Role of Dietary Supplements and Probiotics in Modulating Microbiota and Bone Health: The Gut-Bone Axis
Abstract
:1. Introduction
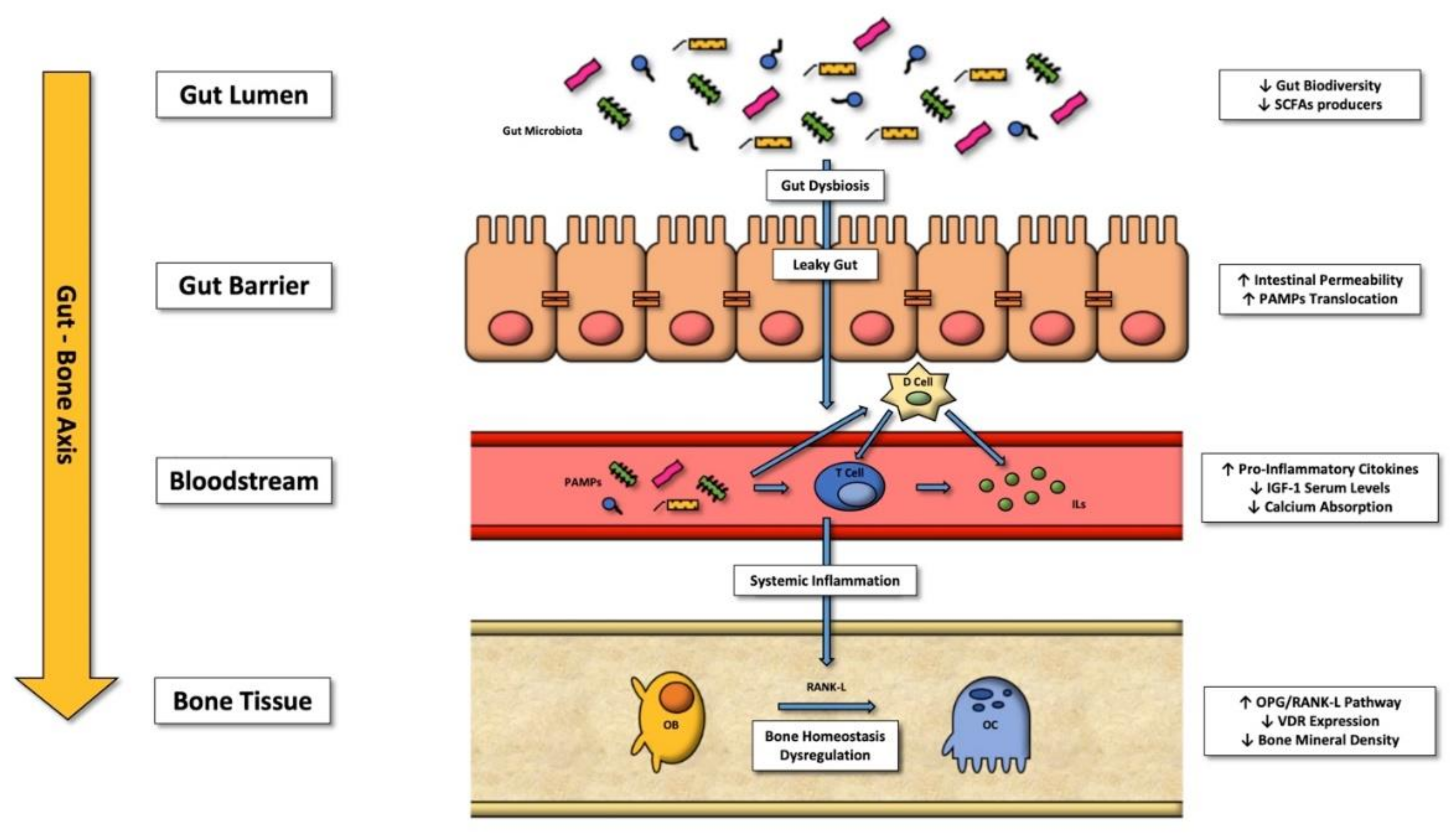
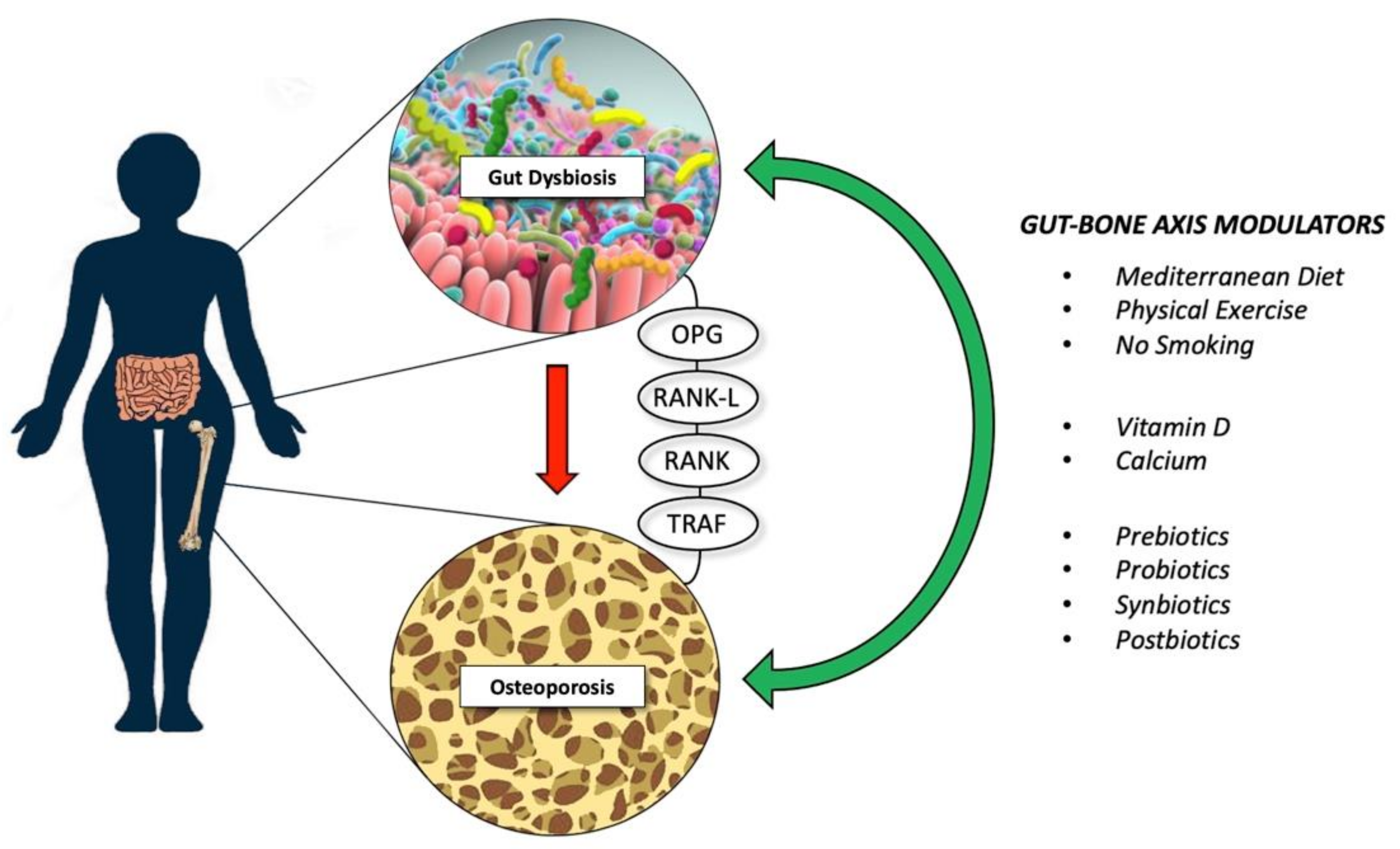
2. The Gut-Bone Axis
3. Effects of Dietary Supplements on the Gut Microbiota and Bone Health
3.1. Roles of Prebiotics
3.2. Proteins, Peptides, and Amino Acids
3.3. Micronutrients
4. Impact of Probiotics on the Gut Microbiota
5. Interventional Microbiota Modulation to Reduce Bone Loss
6. Future Research Avenues and Perspectives for Osteoporosis Treatment
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marini, F.; Cianferotti, L.; Brandi, M.L. Epigenetic Mechanisms in Bone Biology and Osteoporosis: Can They Drive Therapeutic Choices? Int. J. Mol. Sci. 2016, 17, 1329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisani, P.; Renna, M.D.; Conversano, F.; Casciaro, E.; Di Paola, M.; Quarta, E.; Muratore, M.; Casciaro, S. Major osteoporotic fragility fractures: Risk factor updates and societal impact. World J. Orthop. 2016, 7, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Norton, N.; Harvey, N.C.; Jacobson, T.; Johansson, H.; Lorentzon, M.; McCloskey, E.V.; Willers, C.; Borgström, F. SCOPE 2021: A new scorecard for osteoporosis in Europe. Arch. Osteoporos. 2021, 16, 82. [Google Scholar] [CrossRef] [PubMed]
- Compston, J.E.; McClung, M.R.; Leslie, W.D. Osteoporosis. Lancet 2019, 393, 364–376. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, Y.; Wang, Y.; Ren, X.; Han, J. The impact of the intestinal microbiome on bone health. Intractable Rare Dis. Res. 2018, 7, 148–155. [Google Scholar] [CrossRef] [Green Version]
- Peterson, J.; Garges, S.; Giovanni, M.; McInnes, P.; Wang, L.; Schloss, J.A.; Bonazzi, V.; McEwen, J.E.; Wetterstrand, K.A.; et al.; NIH HMP Working Group The NIH Human Microbiome Project. Genome Res. 2009, 19, 2317–2323. [Google Scholar] [CrossRef] [Green Version]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagpal, R.; Yadav, H.; Marotta, F. Gut microbiota: The next-gen frontier in preventive and therapeutic medicine? Front. Med. 2014, 1, 15. [Google Scholar] [CrossRef] [Green Version]
- Frost, F.; Kacprowski, T.; Rühlemann, M.; Pietzner, M.; Bang, C.; Franke, A.; Nauck, M.; Völker, U.; Völzke, H.; Dörr, M.; et al. Long-term instability of the intestinal microbiome is associated with metabolic liver disease, low microbiota diversity, diabetes mellitus and impaired exocrine pancreatic function. Gut 2021, 70, 522–530. [Google Scholar] [CrossRef]
- Wright, E.K.; Kamm, M.A.; Teo, S.M.; Inouye, M.; Wagner, J.; Kirkwood, C.D. Recent advances in characterizing the gastrointestinal microbiome in Crohn’s disease: A systematic review. Inflamm. Bowel Dis. 2015, 21, 1219–1228. [Google Scholar] [CrossRef] [Green Version]
- Ussar, S.; Fujisaka, S.; Kahn, C.R. Interactions between host genetics and gut microbiome in diabetes and metabolic syndrome. Mol. Metab. 2016, 5, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Minemura, M.; Shimizu, Y. Gut microbiota and liver diseases. World J. Gastroenterol. 2015, 21, 1691–1702. [Google Scholar] [CrossRef] [PubMed]
- Pevsner-Fischer, M.; Blacher, E.; Tatirovsky, E.; Ben-Dov, I.Z.; Elinav, E. The gut microbiome and hypertension. Curr. Opin. Nephrol. Hypertens. 2017, 26, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rosser, E.C.; Mauri, C. A clinical update on the significance of the gut microbiota in systemic autoimmunity. J. Autoimmun. 2016, 74, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, D.; Kubota, R.; Maeno, T.; Abdelhakim, M.; Hitosugi, N. Association between gut microbiota, bone metabolism, and fracture risk in postmenopausal Japanese women. Osteoporos. Int. 2021, 32, 145–156. [Google Scholar] [CrossRef]
- McKnite, A.M.; Perez-Munoz, M.E.; Lu, L.; Williams, E.G.; Brewer, S.; Andreux, P.A.; Bastiaansen, J.W.; Wang, X.; Kachman, S.D.; Auwerx, J.; et al. Murine gut microbiota is defined by host genetics and modulates variation of metabolic traits. PLoS ONE 2012, 7, e39191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, J.; Herzog, J.W.; Tsang, K.; Brennan, C.A.; Bower, M.A.; Garrett, W.S.; Sartor, B.R.; Aliprantis, A.O.; Charles, J.F. Gut microbiota induce IGF-1 and promote bone formation and growth. Proc. Natl. Acad. Sci. USA 2016, 113, E7554–E7563. [Google Scholar] [CrossRef] [Green Version]
- Hathaway-Schrader, J.D.; Poulides, N.A.; Carson, M.D.; Kirkpatrick, J.E.; Warner, A.J.; Swanson, B.A.; Taylor, E.V.; Chew, M.E.; Reddy, S.V.; Liu, B.; et al. Specific Commensal Bacterium Critically Regulates Gut Microbiota Osteoimmunomodulatory Actions During Normal Postpubertal Skeletal Growth and Maturation. JBMR Plus 2020, 4, e10338. [Google Scholar] [CrossRef]
- Castaneda, M.; Smith, K.M.; Nixon, J.C.; Hernandez, C.J.; Rowan, S. Alterations to the gut microbiome impair bone tissue strength in aged mice. Bone Rep. 2021, 14, 101065. [Google Scholar] [CrossRef]
- Novince, C.M.; Whittow, C.R.; Aartun, J.D.; Hathaway, J.D.; Poulides, N.; Chavez, M.B.; Steinkamp, H.M.; Kirkwood, K.A.; Huang, E.; Westwater, C.; et al. Commensal Gut Microbiota Immunomodulatory Actions in Bone Marrow and Liver have Catabolic Effects on Skeletal Homeostasis in Health. Sci. Rep. 2017, 7, 5747. [Google Scholar] [CrossRef] [Green Version]
- Lahiri, S.; Kim, H.; Garcia-Perez, I.; Reza, M.M.; Martin, K.A.; Kundu, P.; Cox, L.M.; Selkrig, J.; Posma, J.M.; Zhang, H.; et al. The gut microbiota influences skeletal muscle mass and function in mice. Sci. Transl. Med. 2019, 11, eaan5662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manickam, R.; Oh, H.Y.P.; Tan, C.K.; Paramalingam, E.; Wahli, W. Metronidazole Causes Skeletal Muscle Atrophy and Modulates Muscle Chronometabolism. Int. J. Mol. Sci. 2018, 19, 2418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ratajczak, A.E.; Rychter, A.M.; Zawada, A.; Dobrowolska, A.; Krela-Kaźmierczak, I. Do Only Calcium and Vitamin D Matter? Micronutrients in the Diet of Inflammatory Bowel Diseases Patients and the Risk of Osteoporosis. Nutrients 2021, 13, 525. [Google Scholar] [CrossRef] [PubMed]
- Behera, J.; Ison, J.; Tyagi, S.C.; Tyagi, N. The role of gut microbiota in bone homeostasis. Bone 2020, 135, 115317. [Google Scholar] [CrossRef] [PubMed]
- Floch, M.H. Probiotics and Prebiotics. Gastroenterol. Hepatol. 2014, 10, 680–681. [Google Scholar]
- Rizzoli, R. Microbiota and Bone Health: The Gut-Musculoskeletal Axis. Calcif. Tissue Int. 2018, 102, 385–386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkïla, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Heiskanen, J.T.; Kröger, H.; Pääkkönen, M.; Parviainen, M.T.; Lamberg- Allardt, C.; Alhava, E. Bone mineral metabolism after total gastrectomy. Bone 2001, 28, 123–127. [Google Scholar] [CrossRef]
