Arsenic Accumulation in Grafted Melon Plants: Role of Rootstock in Modulating Root-To-Shoot Translocation and Physiological Response
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site, Plant Material, and Management Practices
2.2. Leaf Relative Chlorophyll (Chl) Content and Gas Exchange Measurements
2.3. Plant Growth and Development Measurements
2.4. Arsenic and Phosphorous Determination in Plant Tissues
2.5. Bioaccumulation and Translocation Factors
2.6. Statistical Procedures
2.7. Microclimate Conditions Inside the Greenhouse
3. Results
3.1. Aboveground Plant Biomass and Partitioning
3.2. Leaf Growth Variables
3.3. Leaf Relative Chl Content and Gas Exchanges
3.4. Arsenic Accumulation in Plant Fractions
3.5. Arsenic Speciation in Root
3.6. Arsenic Bioaccumulation and Translocation Factors
3.7. Phosphorus Accumulation in Plant Fractions
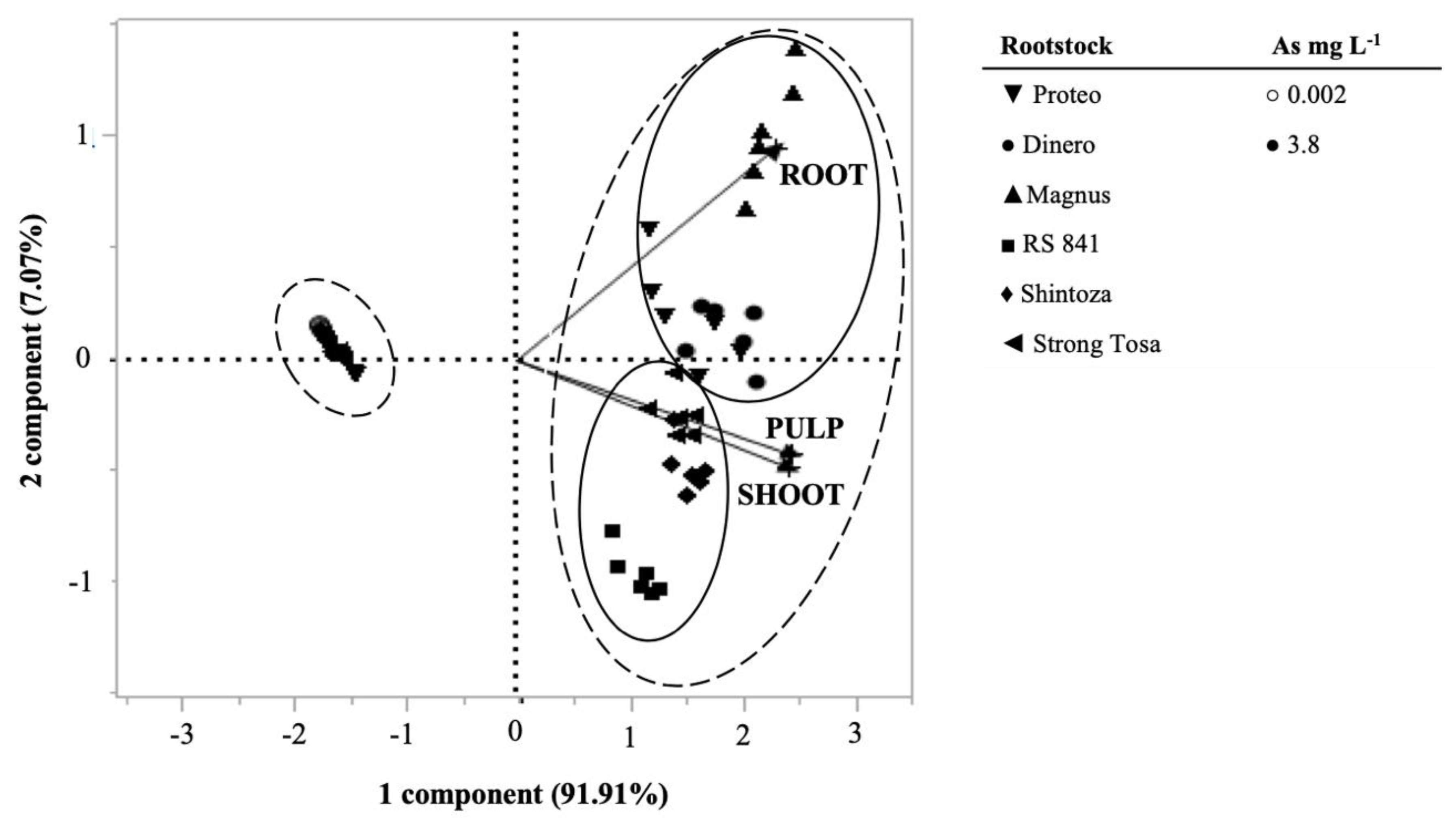
3.8. Principal Component Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Calatayud, Á.; San Bautista, A.; Pascual, B.; Maroto, J.V.; López-Galarza, S. Use of Chlorophyll Fluorescence Imaging as Diagnostic Technique to Predict Compatibility in Melon Graft. Sci. Hortic. (Amsterdam) 2013, 149, 13–18. [Google Scholar] [CrossRef]
- Gupta, S.; Satpati, S.; Nayek, S.; Garai, D. Effect of Wastewater Irrigation on Vegetables in Relation to Bioaccumulation of Heavy Metals and Biochemical Changes. Environ. Monit. Assess. 2010, 165, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Muchuweti, M.; Birkett, J.W.; Chinyanga, E.; Zvauya, R.; Scrimshaw, M.D.; Lester, J.N. Heavy Metal Content of Vegetables Irrigated with Mixtures of Wastewater and Sewage Sludge in Zimbabwe: Implications for Human Health. Agric. Ecosyst. Environ. 2006, 112, 41–48. [Google Scholar] [CrossRef]
- Verkleij, J.A.C.; Golan-Goldhirsh, A.; Antosiewisz, D.M.; Schwitzguébel, J.P.; Schröder, P. Dualities in Plant Tolerance to Pollutants and Their Uptake and Translocation to the Upper Plant Parts. Environ. Exp. Bot. 2009, 67, 10–22. [Google Scholar] [CrossRef]
- Meharg, A.A.; Macnair, M.R. Relationship between Plant Phosphorus Status and the Kinetics of Arsenate Influx in Clones of Deschampsia cespitosa (L.) Beauv. That Differ in Their Tolerance to Arsenate. Plant Soil 1994, 162, 99–106. [Google Scholar] [CrossRef]
- Hughes, M.F.; Beck, B.D.; Chen, Y.; Lewis, A.S.; Thomas, D.J. Arsenic Exposure and Toxicology: A Historical Perspective. Toxicol. Sci. 2011, 123, 305–332. [Google Scholar] [CrossRef]
