Enhancing Greenhouse Tomato-Crop Productivity by Using Brassica macrocarpa Guss. Leaves for Controlling Root-Knot Nematodes
Abstract
:1. Introduction
2. Materials and Methods
2.1. B. Macrocarpa Characterization and Production
2.2. Glucosinolate Quantitative Analysis
2.3. Tomato Experiment Field
2.4. Sinigrin Effect for Controlling Root Knot Nematodes (Meloidogyne spp.)
2.5. Root Nematode Infestation and Fruit Yield and Quality
2.6. Statistical Analysis
3. Results
3.1. B. Macrocarpa Characterization and Production
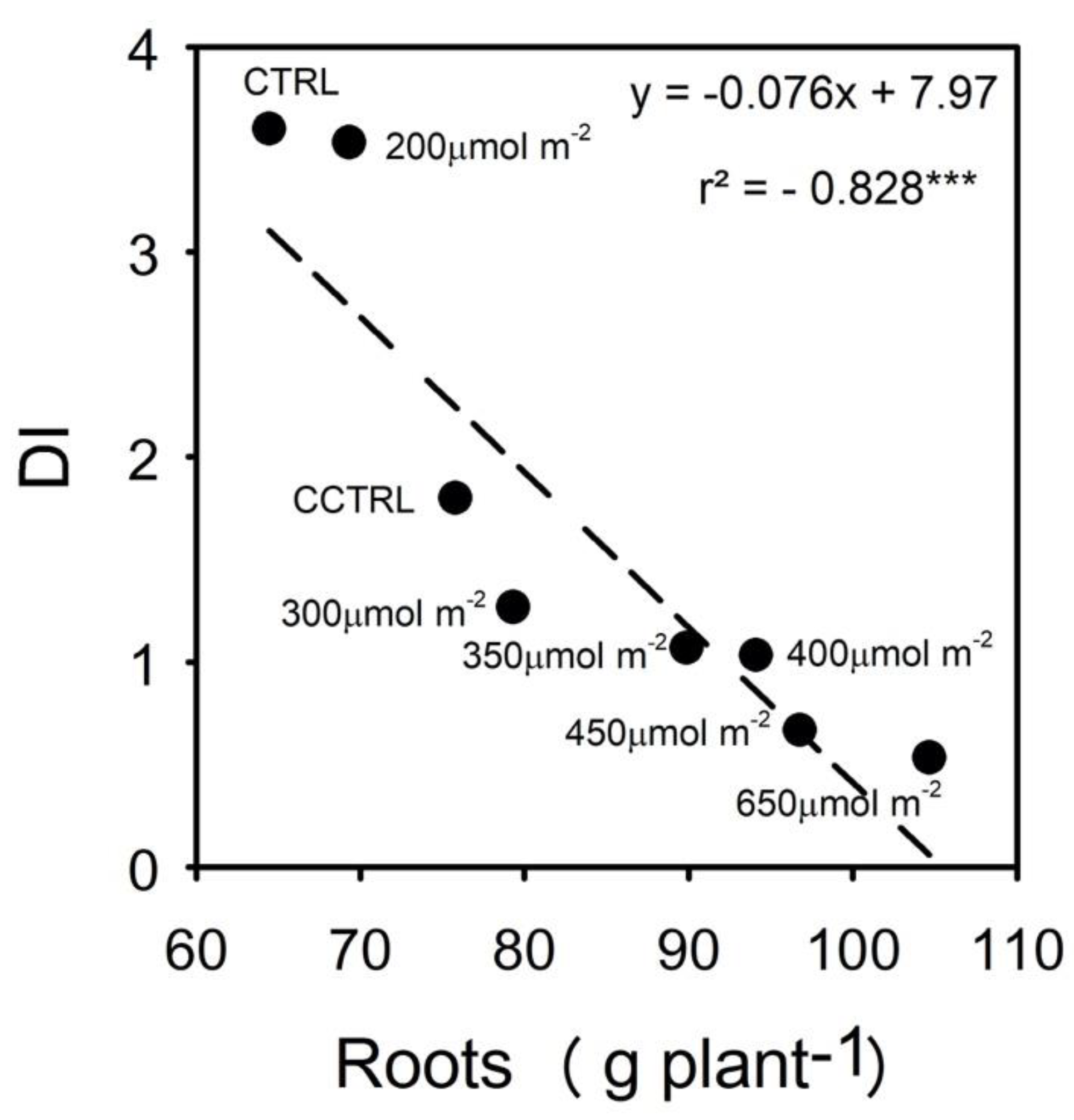
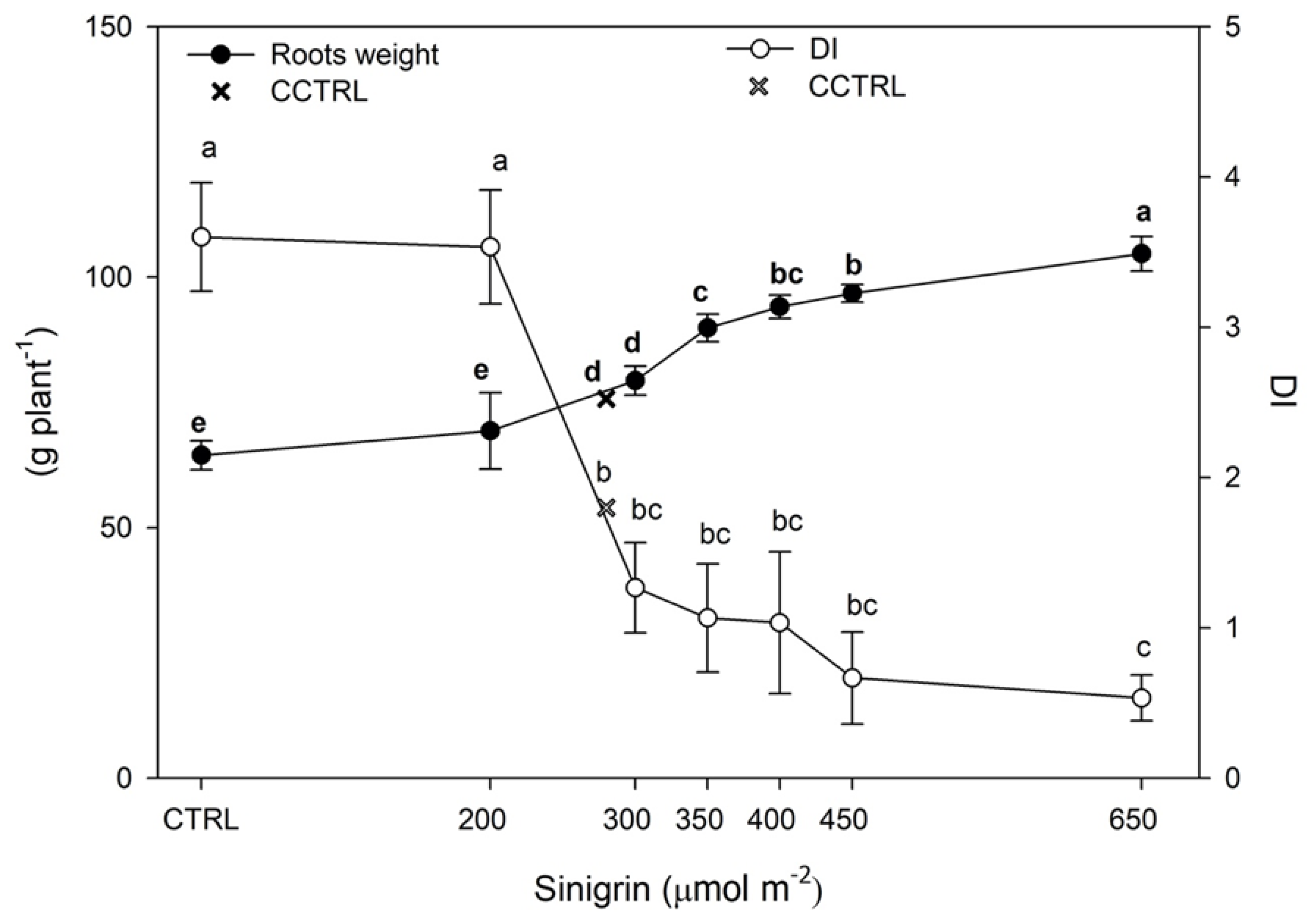
3.2. Root Nematodes Infestation and Fruit Yield and Quality
3.3. Fruit-Quality Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Faostat. Available online: http://www.fao.org/faostat/en/#home (accessed on 9 October 2019).
- ISTAT. Available online: https://www.istat.it/ (accessed on 9 October 2019).
