is the ability of individual microRNAs to coordinate and regulate multiple target genes encoding proteins with related functions (e.g., stem cell differentiation, neurogenesis, skeletal and cardiac muscle development and function) [
6]. Furthermore, individual mRNAs can be targeted by multiple microRNAs, allowing for enormous combinatorial complexity and regulatory potential.
Recently, it has become apparent that mammalian cardiac development depends on the correct spatiotemporal expression of specific microRNAs [
7]. For example, miR-1 promotes embryonic stem cell differentiation toward a cardiomyocyte lineage, whereas miR-133 inhibits differentiation into cardiac muscle. MiR-1 targets the Notch ligand Delta-like-1 (Dll-1) [
8] and the transcription factor Hand2 [
9] which is required for right ventricular growth and cardiomyocyte expansion. Furthermore, miR-1 knockout mice had thickened ventricles resulting from hyperplasia. Deletion of miR-1-2 causes lethal VSDs [
10]. MiR-133a-1/133a-2 double knockout in mice results in inappropriate expression of SRF and cyclin D2 leading to late embryonic neonatal lethality due to VSDs and chamber dilatation [
11]. Deletion of either miR-106b-25 or miR-106a-363, combined with the miR-17-92 null allele, can result in embryonic lethality accompanied by severe VSDs, atrial septal defects, and thin walled myocardium [
12].
A few miRNAs have been associated with CHDs. Kuhn