- Carbonell-Barrachina, A.A.; Aarabi, M.A.; DeLaune, R.D.; Gambrell, R.P.; Patrick, W.H. The Influence of Arsenic Chemical Form and Concentration on Spartina Patens and Spartina Alterniflora Growth and Tissue Arsenic Concentration. Plant Soil 1998, 198, 33–43. [Google Scholar] [CrossRef]
- Stazi, S.R.; Marabottini, R.; Papp, R.; Moscatelli, M.C. Arsenic in Soil: Availability and Interactions with Soil Microorganisms. In Heavy Metal Contamination of Soils; Sherameti, I., Varma, A., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 113–126. [Google Scholar] [CrossRef]
- Garg, N.; Singla, P. Arsenic Toxicity in Crop Plants: Physiological Effects and Tolerance Mechanisms. Environ. Chem. Lett. 2011, 9, 303–321. [Google Scholar] [CrossRef]
- Finnegan, P.M.; Chen, W. Arsenic Toxicity: The Effects on Plant Metabolism. Front. Physiol. 2012, 3, 1–18. [Google Scholar] [CrossRef]
- Maurel, C.; Verdoucq, L.; Luu, D.T.; Santoni, V. Plant Aquaporins: Membrane Channels with Multiple Integrated Functions. Annu. Rev. Plant Biol. 2008, 59, 595–624. [Google Scholar] [CrossRef]
- Allevato, E.; Stazi, S.R.; Marabottini, R.; D’Annibale, A. Mechanisms of arsenic assimilation by plants and countermeasures to attenuate its accumulation in crops other than rice. Ecotoxicol. Environ. Saf. 2019, 185, 109701. [Google Scholar] [CrossRef]
- Abbas, G.; Murtaza, B.; Bibi, I.; Shahid, M.; Niazi, N.K.; Khan, M.I.; Amjad, M.; Hussain, M.; Natasha. Arsenic Uptake, Toxicity, Detoxification, and Speciation in Plants: Physiological, Biochemical, and Molecular Aspects. Int. J. Environ. Res. Public Health 2018, 15, 59. [Google Scholar] [CrossRef] [PubMed]
- Verbruggen, N.; Hermans, C.; Schat, H. Mechanisms to Cope with Arsenic or Cadmium Excess in Plants. Curr. Opin. Plant Biol. 2009, 12, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Kyriacou, M.C.; Rouphael, Y.; Colla, G.; Zrenner, R.; Schwarz, D. Vegetable Grafting: The Implications of a Growing Agronomic Imperative for Vegetable Fruit Quality and Nutritive Value. Front. Plant Sci. 2017, 8, 1–23. [Google Scholar] [CrossRef]
- Rouphael, Y.; Kyriacou, M.C.; Colla, G. Vegetable Grafting: A Toolbox for Securing Yield Stability under Multiple Stress Conditions. Front. Plant Sci. 2018, 8, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, L.; Iapichino, G.; D’Anna, F.; Palazzolo, E.; Mennella, G.; Rotino, G.L. Hybrids and Allied Species as Potential Rootstocks for Eggplant: Effect of Grafting on Vigour, Yield and Overall Fruit Quality Traits. Sci. Hortic. Amsterdam 2018, 228, 81–90. [Google Scholar] [CrossRef]
