Subarachnoid Hemorrhage Depletes Calcitonin Gene-Related Peptide Levels of Trigeminal Neurons in Rat Dura Mater
Abstract
1. Introduction
2. Materials and Methods
2.1. Surgical Procedure and SAH Induction
2.2. Retrograde Labeling of Trigeminal Afferents
2.3. Experimental Groups
2.4. Neurological Examination
2.5. Immunohistochemistry
2.6. Immunohistochemical Imaging and Image Analysis
2.6.1. Image Acquisition and Line Scan Analysis in Dura Mater Preparation
2.6.2. Immunohistochemical Analysis in the TG
2.6.3. Immunohistochemical Analysis in the TNC
2.7. RNA Extraction and Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
2.8. Measuring CGRP Release from Meningeal Afferents
2.9. Statistics
3. Results
3.1. Neurological Evaluation of the Single-Injection SAH Model
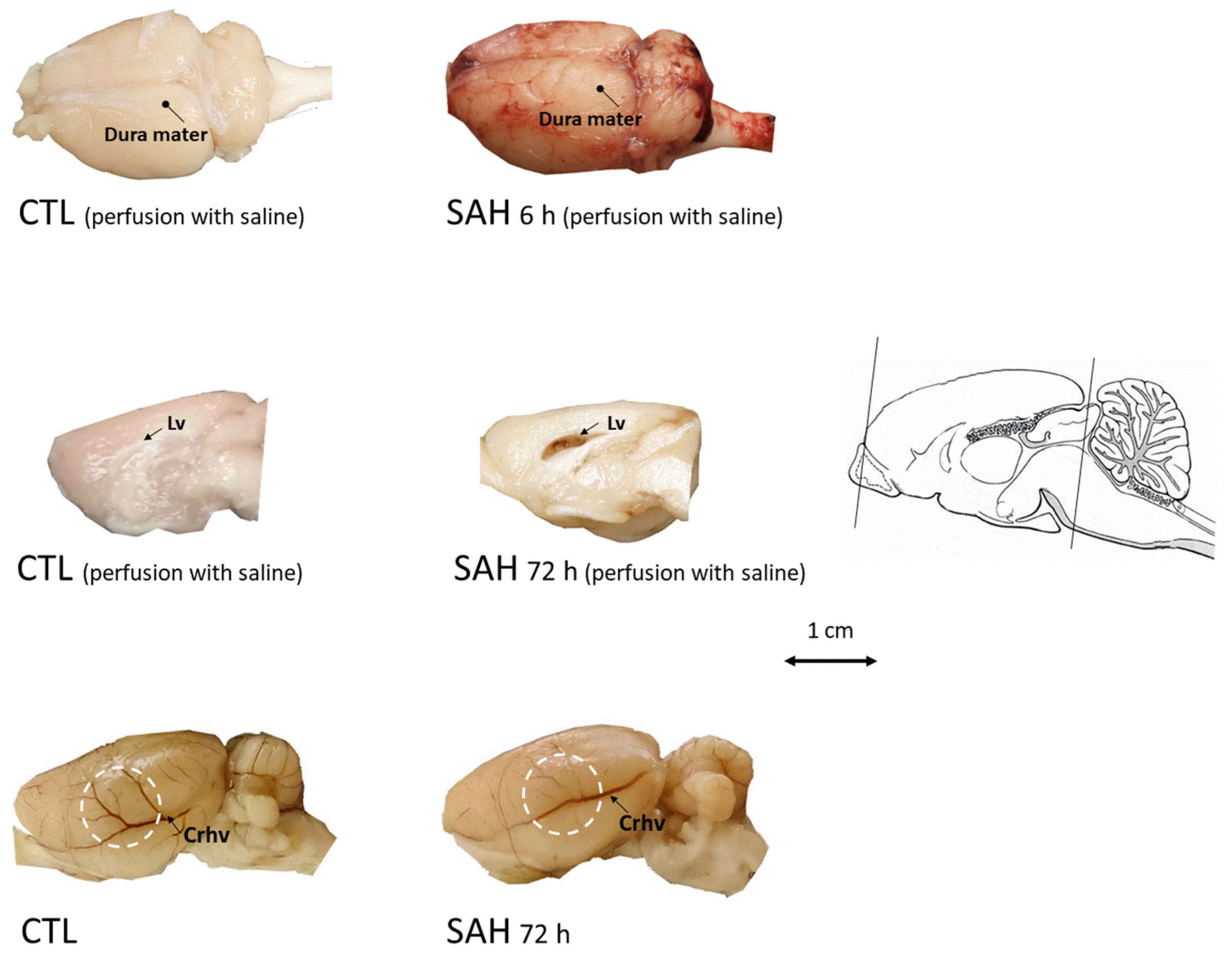
3.2. Macroscopic Inspection of the Brain after SAH
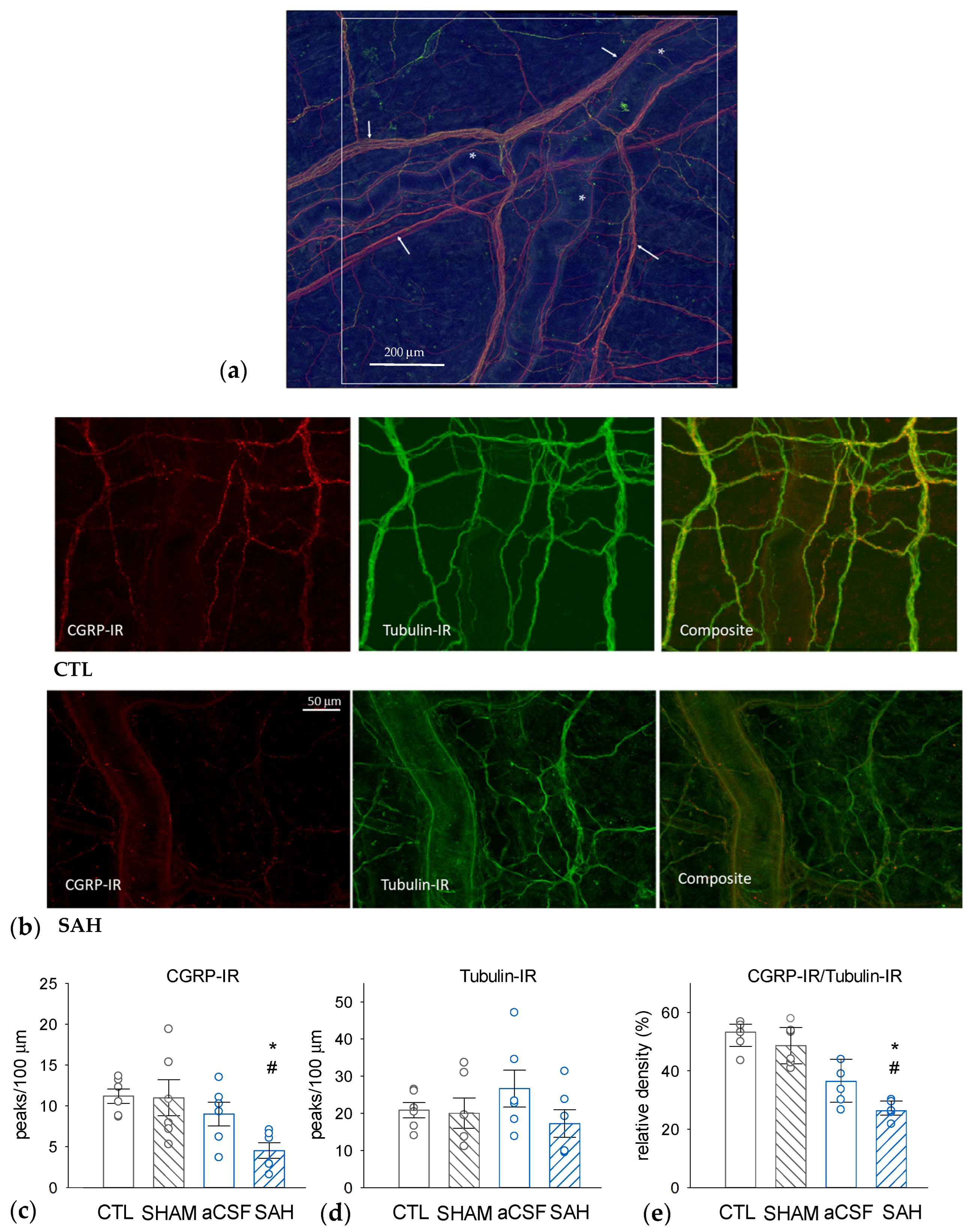
3.3. SAH Depletes CGRP in Perivascular Nerves in the Rat Dura Mater
3.4. SAH Reduces CGRP Release from the Rat Dura Mater
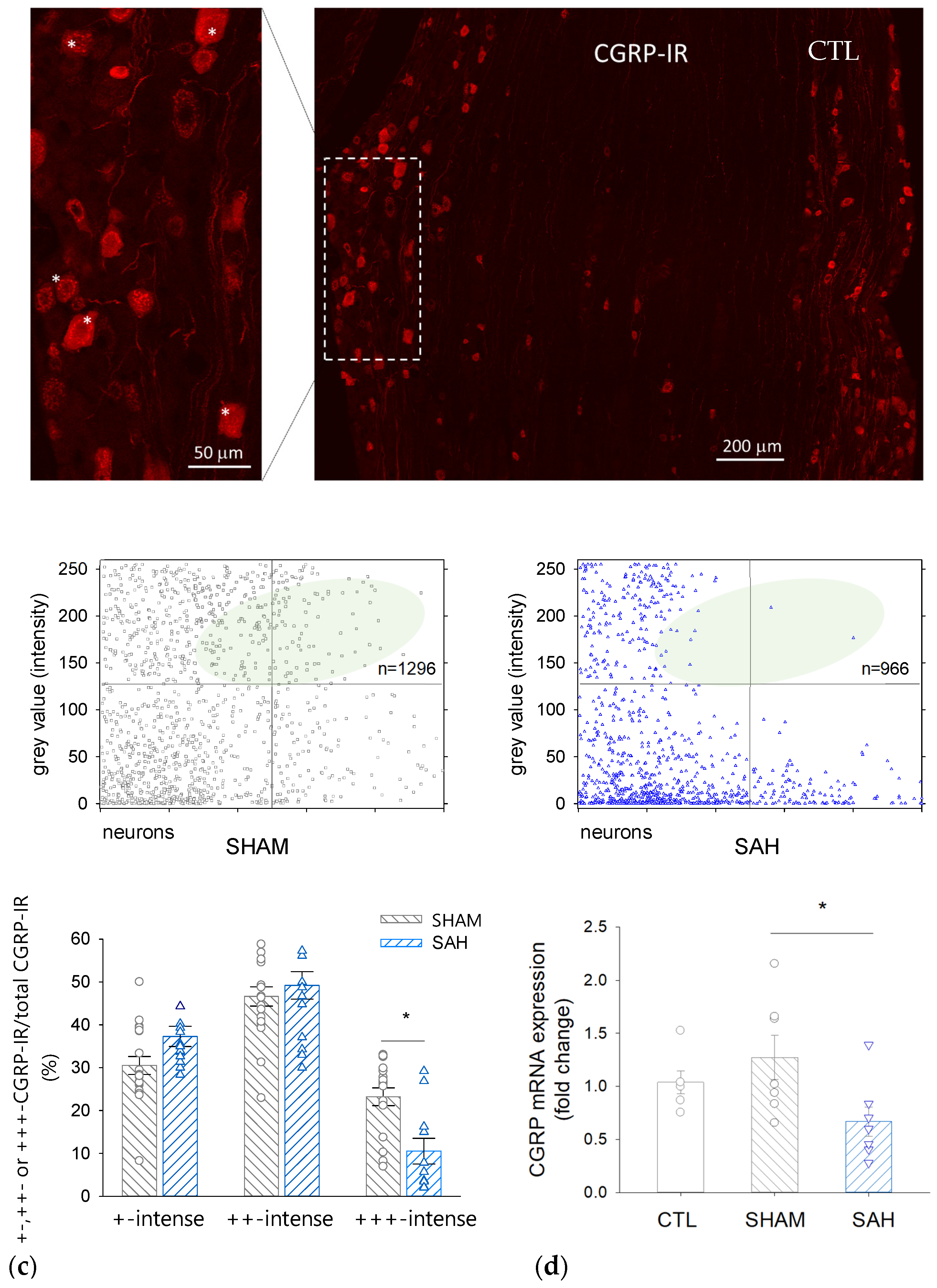
3.5. SAH Reduces the Proportion of Intensively CGRP-IR Neuronal Cell Bodies in the TG
3.6. SAH Attenuates CGRP mRNA Expression in the TG
3.7. SAH Depletes CGRP also in the Central Trigeminal Projections in the Trigeminal Nucleus Caudalis (TNC)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CGRP | calcitonin gene-related peptide |
| CSF | cerebrospinal fluid |
| CV | cerebral vasospasm |
| ELISA | Enzyme-linked Immunosorbent Assay |
| ICP | intracranial pressure |
| IR | immuno-reactive |
| qRT-PCR | Quantitative Real-Time Polymerase Chain Reaction |
| SEM | standard error of the mean |
| SIF | synthetic interstitial fluid |
| SAH | subarachnoid hemorrhage |
| TNC | trigeminal nucleus caudalis |
References
- Feindel, W.; Penfield, W.; McNaughton, F. The tentorial nerves and Iocalization of intracranial pain in man. Neurology 1960, 10, 555–563. [Google Scholar] [CrossRef]
