Small-Scale Mechanical Harvesting and Tractor-Caused Soil Compaction Reduce Early Growth in Sugarcane
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Sites, Plant Materials, and Experimental Conditions
2.2. Harvest and Compaction Treatment
2.3. Measurements of the Traits Related to Early Growth of Sugarcane
2.4. Yield Component and Cane Yield Measurement
2.5. Statistical Analysis
3. Results
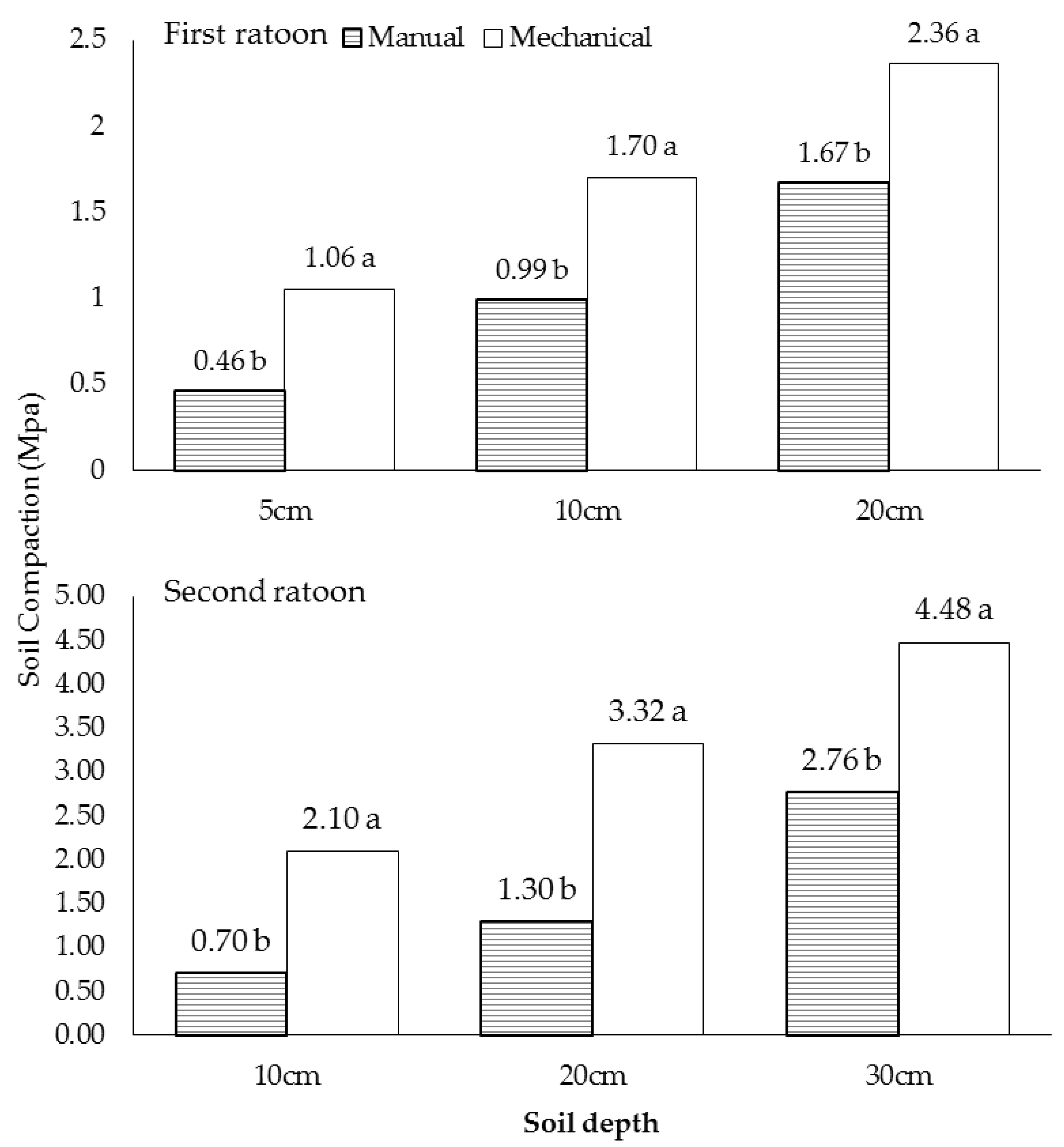
3.1. Soil Compaction
3.2. Stool Damage and Gaps
3.3. Underground Bud Bank, Ratoon Sprouting, and Their Correlations
3.4. Plant Height and Height Uniformity at Grand Growth and Maturing Stages
3.5. Cane Yield and Its Components
4. Discussion
4.1. Soil Compaction, Stool Damage, and Gaps
4.2. Underground Bud Banks and Sprouting
4.3. Plant Height and Height Uniformity
4.4. Cane Yield and Its Components Responses to Small-Scale Mechanical Harvest
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. FAOstat. Available online: http://www.fao.org/statistics/zh/ (accessed on 11 June 2019).
