Mesenchymal Stem or Stromal Cells from Amnion and Umbilical Cord Tissue and Their Potential for Clinical Applications
Abstract
:1. Introduction
2. Isolation, Expansion and Characterization of hAMSC
| basal | % FCS | further supplements | |
|---|---|---|---|
| Bilic [47,57] | DMEM:Ham'sF12 1:1 | 10 | 50 ng/mL EGF, 2.5 μg/mL insulin, 5 μg/mL transferrin, 0.1 ng/mL tri-iodothyronine |
| Bilic [2] | DMEM:Ham'sF12 1:1 | 10 | for some experiments 10 ng/mL EGF |
| Diaz-Prado [4] | DMEM | 20 | |
| In't Anker [8] | M199 | 10 | 20 μg/mL EGF, 8 U/mL heparin |
| Kang [10] | alphaMEM | 10 | |
| Kim [11] | DMEM | 10 | 3.7 mg/mL sodium bicarbonate |
| König [12] | DMEM | 15 | |
| Lisi [14] | DMEM | 10 | 10 ng/mL EGF, 55 μM b-ME |
| Paracchini [18] | DMEM | 10 | 1% sodium pyruvate, 1% NEAA, 55 μM b-ME, 10 ng/mL EGF |
| Stadler [21] | DMEM | 10 | 1% NEAA, 55 μM 2-mercaptoethanol, 1 mM sodium pyruvate |
| Sudo [56] | alphaMEM | 10 | without any further supplements or 10 ng/mL EGF or 10 ng/mL EGF + 105 U/mL LIF |
| Tamagawa [53] | alphaMEM | 10 | 10 ng/mL EGF, 10 ng/mL LIF |
| Whittle [54] | DMEM | 10 | |
| Zhao [25] | DMEM | 10 |
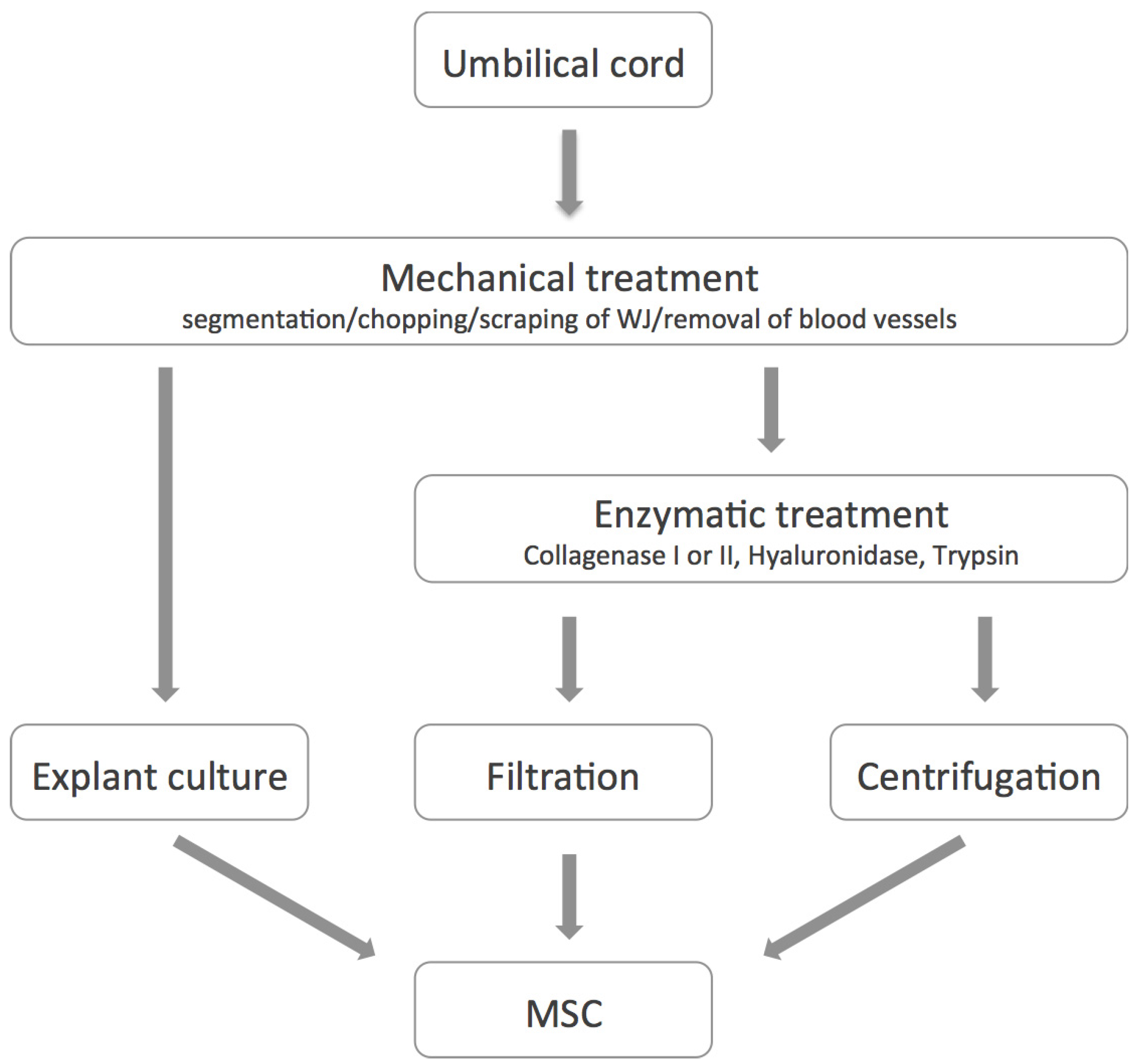
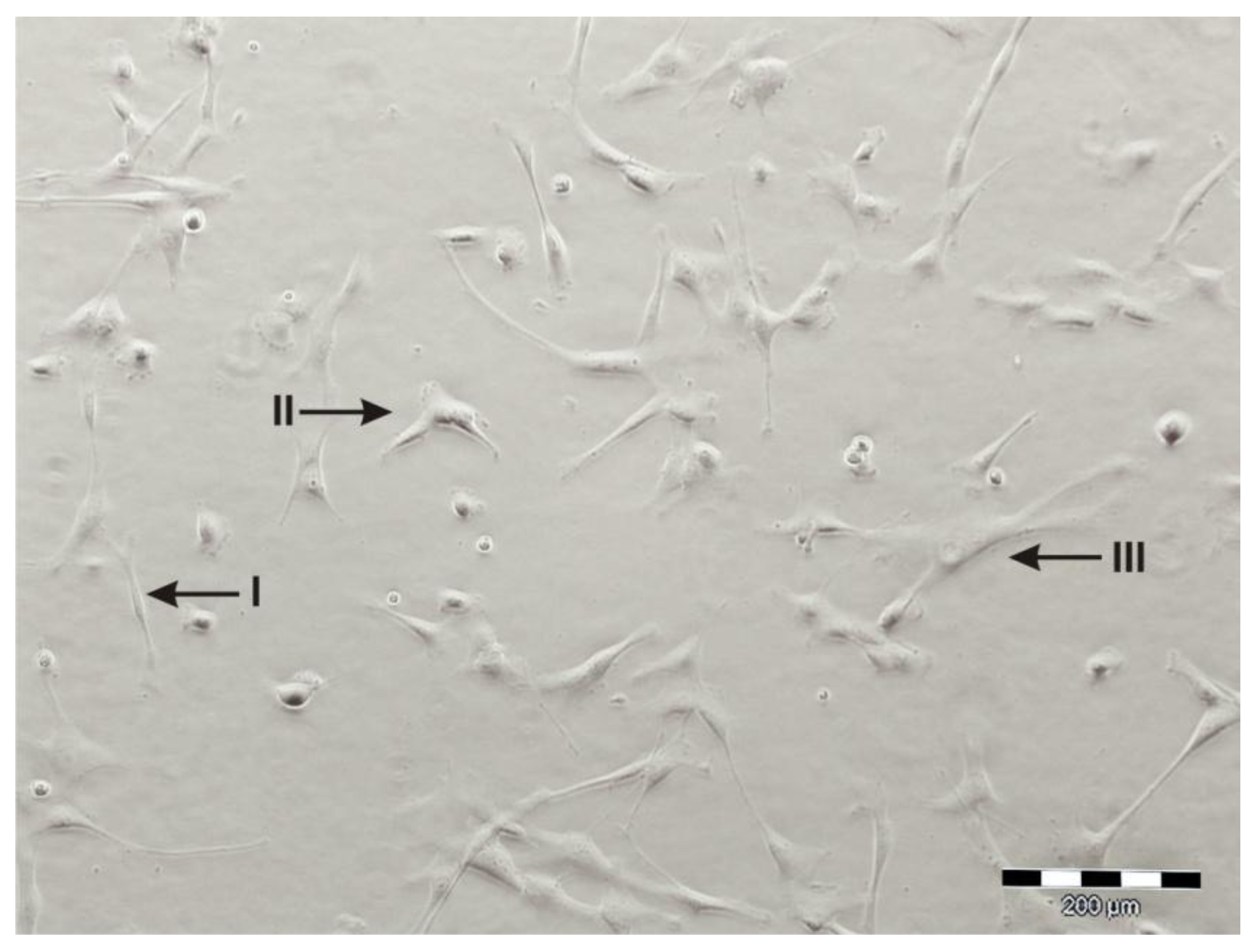
3. Isolation, Expansion and Characterization of UC-MSC
4. Differentiation Potential of hAMSC
| lineage | reference | media description | protocol |
|---|---|---|---|
| osteogenic | Pittenger [82] | DMEM-LG, 10% FCS, 10−7 M dexa, 50 μM AAP, 10 mM b-GP, 10 mM vit D3 | |
| In't Anker [8] | alphaMEM, 10% FCS, 10−7 M dexa, 50 μg/mL AAP, 5 mM b-GP | b-GP was added from day 7 on, 21 days | |
| Portmann-Lanz [50] | DMEM-HG, 10% FCS, 10 μmol/L dexa, 50 μg/mL AAP, 10 mol/L b-GP, 10 nmol/L vit D3 | 18–21 days | |
| Alviano [1] | DMEM, 10% FCS, 10−8 M dexa, 0.2 mM AAP, 10 mM b-GP | 21–28 days | |
| Sudo [56] | DMEM-HG, 10% FCS, 10−7 M dexa, 0.5 μM AAP, 10 mM b-GP | 28 days | |
| Chen [81] | DMEM, 10% FCS, 100nM dexa, 0.1 μM AAP, 10 mM b-GP, 5 × 10−5 M b-ME | induction on day 8 for 20 days | |
| Bilic [2], Diaz-Prado [4], Paracchini [18] | hMSC Mesenchymal Stem Cell Osteogenic Differentiation Medium (Lonza) | 14 (Bilic) or 21 days (Diaz-Prado, Paracchini) | |
| Stadler [21], Wolbank [24] | MesenCult Osteogenic Stimulatory Kit (OKit, Stemcell Technologies) | 21 (Wolbank) or 28 (Stadler) days | |
| Manochantr [57], Kang [10] | NH Osteodiff medium (Miltenyi) | 14 days (Kang) | |
| adipogenic | Pittenger [82] | medium 1: DMEM-HG, 10% FCS, 0.5 mM IBMX, 0.1 μM dexa, 10 μg/mL insulin, 100 μM IM, | medium 1: 48-72h, medium 2: 24h; 3 repeated cycles; |
| medium 2; DMEM-HG, 10% FCS, 10 μg/mL insulin | 28 days (Stadler) | ||
| In't Anker [8] | alphaMEM, 10% FCS, 0.5 mM IBMX, 10−7 M dexa, 1.60 μM insulin, 50 μM IM, 50 μg/mL AAP | 21 (In’t Anker) or 28 days (Stadler) | |
| Portmann-Lanz [50] | DMEM-HG, 10% FCS, 0.5 mmoL/L IBMX, 1 μmol/L dexa, 10 μmol/L insulin, 200 μmol/L IM | 18–21d (Portmann-Lanz, Wolbank) or 28 days (Stadler) | |
| Alviano [1] | DMEM, 10% FCS, 0.5 mM IBMX, 10−6 M dexa, 10 μg/mL insulin, 200 μM IM | 14–21 days | |
| Sudo [56] | medium 1: DMEM-HG, 10% FCS, 0.5 mM IBMX, 1 μM dexa, 10 ng/mL insulin, 0.2 mM IM | medium 1: 3 days, medium 2: 3 days; repeated cylces for 24–30 days | |
| medium 2; DMEM-HG, 10% FCS, 10 ng/mL insulin | |||
| Kang [10] | DMEM-LG, 10% FCS, 1mM dexa, 0.5 mM IBMX, 1 μg/mL insulin, 200 μM IM | 21 days | |
| Diaz-Prado [4], Paracchini [18] | hMSC Mesenchymal Stem Cell Adipogenic Differentiation Medium (Lonza) | 21 days | |
| Bilic [2], Manochantr [57] | NH Adipodiff medium (Miltenyi) | 21 days | |
| chondrogenic | Sudo [56] | alphaMEM, 3.5 g/mL glucose, 1% ITS, 100 μg/mL sodium pyruvate, 0.2 mM AAP, 10−7 M dexa, 10 ng/mL TGF-b3 | 28–30 days, pellet culture |
| Alviano [1] | DMEM, 1 mM sodium pyruvate, 0.1 mM AAP, 10−7 M dexa, 10 ng/mL TGF-b3, 6.25 μg/mL insulin, 6.25 μg/mL transferrin, 6.25 μg/mL selenous acid, 5.33 μg/mL linolenic acid, 0.35 mM proline, 1.25 mg/mL BSA, 1/1.000 monotioglycerol | 21–28 days, pellet culture | |
| Diaz-Prado [4] | medium 1: DMEM, 15% FCS, 5 mg/mL AAP | first 2 days medium 1, | |
| medium 2: DMEM, 15% knockout serum, 1 μL/mL AAP, 10 μM dexa, 1 ng/mL TGF-b3, 6 μL/mL transferrin, 107 M retinoic acid | followed by 21 days medium 2; pellet culture | ||
| Kang [10] | NH chondrogenic medium (Miltenyi) | 21 days, pellet culture | |
| Portmann-Lanz [50] | DMEM-HG, 1% FCS, 50 ng/mL AAP, 10 ng/mL TGF-b1, 6.25 μg/mL insulin | 18–21 days, monolayer | |
| Wei [23] | DMEM. 10% FCS, 200 ng/mL rhuBMP-2 | induction 21 days after confluence, 14 days | |
| myogenic | Portmann-Lanz [50] | DMEM-HG, 10% FCS, 50 μmol/L hydrocortison, 0.1 μmol/L dexa | 18–21 days |
| Alviano [1] | DMEM, 5% FCS, 40% MCDB-201, 10−8 M dexa, ITS-LA*BSA 1×, 10−4 M AAP, 10 ng/mL bFGF, 10 ng/mL VEGF, 10 ng/mL IGF-1 | 21 days | |
| Bilic [2] | DMEM-HG, 10% FCS, 5% horse serum, 50 μM hydrocortison, 0.1 μM dexa | 7 days | |
| angiogenic | Alviano [1] | DMEM, 2% FCS, 50 ng/mL VEGF | 7 days, followed by matrigel assay |
| König [12] | Endothelial Growth Medium-2 (Lonza) +/− 50 ng/mL VEGF | at least 10 days, followed by matrigel assay | |
| Kim SW [83] | Endothelial Growth Medium-2 (Lonza) | 10–20 days | |
| cardiomyogenic | Zhao [25] | DMEM, 10% FCS, 2nd d addition of 10 ng/mL bFGF or 50 ng/mL activin A | (a) 7 days after addition of growth factors; (b) coculture with rat heart explants |
| Tsuji [58] | unknown | coculture with fetal murine cardiomyocytes | |
| Lisi [14] | DMEM, 10% FCS, 10 ng/mL EGF, 55 μM b-ME, d2-4: + 0.4 μM S-nitoso-N-acetylpenicillamine | seeded on s-IPN PRtU-PDMS/fibrin scaffold, 14 days | |
| hepatic | Tamagawa [53] | alphaMEM, 10% FCS, 0.1 mmoL/L dexa, 20 ng/mL hHGF, 10 ng/mL hFGF, 10 ng/mL oncostatin | 21 days |
| Paracchini [18] | medium 1: DMEM, 10% FCS, 1% NEAA, 55 μM b-ME, 10 ng/mL EGF | first 8 days medium 1, | |
| medium 2: IMEM, 10% FCS, 10−7 M dexa, 1% NEAA, 55 μM b-ME, 10 ng/mL EGF | followed by medium 2 until day 21 | ||
| pancreatic | Wei [84] | DMEM, N2 supplement, 0.1 mmoL/L nicotinamide | |
| Tamagawa [22] | medium 1: DMEM-HG, 10−6 M retinoic acid | first 2 days medium 1, medium 2 and 3 in matrigel | |
