Endothelial Glycocalyx Morphology in Different Flow Regions of the Aqueous Outflow Pathway of Normal and Laser-Induced Glaucoma Monkey Eyes
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Intraocular Pressure
3.2. Outflow Facility
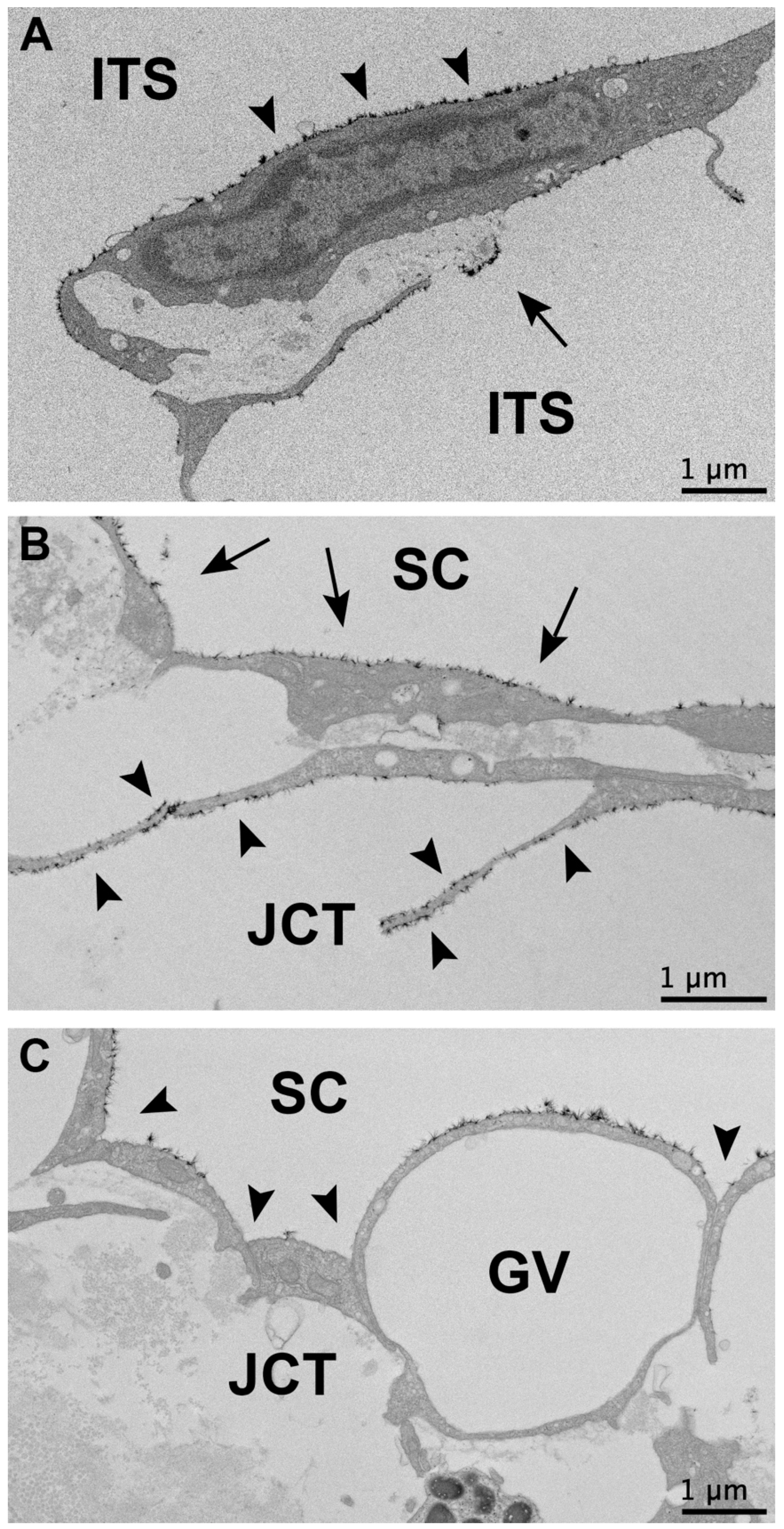
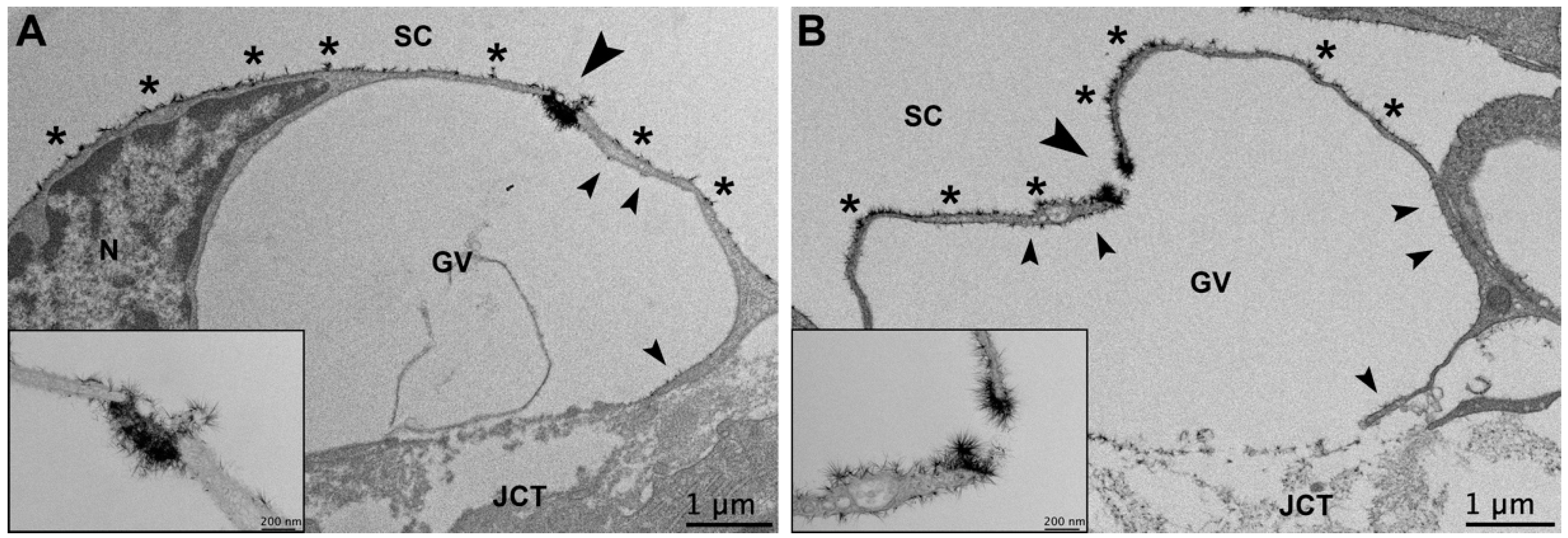
3.3. Glycocalyx Morphology
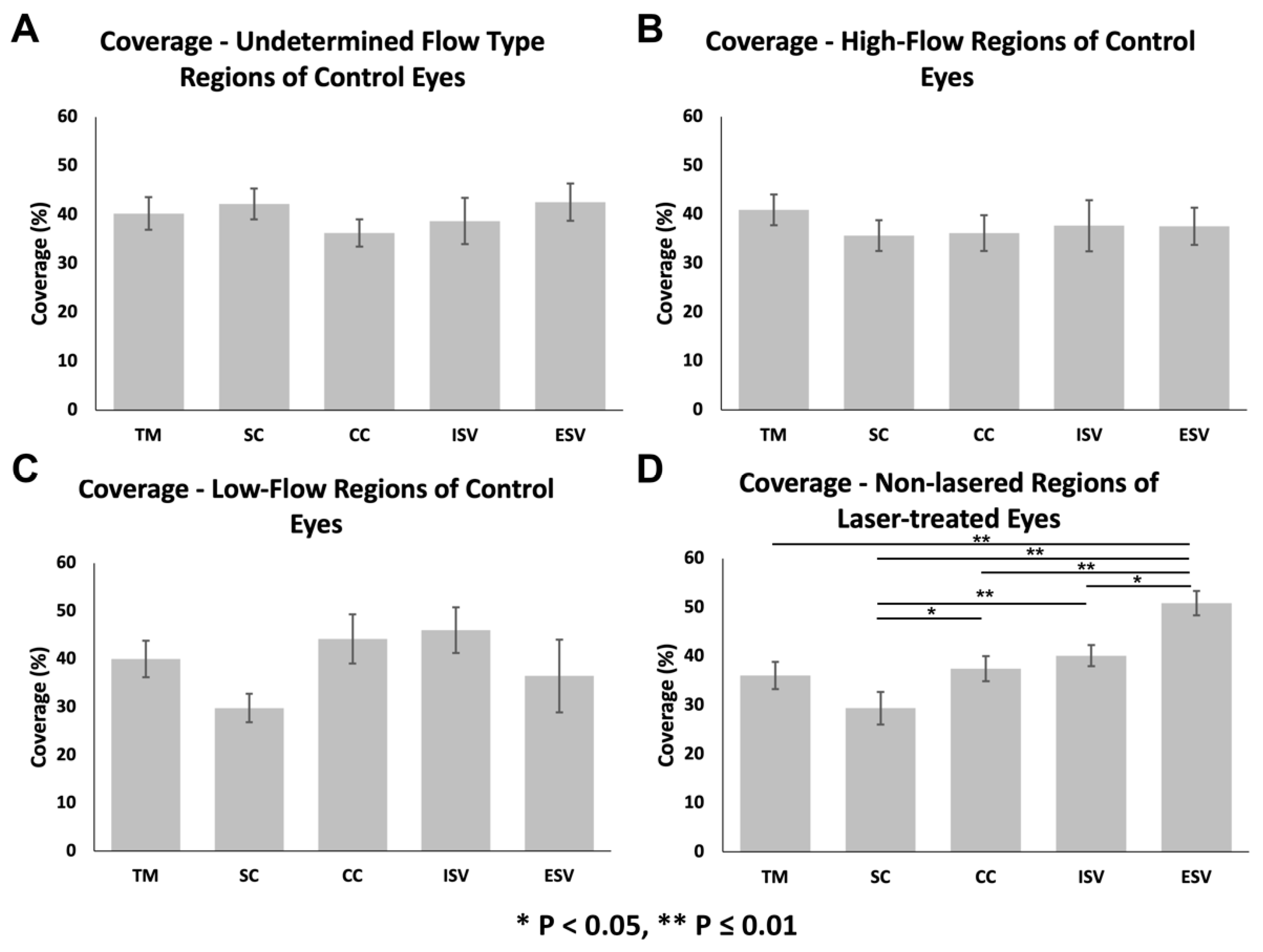
3.3.1. Glycocalyx Coverage
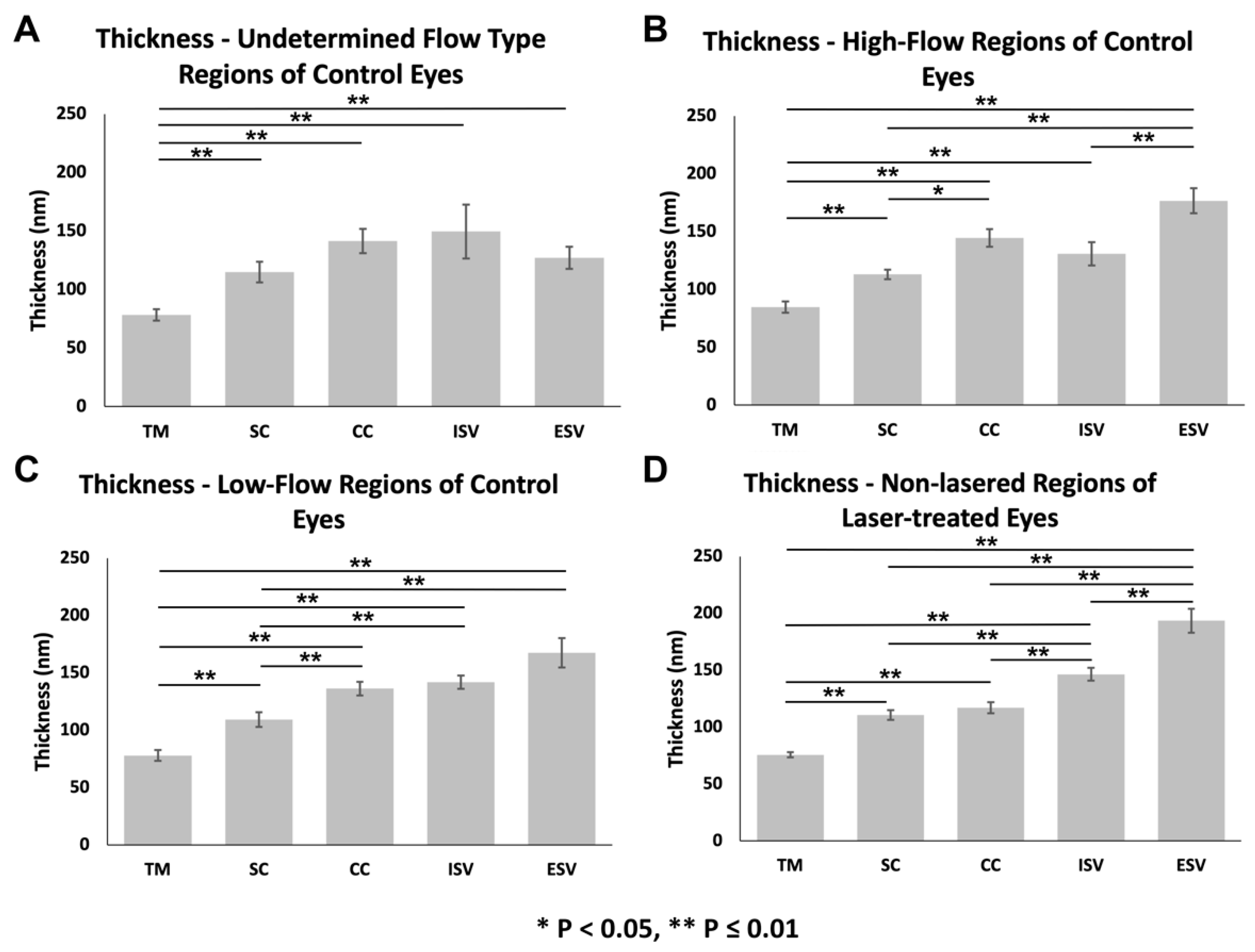
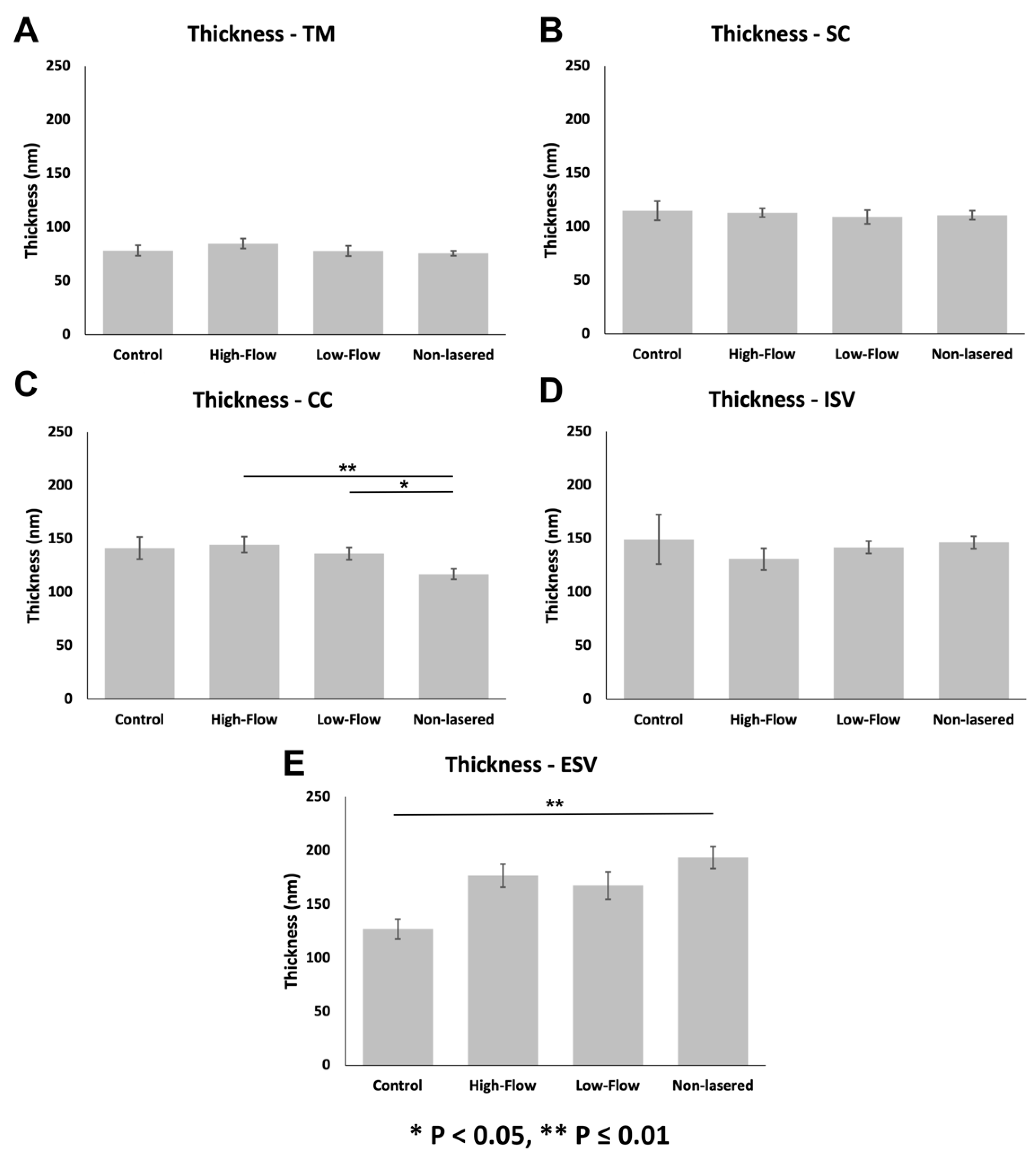
3.3.2. Glycocalyx Thickness
3.3.3. Glycocalyx Associated with Giant Vacuoles and I-Pores
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ederer, F.; Gaasterland, D.E.; Sullivan, E.K.; Beck, A.; E Prum, B.; Cyrlin, M.N.; Weiss, H. The advanced glaucoma intervention study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am. J. Ophthalmol. 2000, 130, 429–440. [Google Scholar] [CrossRef]
- Weinreb, R.N.; Leung, C.K.S.; Crowston, J.G.; Medeiros, F.A.; Friedman, D.S.; Wiggs, J.L.; Martin, K.R. Primary Open-Angle Glaucoma. Nat. Rev. Dis. Prim. 2016, 2, 16067. [Google Scholar] [CrossRef]
- Bill, A. Conventional and uveo-scleral drainage of aqueous humour in the cynomolgus monkey (Macaca irus) at normal and high intraocular pressures. Exp. Eye Res. 1966, 5, 45–54. [Google Scholar] [CrossRef]
- Ethier, C.R.; Read, A.T.; Chan, D. Biomechanics of Schlemm’s Canal Endothelial Cells: Influence on F-Actin Architecture. Biophys. J. 2004, 87, 2828–2837. [Google Scholar] [CrossRef]
- Stamer, W.D.; Lei, Y.; Boussommier-Calleja, A.; Overby, D.R.; Ethier, C.R. eNOS, a Pressure-Dependent Regulator of Intraocular Pressure. Investig. Opthalmol. Vis. Sci. 2011, 52, 9438–9444. [Google Scholar] [CrossRef]
- McDonnell, F.; Dismuke, W.M.; Overby, D.; Stamer, W.D. Pharmacological regulation of outflow resistance distal to Schlemm’s canal. Am. J. Physiol. Cell Physiol. 2018, 315, C44–C51. [Google Scholar] [CrossRef]