- Tovey, F.I.; Hall, M.L.; Ell, P.J.; Hobsley, M. A review of postgastrectomy bone disease. J. Gastroenterol. Hepatol. 1992, 7, 639–645. [Google Scholar] [CrossRef]
- Schinke, T.; Schilling, A.F.; Baranowsky, A.; Seitz, S.; Marshall, R.P.; Linn, T.; Blaeker, M.; Huebner, A.K.; Schulz, A.; Simon, R.; et al. Impaired gastric acidification negatively affects calcium homeostasis and bone mass. Nat. Med. 2009, 15, 674–681. [Google Scholar] [CrossRef]
- Hansen, M.S.; Frost, M. Alliances of the gut and bone axis. Semin. Cell Dev. Biol. 2021, 123, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Karsenty, G.; Yadav, V.K. Regulation of bone mass by serotonin: Molecular biology and therapeutic implications. Annu. Rev. Med. 2011, 62, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Greenbaum, J.; Shen, H.; Deng, H.W. Association Between Gut Microbiota and Bone Health: Potential Mechanisms and Prospective. J. Clin. Endocrinol. Metab. 2017, 102, 3635–3646. [Google Scholar] [CrossRef] [PubMed]
- Peterson, C.T.; Sharma, V.; Elmén, L.; Peterson, S.N. Immune homeostasis, dysbiosis and therapeutic modulation of the gut microbiota. Clin. Exp. Immunol. 2015, 179, 363–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, M.; Cronin, O.; Keohane, D.M.; Cormac, E.M.; Nugent, H.; Nugent, M.; Molloy, C.; O’Toole, P.W.; Shanahan, F.; Molloy, M.G.; et al. Gut microbiota alterations associated with reduced bone mineral density in older adults. Rheumatology 2019, 58, 2295–2304. [Google Scholar] [CrossRef] [Green Version]
- Rafii, F. The role of colonic bacteria in the metabolism of the natural isoflavone daidzin to equol. Metabolites 2015, 5, 56–73. [Google Scholar] [CrossRef] [Green Version]
- Tu, Y.; Yang, R.; Xu, X.; Zhou, X. The microbiota-gut-bone axis and bone health. J. Leukoc. Biol. 2021, 110, 525–537. [Google Scholar] [CrossRef]
- Liu, Y.; Guo, Y.L.; Meng, S.; Gao, H.; Sui, L.J.; Jin, S.; Li, Y.; Fan, S.G. Gut microbiota-dependent trimethylamine N-Oxide are related with hip fracture in postmenopausal women: A matched case-control study. Aging 2020, 12, 10633–10641. [Google Scholar] [CrossRef]
- Claesson, M.J.; Jeffery, I.B.; Conde, S.; Power, S.E.; O’Connor, E.M.; Cusack, S.; Harris, H.M.; Coakley, M.; Lakshminarayanan, B.; O’Sullivan, O.; et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 2012, 488, 178–184. [Google Scholar] [CrossRef]
- Suez, J.; Korem, T.; Zeevi, D.; Zilberman-Schapira, G.; Thaiss, C.A.; Maza, O.; Israeli, D.; Zmora, N.; Gilad, S.; Weinberger, A.; et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 2014, 514, 181–186. [Google Scholar] [CrossRef]
- Gentile, C.L.; Weir, T.L. The gut microbiota at the intersection of diet and human health. Science 2018, 362, 776–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Byberg, L.; Bellavia, A.; Larsson, S.C.; Orsini, N.; Wolk, A.; Michaëlsson, K. Mediterranean Diet and Hip Fracture in Swedish Men and Women. J. Bone Miner. Res. 2016, 31, 2098–2105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef]
- Palomeras-Vilches, A.; Viñals-Mayolas, E.; Bou-Mias, C.; Jordà-Castro, M.; Agüero-Martínez, M.; Busquets-Barceló, M.; Pujol-Busquets, G.; Carrion, C.; Bosque-Prous, M.; Serra-Majem, L.; et al. Adherence to the Mediterranean Diet and Bone Fracture Risk in Middle-Aged Women: A Case Control Study. Nutrients 2019, 11, 2508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noel, S.E.; Mangano, K.M.; Mattei, J.; Griffith, J.L.; Dawson-Hughes, B.; Bigornia, S.; Tucker, K.L. Dietary Approaches to Stop Hypertension, Mediterranean, and Alternative Healthy Eating indices are associated with bone health among Puerto Rican adults from the Boston Puerto Rican Osteoporosis Study. Am. J. Clin. Nutr. 2020, 111, 1267–1277. [Google Scholar] [CrossRef]
- Zaiss, M.M.; Jones, R.M.; Schett, G.; Pacifici, R. The gut-bone axis: How bacterial metabolites bridge the distance. J. Clin. Investig. 2019, 129, 3018–3028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whisner, C.M.; Castillo, L.F. Prebiotics, Bone and Mineral Metabolism. Calcif. Tissue Int. 2018, 102, 443–479. [Google Scholar] [CrossRef] [Green Version]
- Schepper, J.D.; Collins, F.L.; Rios-Arce, N.D.; Raehtz, S.; Schaefer, L.; Gardinier, J.D.; Britton, R.A.; Parameswaran, N.; McCabe, L.R. Probiotic Lactobacillus reuteri Prevents Postantibiotic Bone Loss by Reducing Intestinal Dysbiosis and Preventing Barrier Disruption. J. Bone Min. Res. 2019, 34, 681–698. [Google Scholar] [CrossRef]
- Zhang, J.; Motyl, K.J.; Irwin, R.; MacDougald, O.A.; Britton, R.A.; McCabe, L.R. Loss of Bone and Wnt10b Expression in Male Type 1 Diabetic Mice Is Blocked by the Probiotic Lactobacillus reuteri. Endocrinology 2015, 156, 3169–3182. [Google Scholar] [CrossRef] [Green Version]
- Weaver, C.M.; Martin, B.R.; Nakatsu, C.H.; Armstrong, A.P.; Clavijo, A.; McCabe, L.D.; McCabe, G.P.; Duignan, S.; Schoterman, M.H.; van den Heuvel, E.G. Galactooligosaccharides improve mineral absorption and bone properties in growing rats through gut fermentation. J. Agric. Food Chem. 2011, 59, 6501–6510. [Google Scholar] [CrossRef]
- Van den Heuvel, E.G.; Schoterman, M.H.; Muijs, T. Transgalactooligosaccharides stimulate calcium absorption in postmenopausal women. J. Nutr. 2000, 130, 2938–2942. [Google Scholar] [CrossRef] [PubMed]
- Whisner, C.M.; Martin, B.R.; Schoterman, M.H.; Nakatsu, C.H.; McCabe, L.D.; McCabe, G.P.; Wastney, M.E.; van den Heuvel, E.G.; Weaver, C.M. Galacto-oligosaccharides increase calcium absorption and gut bifidobacteria in young girls: A double-blind cross-over trial. Br. J. Nutr. 2013, 110, 1292–1303. [Google Scholar] [CrossRef] [PubMed]
- Bornet, F.R.; Brouns, F.; Tashiro, Y.; Duvillier, V. Nutritional aspects of short-chain fructooligosaccharides: Natural occurrence, chemistry, physiology and health implications. Dig. Liver Dis. 2002, 34, S111–S120. [Google Scholar] [CrossRef]
- Yan, C.; Zhang, S.; Wang, C.; Zhang, Q. A fructooligosaccharide from Achyranthes bidentata inhibits osteoporosis by stimulating bone formation. Carbohydr. Polym. 2019, 210, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Mathey, J.; Puel, C.; Kati-Coulibaly, S.; Bennetau-Pelissero, C.; Davicco, M.J.; Lebecque, P.; Horcajada, M.N.; Coxam, V. Fructooligosaccharides maximize bone-sparing effects of soy isoflavone-enriched diet in the ovariectomized rat. Calcif. Tissue Int. 2004, 75, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, K.; Nakamura, S.; Moriyama-Hashiguchi, M.; Kitajima, M.; Ejima, H.; Imori, C.; Oku, T. Dietary Fructooligosaccharide and Glucomannan Alter Gut Microbiota and Improve Bone Metabolism in Senescence-Accelerated Mouse. J. Agric. Food Chem. 2019, 67, 867–874. [Google Scholar] [CrossRef]