| medium 2: DMEM-LG, 10% FCS, 1 × N2 supplement, 10 mM nicotinamide, 20 ng/mL hEGF | |||
| medium 3: DMEM-LG, 10 nM exendin-4 | |||
| Kim J [11] | medium 1: DMEM-HG, 10% FCS, 10 mmoL nicotinamide, 4 nmol activin A, 10 nmol GLP-1 | first 7 days medium 1, | |
| medium 2: DMEM-LG, 10% FCS, 10 mmoL nicotinamide, 4 nmol activin A, 10 nmol GLP-1 | followed by 14 days medium 2 | ||
| neural | Portmann-Lanz [50] | DMEM-HG, 10% FCS, 30 μmol/L all trans retinoic acid | 18–21 days |
| Sakuragawa [51] | DMEM, 100 μM butylated hydroxianisole, 10 μM forskolin, 2% DMSO, 5 U/mL heparin, 5 nM K252a, 25 mM KCl, 2 mM valporic acid, 1 × N2 supplement, 10 ng/mL bFGF, 10 ng/mL PDGF | ||
| Tamagawa [85] | medium 1: alphaMEM, 10% FCS, 1 × N2 supplement, 10 ng/mL bFGF, 10 ng/mL EGF | 48 h medium 1, followed by 96 h medium 2 | |
| medium 2: alphaMEM, 1 μM all trans retinoic acid, 200 μM butylated hydroxianisole, 1× N2 supplement, 1 mM dibutyryl cyclic AMP, 0.5 mM IBMX | followed by 96 h medium 2 | ||
| Manochantr [57] | AdvancesSTEM Neural Differentiation Medium (HyClone) | until neural like cells were observed |
5. Differentiation potential of UC-MSC
| lineage | reference |
|---|---|
| osteogenic | [27,29,33,38,40,63,68,70,86,87,88,89,90,91,93,94,96,98,99,103,104,106,108,109], and osteocytes [26,27,28,30,33,38,40,41,63,64,65,67,68,73,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,108,110,111,112,113,114] |
| adipogenic | [26,27,30,33,38,40,63,64,65,67,68,70,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108] |
| chondrogenic | [27,29,33,38,40,63,68,70,86,87,88,89,90,91,93,94,96,98,99,103,104,106,108,109] |
| myogenic | [30] |
| angiogenic | [86,93,101,115] |
| cardiomyogenic | [38,68,92,96,116,117,118] |
| hepatic | [109,119,120] |
| pancreatic | [121,122,123,124] |
| neural | [31,32,33,35,36,65,68,91,104,125,126,127,128] |
6. Immunological properties of hAMSC and UC-MSC
7. Immortalization of hAMSC and UC-MSC
8. Prospective Clinical Applications: Current State and Outlook
9. Conclusion
Conflict of Interest
References and Notes
- Alviano, F.; Fossati, V.; Marchionni, C.; Arpinati, M.; Bonsi, L.; Franchina, M.; Lanzoni, G.; Cantoni, S.; Cavallini, C.; Bianchi, F.; et al. Term amniotic membrane is a high throughput source for multipotent mesenchymal stem cells with the ability to differentiate into endothelial cells in vitro. BMC. Dev. Biol. 2007, 7, 11. [Google Scholar] [CrossRef]
- Bilic, G.; Zeisberger, S.M.; Mallik, A.S.; Zimmermann, R.; Zisch, A.H. Comparative characterization of cultured human term amnion epithelial and mesenchymal stromal cells for application in cell therapy. Cell Transplant. 2008, 17, 955–968. [Google Scholar] [CrossRef] [Green Version]
- Cargnoni, A.; Ressel, L.; Rossi, D.; Poli, A.; Arienti, D.; Lombardi, G.; Parolini, O. Conditioned medium from amniotic mesenchymal tissue cells reduces progression of bleomycin-induced lung fibrosis. Cytotherapy 2012, 14, 153–161. [Google Scholar] [CrossRef]
- Diaz-Prado, S.; Muinos-Lopez, E.; Hermida-Gomez, T.; Rendal-Vazquez, M.E.; Fuentes-Boquete, I.; de Toro, F.J.; Blanco, F.J. Multilineage differentiation potential of cells isolated from the human amniotic membrane. J. Cell Biochem. 2010, 111, 846–857. [Google Scholar] [CrossRef]
- Diaz-Prado, S.; Muinos-Lopez, E.; Hermida-Gomez, T.; Cicione, C.; Rendal-Vazquez, M.E.; Fuentes-Boquete, I.; de Toro, F.J.; Blanco, F.J. Human amniotic membrane as an alternative source of stem cells for regenerative medicine. Differentiation 2011, 81, 162–171. [Google Scholar] [CrossRef]
- Evangelista, M.; Soncini, M.; Parolini, O. Placenta-derived stem cells: New hope for cell therapy? Cytotechnology 2008, 2008, 33–42. [Google Scholar]
- Ilancheran, S.; Moodley, Y.; Manuelpillai, U. Human fetal membranes: A source of stem cells for tissue regeneration and repair? Placenta 2009, 30, 2–10. [Google Scholar]
- In 't Anker, P.S.; Scherjon, S.A.; Kleijburg-van der Keur, C.; de Groot-Swings, G.M.; Claas, F.H.; Fibbe, W.E.; Kanhai, H.H. Isolation of mesenchymal stem cells of fetal or maternal origin from human placenta. Stem Cells 2004, 22, 1338–1345. [Google Scholar] [CrossRef]
- Insausti, C.L.; Blanquer, M.; Bleda, P.; Iniesta, P.; Majado, M.J.; Castellanos, G.; Moraleda, J.M. The amniotic membrane as a source of stem cells. Histol. Histopathol. 2010, 25, 91–98. [Google Scholar]
- Kang, J.W.; Koo, H.C.; Hwang, S.Y.; Kang, S.K.; Ra, J.C.; Lee, M.H.; Park, Y.H. Immunomodulatory effects of human amniotic membrane-derived mesenchymal stem cells. J. Vet. Sci. 2012, 13, 23–31. [Google Scholar] [CrossRef]
- Kim, J.; Park, S.; Kang, H.M.; Ahn, C.W.; Kwon, H.C.; Song, J.H.; Lee, Y.J.; Lee, K.H.; Yang, H.; Baek, S.Y.; et al. Human insulin secreted from insulinogenic xenograft restores normoglycemia in type i diabetic mice without immunosuppression. Cell Transplant. 2012. [Google Scholar] [CrossRef]
- König, J.; Huppertz, B.; Desoye, G.; Parolini, O.; Frohlich, J.D.; Weiss, G.; Dohr, G.; Sedlmayr, P.; Lang, I. Amnion-derived mesenchymal stromal cells show angiogenic properties but resist differentiation into mature endothelial cells. Stem Cells Dev. 2012, 21, 1309–1320. [Google Scholar] [CrossRef]
- Kronsteiner, B.; Wolbank, S.; Peterbauer, A.; Hackl, C.; Redl, H.; van, G.M.; Gabriel, C. Human mesenchymal stem cells from adipose tissue and amnion influence t-cells depending on stimulation method and presence of other immune cells. Stem Cells Dev. 2011, 20, 2115–2126. [Google Scholar] [CrossRef]
- Lisi, A.; Briganti, E.; Ledda, M.; Losi, P.; Grimaldi, S.; Marchese, R.; Soldani, G. A combined synthetic-fibrin scaffold supports growth and cardiomyogenic commitment of human placental derived stem cells. PLoS One 2012, 7, e34284. [Google Scholar]
- Magatti, M.; De, M.S.; Vertua, E.; Nassauto, C.; Albertini, A.; Wengler, G.S.; Parolini, O. Amniotic mesenchymal tissue cells inhibit dendritic cell differentiation of peripheral blood and amnion resident monocytes. Cell Transplant. 2009, 18, 899–914. [Google Scholar] [CrossRef]
- Manuelpillai, U.; Moodley, Y.; Borlongan, C.V.; Parolini, O. Amniotic membrane and amniotic cells: Potential therapeutic tools to combat tissue inflammation and fibrosis? Placenta 2011, 32 Suppl. 4, S320–S325. [Google Scholar] [CrossRef]
