Hematopoietic Cell Transplantation for Systemic Sclerosis—A Review
Abstract
1. Introduction
2. Proposed Mechanisms of Action of Autohct on SSc
2.1. Partial Lymphoablation Followed by the Crowding out of the Persisting Autoreactive T Cells by De Novo Generated T Cells
2.2. Partial Lymphoablation Followed by the Functional Tolerization of Autoreactive T Cells
2.3. Fast Reconstitution of Tregs
2.4. Less Proinflammatory/More Anti-Inflammatory Myeloid Cells?
2.5. Role of B Cell?
2.6. Non-Immunologic Mechanisms?
3. Clinical Results
4. Patient Selection
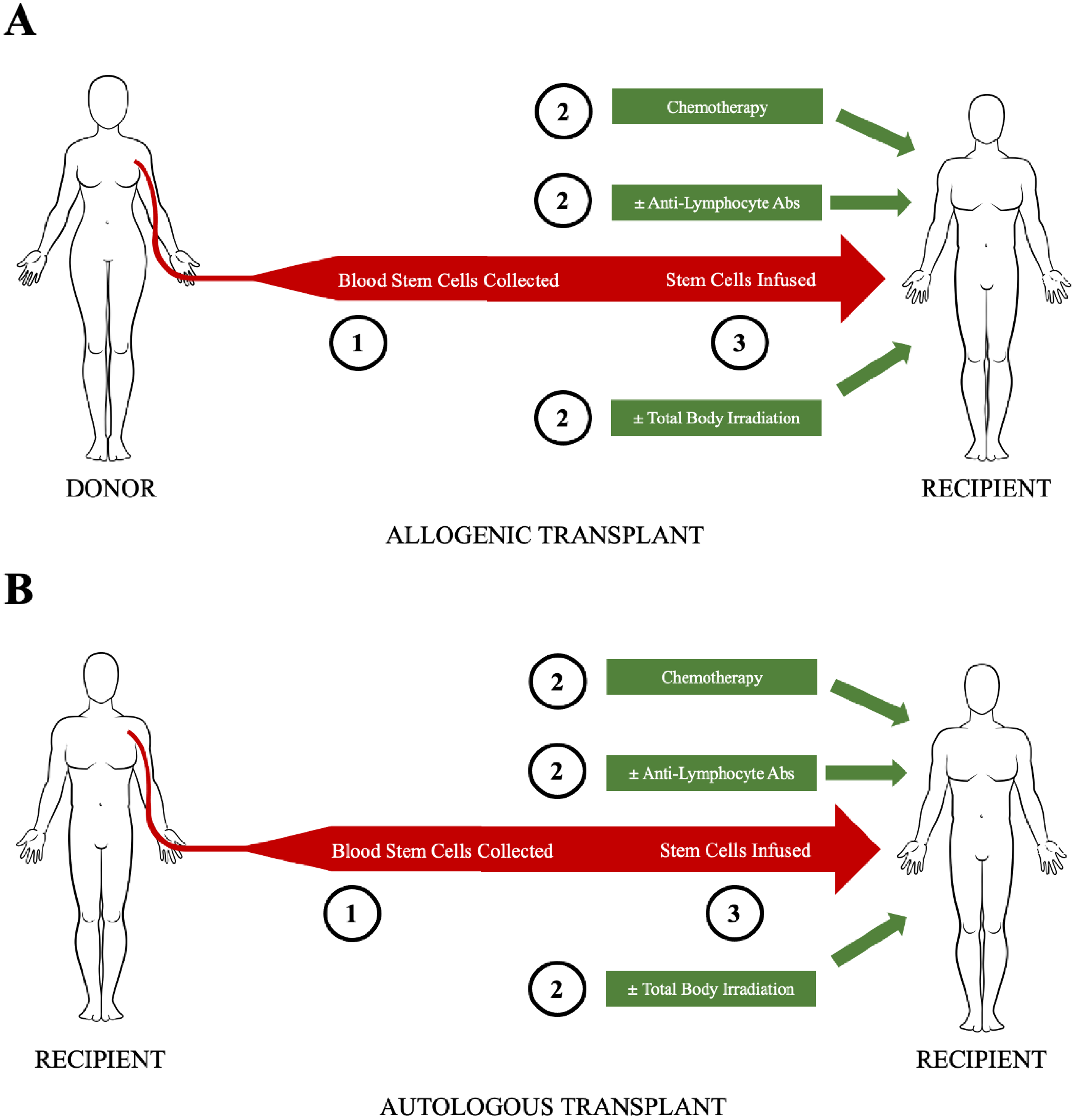
5. Transplant Protocols
5.1. Mobilization
5.2. Graft Manipulation/CD34 Cell Selection
5.3. Conditioning
5.4. Supportive Care
6. Complications of HCT
6.1. Acute Complications
6.2. Long-Term Complications
6.2.1. Infertility/Premature Menopause
6.2.2. Malignancy
6.2.3. New Autoimmune Disease
7. Relapse/SSc Progression
7.1. Definition
7.2. Frequency of Relapse
7.3. Treatment of Relapse
8. Allogeneic HCT
9. Future Works
9.1. HCT as First vs. Next Line Treatment?
9.2. Refining Patient Selection
9.3. Comparison of VARIABLES in HCT Techniques and Development of New HCT Techniques
9.4. Role of Maintenance Therapy
9.5. Cellular Therapies Other Than AutoHCT
10. Summary
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ingegnoli, F.; Ughi, N.; Mihai, C. Update on the Epidemiology, Risk Factors, and Disease Outcomes of Systemic Sclerosis. Best Pract Res. Clin. Rheumatol. 2018, 32, 223–240. [Google Scholar] [CrossRef]
- Al-Dhaher, F.F.; Pope, J.E.; Ouimet, J.M. Determinants of Morbidity and Mortality of Systemic Sclerosis in Canada. Semin. Arthritis Rheum. 2010, 39, 269–277. [Google Scholar] [CrossRef]
- Hudson, M.; Thombs, B.; Baron, M. Time to Diagnosis in Systemic Sclerosis: Is Sex a Factor? Arthritis Care Res. (Hoboken) 2009, 61, 274–278. [Google Scholar] [CrossRef]
- Becker, M.; Graf, N.; Sauter, R.; Allanore, Y.; Curram, J.; Denton, C.P.; Khanna, D.; Matucci-Cerinic, M.; de Oliveira Pena, J.; Pope, J.E.; et al. Predictors of Disease Worsening Defined by Progression of Organ Damage in Diffuse Systemic Sclerosis: A European Scleroderma Trials and Research (EUSTAR) Analysis. Ann. Rheum. Dis. 2019, 78, 1242–1248. [Google Scholar] [CrossRef]
- Wu, W.; Jordan, S.; Graf, N.; de Oliveira Pena, J.; Curram, J.; Allanore, Y.; Matucci-Cerinic, M.; Pope, J.E.; Denton, C.P.; Khanna, D.; et al. Progressive Skin Fibrosis Is Associated with a Decline in Lung Function and Worse Survival in Patients with Diffuse Cutaneous Systemic Sclerosis in the European Scleroderma Trials and Research (EUSTAR) Cohort. Ann. Rheum. Dis. 2019, 78, 648–656. [Google Scholar] [CrossRef]
- Herrick, A.L.; Peytrignet, S.; Lunt, M.; Pan, X.; Hesselstrand, R.; Mouthon, L.; Silman, A.J.; DInsdale, G.; Brown, E.; Czirják, L.; et al. Patterns and Predictors of Skin Score Change in Early Diffuse Systemic Sclerosis from the European Scleroderma Observational Study. Ann. Rheum. Dis. 2018, 77, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Ledoult, E.; Launay, D.; Béhal, H.; Mouthon, L.; Pugnet, G.; Lega, J.C.; Agard, C.; Allanore, Y.; Jego, P.; Fauchais, A.L.; et al. Early Trajectories of Skin Thickening Are Associated with Severity and Mortality in Systemic Sclerosis. Arthritis Res. Ther. 2020, 22. [Google Scholar] [CrossRef]
- Bernatsky, S.; Joseph, L.; Pineau, C.A.; Belisle, P.; Hudson, M.; Clarke, A.E. Scleroderma Prevalence: Demographic Variations in a Population-Based Sample. Arthritis Rheum. 2009, 61, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Bergamasco, A.; Hartmann, N.; Wallace, L.; Verpillat, P. Epidemiology of Systemic Sclerosis and Systemic Sclerosis-Associated Interstitial Lung Disease. Clin. Epidemiol. 2019, 11, 257–273. [Google Scholar] [CrossRef] [PubMed]
- Kowal-Bielecka, O.; Fransen, J.; Avouac, J.; Becker, M.; Kulak, A.; Allanore, Y.; Distler, O.; Clements, P.; Cutolo, M.; Czirjak, L.; et al. Update of EULAR Recommendations for the Treatment of Systemic Sclerosis. Ann. Rheum. Dis. 2017, 76, 1327–1339. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Rivas, M.; Royo, C.; Simeón, C.P.; Corbella, X.; Fonollosa, V. Mortality and Survival in Systemic Sclerosis: Systematic Review and Meta-Analysis. Semin. Arthritis Rheum. 2014, 44, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Poudel, D.R.; Derk, C.T. Mortality and Survival in Systemic Sclerosis: A Review of Recent Literature. Curr. Opin. Rheumatol 2018, 30, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Farge, D.; Ait Abdallah, N.; Marjanovic, Z.; del Papa, N. Autologous Stem Cell Transplantation in Scleroderma. Presse. Med. 2021, 50. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Hudson, M.; Baron, M.; Carreira, P.; Stevens, W.; Rabusa, C.; Tatibouet, S.; Carmona, L.; Joven, B.E.; Huq, M.; et al. Early Mortality in a Multinational Systemic Sclerosis Inception Cohort. Arthritis Rheumatol. 2017, 69, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Altman, R.D.; Medsger, T.A.; Bloch, D.A.; Michel, B.A. Predictors of Survival in Systemic Sclerosis (Scleroderma). Arthritis Rheum. 1991, 34, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Copelan, E.A. Hematopoietic Stem-Cell Transplantation. New Engl. J. Med. 2006, 354, 1813–1826. [Google Scholar] [CrossRef]
- Binks, M.; Passweg, J.R.; Furst, D.; McSweeney, P.; Sullivan, K.; Besenthal, C.; Finke, J.; Peter, H.H.; van Laar, J.; Breedveld, F.C.; et al. Phase I/II Trial of Autologous Stem Cell Transplantation in Systemic Sclerosis: Procedure Related Mortality and Impact on Skin Disease. Ann. Rheum. Dis. 2001, 60, 577–584. [Google Scholar] [CrossRef]
- Tyndall, A.; Gratwohl, A. Haemopoietic Stem and Progenitor Cells in the Treatment of Severe Autoimmune Diseases. Ann. Rheum. Dis. 1996, 55, 149–151. [Google Scholar] [CrossRef]
- Nash, R.A.; McSweeney, P.A.; Nelson, J.L.; Wener, M.; Georges, G.E.; Langston, A.A.; Shulman, H.; Sullivan, K.M.; Lee, J.; Henstorf, G.; et al. Allogeneic Marrow Transplantation in Patients with Severe Systemic Sclerosis: Resolution of Dermal Fibrosis. Arthritis Rheum. 2006, 54, 1982–1986. [Google Scholar] [CrossRef]
- Daikeler, T.; Hügle, T.; Farge, D.; Andolina, M.; Gualandi, F.; Baldomero, H.; Bocelli-Tyndall, C.; Brune, M.; Dalle, J.H.; Ehninger, G.; et al. Allogeneic Hematopoietic SCT for Patients with Autoimmune Diseases. Bone Marrow Transplant. 2009, 44, 27–33. [Google Scholar] [CrossRef]
- Burt, R.K.; Shah, S.J.; Dill, K.; Grant, T.; Gheorghiade, M.; Schroeder, J.; Craig, R.; Hirano, I.; Marshall, K.; Ruderman, E.; et al. Autologous Non-Myeloablative Haemopoietic Stem-Cell Transplantation Compared with Pulse Cyclophosphamide Once per Month for Systemic Sclerosis (ASSIST): An Open-Label, Randomised Phase 2 Trial. Lancet 2011, 378, 498–506. [Google Scholar] [CrossRef] [PubMed]
