Screening and Evaluation of Essential Oils from Mediterranean Aromatic Plants against the Mushroom Cobweb Disease, Cladobotryum mycophilum
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Essential Oil Extraction
2.3. Components Identification and Quantification
2.4. Fungal Isolates
2.5. In Vitro Antifungal Activity and Selectivity Index of the Essential Oils on Mycelium Growth of Cladobotryum Mycophilum and Agaricus bisporus
2.6. Effectiveness of EOs Treatments in Mushroom Growing Trials with Cladobotryum Mycophilum Inoculation
2.7. Effect of EOs Treatments on Agaricus bisporus Yield
3. Results
3.1. Chemical Composition of the Essential Oils
3.2. In Vitro Antifungal Activity and Selectivity Index of the Essential Oils on Mycelium Growth of Cladobotryum mycophilum and Agaricus bisporus
3.3. Effectiveness of EOs Treatments in Mushroom Growing Trials with Cladobotryum mycophilum Inoculation
3.4. Effect of EOs Treatments on Agaricus bisporus Yield
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grogan, H.M.; Gaze, R.H. Fungicide resistance among Cladobotryum spp.–causal agents of cobweb disease of the edible mushroom Agaricus bisporus. Mycol. Res. 2000, 104, 357–364. [Google Scholar] [CrossRef]
- Fletcher, J.T.; Gaze, R.H. Mushroom Pest and Disease Control; Manson Publishing: London, UK, 2008. [Google Scholar]
- Largeteau, M.L.; Savoie, J.M. Microbially induced diseases of Agaricus bisporus: Biochemical mechanisms and impact on commercial mushroom production. Appl. Microbiol. Biotechnol. 2010, 86, 63–73. [Google Scholar] [CrossRef]
- Carrasco, J.; Navarro, M.J.; Gea, F.J. Cobweb, a serious pathology in mushroom crops: A review. Span. J. Agric. Res. 2017, 15, 19. [Google Scholar] [CrossRef]
- Chakwiya, A.; Van der Linde, E.J.; Chidamba, L.; Korsten, L. Diversity of Cladobotryum mycophilum isolates associated with cobweb disease of Agaricus bisporus in the south African mushroom industry. Eur. J. Plant Pathol. 2019, 154, 767–776. [Google Scholar] [CrossRef]
- Back, C.G.; Lee, C.Y.; Seo, G.S.; Jung, H.Y. Characterization of species of Cladobotryum which cause cobweb disease in edible mushrooms grown in Korea. Mycobiology 2012, 40, 189–194. [Google Scholar] [CrossRef]
- Kim, M.K.; Lee, Y.H.; Cho, K.M.; Lee, J.Y. First report of cobweb disease caused by Cladobotryum mycophilum on the edible mushroom Pleurotus eryngii in Korea. Plant Dis. 2012, 96, 1374. [Google Scholar] [CrossRef] [PubMed]
- Gea, F.J.; Carrasco, J.; Suz, L.M.; Navarro, M.J. Characterization and pathogenicity of Cladobotryum mycophilum in Spanish Pleurotus eryngii mushroom crops and their sensitivity to fungicides. Eur. J. Plant Pathol. 2017, 147, 129–139. [Google Scholar] [CrossRef]
- Gea, F.J.; Navarro, M.J.; Suz, L.M. Cobweb disease on oyster culinary-medicinal mushroom (Pleurotus ostreatus) caused by the mycoparasite Cladobotryum mycophilum. J. Plant Pathol. 2019, 101, 349–354. [Google Scholar] [CrossRef]
- Carrasco, J.; Navarro, M.J.; Santos, M.; Diánez, F.; Gea, F.J. Identification, incidence and pathogenicity of Cladobotryum mycophilum, causal agent of cobweb disease on Agaricus bisporus mushroom crops in Spain. Ann. Appl. Biol. 2016, 168, 214–224. [Google Scholar] [CrossRef]
- Adie, B.; Grogan, H.; Archer, S.; Mills, P. Temporal and spatial dispersal of Cladobotryum conidia in the controlled environmental of a mushroom growing room. Appl. Environ. Microbiol. 2006, 72, 7212–7217. [Google Scholar] [CrossRef] [PubMed]
- Pyck, N.; Grogan, H. Fungal Diseases of Mushrooms and Their Control, MushTV Publications (Factsheet 04/15). 2015. Available online: http://horticulture.ahdb.org.uk/mushtv-publications (accessed on 12 May 2017).
