3.1. Assessment of Sweet Basil Development
Basil grown in our experiment using organic farming methods developed well, both in the open field and under the foil tunnel. Both cultivation variants, during the period of two months (from the end of June to the end of August), enabled four cuts to be carried out (
Table 2). Earlier trials showed that only two–three cuts are possible during one vegetation period [
9]. The productivity of plants from our experiment was comparable to or even higher than those cultivated in Greece, where two–four harvests were performed, depending on the cultivation system [
5]. In our study, plants grown in the foil tunnel were taller and produced a greater number of shoots per plant than those in the open field (
Table 2). Thus, they were characterized by a higher cumulative mass of herb (
Table 3).
The fast regrowth of basil herb observed in our experiment could be associated with its high light requirements, and in particular the radiation intensity and length of the day. In the mentioned period, from the end of June until August, in the conditions of central Europe, the highest monthly sum of radiation is noted, and the day reaches lengths of up to 18 hours. It was proven that such a long day is one of the factors accelerating the development of basil [
9,
36]. Another factor important in basil cultivation is temperature. It is well known that the species is susceptible not only to frost but also to cold (from 2 to 0 °C). According to some chamber experiments, the fastest growth of basil was noted at 27 °C with seven harvests performed [
9]. It seems that intense cutting of the herb stimulates plants to regrow and enables quick vegetative shoot production, which was visible especially for plants grown under the foil tunnel. These plants, with a temperature about 4–6 °C higher than in the open field, produced a distinctly higher mass of herb (
Table 3).
3.2. Chemical Analysis of Raw Materials
The total content of essential oil in fresh (0.02–0.17 g∙100 g
−1) and dry (0.21–1.16 g∙100 g
−1) basil varied depending on the term of harvest. The highest content, irrespective of cultivation method, was observed in the herb from the last cut, and the lowest was observed from the first cut (
Table 4 and
Table 5). These results are in agreement with the previously published results concerning the accumulation of essential oil in fresh basil collected successively from plants cultivated in a greenhouse in Greece [
5]. According to Lemberkovics et al. [
37], the essential oil content increases during basil ontogenesis. With regard to cultivation method, differences in the content of essential oil between plants grown in the open field and under the foil tunnel were observed especially concerning air-dried leaves, in which the content was significantly higher in the plants collected from the tunnel, especially in the third and fourth cuts (
Table 6). The results obtained in this study indicate that the accumulation of essential oil in basil depends on both plant developmental phase and climatic factors, among which temperature seems to be particularly important.
In total, 24 substances were detected in the essential oil obtained from the fresh herb, and these formed 87.51%–97.68% of the identified fractions (
Table 4). Among these, the oxygenated monoterpene fraction formed up to 84.85%, with linalool as a dominant compound. This monoterpene alcohol exhibits antihyperalgesic and antinociceptive activity [
38]. Along with methyl chavicol, it is considered the most important sweet basil essential oil component, forming up to 90% of the oil. Both compounds are also related to the fine basil aroma [
9]. In our study, linalool content increased in tandem with the subsequent four herbal cuts. In the case of plants cultivated in the open field, the content grew from 53.52% (first cut) to 59.50% (fourth cut), and, in thecase of plants cultivated in the foil tunnel, the content grew from 48.50% (first cut) to 54.01% (fourth cut) (
Table 4). The increase in linalool content during successive harvests of the fresh herb was described earlier by Tsasi et al. [
5]. However, it was observed for one out of four tested varieties of sweet basil. This phenomenon may, therefore, be related to the specific genotype. Other oxygenated monoterpene compounds, identified in our experiment, such as
trans-methyl cinnamate and 1,8-cineole, were present in basil essential oil in much lower amounts (from 6.46% to 17.17% and from 5.28% to 13.39%, respectively). Methyl chavicol was also observed only in small quantities (from 0.91% to 1.54%). There was no relationship between term of harvest and the content of these compounds in the analyzed essential oils. According to Lemberkovics et al. [
37], during basil ontogenesis, the composition of essential oil does not fluctuate as clearly as its content. In turn, Klimánková et al. [
39] claimed that no distinct differences are observed in basil volatile content and their composition between plants under organic and conventional cultivation. Thus, it seems that the composition is related to the genotype, rather than to external factors.