- Porwal, K.; Pal, S.; Kulkarni, C.; Singh, P.; Sharma, S.; Singh, P.; Prajapati, G.; Gayen, J.R.; Ampapathi, R.S.; Mullick, A.; et al. A prebiotic, short-chain fructo-oligosaccharides promotes peak bone mass and maintains bone mass in ovariectomized rats by an osteogenic mechanism. Biomed. Pharmacother. 2020, 129, 110448. [Google Scholar] [CrossRef]
- Slevin, M.M.; Allsopp, P.J.; Magee, P.J.; Bonham, M.P.; Naughton, V.R.; Strain, J.J.; Duffy, M.E.; Wallace, J.M.; Mc Sorley, E.M. Supplementation with calcium and short-chain fructo-oligosaccharides affects markers of bone turnover but not bone mineral density in postmenopausal women. J. Nutr. 2014, 144, 297–304. [Google Scholar] [CrossRef] [Green Version]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef]
- Tousen, Y.; Abe, F.; Ishida, T.; Uehara, M.; Ishimi, Y. Resistant starch promotes equol production and inhibits tibial bone loss in ovariectomized mice treated with daidzein. Metabolism 2011, 60, 1425–1432. [Google Scholar] [CrossRef]
- Tousen, Y.; Matsumoto, Y.; Matsumoto, C.; Nishide, Y.; Nagahata, Y.; Kobayashi, I.; Ishimi, Y. The combined effects of soya isoflavones and resistant starch on equol production and trabecular bone loss in ovariectomised mice. Br. J. Nutr. 2016, 116, 247–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tousen, Y.; Matsumoto, Y.; Nagahata, Y.; Kobayashi, I.; Inoue, M.; Ishimi, Y. Resistant Starch Attenuates Bone Loss in Ovariectomised Mice by Regulating the Intestinal Microbiota and Bone-Marrow Inflammation. Nutrients 2019, 11, 297. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Summanen, P.H.; Komoriya, T.; Finegold, S.M. In vitro study of the prebiotic xylooligosaccharide (XOS) on the growth of Bifidobacterium spp and Lactobacillus spp. Int. J. Food Sci. Nutr. 2015, 66, 919–922. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Zhou, Z. Effect of Xylo-Oligosaccharides Supplementation by Drinking Water on the Bone Properties and Related Calcium Transporters in Growing Mice. Nutrients 2020, 12, 3542. [Google Scholar] [CrossRef]
- Eaimworawuthikul, S.; Tunapong, W.; Chunchai, T.; Suntornsaratoon, P.; Charoenphandhu, N.; Thiennimitr, P.; Chattipakorn, N.; Chattipakorn, S.C. Altered gut microbiota ameliorates bone pathology in the mandible of obese-insulin-resistant rats. Eur. J. Nutr. 2020, 59, 1453–1462. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Z.; Hu, Y.; Cui, J.; Zhi, X.; Li, X.; Jiang, H.; Wang, Y.; Gu, Z.; Qiu, Z.; et al. Lactulose Suppresses Osteoclastogenesis and Ameliorates Estrogen Deficiency-Induced Bone Loss in Mice. Aging Dis. 2020, 11, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Karakan, T.; Tuohy, K.M.; Janssen-van Solingen, G. Low-Dose Lactulose as a Prebiotic for Improved Gut Health and Enhanced Mineral Absorption. Front. Nutr. 2021, 8, 672925. [Google Scholar] [CrossRef] [PubMed]
- Seki, N.; Hamano, H.; Iiyama, Y.; Asano, Y.; Kokubo, S.; Yamauchi, K.; Tamura, Y.; Uenishi, K.; Kudou, H. Effect of lactulose on calcium and magnesium absorption: A study using stable isotopes in adult men. J. Nutr. Sci. Vitam. 2007, 53, 5–12. [Google Scholar] [CrossRef] [Green Version]
- Scholz-Ahrens, K.E.; Schaafsma, G.; van den Heuvel, E.G.; Schrezenmeir, J. Effects of prebiotics on mineral metabolism. Am. J. Clin. Nutr. 2001, 73 (Suppl. S2), 459S–464S. [Google Scholar] [CrossRef] [Green Version]
- McCabe, L.; Britton, R.A.; Parameswaran, N. Prebiotic and Probiotic Regulation of Bone Health: Role of the Intestine and its Microbiome. Curr. Osteoporos. Rep. 2015, 13, 363–371. [Google Scholar] [CrossRef] [Green Version]
- Wolever, T.M.; Trinidad, T.P.; Thompson, L.U. Short chain fatty acid absorption from the human distal colon: Interactions between acetate, propionate and calcium. J. Am. Coll. Nutr. 1995, 14, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Nath, A.; Molnár, M.A.; Csighy, A.; Kőszegi, K.; Galambos, I.; Huszár, K.P.; Koris, A.; Vatai, G. Biological Activities of Lactose-Based Prebiotics and Symbiosis with Probiotics on Controlling Osteoporosis, Blood-Lipid and Glucose Levels. Medicina 2018, 54, 98. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Sun, S.; Guo, B.; Miao, B.; Luo, Z.; Xia, Z.; Ying, D.; Liu, F.; Guo, B.; Tang, J.; et al. Bioactive peptide isolated from casein phosphopeptides promotes calcium uptake in vitro and in vivo. Food Funct. 2018, 9, 2251–2260. [Google Scholar] [CrossRef] [PubMed]
- Dawson-Hughes, B.; Harris, S.S.; Rasmussen, H.M.; Dallal, G.E. Comparative effects of oral aromatic and branched-chain amino acids on urine calcium excretion in humans. Osteoporos. Int. 2007, 18, 955–961. [Google Scholar] [CrossRef]
- Biver, E.; Durosier-Izart, C.; Merminod, F.; Chevalley, T.; van Rietbergen, B.; Ferrari, S.L.; Rizzoli, R. Fermented dairy products consumption is associated with attenuated cortical bone loss independently of total calcium, protein, and energy intakes in healthy postmenopausal women. Osteoporos. Int. 2018, 29, 1771–1782. [Google Scholar] [CrossRef]
- Laird, E.; Molloy, A.M.; McNulty, H.; Ward, M.; McCarroll, K.; Hoey, L.; Hughes, C.F.; Cunningham, C.; Strain, J.J.; Casey, M.C. Greater yogurt consumption is associated with increased bone mineral density and physical function in older adults. Osteoporos. Int. 2017, 28, 2409–2419. [Google Scholar] [CrossRef]
- Ong, A.M.; Kang, K.; Weiler, H.A.; Morin, S.N. Fermented Milk Products and Bone Health in Postmenopausal Women: A Systematic Review of Randomized Controlled Trials, Prospective Cohorts, and Case-Control Studies. Adv. Nutr. 2020, 11, 251–265. [Google Scholar] [CrossRef]
- Rizzoli, R. Dairy products and bone health. Aging Clin. Exp. Res. 2022, 34, 9–24. [Google Scholar] [CrossRef]
- Chen, H.L.; Tung, Y.T.; Chuang, C.H.; Tu, M.Y.; Tsai, T.C.; Chang, S.Y.; Chen, C.M. Kefir improves bone mass and microarchitecture in an ovariectomized rat model of postmenopausal osteoporosis. Osteoporos. Int. 2015, 26, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.Y.; Han, K.Y.; Chang, G.R.; Lai, G.D.; Chang, K.Y.; Chen, C.F.; Lai, J.C.; Lai, C.Y.; Chen, H.L.; Chen, C.M. Kefir Peptides Prevent Estrogen Deficiency-Induced Bone Loss and Modulate the Structure of the Gut Microbiota in Ovariectomized Mice. Nutrients 2020, 12, 3432. [Google Scholar] [CrossRef] [PubMed]