- Kumar, P.; Edelstein, M.; Cardarelli, M.; Ferri, E.; Colla, G. Grafting Affects Growth, Yield, Nutrient Uptake, and Partitioning under Cadmium Stress in Tomato. HortScience 2015, 50, 1654–1661. [Google Scholar] [CrossRef]
- Stazi, S.R.; Cassaniti, C.; Marabottini, R.; Giuffrida, F.; Leonardi, C. Arsenic Uptake and Partitioning in Grafted Tomato Plants. Hortic. Environ. Biotechnol. 2016, 57, 241–247. [Google Scholar] [CrossRef]
- Savvas, D.; Colla, G.; Rouphael, Y.; Schwarz, D. Amelioration of Heavy Metal and Nutrient Stress in Fruit Vegetables by Grafting. Sci. Hortic. 2010, 127, 156–161. [Google Scholar] [CrossRef]
- Hoel, B.O.; Solhaug, K.A. Effect of irradiance on chlorophyll estimation with the Minolta SPAD-502 leaf chlorophyll meter. Ann. Bot. 1998, 82, 389–392. [Google Scholar] [CrossRef]
- Martínez, D.E.; Guiamet, J.J. Distortion of the SPAD 502 chlorophyll meter readings by changes in irradiance and leaf water status. Agronomie 2004, 24, 41–46. [Google Scholar] [CrossRef]
- Al-hamdani, S.H.; Murphy, J.M.; Todd, G.W. Stomatal conductance and CO2 assimilation as screening tools for drought resistance in sorghum. Can. J. Plant Sci. 1991, 71, 689–694. [Google Scholar] [CrossRef]
- Rintala, E.M.; Ekholm, P.; Koivisto, P.; Peltonen, K.; Venäläinen, E.R. The intake of inorganic arsenic from long grain rice and rice-based baby food in Finland—Low safety margin warrants follow up. Food Chem. 2014, 150, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Welna, M.; Pohl, P.; Szymczycha-Madeja, A. Non-chromatographic Speciation of Inorganic Arsenic in Rice by Hydride Generation Inductively Coupled Plasma Optical Emission Spectrometry. Food Anal. Methods 2019, 12, 581–594. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, X.H.; Li, T.Y.; Wu, Q.X.; Jin, Z.J. Soil characteristics and heavy metal accumulation by native plants in a Mn mining area of Guangxi, South China. Environ. Monit. Assess. 2014, 186, 2269–2279. [Google Scholar] [CrossRef]
- de Campos, F.V.; de Oliveira, J.A.; da Silva, A.A.; Ribeiro, C.; dos Santos Farnese, F. Phytoremediation of arsenite-contaminated environments: Is Pistia stratiotes L. a useful tool? Ecol. Indic. 2019, 104, 794–801. [Google Scholar] [CrossRef]
- Pandey, J.; Verma, R.K.; Singh, S. Screening of most potential candidate among different lemongrass varieties for phytoremediation of tannery sludge contaminated sites. Int. J. Phytoremediat. 2019, 21, 600–609. [Google Scholar] [CrossRef]