- van Laar, J.M.; Farge, D.; Sont, J.K.; Naraghi, K.; Marjanovic, Z.; Larghero, J.; Schuerwegh, A.J.; Marijt, E.W.A.; Vonk, M.C.; Schattenberg, A.V.; et al. Autologous Hematopoietic Stem Cell Transplantation vs Intravenous Pulse Cyclophosphamide in Diffuse Cutaneous Systemic Sclerosis: A Randomized Clinical Trial. JAMA 2014, 311, 2490–2498. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.M.; Goldmuntz, E.A.; Keyes-Elstein, L.; McSweeney, P.A.; Pinckney, A.; Welch, B.; Mayes, M.D.; Nash, R.A.; Crofford, L.J.; Eggleston, B.; et al. Myeloablative Autologous Stem-Cell Transplantation for Severe Scleroderma. New Engl. J. Med. 2018, 378, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Eyraud, A.; Scouppe, L.; Barnetche, T.; Forcade, E.; Lazaro, E.; Duffau, P.; Richez, C.; Seneschal, J.; Truchetet, M.E. Efficacy and Safety of Autologous Haematopoietic Stem Cell Transplantation in Systemic Sclerosis: A Systematic Review of the Literature. Br. J. Dermatol. 2018, 178, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Shouval, R.; Furie, N.; Raanani, P.; Nagler, A.; Gafter-Gvili, A. Autologous Hematopoietic Stem Cell Transplantation for Systemic Sclerosis: A Systematic Review and Meta-Analysis. Biol. Blood Marrow Transplant. 2018, 24, 937–944. [Google Scholar] [CrossRef]
- Host, L.; Nikpour, M.; Calderone, A.; Cannell, P.; Roddy, J. Autologous Stem Cell Transplantation in Systemic Sclerosis: A Systematic Review. Clin. Exp. Rheumatol. 2017, 35, S198–S207. [Google Scholar]
- Panopoulos, S.T.; Tektonidou, M.G.; Bournia, V.K.; Laskari, K.; Sfikakis, P.P. Outcomes of Conventionally-Treated Systemic Sclerosis Patients Eligible for Autologous Haematopoietic Stem Cell Transplantation. Clin. Exp. Rheumatol. 2021, 39 (Suppl. S131), S29–S33. [Google Scholar] [CrossRef]
- Burt, R.K.; Han, X.; Quigley, K.; Arnautovic, I.; Shah, S.J.; Lee, D.C.; Freed, B.H.; Jovanovic, B.; Helenowski, I.B. Cardiac Safe Hematopoietic Stem Cell Transplantation for Systemic Sclerosis with Poor Cardiac Function: A Pilot Safety Study That Decreases Neutropenic Interval to 5 Days. Bone Marrow Transplant. 2020 56:1 2020, 56, 50–59. [Google Scholar] [CrossRef]
- Alexander, T.; Greco, R.; Snowden, J.A. Hematopoietic Stem Cell Transplantation for Autoimmune Disease. Annu. Rev. Med. 2021, 72, 215–228. [Google Scholar] [CrossRef]
- Georges, G.; Sullivan, K.; Khanna, D.; Mei, M.; Mayes, M.; Furst, D.; Wener, M.; Kafaja, S.; Long, G.; Sanchorawala, V.; et al. Autologous Hematopoietic Stem Cell (HSC) Transplantation for Systemic Sclerosis, North American Registry: Updated Outcomes and the Impact of CD34+ HSC Enrichment. In Proceedings of the ACR Convergence 2021, online, 8 November 2021. [Google Scholar]
- Storek, J.; Zhao, Z.; Lin, E.; Berger, T.; McSweeney, P.A.; Nash, R.A.; Akatsuka, Y.; Metcalf, M.D.; Lu, H.; Kalina, T.; et al. Recovery from and Consequences of Severe Iatrogenic Lymphopenia (Induced to Treat Autoimmune Diseases). Clin. Immun. 2004, 113, 285–298. [Google Scholar] [CrossRef]
- Bosch, M.; Khan, F.M.; Storek, J. Immune Reconstitution after Hematopoietic Cell Transplantation. Curr. Opin. Hematol. 2012, 19, 324–355. [Google Scholar] [CrossRef] [PubMed]
- Malmegrim, K.C.R.; Lima-Júnior, J.R.; Arruda, L.C.M.; de Azevedo, J.T.C.; de Oliveira, G.L.V.; Oliveira, M.C. Autologous Hematopoietic Stem Cell Transplantation for Autoimmune Diseases: From Mechanistic Insights to Biomarkers. Front. Immunol. 2018, 9, 2602. [Google Scholar] [CrossRef]
- Henes, J.; Glaeser, L.; Kötter, I.; Vogel, W.; Kanz, L.; Klein, R. Analysis of Anti–Topoisomerase I Antibodies in Patients with Systemic Sclerosis before and after Autologous Stem Cell Transplantation. Rheumatology 2017, 56, 451–456. [Google Scholar] [CrossRef][Green Version]
- Glaeser, L.; Henes, J.; Kötter, I.; Vogel, W.; Kanz, L.; Klein, R. Molecular Recognition Patterns of Anti-Topoisomerase I-Antibodies in Patients with Systemic Sclerosis before and after Autologous Stem Cell Transplantation. Clinexprheumatol. Org. 2018, 36, 28–35. [Google Scholar]
- Tsukamoto, H.; Nagafuji, K.; Horiuchi, T.; Mitoma, H.; Niiro, H.; Arinobu, Y.; Inoue, Y.; To, K.; Miyamoto, T.; Iwasaki, H.; et al. Analysis of Immune Reconstitution after Autologous CD34+ Stem/Progenitor Cell Transplantation for Systemic Sclerosis: Predominant Reconstitution of Th1 CD4+ T Cells. Rheumatology 2011, 50, 944–952. [Google Scholar] [CrossRef] [PubMed]
- Roumm, A. Lymphocytes in the Skin of Patients with Progressive Systemic Sclerosis. Arthritis Rheumat. 1984, 27, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, M.L.; Finlay, D.R.; Murray, J.I.; Troyanskaya, O.G.; Chi, J.T.; Pergamenschikov, A.; McCalmont, T.H.; Brown, P.O.; Botstein, D.; Connolly, M.K. Systemic and Cell Type-Specific Gene Expression Patterns in Scleroderma Skin. Proc. Natl. Acad. Sci. USA 2003, 100, 12319–12324. [Google Scholar] [CrossRef]
- Maul, G.G.; Jimenez, S.A. Mononuclear Cellular Infiltrates in Clinically Involved Skin from Patients with Systemic Sclerosis of Recent Onset Predominantly Consist of Monocytes/Macrophages. Pathobiology 1995, 63, 48–56. [Google Scholar] [CrossRef]
- Sakkas, L.I.; Xu, B.; Artlett, C.M.; Lu, S.; Jimenez, S.A.; Platsoucas, C.D. Oligoclonal T Cell Expansion in the Skin of Patients with Systemic Sclerosis. J. Immunol. 2002, 168, 3649–3659. [Google Scholar] [CrossRef]
- Storek, J.; Zhao, Z.; Liu, Y.; Nash, R.; McSweeney, P.; Maloney, D.G. Early Recovery of CD4 T Cell Receptor Diversity after “Lymphoablative” Conditioning and Autologous CD34 Cell Transplantation. Biol. Blood Marrow Transplant. 2008, 14, 1373–1379. [Google Scholar] [CrossRef][Green Version]
- Dubinsky, A.N.; Burt, R.K.; Martin, R.; Muraro, P.A. T-Cell Clones Persisting in the Circulation after Autologous Hematopoietic SCT Are Undetectable in the Peripheral CD34+ Selected Graft. Bone Marrow Transplant. 2010 45:2 2009, 45, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Muraro, P.A.; Douek, D.C.; Packer, A.; Chung, K.; Guenaga, F.J.; Cassiani-Ingoni, R.; Campbell, C.; Memon, S.; Nagle, J.W.; Hakim, F.T.; et al. Thymic Output Generates a New and Diverse TCR Repertoire after Autologous Stem Cell Transplantation in Multiple Sclerosis Patients. J. Exp. Med. 2005, 201, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Henes, J.; Oliveira, M.C.; Labopin, M.; Badoglio, M.; Scherer, H.U.; del Papa, N.; Daikeler, T.; Schmalzing, M.; Schroers, R.; Martin, T.; et al. Autologous Stem Cell Transplantation for Progressive Systemic Sclerosis: A Prospective Non-Interventional Study from the European Society for Blood and Marrow Transplantation Autoimmune Disease Working Party. Haematologica 2021, 106, 375. [Google Scholar] [CrossRef] [PubMed]