- Collange, B.; Navarrete, M.; Peyre, G.; Mateille, T.; Tchamitchian, M. Root-knot nematode (Meloidogyne) management in vegetable crop production: The challenge of an agronomic system analysis. Crop Prot. 2011, 30, 1251–1262. [Google Scholar] [CrossRef]
- Argento, S.; Raccuia, S.A.; Melilli, M.G.; Toscano, V.; Branca, F. Brassicas and their glucosinolate content for the biological control of root-knot nematodes in protected cultivation. Acta Hortic. 2013, 1005, 539–544. [Google Scholar] [CrossRef]
- Onkendi, E.M.; Kariuki, G.M.; Marais, M.; Moleleki, L.N. The threat of root-knot nematodes (Meloidogyne spp.) in Africa: A review. Plant Pathol. 2014, 63, 727–737. [Google Scholar] [CrossRef]
- Mocali, S.; Landi, S.; Curto, G.; Dallavalle, E.; Infantino, A.; Colzi, C.; d’Errico, G.; Roversi, P.F.; D’Avino, L.; Lazzeri, L. Resilience of soil microbial and nematode communities after biofumigant treatment with defatted seed meals. Ind. Crops Prod. 2015, 75, 79–90. [Google Scholar] [CrossRef]
- Zhou, L.; Yuen, G.; Wang, Y.; Wei, L.; Ji, G. Evaluation of bacterial biological control agents for control of root-knot nematode disease on tomato. Crop Prot. 2016, 84, 8–13. [Google Scholar] [CrossRef]
- Ntalli, N.; Caboni, P. A review of isothiocyanates biofumigation activity on plant parasitic nematodes. Phytochem. Rev. 2017, 16, 827–834. [Google Scholar] [CrossRef]
- Matthiessen, J.; Kirkegaard, J. Biofumigation and enhanced biodegradation: Opportunity and challenge in soilborne pest and disease management. CRC Crit. Rev. Plant Sci. 2006, 25, 235–265. [Google Scholar] [CrossRef]
- Villalta, O.; Wite, D.; Riches, D.A.; Guiano, J.; Chandolu, V.; Scoble, C.; Donald, C.; Porter, I.J.; Mattner, S.W. The Concentration of 2-Propenyl Glucosinolate in Biofumigant Crops Influences Their Anti-Fungal Activity (In-Vitro) against Soil-Borne Pathogens. J. Agric. Chem. Environ. 2016, 5, 38–45. [Google Scholar] [CrossRef]
- D’Addabbo, T.; Laquale, S.; Lovelli, S.; Candido, V.; Avato, P. Biocide plants as a sustainable tool for the control of pests and pathogens in vegetable cropping systems. Ital. J. Agron. 2014, 9, 137–145. [Google Scholar] [CrossRef]
- Lazzeri, L.; Leoni, O.; Manici, L.M. Biocidal plant dried pellets for biofumigation. Ind. Crops Prod. 2004, 20, 59–65. [Google Scholar] [CrossRef]
- Youssef, M.M.A.; Lashein, A.M.S. Effect of cabbage (brassica oleracea) leaf residue as a biofumigant, on root knot nematode, meloidogyne incognita infecting tomato. J. Plant Prot. Res. 2013, 53, 271–274. [Google Scholar] [CrossRef]
- Mojtahedi, H.; Santo, G.S.; Hang, A.N.; Wilson, J.H. Suppression of Root-knot Nematode Populations with Selected Rapeseed Cultivars as Green Manure 1. J. Nematol. 1991, 23, 170–174. [Google Scholar] [PubMed]
- Bangarwa, S.K.; Norsworthy, J.K. Brassicaceae Cover-Crop Effects on Weed Management in Plasticulture Tomato. J. Crop Improv. 2014, 28, 145–158. [Google Scholar] [CrossRef]
- Salem, M.F.; Mahdy, M.E. Suppression of root-knot nematode through innovative mustard biofumigation. Future Food J. Food Agric. Soc. 2015, 3, 41–50. [Google Scholar]