- Li, R.D.; Zhang, Y.B.; Yang, D.T.; Ou, Y.G.; Guo, J.W. Study on development potential of full mechanization in diversity terrain of Yunnan Sugarcane Region. Chin. Agric. Mech. 2012, 242, 51, 71–74. (In Chinese) [Google Scholar]
- Wang, S.L.; Xin, X. Analysis of sugarcane planting mechanization factors and its interactive effects. China Agric. Univ. J. Soc. Sci. Ed. 2017, 34, 84–93. (In Chinese) [Google Scholar]
- Ou, Y.G. Present situation and countermeasure of whole-process mechanization of sugarcane production in China. Mod. Agric. Equip. 2019, 40, 3–8, 42. (In Chinese) [Google Scholar]
- Pongpat, P.; Gheewala, S.H.; Silalertruksa, T. An assessment of harvesting practices of sugarcane in the central region of Thailand. J. Clean. Prod. 2017, 142, 1138–1147. [Google Scholar] [CrossRef]
- Guo, Y.S.; Xie, W.Z.; Liang, B.; Xu, G.Q.; Huang, X.Y.; Gao, J.Y. The small machine harvest and transportation model of raw cane. Guangxi Sugar Ind. 2016, 88, 47–48. (In Chinese) [Google Scholar]
- Cardoso, T.F.; Watanabe, M.D.B.; Souza, A.; Chagas, M.F.; Gavalett, O.; Morais, E.R.; Nogueira, L.A.H.; Leal, M.R.L.V.; Braunbeck, O.A.; Cortez, L.A.B.; et al. A regional approach to determine economic, environmental and social impacts of different sugarcane production system in Brazil. Biomass Bioenergy 2019, 120, 9–20. [Google Scholar] [CrossRef]
- Lozano, N.; Rolim, M.M.; Oliveira, V.S.; Tavares, U.E.; Pedrosa, E.M.R. Evaluation of soil compaction by modeling field vehicle traffic with SoilFlex during sugarcane harvest. Soil Tillage Res. 2013, 129, 61–68. [Google Scholar] [CrossRef]
- Esteban, D.A.A.; Souza, Z.M.; Tormena, C.A.; Lovera, L.H.; Lima, E.S.; Oliveira, I.N.; Ribeiro, N.P. Soil compaction, root system and productivity of sugarcane under different row spacing and controlled traffic at harvest. Soil Tillage Res. 2019, 187, 60–71. [Google Scholar] [CrossRef]
- Chen, C.J.; Liang, H.; He, Z.F.; Mo, Q.G.; Huang, Y.; Kuang, W.S.; Li, T.S.; Lu, G.Y. Effect of mechanical harvesting on sugarcane stubble quality and growth of ratoon. Asian Agric. Res. 2012, 4, 84–89. [Google Scholar]
- Li, Y.J.; Liang, Q.; Dong, W.B.; Chen, Q.; Liu, X.Y.; Xie, J.L.; Li, C.N.; Wang, W.Z.; Li, Y.R. Effect of mechanical compaction on seedling emergence and roots formation of ratoon sugarcane. Southwest China J. Agric. Sci. 2017, 30, 2041–2047. (In Chinese) [Google Scholar]
- Trouse, A.C., Jr.; Humbert, R.P. Some effects of soil compaction on the development of sugar cane roots. Soil Sci. 1961, 91, 208–217. [Google Scholar] [CrossRef]
- Silva, R.B.; Lancas, K.P.; Miranda, E.E.V.; Miranda, E.E.V.; Silva, F.A.M.; Baio, F.H.R. Estimation and evaluation of dynamic properties as indicators of changes on soil structure in sugarcane fields of Sao Paulo State–Brazil. Soil Tillage Res. 2009, 103, 265–270. [Google Scholar] [CrossRef]
- Braunack, M.V.; Arvidsson, J.; Håkansson, I. Effect of harvest traffic position on soil conditions and sugarcane (Saccharum officinarum) response to environmental conditions in Queensland, Australia. Soil Tillage Res. 2006, 89, 103–121. [Google Scholar] [CrossRef]
- Klimes Ová, J.; Klimeš, L. Bud banks and their role in vegetative regeneration—A literature review and proposal for simple classification and assessment. Perspect. Plant Ecol. Evol. Syst. 2007, 8, 115–129. [Google Scholar] [CrossRef]