- van Bijnen, S.; de Vries-Bouwstra, J.; van den Ende, C.H.; Boonstra, M.; Kroft, L.; Geurts, B.; Snoeren, M.; Schouffoer, A.; Spierings, J.; van Laar, J.M.; et al. Predictive Factors for Treatment-Related Mortality and Major Adverse Events after Autologous Haematopoietic Stem Cell Transplantation for Systemic Sclerosis: Results of a Long-Term Follow-up Multicentre Study. Ann. Rheum. Dis. 2020, 79, 1084–1089. [Google Scholar] [CrossRef]
- Mancardi, G.L.; Sormani, M.P.; di Gioia, M.; Vuolo, L.; Gualandi, F.; Amato, M.P.; Capello, E.; Currò, D.; Uccelli, A.; Bertolotto, A.; et al. Autologous Haematopoietic Stem Cell Transplantation with an Intermediate Intensity Conditioning Regimen in Multiple Sclerosis: The Italian Multi-Centre Experience. Mult. Scler. 2012, 18, 835–842. [Google Scholar] [CrossRef]
- Krasulová, E.; Trněný, M.; Kozák, T.; Vacková, B.; Pohlreich, D.; Kemlink, D.; Kobylka, P.; Kovářová, I.; Lhotáková, P.; Havrdová, E. High-Dose Immunoablation with Autologous Haematopoietic Stem Cell Transplantation in Aggressive Multiple Sclerosis: A Single Centre 10-Year Experience. Mult. Scler. 2010, 16, 685–693. [Google Scholar] [CrossRef]
- Arruda, L.C.M.; Malmegrim, K.C.R.; Lima-Junior, J.R.; Clave, E.; Dias, J.B.E.; Moraes, D.A.; Douay, C.; Fournier, I.; Moins-Teisserenc, H.; Alberdi, A.J.; et al. Immune Rebound Associates with a Favorable Clinical Response to autologous HSCT in Systemic Sclerosis Patients. Blood Adv. 2018, 2, 126. [Google Scholar] [CrossRef]
- Arruda, L.C.M.; Clave, E.; Douay, C.; Lima-Júnior, J.R.; Slavov, S.N.; Malmegrim, K.C.R.; Alberdi, A.J.; Oliveira, M.C.; Toubert, A. CMV-Specific Clones May Lead to Reduced TCR Diversity and Relapse in Systemic Sclerosis Patients Treated with AHSCT. Rheumatology 2020, 59, e38–e40. [Google Scholar] [CrossRef]
- Muraro, P.A.; Robins, H.; Malhotra, S.; Howell, M.; Phippard, D.; Desmarais, C.; de Paula Alves Sousa, A.; Griffith, L.M.; Lim, N.; Nash, R.A.; et al. T Cell Repertoire Following Autologous Stem Cell Transplantation for Multiple Sclerosis. J. Clin. Investig. 2014, 124, 1168–1172. [Google Scholar] [CrossRef]
- Farge, D.; Arruda, L.C.M.; Brigant, F.; Clave, E.; Douay, C.; Marjanovic, Z.; Deligny, C.; Maki, G.; Gluckman, E.; Toubert, A.; et al. Long-Term Immune Reconstitution and T Cell Repertoire Analysis after Autologous Hematopoietic Stem Cell Transplantation in Systemic Sclerosis Patients. J. Hematol. Oncol. 2017, 10, 21. [Google Scholar] [CrossRef]
- van der Velden, A.M.T.; Claessen, A.M.E.; van Velzen-Blad, H.; Biesma, D.H.; Rijkers, G.T. Development of T Cell-Mediated Immunity after Autologous Stem Cell Transplantation: Prolonged Impairment of Antigen-Stimulated Production of γ-Interferon. Bone Marrow Transplant. 2007 40:3 2007, 40, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Delemarre, E.M.; van den Broek, T.; Mijnheer, G.; Meerding, J.; Wehrens, E.J.; Olek, S.; Boes, M.; van Herwijnen, M.J.C.; Broere, F.; van Royen, A.V.; et al. Autologous Stem Cell Transplantation Aids Autoimmune Patients by Functional Renewal and TCR Diversification of Regulatory T Cells. Blood 2016, 127, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Assassi, S.; Wang, X.; Chen, G.; Goldmuntz, E.; Keyes-Elstein, L.; Ying, J.; Wallace, P.K.; Turner, J.; Zheng, W.J.; Pascual, V.; et al. Myeloablation Followed by Autologous Stem Cell Transplantation Normalises Systemic Sclerosis Molecular Signatures. Ann. Rheum. Dis. 2019, 78, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Hendrawan, K.; Visweswaran, M.; Ma, D.D.F.; Moore, J.J. Tolerance Regeneration by T Regulatory Cells in Autologous Haematopoietic Stem Cell Transplantation for Autoimmune Diseases. Bone Marrow Transplant. 2019, 55, 857–866. [Google Scholar] [CrossRef]
- Baraut, J.; Grigore, E.I.; Jean-Louis, F.; Khelifa, S.H.; Durand, C.; Verrecchia, F.; Farge, D.; Michel, L. Peripheral Blood Regulatory T Cells in Patients with Diffuse Systemic Sclerosis (SSc) before and after Autologous Hematopoietic SCT: A Pilot Study. Bone Marrow Transplant. 2014, 49, 349–354. [Google Scholar] [CrossRef]
- Hainz, U.; Obexer, P.; Winkler, C.; Sedlmayr, P.; Takikawa, O.; Greinix, H.; Lawitschka, A.; Pötschger, U.; Fuchs, D.; Ladisch, S.; et al. Monocyte-Mediated T-Cell Suppression and Augmented Monocyte Tryptophan Catabolism after Human Hematopoietic Stem-Cell Transplantation. Blood 2005, 105, 4127–4134. [Google Scholar] [CrossRef]
- Lima-Júnior, J.R.; Arruda, L.C.M.; Gonçalves, M.S.; Dias, J.B.E.; Moraes, D.A.; Covas, D.T.; Simões, B.P.; Oliveira, M.C.; Malmegrim, K.C.R. Autologous Haematopoietic Stem Cell Transplantation Restores the Suppressive Capacity of Regulatory B Cells in Systemic Sclerosis Patients. Rheumatology 2021, 60, 5538–5548. [Google Scholar] [CrossRef]
- Gernert, M.; Tony, H.P.; Schwaneck, E.C.; Gadeholt, O.; Schmalzing, M. Autologous Hematopoietic Stem Cell Transplantation in Systemic Sclerosis Induces Long-Lasting Changes in B Cell Homeostasis toward an Anti-Inflammatory B Cell Cytokine Pattern. Arthritis Res. Ther. 2019, 21, 106. [Google Scholar] [CrossRef]
- Gernert, M.; Tony, H.P.; Fröhlich, M.; Schwaneck, E.C.; Schmalzing, M. Immunosuppressive Therapy After Autologous Hematopoietic Stem Cell Transplantation in Systemic Sclerosis Patients-High Efficacy of Rituximab. Front. Immunol. 2022, 12, 817893. [Google Scholar] [CrossRef]
- Moore, J.; Ma, D.; Will, R.; Cannell, P.; Handel, M.; Milliken, S. A Phase II Study of Rituximab in Rheumatoid Arthritis Patients with Recurrent Disease Following Haematopoietic Stem Cell Transplantation. Bone Marrow Transplant. 2004, 34, 241–247. [Google Scholar] [CrossRef][Green Version]
- Pescovitz, M.D.; Torgerson, T.R.; Ochs, H.D.; Ocheltree, E.; McGee, P.; Krause-Steinrauf, H.; Lachin, J.M.; Canniff, J.; Greenbaum, C.; Herold, K.C.; et al. Effect of Rituximab on Human in Vivo Antibody Immune Responses. J. Allergy Clin. Immunol. 2011, 128, 1295–1302.e5. [Google Scholar] [CrossRef] [PubMed]
- Gniadecki, R.; Iyer, A.; Hennessey, D.; Khan, L.; O’Keefe, S.; Redmond, D.; Storek, J.; Durand, C.; Cohen-Tervaert, J.W.; Osman, M. Genomic Instability in Early Systemic Sclerosis. J. Autoimmun. 2022, 131, 102847. [Google Scholar] [CrossRef] [PubMed]
- Aringer, M.; Riemekasten, G. Relevance of Immunomodulatory Therapy for Interstitial Lung Disease in Systemic Sclerosis. Best Pract. Res. Clin. Rheumatol. 2021, 35, 101672. [Google Scholar] [CrossRef] [PubMed]