- Miki, T.; Strom, S.C. Amnion-derived pluripotent/multipotent stem cells. Stem Cell Rev. 2006, 2, 133–142. [Google Scholar] [CrossRef]
- Paracchini, V.; Carbone, A.; Colombo, F.; Castellani, S.; Mazzucchelli, S.; Gioia, S.D.; Degiorgio, D.; Seia, M.; Porretti, L.; Colombo, C.; et al. Amniotic mesenchymal stem cells: A new source for hepatocyte-like cells and induction of cftr expression by coculture with cystic fibrosis airway epithelial cells. J. Biomed. Biotechnol. 2012, 2012, 575471. [Google Scholar]
- Parolini, O.; Alviano, F.; Bagnara, G.P.; Bilic, G.; Buhring, H.J.; Evangelista, M.; Hennerbichler, S.; Liu, B.; Magatti, M.; Mao, N.; et al. Concise review: Isolation and characterization of cells from human term placenta: Outcome of the first international workshop on placenta derived stem cells. Stem Cells 2008, 26, 300–311. [Google Scholar] [CrossRef] [Green Version]
- Roubelakis, M.G.; Trohatou, O.; Anagnou, N.P. Amniotic fluid and amniotic membrane stem cells: Marker discovery. Stem Cells Int. 2012, 2012, 107836. [Google Scholar]
- Stadler, G.; Hennerbichler, S.; Lindenmair, A.; Peterbauer, A.; Hofer, K.; van, G.M.; Gabriel, C.; Redl, H.; Wolbank, S. Phenotypic shift of human amniotic epithelial cells in culture is associated with reduced osteogenic differentiation in vitro. Cytotherapy. 2008, 10, 743–752. [Google Scholar] [CrossRef]
- Tamagawa, T.; Ishiwata, I.; Sato, K.; Nakamura, Y. Induced in vitro differentiation of pancreatic-like cells from human amnion-derived fibroblast-like cells. Hum. Cell 2009, 22, 55–63. [Google Scholar] [CrossRef]
- Wei, J.P.; Nawata, M.; Wakitani, S.; Kametani, K.; Ota, M.; Toda, A.; Konishi, I.; Ebara, S.; Nikaido, T. Human amniotic mesenchymal cells differentiate into chondrocytes. Cloning Stem Cells 2009, 11, 19–26. [Google Scholar] [CrossRef]
- Wolbank, S.; Stadler, G.; Peterbauer, A.; Gillich, A.; Karbiener, M.; Streubel, B.; Wieser, M.; Katinger, H.; van, G.M.; Redl, H.; et al. Telomerase immortalized human amnion- and adipose-derived mesenchymal stem cells: Maintenance of differentiation and immunomodulatory characteristics. Tissue Eng. Part A 2009, 15, 1843–1854. [Google Scholar] [CrossRef]
- Zhao, P.; Ise, H.; Hongo, M.; Ota, M.; Konishi, I.; Nikaido, T. Human amniotic mesenchymal cells have some characteristics of cardiomyocytes. Transplantation 2005, 79, 528–535. [Google Scholar] [CrossRef]
- Covas, D.T.; Siufi, J.L.; Silva, A.R.; Orellana, M.D. Isolation and culture of umbilical vein mesenchymal stem cells. Braz. J. Med. Biol. Res. 2003, 36, 1179–1183. [Google Scholar] [CrossRef]
- Panepucci, R.A.; Siufi, J.L.; Silva, W.A., Jr.; Proto-Siquiera, R.; Neder, L.; Orellana, M.; Rocha, V.; Covas, D.T.; Zago, M.A. Comparison of gene expression of umbilical cord vein and bone marrow-derived mesenchymal stem cells. Stem Cells 2004, 22, 1263–1278. [Google Scholar] [CrossRef]
- Romanov, Y.A.; Svintsitskaya, V.A.; Smirnov, V.N. Searching for alternative sources of postnatal human mesenchymal stem cells: Candidate msc-like cells from umbilical cord. Stem Cells 2003, 21, 105–110. [Google Scholar] [CrossRef]
- Bailey, M.M.; Wang, L.; Bode, C.J.; Mitchell, K.E.; Detamore, M.S. A comparison of human umbilical cord matrix stem cells and temporomandibular joint condylar chondrocytes for tissue engineering temporomandibular joint condylar cartilage. Tissue Eng 2007, 13, 2003–2010. [Google Scholar] [CrossRef]
- Conconi, M.T.; Burra, P.; Di Liddo, R.; Calore, C.; Turetta, M.; Bellini, S.; Bo, P.; Nussdorfer, G.G.; Parnigotto, P.P. Cd105(+) cells from wharton's jelly show in vitro and in vivo myogenic differentiative potential. Int. J. Mol. Med. 2006, 18, 1089–1096. [Google Scholar]
- Fu, Y.S.; Cheng, Y.C.; Lin, M.Y.; Cheng, H.; Chu, P.M.; Chou, S.C.; Shih, Y.H.; Ko, M.H.; Sung, M.S. Conversion of human umbilical cord mesenchymal stem cells in wharton's jelly to dopaminergic neurons in vitro: Potential therapeutic application for parkinsonism. Stem Cells 2006, 24, 115–124. [Google Scholar] [CrossRef]
- Fu, Y.S.; Shih, Y.T.; Cheng, Y.C.; Min, M.Y. Transformation of human umbilical mesenchymal cells into neurons in vitro. J. Biomed. Sci. 2004, 11, 652–660. [Google Scholar] [CrossRef]
- Karahuseyinoglu, S.; Cinar, O.; Kilic, E.; Kara, F.; Akay, G.G.; Demiralp, D.O.; Tukun, A.; Uckan, D.; Can, A. Biology of stem cells in human umbilical cord stroma: In situ and in vitro surveys. Stem Cells 2007, 25, 319–331. [Google Scholar] [CrossRef]
- Lund, R.D.; Wang, S.; Lu, B.; Girman, S.; Holmes, T.; Sauve, Y.; Messina, D.J.; Harris, I.R.; Kihm, A.J.; Harmon, A.M.; et al. Cells isolated from umbilical cord tissue rescue photoreceptors and visual functions in a rodent model of retinal disease. Stem Cells 2007, 25, 602–611. [Google Scholar]
- Ma, L.; Feng, X.Y.; Cui, B.L.; Law, F.; Jiang, X.W.; Yang, L.Y.; Xie, Q.D.; Huang, T.H. Human umbilical cord wharton's jelly-derived mesenchymal stem cells differentiation into nerve-like cells. Chin. Med. J. (Engl) 2005, 118, 1987–1993. [Google Scholar]
- Mitchell, K.E.; Weiss, M.L.; Mitchell, B.M.; Martin, P.; Davis, D.; Morales, L.; Helwig, B.; Beerenstrauch, M.; Abou-Easa, K.; Hildreth, T.; et al. Matrix cells from wharton's jelly form neurons and glia. Stem Cells 2003, 21, 50–60. [Google Scholar] [CrossRef]
- Schmidt, D.; Mol, A.; Odermatt, B.; Neuenschwander, S.; Breymann, C.; Gossi, M.; Genoni, M.; Zund, G.; Hoerstrup, S.P. Engineering of biologically active living heart valve leaflets using human umbilical cord-derived progenitor cells. Tissue. Eng. 2006, 12, 3223–3232. [Google Scholar] [CrossRef]
- Wang, H.S.; Hung, S.C.; Peng, S.T.; Huang, C.C.; Wei, H.M.; Guo, Y.J.; Fu, Y.S.; Lai, M.C.; Chen, C.C. Mesenchymal stem cells in the wharton's jelly of the human umbilical cord. Stem Cells 2004, 22, 1330–1337. [Google Scholar] [CrossRef]
- Weiss, M.L.; Medicetty, S.; Bledsoe, A.R.; Rachakatla, R.S.; Choi, M.; Merchav, S.; Luo, Y.; Rao, M.S.; Velagaleti, G.; Troyer, D. Human umbilical cord matrix stem cells: Preliminary characterization and effect of transplantation in a rodent model of parkinson's disease. Stem Cells 2006, 24, 781–792. [Google Scholar] [CrossRef]