- Ahmad, N.; Hassan, F.U.; Belford, R.K. Effect of soil compaction in the sub-humid cropping environment in Pakistan on uptake of NPK and grain yield in wheat (Triticum aestivum): I. Compaction. Field Crops Res. 2009, 110, 54–60. [Google Scholar] [CrossRef]
- Brereton, J.C.; McGowan, M.; Dawkins, T.C.K. The relative sensitivity of spring barley, spring field beans and sugar beet crops to soil compaction. Field Crops Res. 1986, 13, 223–237. [Google Scholar] [CrossRef]
- Otto, R.; Silva, A.P.; Franco, H.C.J.; Oliveira, E.C.A.; Trivelin, P.C.O. High soil penetration resistance reduces sugarcane root system development. Soil Tillage Res. 2011, 117, 201–210. [Google Scholar] [CrossRef]
- Basnayake, J.; Jackson, P.A.; Inman-Bamber, N.G.; Lakshmanan, P. Sugarcane for water-limited environments. Genetic variation in cane yield and sugar content in response to water stress. J. Exp. Bot. 2012, 63, 6023–6033. [Google Scholar] [CrossRef]
- Liu, J.Y.; Basnayake, J.; Jackson, P.A.; Chen, X.K.; Zhao, J.; Zhao, P.F.; Yang, L.H.; Bai, Y.D.; Xia, H.M.; Zan, F.G.; et al. Growth and yield of sugarcane genotypes are strongly correlated across irrigated and rainfed environments. Field Crops Res. 2016, 196, 418–425. [Google Scholar] [CrossRef]
- Zhao, P.F.; Jackson, P.A.; Basnayake, J.; Liu, J.Y.; Chen, X.K.; Zhao, J.; Zhao, X.D.; Bai, Y.D.; Yang, L.H.; Zan, F.G.; et al. Genetic variation in sugarcane for leaf functional traits and relationships with cane yield, in environments with varying water stress. Field Crops Res. 2017, 213, 143–153. [Google Scholar] [CrossRef]
- Zhao, P.F.; Liu, J.Y.; Yang, K.; Xia, H.M.; Wu, C.W.; Chen, X.K.; Zhao, J.; Yang, H.C.; Li, J.; Zan, F.G.; et al. Registration of ‘YZ05-51’ sugarcane. J. Plant Regist. 2015, 9, 172–178. [Google Scholar] [CrossRef]
- Ma, S.; Scharf, P.A.; Zhang, Q.; Karkee, M.; Tong, J.; Yu, L. Effect of cane stool density and stubble height on sugarcane stubble damage in Hawaii fields. Trans. ASABE 2016, 59, 813–820. [Google Scholar]
- Zhao, P.F.; Dai, J.J.; Liu, G.Y.; Gao, X.X.; Yang, L.T.; Li, Y.R.; Guo, J.W. A primary report on the relationships between fiber components of bottom stems and stool damage by mechanical harvest in sugarcane. Chin. J. Trop. Agric. 2017, 37, 104–108, 116. (In Chinese) [Google Scholar]
- Ma, D.L.; Xie, R.Z.; Zhai, L.C.; Ming, B.; Li, S.K. Changes in population uniformity among maize varieties from different eras. J. Maize Sci. 2017, 25, 1–6. (In Chinese) [Google Scholar]
- Jackson, P.; Braunack, M.; Foreman, J.; Peatey, T. Genetic variation in sugarcane for ratooning after harvester damage in wet soil. Euphytica 2000, 111, 1–8. [Google Scholar] [CrossRef]
- Li, W.F.; Fan, Y.H.; Chen, X.K.; Xia, H.M.; Li, F.Q.; Wang, Y.Y. Rapid determination method of cane sugar content. Sugar Crops China 2009, 31, 14–15. (In Chinese) [Google Scholar]
- Viator, R.P.; Dalley, C.D.; Johnson, R.M.; Richard, E.P., Jr. Early harvest affects sugarcane ratooning ability in Louisiana. Sugar Cane Int. 2010, 28, 123–127. [Google Scholar]
- Statistix. Analytical Software. v. 8.0. Available online: http://www.statistix.com/ (accessed on 6 May 2014).