- Herrick, A.L.; Pan, X.; Peytrignet, S.; Lunt, M.; Hesselstrand, R.; Mouthon, L.; Silman, A.; Brown, E.; Czirják, L.; Distler, J.H.W.; et al. Treatment Outcome in Early Diffuse Cutaneous Systemic Sclerosis: The European Scleroderma Observational Study (ESOS). Ann. Rheum. Dis. 2017, 76, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- del Papa, N.; Onida, F.; Zaccara, E.; Saporiti, G.; Maglione, W.; Tagliaferri, E.; Andracco, R.; Vincenti, D.; Montemurro, T.; Mircoli, L.; et al. Autologous Hematopoietic Stem Cell Transplantation Has Better Outcomes than Conventional Therapies in Patients with Rapidly Progressive Systemic Sclerosis. Bone Marrow Transplant. 2017, 52, 53–58. [Google Scholar] [CrossRef]
- Ait Abdallah, N.; Wang, M.; Lansiaux, P.; Puyade, M.; Berthier, S.; Terriou, L.; Charles, C.; Burt, R.K.; Hudson, M.; Farge, D. Long Term Outcomes of the French ASTIS Systemic Sclerosis Cohort Using the Global Rank Composite Score. Bone Marrow Transplant. 2021, 56, 2259–2267. [Google Scholar] [CrossRef]
- Keyes-Elstein, L.; Pinckney, A.; Goldmuntz, E.; Welch, B.; Franks, J.M.; Martyanov, V.; Wood, T.A.; Crofford, L.; Mayes, M.; McSweeney, P.; et al. Clinical and Molecular Findings after Autologous Stem Cell Transplantation or Cyclophosphamide for Scleroderma: Handling Missing Longitudinal Data. Arthritis Care Res. (Hoboken) 2021, 0, 1–10. [Google Scholar] [CrossRef]
- Nash, R.A.; McSweeney, P.A.; Crofford, L.J.; Abidi, M.; Chen, C.S.; Godwin, J.D.; Gooley, T.A.; Holmberg, L.; Henstorf, G.; LeMaistre, C.F.; et al. High-Dose Immunosuppressive Therapy and Autologous Hematopoietic Cell Transplantation for Severe Systemic Sclerosis: Long-Term Follow-up of the US Multicenter Pilot Study. Blood 2007, 110, 1388–1396. [Google Scholar] [CrossRef]
- Snowden, J.A.; Badoglio, M.; Labopin, M.; Giebel, S.; McGrath, E.; Marjanovic, Z.; Burman, J.; Moore, J.; Rovira, M.; Wulffraat, N.M.; et al. Evolution, trends, outcomes, and economics of hematopoietic stem cell transplantation in severe autoimmune diseases. Blood Adv. 2017, 1, 2742–2755. [Google Scholar] [CrossRef]
- Maltez, N.; Puyade, M.; Wang, M.; Lansiaux, P.; Marjanovic, Z.; Charles, C.; Steele, R.; Baron, M.; Colmegna, I.; Hudson, M.; et al. Association of Autologous Hematopoietic Stem Cell Transplantation in Systemic Sclerosis With Marked Improvement in Health-Related Quality of Life. Arthritis Rheumatol. 2021, 73, 305–314. [Google Scholar] [CrossRef]
- Puyade, M.; Maltez, N.; Lansiaux, P.; Pugnet, G.; Roblot, P.; Colmegna, I.; Hudson, M.; Farge, D. Health-Related Quality of Life in Systemic Sclerosis before and after Autologous Haematopoietic Stem Cell Transplant-a Systematic Review. Rheumatology (Oxford) 2020, 59, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Hudson, M.; Thombs, B.D.; Steele, R.; Panopalis, P.; Newton, E.; Baron, M. Quality of Life in Patients with Systemic Sclerosis Compared to the General Population and Patients with Other Chronic Conditions. J. Rheumatol. 2009, 36, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Furst, D.E.; Clements, P.J.; Allanore, Y.; Baron, M.; Czirjak, L.; Distler, O.; Foeldvari, I.; Kuwana, M.; Matucci-Cerinic, M.; et al. Standardization of the Modified Rodnan Skin Score for Use in Clinical Trials of Systemic Sclerosis. J. Scleroderma Relat. Disord. 2017, 2, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Odani, T.; Yasuda, S.; Noguchi, A.; Fujieda, Y.; Kato, M.; Oku, K.; Bohgaki, T.; Sugita, J.; Endo, T.; et al. Autologous Haematopoietic Stem Cell Transplantation for Japanese Patients with Systemic Sclerosis: Long-Term Follow-up on a Phase II Trial and Treatment-Related Fatal Cardiomyopathy. Mod. Rheumatol. 2018, 28, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Zanin-Silva, D.C.; Santana-Gonçalves, M.; Kawashima-Vasconcelos, M.Y.; Lima-Júnior, J.R.; Dias, J.B.E.; Moraes, D.A.; Covas, D.T.; Malmegrim, K.C.R.; Ramalho, L.; Oliveira, M.C. Autologous Hematopoietic Stem Cell Transplantation Promotes Connective Tissue Remodeling in Systemic Sclerosis Patients. Arthritis Res. Ther. 2022, 24. [Google Scholar] [CrossRef] [PubMed]
- Costa-Pereira, K.R.; Guimarães, A.L.; Moraes, D.A.; Dias, J.B.E.; Garcia, J.T.; de Oliveira-Cardoso, E.A.; Zombrilli, A.; Leopoldo, V.; Costa, T.M.; Simões, B.P.; et al. Hematopoietic Stem Cell Transplantation Improves Functional Outcomes of Systemic Sclerosis Patients. J. Clin. Rheumatol. 2020, 26, S131–S138. [Google Scholar] [CrossRef] [PubMed]
- Burt, R.K.; Oliveira, M.C.; Shah, S.J.; Moraes, D.A.; Simoes, B.; Gheorghiade, M.; Schroeder, J.; Ruderman, E.; Farge, D.; Chai, Z.J.; et al. Cardiac Involvement and Treatment-Related Mortality after Non-Myeloablative Haemopoietic Stem-Cell Transplantation with Unselected Autologous Peripheral Blood for Patients with Systemic Sclerosis: A Retrospective Analysis. Lancet 2013, 381, 1116–1124. [Google Scholar] [CrossRef] [PubMed]
- Santana-Gonçalves, M.; Zanin-Silva, D.; Henrique-Neto, Á.; Moraes, D.A.; Kawashima- Vasconcelos, M.Y.; Lima-Júnior, J.R.; Dias, J.B.E.; Bragagnollo, V.; de Azevedo, J.T.C.; Covas, D.T.; et al. Autologous Hematopoietic Stem Cell Transplantation Modifies Specific Aspects of Systemic Sclerosis-Related Microvasculopathy. Ther. Adv. Musculoskelet Dis. 2022, 14. [Google Scholar] [CrossRef]
- Schieir, O.; Thombs, B.D.; Hudson, M.; Boivin, J.F.; Steele, R.; Bernatsky, S.; Hanley, J.; Baron, M.; Pope, J.; Markland, J.; et al. Prevalence, Severity, and Clinical Correlates of Pain in Patients with Systemic Sclerosis. Arthritis Care Res. (Hoboken) 2010, 62, 409–417. [Google Scholar] [CrossRef]
- Tuffanelli, D.L.; Winkelmann, R.K. Systemic Scleroderma: A Clinical Study of 727 Cases. Arch. Dermatol. 1961, 84, 359–371. [Google Scholar] [CrossRef]
- Baron, M.; Lee, P.; Keystone, E.C. The Articular Manifestations of Progressive Systemic Sclerosis (Scleroderma). Ann. Rheum. Dis. 1982, 41, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Steen, V.D.; Conte, C.; Owens, G.R.; Medsger, T.A. Severe Restrictive Lung Disease in Systemic Sclerosis. Arthritis Rheum 1994, 37, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Solomon, J.J.; Olson, A.; Fischer, A.; Bull, T.; Brown, K.K.; Raghu, G. Scleroderma lung disease. Eur. Respir. Rev. 2013, 22, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Kloth, C.; Henes, J.; Xenitidis, T.; Thaiss, W.M.; Blum, A.C.; Fritz, J.; Nikolaou, K.; Horger, M.; Ioanoviciu, S.D. Chest CT Texture Analysis for Response Assessment in Systemic Sclerosis. Eur. J. Radiol. 2018, 101, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Wada, D.T.; de Almeida, F.A.; de Moraes, D.A.; Dias, J.B.E.; Baddini-Martinez, J.; Oliveira, M.C.; Koenigkam-Santos, M. Automatic Quantitative Computed Tomography Evaluation of the Lungs in Patients With Systemic Sclerosis Treated With Autologous Stem Cell Transplantation. J. Clin. Rheumatol. 2020, 26, S158–S164. [Google Scholar] [CrossRef]