- Wirth, F.P.; Van Buren, J.M. Referral of pain from dural stimulation in man. J. Neurosurg. 1971, 34, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Mayberg, M.; Langer, R.S.; Zervas, N.T.; Moskowitz, M.A. Perivascular meningeal projections from cat trigeminal ganglia: Possible pathway for vascular headaches in man. Science 1981, 213, 228–230. [Google Scholar] [CrossRef] [PubMed]
- May, A.; Goadsby, P.J. The trigeminovascular system in humans: Pathophysiologic implications for primary headache syndromes of the neural influences on the cerebral circulation. J. Cereb. Blood Flow Metab. 1999, 19, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Edvinsson, L.; Ekman, R. Release of vasoactive peptides in the extracerebral circulation of humans and the cat during activation of the trigeminovascular system. Ann. Neurol. 1988, 23, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Zagami, A.S.; Goadsby, P.J.; Edvinsson, L. Stimulation of the superior sagittal sinus in the cat causes release of vasoactive peptides. Neuropeptides 1990, 16, 69–75. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, T.P.; van der Kooy, D. Enrichment of a vasoactive neuropeptide (calcitonin gene related peptide) in the trigeminal sensory projection to the intracranial arteries. J. Neurosci. 1988, 8, 2468–2476. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, M.A.; Buzzi, M.G.; Sakas, D.E.; Linnik, M.D. Pain mechanisms underlying vascular headaches. Progress Report 1989. Rev. Neurol. 1989, 145, 181–193. [Google Scholar] [PubMed]
- Edvinsson, L.; Goadsby, P.J. Neuropeptides in migraine and cluster headache. Cephalalgia Int. J. Headache 1994, 14, 320–327. [Google Scholar] [CrossRef]
- Naghdi, S.; Underwood, M.; Madan, J.; Brown, A.; Duncan, C.; Matharu, M.; Aksentyte, A.; Davies, N.; Rees, S.; Cooklin, A.; et al. Clinical effectiveness of pharmacological interventions for managing chronic migraine in adults: A systematic review and network meta-analysis. J. Headache Pain 2023, 24, 164. [Google Scholar] [CrossRef]
