Winter Wheat Grain Quality, Zinc and Iron Concentration Affected by a Combined Foliar Spray of Zinc and Iron Fertilizers
Abstract
1. Introduction
2. Materials and Methods
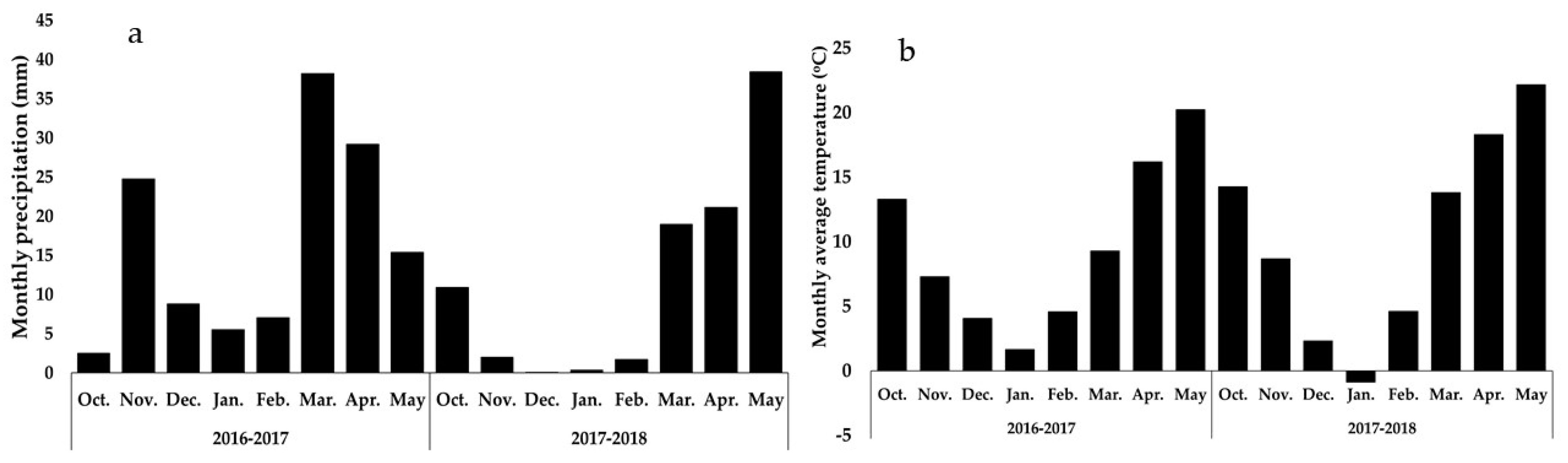
2.1. Field Location
2.2. Experimental Design and Treatments
2.3. Sampling Procedures and Analysis
2.4. Statistical Analysis
3. Results
4. Discussion
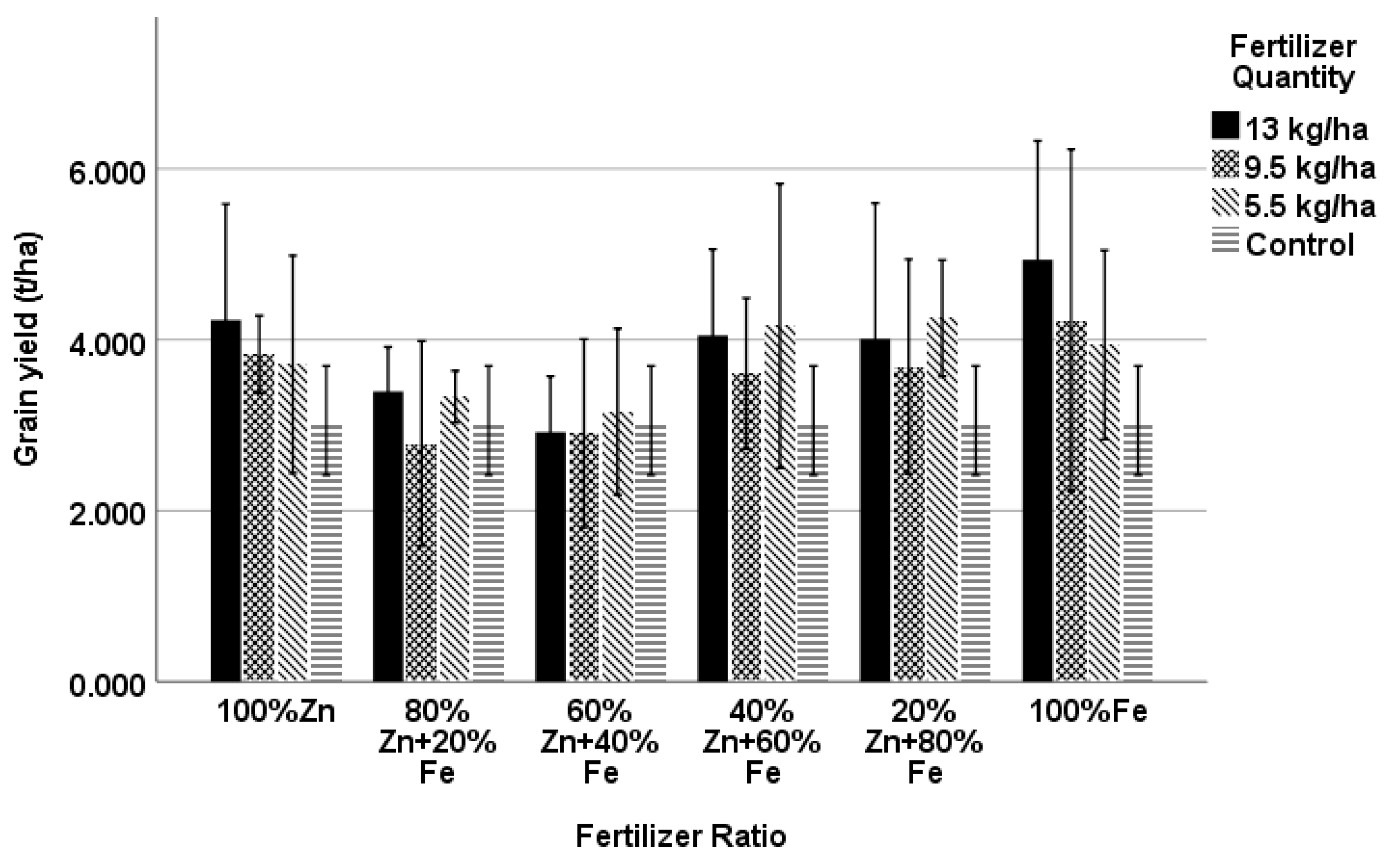
4.1. Yield and Yield Components
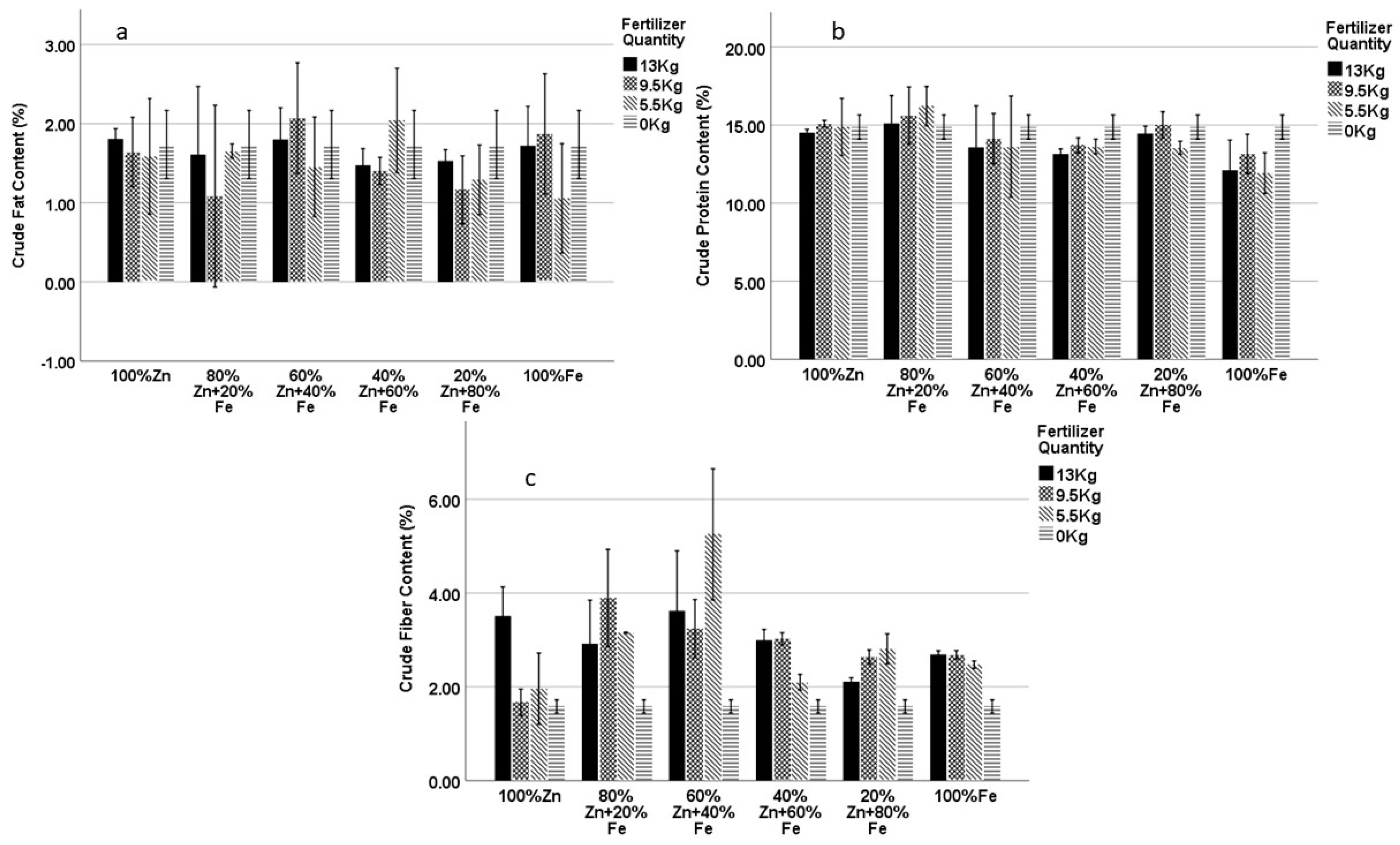
4.2. Crude Fat
4.3. Crude Fiber
4.4. Crude Protein
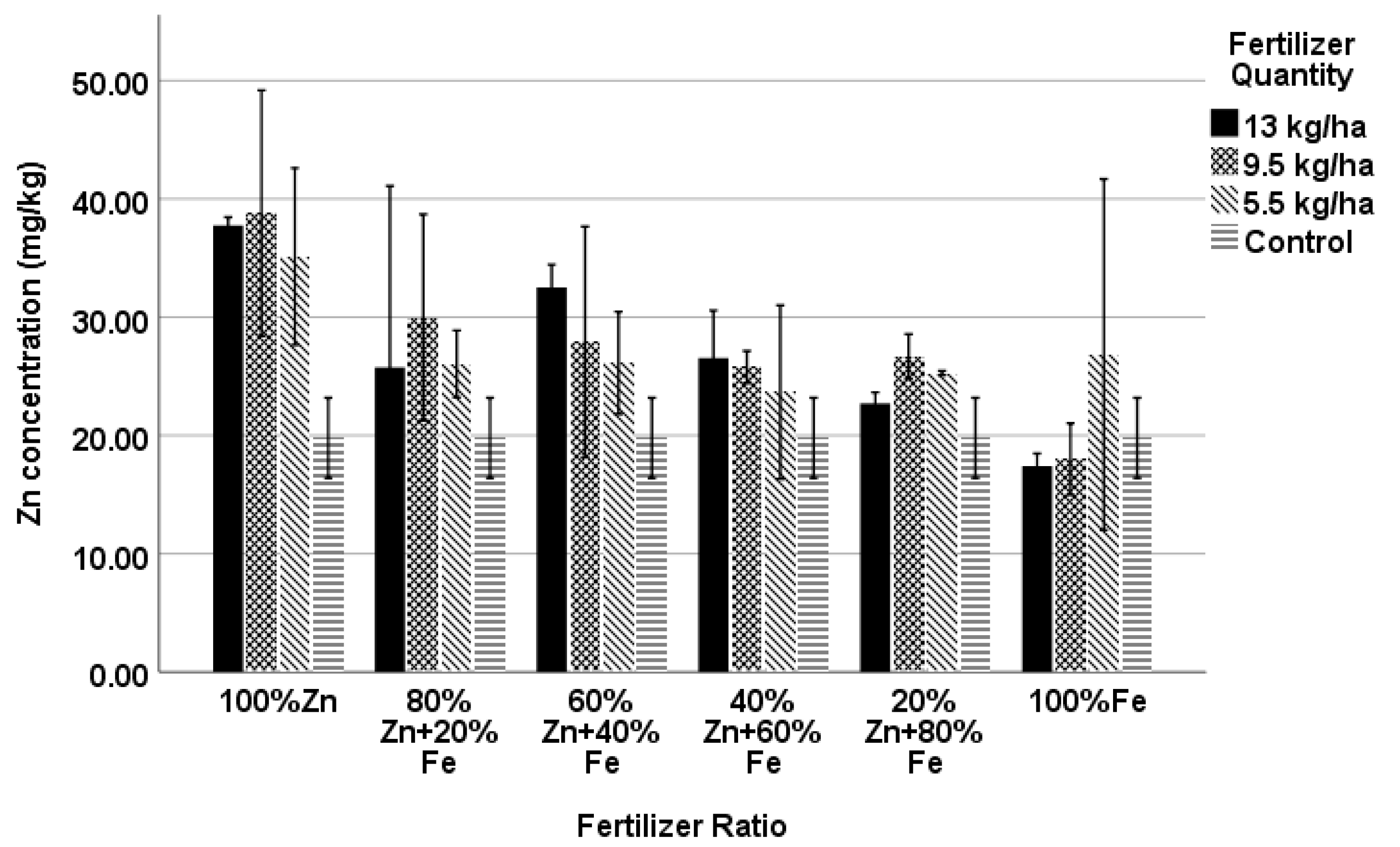
4.5. Zn Concentration
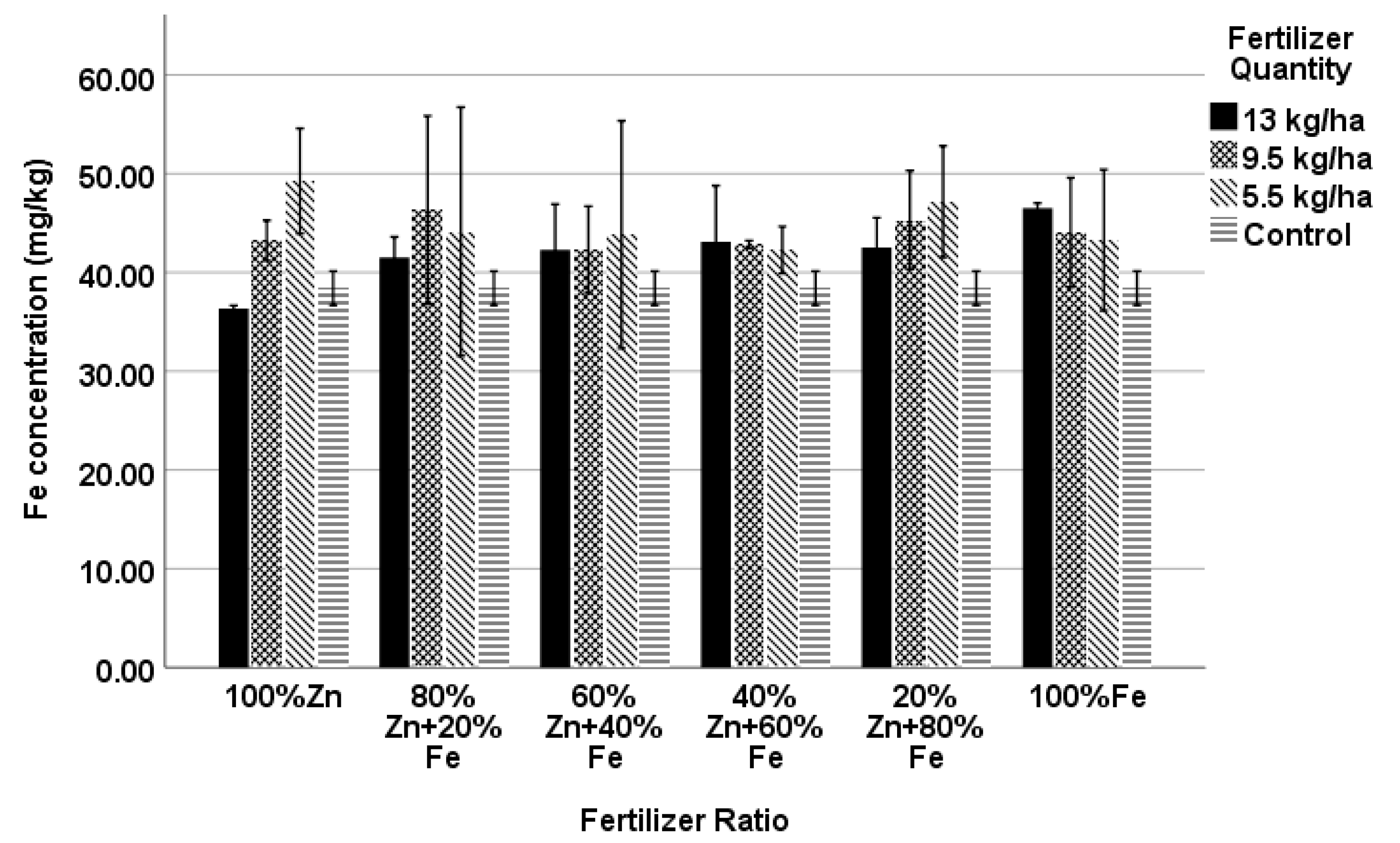
4.6. Fe Concentration
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- FAO. FAO Statistical Pocket Book: World Food and Agriculture; FAO: Rome, Italy, 2015. [Google Scholar]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671. [Google Scholar] [CrossRef] [PubMed]
- World Bank. World Bank Country and Lending Groups. Available online: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups (accessed on 17 April 2019).