- Saldaña-Robles, A.; Abraham-Juárez, M.R.; Saldaña-Robles, A.L.; Saldaña-Robles, N.; Ozuna, C.; Gutiérrez-Chávez, A.J. The Negative Effect of Arsenic in Agriculture: Irrigation Water, Soil and Crops, State of the Art. Appl. Ecol. Environ. Res. 2018, 16, 1533–1551. [Google Scholar] [CrossRef]
- Stazi, S.R.; Allevato, E.; Marabottini, R. Contaminazione Dei Prodotti Agricoli Da Arsenico: Assimilazione e Strategie Di Mitigazione. Italus Hortus 2016, 23, 19–32. [Google Scholar]
- Ma, L.Q.; Komar, K.M.; Tu, C.; Zhang, W.; Cai, Y.; Kennelley, E.D. A Fern That Hyperaccumulates Arsenic. Nature 2001, 409, 579. [Google Scholar] [CrossRef]
- Gomes, M.P.; Carvalho, M.; Carvalho, G.S.; Marques, T.C.L.L.S.M.; Garcia, Q.S.; Guilherme, L.R.G.; Soares, A.M. Phosphorus Improves Arsenic Phytoremediation by Anadenanthera peregrina by Alleviating Induced Oxidative Stress. Int. J. Phytoremediat. 2013, 15, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, T.; Bibi, I.; Shahid, M.; Niazi, N.K.; Murtaza, B.; Wang, H.; Ok, Y.S.; Sarkar, B.; Javed, M.T.; Murtaza, G. Effect of Compost Addition on Arsenic Uptake, Morphological and Physiological Attributes of Maize Plants Grown in Contrasting Soils. J. Geochem. Explor. 2017, 178, 83–91. [Google Scholar] [CrossRef]
- Koyama, K.; Kikuzawa, K. Is Whole-Plant Photosynthetic Rate Proportional to Leaf Area? A Test of Scalings and a Logistic Equation by Leaf Demography Census. Am. Nat. 2009, 173, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Ropokis, A.; Ntatsi, G.; Kittas, C.; Katsoulas, N.; Savvas, D. Impact of Cultivar and Grafting on Nutrient and Water Uptake by Sweet Pepper (Capsicum annuum L.) Grown Hydroponically Under Mediterranean Climatic Conditions. Front. Plant Sci. 2018, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.M.; Hasegawa, H.; Mahfuzur Rahman, M.; Nazrul Islam, M.; Majid Miah, M.A.; Tasmen, A. Effect of Arsenic on Photosynthesis, Growth and Yield of Five Widely Cultivated Rice (Oryza sativa L.) Varieties in Bangladesh. Chemosphere 2007, 67, 1072–1079. [Google Scholar] [CrossRef]
- Zu, Y.Q.; Sun, J.J.; He, Y.M.; Wu, J.; Feng, G.Q.; Li, Y. Effects of Arsenic on Growth, Photosynthesis and Some Antioxidant Parameters of Panax Notoginseng Growing in Shaded Conditions. Int. J. Adv. Agric. Res. 2016, 4, 78–88. [Google Scholar]
- Srivastava, S.; Akkarakaran, J.J.; Sounderajan, S.; Shrivastava, M.; Suprasanna, P. Arsenic Toxicity in Rice (Oryza sativa L.) Is Influenced by Sulfur Supply: Impact on the Expression of Transporters and Thiol Metabolism. Geoderma 2016, 270, 33–42. [Google Scholar] [CrossRef]