- Baksh, D.; Yao, R.; Tuan, R.S. Comparison of proliferative and multilineage differentiation potential of human mesenchymal stem cells derived from umbilical cord and bone marrow. Stem Cells 2007, 25, 1384–1392. [Google Scholar] [CrossRef]
- Sarugaser, R.; Lickorish, D.; Baksh, D.; Hosseini, M.M.; Davies, J.E. Human umbilical cord perivascular (hucpv) cells: A source of mesenchymal progenitors. Stem Cells 2005, 23, 220–229. [Google Scholar] [CrossRef]
- Bieback, K.; Kluter, H. Mesenchymal stromal cells from umbilical cord blood. Curr. Stem. Cell Res. Ther. 2007, 2, 310–323. [Google Scholar] [CrossRef]
- Gang, E.J.; Jeong, J.A.; Han, S.; Yan, Q.; Jeon, C.J.; Kim, H. In vitro endothelial potential of human uc blood-derived mesenchymal stem cells. Cytotherapy 2006, 8, 215–227. [Google Scholar] [CrossRef]
- Gang, E.J.; Jeong, J.A.; Hong, S.H.; Hwang, S.H.; Kim, S.W.; Yang, I.H.; Ahn, C.; Han, H.; Kim, H. Skeletal myogenic differentiation of mesenchymal stem cells isolated from human umbilical cord blood. Stem Cells 2004, 22, 617–624. [Google Scholar] [CrossRef]
- Hou, L.; Cao, H.; Wang, D.; Wei, G.; Bai, C.; Zhang, Y.; Pei, X. Induction of umbilical cord blood mesenchymal stem cells into neuron-like cells in vitro. Int. J. Hematol. 2003, 78, 256–261. [Google Scholar] [CrossRef]
- Lee, O.K.; Kuo, T.K.; Chen, W.M.; Lee, K.D.; Hsieh, S.L.; Chen, T.H. Isolation of multipotent mesenchymal stem cells from umbilical cord blood. Blood 2004, 103, 1669–1675. [Google Scholar] [CrossRef]
- Bilic, G.; Ochsenbein-Kolble, N.; Hall, H.; Huch, R.; Zimmermann, R. In vitro lesion repair by human amnion epithelial and mesenchymal cells. Am. J. Obstet. Gynecol. 2004, 190, 87–92. [Google Scholar] [CrossRef]
- Casey, M.L.; MacDonald, P.C. Keratinocyte growth factor expression in the mesenchymal cells of human amnion. J. Clin. Endocrinol. Metab 1997, 82, 3319–3323. [Google Scholar] [CrossRef]
- Magatti, M.; De, M.S.; Vertua, E.; Gibelli, L.; Wengler, G.S.; Parolini, O. Human amnion mesenchyme harbors cells with allogeneic t-cell suppression and stimulation capabilities. Stem Cells 2008, 26, 182–192. [Google Scholar] [CrossRef]
- Portmann-Lanz, C.B.; Schoeberlein, A.; Huber, A.; Sager, R.; Malek, A.; Holzgreve, W.; Surbek, D.V. Placental mesenchymal stem cells as potential autologous graft for pre- and perinatal neuroregeneration. Am. J. Obstet. Gynecol. 2006, 194, 664–673. [Google Scholar] [CrossRef]
- Sakuragawa, N.; Kakinuma, K.; Kikuchi, A.; Okano, H.; Uchida, S.; Kamo, I.; Kobayashi, M.; Yokoyama, Y. Human amnion mesenchyme cells express phenotypes of neuroglial progenitor cells. J. Neurosci. Res. 2004, 78, 208–214. [Google Scholar] [CrossRef]
- Soncini, M.; Vertua, E.; Gibelli, L.; Zorzi, F.; Denegri, M.; Albertini, A.; Wengler, G.S.; Parolini, O. Isolation and characterization of mesenchymal cells from human fetal membranes. J. Tissue Eng. Regen. Med. 2007, 1, 296–305. [Google Scholar] [CrossRef]
- Tamagawa, T.; Oi, S.; Ishiwata, I.; Ishikawa, H.; Nakamura, Y. Differentiation of mesenchymal cells derived from human amniotic membranes into hepatocyte-like cells in vitro. Hum. Cell 2007, 20, 77–84. [Google Scholar] [CrossRef]
- Whittle, W.L.; Gibb, W.; Challis, J.R. The characterization of human amnion epithelial and mesenchymal cells: The cellular expression, activity and glucocorticoid regulation of prostaglandin output. Placenta 2000, 21, 394–401. [Google Scholar] [CrossRef]
- Wolbank, S.; Peterbauer, A.; Fahrner, M.; Hennerbichler, S.; van, G.M.; Stadler, G.; Redl, H.; Gabriel, C. Dose-dependent immunomodulatory effect of human stem cells from amniotic membrane: A comparison with human mesenchymal stem cells from adipose tissue. Tissue Eng 2007, 13, 1173–1183. [Google Scholar] [CrossRef]
- Sudo, K.; Kanno, M.; Miharada, K.; Ogawa, S.; Hiroyama, T.; Saijo, K.; Nakamura, Y. Mesenchymal progenitors able to differentiate into osteogenic, chondrogenic, and/or adipogenic cells in vitro are present in most primary fibroblast-like cell population. Stem Cells 2007, 25, 1610–1617. [Google Scholar] [CrossRef]
- Manochantr, S.; Tantrawatpan, C.; Kheolamai, P.; Pratya, Y.; Supokawej, A.; Issaragrisil, S. Isolation, characterization and neural differentiation potential of amnion derived mesenchymal stem cells. J. Med. Assoc. Thai. 2010, 93 Suppl. 7, S183–S191. [Google Scholar]
- Tsuji, H.; Miyoshi, S.; Ikegami, Y.; Hida, N.; Asada, H.; Togashi, I.; Suzuki, J.; Satake, M.; Nakamizo, H.; Tanaka, M.; et al. Xenografted human amniotic membrane-derived mesenchymal stem cells are immunologically tolerated and transdifferentiated into cardiomyocytes. Circ. Res. 2010, 106, 1613–1623. [Google Scholar] [CrossRef]
- Miki, T.; Mitamura, K.; Ross, M.A.; Stolz, D.B.; Strom, S.C. Identification of stem cell marker-positive cells by immunofluorescence in term human amnion. J. Reprod. Immunol. 2007, 75, 91–96. [Google Scholar] [CrossRef]
- Bilic, G.; Hall, H.; Bittermann, A.G.; Zammeretti, P.; Burkhart, T.; Ochsenbein-Kolble, N.; Zimmermann, R. Human preterm amnion cells cultured in 3-dimensional collagen i and fibrin matrices for tissue engineering purposes. Am. J. Obstet. Gynecol. 2005, 193, 1724–1732. [Google Scholar]
- Pasquinelli, G.; Tazzari, P.; Ricci, F.; Vaselli, C.; Buzzi, M.; Conte, R.; Orrico, C.; Foroni, L.; Stella, A.; Alviano, F.; et al. Ultrastructural characteristics of human mesenchymal stromal (stem) cells derived from bone marrow and term placenta. Ultrastruct. Pathol. 2007, 31, 23–31. [Google Scholar] [CrossRef]
- Kadam, S.S.; Bhonde, R.R. Islet neogenesis from the constitutively nestin expressing human umbilical cord matrix derived mesenchymal stem cells. Islets 2010, 2, 112–120. [Google Scholar] [CrossRef]
- Ishige, I.; Nagamura-Inoue, T.; Honda, M.; Harnprasopwat, R.; Kido, M.; Sugimoto, M.; Nakauchi, H.; Tojo, A. Comparison of mesenchymal stem cells derived from arterial, venous, and wharton’s jelly explants of human umbilical cord. Int. J. Hematol. 2009, 90, 261. [Google Scholar] [CrossRef]