et al. reported that five human chromosome 21-derived microRNAs (miR-99a, let-7c, miR-125b-2, miR-155, and miR-802) are overexpressed in hearts from subjects with Down syndrome and CHD [
13]. Also, miR-196a, which is an upstream regulator of Hoxb8 and Sonic hedgehog (Shh), has been associated with sporadic CHD [
14]. We recently evaluated microRNA expression in right ventricular tissue from children with tetralogy of Fallot compared to tissue from normally developing age-matched comparison subjects [
15]. We found multiple microRNAs with altered expression, accompanied by an inverse expression pattern of many genes predicted to be targeted by these microRNAs. In addition, we evaluated gene expression patterns in the RV of infants with TOF and found a significant suppression of both the Notch and Wnt pathways relative to normally developing tissue [
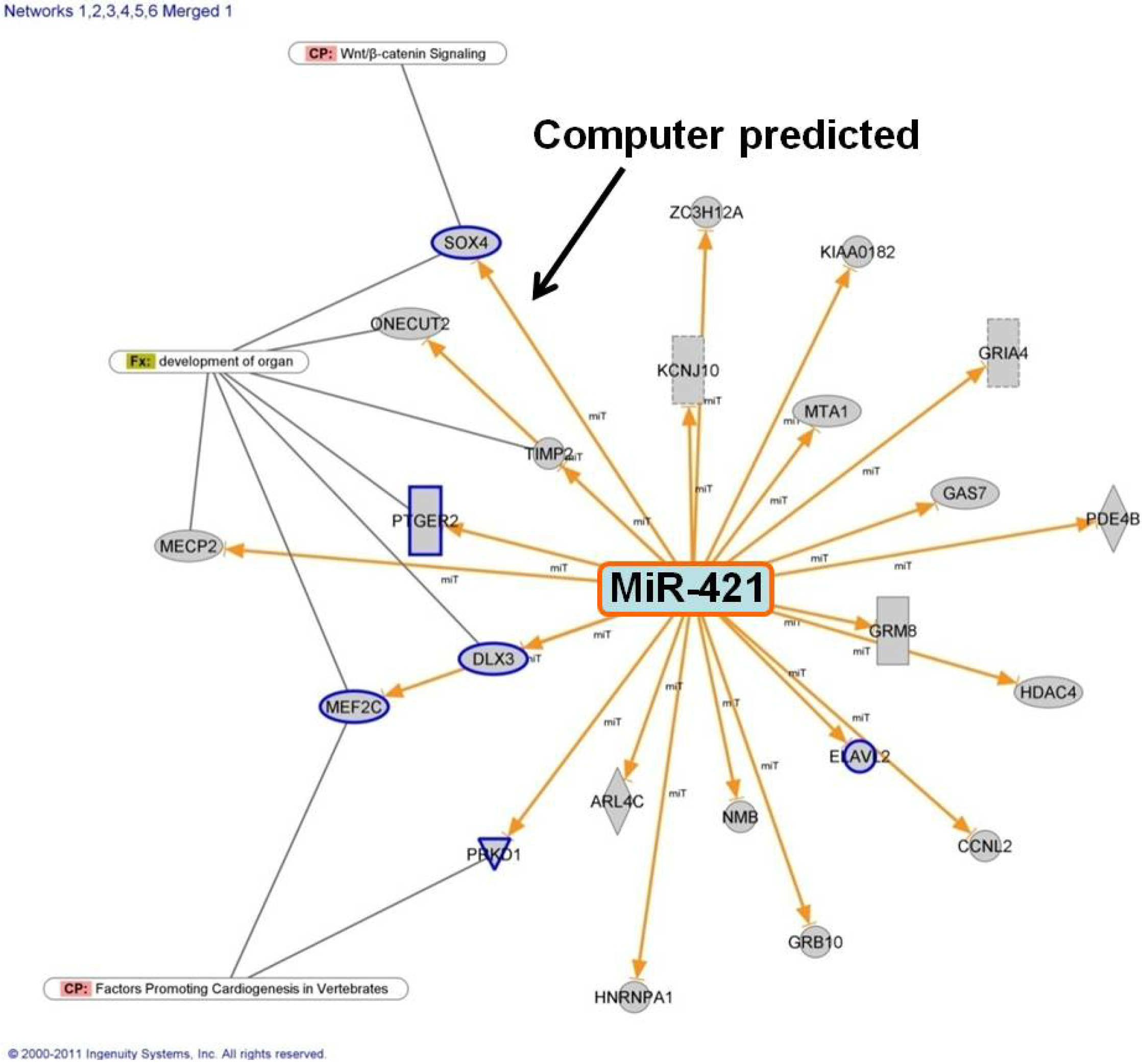
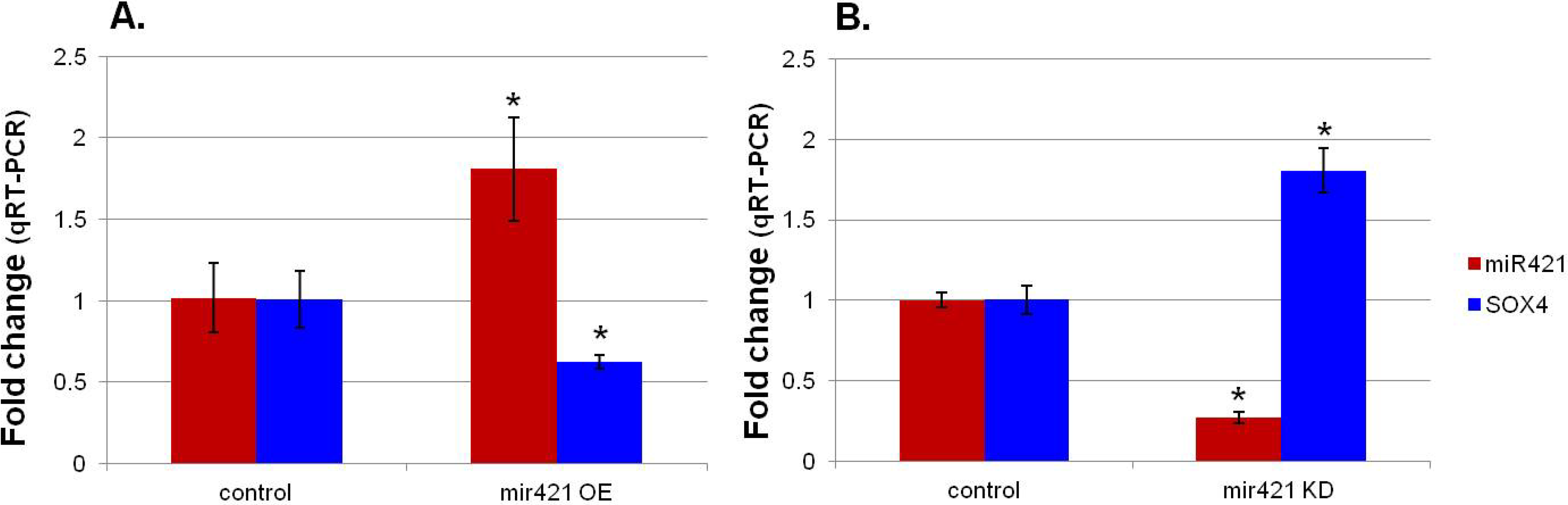
16]. Intriguingly, many of the microRNAs we identified with altered expression in the RV of infants with TOF are predicted to interact with genes in the Notch or Wnt pathways. Of particular interest was SOX4, predicted to be targeted by several of the microRNAs and shown to interact with several key developmental pathways including the Notch and Wnt pathways [
17]. SOX4 has been shown to be essential for cardiac outflow track formation [
18,
19]. Taken together, these studies support a significant role for microRNAs in mammalian heart development. Never the less, the extent and mechanism by which microRNAs contribute to human CHD remains poorly investigated.