- Avato, P.; D’Addabbo, T.; Leonetti, P.; Argentieri, M.P. Nematicidal potential of Brassicaceae. Phytochem. Rev. 2013, 12, 791–802. [Google Scholar] [CrossRef]
- Galletti, S.; Bagatta, M.; Branca, F.; Argento, S.; De Nicola, G.R.; Cianchetta, S.; Iori, R.; Ninfali, P. Isatis canescens is a rich source of glucobrassicin and other health-promoting compounds. J. Sci. Food Agric. 2015, 95, 158–164. [Google Scholar] [CrossRef]
- Al Shahawany, A.W.; Al Hattab, Z.N.; Al Tahhan, S.F. Qualitative and Quantitative Analysis of Sinigrin in Different Parts In Vitro and In Vivo of Brassica nigra Plants. Biomedicine 2016, 4, 19–24. [Google Scholar]
- Hanschen, F.S.; Yim, B.; Winkelmann, T.; Smalla, K.; Schreiner, M. Degradation of biofumigant isothiocyanates and allyl glucosinolate in soil and their effects on the microbial community composition. PLoS ONE 2015, 10, e0132931. [Google Scholar] [CrossRef]
- Dutta, T.K.; Khan, M.R.; Phani, V. Plant-parasitic nematode management via biofumigation using brassica and non-brassica plants: Current status and future prospects. Curr. Plant Biol. 2019, 17, 17–32. [Google Scholar] [CrossRef]
- Gimsing, A.L.; Kirkegaard, J.A. Glucosinolates and biofumigation: Fate of glucosinolates and their hydrolysis products in soil. Phytochem. Rev. 2009, 8, 299–310. [Google Scholar] [CrossRef]
- De Nicola, G.R.; D’Avino, L.; Curto, G.; Malaguti, L.; Ugolini, L.; Cinti, S.; Patalano, G.; Lazzeri, L. A new biobased liquid formulation with biofumigant and fertilising properties for drip irrigation distribution. Ind. Crops Prod. 2013, 42, 113–118. [Google Scholar] [CrossRef]
- Sukovata, L.; Jaworski, T.; Kolk, A. Efficacy of Brassica juncea granulated seed meal against Melolontha grubs. Ind. Crops Prod. 2015, 70, 260–265. [Google Scholar] [CrossRef]
- Aydınlı, G.; Mennan, S. Biofumigation Studies by Using Raphanus sativus and Eruca sativa as a Winter Cycle Crops to Control Root-knot Nematodes. Braz. Arch. Biol. Technol. 2018, 61. [Google Scholar] [CrossRef]
- Branca, F.; Li, G.; Goyal, S.; Quiros, C.F. Survey of aliphatic glucosinolates in Sicilian wild and cultivated Brassicaceae. Phytochemistry 2002, 59, 717–724. [Google Scholar] [CrossRef]
- Branca, F.; Argento, S.; Tribulato, A. Assessing genetic reserves in Sicily (Italy): The Brassica wild relatives case study. In Agrobiodiversity Conservation Securing the Diversity of Crop Wild Relatives and Landraces; CABI: Wallingford, UK, 2012; pp. 52–58. [Google Scholar]
- Maxted, N.; Ehsan Dulloo, M.; Ford-Lloyd, B.V.; Frese, L.; Iriondo, J.M.; Pinheiro de Carvalho, M.A.A. (Eds.) Agrobiodiversity Conservation: Securing the Diversity of Crop Wild Relatives and Landraces; CABI: Wallingford, UK, 2012; ISBN 9781845938512. [Google Scholar]
- Branca, F.; Chiarenza, G.L.; Ragusa, L.; Argento, S. Morphological characterization of the ECPGR wild Brassica species collection. Acta Hortic. 2013, 1005, 157–164. [Google Scholar] [CrossRef]
- Mitsiogianni, M.; Koutsidis, G.; Mavroudis, N.; Trafalis, D.T.; Botaitis, S.; Franco, R.; Zoumpourlis, V.; Amery, T.; Galanis, A.; Pappa, A.; et al. The Role of Isothiocyanates as Cancer Chemo-Preventive, Chemo-Therapeutic and Anti-Melanoma Agents. Antioxidants 2019, 8, 106. [Google Scholar] [CrossRef]