- Dewey, C.F. Effects of Fluid Flow on Living Vascular Cells. J. Biomech. Eng. 1984, 106, 31–35. [Google Scholar] [CrossRef]
- Yao, Y.; Rabodzey, A.; Dewey, J.C.F. Glycocalyx modulates the motility and proliferative response of vascular endothelium to fluid shear stress. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H1023–H1030. [Google Scholar] [CrossRef]
- Pries, A.R.; Secomb, T.W.; Gaehtgens, P. The endothelial surface layer. Pflug. Arch. 2000, 440, 653–666. [Google Scholar] [CrossRef]
- Weinbaum, S.; Tarbell, J.M.; Damiano, E.R. The Structure and Function of the Endothelial Glycocalyx Layer. Annu. Rev. Biomed. Eng. 2007, 9, 121–167. [Google Scholar] [CrossRef]
- Florian, J.A.; Kosky, J.R.; Ainslie, K.; Pang, Z.; Dull, R.O.; Tarbell, J.M. Heparan Sulfate Proteoglycan Is a Mechanosensor on Endothelial Cells. Circ. Res. 2003, 93, e136–e142. [Google Scholar] [CrossRef]
- Mochizuki, S.; Vink, H.; Hiramatsu, O.; Kajita, T.; Shigeto, F.; Spaan, J.A.E.; Kajiya, F. Role of hyaluronic acid glycosaminoglycans in shear-induced endothelium-derived nitric oxide release. Am. J. Physiol. Circ. Heart Physiol. 2003, 285, H722–H726. [Google Scholar] [CrossRef]
- Yang, C.-Y.C.; Huynh, T.; Johnson, M.; Gong, H. Endothelial glycocalyx layer in the aqueous outflow pathway of bovine and human eyes. Exp. Eye Res. 2014, 128, 27–33. [Google Scholar] [CrossRef]
- Cha, E.D.; Xu, J.; Gong, L.; Gong, H. Variations in active outflow along the trabecular outflow pathway. Exp. Eye Res. 2016, 146, 354–360. [Google Scholar] [CrossRef]
- Hann, C.R.; Bahler, C.K.; Johnson, D.H. Cationic Ferritin and Segmental Flow through the Trabecular Meshwork. Investig. Opthalmol. Vis. Sci. 2005, 46, 1–7. [Google Scholar] [CrossRef]
- Ren, R.; Li, G.; Le, T.D.; Kopczynski, C.; Stamer, W.D.; Gong, H. Netarsudil Increases Outflow Facility in Human Eyes Through Multiple Mechanisms. Investig. Opthalmol. Vis. Sci. 2016, 57, 6197–6209. [Google Scholar] [CrossRef]