- Graham, R.D.; Welch, R.M.; Saunders, D.A.; Ortiz-Monasterio, I.; Bouis, H.E.; Bonierbale, M.; De Haan, S.; Burgos, G.; Thiele, G.; Liria, R. Nutritious subsistence food systems. Adv. Agron. 2007, 92, 1–74. [Google Scholar]
- Who, J.; Consultation, F.E. Diet, Nutrition and the Prevention of Chronic Diseases; WHO Technicl Report Series, No.916; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Müller, O.; Krawinkel, M. Malnutrition and health in developing countries. CMAJ 2005, 173, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Welch, R.M.; Graham, R.D. Breeding for micronutrients in staple food crops from a human nutrition perspective. J. Exp. Bot. 2004, 55, 353–364. [Google Scholar] [CrossRef]
- Singh, B.R.; Timsina, Y.N.; Lind, O.C.; Cagno, S.; Janssens, K. Zinc and iron concentration as affected by nitrogen fertilization and their localization in wheat grain. Front. Plant Sci. 2018, 9, 307. [Google Scholar] [CrossRef]
- Hotz, C.; Brown, K.H. Assessment of the Risk of Zinc Deficiency in Populations and Options for Its Control. Food Nutr. Bull. 2004, 25, S99–S203. [Google Scholar]
- WHO. World Health Statistics; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Murray, C.J.; Lopez, A.D. Measuring the global burden of disease. N. Engl. J. Med. 2013, 369, 448–457. [Google Scholar] [CrossRef]
- WHO. World Health Statistics; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Balk, J.; Connorton, J.; Wan, Y.; Lovegrove, A.; Moore, K.; Uauy, C.; Sharp, P.; Shewry, P. Improving wheat as a source of iron and zinc for global nutrition. Nutr. Bull. 2019, 44, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.; Steer, T.; Maplethorpe, N.; Cox, L.; Meadows, S.; Nicholson, S.; Page, P.; Swan, G. National Diet and Nutrition Survey: Results from Years 7 and 8 (Combined) of the Rolling Programme (2014/2015–2015/2016); Food Standards Agency: London, UK, 2018.
