Cell and Gene Therapy Approaches for Cardiac Vascularization
Abstract
:1. Introduction
2. The Microenvironmental Regulation of Angiogenesis
3. Revascularization Strategies for Ischemic Myocardium
3.1. Cell Therapy
3.2. Gene Therapy
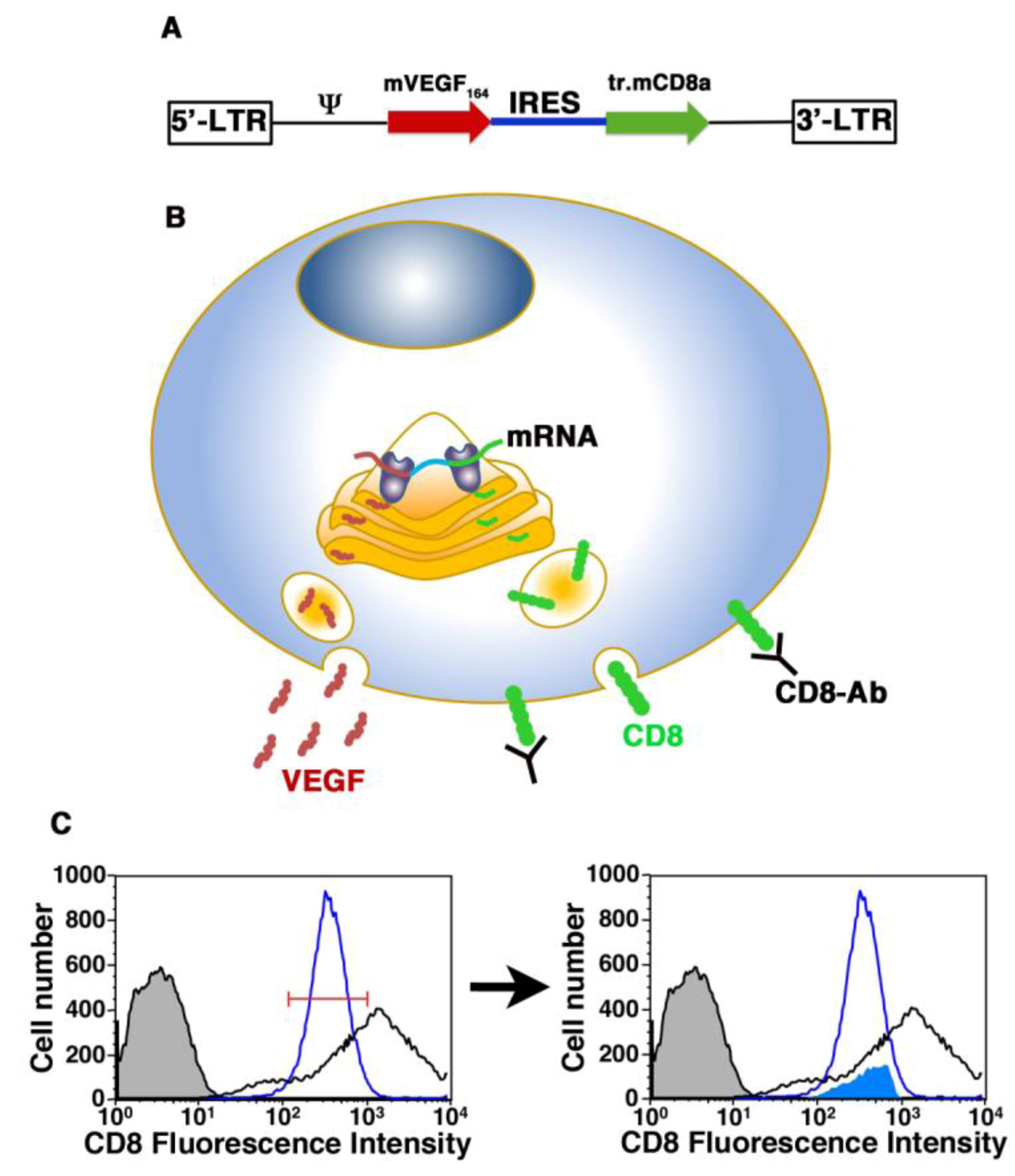
3.3. Cell-based Gene Therapy
4. Vascularization of Engineered Cardiac Tissue
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Benjamin, E.J.; Berry, J.D.; Borden, W.B.; Bravata, D.M.; Dai, S.; Ford, E.S.; Fox, C.S.; et al. Heart disease and stroke statistics—2012 update: A report from the American Heart Association. Circulation 2012, 125, e2–e220. [Google Scholar]
- Pries, A.R.; Höpfner, M.; le Noble, F.; Dewhirst, M.W.; Secomb, T.W. The shunt problem: control of functional shunting in normal and tumour vasculature. Nat. Rev. Cancer 2010, 10, 587–593. [Google Scholar] [CrossRef]
- Rissanen, T.T.; Korpisalo, P.; Markkanen, J.E.; Liimatainen, T.; Ordén, M.R.; Kholová, I.; de Goede, A.; Heikura, T.; Gröhn, O.H.; Ylä-Herttuala, S. Blood flow remodels growing vasculature during vascular endothelial growth factor gene therapy and determines between capillary arterialization and sprouting angiogenesis. Circulation 2005, 112, 3937–3946. [Google Scholar] [CrossRef]
- Giacca, M.; Zacchigna, S. VEGF gene therapy: Therapeutic angiogenesis in the clinic and beyond. Gene Ther. 2012, 19, 622–629. [Google Scholar] [CrossRef]
- Ferrara, N.; Carver-Moore, K.; Chen, H.; Dowd, M.; Lu, L.; O'Shea, K.S.; Powell-Braxton, L.; Hillan, K.J.; Moore, M.W. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 1996, 380, 439–442. [Google Scholar] [CrossRef]
- Haigh, J.J.; Gerber, H.P.; Ferrara, N.; Wagner, E.F. Conditional inactivation of VEGF-A in areas of collagen2a1 expression results in embryonic lethality in the heterozygous state. Development 2000, 127, 1445–1453. [Google Scholar]
- Mitsos, S.; Katsanos, K.; Koletsis, E.; Kagadis, G.C.; Anastasiou, N.; Diamantopoulos, A.; Karnabatidis, D.; Dougenis, D. Therapeutic angiogenesis for myocardial ischemia revisited: basic biological concepts and focus on latest clinical trials. Angiogenesis. 2012, 15, 1–22. [Google Scholar] [CrossRef]
- Gerhardt, H.; Golding, M.; Fruttiger, M.; Ruhrberg, C.; Lundkvist, A.; Abramsson, A.; Jeltsch, M.; Mitchell, C.; Alitalo, K.; Shima, D.; et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell. Biol. 2003, 161, 1163–1177. [Google Scholar] [CrossRef]