- La Rocca, G.; Anzalone, R.; Corrao, S.; Magno, F.; Loria, T.; Lo Iacono, M.; Di Stefano, A.; Giannuzzi, P.; Marasa, L.; Cappello, F.; et al. Isolation and characterization of oct-4+/hla-g+ mesenchymal stem cells from human umbilical cord matrix: Differentiation potential and detection of new markers. Histochem. Cell. Biol. 2009, 131, 267–282. [Google Scholar] [CrossRef]
- Lu, L.L.; Liu, Y.J.; Yang, S.G.; Zhao, Q.J.; Wang, X.; Gong, W.; Han, Z.B.; Xu, Z.S.; Lu, Y.X.; Liu, D.; et al. Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials. Haematologica 2006, 91, 1017–1026. [Google Scholar]
- Schneider, R.K.; Pullen, A.; Kramann, R.; Bornemann, J.; Knuchel, R.; Neuss, S.; Perez-Bouza, A. Long-term survival and characterisation of human umbilical cord-derived mesenchymal stem cells on dermal equivalents. Differentiation 2010, 79, 182–193. [Google Scholar] [CrossRef]
- Tsagias, N.; Koliakos, I.; Karagiannis, V.; Eleftheriadou, M.; Koliakos, G.G. Isolation of mesenchymal stem cells using the total length of umbilical cord for transplantation purposes. Transfus. Med. 2011, 21, 253–261. [Google Scholar] [CrossRef]
- Gonzalez, R.; Griparic, L.; Umana, M.; Burgee, K.; Vargas, V.; Nasrallah, R.; Silva, F.; Patel, A. An efficient approach to isolation and characterization of pre- and postnatal umbilical cord lining stem cells for clinical applications. Cell Transplant. 2010, 19, 1439–1449. [Google Scholar] [CrossRef]
- Kadner, A.; Zund, G.; Maurus, C.; Breymann, C.; Yakarisik, S.; Kadner, G.; Turina, M.; Hoerstrup, S.P. Human umbilical cord cells for cardiovascular tissue engineering: A comparative study. Eur. J. Cardiothorac. Surg. 2004, 25, 635–641. [Google Scholar] [CrossRef]
- Majore, I.; Moretti, P.; Stahl, F.; Hass, R.; Kasper, C. Growth and differentiation properties of mesenchymal stromal cell populations derived from whole human umbilical cord. Stem. Cell. Rev. 2010, 7, 17–31. [Google Scholar]
- Petsa, A.; Gargani, S.; Felesakis, A.; Grigoriadis, N.; Grigoriadis, I. Effectiveness of protocol for the isolation of wharton's jelly stem cells in large-scale applications. In vitro Cell. Dev. Biol. Anim. 2009, 45, 573–576. [Google Scholar] [CrossRef]
- Moretti, P.; Hatlapatka, T.; Marten, D.; Lavrentieva, A.; Majore, I.; Hass, R.; Kasper, C. Mesenchymal stromal cells derived from human umbilical cord tissues: Primitive cells with potential for clinical and tissue engineering applications. Adv. Biochem. Eng. Biotechnol. 2010, 123, 29–54. [Google Scholar]
- Majore, I.; Moretti, P.; Hass, R.; Kasper, C. Identification of subpopulations in mesenchymal stem cell-like cultures from human umbilical cord. Cell. Commun. Signal. 2009, 7, 6. [Google Scholar] [CrossRef] [Green Version]
- Sivasubramaniyan, K.; Lehnen, D.; Ghazanfari, R.; Sobiesiak, M.; Harichandan, A.; Mortha, E.; Petkova, N.; Grimm, S.; Cerabona, F.; de Zwart, P.; et al. Phenotypic and functional heterogeneity of human bone marrow- and amnion-derived msc subsets. Ann. NY Acad. Sci. 2012, 1266, 94–106. [Google Scholar]
- Battula, V.L.; Treml, S.; Bareiss, P.M.; Gieseke, F.; Roelofs, H.; de Zwart, P.; Muller, I.; Schewe, B.; Skutella, T.; Fibbe, W.E.; et al. Isolation of functionally distinct mesenchymal stem cell subsets using antibodies against cd56, cd271, and mesenchymal stem cell antigen-1. Haematologica 2009, 94, 173–184. [Google Scholar] [CrossRef]
- Buhring, H.J.; Battula, V.L.; Treml, S.; Schewe, B.; Kanz, L.; Vogel, W. Novel markers for the prospective isolation of human msc. Ann. NY Acad. Sci. 2007, 1106, 262–271. [Google Scholar]
- Gang, E.J.; Bosnakovski, D.; Figueiredo, C.A.; Visser, J.W.; Perlingeiro, R.C. Ssea-4 identifies mesenchymal stem cells from bone marrow. Blood 2007, 109, 1743–1751. [Google Scholar] [CrossRef]
- Kawanabe, N.; Murata, S.; Fukushima, H.; Ishihara, Y.; Yanagita, T.; Yanagita, E.; Ono, M.; Kurosaka, H.; Kamioka, H.; Itoh, T.; et al. Stage-specific embryonic antigen-4 identifies human dental pulp stem cells. Exp. Cell. Res. 2012, 318, 453–463. [Google Scholar] [CrossRef]
- Martinez, C.; Hofmann, T.J.; Marino, R.; Dominici, M.; Horwitz, E.M. Human bone marrow mesenchymal stromal cells express the neural ganglioside gd2: A novel surface marker for the identification of mscs. Blood 2007, 109, 4245–4248. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Liao, W.; Gu, D.; Liang, L.; Liu, M.; Du, W.; Liu, P.; Zhang, L.; Lu, S.; Dong, C.; et al. Neural ganglioside gd2 identifies a subpopulation of mesenchymal stem cells in umbilical cord. Cell. Physiol. Biochem. 2009, 23, 415–424. [Google Scholar] [CrossRef]
- Cheng, H.; Qiu, L.; Ma, J.; Zhang, H.; Cheng, M.; Li, W.; Zhao, X.; Liu, K. Replicative senescence of human bone marrow and umbilical cord derived mesenchymal stem cells and their differentiation to adipocytes and osteoblasts. Mol. Biol. Rep. 2011, 38, 5161–5168. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef]
- Kim, S.W.; Zhang, H.Z.; Kim, C.E.; An, H.S.; Kim, J.M.; Kim, M.H. Amniotic mesenchymal stem cells have robust angiogenic properties and are effective in treating hindlimb ischaemia. Cardiovasc. Res. 2012, 93, 525–534. [Google Scholar] [CrossRef]
- Wei, J.P.; Zhang, T.S.; Kawa, S.; Aizawa, T.; Ota, M.; Akaike, T.; Kato, K.; Konishi, I.; Nikaido, T. Human amnion-isolated cells normalize blood glucose in streptozotocin-induced diabetic mice. Cell Transplant. 2003, 12, 545–552. [Google Scholar]
- Tamagawa, T.; Ishiwata, I.; Ishikawa, H.; Nakamura, Y. Induced in-vitro differentiation of neural-like cells from human amnion-derived fibroblast-like cells. Hum. Cell 2008, 21, 38–45. [Google Scholar] [CrossRef]
- Chen, M.Y.; Lie, P.C.; Li, Z.L.; Wei, X. Endothelial differentiation of wharton's jelly-derived mesenchymal stem cells in comparison with bone marrow-derived mesenchymal stem cells. Exp. Hematol. 2009, 37, 629–640. [Google Scholar] [CrossRef]
- Ciavarella, S.; Dammacco, F.; De Matteo, M.; Loverro, G.; Silvestris, F. Umbilical cord mesenchymal stem cells: Role of regulatory genes in their differentiation to osteoblasts. Stem Cells Dev. 2009, 18, 1211–1220. [Google Scholar] [CrossRef]