- Diether, N.E.; Willing, B.P. Microbial Fermentation of Dietary Protein: An Important Factor in Diet-Microbe-Host Interaction. Microorganisms 2019, 7, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, Y.; Xie, C.; Li, X.; Yang, J.; Yu, T.; Zhang, R.; Zhang, T.; Saxena, D.; Snyder, M.; Wu, Y.; et al. Succinate and its G-protein-coupled receptor stimulates osteoclastogenesis. Nat. Commun. 2017, 8, 15621. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhang, L.; Xia, B.; Tang, S.; Liu, L.; Xie, J.; Zhang, H. Bioregional Alterations in Gut Microbiome Contribute to the Plasma Metabolomic Changes in Pigs Fed with Inulin. Microorganisms 2020, 8, 111. [Google Scholar] [CrossRef] [Green Version]
- Jennings, A.; MacGregor, A.; Spector, T.; Cassidy, A. Amino Acid Intakes Are Associated with Bone Mineral Density and Prevalence of Low Bone Mass in Women: Evidence from Discordant Monozygotic Twins. J. Bone Min. Res. 2016, 31, 326–335. [Google Scholar] [CrossRef] [Green Version]
- Ling, C.W.; Miao, Z.; Xiao, M.L.; Zhou, H.; Jiang, Z.; Fu, Y.; Xiong, F.; Zuo, L.S.; Liu, Y.P.; Wu, Y.Y.; et al. The Association of Gut Microbiota with Osteoporosis Is Mediated by Amino Acid Metabolism: Multiomics in a Large Cohort. J. Clin. Endocrinol. Metab. 2021, 106, e3852–e3864. [Google Scholar] [CrossRef]
- Karaaslan, F.; Mutlu, M.; Mermerkaya, M.U.; Karaoğlu, S.; Saçmaci, Ş.; Kartal, Ş. Comparison of bone tissue trace-element concentrations and mineral density in osteoporotic femoral neck fractures and osteoarthritis. Clin. Interv. Aging 2014, 9, 1375–1382. [Google Scholar] [CrossRef] [Green Version]
- Rayman, M.P. Selenium intake, status, and health: A complex relationship. Hormones 2020, 19, 9–14. [Google Scholar] [CrossRef] [Green Version]
- Beukhof, C.M.; Medici, M.; van den Beld, A.W.; Hollenbach, B.; Hoeg, A.; Visser, W.E.; de Herder, W.W.; Visser, T.J.; Schomburg, L.; Peeters, R.P. Selenium Status Is Positively Associated with Bone Mineral Density in Healthy Aging European Men. PLoS ONE 2016, 11, e0152748. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Xie, D.; Li, J.; Long, H.; Wu, J.; Wu, Z.; He, H.; Wang, H.; Yang, T.; Wang, Y. Association between dietary selenium intake and the prevalence of osteoporosis: A cross-sectional study. BMC Musculoskelet. Disord. 2019, 20, 585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasaikina, M.V.; Kravtsova, M.A.; Lee, B.C.; Seravalli, J.; Peterson, D.A.; Walter, J.; Legge, R.; Benson, A.K.; Hatfield, D.L.; Gladyshev, V.N. Dietary selenium affects host selenoproteome expression by influencing the gut microbiota. FASEB J. 2011, 25, 2492–2499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palacios, C. The role of nutrients in bone health, from A to Z. Crit. Rev. Food Sci. Nutr. 2006, 46, 621–628. [Google Scholar] [CrossRef]
- Mutlu, M.; Argun, M.; Kilic, E.; Saraymen, R.; Yazar, S. Magnesium, zinc and copper status in osteoporotic, osteopenic and normal post-menopausal women. J. Int. Med. Res. 2007, 35, 692–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Starke, I.C.; Pieper, R.; Neumann, K.; Zentek, J.; Vahjen, W. The impact of high dietary zinc oxide on the development of the intestinal microbiota in weaned piglets. FEMS Microbiol. Ecol. 2014, 87, 416–427. [Google Scholar] [CrossRef] [Green Version]
- Reed, S.; Neuman, H.; Moscovich, S.; Glahn, R.P.; Koren, O.; Tako, E. Chronic Zinc Deficiency Alters Chick Gut Microbiota Composition and Function. Nutrients 2015, 7, 9768–9784. [Google Scholar] [CrossRef]
- Balogh, E.; Paragh, G.; Jeney, V. Influence of Iron on Bone Homeostasis. Pharmaceuticals 2018, 11, 107. [Google Scholar] [CrossRef] [Green Version]
- Imel, E.A.; Liu, Z.; McQueen, A.K.; Acton, D.; Acton, A.; Padgett, L.R.; Peacock, M.; Econs, M.J. Serum fibroblast growth factor 23, serum iron and bone mineral density in premenopausal women. Bone 2016, 86, 98–105. [Google Scholar] [CrossRef] [Green Version]
- Hu, L.; Andersson, G.; Jonsson, K.B.; Melhus, H.; Lind, T. Adamts1 is highly induced in rachitic bones of FGF23 transgenic mice and participates in degradation of non-mineralized bone matrix collagen. Biochem. Biophys. Res. Commun. 2013, 430, 901–906. [Google Scholar] [CrossRef]
- Kaneko, I.; Segawa, H.; Ikuta, K.; Hanazaki, A.; Fujii, T.; Tatsumi, S.; Kido, S.; Hasegawa, T.; Amizuka, N.; Saito, H.; et al. Eldecalcitol Causes FGF23 Resistance for Pi Reabsorption and Improves Rachitic Bone Phenotypes in the Male Hyp Mouse. Endocrinology 2018, 159, 2741–2758. [Google Scholar] [CrossRef]
- Harris, M.M.; Houtkooper, L.B.; Stanford, V.A.; Parkhill, C.; Weber, J.L.; Flint-Wagner, H.; Weiss, L.; Going, S.B.; Lohman, T.G. Dietary iron is associated with bone mineral density in healthy postmenopausal women. J. Nutr. 2003, 133, 3598–3602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qasem, W.; Azad, M.B.; Hossain, Z.; Azad, E.; Jorgensen, S.; Castillo San Juan, S.; Cai, C.; Khafipour, E.; Beta, T.; Roberts, L.J., 2nd; et al. Assessment of complementary feeding of Canadian infants: Effects on microbiome & oxidative stress, a randomized controlled trial. BMC Pediatr. 2017, 17, 54. [Google Scholar] [CrossRef] [Green Version]
- Seura, T.; Yoshino, Y.; Fukuwatari, T. The Relationship between Habitual Dietary Intake and Gut Microbiota in Young Japanese Women. J. Nutr. Sci. Vitam. 2017, 63, 396–404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, S.; Zhuo, Z.; Yu, X.; Wang, H.; Feng, J. Oral administration of liquid iron preparation containing excess iron induces intestine and liver injury, impairs intestinal barrier function and alters the gut microbiota in rats. J. Trace Elem. Med. Biol. 2018, 47, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Kortman, G.A.; Dutilh, B.E.; Maathuis, A.J.; Engelke, U.F.; Boekhorst, J.; Keegan, K.P.; Nielsen, F.G.; Betley, J.; Weir, J.C.; Kingsbury, Z.; et al. Microbial Metabolism Shifts Towards an Adverse Profile with Supplementary Iron in the TIM-2 In vitro Model of the Human Colon. Front. Microbiol. 2016, 6, 1481. [Google Scholar] [CrossRef] [Green Version]
- Araya, M.; Morelli, L.; Reid, G.; Sanders, M.; Stanton, C. Guidelines for the Evaluation of Probiotics in Food; FAO; WHO Working Group: London, ON, Canada, 2002; Available online: https://www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf (accessed on 18 December 2021).
- Collins, F.L.; Rios-Arce, N.D.; Schepper, J.D.; Parameswaran, N.; McCabe, L.R. The Potential of Probiotics as a Therapy for Osteoporosis. Microbiol. Spectr. 2017, 5, 4. [Google Scholar] [CrossRef]
- Rizzoli, R.; Biver, E. Are Probiotics the New Calcium and Vitamin D for Bone Health? Curr. Osteoporos. Rep. 2020, 18, 273–284. [Google Scholar] [CrossRef]
- Wieërs, G.; Belkhir, L.; Enaud, R.; Leclercq, S.; Philippart de Foy, J.M.; Dequenne, I.; de Timary, P.; Cani, P.D. How Probiotics Affect the Microbiota. Front. Cell Infect. Microbiol. 2020, 9, 454. [Google Scholar] [CrossRef] [Green Version]