- Zhao, F.J.; Ma, J.F.; Meharg, A.A.; McGrath, S.P. Arsenic Uptake and Metabolism in Plants. New Phytol. 2009, 181, 777–794. [Google Scholar] [CrossRef]
- Raab, A.; Schat, H.; Meharg, A.A.; Feldmann, J. Uptake, Translocation and Transformation of Arsenate and Arsenite in Sunflower (Helianthus annuus): Formation of Arsenic-Phytochelatin Complexes during Exposure to High Arsenic Concentrations. New Phytol. 2005, 168, 551–558. [Google Scholar] [CrossRef]
- Pickering, I.J.; Prince, R.C.; George, M.J.; Smith, R.D.; George, G.N.; Salt, D.E. Reduction and Coordination of Arsenic in Indian Mustard 1. Plant Physiol. 2000, 122, 1171–1177. [Google Scholar] [CrossRef]
- Ouyang, Y. Phytoextraction: Simulating Uptake and Translocation of Arsenic in a Soil-Plant System. Int. J. Phytoremediat. 2005, 7, 3–17. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, F.J.; Meharg, A.A.; Raab, A.; Feldmann, J.; McGrath, S.P. Mechanisms of Arsenic Hyperaccumulation in Pteris Vittata. Uptake Kinetics, Interactions with Phosphate, and Arsenic Speciation. Plant Physiol. 2002, 130, 1552–1561. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, I. Effects of Arsenic Concentration and Forms on Growth and Arsenic Uptake and Accumulation by Indian Mustard (Brassica juncea L.) Genotyopes. J. Cent. Eur. Agric. 2006, 7, 31–40. [Google Scholar]
- Ierna, A.; Mauro, R.P.; Mauromicale, G. Improved Yield and Nutrient Efficiency in Two Globe Artichoke Genotypes by Balancing Nitrogen and Phosphorus Supply. Agron. Sustain. Dev. 2012, 32, 773–780. [Google Scholar] [CrossRef]
- Tu, C.; Ma, L.Q. Effects of Arsenate and Phosphate on Their Accumulation by an Arsenic-Hyperaccumulator Pteris vittata L. Plant Soil 2003, 249, 373–382. [Google Scholar] [CrossRef]
- Gunes, A.; Pilbeam, D.J.; Inal, A. Effect of Arsenic-Phosphorus Interaction on Arsenic-Induced Oxidative Stress in Chickpea Plants. Plant Soil 2009, 314, 211–220. [Google Scholar] [CrossRef]
- Suriyagoda, L.D.B.; Dittert, K.; Lambers, H. Mechanism of Arsenic Uptake, Translocation and Plant Resistance to Accumulate Arsenic in Rice Grains. Agric. Ecosyst. Environ. 2018, 253, 23–37. [Google Scholar] [CrossRef]
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo” (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| Plant | As− | 235.0 | 251.7 | 207.0 | 300.3 | 247.7 | 317.0 | 259.8 a | 45.1 |
| As+ | 220.0 | 198.7 | 226.3 | 278.7 | 268.3 | 239.3 | 238.6 b | ||
| Mean | 227 bc | 225 c | 217 c | 289 a | 258 ab | 278 a | |||
| Stem | As− | 29.7 | 31.2 | 31.4 | 29.6 | 30.8 | 28.0 | 30.1 a | NS |