In recent years, much attention was paid to the phenolic profile of aromatic plants. These substances, revealing wide pro-health or even medicinal activity, play an important role for the plants themselves. It was proven that phenolics, especially flavonoids, protect DNA against UV-B radiation [
40] and inactivate free radicals responsible for oxidative stress [
41]. They can have the characteristics of signaling molecules affecting the expression of genes responsible for plant defense reactions against stress (e.g., salicylic acid). Some polyphenols show allelopathic activity (e.g., chlorogenic,
p-coumaric, and vanillic acids), inhibit germination of seeds, or limit the development of other species. They may also play an important role in the interaction between the plant and the pathogen [
42].
In our experiment, the total flavonoid and phenolic acid contents were determined. For both cultivation methods, the content of flavonoids was the highest in the herb from the last harvest, whereas the amount of phenolic acids was the highest at the first cut. As for flavonoids, their content increased from the first to the last harvest and was higher in plants grown under the foil tunnel (
Table 5). According to Mattila et al. [
43], the production of flavonols in plant tissues takes place during daytime and is enhanced by light intensity. This, in turn, is combined with their function in plants as quenchers of singlet oxygen. In experiments on rye, it was also observed that, in the early stage of plant development, a protective role against UV-B is played by some phenolic acids. However, during plant development and acclimation to stress, they are successively replaced in this function by flavonoids [
44]. This may explain the pattern of accumulation of phenolics as protective substances in plants from our experiment. It was also described that some flavonoids influence the movement of auxin and, consequently, regulate the development of some organs or even the whole plant [
45]. Thus, the increase in the content of flavonoids in basil may be related to its quick regrowth.
It is worth mentioning that the content of phenolics in basil depends on other factors, including the level of nutrient compounds in the soil during development [
46], presowing treatment of its seeds [
47], or light quality during cultivation [
48]. Other type of stress, including water shortage, may also influence the accumulation of phenolics and chlorophyll a and b in basil leaves [
15,
49]. In our experiment, similar to flavonoids, the content of chlorophyll a and b increased significantly from the first to the last term of harvest. However, their amount was distinctly higher in the herbs collected from plants grown under the foil tunnel than in the open field (
Table 5). The content of chlorophyll a and b depends on plant species, light regime and its quality, and mineral availability during growth [
50]. This is taken into consideration when assessing the overall plant condition and is especially important for production of plants consumed in fresh form. According to Kopsellet et al. [
51], the accumulation of chlorophyll a and b in fresh basil depends on both the genotype used and the method of cultivation, wherein cultivation in the greenhouse, with sphagnum peat moss as a medium, resulted in a lower content of pigments in the herb in comparison to open-field cultivation. In our experiments carried out in accordance with an organic farming system, where the same soil was used in both cultivation methods (open field and foil tunnel), a higher content of chlorophyll a and b was detected in plants cultivated under the foil tunnel (
Table 5).