- Hellstrom, M.; Phng, L.K.; Hofmann, J.J.; Wallgard, E.; Coultas, L.; Lindblom, P.; Alva, J.; Nilsson, A.K.; Karlsson, L.; Gaiano, N.; et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 2007, 445, 776–780. [Google Scholar] [CrossRef]
- Park, J.E.; Keller, G.A.; Ferrara, N. The vascular endothelial growth factor (VEGF) isoforms: differential deposition into the subepithelial extracellular matrix and bioactivity of extracellular matrix-bound VEGF. Mol. Biol. Cell. 1993, 4, 1317–1326. [Google Scholar]
- Gaengel, K.; Genove, G.; Armulik, A.; Betsholtz, C. Endothelial-mural cell signaling in vascular development and angiogenesis. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 630–638. [Google Scholar] [CrossRef]
- Reginato, S.; Gianni-Barrera, R.; Banfi, A. Taming of the wild vessel: Promoting vessel stabilization for safe therapeutic angiogenesis. Biochem. Soc. Trans. 2011, 39, 1654–1658. [Google Scholar] [CrossRef]
- Zacchigna, S.; Pattarini, L.; Zentilin, L.; Moimas, S.; Carrer, A.; Sinigaglia, M.; Arsic, N.; Tafuro, S.; Sinagra, G.; Giacca, M. Bone marrow cells recruited through the neuropilin-1 receptor promote arterial formation at the sites of adult neoangiogenesis in mice. J. Clin. Invest. 2008, 118, 2062–2075. [Google Scholar]
- Ozawa, C.R.; Banfi, A.; Glazer, N.L.; Thurston, G.; Springer, M.L.; Kraft, P.E.; McDonald, D.M.; Blau, H.M. Microenvironmental VEGF concentration, not total dose, determines a threshold between normal and aberrant angiogenesis. J. Clin. Invest. 2004, 113, 516–527. [Google Scholar]
- Pettersson, A.; Nagy, J.A.; Brown, L.F.; Sundberg, C.; Morgan, E.; Jungles, S.; Carter, R.; Krieger, J.E.; Manseau, E.J.; Harvey, V.S.; et al. Heterogeneity of the angiogenic response induced in different normal adult tissues by vascular permeability factor/vascular endothelial growth factor. Lab Invest. 2000, 80, 99–115. [Google Scholar] [CrossRef]
- Springer, M.L.; Chen, A.S.; Kraft, P.E.; Bednarski, M.; Blau, H.M. VEGF gene delivery to muscle: potential role for vasculogenesis in adults. Mol. Cell. 1998, 2, 549–558. [Google Scholar] [CrossRef]
- Dor, Y.; Djonov, V.; Abramovitch, R.; Itin, A.; Fishman, G.I.; Carmeliet, P.; Goelman, G.; Keshet, E. Conditional switching of VEGF provides new insights into adult neovascularization and pro-angiogenic therapy. EMBO J. 2002, 21, 1939–1947. [Google Scholar] [CrossRef]
- Carmeliet, P. VEGF gene therapy: stimulating angiogenesis or angioma-genesis? Nat. Med. 2000, 6, 1102–1103. [Google Scholar] [CrossRef]