- Pugnet, G.; Petermann, A.; Collot, S.; Otal, P.; Lansiaux, P.; Maria, A.; Abdallah, N.A.; Lorillon, G.; Resch-Rigon, M.; Marjanovic, Z.; et al. Longitudinal Changes on Quantitative Chest High-Resolution Computed Tomography in Early Diffuse Systemic Sclerosis-Related Interstitial Lung Disease After Autologous Hematopoietic Stem Cell Transplantation. Ann. Rheum. Dis. 2022, 81, 251. [Google Scholar] [CrossRef]
- Nihtyanova, S.I.; Schreiber, B.E.; Ong, V.H.; Rosenberg, D.; Moinzadeh, P.; Coghlan, J.G.; Wells, A.U.; Denton, C.P. Prediction of Pulmonary Complications and Long-Term Survival in Systemic Sclerosis. Arthritis Rheumatol. 2014, 66, 1625–1635. [Google Scholar] [CrossRef]
- Henes, J.C.; Koetter, I.; Horger, M.; Schmalzing, M.; Mueller, K.; Eick, C.; Bauer, A.; Vogel, W.; Kanz, L. Autologous Stem Cell Transplantation with Thiotepa-Based Conditioning in Patients with Systemic Sclerosis and Cardiac Manifestations. Rheumatology (Oxford) 2014, 53, 919–922. [Google Scholar] [CrossRef]
- Kloth, C.; Maximilian Thaiss, W.; Preibsch, H.; Mark, K.; Kötter, I.; Hetzel, J.; Nikolaou, K.; Henes, J.; Horger, M. Quantitative Chest CT Analysis in Patients with Systemic Sclerosis before and after Autologous Stem Cell Transplantation: Comparison of Results with Those of Pulmonary Function Tests and Clinical Tests. Rheumatology (Oxford) 2016, 55, 1763–1770. [Google Scholar] [CrossRef]
- Ciaffi, J.; van Leeuwen, N.M.; Boonstra, M.; Kroft, L.J.M.; Schouffoer, A.A.; Ninaber, M.K.; Huizinga, T.W.J.; de Vries-Bouwstra, J.K. Evolution of Systemic Sclerosis–Associated Interstitial Lung Disease One Year After Hematopoietic Stem Cell Transplantation or Cyclophosphamide. Arthritis Care Res. (Hoboken) 2022, 74, 433–441. [Google Scholar] [CrossRef]
- Gotschy, A.; Jordan, S.; Stoeck, C.T.; von Deuster, C.; Peer, T.; Gastl, M.; Vishnevskiy, V.; Wissmann, L.; Dobrota, R.; Mihai, C.; et al. Diffuse Myocardial Fibrosis Precedes Subclinical Functional Myocardial Impairment and Provides Prognostic Information in Systemic Sclerosis. Eur. Heart J. Cardiovasc. Imaging 2022. [Google Scholar] [CrossRef]
- Faludi, R.; Költo, G.; Bartos, B.; Csima, G.; Czirják, L.; Komócsi, A. Five-Year Follow-up of Left Ventricular Diastolic Function in Systemic Sclerosis Patients: Determinants of Mortality and Disease Progression. Semin. Arthritis Rheum. 2014, 44, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Hachulla, A.L.; Launay, D.; Gaxotte, V.; de Groote, P.; Lamblin, N.; Devos, P.; Hatron, P.Y.; Beregi, J.P.; Hachulla, E. Extended Report: Cardiac Magnetic Resonance Imaging in Systemic Sclerosis: A Cross-Sectional Observational Study of 52 Patients. Ann. Rheum. Dis. 2009, 68, 1878. [Google Scholar] [CrossRef]
- Sullivan, K.; Pinckney, A.; Goldmuntz, E.; Welch, B.; Khanna, D.; Simms, R.; Kafaja, S.; Georges, G.; Storek, J.; Csuka, M.E.; et al. Myeloablative Autologous Hematopoietic Stem Cell Transplantation for Severe Scleroderma: Long-Term Outcomes 6-11 Years after Entry on a Randomized Study Comparing Transplantation and Cyclophosphamide. In Proceedings of the 2018 ACR/ARHP Annual Meeting, Chicago, IL, USA, 19–24 October 2018. [Google Scholar]
- Pugnet, G.; Petermann, A.; Collot, S.; Otal, P.; Lansiaux, P.; Ait Abdallah, N.; Lorillon, G.; Resche-Rigon, M.; Borel, C.; Marjanovic, Z.; et al. Changes on Chest HRCT in Systemic Sclerosis-Related Interstitial Lung Disease after Autologous Haematopoietic Stem Cell Transplantation. Rheumatology 2022. [Google Scholar] [CrossRef] [PubMed]
- Denaxas, K.; Ladas, S.D.; Karamanolis, G.P. Evaluation and Management of Esophageal Manifestations in Systemic Sclerosis. Ann. Gastroenterol. 2018, 31, 165. [Google Scholar] [CrossRef] [PubMed]
- Hudson, M.; Baron, M.; Tatibouet, S.; Furst, D.E.; Khanna, D.; Hummers, L.; Hachulla, E.; Medsger, T.; Steen, V.; Alkassab, F.; et al. Exposure to ACE Inhibitors Prior to the Onset of Scleroderma Renal Crisis-Results from the International Scleroderma Renal Crisis Survey. Semin. Arthritis Rheum. 2014, 43, 666–672. [Google Scholar] [CrossRef]
- Gadioli, L.P.; Costa-Pereira, K.; Dias, J.B.E.; Moraes, D.A.; Crescêncio, J.C.; Schwartzmann, P.V.; Gallo-Júnior, L.; Schmidt, A.; Oliveira, M.C. Autologous Stem Cell Transplantation Improves Cardiopulmonary Exercise Testing Outcomes in Systemic Sclerosis Patients. Rheumatology 2022. [Google Scholar] [CrossRef]
- Penglase, R.; Englert, H.; Tymms, K.; Carr, A.; Ma, D.; Milliken, S.; Moore, J. Successful Lymphoablative Autologous Haematopoietic Stem Cell Transplantation in Three Cases of Severe Autoimmune Disease despite Reduced Dose Cyclophosphamide Conditioning—Do We Need 200 Mg/Kg Cyclophosphamide? Bone Marrow Transplant. 2022, 57, 1207–1209. [Google Scholar] [CrossRef]
- Almehmadi, B.A.; To, F.Z.; Anderson, M.A.; Johnson, S.R. Epidemiology and Treatment of Peripheral Neuropathy in Systemic Sclerosis. J. Rheumatol. 2021, 48, 1839–1849. [Google Scholar] [CrossRef]
- Sulli, A.; Pizzorni, C.; Smith, V.; Zampogna, G.; Ravera, F.; Cutolo, M. Timing of Transition between Capillaroscopic Patterns in Systemic Sclerosis. Arthritis Rheum. 2012, 64, 821–825. [Google Scholar] [CrossRef]
- Aschwanden, M.; Daikeler, T.; Jaeger, K.A.; Thalhammer, C.; Gratwohl, A.; Matucci-Cerinic, M.; Tyndall, A. Rapid Improvement of Nailfold Capillaroscopy after Intense Immunosuppression for Systemic Sclerosis and Mixed Connective Tissue Disease. Ann. Rheum. Dis. 2008, 67, 1057–1059. [Google Scholar] [CrossRef] [PubMed]
- Daikeler, T.; Kump, E.; Stern, M.; Hügle, T.; Hij, A.; Haeuserman, P.; Farge, D. Autologous Hematopoietic Stem Cell Transplantation Reverses Skin Fibrosis but Does Not Change Skin Vessel Density in Patients with Systemic Sclerosis. Pathol. Biol. (Paris) 2015, 63, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, A.; Khalid, M.; Ishaq, Q.; Donnelly, S. Scleroderma-Associated Cutaneous Calcinosis: A Remarkable Regression Post Stem Cell Transplantation. Clin. Rheumatol. 2021, 40, 789–790. [Google Scholar] [CrossRef]
- de Pietri, L.; Serra, V.; Preziosi, G.; Rompianesi, G.; Begliomini, B. Perioperative Effects of High Doses of Intraoperative Thymoglobulin Induction in Liver Transplantation. World J. Transplant. 2015, 5, 320. [Google Scholar] [CrossRef] [PubMed]