- Yang, C.-Y.C.; Liu, Y.; Lu, Z.; Ren, R.; Gong, H. Effects of Y27632 on Aqueous Humor Outflow Facility with Changes in Hydrodynamic Pattern and Morphology in Human Eyes. Investig. Opthalmol. Vis. Sci. 2013, 54, 5859–5870. [Google Scholar] [CrossRef]
- Lu, Z.; Zhang, Y.; Freddo, T.F.; Gong, H. Similar hydrodynamic and morphological changes in the aqueous humor outflow pathway after washout and Y27632 treatment in monkey eyes. Exp. Eye Res. 2011, 93, 397–404. [Google Scholar] [CrossRef]
- Sabanay, I.; Gabelt, B.T.; Tian, B.; Kaufman, P.L.; Geiger, B. H-7 effects on the structure and fluid conductance of monkey trabecular meshwork. Arch. Ophthalmol. 2000, 118, 955–962. [Google Scholar]
- Gouverneur, M.; Berg, B.; Nieuwdorp, M.; Stroes, E.; Vink, H. Vasculoprotective properties of the endothelial glycocalyx: Effects of fluid shear stress. J. Intern. Med. 2006, 259, 393–400. [Google Scholar] [CrossRef]
- Arisaka, T.; Mitsumata, M.; Kawasumi, M.; Tohjima, T.; Hirose, S.; Yoshida, Y. Effects of Shear Stress on Glycosaminoglycan Synthesis in Vascular Endothelial Cellsa. Ann. New York Acad. Sci. 1995, 748, 543–554. [Google Scholar] [CrossRef]
- Pries, A.R.; Secomb, T.W.; Gessner, T.; Sperandio, M.B.; Gross, J.F.; Gaehtgens, P. Resistance to blood flow in microvessels in vivo. Circ. Res. 1994, 75, 904–915. [Google Scholar] [CrossRef]
- Gaasterland, D.; Kupfer, C. Experimental glaucoma in the rhesus monkey. Investig. Ophthalmol. 1974, 13, 455–457. [Google Scholar]
- Pederson, J.E.; Gaasterland, D.E. Laser-Induced Primate Glaucoma. I. Progression of Cupping. Arch. Ophthalmol. 1984, 102, 1689–1692. [Google Scholar] [CrossRef]
- Radius, R.L.; Pederson, J.E. Laser-Induced Primate Glaucoma. II. Histopathology. Arch. Ophthalmol. 1984, 102, 1693–1698. [Google Scholar] [CrossRef]