3.3. Sensory Analysis of Fresh Herb
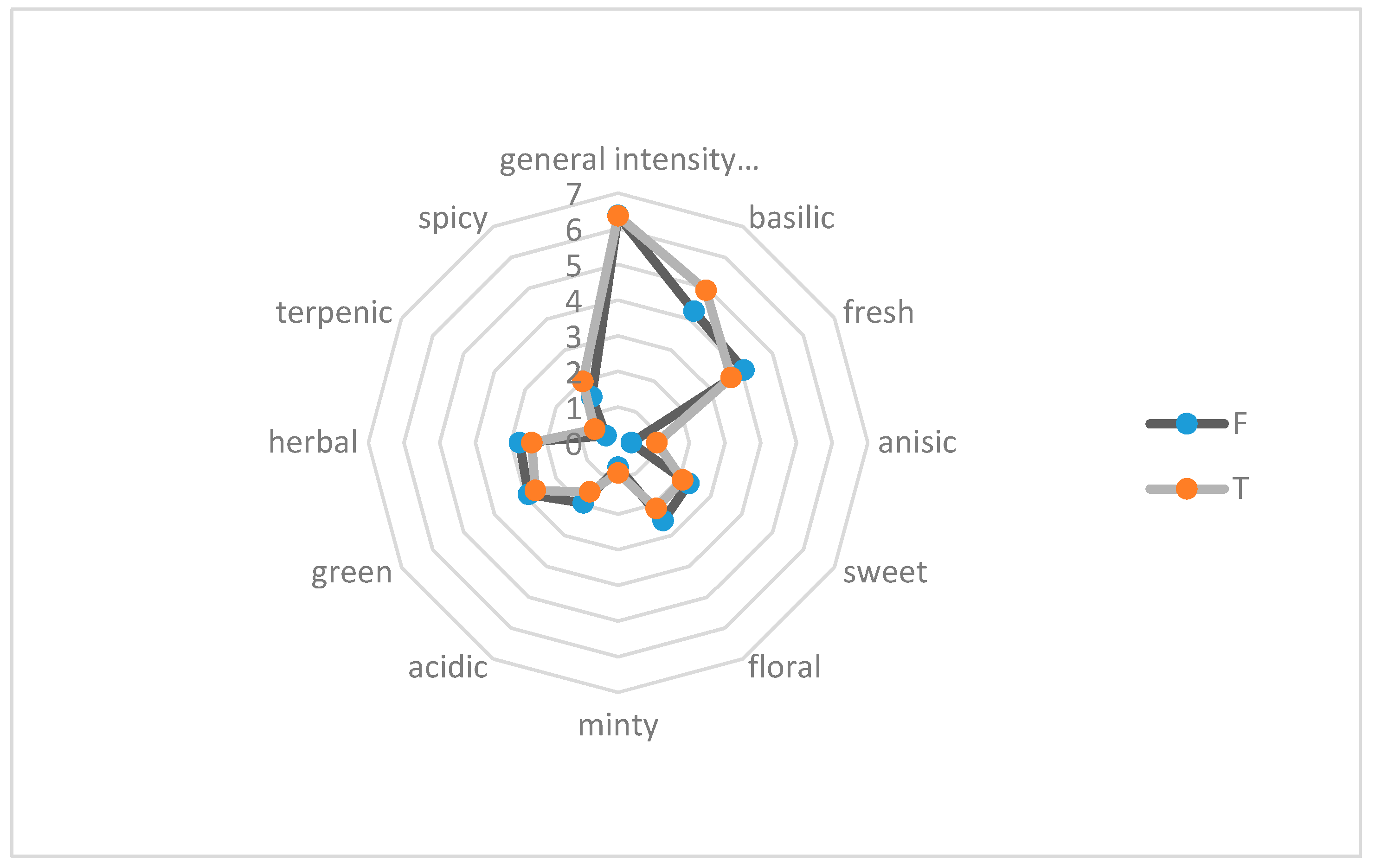
The genotype used in our experiment was represented by a linalool chemotype (
Table 4), which influenced the sensory profile of the fresh herb. According to D’Antuono et al. (2007), among the most valuable components of sweet basil essential oil, i.e., linalool, eugenol, and 1,8-cineole, only linalool generated positive acceptance of panelists assessing their odor value. However, the balance between the three compounds plays an important role in the appreciation of the odor of basil essential oil. In our experiment, the general intensity of fresh basil herbs collected from both open field and tunnel cultivation was similar. The rate of the intensity of perception for most analyzed odor notes, i.e., fresh, sweet, floral, minty, acidic, green, herbal, terpenic, and spicy, was also similar. The distinct differences between herbs obtained from the open air and foil tunnel cultivation concerned basilic, anisic, and spicy odor notes, which were higher for the herb collected from plants cultivated under the foil tunnel (
Figure 1,
Table 6). Of the 14 taste notes, only two differentiated fresh basil, i.e., basilic and spicy. The intensity of their perception was also higher for the raw material collected from plants grown under the foil tunnel (
Figure 2,
Table 7). This was probably connected with the higher content of the essential oil in this raw material.