- Covas, D.T.; Panepucci, R.A.; Fontes, A.M.; Silva, W.A., Jr.; Orellana, M.D.; Freitas, M.C.; Neder, L.; Santos, A.R.; Peres, L.C.; Jamur, M.C.; et al. Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with cd146+ perivascular cells and fibroblasts. Exp. Hematol. 2008, 36, 642–654. [Google Scholar] [CrossRef]
- Diao, Y.; Ma, Q.; Cui, F.; Zhong, Y. Human umbilical cord mesenchymal stem cells: Osteogenesis in vivo as seed cells for bone tissue engineering. J. Biomed. Mater. Res. A 2008, 91, 123–131. [Google Scholar]
- Jo, C.H.; Kim, O.S.; Park, E.Y.; Kim, B.J.; Lee, J.H.; Kang, S.B.; Lee, J.H.; Han, H.S.; Rhee, S.H.; Yoon, K.S. Fetal mesenchymal stem cells derived from human umbilical cord sustain primitive characteristics during extensive expansion. Cell Tissue Res. 2008, 334, 423–433. [Google Scholar] [CrossRef]
- Kadam, S.S.; Tiwari, S.; Bhonde, R.R. Simultaneous isolation of vascular endothelial cells and mesenchymal stem cells from the human umbilical cord. In Vitro Cell Dev. Biol. Anim. 2009, 45, 23–27. [Google Scholar] [CrossRef]
- Kadivar, M.; Khatami, S.; Mortazavi, Y.; Shokrgozar, M.A.; Taghikhani, M.; Soleimani, M. In vitro cardiomyogenic potential of human umbilical vein-derived mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2006, 340, 639–647. [Google Scholar] [CrossRef]
- Kestendjieva, S.; Kyurkchiev, D.; Tsvetkova, G.; Mehandjiev, T.; Dimitrov, A.; Nikolov, A.; Kyurkchiev, S. Characterization of mesenchymal stem cells isolated from the human umbilical cord. Cell. Biol. Int. 2008, 32, 724–732. [Google Scholar] [CrossRef]
- Liu, X.D.; Liu, B.; Li, X.S.; Mao, N. Isolation and identification of mesenchymal stem cells from perfusion of human umbilical cord vein. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2007, 15, 1019–1022. [Google Scholar]
- Lu, L.L.; Song, Y.P.; Wei, X.D.; Fang, B.J.; Zhang, Y.L.; Li, Y.F. comparative characterization of mesenchymal stem cells from human umbilical cord tissue and bone marrow. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2008, 16, 140–146. [Google Scholar]
- Martin-Rendon, E.; Sweeney, D.; Lu, F.; Girdlestone, J.; Navarrete, C.; Watt, S.M. 5-azacytidine-treated human mesenchymal stem/progenitor cells derived from umbilical cord, cord blood and bone marrow do not generate cardiomyocytes in vitro at high frequencies. Vox Sang 2008, 95, 137–148. [Google Scholar] [CrossRef]
- Qiao, C.; Xu, W.; Zhu, W.; Hu, J.; Qian, H.; Yin, Q.; Jiang, R.; Yan, Y.; Mao, F.; Yang, H.; et al. Human mesenchymal stem cells isolated from the umbilical cord. Cell. Biol. Int. 2008, 32, 8–15. [Google Scholar] [CrossRef]
- Sudo, K.; Kanno, M.; Miharada, K.; Ogawa, S.; Hiroyama, T.; Saijo, K.; Nakamura, Y. Mesenchymal progenitors able to differentiate into osteogenic, chondrogenic, and/or adipogenic cells in vitro are present in most primary fibroblast-like cell populations. Stem Cells 2007, 25, 1610–1617. [Google Scholar] [CrossRef]
- Suzdal'tseva, Y.G.; Burunova, V.V.; Vakhrushev, I.V.; Yarygin, V.N.; Yarygin, K.N. Capability of human mesenchymal cells isolated from different sources to differentiation into tissues of mesodermal origin. Bull. Exp. Biol. Med. 2007, 143, 114–121. [Google Scholar] [CrossRef]
- Tian, X.; Fu, R.Y.; Chen, Y.; Yuan, L.X. isolation of multipotent mesenchymal stem cells from the tissue of umbilical cord for osteoblasts and adipocytes differentiation. Sichuan Da Xue Xue Bao Yi Xue Ban 2008, 39, 26–29. [Google Scholar]
- Wu, K.H.; Zhou, B.; Lu, S.H.; Feng, B.; Yang, S.G.; Du, W.T.; Gu, D.S.; Han, Z.C.; Liu, Y.L. In vitro and in vivo differentiation of human umbilical cord derived stem cells into endothelial cells. J. Cell. Biochem. 2007, 100, 608–616. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Teoh, S.H.; Chong, M.S.; Schantz, J.T.; Fisk, N.M.; Choolani, M.A.; Chan, J. Superior osteogenic capacity for bone tissue engineering of fetal compared with perinatal and adult mesenchymal stem cells. Stem Cells 2009, 27, 126–137. [Google Scholar] [CrossRef]
- Farias, V.A.; Linares-Fernandez, J.L.; Penalver, J.L.; Paya Colmenero, J.A.; Ferron, G.O.; Duran, E.L.; Fernandez, R.M.; Olivares, E.G.; O'Valle, F.; Puertas, A.; et al. Human umbilical cord stromal stem cell express cd10 and exert contractile properties. Placenta 2010, 32, 86–95. [Google Scholar]
- Montanucci, P.; Basta, G.; Pescara, T.; Pennoni, I.; Di Giovanni, F.; Calafiore, R. New simple and rapid method for purification of mesenchymal stem cells from the human umbilical cord wharton jelly. Tissue Eng. Part A 2011, 17, 2651–2661. [Google Scholar] [CrossRef]
- Salehinejad, P.; Alitheen, N.B.; Nematollahi-Mahani, S.N.; Ali, A.M.; Omar, A.R.; Janzamin, E.; Hajghani, M. Effect of culture media on expansion properties of human umbilical cord matrix-derived mesenchymal cells. Cytotherapy 2012, 14, 948–953. [Google Scholar] [CrossRef]
- Struys, T.; Moreels, M.; Martens, W.; Donders, R.; Wolfs, E.; Lambrichts, I. Ultrastructural and immunocytochemical analysis of multilineage differentiated human dental pulp- and umbilical cord-derived mesenchymal stem cells. Cells Tissues Organs 2011, 193, 366–378. [Google Scholar] [CrossRef]
- Xu, Z.F.; Pan, A.Z.; Yong, F.; Shen, C.Y.; Chen, Y.W.; Wu, R.H. Human umbilical mesenchymal stem cell and its adipogenic differentiation: Profiling by nuclear magnetic resonance spectroscopy. World J. Stem. Cells 2012, 4, 21–27. [Google Scholar] [CrossRef]
- Qiao, S.M.; Chen, G.H.; Wang, Y.; Wu, D.P. ultrastructure of human umbilical cord mesenchymal stem cells. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2012, 20, 443–447. [Google Scholar]
- Zhang, Y.N.; Lie, P.C.; Wei, X. Differentiation of mesenchymal stromal cells derived from umbilical cord wharton's jelly into hepatocyte-like cells. Cytotherapy 2009, 11, 548–558. [Google Scholar] [CrossRef]
- Honsawek, S.; Dhitiseith, D.; Phupong, V. Effects of demineralized bone matrix on proliferation and osteogenic differentiation of mesenchymal stem cells from human umbilical cord. J. Med. Assoc. Thai 2006, 89 Suppl. 3, S189–S195. [Google Scholar]
- Hou, T.; Xu, J.; Wu, X.; Xie, Z.; Luo, F.; Zhang, Z.; Zeng, L. Umbilical cord wharton's jelly: A new potential cell source of mesenchymal stromal cells for bone tissue engineering. Tissue Eng Part A 2009, 15, 2325–2334. [Google Scholar] [CrossRef]