- Lee, P.-Y.; Podos, S.M.; Serle, J.B.; Camras, C.B.; Severin, C.H. Intraocular Pressure Effects of Multiple Doses of Drugs Applied to Glaucomatous Monkey Eyes. Arch. Ophthalmol. 1987, 105, 249–252. [Google Scholar] [CrossRef]
- Melamed, S.; Epstein, D.L. Alterations of aqueous humour outflow following argon laser trabeculoplasty in monkeys. Br. J. Ophthalmol. 1987, 71, 776–781. [Google Scholar] [CrossRef]
- Zhang, Y.; Toris, C.B.; Liu, Y.; Ye, W.; Gong, H. Morphological and hydrodynamic correlates in monkey eyes with laser induced glaucoma. Exp. Eye Res. 2009, 89, 748–756. [Google Scholar] [CrossRef]
- de Kater, A.W.; Melamed, S.; Epstein, D.L. Patterns of Aqueous Humor Outflow in Glaucomatous and Nonglaucomatous Human Eyes. A Tracer Study Using Cationized Ferritin. Arch. Ophthalmol. 1989, 107, 572–576. [Google Scholar] [CrossRef]
- Sosnowik, S.; Swain, D.L.; Fan, S.; Toris, C.B.; Gong, H. Morphological changes to Schlemm’s canal and the distal aqueous outflow pathway in monkey eyes with laser-induced ocular hypertension. Exp. Eye Res. 2022, 219, 109030. [Google Scholar] [CrossRef]
- Dvorak-Theobald, G.; Kirk, H.Q. Aqueous pathways in some cases of glaucoma. Trans. Am. Ophthalmol. Soc. 1955, 53, 301–319. [Google Scholar]
- Bhatt, K.; Gong, H.; Freddo, T.F. Freeze-fracture studies of interendothelial junctions in the angle of the human eye. Investig. Ophthalmol. Vis. Sci. 1995, 36, 1379–1389. [Google Scholar]
- Raviola, G.; Raviola, E. Paracellular route of aqueous outflow in the trabecular meshwork and canal of Schlemm. A freeze-fracture study of the endothelial junctions in the sclerocorneal angel of the macaque monkey eye. Investig. Ophthalmol. Vis. Sci. 1981, 21, 52–72. [Google Scholar]
- Ethier, C.R.; Coloma, F.M.; Sit, A.J.; Johnson, M. Two pore types in the inner-wall endothelium of Schlemm’s canal. Investig. Ophthalmol. Vis. Sci. 1998, 39, 2041–2048. [Google Scholar]