- Burnstock, G. Local mechanisms of blood flow control by perivascular nerves and endothelium. J. Hypertens. Suppl. 1990, 8, S95–S106. [Google Scholar]
- Wahl, M.; Schilling, L.; Parsons, A.A.; Kaumann, A. Involvement of calcitonin gene-related peptide (CGRP) and nitric oxide (NO) in the pial artery dilatation elicited by cortical spreading depression. Brain Res. 1994, 637, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Lenz, I.J.; Plesnila, N.; Terpolilli, N.A. Role of endothelial nitric oxide synthase for early brain injury after subarachnoid hemorrhage in mice. J. Cereb. Blood Flow Metab. 2021, 41, 1669–1681. [Google Scholar] [CrossRef] [PubMed]
- Hamann, G.; Isenberg, E.; Strittmatter, M.; Schimrigk, K. Absence of elevation of big endothelin in subarachnoid hemorrhage. Stroke 1993, 24, 383–386. [Google Scholar] [CrossRef][Green Version]
- Nozaki, K.; Uemura, Y.; Okamoto, S.; Kikuchi, H.; Mizuno, N. Relaxant effect of calcitonin gene-related peptide on cerebral arterial spasm induced by experimental subarachnoid hemorrhage in dogs. J. Neurosurg. 1989, 71, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Edvinsson, L.; Ekman, R.; Jansen, I.; McCulloch, J.; Mortensen, A.; Uddman, R. Reduced levels of calcitonin gene-related peptide-like immunoreactivity in human brain vessels after subarachnoid haemorrhage. Neurosci. Lett. 1991, 121, 151–154. [Google Scholar] [CrossRef]
- Inoue, T.; Shimizu, H.; Kaminuma, T.; Tajima, M.; Watabe, K.; Yoshimoto, T. Prevention of cerebral vasospasm by calcitonin gene-related peptide slow-release tablet after subarachnoid hemorrhage in monkeys. Neurosurgery 1996, 39, 984–990. [Google Scholar] [CrossRef]
- Sun, B.; Shen, F.; Wu, Q.; Chi, S.; Yang, M.; Yuan, H.; Xie, F.; Zhang, Y.; Chen, J.; Zhang, F. Intranasal delivery of calcitonin gene-related peptide reduces cerebral vasospasm in rats. Front. Biosci. Elite Ed. 2010, 2, 1502–1513. [Google Scholar] [CrossRef]
- Chen, J.; Wang, L.; Xu, H.; Xing, L.; Zhuang, Z.; Zheng, Y.; Li, X.; Wang, C.; Chen, S.; Guo, Z.; et al. Meningeal lymphatics clear erythrocytes that arise from subarachnoid hemorrhage. Nat. Commun. 2020, 11, 3159. [Google Scholar] [CrossRef]