- Hill, C.; Guarner, F.; Reid, G. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [Green Version]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Satokari, R. Modulation of Gut Microbiota for Health by Current and Next-Generation Probiotics. Nutrients 2019, 8, 1921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Britton, R.A.; Irwin, R.; Quach, D.; Schaefer, L.; Zhang, J.; Lee, T.; Parameswaran, N.; McCabe, L.R. Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model. J. Cell Physiol. 2014, 229, 1822–1830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, S.S.; Pan, T.M. Antiosteoporotic effects of Lactobacillus-fermented soy skim milk on bone mineral density and the microstructure of femoral bone in ovariectomized mice. J. Agric. Food Chem. 2011, 59, 7734–7742. [Google Scholar] [CrossRef] [PubMed]
- Narva, M.; Rissanen, J.; Halleen, J.; Vapaatalo, H.; Vaananen, K.; Korpela, R. Effects of bioactive peptide, valyl-prolyl-proline (VPP), and Lactobacillus helveticus fermented milk containing VPP on bone loss in ovariectomized rats. Ann. Nutr. Metab. 2007, 51, 65–74. [Google Scholar] [CrossRef]
- Ohlsson, C.; Engdahl, C.; Fak, F.; Andersson, A.; Windahl, S.H.; Farman, H.H.; Movérare-Skrtic, S.; Islander, U.; Sjögren, K. Probiotics protect mice from ovariectomy-induced cortical bone loss. PLoS ONE 2014, 9, e92368. [Google Scholar] [CrossRef]
- Malmir, H.; Ejtahed, H.S.; Soroush, A.R.; Mortazavian, A.M.; Fahimfar, N.; Ostovar, A.; Esmaillzadeh, A.; Larijani, B.; Hasani-Ranjbar, S. Probiotics as a New Regulator for Bone Health: A Systematic Review and Meta-Analysis. Evid.-Based Complement. Altern. Med. 2021, 2021, 3582989. [Google Scholar] [CrossRef]
- Schepper, J.D.; Collins, F.; Rios-Arce, N.D.; Kang, H.J.; Schaefer, L.; Gardinier, J.D.; Raghuvanshi, R.; Quinn, R.A.; Britton, R.; Parameswaran, N.; et al. Involvement of the Gut Microbiota and Barrier Function in Glucocorticoid-Induced Osteoporosis. J. Bone Min. Res. 2020, 35, 801–820. [Google Scholar] [CrossRef]
- Bennett, C.N.; Longo, K.A.; Wright, W.S.; Suva, L.J.; Lane, T.F.; Hankenson, K.D.; MacDougald, O.A. Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc. Natl. Acad. Sci. USA 2005, 102, 3324–3329. [Google Scholar] [CrossRef] [Green Version]
- Jafarnejad, S.; Djafarian, K.; Fazeli, M.R.; Yekaninejad, M.S.; Rostamian, A.; Keshavarz, S.A. Effects of a Multispecies Probiotic Supplement on Bone Health in Osteopenic Postmenopausal Women: A Randomized, Double-blind, Controlled Trial. J. Am. Coll. Nutr. 2017, 36, 497–506. [Google Scholar] [CrossRef]
- Lambert, M.N.T.; Thybo, C.B.; Lykkeboe, S.; Rasmussen, L.M.; Frette, X.; Christensen, L.P.; Jeppesen, P.B. Combined bioavailable isoflavones and probiotics improve bone status and estrogen metabolism in postmenopausal osteopenic women: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 106, 909–920. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, A.G.; Sundh, D.; Bäckhed, F.; Lorentzon, M. Lactobacillus reuteri reduces bone loss in older women with low bone mineral density: A randomized, placebo-controlled, double-blind, clinical trial. J. Intern. Med. 2018, 284, 307–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seely, K.D.; Kotelko, C.A.; Douglas, H.; Bealer, B.; Brooks, A.E. The Human Gut Microbiota: A Key Mediator of Osteoporosis and Osteogenesis. Int. J. Mol. Sci. 2021, 22, 9452. [Google Scholar] [CrossRef]
- Felizardo, R.J.F.; Watanabe, I.K.M.; Dardi, P.; Rossoni, L.V.; Câmara, N.O.S. The interplay among gut microbiota, hypertension and kidney diseases: The role of short-chain fatty acids. Pharmacol. Res. 2019, 141, 366–377. [Google Scholar] [CrossRef]
- Gimigliano, F.; Moretti, A.; de Sire, A.; Calafiore, D.; Iolascon, G. The combination of vitamin D deficiency and overweight affects muscle mass and function in older post-menopausal women. Aging Clin. Exp. Res. 2018, 30, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Quach, D.; Britton, R.A. Gut Microbiota and Bone Health. Adv. Exp. Med. Biol. 2017, 1033, 47–58. [Google Scholar] [CrossRef] [PubMed]
- de Sire, R.; Talocco, C.; Petito, V.; Lopetuso, L.R.; Graziani, C.; Gasbarrini, A.; Scaldaferri, F. Microbiota and inflammatory bowel disease: An update. Recenti Progress. Med. 2018, 109, 570–573. [Google Scholar] [CrossRef] [PubMed]
- de Sire, A.; de Sire, R.; Petito, V.; Masi, L.; Cisari, C.; Gasbarrini, A.; Scaldaferri, F.; Invernizzi, M. Gut-Joint Axis: The Role of Physical Exercise on Gut Microbiota Modulation in Older People with Osteoarthritis. Nutrients 2020, 12, 574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biver, E.; Berenbaum, F.; Valdes, A.M.; Araujo de Carvalho, I.; Bindels, L.B.; Brandi, M.L.; Calder, P.C.; Castronovo, V.; Cavalier, E.; Cherubini, A.; et al. Gut microbiota and osteoarthritis management: An expert consensus of the European society for clinical and economic aspects of osteoporosis, osteoarthritis and musculoskeletal diseases (ESCEO). Ageing Res. Rev. 2019, 55, 100946. [Google Scholar] [CrossRef] [PubMed]
- de Sire, R.; Rizzatti, G.; Ingravalle, F.; Pizzoferrato, M.; Petito, V.; Lopetuso, L.; Graziani, C.; de Sire, A.; Mentella, M.C.; Mele, M.C.; et al. Skeletal muscle-gut axis: Emerging mechanisms of sarcopenia for intestinal and extra intestinal diseases. Minerva Gastroenterol. Dietol. 2018, 64, 351–362. [Google Scholar] [CrossRef]
- Pizzoferrato, M.; de Sire, R.; Ingravalle, F.; Mentella, M.C.; Petito, V.; Martone, A.M.; Landi, F.; Miggiano, G.; Mele, M.C.; Lopetuso, L.R.; et al. Characterization of Sarcopenia in an IBD Population Attending an Italian Gastroenterology Tertiary Center. Nutrients 2019, 11, 2281. [Google Scholar] [CrossRef] [Green Version]
- Nardone, O.M.; de Sire, R.; Petito, V.; Testa, A.; Villani, G.; Scaldaferri, F.; Castiglione, F. Inflammatory Bowel Diseases and Sarcopenia: The Role of Inflammation and Gut Microbiota in the Development of Muscle Failure. Front. Immunol. 2021, 12, 694217. [Google Scholar] [CrossRef] [PubMed]
- Ianiro, G.; Bibbò, S.; Gasbarrini, A.; Cammarota, G. Therapeutic modulation of gut microbiota: Current clinical applications and future perspectives. Curr. Drug Targe. 2014, 15, 762–770. [Google Scholar] [CrossRef] [PubMed]
- Cammarota, G.; Ianiro, G.; Bibbò, S.; Gasbarrini, A. Gut microbiota modulation: Probiotics, antibiotics or fecal microbiota transplantation? Intern. Emerg. Med. 2014, 9, 365–373. [Google Scholar] [CrossRef]
- Mullish, B.H.; Quraishi, M.N.; Segal, J.P.; Ianiro, G.; Iqbal, T.H. The gut microbiome: What every gastroenterologist needs to know. Frontline Gastroenterol. 2020, 12, 118–127. [Google Scholar] [CrossRef] [Green Version]