- Henrique-Neto, Á.; Vasconcelos, M.Y.K.; Dias, J.B.E.; de Moraes, D.A.; Gonçalves, M.S.; Zanin-Silva, D.C.; Zucoloto, T.G.; de Oliveira, M.d.F.C.; Dotoli, G.M.; Weffort, L.F.; et al. Hematopoietic Stem Cell Transplantation for Systemic Sclerosis: Brazilian Experience. Adv. Rheumatol. 2021, 61, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Farge, D.; Burt, R.K.; Oliveira, M.C.; Mousseau, E.; Rovir, M.; Marjanovi, Z.; de Vries-Bouwstr, J.; del Pap, N.; Saccard, R.; Shah, S.J.; et al. Cardiopulmonary Assessment of Patients with Systemic Sclerosis for Hematopoietic Stem Cell Transplantation: Recommendations from the European Society for Blood and Marrow Transplantation Autoimmune Diseases Working Party and Collaborating Partners. Bone Marrow Transplant. 2017, 52, 1495–1503. [Google Scholar] [CrossRef]
- Shah, A.; Spierings, J.; van Laar, J.M.; Sullivan, K.M. Re-Evaluating Inclusion Criteria for Autologous Hematopoietic Stem Cell Transplantation in Advanced Systemic Sclerosis: Three Successful Cases and Review of the Literature. J. Scleroderma Relat. Disord. 2021, 6, 199–205. [Google Scholar] [CrossRef]
- González García, A.; Fabregate, M.; Manzano, L.; Guillén del Castillo, A.; Rubio Rivas, M.; Argibay, A.; Marín Ballvé, A.; Rodríguez Pintó, I.; Pla Salas, X.; Marí-Alfonso, B.; et al. Left Ventricular Diastolic Dysfunction in Systemic Sclerosis: Clinical, Immunological and Survival Differences in the Spanish RESCLE Registry. Semin. Arthritis Rheum. 2022, 55, 152033. [Google Scholar] [CrossRef]
- Franks, J.M.; Martyanov, V.; Wang, Y.; Wood, T.A.; Pinckney, A.; Crofford, L.J.; Keyes-Elstein, L.; Furst, D.E.; Goldmuntz, E.; Mayes, M.D.; et al. Machine Learning Predicts Stem Cell Transplant Response in Severe Scleroderma. Ann. Rheum. Dis. 2020, 79, 1608–1615. [Google Scholar] [CrossRef]
- Boukouaci, W.; Lansiaux, P.; Lambert, N.C.; Picard, C.; Clave, E.; Cras, A.; Marjanovic, Z.; Farge, D.; Tamouza, R. Non-Classical HLA Determinants of the Clinical Response after Autologous Stem Cell Transplantation for Systemic Sclerosis. Int. J. Mol. Sci. 2022, 23, 7223. [Google Scholar] [CrossRef]
- Jury, E.C.; D’Cruz, D.; Morrow, W.J.W. Autoantibodies and Overlap Syndromes in Autoimmune Rheumatic Disease. J. Clin. Pathol. 2001, 54, 340–347. [Google Scholar] [CrossRef]
- Ramalingam, S.; Shah, A. Stem Cell Therapy as a Treatment for Autoimmune Disease—Updates in Lupus, Scleroderma, and Multiple Sclerosis. Curr. Allergy Asthma Rep. 2021, 21, 22. [Google Scholar] [CrossRef] [PubMed]
- Pecher, A.-C.; Ach, K.R.; Vogel, W.; Henes, J.C. Mobilization with Reduced Cyclophosphamide for Autologous Stem Cell Transplantation Is Feasible in Patients with Systemic Sclerosis. Rheumatology 2022. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.C.; Labopin, M.; Henes, J.; Moore, J.; Papa, N.D.; Cras, A.; Sakellari, I.; Schroers, R.; Scherer, H.U.; Cuneo, A.; et al. Does Ex Vivo CD34+ Positive Selection Influence Outcome after Autologous Hematopoietic Stem Cell Transplantation in Systemic Sclerosis Patients? Bone Marrow Transplant. 2016, 51, 501–505. [Google Scholar] [CrossRef]
- Ayano, M.; Tsukamoto, H.; Mitoma, H.; Kimoto, Y.; Akahoshi, M.; Arinobu, Y.; Miyamoto, T.; Horiuchi, T.; Niiro, H.; Nagafuji, K.; et al. CD34-Selected versus Unmanipulated Autologous Haematopoietic Stem Cell Transplantation in the Treatment of Severe Systemic Sclerosis: A Post Hoc Analysis of a Phase I/II Clinical Trial Conducted in Japan. Arthritis Res. Ther. 2019, 21, 30. [Google Scholar] [CrossRef]
- Snowden, J.A.; Saccardi, R.; Allez, M.; Ardizzone, S.; Arnold, R.; Cervera, R.; Denton, C.; Hawkey, C.; Labopin, M.; Mancardi, G.; et al. Haematopoietic SCT in Severe Autoimmune Diseases: Updated Guidelines of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 2012, 47, 770–790. [Google Scholar] [CrossRef]
- Matteson, E.L.; Shbeeb, M.I.; McCarthy, T.G.; Calamia, K.T.; Mertz, L.E.; Goronzy, J.J. Pilot Study of Antithymocyte Globulin in Systemic Sclerosis. Arthritis Rheum. 1996, 39, 1132–1137. [Google Scholar] [CrossRef] [PubMed]
- Stratton, R.J.; Wilson, H.; Black, C.M. Pilot Study of Anti-thymocyte Globulin plus Mycophenolate Mofetil in Recent-onset Diffuse Scleroderma. Rheumatology 2001, 40, 84–88. [Google Scholar] [CrossRef]
- Balaban, E.P.; Zashin, S.J.; Geppert, T.D.; Lipsky, P.E.; Condie, R.M. Treatment of Systemic Sclerosis with Antithymocyte Globulin. Arthritis Rheum. 1991, 34, 244–245. [Google Scholar] [CrossRef]
- Jaime-Pérez, J.C.; González-Treviño, M.; Meléndez-Flores, J.D.; Ramos-Dávila, E.M.; Cantú-Rodriguez, O.G.; Gutiérrez-Aguirre, C.H.; Galarza-Delgado, D.A.; Gómez-Almaguer, D. Autologous ATG-Free Hematopoietic Stem Cell Transplantation for Refractory Autoimmune Rheumatic Diseases: A Latin American Cohort. Clin. Rheumatol. 2022, 41, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Helbig, G.; Widuchowska, M.; Koclęga, A.; Kopińska, A.; Kopeć-Mędrek, M.; Gaweł, W.B.; Spałek, A.; Żak, J.; Grygoruk-Wiśniowska, I.; Liwoch, R.; et al. Safety Profile of Autologous Hematopoietic Stem Cell Mobilization and Transplantation in Patients with Systemic Sclerosis. Clin. Rheumatol. 2018, 37, 1709–1714. [Google Scholar] [CrossRef]
- Loh, Y.; Oyama, Y.; Statkute, L.; Quigley, K.; Yaung, K.; Gonda, E.; Barr, W.; Jovanovic, B.; Craig, R.; Stefoski, D.; et al. Development of a Secondary Autoimmune Disorder after Hematopoietic Stem Cell Transplantation for Autoimmune Diseases: Role of Conditioning Regimen Used. Blood 2007, 109, 2643. [Google Scholar] [CrossRef]
- Droller, M.J.; Saral, R.; Santos, G. Prevention of Cyclophosphamide-Induced Hemorrhagic Cystitis. Urology 1982, 20, 256–258. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, J.D.; Pringle, L.E.; Barnett, M.J.; Klingemann, H.G.; Reece, D.E.; Phillips, G.L. Mesna versus Hyperhydration for the Prevention of Cyclophosphamide-Induced Hemorrhagic Cystitis in Bone Marrow Transplantation. J. Clin. Oncol. 2016, 9, 2016–2020. [Google Scholar] [CrossRef] [PubMed]
- Daikeler, T.; Tichelli, A.; Passweg, J. Complications of Autologous Hematopoietic Stem Cell Transplantation for Patients with Autoimmune Diseases. Pediatric Res. 2012, 71, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Fornecker, L.M.; Marcellin, L.; Mousseaux, E.; Hij, A.; Snowden, J.A.; Farge, D.; Martin, T. Acute and Fatal Cardiotoxicity Following High-Dose Cyclophosphamide in a Patient Undergoing Autologous Stem Cell Transplantation for Systemic Sclerosis despite Satisfactory Cardiopulmonary Screening. Bone Marrow Transplant. 2017, 52, 1674–1677. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, C.; Gingrich, R.; hohl, R.; Ajram, K.A. Engraftment Syndrome in Autologous Bone Marrow and Peripheral Stem Cell Transplantation. Bone Marrow Transplant. 1995, 16, 175–182. [Google Scholar]
- Cornell, R.F.; Hari, P.; Drobyski, W.R. Engraftment Syndrome after Autologous Stem Cell Transplantation: An Update Unifying the Definition and Management Approach. Biol. Blood Marrow Transplant. 2015, 21, 2061–2068. [Google Scholar] [CrossRef]