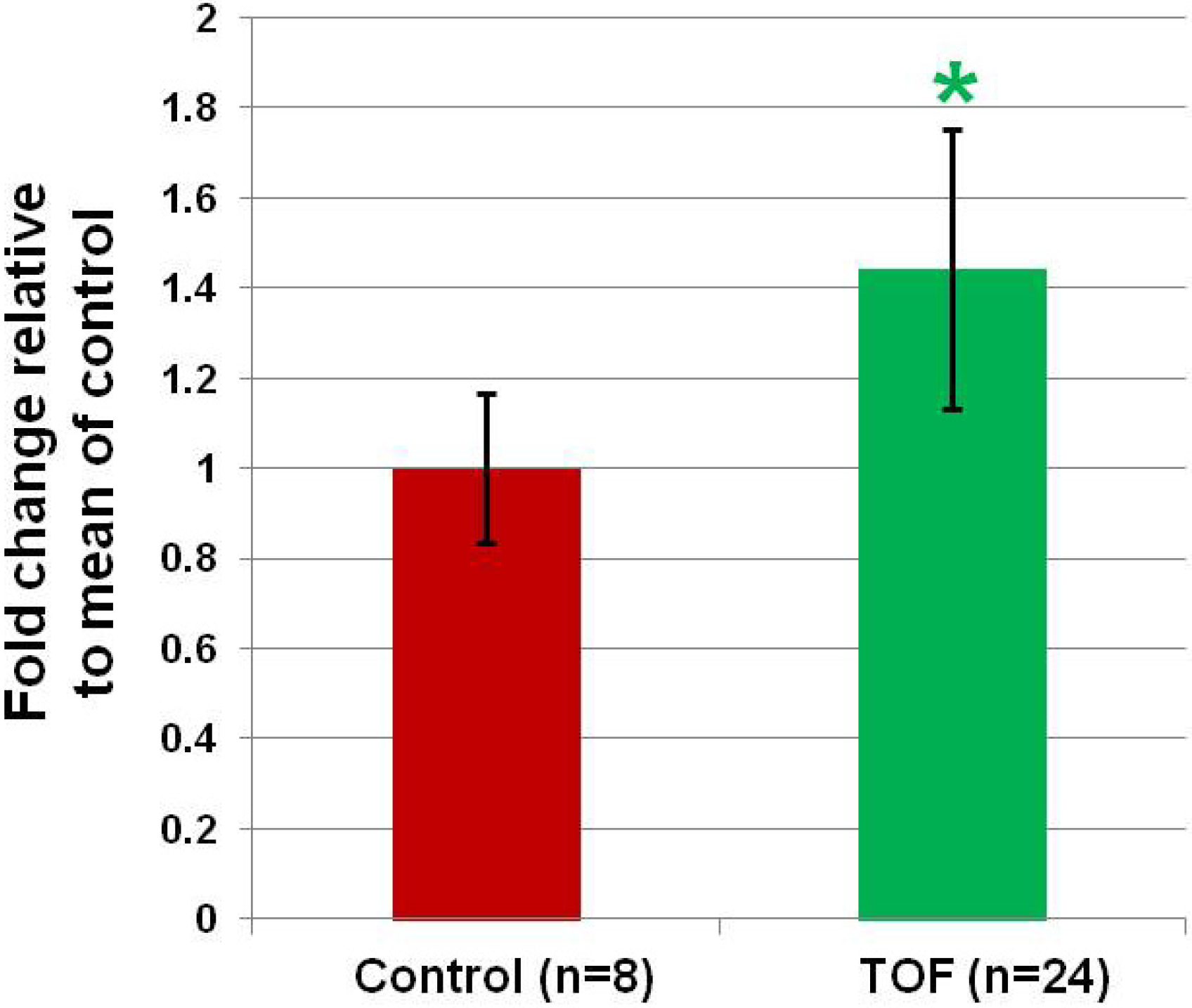
In our previous study, the microRNA with greatest change in expression was miR-421.