- Lee, R.J.; Springer, M.L.; Blanco-Bose, W.E.; Shaw, R.; Ursell, P.C.; Blau, H.M. VEGF gene delivery to myocardium: deleterious effects of unregulated expression. Circulation 2000, 102, 898–901. [Google Scholar] [CrossRef]
- Schwarz, E.R.; Speakman, M.T.; Patterson, M.; Hale, S.S.; Isner, J.M.; Kedes, L.H.; Kloner, R.A. Evaluation of the effects of intramyocardial injection of DNA expressing vascular endothelial growth factor (VEGF) in a myocardial infarction model in the rat--angiogenesis and angioma formation. J. Am. Coll. Cardiol. 2000, 35, 1323–1330. [Google Scholar] [CrossRef]
- Banfi, A.; von, D.G.; Blau, H.M. Critical role of microenvironmental factors in angiogenesis. Curr. Atheroscler. Rep. 2005, 7, 227–234. [Google Scholar] [CrossRef]
- Karvinen, H.; Yla-Herttuala, S. New aspects in vascular gene therapy. Curr. Opin. Pharmacol. 2010, 10, 208–211. [Google Scholar] [CrossRef]
- Yla-Herttuala, S.; Markkanen, J.E.; Rissanen, T.T. Gene therapy for ischemic cardiovascular diseases: Some lessons learned from the first clinical trials. Trends Cardiovasc. Med. 2004, 14, 295–300. [Google Scholar] [CrossRef]
- von Degenfeld, G.; Banfi, A.; Springer, M.L.; Wagner, R.A.; Jacobi, J.; Ozawa, C.R.; Merchant, M.J.; Cooke, J.P.; Blau, H.M. Microenvironmental VEGF distribution is critical for stable and functional vessel growth in ischemia. FASEB J. 2006, 20, 2657–2659. [Google Scholar] [CrossRef]
- Gianni-Barrera, R.; Trani, M.; Fontanellaz, C.; Heberer, M.; Djonov, V.; Hlushchuk, R.; Banfi, A. VEGF over-expression in skeletal muscle induces angiogenesis by intussusception rather than sprouting. Angiogenesis 2012. [Google Scholar]
- Tafuro, S.; Ayuso, E.; Zacchigna, S.; Zentilin, L.; Moimas, S.; Dore, F.; Giacca, M. Inducible adeno-associated virus vectors promote functional angiogenesis in adult organisms via regulated vascular endothelial growth factor expression. Cardiovasc. Res. 2009, 83, 663–671. [Google Scholar] [CrossRef]
- Ptaszek, L.M.; Mansour, M.; Ruskin, J.N.; Chien, K.R. Towards regenerative therapy for cardiac disease. Lancet 2012, 379, 933–942. [Google Scholar] [CrossRef]
- Cashman, T.J.; Gouon-Evans, V.; Costa, K.D. Mesenchymal Stem Cells for Cardiac Therapy: Practical Challenges and Potential Mechanisms. Stem Cell Rev. 2012. [Google Scholar]
- Botta, R.; Gao, E.; Stassi, G.; Bonci, D.; Pelosi, E.; Zwas, D.; Patti, M.; Colonna, L.; Baiocchi, M.; Coppola, S.; et al. Heart infarct in NOD-SCID mice: therapeutic vasculogenesis by transplantation of human CD34+ cells and low dose CD34+KDR+ cells. FASEB J. 2004, 18, 1392–1394. [Google Scholar]