| As+ | 22.2 | 21.9 | 22.7 | 25.0 | 28.4 | 23.39 | 24.0 b | ||
| Mean | 25.9 a | 26.5 a | 27.1 a | 27.3 a | 29.6 a | 25.9 a | |||
| Leaf | As− | 56.8 | 66.4 | 56.7 | 66.3 | 55.8 | 58.0 | 60.0 a | 10.3 |
| As+ | 47.6 | 42.0 | 44.5 | 52.1 | 58.0 | 44.4 | 48.1 b | ||
| Mean | 52.2 a | 54.2 a | 50.6 a | 59.2 a | 56.9 a | 51.2 a | |||
| Fruit | As− | 148.8 | 154.0 | 119.9 | 204.6 | 161.1 | 231.1 | 169.9 a | 37.3 |
| As+ | 150.3 | 134.6 | 159.1 | 201.5 | 182.0 | 170.9 | 166.4 a | ||
| Mean | 149.6 c | 144.3 c | 139.5 c | 203.0 a | 171.5 b | 201.0 a | |||
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo” (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| LN (n. plant−1) | As− | 163 | 178 | 187 | 158 | 157 | 176 | 170 a | 22 |
| As+ | 159 | 135 | 145 | 140 | 162 | 149 | 148 b | ||
| Mean | 161 a | 156 a | 166 a | 149 a | 160 a | 162 a | |||
| LA (dm2 plant−1) | As− | 74.1 | 91.5 | 86.9 | 75.9 | 83.6 | 86.3 | 83.1 a | 14.5 |
| As+ | 66.9 | 57.1 | 59.7 | 63.3 | 83.3 | 67.0 | 66.2 b | ||
| Mean | 70.5 a | 74.3 a | 73.3 a | 69.6 a | 83.4 a | 76.7 a | |||
| LAR (cm2 g−1 DW) | As− | 31.7 | 36.4 | 42.4 | 25.3 | 34.2 | 27.2 | 32.9 a | 7.1 |
| As+ | 30.7 | 28.8 | 26.4 | 22.7 | 31.5 | 28.3 | 28.1 b | ||
| Mean | 31.2 ab | 32.6 ab | 34.4 a | 24.0 c | 32.8 a | 27.7 bc | |||
| LWR (g g−1 DW) | As− | 0.243 | 0.267 | 0.270 | 0.223 | 0.230 | 0.183 | 0.236 a | 0.034 |
| As+ | 0.217 | 0.213 | 0.197 | 0.190 | 0.217 | 0.187 | 0.203 b | ||
| Mean | 0.230 ab | 0.240 a | 0.233 a | 0.207 ab | 0.223 bc | 0.185 c | |||
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo” (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| SPAD | As− | 55.7 | 56.4 | 54.7 | 56.1 | 52.9 | 55.9 | 55.3 b | NS |
| As+ | 57.5 | 59.3 | 58.1 | 61.7 | 58.6 | 60.8 | 59.3 a | ||
| Mean | 56.6 a | 57.8 a | 56.4 a | 58.9 a | 55.7 a | 58.4 a | |||
| AN (μmol CO2 m−2 s−1) | As− | 25.3 | 25.0 | 26.7 | 27.7 | 25.1 | 27.7 | 26.2 a | 2.2 |
| As+ | 27.7 | 23.2 | 25.6 | 22.7 | 25.4 | 29.1 | 25.6 a | ||
| Mean | 26.5 b | 24.1 c | 26.1 b | 25.2 bc | 25.2 bc | 28.4 a | |||
| Gs (μmol CO2 m−2 s−1) | As− | 240 | 273 | 275 | 255 | 190 | 330 | 261 a | 43 |
| As+ | 247 | 385 | 207 | 230 | 245 | 300 | 269 a | ||
| Mean | 243 b | 329 a | 241 b | 243 b | 218 b | 315 a | |||
| WUE (μmol CO2 μmol−1 H2O m−2 s−1) | As− | 3.0 | 3.0 | 2.8 | 3.5 | 3.0 | 2.8 | 3.0 b | 0.3 |
| As+ | 3.4 | 2.8 | 3.6 | 3.0 | 3.2 | 3.3 | 3.2 a | ||
| Mean | 3.2 a | 2.9 b | 3.2 a | 3.3 a | 3.1 ab | 3.1 ab | |||
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo” (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| Asroot | As− | 11.84 | 7.10 | 11.65 | 6.26 | 8.09 | 8.35 | 8.88 b | 77.33 |