- Milani, C.; Ferrario, C.; Turroni, F.; Duranti, S.; Mangifesta, M.; van Sinderen, D.; Ventura, M. The human gut microbiota and its interactive connections to diet. J. Hum. Nutr. Diet. 2016, 29, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 1, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, E.A.; Velazquez, K.T.; Herbert, K.M. Influence of high-fat diet on gut microbiota: A driving force for chronic disease risk. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 515–520. [Google Scholar] [CrossRef]
- Graziani, C.; Talocco, C.; de Sire, R.; Petito, V.; Lopetuso, L.R.; Gervasoni, J.; Persichilli, S.; Franceschi, F.; Ojetti, V.; Gasbarrini, A.; et al. Intestinal permeability in physiological and pathological conditions: Major determinants and assessment modalities. Eur. Rev. Med. Pharm. Sci. 2019, 23, 795–810. [Google Scholar] [CrossRef]
- Li, L.; Rao, S.; Cheng, Y.; Zhuo, X.; Deng, C.; Xu, N.; Zhang, H.; Yang, L. Microbial osteoporosis: The interplay between the gut microbiota and bones via host metabolism and immunity. MicrobiologyOpen 2019, 8, e00810. [Google Scholar] [CrossRef] [Green Version]
- Ticinesi, A.; Lauretani, F.; Milani, C.; Nouvenne, A.; Tana, C.; Del Rio, D.; Maggio, M.; Ventura, M.; Meschi, T. Aging Gut Microbiota at the Cross-Road between Nutrition, Physical Frailty, and Sarcopenia: Is There a Gut-Muscle Axis? Nutrients 2017, 30, 1303. [Google Scholar] [CrossRef] [Green Version]
- Rizzoli, R.; Biver, E.; Brennan-Speranza, T.C. Nutritional intake and bone health. Lancet Diabetes Endocrinol. 2021, 9, 606–621. [Google Scholar] [CrossRef]
- Fong, W.; Li, Q.; Yu, J. Gut microbiota modulation: A novel strategy for prevention and treatment of colorectal cancer. Oncogene 2020, 39, 4925–4943. [Google Scholar] [CrossRef] [PubMed]
- Dewulf, E.M.; Cani, P.D.; Claus, S.P.; Fuentes, S.; Puylaert, P.G.; Neyrinck, A.M.; Bindels, L.B.; de Vos, W.M.; Gibson, G.R.; Thissen, J.P.; et al. Insight into the prebiotic concept: Lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut 2013, 62, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Azcarate-Peril, M.A.; Ritter, A.J.; Savaiano, D.; Monteagudo-Mera, A.; Anderson, C.; Magness, S.T.; Klaenhammer, T.R. Impact of short-chain galactooligosaccharides on the gut microbiome of lactose-intolerant individuals. Proc. Natl. Acad. Sci. USA 2017, 114, E367–E375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maier, T.V.; Lucio, M.; Lee, L.H.; VerBerkmoes, N.C.; Brislawn, C.J.; Bernhardt, J.; Lamendella, R.; McDermott, J.E.; Bergeron, N.; Heinzmann, S.S.; et al. Impact of Dietary Resistant Starch on the Human Gut Microbiome, Metaproteome, and Metabolome. mBio 2017, 8, e01343-17. [Google Scholar] [CrossRef] [Green Version]
- Shoaf, K.; Mulvey, G.L.; Armstrong, G.D.; Hutkins, R.W. Prebiotic galactooligosaccharides reduce adherence of enteropathogenic Escherichia coli to tissue culture cells. Infect. Immun. 2006, 74, 6920–6928. [Google Scholar] [CrossRef] [Green Version]
- Monteagudo-Mera, A.; Rastall, R.A.; Gibson, G.R.; Charalampopoulos, D.; Chatzifragkou, A. Adhesion mechanisms mediated by probiotics and prebiotics and their potential impact on human health. Appl. Microbiol. Biotechnol. 2019, 103, 6463–6472. [Google Scholar] [CrossRef] [Green Version]
- Orchard, T.S.; Larson, J.C.; Alghothani, N.; Bout-Tabaku, S.; Cauley, J.A.; Chen, Z.; LaCroix, A.Z.; Wactawski-Wende, J.; Jackson, R.D. Magnesium intake, bone mineral density, and fractures: Results from the Women’s Health Initiative Observational Study. Am. J. Clin. Nutr. 2014, 99, 926–933. [Google Scholar] [CrossRef] [Green Version]
- Alexy, U.; Remer, T.; Manz, F.; Neu, C.M.; Schoenau, E. Long-term protein intake and dietary potential renal acid load are associated with bone modeling and remodeling at the proximal radius in healthy children. Am. J. Clin. Nutr. 2005, 82, 1107–1114. [Google Scholar] [CrossRef] [Green Version]
- Durosier-Izart, C.; Biver, E.; Merminod, F.; van Rietbergen, B.; Chevalley, T.; Herrmann, F.R.; Ferrari, S.L.; Rizzoli, R. Peripheral skeleton bone strength is positively correlated with total and dairy protein intakes in healthy postmenopausal women. Am. J. Clin. Nutr. 2017, 105, 513–525. [Google Scholar] [CrossRef]
- Langsetmo, L.; Peters, K.W.; Burghardt, A.J.; Ensrud, K.E.; Fink, H.A.; Cawthon, P.M.; Cauley, J.A.; Schousboe, J.T.; Barrett-Connor, E.; Orwoll, E.S.; et al. Volumetric bone mineral density and failure load of distal limbs predict incident clinical fracture independent HR-pQCT BMD and failure load predicts incident clinical fracture of FRAX and clinical risk factors among older men. J. Bone Min. Res. 2018, 33, 1302–1311. [Google Scholar] [CrossRef]
- Wegh, C.A.M.; Geerlings, S.Y.; Knol, J.; Roeselers, G.; Belzer, C. Postbiotics and Their Potential Applications in Early Life Nutrition and Beyond. Int. J. Mol. Sci. 2019, 20, 4673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montazeri-Najafabady, N.; Ghasemi, Y.; Dabbaghmanesh, M.H.; Ashoori, Y.; Talezadeh, P.; Koohpeyma, F.; Abootalebi, S.N.; Gholami, A. Exploring the bone sparing effects of postbiotics in the post-menopausal rat model. BMC Complement. Med. Ther. 2021, 21, 155. [Google Scholar] [CrossRef] [PubMed]
- Jang, A.R.; Park, J.S.; Kim, D.K.; Park, J.Y.; Ahn, J.H.; Kim, D.Y.; Lee, T.S.; Chang, J.Y.; Choi, J.H.; Park, J.H. Cell-free culture supernatant of Lactobacillus curvatus Wikim38 inhibits RANKL-induced osteoclast differentiation and ameliorates bone loss in ovariectomized mice. Lett. Appl. Microbiol. 2021, 73, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Ianiro, G.; Tilg, H.; Gasbarrini, A. Antibiotics as deep modulators of gut microbiota: Between good and evil. Gut 2016, 65, 1906–1915. [Google Scholar] [CrossRef]
- Cho, I.; Yamanishi, S.; Cox, L.; Methé, B.A.; Zavadil, J.; Li, K.; Gao, Z.; Mahana, D.; Raju, K.; Teitler, I.; et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 2012, 488, 621–626. [Google Scholar] [CrossRef]
- Cox, L.M.; Yamanishi, S.; Sohn, J.; Alekseyenko, A.V.; Leung, J.M.; Cho, I.; Kim, S.G.; Li, H.; Gao, Z.; Mahana, D.; et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 2014, 158, 705–721. [Google Scholar] [CrossRef] [Green Version]
- Williams, S.; Wakisaka, A.; Zeng, Q.Q.; Barnes, J.; Martin, G.; Wechter, W.J.; Liang, C.T. Minocycline prevents the decrease in bone mineral density and trabecular bone in ovariectomized aged rats. Bone 1996, 19, 637–644. [Google Scholar] [CrossRef]
- Bibbò, S.; Ianiro, G.; Gasbarrini, A.; Cammarota, G. Fecal microbiota transplantation: Past, present and future perspectives. Minerva Gastroenterol. Dietol. 2017, 63, 420–430. [Google Scholar] [CrossRef]