- Passeri, S.; Nocchi, F.; Lamanna, R.; Lapi, S.; Miragliotta, V.; Giannessi, E.; Abramo, F.; Stornelli, M.R.; Matarazzo, M.; Plenteda, D.; et al. Isolation and expansion of equine umbilical cord-derived matrix cells (eucmcs). Cell. Biol. Int. 2009, 33, 100–105. [Google Scholar] [CrossRef]
- Baba, K.; Yamazaki, Y.; Ikemoto, S.; Aoyagi, K.; Takeda, A.; Uchinuma, E. Osteogenic potential of human umbilical cord-derived mesenchymal stromal cells cultured with umbilical cord blood-derived autoserum. J. Craniomaxillofac. Surg. 2012, in press. [Google Scholar]
- Kang, M.N.; Yoon, H.H.; Seo, Y.K.; Park, J.K. Effect of mechanical stimulation on the differentiation of cord stem cells. Connect. Tissue Res. 2012, 53, 149–159. [Google Scholar] [CrossRef]
- Xu, Y.; Meng, H.; Li, C.; Hao, M.; Wang, Y.; Yu, Z.; Li, Q.; Han, J.; Zhai, Q.; Qiu, L. Umbilical cord-derived mesenchymal stem cells isolated by a novel explantation technique can differentiate into functional endothelial cells and promote revascularization. Stem Cells Dev. 2010, 19, 1511–1522. [Google Scholar] [CrossRef]
- Pereira, W.C.; Khushnooma, I.; Madkaikar, M.; Ghosh, K. Reproducible methodology for the isolation of mesenchymal stem cells from human umbilical cord and its potential for cardiomyocyte generation. J. Tissue Eng. Regen. Med. 2008, 2, 394–399. [Google Scholar] [CrossRef]
- Qian, Q.; Qian, H.; Zhang, X.; Zhu, W.; Yan, Y.; Ye, S.; Peng, X.; Li, W.; Xu, Z.; Sun, L.; et al. 5-azacytidine induces cardiac differentiation of human umbilical cord-derived mesenchymal stem cells by activating extracellular regulated kinase. Stem Cells Dev. 2012, 21, 67–75. [Google Scholar] [CrossRef]
- Zhao, Z.; Chen, Z.; Zhao, X.; Pan, F.; Cai, M.; Wang, T.; Zhang, H.; Lu, J.R.; Lei, M. Sphingosine-1-phosphate promotes the differentiation of human umbilical cord mesenchymal stem cells into cardiomyocytes under the designated culturing conditions. J. Biomed. Sci. 2011, 18, 37. [Google Scholar] [CrossRef]
- Campard, D.; Lysy, P.A.; Najimi, M.; Sokal, E.M. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology 2008, 134, 833–848. [Google Scholar]
- Zhao, Q.; Ren, H.; Li, X.; Chen, Z.; Zhang, X.; Gong, W.; Liu, Y.; Pang, T.; Han, Z.C. Differentiation of human umbilical cord mesenchymal stromal cells into low immunogenic hepatocyte-like cells. Cytotherapy 2009, 11, 414–426. [Google Scholar] [CrossRef]
- Wang, H.W.; Lin, L.M.; He, H.Y.; You, F.; Li, W.Z.; Huang, T.H.; Ma, G.X.; Ma, L. Human umbilical cord mesenchymal stem cells derived from wharton's jelly differentiate into insulin-producing cells in vitro. Chin. Med. J. (Engl) 2011, 124, 1534–1539. [Google Scholar]
- Wang, H.S.; Shyu, J.F.; Shen, W.S.; Hsu, H.C.; Chi, T.C.; Chen, C.P.; Huang, S.W.; Shyr, Y.M.; Tang, K.T.; Chen, T.H. Transplantation of insulin-producing cells derived from umbilical cord stromal mesenchymal stem cells to treat nod mice. Cell Transplant. 2011, 20, 455–466. [Google Scholar] [CrossRef]
- Chao, K.C.; Chao, K.F.; Fu, Y.S.; Liu, S.H. Islet-like clusters derived from mesenchymal stem cells in wharton's jelly of the human umbilical cord for transplantation to control type 1 diabetes. PLoS ONE 2008, 3, e1451. [Google Scholar] [CrossRef]
- Wu, L.F.; Wang, N.N.; Liu, Y.S.; Wei, X. Differentiation of wharton's jelly primitive stromal cells into insulin-producing cells in comparison with bone marrow mesenchymal stem cells. Tissue Eng Part A 2009, 15, 2865–2873. [Google Scholar] [CrossRef]
- Chou, S.C.; Ko, T.L.; Fu, Y.Y.; Wang, H.W.; Fu, Y.S. Identification of genetic networks during mesenchymal stem cell transformation into neurons. Chin. J. Physiol. 2008, 51, 230–246. [Google Scholar]
- Ma, L.; Cui, B.L.; Feng, X.Y.; Law, F.D.; Jiang, X.W.; Yang, L.Y.; Xie, Q.D.; Huang, T.H. biological characteristics of human umbilical cord-derived mesenchymal stem cells and their differentiation into neurocyte-like cells. Zhonghua Er Ke Za Zhi 2006, 44, 513–517. [Google Scholar]
- Matsuse, D.; Kitada, M.; Kohama, M.; Nishikawa, K.; Makinoshima, H.; Wakao, S.; Fujiyoshi, Y.; Heike, T.; Nakahata, T.; Akutsu, H.; et al. Human umbilical cord-derived mesenchymal stromal cells differentiate into functional schwann cells that sustain peripheral nerve regeneration. J. Neuropathol. Exp. Neurol. 2010, 69, 973–985. [Google Scholar] [CrossRef]
- Cho, H.; Seo, Y.K.; Jeon, S.; Yoon, H.H.; Choi, Y.K.; Park, J.K. Neural differentiation of umbilical cord mesenchymal stem cells by sub-sonic vibration. Life Sci. 2012, 90, 591–599. [Google Scholar] [CrossRef]
- Chen, K.; Wang, D.; Du, W.T.; Han, Z.B.; Ren, H.; Chi, Y.; Yang, S.G.; Zhu, D.; Bayard, F.; Han, Z.C. Human umbilical cord mesenchymal stem cells huc-mscs exert immunosuppressive activities through a pge2-dependent mechanism. Clin. Immunol. 2010, 135, 448–458. [Google Scholar] [CrossRef]
- Weiss, M.L.; Anderson, C.; Medicetty, S.; Seshareddy, K.B.; Weiss, R.J.; VanderWerff, I.; Troyer, D.; McIntosh, K.R. Immune properties of human umbilical cord wharton's jelly-derived cells. Stem Cells 2008, 26, 2865–2874. [Google Scholar] [CrossRef]
- Zhou, C.; Yang, B.; Tian, Y.; Jiao, H.; Zheng, W.; Wang, J.; Guan, F. Immunomodulatory effect of human umbilical cord wharton's jelly-derived mesenchymal stem cells on lymphocytes. Cell Immunol. 2011, 272, 33–38. [Google Scholar] [CrossRef]
- Bailo, M.; Soncini, M.; Vertua, E.; Signoroni, P.B.; Sanzone, S.; Lombardi, G.; Arienti, D.; Calamani, F.; Zatti, D.; Paul, P.; et al. Engraftment potential of human amnion and chorion cells derived from term placenta. Transplantation 2004, 78, 1439–1448. [Google Scholar]
- Weiss, M.L.; Mitchell, K.E.; Hix, J.E.; Medicetty, S.; El-Zarkouny, S.Z.; Grieger, D.; Troyer, D.L. Transplantation of porcine umbilical cord matrix cells into the rat brain. Exp. Neurol. 2003, 182, 288–299. [Google Scholar] [CrossRef]
- Liao, W.; Xie, J.; Zhong, J.; Liu, Y.; Du, L.; Zhou, B.; Xu, J.; Liu, P.; Yang, S.; Wang, J.; et al. Therapeutic effect of human umbilical cord multipotent mesenchymal stromal cells in a rat model of stroke. Transplantation 2009, 87, 350–359. [Google Scholar] [CrossRef]