- Branca, F.; Ferrari, M. Impact of micronutrient deficiencies on growth: The stunting syndrome. Ann. Nutr. Metab. 2002, 46, 8–17. [Google Scholar] [CrossRef]
- Golden, M.H. The nature of nutritional deficiency in relation to growth failure and poverty. Acta Paediatr. 1991, 80, 95–110. [Google Scholar] [CrossRef]
- Welch, R.M. Micronutrients, Agriculture and Nutrition; Linkages for Improved Health and Well Being. In Perspectives on the Micronutrient Nutrition of Crops; Scientific Publishers: Jodhpur, India, 2001; pp. 247–289. [Google Scholar]
- Cakmak, I.; Kutman, U.B. Agronomic biofortification of cereals with zinc: A review. Eur. J. Soil Sci. 2018, 69, 172–180. [Google Scholar] [CrossRef]
- Garvin, D.F.; Welch, R.M.; Finley, J.W. Historical shifts in the seed mineral micronutrient concentration of US hard red winter wheat germplasm. J. Sci. Food Agric. 2006, 86, 2213–2220. [Google Scholar] [CrossRef]
- Shewry, P.R.; Pellny, T.K.; Lovegrove, A. Is modern wheat bad for health. Nat. Plants 2016, 2, 16097. [Google Scholar] [CrossRef]
- Cakmak, I.; Kalaycı, M.; Ekiz, H.; Braun, H.; Kılınç, Y.; Yılmaz, A. Zinc deficiency as a practical problem in plant and human nutrition in Turkey: A NATO-science for stability project. Field Crops Res. 1999, 60, 175–188. [Google Scholar] [CrossRef]
- Cakmak, I.; Pfeiffer, W.; McClafferty, B. Review: Biofortification of Durum Wheat with Zinc and Iron. Cereal Chem. 2010, 87, 10–20. [Google Scholar] [CrossRef]
- Cakmak, I. Enrichment of cereal grains with zinc: Agronomic or genetic biofortification? Plant Soil 2008, 302, 1–17. [Google Scholar] [CrossRef]
- Nair, M.K.; Augustine, L.F.; Konapur, A. Food-Based Interventions to Modify Diet Quality and Diversity to Address Multiple Micronutrient Deficiency. Front. Public Health 2016, 3. [Google Scholar] [CrossRef]
- Nestel, P.; Bouis, H.E.; Meenakshi, J.; Pfeiffer, W. Biofortification of staple food crops. J. Nutr. 2006, 136, 1064–1067. [Google Scholar] [CrossRef]
- Rengel, Z.; Batten, G.; Crowley, D.D. Agronomic approaches for improving the micronutrient density in edible portions of field crops. Field Crops Res. 1999, 60, 27–40. [Google Scholar] [CrossRef]
- Cakmak, I.; Kalayci, M.; Kaya, Y.; Torun, A.A.; Aydin, N.; Wang, Y.; Arisoy, Z.; Erdem, H.; Yazici, A.; Gokmen, O.; et al. Biofortification and localization of zinc in wheat grain. J. Agric. Food Chem. 2010, 58, 9092–9102. [Google Scholar] [CrossRef]
- Zhao, F.; Su, Y.; Dunham, S.; Rakszegi, M.; Bedo, Z.; McGrath, S.; Shewry, P. Variation in mineral micronutrient concentrations in grain of wheat lines of diverse origin. J. Cereal Sci. 2009, 49, 290–295. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R. Biofortifying crops with essential mineral elements. Trends Plant Sci. 2005, 10, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Kutman, U.B.; Yildiz, B.; Cakmak, I. Improved nitrogen status enhances zinc and iron concentrations both in the whole grain and the endosperm fraction of wheat. J. Cereal Sci. 2011, 53, 118–125. [Google Scholar] [CrossRef]
- Barunawati, N.; Hettwer Giehl, R.F.; Bauer, B.; Von Wirén, N. The influence of inorganic nitrogen fertilizer forms on micronutrient retranslocation and accumulation in grains of winter wheat. Front. Plant Sci. 2013, 4, 320. [Google Scholar] [CrossRef] [PubMed]
- Kutman, U.B.; Yildiz, B.; Cakmak, I. Effect of nitrogen on uptake, remobilization and partitioning of zinc and iron throughout the development of durum wheat. Plant Soil 2011, 342, 149–164. [Google Scholar] [CrossRef]
- Erenoglu, E.B.; Kutman, U.B.; Ceylan, Y.; Yildiz, B.; Cakmak, I. Improved nitrogen nutrition enhances root uptake, root-to-shoot translocation and remobilization of zinc (65Zn) in wheat. New Phytol. 2011, 189, 438–448. [Google Scholar] [CrossRef]
- Kutman, U.B.; Kutman, B.Y.; Ceylan, Y.; Ova, E.A.; Cakmak, I. Contributions of root uptake and remobilization to grain zinc accumulation in wheat depending on post-anthesis zinc availability and nitrogen nutrition. Plant Soil 2012, 361, 177–187. [Google Scholar] [CrossRef]
- Persson, D.; de Bang, T.; Pedas, P.; Kutman, U.; Cakmak, I.; Andersen, B.; Finnie, C.; K Schjoerring, J.; Husted, S. Molecular speciation and tissue compartmentation of zinc in durum wheat grains with contrasting nutritional status. New Phytol. 2016, 211, 1255–1265. [Google Scholar] [CrossRef]
- Shi, R.; Zhang, Y.; Chen, X.; Sun, Q.; Zhang, F.; Römheld, V.; Zou, C. Influence of long-term nitrogen fertilization on micronutrient density in grain of winter wheat (Triticum aestivum L.). J. Cereal Sci. 2010, 51, 165–170. [Google Scholar] [CrossRef]
- Astolfi, S.; Cesco, S.; Zuchi, S.; Neumann, G.; Roemheld, V. Sulphur starvation reduces phytosiderophores release by Fe-deficient barley plants. Soil Sci. Plant Nutr. 2006, 52, 43–48. [Google Scholar] [CrossRef]