- Waller, K.M.J.; Leong, R.W.; Paramsothy, S. An update on FMT for the treatment of gastrointestinal diseases. J. Gastroenterol. Hepatol. 2021. online ahead of print. [Google Scholar] [CrossRef]
- Ma, S.; Wang, N.; Zhang, P.; Wu, W.; Fu, L. Fecal microbiota transplantation mitigates bone loss by improving gut microbiome composition and gut barrier function in aged rats. PeerJ 2021, 9, e12293. [Google Scholar] [CrossRef] [PubMed]
- Biver, E.; Pepe, J.; de Sire, A.; Chevalley, T.; Ferrari, S. Associations between radius low-frequency axial ultrasound velocity and bone fragility in elderly men and women. Osteoporos. Int. 2019, 30, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Bone, H.G.; Wagman, R.B.; Brandi, M.L.; Brown, J.P.; Chapurlat, R.; Cummings, S.R.; Czerwiński, E.; Fahrleitner-Pammer, A.; Kendler, D.L.; Lippuner, K.; et al. 10 years of denosumab treatment in postmenopausal women with osteoporosis: Results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol. 2017, 5, 513–523. [Google Scholar] [CrossRef]
- Iolascon, G.; de Sire, A.; Curci, C.; Paoletta, M.; Liguori, S.; Calafiore, D.; Gimigliano, F.; Moretti, A. Osteoporosis guidelines from a rehabilitation perspective: Systematic analysis and quality appraisal using AGREE II. Eur. J. Phys. Rehabil. Med. 2021, 57, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Yahiro, Y.; Maeda, S.; Morikawa, M.; Koinuma, D.; Jokoji, G.; Ijuin, T.; Komiya, S.; Kageyama, R.; Miyazono, K.; Taniguchi, N. BMP-induced Atoh8 attenuates osteoclastogenesis by suppressing Runx2 transcriptional activity and reducing the Rankl/Opg expression ratio in osteoblasts. Bone Res. 2020, 8, 32. [Google Scholar] [CrossRef]
- Moretti, A.; de Sire, A.; Curci, C.; Toro, G.; Gimigliano, F.; Iolascon, G. Effectiveness of denosumab on back pain-related disability and quality-of-life in patients with vertebral fragility fractures. Curr. Med. Res. Opin. 2019, 35, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Migliaccio, S.; Francomano, D.; Romagnoli, E.; Marocco, C.; Fornari, R.; Resmini, G.; Buffa, A.; Di Pietro, G.; Corvaglia, S.; Gimigliano, F.; et al. Persistence with denosumab therapy in women affected by osteoporosis with fragility fractures: A multicenter observational real practice study in Italy. J. Endocrinol. Investig. 2017, 40, 1321–1326. [Google Scholar] [CrossRef]
- Lin, T.H.; Yang, R.S.; Tu, H.J.; Liou, H.C.; Lin, Y.M.; Chuang, W.J.; Fu, W.M. Inhibition of osteoporosis by the αvβ3 integrin antagonist of rhodostomin variants. Eur. J. Pharmacol. 2017, 804, 94–101. [Google Scholar] [CrossRef]
- Zur, Y.; Rosenfeld, L.; Keshelman, C.A.; Dalal, N.; Guterman-Ram, G.; Orenbuch, A.; Einav, Y.; Levaot, N.; Papo, N. A dual-specific macrophage colony-stimulating factor antagonist of c-FMS and αvβ3 integrin for osteoporosis therapy. PLoS Biol. 2018, 16, e2002979. [Google Scholar] [CrossRef]
- Fan, G.; Zhao, Q.; Lu, P.; Chen, H.; Tan, W.; Guo, W.; Liu, C.; Liu, J. Comparison between teriparatide and bisphosphonates for improving bone mineral density in postmenopausal osteoporosis patients: A meta-analysis. Medicine 2020, 99, e18964. [Google Scholar] [CrossRef]
- Lau, E.; Dinavahi, R.; Woo, Y.C.; Wu, C.H.; Guan, J.; Maddox, J.; Tolman, C.; Yang, W.; Shin, C.S. Romosozumab or alendronate for fracture prevention in East Asian patients: A subanalysis of the phase III, randomized ARCH study. Osteoporos. Int. 2020, 31, 677–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, H.; Shi, S.; Xiao, F.; Huang, Z.; Xu, J.; Chen, G.; Zhou, K.; Lu, L.; Yin, X. MiR-1-3p regulates the differentiation of mesenchymal stem cells to prevent osteoporosis by targeting secreted frizzled-related protein 1. Bone 2020, 137, 115444. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Baldridge, M.T.; King, K.Y. Hematopoiesis and the bacterial microbiome. Blood 2018, 132, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Zhang, F.; Yin, X.; Cao, H.; Wang, X.; Liu, D.; Chen, J.; Chen, X. Bone Homeostasis and Gut Microbial-Dependent Signaling Pathways. J. Microbiol. Biotechnol. 2021, 31, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Liu, W.; Zhang, X.; Zhao, M.; Zhu, B.; Hou, T.; He, H. Duck Egg White-Derived Peptide VSEE (Val-Ser-Glu-Glu) Regulates Bone and Lipid Metabolisms by Wnt/β-Catenin Signaling Pathway and Intestinal Microbiota. Mol. Nutr. Food Res. 2019, 63, e1900525. [Google Scholar] [CrossRef] [PubMed]
- Goodrich, J.K.; Di Rienzi, S.C.; Poole, A.C.; Koren, O.; Walters, W.A.; Caporaso, J.G.; Knight, R.; Ley, R.E. Conducting a microbiome study. Cell 2014, 158, 250–262. [Google Scholar] [CrossRef] [Green Version]
| Authors | Journal | Year | Dietary Supplement | Subjects | Pathways Investigated | Gut Microbiota Modulation | Main Findings |
|---|---|---|---|---|---|---|---|
| Porwal et al. [55] | Biomedicine & Pharmacotherapy | 2020 | Fructooligosaccharides | Animals | SCFA production | Not investigated | Fructooligosaccharides prevent ovariectomy-induced bone loss |
| Yan et al. [52] | Carbohydrate Polymers | 2019 | Fructooligosaccharides | Animals | Osteoblast stimulation | Not investigated | Fructooligosaccharides increase bone mineral density and strength |
| Tanabe et al. [54] | Journal of Agriculture and Food Chemistry | 2019 | Fructooligosaccharides | Animals | ↓ Serum CRP; SCFA production | ↑ Lactobacillus ↑ Bacteroides ↓ Clostridium | Fructooligosaccharides reduce bone resorption and systemic inflammation |
| Slevin et al. [56] | The Journal of Nutrition | 2014 | Fructooligosaccharides | Humans | Reduction in the number of bone turnover markers | Not investigated | Fructooligosaccharides reduce postmenopausal bone loss |
| Mathey et al. [53] | Calcified Tissue International | 2004 | Fructooligosaccharides | Animals | SCFA production | Not investigated | Fructooligosaccharides increase bone mineral density and strength |
| Whisner et al. [50] | British Journal of Nutrition | 2013 | Galactooligosaccharides | Humans | SCFA production | ↑ Bifidobacteria | Galactooligosaccharides increase calcium absorption |
| Weaver et al. [48] | Journal of Agricultural and Food Chemistry | 2011 | Galactooligosaccharides | Animals | SCFA production | ↑ Bifidobacteria | Galactooligosaccharides increase calcium absorption |
| Van den Heuvel et al. [49] | The Journal of Nutrition | 2000 | Galactooligosaccharides | Humans | SCFA production | Not investigated | Galactooligosaccharides increase calcium absorption |
| Karakan et al. [65] | Frontiers in Nutrition | 2021 | Lactulose | Humans | SCFA production | ↑ Bifidobacteria ↑ Lactobacillus ↓ Clostridium | Lactulose increases calcium absorption |