- Medicetty, S.; Bledsoe, A.R.; Fahrenholtz, C.B.; Troyer, D.; Weiss, M.L. Transplantation of pig stem cells into rat brain: Proliferation during the first 8 weeks. Exp. Neurol. 2004, 190, 32–41. [Google Scholar] [CrossRef]
- Ennis, J.; Gotherstrom, C.; Le Blanc, K.; Davies, J.E. In vitro immunologic properties of human umbilical cord perivascular cells. Cytotherapy 2008, 10, 174–181. [Google Scholar] [CrossRef]
- Hatlapatka, T.; Moretti, P.; Lavrentieva, A.; Hass, R.; Marquardt, N.; Jacobs, R.; Kasper, C. Optimization of culture conditions for the expansion of umbilical cord-derived mesenchymal stem or stromal cell-like cells using xeno-free culture conditions. Tissue Eng. Part C Methods 2011, 17, 485–493. [Google Scholar] [CrossRef]
- Girdlestone, J.; Limbani, V.A.; Cutler, A.J.; Navarrete, C.V. Efficient expansion of mesenchymal stromal cells from umbilical cord under low serum conditions. Cytotherapy 2009, 11, 738–748. [Google Scholar] [CrossRef]
- Yoo, K.H.; Jang, I.K.; Lee, M.W.; Kim, H.E.; Yang, M.S.; Eom, Y.; Lee, J.E.; Kim, Y.J.; Yang, S.K.; Jung, H.L.; et al. Comparison of immunomodulatory properties of mesenchymal stem cells derived from adult human tissues. Cell Immunol. 2009, 259, 150–156. [Google Scholar] [CrossRef]
- Ramasamy, R.; Tong, C.K.; Yip, W.K.; Vellasamy, S.; Tan, B.C.; Seow, H.F. Basic fibroblast growth factor modulates cell cycle of human umbilical cord-derived mesenchymal stem cells. Cell Prolif. 2012, 45, 132–139. [Google Scholar] [CrossRef]
- Cutler, A.J.; Limbani, V.; Girdlestone, J.; Navarrete, C.V. Umbilical cord-derived mesenchymal stromal cells modulate monocyte function to suppress t cell proliferation. J. Immunol. 2010, 185, 6617–6623. [Google Scholar] [CrossRef]
- Che, N.; Li, X.; Zhou, S.; Liu, R.; Shi, D.; Lu, L.; Sun, L. Umbilical cord mesenchymal stem cells suppress b-cell proliferation and differentiation. Cell Immunol. 2012, 274, 46–53. [Google Scholar] [CrossRef]
- Kronsteiner, B.; Peterbauer-Scherb, A.; Grillari-Voglauer, R.; Redl, H.; Gabriel, C.; van, G.M.; Wolbank, S. Human mesenchymal stem cells and renal tubular epithelial cells differentially influence monocyte-derived dendritic cell differentiation and maturation. Cell Immunol. 2011, 267, 30–38. [Google Scholar] [CrossRef]
- Jiang, X.X.; Zhang, Y.; Liu, B.; Zhang, S.X.; Wu, Y.; Yu, X.D.; Mao, N. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 2005, 105, 4120–4126. [Google Scholar] [CrossRef]
- Nauta, A.J.; Kruisselbrink, A.B.; Lurvink, E.; Willemze, R.; Fibbe, W.E. Mesenchymal stem cells inhibit generation and function of both cd34+-derived and monocyte-derived dendritic cells. J. Immunol. 2006, 177, 2080–2087. [Google Scholar]
- Ramasamy, R.; Fazekasova, H.; Lam, E.W.; Soeiro, I.; Lombardi, G.; Dazzi, F. Mesenchymal stem cells inhibit dendritic cell differentiation and function by preventing entry into the cell cycle. Transplantation 2007, 83, 71–76. [Google Scholar] [CrossRef]
- van den Berk, L.C.; Roelofs, H.; Huijs, T.; Siebers-Vermeulen, K.G.; Raymakers, R.A.; Kogler, G.; Figdor, C.G.; Torensma, R. Cord blood mesenchymal stem cells propel human dendritic cells to an intermediate maturation state and boost interleukin-12 production by mature dendritic cells. Immunology 2009, 128, 564–572. [Google Scholar] [CrossRef]
- Kermani, A.J.; Fathi, F.; Mowla, S.J. Characterization and genetic manipulation of human umbilical cord vein mesenchymal stem cells: Potential application in cell-based gene therapy. Rejuvenation Res. 2008, 11, 379–386. [Google Scholar] [CrossRef]
- Liang, X.J.; Chen, X.J.; Yang, D.H.; Huang, S.M.; Sun, G.D.; Chen, Y.P. Differentiation of human umbilical cord mesenchymal stem cells into hepatocyte-like cells by htert gene transfection in vitro. Cell. Biol. Int. 2012, 36, 215–221. [Google Scholar] [CrossRef]
- The U.S. National Institutes of Health and the National Library of Medicine, Clinicaltrials.Gov: A database of clinical studies of human participants. Available online: http://clinicaltrials.gov/ct2/home (accessed on 22 August 2012),.
- The U.S. National Institutes of Health and the National Library of Medicine, Clinicaltrials.Gov identifier: Nct01291329. Available online: http://clinicaltrials.gov/ct2/home (accessed on 22 August 2012),.
- Breymann, C.; Schmidt, D.; Hoerstrup, S.P. Umbilical cord cells as a source of cardiovascular tissue engineering. Stem. Cell. Rev. 2006, 2, 87–92. [Google Scholar] [CrossRef]
- Hua, J.; Qiu, P.; Zhu, H.; Cao, H.; Wang, F.; Li, W. Multipotent mesenchymal stem cells (mscs) from human umbilical cord: Potential differentiation of germ cells. Afr. J. Biochem. Res. 2011, 5, 113–123. [Google Scholar]
- Hilfiker, A.; Kasper, C.; Hass, R.; Haverich, A. Mesenchymal stem cells and progenitor cells in connective tissue engineering and regenerative medicine: Is there a future for transplantation? Langenbecks Arch. Surg. 2011, 396, 489–497. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lindenmair, A.; Hatlapatka, T.; Kollwig, G.; Hennerbichler, S.; Gabriel, C.; Wolbank, S.; Redl, H.; Kasper, C. Mesenchymal Stem or Stromal Cells from Amnion and Umbilical Cord Tissue and Their Potential for Clinical Applications. Cells 2012, 1, 1061-1088. https://doi.org/10.3390/cells1041061
Lindenmair A, Hatlapatka T, Kollwig G, Hennerbichler S, Gabriel C, Wolbank S, Redl H, Kasper C. Mesenchymal Stem or Stromal Cells from Amnion and Umbilical Cord Tissue and Their Potential for Clinical Applications. Cells. 2012; 1(4):1061-1088. https://doi.org/10.3390/cells1041061
Chicago/Turabian StyleLindenmair, Andrea, Tim Hatlapatka, Gregor Kollwig, Simone Hennerbichler, Christian Gabriel, Susanne Wolbank, Heinz Redl, and Cornelia Kasper. 2012. "Mesenchymal Stem or Stromal Cells from Amnion and Umbilical Cord Tissue and Their Potential for Clinical Applications" Cells 1, no. 4: 1061-1088. https://doi.org/10.3390/cells1041061
APA StyleLindenmair, A., Hatlapatka, T., Kollwig, G., Hennerbichler, S., Gabriel, C., Wolbank, S., Redl, H., & Kasper, C. (2012). Mesenchymal Stem or Stromal Cells from Amnion and Umbilical Cord Tissue and Their Potential for Clinical Applications. Cells, 1(4), 1061-1088. https://doi.org/10.3390/cells1041061