- Jain, R.; Shrivastava, A.K.; Solomon, S.; Yadav, R.L. Low temperature stress-induced biochemical changes affect stubble bud sprouting in sugarcane (Saccharum spp. hybrid). Plant Growth Regul. 2007, 53, 17–23. [Google Scholar] [CrossRef]
- Wu, X.L.; Tang, Y.L.; Li, C.S.; McHugh, A.D.; Li, Z.; Wu, C. Individual and combined effects of soil waterlogging and compaction on physiological characteristics of wheat in southwestern China. Field Crops Res. 2018, 215, 163–172. [Google Scholar] [CrossRef]
- Su, J.B.; Kong, R.; Luo, L.F.; Li, D.L. Analysis of sugarcane ratooning performance after mechanized harvest. Sugarcane Canesugar 2016, 6, 22–28. (In Chinese) [Google Scholar]
- Bengough, A.G.; Mullins, C.E. Mechanical impedance to root growth: A review of experimental techniques and root growth responses. J. Soil Sci. 1990, 41, 341–358. [Google Scholar] [CrossRef]
- Lei, C.H. Effect of machinery harvest on the growth of ratoon cane. Agric. Res. Appl. 2015, 156, 26–29. (In Chinese) [Google Scholar]
- Vasantha, S.; Shekinah, E.D.; Gupta, C.; Rakkiyappan, P. Tiller production, regulation and senescence in sugarcane (Saccharum species hybrid) genotypes. Sugar Tech 2012, 14, 156–160. [Google Scholar] [CrossRef]
- Zhao, P.F.; Liu, J.Y.; Wu, C.W.; Yang, H.C.; Zhao, J.; Chen, X.K.; Xia, H.M.; Zan, F.G.; Li, J.; Yang, K.; et al. Registration of ‘YZ01-1413’ sugarcane. J. Plant Regist. 2017, 11, 129–134. [Google Scholar] [CrossRef]
- Marin, F.R.; Edreird, J.L.R.; Andrade, J.; Grassini, P. On-farm sugarcane yield and yield components as influenced by number of harvests. Field Crops Res. 2019, 240, 134–142. [Google Scholar] [CrossRef]
- Ramburan, S.; Wettergreen, T.; Berry, S.D.; Shongwe, B. Genetic, environmental and management contributions to ratoon decline in sugarcane. Field Crops Res. 2013, 146, 105–112. [Google Scholar] [CrossRef]
- Berding, N.; Hurney, A.P.; Salter, B.; Bonnett, G.D. Agronomic impact of sucker development in sugarcane under different environmental conditions. Field Crops Res. 2005, 92, 203–217. [Google Scholar] [CrossRef]

| Attributes | Crop | ||
|---|---|---|---|
| PC | First RC | Second RC | |
| Location | 103°15′ E, 23°42′ N | ||
| Soil texture | Clay loam | ||
| Accumulated rainfall (mm) | 737.75 | 939.90 | 1038.40 |
| Mean monthly max temp (°C) | 31.43 | 30.98 | 30.80 |
| Mean monthly min temp (°C) | 11.37 | 10.77 | 11.11 |
| Planting date | 2 February 2016 | ||
| Harvest dates | 22 February 2017 | 15 March 2018 | 5 March 2019 |
| Growth period (day) | 386 | 386 | 355 |
| Crop | Treatment | Mean Square | ||||
|---|---|---|---|---|---|---|
| MH | SMH | Treatment | Genotype | GT | Error | |
| Stool damage (%) | ||||||
| First RC | 5.22 | 36.88 | 10,024.20 *** | 89.60 * | 93.50 * | 28.30 |
| Second RC | 9.12 | 74.02 | 42,118.80 *** | 92.10 * | 134.10 ** | 30.90 |