- Strunz, P.P.; Froehlich, M.; Gernert, M.; Schwaneck, E.C.; Fleischer, A.; Pecher, A.C.; Tony, H.P.; Henes, J.C.; Schmalzing, M. Immunological Adverse Events After Autologous Hematopoietic Stem Cell Transplantation in Systemic Sclerosis Patients. Front. Immunol. 2021, 12, 3526. [Google Scholar] [CrossRef]
- Carter, A.; Robison, L.L.; Francisco, L.; Smith, D.; Grant, M.; Baker, K.S.; Gurney, J.G.; McGlave, P.B.; Weisdorf, D.J.; Forman, S.J.; et al. Prevalence of Conception and Pregnancy Outcomes after Hematopoietic Cell Transplantation: Report from the Bone Marrow Transplant Survivor Study. Bone Marrow Transplant. 2006, 37, 1023–1029. [Google Scholar] [CrossRef]
- Massarotti, C.; Sbragia, E.; Boffa, G.; Vercelli, C.; Zimatore, G.B.; Cottone, S.; Frau, J.; Raiola, A.; Varaldo, R.; Mancardi, G.; et al. Menstrual Cycle Resumption and Female Fertility after Autologous Hematopoietic Stem Cell Transplantation for Multiple Sclerosis. Mult. Scler. J. 2021, 27, 2103–2107. [Google Scholar] [CrossRef]
- Maciejewska, M.; Snarski, E.; Wiktor-Jędrzejczak, W. A Preliminary Online Study on Menstruation Recovery in Women After Autologous Hematopoietic Stem Cell Transplant for Autoimmune Diseases. Exp. Clin. Transplant. 2016, 14, 665–669. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Massenkeil, G.; Alexander, T.; Rosen, O.; Dörken, B.; Burmester, G.; Radbruch, A.; Hiepe, F.; Arnold, R. Long-Term Follow-up of Fertility and Pregnancy in Autoimmune Diseases after Autologous Haematopoietic Stem Cell Transplantation. Rheumatol. Int. 2016, 36, 1563–1568. [Google Scholar] [CrossRef] [PubMed]
- Danylesko, I.; Shimoni, A. Second Malignancies after Hematopoietic Stem Cell Transplantation. Curr. Treat. Options Oncol. 2018, 19, 9. [Google Scholar] [CrossRef]
- Tarella, C.; Passera, R.; Magni, M.; Benedetti, F.; Rossi, A.; Gueli, A.; Patti, C.; Parvis, G.; Ciceri, F.; Gallamini, A.; et al. Risk Factors for the Development of Secondary Malignancy After High-Dose Chemotherapy and Autograft, With or Without Rituximab: A 20-Year Retrospective Follow-Up Study in Patients With Lymphoma. J. Clin. Oncol. 2011, 29, 814–824. [Google Scholar] [CrossRef] [PubMed]
- Bernal-Bello, D.; de Tena, J.G.; Guillén-del Castillo, A.; Selva-O’Callaghan, A.; Callejas-Moraga, E.L.; Marín-Sánchez, A.M.; Fonollosa-Pla, V.; Simeón-Aznar, C.P. Novel Risk Factors Related to Cancer in Scleroderma. Autoimmun. Rev. 2017, 16, 461–468. [Google Scholar] [CrossRef]
- Bonifazi, M.; Tramacere, I.; Pomponio, G.; Gabrielli, B.; Avvedimento, E.V.; la Vecchia, C.; Negri, E.; Gabrielli, A. Systemic Sclerosis (Scleroderma) and Cancer Risk: Systematic Review and Meta-Analysis of Observational Studies. Rheumatology 2013, 52, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Talar-Williams, C.; Hijazi, Y.M.; Walther, M.M.; Linehan, W.M.; Hallahan, C.W.; Lubensky, I.; Kerr, G.S.; Hoffman, G.S.; Fauci, A.S.; Sneller, M.C. Cyclophosphamide-Induced Cystitis and Bladder Cancer in Patients with Wegener Granulomatosis. Ann. Intern. Med. 1996, 124, 477–484. [Google Scholar] [CrossRef]
- Faurschou, M.; Sorensen, I.J.; Mellemkjaer, L.; Loft, A.G.R.; Thomsen, B.S.; Tvede, N.; Baslund, B. Malignancies in Wegener’s Granulomatosis: Incidence and Relation to Cyclophosphamide Therapy in a Cohort of 293 Patients. J. Rheumatol. 2008, 35, 100–105. [Google Scholar]
- Daikeler, T.; Labopin, M.; di Gioia, M.; Abinun, M.; Alexander, T.; Miniati, I.; Gualandi, F.; Fassas, A.; Martin, T.; Schwarze, C.P.; et al. Secondary Autoimmune Diseases Occurring after HSCT for an Autoimmune Disease: A Retrospective Study of the EBMT Autoimmune Disease Working Party. Blood 2011, 118, 1693–1698. [Google Scholar] [CrossRef]
- Guillaume-Jugnot, P.; Badoglio, M.; Labopin, M.; Terriou, L.; Yakoub-Agha, I.; Martin, T.; Lioure, B.; Marjanovic, Z.; Blaise, D.; Nguyen, S.; et al. Autologous Haematopoietic Stem Cell Transplantation (AHSCT) in Autoimmune Disease Adult Patients in France: Analysis of the Long-Term Outcome from the French Society for Bone Marrow Transplantation and Cellular Therapy (SFGM-TC). Clin. Rheumatol. 2019, 38, 1501–1511. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Mittoo, S.; Aggarwal, R.; Proudman, S.M.; Dalbeth, N.; Matteson, E.L.; Brown, K.; Flaherty, K.; Wells, A.U.; Seibold, J.R.; et al. Connective Tissue Disease-Associated Interstitial Lung Diseases (CTD-ILD) - Report from OMERACT CTD-ILD Working Group. J. Rheumatol. 2015, 42, 2168–2171. [Google Scholar] [CrossRef]
- Henes, J.C.; Schmalzing, M.; Vogel, W.; Riemekasten, G.; Fend, F.; Kanz, L.; Koetter, I. Optimization of Autologous Stem Cell Transplantation for Systemic Sclerosis — A Single-Center Longterm Experience in 26 Patients with Severe Organ Manifestations. J. Rheumatol. 2012, 39, 269–275. [Google Scholar] [CrossRef]
- van Rhijn-Brouwer, F.C.C.; Spierings, J.; van Rhenen, A.; Kuball, J.; van Laar, J.M. Second Autologous Haematopoietic Stem Cell Transplantation in Systemic Sclerosis—a Case Report. Rheumatology 2019, 58, 1305–1307. [Google Scholar] [CrossRef]
- Bolaños-Meade, J.; Cooke, K.R.; Gamper, C.J.; Ali, S.A.; Ambinder, R.F.; Borrello, I.M.; Fuchs, E.J.; Gladstone, D.E.; Gocke, C.B.; Huff, C.A.; et al. Effect of Increased Dose of Total Body Irradiation on Graft Failure Associated with HLA-Haploidentical Transplantation in Patients with Severe Haemoglobinopathies: A Prospective Clinical Trial. Lancet. Haematol. 2019, 6, e183–e193. [Google Scholar] [CrossRef]
- DeZern, A.E.; Zahurak, M.; Symons, H.; Cooke, K.; Jones, R.J.; Brodsky, R.A. Alternative Donor Transplantation with High-Dose Post-Transplantation Cyclophosphamide for Refractory Severe Aplastic Anemia. Biol Blood Marrow Transplant. 2017, 23, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Saraf, S.L.; Oh, A.L.; Patel, P.R.; Sweiss, K.; Koshy, M.; Campbell-Lee, S.; Gowhari, M.; Jain, S.; Peace, D.; Quigley, J.G.; et al. Haploidentical Peripheral Blood Stem Cell Transplantation Demonstrates Stable Engraftment in Adults with Sickle Cell Disease. Biol. Blood Marrow Transplant. 2018, 24, 1759–1765. [Google Scholar] [CrossRef] [PubMed]
- DeZern, A.E.; Zahurak, M.L.; Symons, H.J.; Cooke, K.R.; Rosner, G.L.; Gladstone, D.E.; Huff, C.A.; Swinnen, L.J.; Imus, P.; Borrello, I.; et al. Haploidentical BMT for Severe Aplastic Anemia with Intensive GVHD Prophylaxis Including Posttransplant Cyclophosphamide. Blood Adv. 2020, 4, 1770–1779. [Google Scholar] [CrossRef]
- Ozdogu, H.; Boga, C.; Yeral, M.; Kozanoglu, I.; Gereklioglu, C.; Aytan, P.; Kasar, M.; Asma, S.; Buyukkurt, N.; Solmaz, S.; et al. Allogenic Peripheral Stem Cell Transplantation from HLA-Matched Related Donors for Adult Sickle Cell Disease: Remarkable Outcomes from a Single-Center Trial. Bone Marrow Transplant. 2018, 53, 880–890. [Google Scholar] [CrossRef] [PubMed]