- Terzo, M.N.; Pezzino, F.; Amodeo, L.; Catalano, D.; Viola, M.; Tribulato, A.; Travali, S.; Branca, F. Evaluation of a sicilian black broccoli extract on in vitro cell models. Acta Hortic. 2018, 135–142. [Google Scholar] [CrossRef]
- EEC. EEC Regulation No 1864/90, Enclosure VIII, Oilseeds—Determination of glucosinolates—High performance liquid chromatography. Off. J. Eur. Commun. 1990, L170, 27–34. [Google Scholar]
- Wathelet, J.; Iori, R.; Leoni, O.; Quinsac, A.; Palmieri, S. Guidelines for glucosinolate analysis in green tissues used for biofumigation. Agroindustria 2004, 3, 257–266. [Google Scholar]
- Torne, G. Principles of Nematology; McGraw-Hill Book Company: London, UK, 1961; ISBN 9780198565260. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. FAO Irrigation and Drainage Paper No. 56—Crop Evapotranspiration; FAO: Rome, Italy, 1998. [Google Scholar]
- Lamberti, F. Primi risultati di prove di lotta nematocida su tabacchi levantini in provincia di Lecce. Table 1971, 738, 5–10. [Google Scholar]
- Snedecor, G.W.; Cochran, W.G. Statistical Methods, 7th ed.; The Iowa State University Press: Ames, IA, USA, 1989; ISBN 0 8138 1561 6. [Google Scholar]
- Branca, F.; Tribulato, A. Brassica Macrocarpa; The IUCN Red List of Threatened Species: Cambridge, UK, 2011. [Google Scholar]
- Galletti, S.; Fornasier, F.; Cianchetta, S.; Lazzeri, L. Soil incorporation of brassica materials and seed treatment with Trichoderma harzianum: Effects on melon growth and soil microbial activity. Ind. Crops Prod. 2015, 75, 73–78. [Google Scholar] [CrossRef]
- Gimsing, A.L.; Kirkegaard, J.A. Glucosinolate and isothiocyanate concentration in soil following incorporation of Brassica biofumigants. Soil Biol. Biochem. 2006, 38, 2255–2264. [Google Scholar] [CrossRef]
- Henderson, D.R.; Riga, E.; Ramirez, R.A.; Wilson, J.; Snyder, W.E. Mustard biofumigation disrupts biological control by Steinernema spp. nematodes in the soil. Biol. Control 2009, 48, 316–322. [Google Scholar] [CrossRef]
- Daneel, M.; Engelbrecht, E.; Fourie, H.; Ahuja, P. The host status of Brassicaceae to Meloidogyne and their effects as cover and biofumigant crops on root-knot nematode populations associated with potato and tomato under South African field conditions. Crop Prot. 2018, 110, 198–206. [Google Scholar] [CrossRef]
- Ntalli, N.; Monokrousos, N.; Rumbos, C.; Kontea, D.; Zioga, D.; Argyropoulou, M.D.; Menkissoglu-Spiroudi, U.; Tsiropoulos, N.G. Greenhouse biofumigation with Melia azedarach controls Meloidogyne spp. and enhances soil biological activity. J. Pest Sci. 2018, 91, 29–40. [Google Scholar] [CrossRef]



| IBPGR and UPOCV Descriptors [27] | ||
|---|---|---|
| Plant height (cm) | 53.4 ± 7.6 | 65.3 |
| Number of secondary branches (plant−1) | 5.2 ± 2.0 | 7.0 |
| Number of leaves (plant−1) | 49.6 ± 3.1 | 50.6 |
| Leaf-blade width (cm) | 11.3 ± 0.9 | 5.7 |
| Leaf-blade length (cm) | 30.0 ± 4.6 | 26.3 |
| Petiole length (cm) | 13.6 ± 2.7 | 24.4 |
| Number of siliques (plant−1) | 50.0 ± 7.0 | |
| Number of seeds (silique−1) | 10.0 ± 1.4 | |