- Kocher, A.A.; Schuster, M.D.; Szabolcs, M.J.; Takuma, S.; Burkhoff, D.; Wang, J.; Homma, S.; Edwards, N.M.; Itescu, S. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat. Med. 2001, 7, 430–436. [Google Scholar] [CrossRef]
- Bartunek, J.; Vanderheyden, M.; Vandekerckhove, B.; Mansour, S.; De Bruyne, B.; De Bondt, P.; Van Haute, I.; Lootens, N.; Heyndrickx, G.; Wijns, W. Intracoronary injection of CD133-positive enriched bone marrow progenitor cells promotes cardiac recovery after recent myocardial infarction: feasibility and safety. Circulation 2005, 112, I178–I183. [Google Scholar]
- Stamm, C.; Westphal, B.; Kleine, H.D.; Petzsch, M.; Kittner, C.; Klinge, H.; Schumichen, C.; Nienaber, C.A.; Freund, M.; Steinhoff, G. Autologous bone-marrow stem-cell transplantation for myocardial regeneration. Lancet 2003, 361, 45–46. [Google Scholar]
- Choi, Y.H.; Kurtz, A.; Stamm, C. Mesenchymal stem cells for cardiac cell therapy. Hum. Gene Ther. 2011, 22, 3–17. [Google Scholar]
- Asahara, T.; Kawamoto, A.; Masuda, H. Concise review: Circulating endothelial progenitor cells for vascular medicine. Stem Cells 2011, 29, 1650–1655. [Google Scholar] [CrossRef]
- da Silva Meirelles, L.; Caplan, A.I.; Nardi, N.B. In search of the in vivo identity of mesenchymal stem cells. Stem Cells 2008, 26, 2287–2299. [Google Scholar] [CrossRef]
- Li, Z.; Guo, J.; Chang, Q.; Zhang, A. Paracrine role for mesenchymal stem cells in acute myocardial infarction. Biol. Pharm. Bull. 2009, 32, 1343–1346. [Google Scholar] [CrossRef]
- Poncelet, A.J.; Hiel, A.L.; Vercruysse, J.; Hermans, D.; Zech, F.; Gianello, P. Intracardiac allogeneic mesenchymal stem cell transplantation elicits neo-angiogenesis in a fully immunocompetent ischaemic swine model. Eur. J. Cardiothorac. Surg. 2010, 38, 781–787. [Google Scholar] [CrossRef]
- Rissanen, T.T.; Yla-Herttuala, S. Current status of cardiovascular gene therapy. Mol. Ther. 2007, 15, 1233–1247. [Google Scholar] [CrossRef]
- Suzuki, R.; Oda, Y.; Utoguchi, N.; Maruyama, K. Progress in the development of ultrasound-mediated gene delivery systems utilizing nano- and microbubbles. J. Control. Release 2011, 149, 36–41. [Google Scholar] [CrossRef]
- The Journal of Gene Medicine: Gene Therapy Clinical Trials Worldwide. Available online: http://www.abedia.com/wiley/vectors.php (accessed on 15 October 2012).