- Wajima, D.; Sato, F.; Kawamura, K.; Sugiura, K.; Nakagawa, I.; Motoyama, Y.; Park, Y.-S.; Nakase, H. Venous or arterial blood components trigger more brain swelling, tissue death after acute subdural hematoma compared to elderly atrophic brain with subdural effusion (SDE) model rats. Brain Res. 2017, 1670, 165–172. [Google Scholar] [CrossRef]
- Solomon, R.A.; Antunes, J.L.; Chen, R.Y.; Bland, L.; Chien, S. Decrease in cerebral blood flow in rats after experimental subarachnoid hemorrhage: A new animal model. Stroke 1985, 16, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Gundersen, H.J.; Bagger, P.; Bendtsen, T.F.; Evans, S.M.; Korbo, L.; Marcussen, N.; Møller, A.; Nielsen, K.; Nyengaard, J.R.; Pakkenberg, B. The new stereological tools: Disector, fractionator, nucleator and point sampled intercepts and their use in pathological research and diagnosis. APMIS Acta Pathol. Microbiol. Immunol. Scand. 1988, 96, 857–881. [Google Scholar] [CrossRef] [PubMed]
- Bangaru, M.L.Y.; Park, F.; Hudmon, A.; McCallum, J.B.; Hogan, Q.H. Quantification of gene expression after painful nerve injury: Validation of optimal reference genes. J. Mol. Neurosci. MN 2012, 46, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods San Diego Calif 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Ebersberger, A.; Averbeck, B.; Messlinger, K.; Reeh, P.W. Release of substance P, calcitonin gene-related peptide and prostaglandin E2 from rat dura mater encephali following electrical and chemical stimulation in vitro. Neuroscience 1999, 89, 901–907. [Google Scholar] [CrossRef]
- Nozaki, K.; Kikuchi, H.; Mizuno, N. Changes of calcitonin gene-related peptide-like immunoreactivity in cerebrovascular nerve fibers in the dog after experimentally produced subarachnoid hemorrhage. Neurosci. Lett. 1989, 102, 27–32. [Google Scholar] [CrossRef]