- Zuchi, S.; Cesco, S.; Astolfi, S. High S supply improves Fe accumulation in durum wheat plants grown under Fe limitation. Environ. Exp. Botany 2012, 77, 25–32. [Google Scholar] [CrossRef]
- Celletti, S.; Paolacci, A.R.; Mimmo, T.; Pii, Y.; Cesco, S.; Ciaffi, M.; Astolfi, S. The effect of excess sulfate supply on iron accumulation in three graminaceous plants at the early vegetative phase. Environ. Exp. Bot. 2016, 128, 31–38. [Google Scholar] [CrossRef]
- Astolfi, S.; Pii, Y.; Terzano, R.; Mimmo, T.; Celletti, S.; Allegretta, I.; Lafiandra, D.; Cesco, S. Does Fe accumulation in durum wheat seeds benefit from improved whole-plant sulfur nutrition? J. Cereal Sci. 2018, 83, 74–82. [Google Scholar] [CrossRef]
- Celletti, S.; Pii, Y.; Mimmo, T.; Cesco, S.; Astolfi, S. The characterization of the adaptive responses of durum wheat to different Fe availability highlights an optimum Fe requirement threshold. Plant Physiol. Biochem. 2016, 109, 300–307. [Google Scholar] [CrossRef]
- Ciaffi, M.; Paolacci, A.R.; Celletti, S.; Catarcione, G.; Kopriva, S.; Astolfi, S. Transcriptional and physiological changes in the S assimilation pathway due to single or combined S and Fe deprivation in durum wheat (Triticum durum L.) seedlings. J. Exp. Bot. 2013, 64, 1663–1675. [Google Scholar] [CrossRef]
- Zuchi, S.; Watanabe, M.; Hubberten, H.-M.; Bromke, M.; Osorio, S.; Fernie, A.R.; Celletti, S.; Paolacci, A.R.; Catarcione, G.; Ciaffi, M. The interplay between sulfur and iron nutrition in tomato. Plant Physiol. 2015, 169, 2624–2639. [Google Scholar] [CrossRef]
- Borrill, P.; Connorton, J.M.; Balk, J.; Miller, A.J.; Sanders, D.; Uauy, C. Biofortification of wheat grain with iron and zinc: Integrating novel genomic resources and knowledge from model crops. Front. Plant Sci. 2014, 5, 53. [Google Scholar] [CrossRef]
- de Valença, A.W.; Bake, A.; Brouwer, I.D.; Giller, K.E. Agronomic biofortification of crops to fight hidden hunger in sub-Saharan Africa. Glob. Food Secur. 2017, 12, 8–14. [Google Scholar] [CrossRef]
- Frossard, E.; Bucher, M.; Mächler, F.; Mozafar, A.; Hurrell, R. Potential for increasing the content and bioavailability of Fe, Zn and Ca in plants for human nutrition. J. Sci. Food Agric. 2000, 80, 861–879. [Google Scholar] [CrossRef]
- Welch, R.M.; Graham, R.D. A new paradigm for world agriculture: Meeting human needs: Productive, sustainable, nutritious. Field Crops Res. 1999, 60, 1–10. [Google Scholar] [CrossRef]
- Zou, C.Q.; Zhang, Y.Q.; Rashid, A.; Ram, H.; Savasli, E.; Arisoy, R.Z.; Ortiz-Monasterio, I.; Simunji, S.; Wang, Z.H.; Sohu, V.; et al. Biofortification of wheat with zinc through zinc fertilization in seven countries. Plant Soil 2012, 361, 119–130. [Google Scholar] [CrossRef]
- Peleg, Z.; Saranga, Y.; Yazici, A.; Fahima, T.; Ozturk, L.; Cakmak, I. Grain zinc, iron and protein concentrations and zinc-efficiency in wild emmer wheat under contrasting irrigation regimes. Plant Soil 2007, 306, 57–67. [Google Scholar] [CrossRef]
- Li, M.; Wang, S.; Tian, X.; Li, S.; Chen, Y.; Jia, Z.; Liu, K.; Zhao, A. Zinc and iron concentrations in grain milling fractions through combined foliar applications of Zn and macronutrients. Field Crops Res. 2016, 187, 135–141. [Google Scholar] [CrossRef]
- Pataco, I.M.; Lidon, F.C.; Ramos, I.; Oliveira, K.; Guerra, M.; Pessoa, M.F.; Carvalho, M.L.; Ramalho, J.C.; Leitão, A.E.; Santos, J.P.; et al. Biofortification of durum wheat (Triticum turgidum L. ssp. durum (Desf.) Husnot) grains with nutrients. J. Plant Int. 2017, 12, 39–50. [Google Scholar] [CrossRef]
- Chattha, M.U.; Hassan, M.U.; Khan, I.; Chattha, M.B.; Mahmood, A.; Chattha, M.U.; Nawaz, M.; Subhani, M.N.; Kharal, M.; Khan, S. Biofortification of Wheat Cultivars to Combat Zinc Deficiency. Front. Plant Sci. 2017, 8, 281. [Google Scholar] [CrossRef]
- Buchholz, D.D.; Brown, J.R.; Garret, J.; Hanson, R.; Wheaton, H. Soil Test Interpretations and Recommendations Handbook; Division of Plant Sciences, University of Missouri-College of Agriculture: Columbia, MO, USA, 2004. [Google Scholar]
- Zhang, Y.; Shi, R.; Rezaul, K.M.; Zhang, F.; Zou, C. Iron and Zinc Concentrations in Grain and Flour of Winter Wheat As Affected by Foliar Application. J. Agric. Food Chem. 2010, 58, 12268–12274. [Google Scholar] [CrossRef]
- Arlington, V. Official Methods of Analysis of the Association of Official Analytical Chemists; Association of Official Analytical Chemists: Washington, DC, USA, 1921; Volume 2. [Google Scholar]
- ISO 16634-1. Food Products-Determination of the Total Nitrogen Content by Combustion According to the Dumas Principle and Calculation of the Crude Protein Content: Oilseeds Animal Feeding Stuffs; ISO: Geneva, Switzerland, 2008. [Google Scholar]