- Wu, Z.; Asokan, A.; Samulski, R.J. Adeno-associated virus serotypes: Vector toolkit for human gene therapy. Mol Ther 2006, 14, 316–327. [Google Scholar]
- Kay, M.A. State-of-the-art gene-based therapies: the road ahead. Nat. Rev. Genet. 2011, 12, 316–328. [Google Scholar] [CrossRef]
- Pacak, C.A.; Byrne, B.J. AAV vectors for cardiac gene transfer: experimental tools and clinical opportunities. Mol. Ther. 2011, 19, 1582–1590. [Google Scholar] [CrossRef]
- Korpisalo, P.; Yla-Herttuala, S. Stimulation of functional vessel growth by gene therapy. Integr. Biol. (Camb.) 2010, 2, 102–112. [Google Scholar] [CrossRef]
- Korpisalo, P.; Karvinen, H.; Rissanen, T.T.; Kilpijoki, J.; Marjomaki, V.; Baluk, P.; McDonald, D.M.; Cao, Y.; Eriksson, U.; Alitalo, K.; et al. Vascular endothelial growth factor-A and platelet-derived growth factor-B combination gene therapy prolongs angiogenic effects via recruitment of interstitial mononuclear cells and paracrine effects rather than improved pericyte coverage of angiogenic vessels. Circ. Res. 2008, 103, 1092–1099. [Google Scholar] [CrossRef]
- Kupatt, C.; Hinkel, R.; Pfosser, A.; El-Aouni, C.; Wuchrer, A.; Fritz, A.; Globisch, F.; Thormann, M.; Horstkotte, J.; Lebherz, C.; et al. Cotransfection of vascular endothelial growth factor-A and platelet-derived growth factor-B via recombinant adeno-associated virus resolves chronic ischemic malperfusion role of vessel maturation. J. Am. Coll. Cardiol. 2010, 56, 414–422. [Google Scholar]
- Banfi, A.; von Degenfeld, G.; Gianni-Barrera, R.; Reginato, S.; Merchant, M.J.; McDonald, D.M.; Blau, H.M. Therapeutic angiogenesis due to balanced single-vector delivery of VEGF and PDGF-BB. FASEB J. 2012, 26, 2486–2497. [Google Scholar]
- Li, X.; Tjwa, M.; Van Hove, I.; Enholm, B.; Neven, E.; Paavonen, K.; Jeltsch, M.; Juan, T.D.; Sievers, R.E.; Chorianopoulos, E.; et al. Reevaluation of the role of VEGF-B suggests a restricted role in the revascularization of the ischemic myocardium. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1614–1620. [Google Scholar] [CrossRef]
- Lahteenvuo, J.E.; Lahteenvuo, M.T.; Kivela, A.; Rosenlew, C.; Falkevall, A.; Klar, J.; Heikura, T.; Rissanen, T.T.; Vahakangas, E.; Korpisalo, P.; et al. Vascular endothelial growth factor-B induces myocardium-specific angiogenesis and arteriogenesis via vascular endothelial growth factor receptor-1- and neuropilin receptor-1-dependent mechanisms. Circulation 2009, 119, 845–856. [Google Scholar]
- Karpanen, T.; Bry, M.; Ollila, H.M.; Seppanen-Laakso, T.; Liimatta, E.; Leskinen, H.; Kivela, R.; Helkamaa, T.; Merentie, M.; Jeltsch, M.; et al. Overexpression of vascular endothelial growth factor-B in mouse heart alters cardiac lipid metabolism and induces myocardial hypertrophy. Circ. Res. 2008, 103, 1018–1026. [Google Scholar] [CrossRef]
- Zentilin, L.; Puligadda, U.; Lionetti, V.; Zacchigna, S.; Collesi, C.; Pattarini, L.; Ruozi, G.; Camporesi, S.; Sinagra, G.; Pepe, M.; et al. Cardiomyocyte VEGFR-1 activation by VEGF-B induces compensatory hypertrophy and preserves cardiac function after myocardial infarction. Faseb J. 2010, 24, 1467–1478. [Google Scholar] [CrossRef]