| As+ | 1031.53 | 1090.73 | 1633.57 | 369.10 | 732.75 | 805.88 | 943.93 a | ||
| Mean | 521.68 b | 548.91 b | 822.61 a | 187.68 d | 370.42 c | 407.12 c | |||
| Asshoot | As− | 0.71 | 0.86 | 0.77 | 0.59 | 0.47 | 0.60 | 0.67 b | 1.73 |
| As+ | 23.71 | 25.50 | 23.81 | 26.20 | 23.90 | 22.35 | 24.24 a | ||
| Mean | 12.21 ab | 13.18 a | 12.29 ab | 13.39 a | 12.18 ab | 11.48 b | |||
| Aspulp | As− | 0.86 | 0.14 | 0.46 | 0.29 | 0.30 | 0.60 | 0.44 b | 0.25 |
| As+ | 2.72 | 3.16 | 2.99 | 2.98 | 3.46 | 3.19 | 3.08 a | ||
| Mean | 1.79 ab | 1.65 b | 1.72 ab | 1.64 b | 1.88 a | 1.89 a | |||
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo” (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| Asinorganic | As− | 60.1 | 54.1 | 48.0 | 46.0 | 40.1 | 38.2 | 47.8 a | 12.7 |
| As+ | 48.6 | 38.9 | 31.0 | 80.7 | 71.2 | 77.2 | 57.9 b | ||
| Mean | 54.3 bc | 46.5 cd | 39.5 d | 63.4 a | 55.6 ab | 57.7 ab | |||
| AsIIIroot | As− | 1.94 | 0.96 | 1.25 | 0.01 | 0.09 | 0.18 | 0.74 b | 71.89 |
| As+ | 235.98 | 168.31 | 150.07 | 82.26 | 287.48 | 577.42 | 250.25 a | ||
| Mean | 118.96 bc | 84.64 cd | 75.66 cd | 41.14 d | 143.79 b | 288.80 a | |||
| AsVroot | As− | 5.17 | 2.88 | 4.35 | 2.87 | 3.15 | 3.01 | 3.57 b | 102.15 |
| As+ | 264.86 | 254.95 | 350.29 | 215.72 | 233.87 | 44.86 | 227.42 a | ||
| Mean | 135.02 a | 128.91 a | 177.32 a | 109.30 a | 118.51 a | 23.94 b | |||
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| BAFroot | As− | 5917.67 | 3547.86 | 5824.57 | 3130.51 | 4045.17 | 4176.02 | 4440.30 a | 370.18 |
| As+ | 271.45 | 287.04 | 429.89 | 97.13 | 192.83 | 212.07 | 248.40 b | ||
| Mean | 3094.56 a | 1917.45 c | 3127.23 a | 1613.82 d | 2119.00 bc | 2194.05 b | |||
| BAFshoot | As− | 355.46 | 429.53 | 383.63 | 295.42 | 235.89 | 301.22 | 333.52 a | 50.54 |
| As+ | 6.24 | 6.71 | 6.26 | 6.89 | 6.29 | 5.88 | 6.38 b | ||
| Mean | 180.85 bc | 218.12 a | 194.95 ab | 151.16 cd | 121.09 d | 153.55 | |||
| BAFpulp | As− | 427.64 | 68.88 | 229.97 | 146.70 | 151.36 | 299.31 | 220.64 a | 15.50 |
| As+ | 0.72 | 0.83 | 0.79 | 0.78 | 0.91 | 0.84 | 0.81 b | ||
| Mean | 214.18 a | 34.86 e | 115.38 c | 73.74 d | 76.13 d | 150.07 b | |||
| TFshoot | As− | 0.060 | 0.121 | 0.066 | 0.095 | 0.058 | 0.074 | 0.079 a | 0.014 |
| As+ | 0.023 | 0.024 | 0.015 | 0.071 | 0.033 | 0.028 | 0.032 b | ||
| Mean | 0.042 cd | 0.072 b | 0.040 d | 0.083 a | 0.046 cd | 0.051 c | |||
| TFpulp | As− | 0.073 | 0.019 | 0.040 | 0.048 | 0.037 | 0.073 | 0.048 a | 0.007 |