In silico analysis of the potential targets of miR-421 predict it will interact with multiple genes known to regulate heart development, most interestingly SOX4. Furthermore, miR-421 has recently been identified in multiple forms of cancer, apparently playing a role in cell proliferation [
20,
21]. Here we present evidence that miR-421 does indeed modulate the expression of genes of importance to heart development and therefore, could play a role in congenital heart defects.
2. Experimental Section
2.1. Subjects
Our subjects were children less than one year of age with tetralogy of Fallot (TOF) requiring surgical reconstruction. The diagnosis and anatomy were obtained by echocardiography and angiography, and confirmed at the time of surgery. Informed consent was obtained from a parent or legal guardian after reviewing the consent document and having their questions answered. All proper institutional review board approvals were obtained for this study. Microarray analyses were run on samples from 16 infants with nonsyndromic TOF (
i.e., no 22q11.2 deletion) and eight infants with normally developing hearts (
Table 1). Comparison tissues from eight normally developing infants (three males, five females) were obtained from LifeNet Health [
22]. The control subjects were matched for age to the study population, and all control subjects expired due to non-cardiac related causes. The acquisition and pathology of these samples was described in detail previously [
15].
Table 1.
Age and Sex of subjects.
Table 1.
Age and Sex of subjects.
| | n | Gender | Mean Age (Range) | Analyzed by Array | Analyzed by qRT-PCR |
|---|
| TOF | 16 | 11M/5F | 276 days (98–510) | 16 | 16 |
| control | 8 | 3M/5F | 142 days (28–382) | 8 | 8 |
| additional TOF subjects used for validation | 8 | 4M/4F | 292 days (167–425) | 0 | 8 |
2.2. Cell Cultures and Transfection
Primary cells from infants with TOF were derived from right ventricular tissue removed at the time of surgical repair of the conotruncal outflow track. The RV was minced in DMEM (Life Technologies, Grand Island, NY) plus 10% fetal calf serum and 1% penn/strep (Life Technologies), and placed in an incubator with 5% CO2. After cells were established, they were trypsinized and grown in T-75 flasks with 10%/1% DMEM. We obtained a primary neonatal cardiomyocyte cell culture derived from normally developing human neonatal cardiac tissue from Celprogen (Celprogen Inc., Torrence, CA, Cat#36044-21). These cells were grown in the same media as the TOF primary cells. All transfections on primary cells were performed three times, each on a different cell line (genotype) and expression changes were normalized and used for analysis. Transfections on the primary cells from the normally developing infant heart were repeated three times.
The miR-421 expression vector was ordered from Origene (Origene Technologies, Rockville MD,
Figure 1, MIR421, cat #MI0003685,). We sequenced the plasmid after amplification to ensure that the miR-421 sequence was intact. Plasmid transfections were done using PolyMag transfection reagent according to the manufacturer’s instructions (Oz Bioscience Inc., San Diego, CA, USA). Briefly, 2 μg plasmid DNA was diluted in 200 μL of serum/supplement free media and added to 8 μL PolyMag transfection reagent, vortexed and incubated for 20 min at room temperature. The transfection mixture was added dropwise to 3 × 10
5 cells in 1.8 mL of serum containing media in a single well of a 6 well plate. The plate was set on top of a plate magnet (Oz Biosciences) for 20 min, and returned to the incubator. After 72 h, the cells were trypsinized, pelleted and stored at −80 °C until processed for RNA analysis.
Figure 1.
MiR-421 expression vector.
Figure 1.
MiR-421 expression vector.
We ordered flexitube antisense siRNAs for knockdown experiments targeting miR-421 (Qiagen Inc., Valencia, CA, Cat # Hs_MIR421-1). Knockdown experiments were done using HiPerfect transfection reagent (Qiagen, Valencia, CA) following the manufacturer’s instructions. Briefly, 50 μM siRNA diluted in 100 μL serum free media was mixed with 12 μL HiPerfect transfection reagent and incubated for 20 min at room temperature. The transfection mixture was added to 2 × 105 cells in 2.3 mL media with 10% serum in a single well of a 6 well cell culture plate. After 72 h, the cells were pelleted and stored at −80 °C until processing.