- Juul, R.; Aakhus, S.; Björnstad, K.; Gisvold, S.E.; Brubakk, A.O.; Edvinsson, L. Calcitonin gene-related peptide (human alpha-CGRP) counteracts vasoconstriction in human subarachnoid haemorrhage. Neurosci. Lett. 1994, 170, 67–70. [Google Scholar] [CrossRef]
- Imaizumi, S.; Shimizu, H.; Ahmad, I.; Kaminuma, T.; Tajima, M.; Yoshimoto, T. Effect of calcitonin gene-related peptide on delayed cerebral vasospasm after experimental subarachnoid hemorrhage in rabbits. Surg. Neurol. 1996, 46, 263–270; discussion 270–271. [Google Scholar] [CrossRef]
- Sun, Y.; Shen, Q.; Watts, L.T.; Muir, E.R.; Huang, S.; Yang, G.-Y.; Suarez, J.I.; Duong, T.Q. Multimodal MRI characterization of experimental subarachnoid hemorrhage. Neuroscience 2016, 316, 53–62. [Google Scholar] [CrossRef]
- Uhl, E.; Lehmberg, J.; Steiger, H.-J.; Messmer, K. Intraoperative detection of early microvasospasm in patients with subarachnoid hemorrhage by using orthogonal polarization spectral imaging. Neurosurgery 2003, 52, 1307–1315; discussion 1315–1317. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.H. Vascular Neural Network in Subarachnoid Hemorrhage. Transl. Stroke Res. 2014, 5, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Poca, M.A.; Sahuquillo, J.; Mataró, M.; Benejam, B.; Arikan, F.; Báguena, M. Ventricular enlargement after moderate or severe head injury: A frequent and neglected problem. J. Neurotrauma 2005, 22, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Shigeno, T.; Fritschka, E.; Brock, M.; Schramm, J.; Shigeno, S.; Cervoś-Navarro, J. Cerebral edema following experimental subarachnoid hemorrhage. Stroke 1982, 13, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, N.M.; Wang, J.Z.; Pasarikovski, C.R.; Guha, D.; Al-Mufti, F.; Mamdani, M.; Saposnik, G.; Schweizer, T.A.; Macdonald, R.L. Management of raised intracranial pressure in aneurysmal subarachnoid hemorrhage: Time for a consensus? Neurosurg. Focus 2017, 43, E13. [Google Scholar] [CrossRef]
- Kamp, M.A.; Dibué, M.; Sommer, C.; Steiger, H.-J.; Schneider, T.; Hänggi, D. Evaluation of a Murine Single-Blood-Injection SAH Model. PLoS ONE 2014, 9, e114946. [Google Scholar] [CrossRef] [PubMed]