| As+ | 0.003 | 0.003 | 0.002 | 0.008 | 0.005 | 0.004 | 0.004 b | ||
| Mean | 0.038 a | 0.011 d | 0.021 c | 0.028 b | 0.021 c | 0.039 a | |||
| Variable | Rootstock | Mean | LSDinteraction (p = 0.05) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| “Proteo” (Control) | “Dinero” | “Magnus” | “RS841” | “Shintoza” | “Strong Tosa” | ||||
| Proot | As− | 3527 | 3566 | 14362 | 12959 | 12535 | 20875 | 11304 b | 3111 |
| As+ | 31390 | 17059 | 22938 | 10379 | 19210 | 22343 | 20553 a | ||
| Mean | 17458 bc | 10312 d | 18650 b | 11669 d | 15872 c | 21609 a | |||
| Pshoot | As− | 7038 | 7131 | 6871 | 6039 | 6465 | 6538 | 6681 a | 564 |
| As+ | 6434 | 5064 | 4782 | 5563 | 5861 | 5786 | 5582 b | ||
| Mean | 6736 a | 6098 b | 5827 b | 5801 b | 6163 b | 6162 b | |||
| Ppulp | As− | 4745 | 590 | 3827 | 4119 | 4110 | 3996 | 3565 b | 521 |
| As+ | 5151 | 5062 | 5161 | 3797 | 4561 | 5199 | 4822 a | ||
| Mean | 4948 a | 2826 d | 4494 b | 3958 c | 4335 b | 4597 ab | |||
| Trait | Principal Component Coefficients | |
|---|---|---|
| First | Second | |
| Asroot | 0.556 | 0.830 |
| Asshoot | 0.589 | −0.367 |
| Aspulp | 0.586 | −0.419 |
| Eigenvalue | 2.75 | 0.21 |
| Explained Variability (%) | 91.91 | 7.07 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allevato, E.; Mauro, R.P.; Stazi, S.R.; Marabottini, R.; Leonardi, C.; Ierna, A.; Giuffrida, F. Arsenic Accumulation in Grafted Melon Plants: Role of Rootstock in Modulating Root-To-Shoot Translocation and Physiological Response. Agronomy 2019, 9, 828. https://doi.org/10.3390/agronomy9120828
Allevato E, Mauro RP, Stazi SR, Marabottini R, Leonardi C, Ierna A, Giuffrida F. Arsenic Accumulation in Grafted Melon Plants: Role of Rootstock in Modulating Root-To-Shoot Translocation and Physiological Response. Agronomy. 2019; 9(12):828. https://doi.org/10.3390/agronomy9120828
Chicago/Turabian StyleAllevato, Enrica, Rosario Paolo Mauro, Silvia Rita Stazi, Rosita Marabottini, Cherubino Leonardi, Anita Ierna, and Francesco Giuffrida. 2019. "Arsenic Accumulation in Grafted Melon Plants: Role of Rootstock in Modulating Root-To-Shoot Translocation and Physiological Response" Agronomy 9, no. 12: 828. https://doi.org/10.3390/agronomy9120828
APA StyleAllevato, E., Mauro, R. P., Stazi, S. R., Marabottini, R., Leonardi, C., Ierna, A., & Giuffrida, F. (2019). Arsenic Accumulation in Grafted Melon Plants: Role of Rootstock in Modulating Root-To-Shoot Translocation and Physiological Response. Agronomy, 9(12), 828. https://doi.org/10.3390/agronomy9120828