- Inomata, H.; Bill, A.; Smelser, G.K. Aqueous Humor Pathways Through the Trabecular Meshwork and Into Schlemm’S Canal in the Cynomolgus Monkey (Macaca Irus). An Electron Microscopic Study. Am. J. Ophthalmol. 1972, 73, 760–789. [Google Scholar] [CrossRef]
- Tripathi, R.C. Aqueous outflow pathway in normal and glaucomatous eyes. Br. J. Ophthalmol. 1972, 56, 157–174. [Google Scholar] [CrossRef][Green Version]
- Swain, D.L.; Le, T.D.; Yasmin, S.; Fernandes, B.; Lamaj, G.; Dasgupta, I.; Gao, Y.; Gong, H. Morphological factors associated with giant vacuoles with I-pores in Schlemm’s canal endothelial cells of human eyes: A serial block-face scanning electron microscopy study. Exp. Eye Res. 2021, 205, 108488. [Google Scholar] [CrossRef]
- Grierson, I.; Lee, W.R. Proceedings: Pressure Effects on the Outflow Apparatus: Analysis by Transmission Electron Microscopy. Exp. Eye Res. 1975, 20, 181. [Google Scholar] [CrossRef]
- Johnstone, M.A.; Grant, W.M. Pressure-Dependent Changes in Structures of the Aqueous Outflow System of Human and Monkey Eyes. Am. J. Ophthalmol. 1973, 75, 365–383. [Google Scholar] [CrossRef]
- Johnson, M.; Shapiro, A.; Ethier, C.R.; Kamm, R.D. Modulation of Outflow Resistance by the Pores of the Inner Wall Endothelium. Invest. Ophthalmol. Vis. Sci. 1992, 33, 1670–1675. [Google Scholar]
- Berg, B.M.V.D.; Spaan, J.A.E.; Rolf, T.M.; Vink, H. Atherogenic region and diet diminish glycocalyx dimension and increase intima-to-media ratios at murine carotid artery bifurcation. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H915–H920. [Google Scholar] [CrossRef] [PubMed]
- Gulan, U.; Lüthi, B.; Holzner, M.; Liberzon, A.; Tsinober, A.; Kinzelbach, W.; Liberzon, A. An in vitro investigation of the influence of stenosis severity on the flow in the ascending aorta. Med. Eng. Phys. 2014, 36, 1147–1155. [Google Scholar] [CrossRef]
- Johnson, M. ‘What controls aqueous humour outflow resistance?’. Exp. Eye Res. 2006, 82, 545–557. [Google Scholar] [CrossRef]