| Seed yield (g plant−1) | 11.0 ± 1.5 | |
| Fresh-leaf yield (g plant−1) | 473 ± 35 | |
| Dry-matter content (g 100−1 f.w.) | 13.4 ± 0.6 | |
| Dry-leaf sinigrin content (µmol g −1 d.m.) | 5.4 ± 0.05 | |
| Fresh-leaf sinigrin content (µmol g −1 f.w.) | 5.9 ± 0.06 |
| Sinigrin Dosage | Larvae of Second Age | |
|---|---|---|
| T0 | T1 | |
| CTRL | 1416 ± 50.2 | 1250 ± 35.4 a |
| CCTRL | 385 ± 15.2 b | |
| 200 | 1100 ± 28.0 a | |
| 300 | 400 ± 11.3 b | |
| 350 | 370 ± 7.8 b | |
| 400 | 270 ± 3.8 c | |
| 450 | 180 ± 3.1 d | |
| 650 | 100 ± 3.5 e | |
| Mean | 507 ± 14.2 | |
| LSD (p < 0.05) | 14.5 | |
| p | *** | |
| Sinigrin Dosage | Fruits | Clusters § | Fruit Weight | Chromatic Parameters (CIE) | Soluble Sugars | Dry Matter | pH | |
|---|---|---|---|---|---|---|---|---|
| (n) | (n) | (g) | (L) * | a/b | °Brix | % | ||
| CTRL | 13.9 ± 1.1 e | 4.9 ± 0.5 a | 64.5 ± 5.5 b,c | 39.6 ± 3.9 a | 1.69 ± 0.26 a | 7.1 ± 0.2 a,b | 7.8 ± 0.1 b,c | 5.9 ± 0.5 a |
| CCTRL | 26.2 ± 0.2 d | 4.5 ± 0.1 a | 67.8 ± 2.3 a,b | 39.9 ± 1.7 a | 1.90 ± 0.18 a | 6.8 ± 0.1 b | 7.7 ± 0.3 c | 6.4 ± 0.3 a |
| 200 | 15.3 ± 1.2 e | 4.6 ± 0.2 a | 63.7 ± 1.9 b,c | 36.9 ± 1.9 a | 1.50 ± 0.20 a | 7.3 ± 0.1 a,b | 8.2 ± 0.3 a,b,c | 5.7 ± 0.5 a |
| 300 | 30.2 ± 0.3 c,d | 4.5 ± 0.3 a | 74.3 ± 2.6 a | 40.6 ± 3.2 a | 1.58 ± 0.21 a | 7.3 ± 0.5 a,b | 8.3 ± 0.4 a,b,c | 6.5 ± 0.4 a |
| 350 | 40.1 ± 0.7 c | 4.4 ± 0.2 a | 59.1 ± 2.8 c | 38.9 ± 2.6 a | 1.51 ± 0.20 a | 7.5 ± 0.4 a | 8.6 ± 0.5 a,b | 6.7 ± 0.3 a |
| 400 | 38.5 ± 1.7 c | 4.6 ± 0.1 a | 62.6 ± 5.6 b,c | 37.3 ± 3.0 a | 1.59 ± 0.30 a | 7.4 ± 0.5 a,b | 8.5 ± 0.3 a,b,c | 5.9 ± 0.2 a |
| 450 | 44.1 ± 1.7 b | 4.6 ± 0.2 a | 70.1 ± 4.9 a,b | 39.4 ± 4.2 a | 1.75 ± 0.32 a | 7.3 ± 0.2 a,b | 8.3 ± 0.2 a,b,c | 6.1 ± 0.2 a |
| 650 | 54.3 ± 1.0 a | 4.6 ± 0.1 a | 66.0 ± 3.6 b,c | 39.0 ± 2.9 a | 1.72 ± 0.34 a | 7.5 ± 0.4 a | 8.8 ± 0.5 a | 6.3 ± 0.3 a |
| Mean | 32.8 | 4.6 | 66.0 | 38.9 | 1.65 | 7.29 | 8.29 | 6.19 |
| LSD (p <0.05) | 6.96 | 0.43 | 6.78 | 5.27 | 0.45 | 0.58 | 0.80 | 0.96 |
| p | *** | ns | ** | ns | ns | * | * | ns |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Argento, S.; Melilli, M.G.; Branca, F. Enhancing Greenhouse Tomato-Crop Productivity by Using Brassica macrocarpa Guss. Leaves for Controlling Root-Knot Nematodes. Agronomy 2019, 9, 820. https://doi.org/10.3390/agronomy9120820
Argento S, Melilli MG, Branca F. Enhancing Greenhouse Tomato-Crop Productivity by Using Brassica macrocarpa Guss. Leaves for Controlling Root-Knot Nematodes. Agronomy. 2019; 9(12):820. https://doi.org/10.3390/agronomy9120820
Chicago/Turabian StyleArgento, Sergio, Maria Grazia Melilli, and Ferdinando Branca. 2019. "Enhancing Greenhouse Tomato-Crop Productivity by Using Brassica macrocarpa Guss. Leaves for Controlling Root-Knot Nematodes" Agronomy 9, no. 12: 820. https://doi.org/10.3390/agronomy9120820
APA StyleArgento, S., Melilli, M. G., & Branca, F. (2019). Enhancing Greenhouse Tomato-Crop Productivity by Using Brassica macrocarpa Guss. Leaves for Controlling Root-Knot Nematodes. Agronomy, 9(12), 820. https://doi.org/10.3390/agronomy9120820