- Komarek, A.; Manson, H.; Thiex, N. Crude Fiber Determinations Using the ANKOM System; ANKOM Technology Corporation Publication: Macedon, NY, USA, 1996. [Google Scholar]
- Thiex, N.J.; Anderson, S.; Gildemeister, B. Crude fat, diethyl ether extraction, in feed, cereal grain, and forage (Randall/Soxtec/submersion method): Collaborative study. J. AOAC Int. 2003, 86, 888–898. [Google Scholar]
- Isaac, R.A.; Kerber, J.D. Atomic Absorption and Flame Photometry: Techniques and Uses in Soil, Plant, and Water Analysis. In Instrumental Methods for Analysis of Soils and Plant Tissue; Instrumentalmet; Soil Science Society of America: Madison, WI, USA, 1971; pp. 17–37. [Google Scholar]
- Cakmak, I. Possible roles of zinc in protecting plant cells from damage by reactive oxygen species. New Phytol. 2000, 146, 185–205. [Google Scholar] [CrossRef]
- Welch, R.M.; Allaway, W.H.; House, W.A.; Kubota, J. Geographic Distribution of Trace Element Problems. In Micronutrients in Agriculture; Micronutrientsi2; Soil Science Society of America, Inc.: Madison, WI, USA, 1991; pp. 31–57. [Google Scholar]
- Zhang, Y.-Q.; Sun, Y.-X.; Ye, Y.-L.; Karim, M.R.; Xue, Y.-F.; Yan, P.; Meng, Q.-F.; Cui, Z.-L.; Cakmak, I.; Zhang, F.-S. Zinc biofortification of wheat through fertilizer applications in different locations of China. Field Crops Res. 2012, 125, 1–7. [Google Scholar] [CrossRef]
- Gebbing, T.; Schnyder, H. Pre-anthesis reserve utilization for protein and carbohydrate synthesis in grains of wheat. Plant Physiol. 1999, 121, 871–878. [Google Scholar] [CrossRef]
- Alloway, B.J. Zinc in Soils and Crop Nutrition; International Zinc Association: Brussels, Belgium, 2004. [Google Scholar]
- Barak, P.; Helmke, P.A. The Chemistry of Zinc. In Zinc in Soils and Plants; Springer: Berlin, Germany, 1993; pp. 1–13. [Google Scholar]
- Brar, M.S.; Sekhon, G.S. Interaction of Zinc with Other Micronutrient Cations, II. In Effect of Iron on Zinc 65 Absorption by Rice Seedlings and its Translocation within the Plants; Springer: Berlin, Germany, 1976; Volume 45, pp. 145–150. [Google Scholar]
- Verma, T.S.; Tripathi, B.R. Zinc and Iron Interaction in Submerged Paddy; Springer: Berlin, Germany, 1983; Volume 72, pp. 107–116. [Google Scholar]
- Rietra, R.P.; Heinen, M.; Dimkpa, C.O.; Bindraban, P.S. Effects of nutrient antagonism and synergism on yield and fertilizer use efficiency. Commun. Soil Sci. Plant Anal. 2017, 48, 1895–1920. [Google Scholar] [CrossRef]
- Sumner, M.E.; Farina, M.P. Phosphorus Interactions with other Nutrients and Lime in Field Cropping Systems. In Advances in Soil Science; Springer: Berlin, Germany, 1986; pp. 201–236. [Google Scholar]
- Fageria, V. Nutrient interactions in crop plants. J. Plant Nutr. 2001, 24, 1269–1290. [Google Scholar] [CrossRef]
- Bressani, R.; Gonzalez, J.M.; Elias, L.G.; Melgar, M. Effect of fertilizer application on the yield, protein and fat content, and protein quality of raw and cooked grain of three amaranth species. Plant. Foods Hum. Nutr. 1987, 37, 59–67. [Google Scholar] [CrossRef]
- Raninen, K.; Lappi, J.; Mykkänen, H.; Poutanen, K. Dietary fiber type reflects physiological functionality: Comparison of grain fiber, inulin, and polydextrose. Nutr. Rev. 2011, 69, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Nagengast, F.; Hectors, M.; Buys, W.; Tongeren, J.V. Inhibition of secondary bile acid formation in the large intestine by lactulose in healthy subjects of two different age groups. Eur. J. Clin. Investig. 1988, 18, 56–61. [Google Scholar] [CrossRef]
- Moore, K.L.; Zhao, F.-J.; Gritsch, C.S.; Tosi, P.; Hawkesford, M.J.; McGrath, S.P.; Shewry, P.R.; Grovenor, C.R. Localisation of iron in wheat grain using high resolution secondary ion mass spectrometry. J. Cereal Sci. 2012, 55, 183–187. [Google Scholar] [CrossRef]
- Melash, A.A.; Mengistu, D.K.; Aberra, D.A.; Tsegay, A. The influence of seeding rate and micronutrients foliar application on grain yield and quality traits and micronutrients of durum wheat. J. Cereal Sci. 2018, 85, 221–227. [Google Scholar] [CrossRef]
- Wang, S.; Li, M.; Tian, X.; Li, J.; Li, H.; Ni, Y.; Zhao, J.; Chen, Y.; Guo, C.; Zhao, A. Foliar zinc, nitrogen, and phosphorus application effects on micronutrient concentrations in winter wheat. Agron. J. 2015, 107, 61–70. [Google Scholar] [CrossRef]
- Zhang, Y.-Q.; Deng, Y.; Chen, R.-Y.; Cui, Z.-L.; Chen, X.-P.; Yost, R.; Zhang, F.-S.; Zou, C.-Q. The reduction in zinc concentration of wheat grain upon increased phosphorus-fertilization and its mitigation by foliar zinc application. Plant Soil 2012, 361, 143–152. [Google Scholar] [CrossRef]
- Aciksoz, S.; Yazici, A.; Ozturk, L.; Cakmak, I. Biofortification of wheat with iron through soil and foliar application of nitrogen and iron fertilizers. Plant Soil 2011, 349, 215–225. [Google Scholar] [CrossRef]