- Helps, E.P.W.; McDonald, D.A. Observations on laminar flow in veins. J. Physiol. 1954, 124, 631–639. [Google Scholar] [CrossRef]
- Adamson, R.H.; Clough, G. Plasma proteins modify the endothelial cell glycocalyx of frog mesenteric microvessels. J. Physiol. 1992, 445, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Rostgaard, J.; Qvortrup, K. Electron Microscopic Demonstrations of Filamentous Molecular Sieve Plugs in Capillary Fenestrae. Microvasc. Res. 1997, 53, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tarbell, J.M.; Cancel, L.M. The glycocalyx and its significance in human medicine. J. Intern. Med. 2016, 280, 97–113. [Google Scholar] [CrossRef]
- Henry, C.B.S.; Duling, B.R. TNF-α increases entry of macromolecules into luminal endothelial cell glycocalyx. Am. J. Physiol. Circ. Physiol. 2000, 279, H2815–H2823. [Google Scholar] [CrossRef]
- Ramnath, R.; Foster, R.R.; Qiu, Y.; Cope, G.; Butler, M.J.; Salmon, A.H.; Mathieson, P.W.; Coward, R.J.; Welsh, G.I.; Satchell, S.C. Matrix metalloproteinase 9-mediated shedding of syndecan 4 in response to tumor necrosis factor α: A contributor to endothelial cell glycocalyx dysfunction. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2014, 28, 4686–4699. [Google Scholar] [CrossRef]
- Schmidt, E.P.; Yang, Y.; Janssen, W.J.; Gandjeva, A.; Perez, M.J.; Barthel, L.; Zemans, R.L.; Bowman, J.C.; Koyanagi, D.E.; Yunt, Z.X.; et al. The pulmonary endothelial glycocalyx regulates neutrophil adhesion and lung injury during experimental sepsis. Nat. Med. 2012, 18, 1217–1223. [Google Scholar] [CrossRef]
- Subramanian, S.V.; Fitzgerald, M.L.; Bernfield, M. Regulated Shedding of Syndecan-1 and -4 Ectodomains by Thrombin and Growth Factor Receptor Activation. J. Biol. Chem. 1997, 272, 14713–14720. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Adamson, R.H.; Curry, F.-R.E.; Tarbell, J.M. Sphingosine-1-phosphate protects endothelial glycocalyx by inhibiting syndecan-1 shedding. Am. J. Physiol. Heart Circ. Physiol. 2014, 306, H363–H372. [Google Scholar] [CrossRef] [PubMed]