| Gaps (m ha−1) | ||||||
| First RC | 118.49 | 463.54 | 1,190,609 * | 362,506 (ns) | 226,381 (ns) | 158,889 |
| Second RC | 445.30 | 1070.30 | 3,906,250 * | 2,417,772 *** | 1,096,429 ** | 193,244 |
| Crop | Treatment | Mean Square | ||||
|---|---|---|---|---|---|---|
| MH | SMH | Treatment | Genotype | GT | Error | |
| Buds per stool | ||||||
| First RC | 18.99 | 17.33 | 27.81 (ns) | 8.98 (ns) | 17.73 (ns) | 15.45 |
| Second RC | 18.08 | 17.18 | 8.20 (ns) | 25.49 ** | 3.78 (ns) | 5.29 |
| 1000 buds ha−1 | ||||||
| First RC | 1103 | 1124 | 4410 (ns) | 156,430 *** | 5205 (ns) | 9024 |
| Second RC | 1161 | 1086.30 | 55,840 * | 365,136 *** | 6980 (ns) | 5937 |
| Crop | Treatment | Mean Square | ||||
|---|---|---|---|---|---|---|
| MH | SMH | Treatment | Genotype | GT | Error | |
| Seedlings in April (1000 seedlings ha−1) | ||||||
| Plant crop (PC) | 63.96 | 58.82 | 264.30 (ns) | 572.20 *** | 73.54 (ns) | 74.96 |
| First RC | 80.71 | 72.52 | 670.11 * | 3512.84 *** | 81.69 * | 28.16 |
| Second RC | 69.89 | 39.32 | 9341.28 * | 1294.24 *** | 356.98 ** | 54.14 |
| Seedlings in May (1000 seedlings ha−1) | ||||||
| PC | 102.64 | 100.30 | 54.90 (ns) | 2818.30 *** | 441.31 (ns) | 221.62 |
| First RC | 110.15 | 98.99 | 1244.23 * | 4605.70 *** | 277.33 (ns) | 152.94 |
| Second RC | 124.19 | 106.55 | 3113.46 * | 9297.23 *** | 232.66 (ns) | 157.27 |
| Crop | Month of Seedlings Count | Correlation | ||
|---|---|---|---|---|
| All Subplots | MH | SMH | ||
| First RC | April 2017 | 0.3816 * | 0.3960 (ns) | 0.4143 (ns) |
| May 2017 | 0.3937 * | 0.3934 (ns) | 0.4677 * | |
| Second RC | April 2018 | 0.4930 ** | 0.3907 (ns) | 0.6699 ** |
| May 2018 | 0.8453 *** | 0.7970 *** | 0.8743 *** | |
| Crop | Date | Treatment | Mean Square | ||||
|---|---|---|---|---|---|---|---|
| MH | SMH | Treatment | Genotype | GT | Error | ||
| Plant height (cm) | |||||||
| First RC | 30 June 2017 | 95.90 | 81.64 | 2033.48 * | 3981.39 *** | 57.92 (ns) | 61.54 |
| 15 November 2017 | 295.70 | 287.72 | 637.60 (ns) | 16,824.60 *** | 78.60 (ns) | 86.90 | |
| Second RC | 24 May 2018 | 26.79 | 20.17 | 437.91 *** | 154.00 *** | 2.87 (ns) | 5.23 |
| 20 June 2018 | 69.34 | 48.86 | 4194.3 *** | 1393.26 *** | 61.73 (ns) | 52.47 | |
| 25 July 2018 | 119.51 | 98.02 | 4621.21 ** | 3553.72 *** | 518.02 *** | 58.28 | |
| 30 Nov 2018 | 264.91 | 240.36 | 6027 ** | 18,408.80 *** | 109.90 (ns) | 210.9 | |
| Height uniformity | |||||||
| First RC | 30 June 2017 | 7.81 | 5.27 | 64.34 ** | 34.60 *** | 15.75 * | 4.34 |
| 15 November 2017 | 19.54 | 16.93 | 68.00 * | 373.81 *** | 199.89 *** | 22.06 | |
| Second RC | 24 May 2018 | 4.93 | 3.77 | 13.47 * | 4.35 ** | 1.09 (ns) | 0.68 |
| 20 June 2018 | 7.91 | 5.04 | 81.97 ** | 8.99 *** | 2.40 (ns) | 1.16 | |
| 25 July 2018 | 7.16 | 5.73 | 20.56 * | 29.77 *** | 1.23 (ns) | 2.98 | |