- Gomez-Becerra, H.F.; Erdem, H.; Yazici, A.; Tutus, Y.; Torun, B.; Ozturk, L.; Cakmak, I. Grain concentrations of protein and mineral nutrients in a large collection of spelt wheat grown under different environments. J. Cereal Sci. 2010, 52, 342–349. [Google Scholar] [CrossRef]
- Haslett, B.; Reid, R.; Rengel, Z. Zinc mobility in wheat: Uptake and distribution of zinc applied to leaves or roots. Ann. Bot. 2001, 87, 379–386. [Google Scholar] [CrossRef]
- Alam, S.; Kamei, S.; Kawai, S. Effect of iron deficiency on the chemical composition of the xylem sap of barley. Soil Sci. Plant Nutr. 2001, 47, 643–649. [Google Scholar] [CrossRef]
- Ghasemi-Fasaei, R.; Ronaghi, A. Interaction of iron with copper, zinc, and manganese in wheat as affected by iron and manganese in a calcareous soil. J. Plant Nutr. 2008, 31, 839–848. [Google Scholar] [CrossRef]
- Kaya, C.; Higgs, D.; Burton, A. Foliar application of iron as a remedy for zinc toxic tomato plants. J. Plant Nutr. 1999, 22, 1829–1837. [Google Scholar] [CrossRef]
- Pahlavan Rad, M.; Pessarakli, M. Response of Wheat Plants to Zinc, Iron, and Manganese Applications and Uptake and Concentration of Zinc, Iron, and Manganese in Wheat Grains. Commun. Soil Sci. Plant Anal. 2009, 40, 1322–1332. [Google Scholar] [CrossRef]
- Gupta, U.C. Iron status of crops in Prince Edward Island and effect of soil pH on plant iron concentration. Can. J. Soil Sci. 1991, 71, 197–202. [Google Scholar] [CrossRef]
- Curie, C.; Cassin, G.; Couch, D.; Divol, F.; Higuchi, K.; Le Jean, M.; Misson, J.; Schikora, A.; Czernic, P.; Mari, S. Metal movement within the plant: Contribution of nicotianamine and yellow stripe 1-like transporters. Ann. Bot. 2008, 103, 1–11. [Google Scholar] [CrossRef]
| Symbol | Meaning of Symbol and Exact Quantity of Zn and Fe Applied |
|---|---|
| 100% Zn13 | 3.0 kg ha−1 of Zn |
| 100% Zn9.5 | 2.2 kg ha−1 of Zn |
| 100% Zn5.5 | 1.3 kg ha−1 of Zn |
| 80% Zn + 20% Fe13 | 2.40 kg ha−1 of Zn +0.52 kg ha−1 of Fe |
| 80% Zn + 20% Fe9.5 | 1.76 kg ha−1 of Zn +0.38 kg ha−1 of Fe |
| 80% Zn + 20% Fe5.5 | 1.04 kg ha−1 of Zn +0.22 kg ha−1 of Fe |
| 60% Zn + 40% Fe13 | 1.80 kg ha−1 of Zn +1.04 kg ha−1 of Fe |
| 60% Zn + 40% Fe9.5 | 1.32 kg ha−1 of Zn +0.76 kg ha−1 of Fe |
| 60% Zn + 40% Fe5.5 | 0.78 kg ha−1 of Zn +0.44 kg ha−1 of Fe |
| 40% Zn + 60% Fe13 | 1.20 kg ha−1 of Zn +1.56 kg ha−1 of Fe |
| 40% Zn + 60% Fe9.5 | 0.88 kg ha−1 of Zn +1.14 kg ha−1 of Fe |
| 40% Zn + 60% Fe5.5 | 0.52 kg ha−1 of Zn +0.66 kg ha−1 of Fe |
| 20% Zn + 80% Fe13 | 0.60 kg ha−1 of Zn +2.08 kg ha−1 of Fe |
| 20% Zn + 80% Fe9.5 | 0.44 kg ha−1 of Zn +1.52 kg ha−1 of Fe |
| 20% Zn + 80% Fe5.5 | 0.26 kg ha−1 of Zn +0.88 kg ha−1 of Fe |
| 100% Fe13 | 2.6 kg ha−1 of Fe |
| 100% Fe9.5 | 1.9 kg ha−1 of Fe |
| 100% Fe5.5 | 1.1 kg ha−1 of Fe |
| Control | 0 kg ha−1 of Zn + 0 kg ha−1 of Fe |
| Source of Variation | Grain Yield | TKW | Spike Length | Kernels per Spike | Crude Fat | Crude Fiber | Crude Protein | Zn Content | Fe Content |
|---|---|---|---|---|---|---|---|---|---|
| Year (Y) | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| Fertilizer ratio (R) | *** | *** | *** | ns | ns | ns | *** | *** | *** |
| Fertilizer quantity (Q) | ns | ns | ** | ns | ns | *** | ns | *** | ns |
| Y × R | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| Y × Q | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| R × Q | * | * | ** | ns | ns | ns | * | *** | *** |
| Y × R × Q | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| Treatments b | 2016–2017 | 2017–2018 | ||||||
|---|---|---|---|---|---|---|---|---|
| Grain Yield (t ha−1) | TKW (g) | Spike Length (cm) | Kernels per Spike | Grain Yield (t ha−1) | TKW (g) | Spike Length (cm) | Kernels per Spike | |
| 100% Zn13 | 4.228 | 44.07 | 7.88 | 35.20 | 4.106 *** | 44.20 * | 7.91 *** | 35.21 |
| 100% Zn9.5 | 3.829 | 43.96 | 7.79 | 38.85 | 3.800 *** | 44.08 | 7.83 *** | 38.38 *** |
| 100% Zn5.5 | 3.711 | 46.15* | 8.35 | 38.85 | 3.590 ** | 46.01 *** | 8.32 | 38.72 *** |
| 80% Zn + 20% Fe13 | 3.395 | 43.39 | 8.19 | 36.25 | 3.347 | 43.18 | 8.21 | 36.24 |
| 80% Zn + 20% Fe9.5 | 2.785 | 43.62 | 8.43 | 36.75 | 2.682 * | 43.51 | 8.44 ** | 36.62 * |
| 80% Zn + 20% Fe5.5 | 3.332 | 44.25 | 8.49 | 40.10 | 3.361 | 44.08 | 8.43 ** | 39.84 *** |
| 60%Zn + 40% Fe13 | 2.919 | 43.01 | 8.04 | 35.55 | 2.869 | 42.74 | 7.98 ** | 34.99 |
| 60% Zn + 40% Fe9.5 | 2.905 | 42.68 | 7.86 | 34.25 | 3.007 | 42.71 | 7.94 ** | 34.63 |
| 60% Zn + 40% Fe5.5 | 3.157 | 43.28 | 8.03 | 34.85 | 3.064 | 43.32 | 7.92 *** | 34.21 |
| 40% Zn + 60% Fe13 | 4.051 | 41.68 | 8.01 | 38.00 | 3.955 *** | 41.46 ** | 7.94 ** | 37.78 *** |
| 40% Zn + 60% Fe9.5 | 3.604 | 42.26 | 7.99 | 36.75 | 3.619 *** | 42.38 | 8.05 | 36.70 * |