- Kaye, A.H.; Tagari, P.C.; Teddy, P.J.; Adams, C.B.; Blaso, W.P.; Boullin, D.J. CSF smooth-muscle constrictor activity associated with cerebral vasospasm and mortality in SAH patients. J. Neurosurg. 1984, 60, 927–934. [Google Scholar] [CrossRef]
- Winslow, N.; Ehsan, M.; Klopfenstein, J. Delayed ischemic neurologic deficit with vasospasm in aneurysmal subarachnoid hemorrhage after negative post-bleed day 7 angiography. Clin. Neurol. Neurosurg. 2022, 220, 107353. [Google Scholar] [CrossRef] [PubMed]
- Juul, R.; Edvinsson, L.; Gisvold, S.E.; Ekman, R.; Brubakk, A.O.; Fredriksen, T.A. Calcitonin gene-related peptide-LI in subarachnoid haemorrhage in man. Signs of activation of the trigemino-cerebrovascular system? Br. J. Neurosurg. 1990, 4, 171–179. [Google Scholar] [CrossRef]
- Schebesch, K.-M.; Herbst, A.; Bele, S.; Schödel, P.; Brawanski, A.; Stoerr, E.-M.; Lohmeier, A.; Kagerbauer, S.M.; Martin, J.; Proescholdt, M. Calcitonin-gene related peptide and cerebral vasospasm. J. Clin. Neurosci. 2013, 20, 584–586. [Google Scholar] [CrossRef]
- Kondziolka, D.; Bernstein, M.; Spiegel, S.M.; ter Brugge, K. Symptomatic arterial luminal narrowing presenting months after subarachnoid hemorrhage and aneurysm clipping. J. Neurosurg. 1988, 69, 494–499. [Google Scholar] [CrossRef] [PubMed]
- Johansson, S.; Abdolalizade, B.; Sheykhzade, M.; Edvinsson, L.; Sams, A. Vascular pathology of large cerebral arteries in experimental subarachnoid hemorrhage: Vasoconstriction, functional CGRP depletion and maintained CGRP sensitivity. Eur. J. Pharmacol. 2019, 846, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Schwarting, J.; Nehrkorn, K.; Liu, H.; Plesnila, N.; Terpolilli, N.A. Role of Pial Microvasospasms and Leukocyte Plugging for Parenchymal Perfusion after Subarachnoid Hemorrhage Assessed by In Vivo Multi-Photon Microscopy. Int. J. Mol. Sci. 2021, 22, 8444. [Google Scholar] [CrossRef] [PubMed]