| 30 November 2018 | 21.27 | 13.18 | 653.63 * | 159.17 ** | 69.29 * | 13.16 | |
| Crop | Treatment | Mean Square | ||||
|---|---|---|---|---|---|---|
| MH | SMH | Treatment | Genotype | GT | Error | |
| Cane yield (t ha−1) | ||||||
| PC | 106.36 | 103.26 | 95.57 (ns) | 2228.69 *** | 167.71 (ns) | 143.42 |
| First RC | 108.49 | 102.15 | 402.43 (ns) | 4052.10 *** | 42.73 (ns) | 44.09 |
| Second RC | 96.17 | 81.12 | 2265.93 * | 5574.91 *** | 30.47 (ns) | 82.18 |
| Millable stalks (×1000) ha−1 | ||||||
| First RC | 60.76 | 61.90 | 13.12 (ns) | 375.95 *** | 15.63 (ns) | 28.48 |
| Second RC | 65.57 | 61.07 | 203.09 * | 350.53 *** | 29.91 (ns) | 20.41 |
| First RC | 69.53 | 61.54 | 639.12 ** | 1038.24 *** | 90.21 * | 25.28 |
| Plant height (cm) | ||||||
| First RC | 295.70 | 287.72 | 637.60 (ns) | 16824.60 *** | 78.60 (ns) | 86.90 |
| Second RC | 264.91 | 240.36 | 6027 ** | 18408.80 *** | 109.90 (ns) | 210.90 |
| Stalk diameter (cm) | ||||||
| First RC | 2.77 | 2.91 | 0.20 (ns) | 0.13 *** | 0.003 (ns) | 0.01 |
| Second RC | 2.768 | 2.94 | 0.299 * | 0.097 ** | 0.01 (ns) | 0.02 |
| Single stalk weight (kg) | ||||||
| First RC | 1.77 | 1.71 | 0.05 (ns) | 0.43 *** | 0.02 (ns) | 0.05 |
| Second RC | 1.35 | 1.29 | 0.04 (ns) | 0.25 *** | 0.01 (ns) | 0.03 |
| Fiber (%) | ||||||
| First RC | 13.32 | 13.70 | 1.43 (ns) | 4.77 *** | 0.27 (ns) | 0.43 |
| Second RC | 13.27 | 13.61 | 1.16 (ns) | 4.83 *** | 0.74 (ns) | 0.37 |
| Purity (%) | ||||||
| First RC | 86.97 | 86.23 | 5.44 * | 6.60 *** | 0.49 (ns) | 0.53 |
| Second RC | 85.62 | 84.21 | 20.09 * | 18.22 *** | 0.74 (ns) | 2.21 |
| Sucrose content (%) | ||||||
| First RC | 15.21 | 15.42 | 0.43 (ns) | 1.25 *** | 0.22 (ns) | 0.17 |
| Second RC | 13.79 | 13.39 | 1.56 (ns) | 2.91 *** | 0.15 (ns) | 0.25 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, P.; Guo, J.; Gao, X.; Liu, G.; Li, Y.; Burner, D.M.; Yang, L. Small-Scale Mechanical Harvesting and Tractor-Caused Soil Compaction Reduce Early Growth in Sugarcane. Agronomy 2019, 9, 830. https://doi.org/10.3390/agronomy9120830
Zhao P, Guo J, Gao X, Liu G, Li Y, Burner DM, Yang L. Small-Scale Mechanical Harvesting and Tractor-Caused Soil Compaction Reduce Early Growth in Sugarcane. Agronomy. 2019; 9(12):830. https://doi.org/10.3390/agronomy9120830
Chicago/Turabian StyleZhao, Peifang, Jiawen Guo, Xinxin Gao, Gaoyuan Liu, Yangrui Li, David M. Burner, and Litao Yang. 2019. "Small-Scale Mechanical Harvesting and Tractor-Caused Soil Compaction Reduce Early Growth in Sugarcane" Agronomy 9, no. 12: 830. https://doi.org/10.3390/agronomy9120830
APA StyleZhao, P., Guo, J., Gao, X., Liu, G., Li, Y., Burner, D. M., & Yang, L. (2019). Small-Scale Mechanical Harvesting and Tractor-Caused Soil Compaction Reduce Early Growth in Sugarcane. Agronomy, 9(12), 830. https://doi.org/10.3390/agronomy9120830