2.3. RNA Preparation
RNA was extracted from ~10 mg of frozen tissue from the right ventricle or cells using a mirVana microRNA isolation kit (Applied Biosystems/Ambion, Austin, TX, USA) according to manufacturer’s instructions. The control tissues (cryo-preserved pulmonary homografts) were thawed per protocol in sterile conditions. Tissue samples obtained during patient surgery (attending surgeon JEO) were immediately de-identified and frozen. All tissue samples removed during surgery were excised by the performing surgeon for clinical indications utilizing standard of care procedures. While a subset of patients were previously palliated with a modified Blalock-Taussig (BT) shunt, the right ventricular outflow tract region from which the tissue was harvested had not undergone any previous surgical manipulation. One author (JEO) aseptically dissected samples from the normally developing control heart tissue to ensure the tissues were from equivalent locations.
2.4. Microarray Analysis
The microRNA microarrays were Affymetrix GeneChip microRNA-1.0 (Affymetrix Inc. Santa Clara, CA, USA). The GeneChip microRNA-1.0 array contains probes for 847 human microRNAs and 922 human snoRNAs (the snoRNAs are inclusive of scaRNAs). The exon arrays were Affymetrix HuEx-1_0-st-v2. The raw data for the microRNA arrays have been deposited in the Gene Expression Omnibus (microRNA arrays -accession number GSE35490). All arrays were run at the Kansas University Medical Center-Microarray Facility (KUMC-MF) according to the manufacturer’s protocols. The KUMC-MF is supported by the Kansas University-School of Medicine, KUMC Biotechnology Support Facility, the Smith Intellectual and Developmental Disabilities Research Center (HD02528), and the Kansas IDeA Network of Biomedical Research Excellence (RR016475).
All statistical analyses were performed using statistical software: Partek Genomics Suite software version 6.6 (Partek Inc, St. Louis, MO, USA), and Ingenuity Pathways (Ingenuity systems, Inc. Redwood City, CA, USA) as described previously [
15]. Raw data (CEL. files) were uploaded into Partek Genomics Suite for normalization and statistical analysis. Robust Multichip Analysis (RMA) was used for background correction followed by quantile normalization with baseline transformation to the median of the control samples. Only probes with intensity values above 20% of background value, in at least one of the conditions, were included for additional analysis. A Student t-test with a Benjamini and Hochberg multiple test correction for false discovery rate (FDR) was used to determine significance. Probes were filtered using an FDR-adjusted
p- value ≤ 0.05.
The Ingenuity Pathways Analysis (IPA) version 9.0 (Ingenuity Systems, Inc., Redwood City, CA, USA) was used to explore networks, canonical pathways, predefined functional categories, IPA compiles experimentally validated miRNA/mRNA interactions as well as providing predicted microRNA/mRNA interactions using integrated access to TargetScan (Human), version 5; TarBase and MiRecords. Predicted interactions are scored as highly probable or moderately probable. We limited our miRNA target assessment to highly probable interactions only. IPA contains hundreds of pathways and identifies significant associations between the experimental data set and canonical pathways, functional categories or disease associations within the database by comparing the ratio of the number of molecules from the dataset that map to a given pathway, divided by the total number of molecules that map to the pathway. A Fisher’s exact test was used to calculate a p-value determining the probability that the association between the genes in the dataset and the pathway was explained by chance alone. All biological functions and/or diseases in IPA’s database were considered for the analysis without bias. Significance was defined as a p-value ≤ 0.05.
2.5. Real-Time Quantitative Polymerase Chain Reaction
To validate gene expression, quantitative reverse transcription-PCR (qRT-PCR) was performed on a subset of genes/transcripts using Taqman assays (Applied Biosystems. Inc., Life Technologies, Grand Island, NY, USA) according to the manufacturer’s instructions. Briefly, 2 ng of total RNA from each sample was reversed-transcribed using a microRNA specific Taqman microRNA reverse transcription kit. For each sample, real-time qRT-PCR was performed in triplicate on an ABI 7000 sequence detection instrument. The point at which the intensity level crossed the PCR cycle threshold (CT) was used to compare individual reactions. RNU24 and GAPDH were used for normalization using the standard ΔΔCT method. Results were calculated as fold change relative to control subjects.