- Misteli, H.; Wolff, T.; Fuglistaler, P.; Gianni-Barrera, R.; Gurke, L.; Heberer, M.; Banfi, A. High-throughput flow cytometry purification of transduced progenitors expressing defined levels of vascular endothelial growth factor induces controlled angiogenesis in vivo. Stem Cells 2010, 28, 611–619. [Google Scholar]
- Wolff, T.; Mujagic, E.; Gianni-Barrera, R.; Fueglistaler, P.; Helmrich, U.; Misteli, H.; Gurke, L.; Heberer, M.; Banfi, A. FACS-purified myoblasts producing controlled VEGF levels induce safe and stable angiogenesis in chronic hind limb ischemia. J. Cell. Mol. Med. 2012, 16, 107–117. [Google Scholar] [CrossRef]
- Helmrich, U.; Marsano, A.; Melly, L.; Wolff, T.; Christ, L.; Heberer, M.; Scherberich, A.; Martin, I.; Banfi, A. Generation of human adult mesenchymal stromal/stem cells expressing defined xenogenic vascular endothelial growth factor levels by optimized transduction and flow cytometry purification. Tissue Eng. Part C. Methods 2012, 18, 283–292. [Google Scholar] [CrossRef]
- Melly, L.; Marsano, A.; Frobert, A.; Boccardo, S.; Helmrich, U.; Heberer, M.; Eckstein, F.S.; Carrel, T.P.; Giraud, M.; Tevaearai, H.T.; et al. Controlled angiogenesis in the heart by cell-based expression of specific VEGF levels. Hum. Gene Ther. Methods 2012, in press.. [Google Scholar]
- Iyer, R.K.; Chiu, L.L.; Reis, L.A.; Radisic, M. Engineered cardiac tissues. Curr. Opin. Biotechnol. 2011, 22, 706–714. [Google Scholar]
- Miyahara, Y.; Nagaya, N.; Kataoka, M.; Yanagawa, B.; Tanaka, K.; Hao, H.; Ishino, K.; Ishida, H.; Shimizu, T.; Kangawa, K.; et al. Monolayered mesenchymal stem cells repair scarred myocardium after myocardial infarction. Nat. Med. 2006, 12, 459–465. [Google Scholar] [CrossRef]
- Shimizu, T.; Sekine, H.; Yang, J.; Isoi, Y.; Yamato, M.; Kikuchi, A.; Kobayashi, E.; Okano, T. Polysurgery of cell sheet grafts overcomes diffusion limits to produce thick, vascularized myocardial tissues. FASEB J. 2006, 20, 708–710. [Google Scholar]
- Zhang, M.; Methot, D.; Poppa, V.; Fujio, Y.; Walsh, K.; Murry, C.E. Cardiomyocyte grafting for cardiac repair: Graft cell death and anti-death strategies. J. Mol. Cell. Cardiol. 2001, 33, 907–921. [Google Scholar] [CrossRef]
- Zimmermann, W.H.; Didie, M.; Doker, S.; Melnychenko, I.; Naito, H.; Rogge, C.; Tiburcy, M.; Eschenhagen, T. Heart muscle engineering: an update on cardiac muscle replacement therapy. Cardiovasc. Res. 2006, 71, 419–429. [Google Scholar] [CrossRef]
- Caspi, O.; Lesman, A.; Basevitch, Y.; Gepstein, A.; Arbel, G.; Habib, I.H.; Gepstein, L.; Levenberg, S. Tissue engineering of vascularized cardiac muscle from human embryonic stem cells. Circ. Res. 2007, 100, 263–272. [Google Scholar] [CrossRef]
- Laugwitz, K.L.; Moretti, A.; Lam, J.; Gruber, P.; Chen, Y.; Woodard, S.; Lin, L.Z.; Cai, C.L.; Lu, M.M.; Reth, M.; et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 2005, 433, 647–653. [Google Scholar]
- Vunjak-Novakovic, G.; Tandon, N.; Godier, A.; Maidhof, R.; Marsano, A.; Martens, T.P.; Radisic, M. Challenges in cardiac tissue engineering. Tissue Eng. Part B Rev. 2010, 16, 169–187. [Google Scholar] [CrossRef]
- Hamdi, H.; Planat-Benard, V.; Bel, A.; Puymirat, E.; Geha, R.; Pidial, L.; Nematalla, H.; Bellamy, V.; Bouaziz, P.; Peyrard, S.; et al. Epicardial adipose stem cell sheets results in greater post-infarction survival than intramyocardial injections. Cardiovasc. Res. 2011, 91, 483–491. [Google Scholar]