- Mikhailov, N.; Virenque, A.; Koroleva, K.; Eme-Scolan, E.; Teleman, M.; Abdollahzadeh, A.; Giniatullina, R.; Gafurov, O.; Krivoshein, G.; Malm, T.; et al. The role of the meningeal lymphatic system in local meningeal inflammation and trigeminal nociception. Sci. Rep. 2022, 12, 8804. [Google Scholar] [CrossRef] [PubMed]
- Strassman, A.M.; Mineta, Y.; Vos, B.P. Distribution of fos-like immunoreactivity in the medullary and upper cervical dorsal horn produced by stimulation of dural blood vessels in the rat. J. Neurosci. 1994, 14, 3725–3735. [Google Scholar] [CrossRef]
- Messlinger, K. The big CGRP flood—Sources, sinks and signalling sites in the trigeminovascular system. J. Headache Pain 2018, 19, 22. [Google Scholar] [CrossRef]




| Experimental Group | CTL | SHAM | aCSF | SAH | FG |
|---|---|---|---|---|---|
| surgical procedure | - | + | + | + | + |
| intracisternal injection | - | - | 200 μL aCSF | 200 μL blood | 15 μL FG in aCSF |
| survival time | - | 72 h | 72 h | 72 h | 5 days |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masood, T.; Lakatos, S.; Kis, G.; Ignácz, M.; Domoki, F.; Rosta, J. Subarachnoid Hemorrhage Depletes Calcitonin Gene-Related Peptide Levels of Trigeminal Neurons in Rat Dura Mater. Cells 2024, 13, 653. https://doi.org/10.3390/cells13080653
Masood T, Lakatos S, Kis G, Ignácz M, Domoki F, Rosta J. Subarachnoid Hemorrhage Depletes Calcitonin Gene-Related Peptide Levels of Trigeminal Neurons in Rat Dura Mater. Cells. 2024; 13(8):653. https://doi.org/10.3390/cells13080653
Chicago/Turabian StyleMasood, Thannoon, Szandra Lakatos, Gyöngyi Kis, Melissza Ignácz, Ferenc Domoki, and Judit Rosta. 2024. "Subarachnoid Hemorrhage Depletes Calcitonin Gene-Related Peptide Levels of Trigeminal Neurons in Rat Dura Mater" Cells 13, no. 8: 653. https://doi.org/10.3390/cells13080653
APA StyleMasood, T., Lakatos, S., Kis, G., Ignácz, M., Domoki, F., & Rosta, J. (2024). Subarachnoid Hemorrhage Depletes Calcitonin Gene-Related Peptide Levels of Trigeminal Neurons in Rat Dura Mater. Cells, 13(8), 653. https://doi.org/10.3390/cells13080653