| Chen et al. [64] | Aging and Disease | 2020 | Lactulose | Animals | ↓ TNFα ↓ RANKL ↓ IL-17 ↑ IL-10 | ↓ Firmicutes ↑ Bacteroides | Lactulose inhibits osteoclastogenesis and bone resorption |
| Seki et al. [66] | Journal of Nutritional Science and Vitaminology | 2007 | Lactulose | Humans | Not investigated | Not investigated | Lactulose increases calcium and magnesium absorption |
| Tousen et al. [60] | Nutrients | 2019 | Resistant starch | Animals | ↓ IL-7R mRNA↑ IL-10 mRNA↓ RANKL | ↑ Bifidobacteria | Resistant starch prevents post-ovariectomy bone loss |
| Tousen et al. [59] | British Journal of Nutrition | 2016 | Resistant starch | Animals | ↓ IL-7R mRNA | ↑ Bifidobacteria | Resistant starch prevents post ovariectomy bone loss |
| Tousen et al. [58] | Metabolism | 2011 | Resistant starch | Animals | Increased isoflavone availability and estrogenic activities | ↑ Bifidobacteria | Resistant starch prevents post-ovariectomy bone loss |
| Gao et al. [62] | Nutrients | 2020 | Xylooligosaccharides | Animals | ↑ Na+/Ca2+ exchanger 1 ↑ TRPV6 | Not investigated | Xylooligosaccharides increase calcium absorption |
| Eaimworawuthikul et al. [63] | European Journal of Nutrition | 2020 | Xylooligosaccharides | Animals | Osteoclast inhibition | Not investigated | Xylooligosaccharides reduce bone resorption in systemic inflammation |
| Authors | Journal | Year | Dietary Supplement | Subjects | Pathways Investigated | Gut Microbiota Modulation | Main Findings |
|---|---|---|---|---|---|---|---|
| Ling et al. [86] | The Journal of Clinical Endocrinology and Metabolism | 2021 | Amino acids | Humans | Not determined |
↓ Actinobacillus, ↓ Blautia, ↓ Oscillospira | Valine, leucine, and isoleucine serum levels are inversely related to the occurrence of osteoporosis |
| Jennings et al. [85] | Journal of Bone and Mineral Research | 2016 | Amino acids | Humans | ↑ IGF1 | Not investigated | Alanine, arginine, glutamic acid, leucine, lysine, and proline increase BMD |
| Dawson-Hugler et al. [75] | Osteoporosis International | 2007 | Amino acids | Humans | ↑ IGF1 | Not investigated | Phenylalanine and histidine increase calcium absorption |
| Liu et al. [74] | Food & Function | 2018 | Casein Phosphopeptides | Animals | ↑ TRPV6 | Not investigated | Casein phosphopeptides increase calcium absorption and prevent bone resorption |
| Ong et al. [78] | Advances in Nutrition | 2020 | Fermented dairy products | Humans | ↓ TNF-α ↓ IL-6 ↓ RANKL | Not investigated | Fermented dairy products might reduce hip fracture risk |
| Biver et al. [76] | Osteoporosis International | 2018 | Fermented dairyproducts | Humans | Action on calcium balance and decrease in secondary hyper parathyroidism. | Not investigated | Fermented dairy products attenuate postmenopausal bone loss |
| Laird et al. [77] | Osteoporosis International | 2017 | Fermented dairy products | Humans | Modulation of osteoclast numbers and activity | Not investigated | Fermented dairy products increase bone mineral density |
| Tu et al. [81] | Nutrients | 2020 | Kefir peptides | Animals | ↓ TNF-α ↓ RANKL |
↑ Alloprevotella, ↑ Anaerostipes, ↑ Parasutterella, ↑ Romboutsia, ↑ Ruminococcus, ↑ Streptococcus | Kefir peptides prevent ovariectomy-induced bone loss |
| Chen et al. [80] | Osteoporosis International | 2014 | Kefir peptides | Animals | ↑TRPV6 | Not investigated | Kefir peptides prevent ovariectomy-induced bone loss |
| Authors | Journal | Year | Dietary Supplement | Subjects | Pathways Investigated | Gut Microbiota Modulation | Main Findings |
|---|---|---|---|---|---|---|---|
| Qasem et al. [100] | BMC Pediatrics | 2017 | Iron | Humans | Lowering inflammation | ↑ Bifidobacteria ↑ Bacteroides | Favorable effects of iron on bone might be mediated by the gut microbiome |
| Wang et al. [89] | BMC Musculoskeletal Disorders | 2019 | Selenium | Humans | Not determined | ↓ Parabacteroides ↓ Firmicutes | Selenium deficiency is correlated with a higher prevalence of osteoporosis |
| Reed et al. [94] | Nutrients | 2015 | Zinc | Animals | SCFA production Lowering of inflammation | ↑ Lactobacillus | Favorable effects of zinc on bone might be mediated by the gut microbiome |
| Authors | Journal | Year | Dietary Supplement | Subjects | Pathways Investigated | Gut Microbiota Modulation | Main Findings |
|---|---|---|---|---|---|---|---|
| Narva et al. [113] | Annals of Nutrition and Metabolism | 2007 | Lactobacillus helveticus | Animals | Increasing bone formation | Not investigated | Lactobacillus helveticus prevents ovariectomy-induced bone loss |
| Ohlsson et al. [114] | PLoS ONE | 2014 | Lactobacillus paracasei | Animals | ↓ TNF-α ↓ IL-1β ↑ OPG | Not investigated | Lactobacillus paracasei prevents ovariectomy-induced bone loss |
| Schepper et al. [46] | The Journal of Bone and Mineral Research | 2020 | Lactobacillus reuteri | Animals | ↑ Wnt10b | ↓ Clostridium | Lactobacillus reuteri prevents glucocorticoid-induced bone loss |
| Nilsson et al. [120] | Journal of Internal Medicine | 2018 | Lactobacillus reuteri | Humans | Not determined | Not investigated | Lactobacillus reuteri prevents bone loss |
| Zhang et al. [115] | Endocrinology | 2015 | Lactobacillus reuteri | Animals | ↓ TNF-α | Not investigated | Lactobacillus reuteri prevents type-1-diabetes-induced bone loss |
| Britton et al. [111] | Journal of Cellular Physiology | 2014 | Lactobacillus reuteri | Animals | ↓ Trap5 ↓ RANKL ↑ CD4+ T-lymphocytes | Promoting gut microbiota diversity | Lactobacillus reuteri prevents Ovariectomy-induced bone loss |
| Jafarnejad et al. [118] | Journal of the American Nutrition Association | 2017 | Multispecies probiotic | Humans | ↓ PTH ↓ TNF-α | Not investigated | Multispecies probiotic reduces bone turnover |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Sire, A.; de Sire, R.; Curci, C.; Castiglione, F.; Wahli, W. Role of Dietary Supplements and Probiotics in Modulating Microbiota and Bone Health: The Gut-Bone Axis. Cells 2022, 11, 743. https://doi.org/10.3390/cells11040743
de Sire A, de Sire R, Curci C, Castiglione F, Wahli W. Role of Dietary Supplements and Probiotics in Modulating Microbiota and Bone Health: The Gut-Bone Axis. Cells. 2022; 11(4):743. https://doi.org/10.3390/cells11040743
Chicago/Turabian Stylede Sire, Alessandro, Roberto de Sire, Claudio Curci, Fabiana Castiglione, and Walter Wahli. 2022. "Role of Dietary Supplements and Probiotics in Modulating Microbiota and Bone Health: The Gut-Bone Axis" Cells 11, no. 4: 743. https://doi.org/10.3390/cells11040743
APA Stylede Sire, A., de Sire, R., Curci, C., Castiglione, F., & Wahli, W. (2022). Role of Dietary Supplements and Probiotics in Modulating Microbiota and Bone Health: The Gut-Bone Axis. Cells, 11(4), 743. https://doi.org/10.3390/cells11040743