- Leor, J.; Aboulafia-Etzion, S.; Dar, A.; Shapiro, L.; Barbash, I.M.; Battler, A.; Granot, Y.; Cohen, S. Bioengineered cardiac grafts: A new approach to repair the infarcted myocardium? Circulation 2000, 102, III56–III61. [Google Scholar]
- Li, R.K.; Jia, Z.Q.; Weisel, R.D.; Mickle, D.A.; Choi, A.; Yau, T.M. Survival and function of bioengineered cardiac grafts. Circulation 1999, 100, II63–II69. [Google Scholar]
- Dvir, T.; Kedem, A.; Ruvinov, E.; Levy, O.; Freeman, I.; Landa, N.; Holbova, R.; Feinberg, M.S.; Dror, S.; Etzion, Y.; et al. Prevascularization of cardiac patch on the omentum improves its therapeutic outcome. Proc. Natl. Acad. Sci. USA 2009, 106, 14990–14995. [Google Scholar]
- Iyer, R.K.; Chiu, L.L.; Radisic, M. Microfabricated poly(ethylene glycol) templates enable rapid screening of triculture conditions for cardiac tissue engineering. J. Biomed. Mater. Res. Part A 2009, 89, 616–631. [Google Scholar]
- Iyer, R.K.; Chiu, L.L.; Vunjak-Novakovic, G.; Radisic, M. Biofabrication enables efficient interrogation and optimization of sequential culture of endothelial cells, fibroblasts and cardiomyocytes for formation of vascular cords in cardiac tissue engineering. Biofabrication 2012, 4, 035002. [Google Scholar] [CrossRef]
- Lesman, A.; Habib, M.; Caspi, O.; Gepstein, A.; Arbel, G.; Levenberg, S.; Gepstein, L. Transplantation of a tissue-engineered human vascularized cardiac muscle. Tissue Eng. Part A 2010, 16, 115–125. [Google Scholar]
- Madden, L.R.; Mortisen, D.J.; Sussman, E.M.; Dupras, S.K.; Fugate, J.A.; Cuy, J.L.; Hauch, K.D.; Laflamme, M.A.; Murry, C.E.; Ratner, B.D. Proangiogenic scaffolds as functional templates for cardiac tissue engineering. Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 15211–15216. [Google Scholar]
- Miyagi, Y.; Chiu, L.L.; Cimini, M.; Weisel, R.D.; Radisic, M.; Li, R.K. Biodegradable collagen patch with covalently immobilized VEGF for myocardial repair. Biomaterials 2011, 32, 1280–1290. [Google Scholar]
- Marsano, A.; Maidhof, R.; Luo, J.; Fujikara, K.; Konofagou, E.E.; Banfi, A.; Vunjak-Novakovic, G. The effect of controlled expression of VEGF by transduced myoblasts in a cardiac patch on vascularization in a mouse model of myocardial infarction. Biomaterials 2013, 34, 393–401. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Melly, L.; Boccardo, S.; Eckstein, F.; Banfi, A.; Marsano, A. Cell and Gene Therapy Approaches for Cardiac Vascularization. Cells 2012, 1, 961-975. https://doi.org/10.3390/cells1040961
Melly L, Boccardo S, Eckstein F, Banfi A, Marsano A. Cell and Gene Therapy Approaches for Cardiac Vascularization. Cells. 2012; 1(4):961-975. https://doi.org/10.3390/cells1040961
Chicago/Turabian StyleMelly, Ludovic, Stefano Boccardo, Friedrich Eckstein, Andrea Banfi, and Anna Marsano. 2012. "Cell and Gene Therapy Approaches for Cardiac Vascularization" Cells 1, no. 4: 961-975. https://doi.org/10.3390/cells1040961
APA StyleMelly, L., Boccardo, S., Eckstein, F., Banfi, A., & Marsano, A. (2012). Cell and Gene Therapy Approaches for Cardiac Vascularization. Cells, 1(4), 961-975. https://doi.org/10.3390/cells1040961