- Balaiya, S.; Edwards, J.; Tillis, T.; Khetpal, V.; Chalam, K. Tumor necrosis factor-alpha (TNF-α) levels in aqueous humor of primary open angle glaucoma. Clin. Ophthalmol. 2011, 5, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Baudouin, C.; Kolko, M.; Melik-Parsadaniantz, S.; Messmer, E.M. Inflammation in Glaucoma: From the back to the front of the eye, and beyond. Prog. Retin. Eye Res. 2020, 83, 100916. [Google Scholar] [CrossRef]
- Burgos-Blasco, B.; Vidal-Villegas, B.; Saenz-Frances, F.; Morales-Fernandez, L.; Perucho-Gonzalez, L.; Feijoo, J.G.; Martinez-De-La-Casa, J.M. Tear and aqueous humour cytokine profile in primary open-angle glaucoma. Acta Ophthalmol. 2020, 98, e768–e772. [Google Scholar] [CrossRef]
- Markiewicz, L.; Pytel, D.; Mucha, B.; Szymanek, K.; Szaflik, J.; Szaflik, J.P.; Majsterek, I. Altered Expression Levels of MMP1, MMP9, MMP12, TIMP1, and IL-1βas a Risk Factor for the Elevated IOP and Optic Nerve Head Damage in the Primary Open-Angle Glaucoma Patients. BioMed Res. Int. 2015, 2015, 812503. [Google Scholar] [CrossRef]
- Nga, A.D.; Yap, S.-L.; Samsudin, A.; Abdul-Rahman, P.S.; Hashim, O.H.; Mimiwati, Z. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in the aqueous humour of patients with primary angle closure glaucoma—A quantitative study. BMC Ophthalmol. 2014, 14, 33. [Google Scholar] [CrossRef]
- Dismuke, W.M.; Mbadugha, C.C.; Ellis, D.Z. NO-induced regulation of human trabecular meshwork cell volume and aqueous humor outflow facility involve the BKCa ion channel. Am. J. Physiol. Cell Physiol. 2008, 294, C1378–C1386. [Google Scholar] [CrossRef]
- Ellis, D.Z.; Sharif, N.A.; Dismuke, W.M. Endogenous Regulation of Human Schlemm’s Canal Cell Volume by Nitric Oxide Signaling. Investig. Opthalmology Vis. Sci. 2010, 51, 5817–5824. [Google Scholar] [CrossRef]