3.4. Microbiological Analysis of Fresh and Air-Dried Herb
The microbiological quality of aromatic herbs is determined by the hygienic status and environmental conditions of the region they originate from and where they are pre-treated. The method of cultivation may also influence their microbiological quality, especially when natural fertilizers based on animal excrements are used. Therefore, contaminations with undesirable microflora may occur at each stage of their production process, i.e., during cultivation, harvest, and processing, as well as during storage, distribution, or use by consumers [
52].
Table 8 presents the results of microbiological contamination of fresh basil herb and
Table 9 presents the results of the air-dried herb.
Salmonella and coagulase-positive
Staphylococcus were not found in any of the samples.
E. coli was present in samples of fresh basil cultivated both in the open field and foil tunnel. However, their number did not exceed 90 cells in 1 g of herb.
E. coli was not found in the dried basil. The samples of herb collected from plants grown in the open field were characterized by a higher number of mesophilic aerobic bacteria (TPC) of 6.11 log
10 colony-forming unit (CFU)∙g
−1, compared to those from the foil tunnel (5.12 log
10 CFU∙g
−1). The content of mold and yeast in 1 g of fresh basil exceeded 4 log
10 CFU. However, a smaller number of fungi was determined in dried samples, and it did not exceed 3.5 log
10 CFU∙g
−1. The contamination of sulfite-reducing
Clostridium was very low and did not exceed 10
2 CFU∙g
−1. The content of aerobic spore-forming bacteria was 4.37 to 4.87 log
10 CFU∙g
−1 for fresh samples and significantly lower (3.27–4.04 log
10 CFU∙g
−1) for dried ones. In the case of basil cultivated in the open field, a high content of spores (over 56%) consisted of amylolytic bacteria. However, in dried samples, the content decreased to over 11%. The samples of fresh and air-dried basil originating from the foil tunnel contained similar numbers of these bacteria.
European Union (EU) legislation has no definite microbiological standards for herbs [
53]. According to the Codex Code of Hygienic practice [
54], which states that herbs should not contain any toxin-producing elements, all samples of the analyzed basil samples met the requirements. Specifically,
Salmonella spp. should be completely absent in 25 g of the sample [
55], and the maximum levels of
E. coli should not exceed 10
2 CFU∙g
−1 [
56]. The level of mold contamination of the basil also met the requirements of the EU commission [
56]. According to Stankovic et al. [
57], the presence of basil herbs next to black pepper (ground and whole corns) and dill available on sale in healthy food stores and supermarkets, is most often microbiologically unacceptable. Most spices are significantly contaminated with spore-forming bacteria of the genus
Bacillus [
53,
58]. We showed that the method of cultivation did not influence the content of spore-forming aerobic and anaerobic (sulfite-reducing) bacteria in basil. The factor significantly affecting the majority of microbiological parameters was drying. The air-dried raw materials were free from
E. coli, which means that the drying method used and proper storage improved their microbial quality. This is important, especially in the context that some of the foodborne pathogenic strains of
E. coli (EIEC, ETEC, APEC, STEC, and others) indicate a potential health risk associated with contaminated products [
59]. Low water activity (a
w) of the air-dried herb is a factor preventing the development of pathogenic or spoilage microflora, both bacteria and mold. In turn, mold growth carries the risk of mycotoxin contamination, which makes the post-harvest treatment of herbs very important.