| 40% Zn + 60% Fe5.5 | 4.161 | 42.63 | 8.13 | 34.75 | 4.320 *** | 42.72 | 8.09 | 35.23 |
| 20% Zn + 80% Fe13 | 4.007 | 40.54 | 8.68 | 37.50 | 4.159 *** | 40.49 *** | 8.72 *** | 37.12 * |
| 20% Zn + 80% Fe9.5 | 3.685 | 41.81 | 8.65 | 36.05 | 3.805 *** | 41.80 ** | 8.63 *** | 36.49 * |
| 20% Zn + 80% Fe5.5 | 4.251 | 43.76 | 8.39 | 39.85 | 4.316 *** | 43.55 | 8.38 * | 39.29 *** |
| 100% Fe13 | 4.935 * | 43.61 | 8.18 | 36.85 | 5.050 *** | 43.76 | 8.09 | 37.13 ** |
| 100% Fe9.5 | 4.228 | 41.21 | 8.55 | 36.40 | 4.037 *** | 41.03 *** | 8.56 *** | 35.94 |
| 100% Fe5.5 | 3.941 | 44.16 | 8.22 | 37.65 | 4.022 *** | 44.26 * | 8.17 | 37.28 *** |
| Control | 3.054 | 43.07 | 8.19 | 35.67 | 3.081 | 43.02 | 8.19 | 35.15 |
| Treatments b | 2016–2017 | 2017–2018 | ||||
|---|---|---|---|---|---|---|
| Crude Fat (%) | Crude Fiber (%) | Crude Protein (%) | Crude Fat (%) | Crude Fiber (%) | Crude Protein (%) | |
| 100% Zn13 | 1.80 | 3.51 *** | 14.51 | 1.80 | 3.58 *** | 14.49 |
| 100% Zn9.5 | 1.64 | 1.67 | 15.08 | 1.88 | 1.77 | 15.09 |
| 100% Zn5.5 | 1.58 | 1.96 *** | 14.87 | 1.78 | 2.17 | 14.99 |
| 80% Zn + 20% Fe13 | 1.61 | 2.92 *** | 15.11 | 1.71 | 2.96 *** | 15.00 |
| 80% Zn + 20% Fe9.5 | 1.08 | 3.89 *** | 15.60 | 1.46 | 4.13 *** | 15.49 * |
| 80% Zn + 20% Fe5.5 | 1.65 | 3.15 *** | 16.21 | 1.95 | 3.29 *** | 16.28 *** |
| 60%Zn + 40% Fe13 | 1.80 | 3.62 *** | 13.57 | 2.07 | 3.63 *** | 13.40 ** |
| 60% Zn + 40% Fe9.5 | 2.07 | 3.24 *** | 14.12 | 2.02 | 3.31 *** | 14.21 |
| 60% Zn + 40% Fe5.5 | 1.45 | 5.25 *** | 13.62 | 1.86 | 5.53 *** | 13.41 ** |
| 40% Zn + 60% Fe13 | 1.47 | 2.99 *** | 13.15 | 1.57 | 3.15 *** | 13.17 *** |
| 40% Zn + 60% Fe9.5 | 1.40 | 3.02 *** | 13.70 | 1.93 | 3.17 *** | 13.72 * |
| 40% Zn + 60% Fe5.5 | 2.04 | 2.09 | 13.62 | 2.37 | 2.25 | 13.59 * |
| 20% Zn + 80% Fe13 | 1.53 | 2.11 | 14.45 | 1.86 | 2.26 | 14.42 |
| 20% Zn + 80% Fe9.5 | 1.16 | 2.63 ** | 15.01 | 1.35 | 2.75 *** | 15.05 |
| 20% Zn + 80% Fe5.5 | 1.29 | 2.81 *** | 13.54 | 1.75 | 2.91 *** | 13.51 ** |
| 100% Fe13 | 1.72 | 2.69 ** | 12.10 ** | 1.90 | 2.82 *** | 11.98 *** |
| 100% Fe9.5 | 1.87 | 2.68 ** | 13.15 | 1.98 | 2.81 *** | 13.07 *** |
| 100% Fe5.5 | 1.05 | 2.47 * | 11.92 *** | 1.16 | 2.61 ** | 11.84 *** |
| Control | 1.73 | 1.58 | 14.87 | 2.14 | 1.70 | 14.57 |
| Treatments b | 2016–2017 | 2017–2018 | ||
|---|---|---|---|---|
| Zn (mg kg−1) c | Fe (mg kg−1) | Zn (mg kg−1) | Fe (mg kg−1) | |
| 100% Zn13 | 37.74 *** | 36.34 | 37.46 *** | 35.91 |
| 100% Zn9.5 | 38.79 *** | 43.22 | 39.63 *** | 43.02 *** |
| 100% Zn5.5 | 35.12 *** | 49.26 ** | 34.17 *** | 48.32 *** |
| 80% Zn + 20% Fe13 | 25.75 | 41.52 | 27.08 *** | 40.90 * |
| 80% Zn + 20% Fe9.5 | 29.97 * | 46.32 * | 30.64 *** | 46.87 *** |
| 80% Zn + 20% Fe5.5 | 26.04 | 44.14 | 26.12 *** | 45.00 *** |
| 60%Zn + 40% Fe13 | 32.52 ** | 42.27 | 32.51 *** | 41.40 ** |
| 60% Zn + 40% Fe9.5 | 27.90 | 42.30 | 26.72 *** | 41.45 *** |
| 60% Zn + 40% Fe5.5 | 26.12 | 43.83 | 25.49 *** | 42.27 *** |
| 40% Zn + 60% Fe13 | 26.54 | 43.10 | 26.74 *** | 43.27 *** |
| 40% Zn + 60% Fe9.5 | 25.78 | 42.83 | 25.44 *** | 42.47 *** |
| 40% Zn + 60% Fe5.5 | 23.66 | 42.27 | 22.72 ** | 42.10 *** |
| 20% Zn + 80% Fe13 | 22.70 | 42.51 | 22.41 * | 42.41 *** |
| 20% Zn + 80% Fe9.5 | 26.66 | 45.31 | 26.27 *** | 45.40 *** |
| 20% Zn + 80% Fe5.5 | 25.27 | 47.17 * | 25.04 *** | 47.33 *** |
| 100% Fe13 | 17.41 | 46.50 * | 17.10 | 46.15 *** |
| 100% Fe9.5 | 17.99 | 44.05 | 17.49 | 44.20 *** |
| 100% Fe5.5 | 26.84 | 43.28 | 25.15 *** | 42.16 *** |
| Control | 19.79 | 38.41 | 19.25 | 38.18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niyigaba, E.; Twizerimana, A.; Mugenzi, I.; Ngnadong, W.A.; Ye, Y.P.; Wu, B.M.; Hai, J.B. Winter Wheat Grain Quality, Zinc and Iron Concentration Affected by a Combined Foliar Spray of Zinc and Iron Fertilizers. Agronomy 2019, 9, 250. https://doi.org/10.3390/agronomy9050250
Niyigaba E, Twizerimana A, Mugenzi I, Ngnadong WA, Ye YP, Wu BM, Hai JB. Winter Wheat Grain Quality, Zinc and Iron Concentration Affected by a Combined Foliar Spray of Zinc and Iron Fertilizers. Agronomy. 2019; 9(5):250. https://doi.org/10.3390/agronomy9050250
Chicago/Turabian StyleNiyigaba, Etienne, Angelique Twizerimana, Innocent Mugenzi, Wansim Aboubakar Ngnadong, Yu Ping Ye, Bang Mo Wu, and Jiang Bo Hai. 2019. "Winter Wheat Grain Quality, Zinc and Iron Concentration Affected by a Combined Foliar Spray of Zinc and Iron Fertilizers" Agronomy 9, no. 5: 250. https://doi.org/10.3390/agronomy9050250
APA StyleNiyigaba, E., Twizerimana, A., Mugenzi, I., Ngnadong, W. A., Ye, Y. P., Wu, B. M., & Hai, J. B. (2019). Winter Wheat Grain Quality, Zinc and Iron Concentration Affected by a Combined Foliar Spray of Zinc and Iron Fertilizers. Agronomy, 9(5), 250. https://doi.org/10.3390/agronomy9050250