- Greco, R.; Labopin, M.; Badoglio, M.; Veys, P.; Silva, J.M.F.; Abinun, M.; Gualandi, F.; Bornhauser, M.; Ciceri, F.; Saccardi, R.; et al. Allogeneic HSCT for Autoimmune Diseases: A Retrospective Study from the EBMT ADWP, IEWP, and PDWP Working Parties. Front. Immunol. 2019, 10, 1570. [Google Scholar] [CrossRef]
- Loh, Y.; Oyama, Y.; Statkute, L.; Verda, L.; Quigley, K.; Yaung, K.; Barr, W.; Jovanovic, B.; Burt, R.K. Non-Myeloablative Allogeneic Hematopoietic Stem Cell Transplantation for Severe Systemic Sclerosis: Graft-versus-Autoimmunity without Graft-versus-Host Disease? Bone Marrow Transplant. 2007, 39, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Khorshid, O.; Hosing, C.; Bibawi, S.; Ueno, N.; Reveille, J.; Mayes, M.; Champlin, R. Nonmyeloablative Stem Cell Transplant in a Patient with Advanced Systemic Sclerosis and Systemic Lupus Erythematosus. J. Rheumatol. 2004, 31, 2516. [Google Scholar]
- Shifa, I.; Hazlewood, G.S.; Durand, C.; Barr, S.G.; Mydlarski, P.R.; Beck, P.L.; Burton, J.M.; Khan, F.M.; Jamani, K.; Osman, M.; et al. Efficacy of Allogeneic Hematopoietic Cell Transplantation for Autoimmune Diseases. Transplant. Cell Ther. 2021, 27, 489.e1–489.e9. [Google Scholar] [CrossRef] [PubMed]
- Spierings, J.; van Rhenen, A.; Welsing, P.M.W.; Marijnissen, A.C.A.; de Langhe, E.; del Papa, N.; Dierickx, D.; Gheorghe, K.R.; Henes, J.; Hesselstrand, R.; et al. A Randomised, Open-Label Trial to Assess the Optimal Treatment Strategy in Early Diffuse Cutaneous Systemic Sclerosis: The UPSIDE Study Protocol. BMJ Open 2021, 11, e044483. [Google Scholar] [CrossRef] [PubMed]
- Georges, G. Scleroderma Treatment With Autologous Transplant (STAT) Study. Available online: https://clinicaltrials.gov/ct2/show/NCT01413100 (accessed on 26 July 2022).
- Farge, D.; Loisel, S.; Resche-Rigon, M.; Lansiaux, P.; Colmegna, I.; Langlais, D.; Charles, C.; Pugnet, G.; Maria, A.T.J.; Chatelus, E.; et al. Safety and Preliminary Efficacy of Allogeneic Bone Marrow-Derived Multipotent Mesenchymal Stromal Cells for Systemic Sclerosis: A Single-Centre, Open-Label, Dose-Escalation, Proof-of-Concept, Phase 1/2 Study. Lancet. Rheumatol. 2022, 4, e91–e104. [Google Scholar] [CrossRef]
- Huang, H. A Study of CD19/BCMA Chimeric Antigen Receptor T Cells Therapy for Patients With Refractory Scleroderma. Zhejiang. 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT05085431 (accessed on 26 July 2022).

| Manifestation/Aspect of SSc | Effect of HCT | Level of Evidence ** | ||
|---|---|---|---|---|
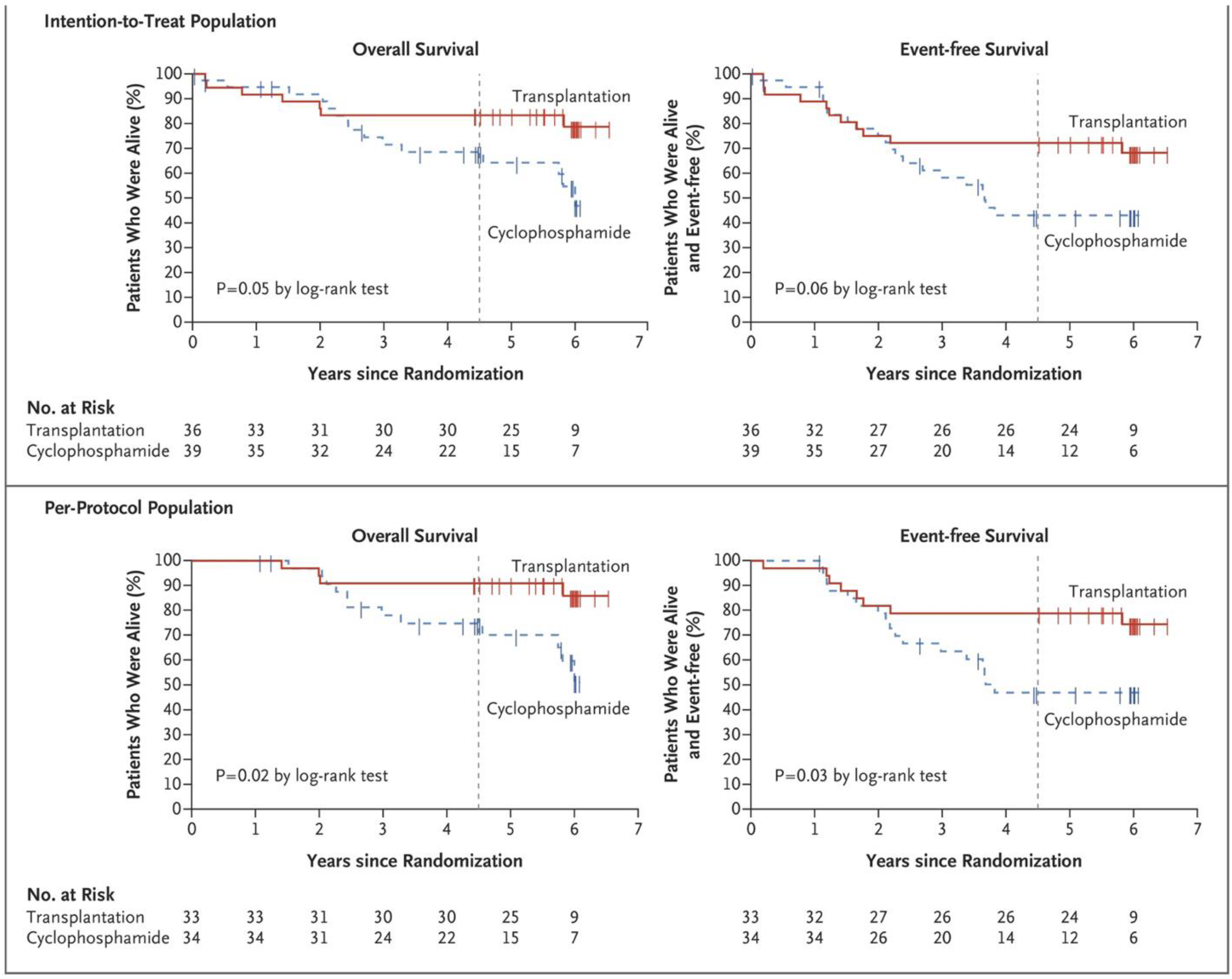
| Mortality | Improvement | High [21,22,23] | ||
| Quality of life | Improvement | High [21,22,23,65,71,72,73] | ||
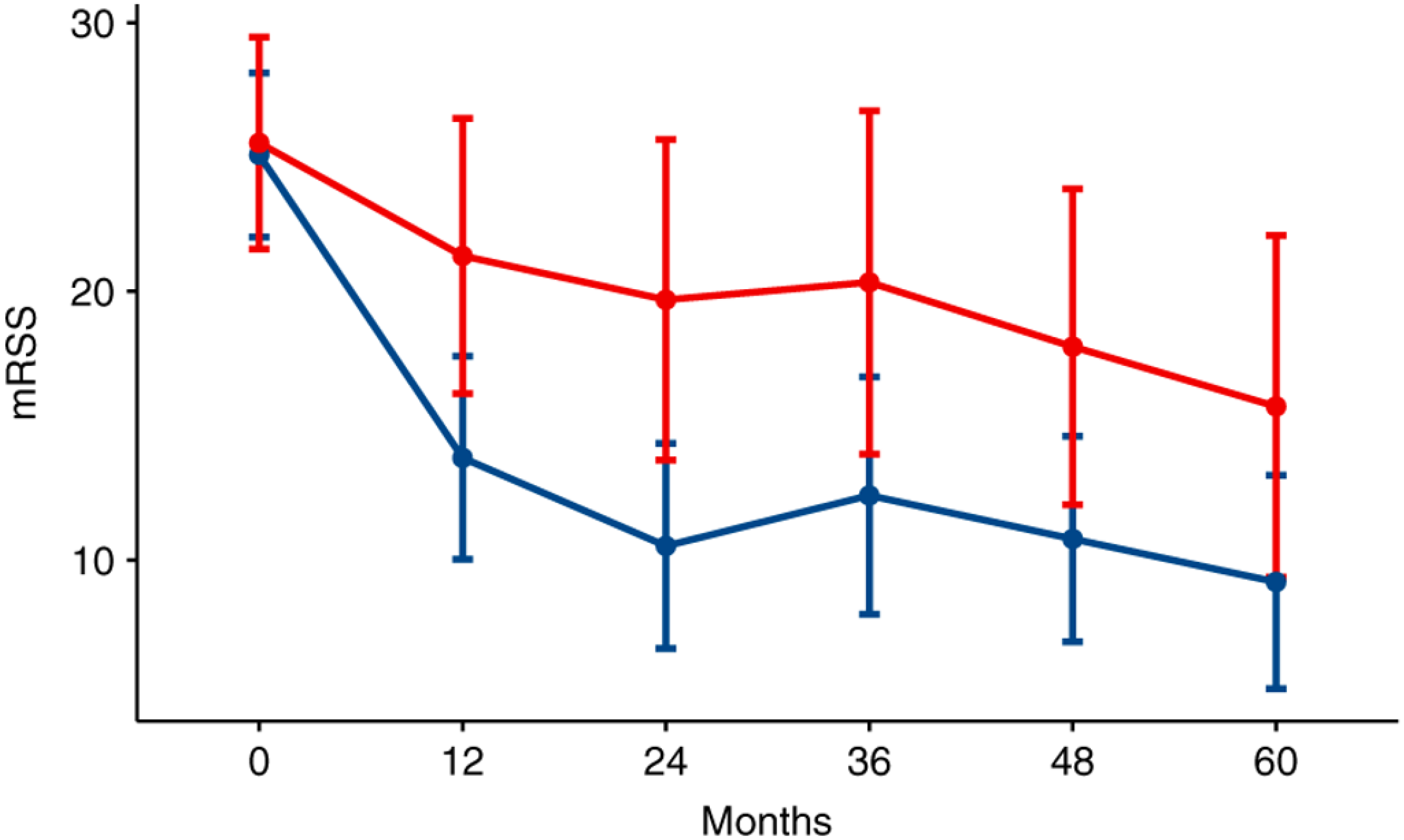
| Skin tightness (mRSS) [74] | Improvement | High [21,22,23,24,25,44,65,66,72,75,76,77,78,79] | ||
| Pain [80,81,82] | Improvement | Medium [71] | ||
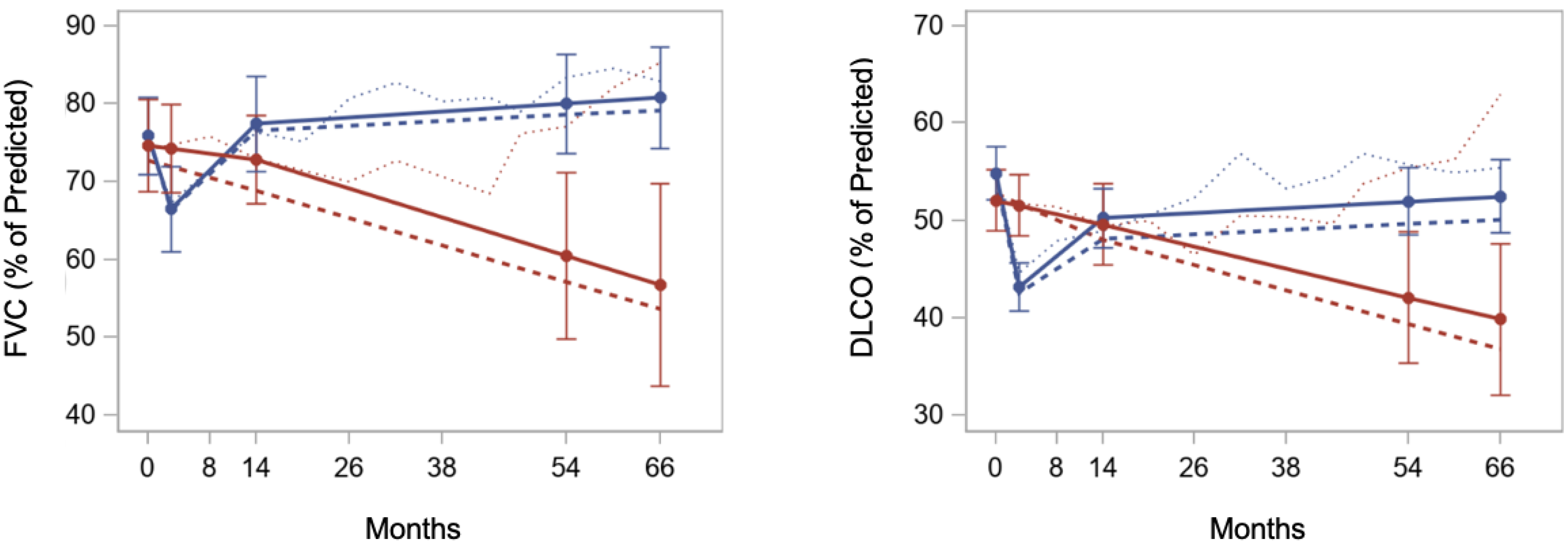
| Lung function (FVC) [83,84] | Mild improvement | High [21,22,23,28,75,78,85,86,87] | ||
| Interstitial lung disease (CT) [88] | Improvement of inflammation, probably not fibrosis | Medium [28,85,86,87,89,90,91] | ||
| Pulmonary arterial hypertension | Preventing PAH; effect on established PAH is unknown | High [23] | ||
| Heart failure [92,93,94] | Preventing or delaying HF; effect on established HF is unknown | High [23,95] | ||
| Esophageal volume/dilation (CT) | Worsening | Medium [96] | ||
| Esophageal motility | Stabilization or mild improvement(?) if pre-transplant hypomotility; no improvement if pre-transplant amotility | Medium, but unpublished [M.W. et al., publication in progress] | ||
| Gastrointestinal symptoms [97] | Improvement | Medium [71] | ||
| Renal function [98] | Worsening | High, but shown in only 1 of 3 randomized studies [22] | ||
| Range of motion of joints | Improvement | Medium [77] | ||
| Hand grip strength | Improvement | Medium [77] | ||
| Exercise capacity | Improvement | Medium [99] | ||
| Myositis | May improve | Low [100] | ||
| Peripheral neuropathy | May improve | Low [101] | ||
| Nailfold capillaries [102] | May improve | Medium [103] | ||
| Capillary density on skin biopsy | No improvement | Medium [104] | ||
| Raynaud’s phenomenon | May improve late after HCT | Medium [71] | ||
| Calcinosis | May improve | Low [105] | ||
| Conventional Criteria | Expanded Criteria | |
|---|---|---|
| Age | 18–65 Years of Age | No Expansion |
| Disease Duration | ≤5 years from first non-Raynaud’s manifestation | Shah et al. 2021 [109]:
|
| SSc Subtype/mRSS | dcSSc
| Burt et al. 2021:
|
| Smoking | Non-current smokers | No expansion |
| Pregnancy | Non-pregnant individuals | No expansion |
| Cardiopulmonary Involvement |
| Shah et al. 2021:
|
| Hepatic Involvement | No evidence of persistent or progressive hepatic impairment defined as persistent increase in rate or twice-normal transaminases/bilirubin | No expansion |
| Renal Involvement | GFR > 40 mL/min | No expansion |
| Hematolymphatic System Status | No evidence of persistent neutropenia (neutrophils <0.5 × 109/L), thrombocytopenia (<50 × 109/L), or CD4 lymphopenia (<200/mm3) | No expansion |
| Nutritional Status | BMI ≥ 18 kg/m2, Albumin ≥ 20 mg/L | No expansion |
| Other | No active neoplasia or concomitant myelodysplasia, no acute or chronic uncontrolled infection, good patient compliance | No expansion |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levin, D.; Osman, M.S.; Durand, C.; Kim, H.; Hemmati, I.; Jamani, K.; Howlett, J.G.; Johannson, K.A.; Weatherald, J.; Woo, M.; et al. Hematopoietic Cell Transplantation for Systemic Sclerosis—A Review. Cells 2022, 11, 3912. https://doi.org/10.3390/cells11233912
Levin D, Osman MS, Durand C, Kim H, Hemmati I, Jamani K, Howlett JG, Johannson KA, Weatherald J, Woo M, et al. Hematopoietic Cell Transplantation for Systemic Sclerosis—A Review. Cells. 2022; 11(23):3912. https://doi.org/10.3390/cells11233912
Chicago/Turabian StyleLevin, Daniel, Mohammed S. Osman, Caylib Durand, Hyein Kim, Iman Hemmati, Kareem Jamani, Jonathan G. Howlett, Kerri A. Johannson, Jason Weatherald, Matthew Woo, and et al. 2022. "Hematopoietic Cell Transplantation for Systemic Sclerosis—A Review" Cells 11, no. 23: 3912. https://doi.org/10.3390/cells11233912
APA StyleLevin, D., Osman, M. S., Durand, C., Kim, H., Hemmati, I., Jamani, K., Howlett, J. G., Johannson, K. A., Weatherald, J., Woo, M., Lee, J., & Storek, J. (2022). Hematopoietic Cell Transplantation for Systemic Sclerosis—A Review. Cells, 11(23), 3912. https://doi.org/10.3390/cells11233912