- Wiederholt, M.; Sturm, A.; Lepple-Wienhues, A. Relaxation of trabecular meshwork and ciliary muscle by release of nitric oxide. Investig. Ophthalmol. Vis. Sci. 1994, 35, 2515–2520. [Google Scholar]



| Control | Laser-Treated | ||||
|---|---|---|---|---|---|
| Monkey | Intermediate IOP (mmHg) | Final IOP (mmHg) | Intermediate IOP (mmHg) | Final IOP (mmHg) | Age at Final IOP (Years) |
| 1 | N/A | 22.0 | N/A | 20.0 | N/A |
| 2 | 26.5 | 22.0 | 35.8 | 31.0 | 15 |
| 3 | 23.7 | 29.9 | 30.0 | 31.5 | 17 |
| 4 | 31.5 | 19.6 | 64.8 | 29.8 | 18 |
| 5 | 22.0 | 27.5 | 28.4 | 31.3 | 18 |
| 6/OS | 28.9 | 25.7 | 15 | ||
| 6/OD | 34.5 | 27.5 | 15 | ||
| 7 | 34.6 | 19.3 | 18 | ||
| 8 | N/A | 24.3 | 19 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sosnowik, S.; Swain, D.L.; Liu, N.; Fan, S.; Toris, C.B.; Gong, H. Endothelial Glycocalyx Morphology in Different Flow Regions of the Aqueous Outflow Pathway of Normal and Laser-Induced Glaucoma Monkey Eyes. Cells 2022, 11, 2452. https://doi.org/10.3390/cells11152452
Sosnowik S, Swain DL, Liu N, Fan S, Toris CB, Gong H. Endothelial Glycocalyx Morphology in Different Flow Regions of the Aqueous Outflow Pathway of Normal and Laser-Induced Glaucoma Monkey Eyes. Cells. 2022; 11(15):2452. https://doi.org/10.3390/cells11152452
Chicago/Turabian StyleSosnowik, Shayna, David L. Swain, Neil Liu, Shan Fan, Carol B. Toris, and Haiyan Gong. 2022. "Endothelial Glycocalyx Morphology in Different Flow Regions of the Aqueous Outflow Pathway of Normal and Laser-Induced Glaucoma Monkey Eyes" Cells 11, no. 15: 2452. https://doi.org/10.3390/cells11152452
APA StyleSosnowik, S., Swain, D. L., Liu, N., Fan, S., Toris, C. B., & Gong, H. (2022). Endothelial Glycocalyx Morphology in Different Flow Regions of the Aqueous Outflow Pathway of Normal and Laser-Induced Glaucoma Monkey Eyes. Cells, 11(15), 2452. https://doi.org/10.3390/cells11152452