- Carrasco, J.; Navarro, M.J.; Santos, M.; Gea, F.J. Effect of five fungicides with different modes of action on cobweb disease (Cladobotryum mycophilum) and mushroom yield. Ann. Appl. Biol. 2017, 171, 62–69. [Google Scholar] [CrossRef]
- Lang, G.; Buchbauer, G. A review on recent research results (2008–2010) on essential oils as antimicrobials and antifungals. A review. Flavour Fragr. J. 2012, 27, 13–39. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Coppola, R.; De Feo, V. Essential oils and antifungal activity. Pharmaceuticals 2017, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Diánez, F.; Santos, M.; Parra, C.; Navarro, M.J.; Blanco, R.; Gea, F.J. Screening of antifungal activity of twelve essential oils against eight pathogenic fungi of vegetables and mushroom. Lett. Appl. Microbiol. 2018, 67, 400–410. [Google Scholar] [CrossRef]
- Soković, M.; van Griensven, L.J.L.D. Antimicrobial activity of essential oils and their components against the three major pathogens of the cultivated button mushroom, Agaricus bisporus. Eur. J. Plant Pathol. 2006, 116, 211–224. [Google Scholar] [CrossRef]
- Mehrparvar, M.; Goltapeh, E.M.; Safaie, N.; Ashkani, S.; Hedesh, R.M. Antifungal activity of essential oils against mycelial growth of Lecanicillium fungicola var. fungicola and Agaricus bisporus. Ind. Crops Prod. 2016, 84, 391–398. [Google Scholar] [CrossRef]
- Santos, T.L.; Belan, L.L.; Zied, D.C.; Dias, E.S.; Alves, E. Essential oils in the control of dry bubble disease in white button mushroom. Cienc. Rural 2017, 47, e20160780. [Google Scholar] [CrossRef]
- Regnier, T.; Combrinck, S. In vitro and in vivo screening of essential oils for the control of wet bubble disease of Agaricus bisporus. S. Afr. J. Bot. 2010, 76, 681–685. [Google Scholar] [CrossRef]
- Idrees, M.; Sossah, F.L.; Yang, Y.; Li, D.; Li, S.; Fu, Y.; Li, Y. Identification of resistance to cobweb disease caused by Cladobotryum mycophilum in wild and cultivated strains of Agaricus bisporus and screening for bioactive botanicals. RSC Adv. 2019, 9, 14758–14765. [Google Scholar]
- Potočnik, I.; Vukojević, J.; Stajić, M.; Rekanović, E.; Stepanović, M.; Milijašević, S.; Todorović, B. Toxicity of biofungicide Timorex 66 EC to Cladobotryum dendroides and Agaricus bisporus. Crop Prot. 2010, 2, 290–294. [Google Scholar] [CrossRef]
- Santana, O.; Cabrera, R.; Giménez, C.; González-Coloma, A.; Sánchez-Vioque, R.; de los Mozos-Pascual, M.; Rodríguez-Conde, M.F.; Laserna-Ruiz, I.; Usano-Alemany, J.; Herraiz, D. Chemical and biological profiles of the essential oils from aromatic plants of agro-industrial interest in Castilla-La Mancha (Spain). Grasas Y Aceites 2012, 63, 214–222. [Google Scholar]
- Ortiz de Elguea-Culebras, G.; Sanchez-Vioque, R.; Berruga, M.I.; Herraiz-Peñalver, D.; González-Coloma, A.; Andrés, M.F.; Santana-Méridas, O. Biocidal potential and chemical composition of industrial essential oils from Hyssopus officinalis, Lavandula x intermedia var. super, and Santolina chamaecyparissus. Chem. Biodivers. 2018, 15, e1700313. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, Z.; Huang, Q.; Zhang, D. Antifungal activity of several essential oils and major components against wood-rot fungi. Ind. Crops Prod. 2017, 108, 278–285. [Google Scholar] [CrossRef]
- Council of Europe. European Pharmacopoeia, 3rd ed.; Council of Europe: Strasbourg, France, 1996. [Google Scholar]
- Adams, R.P. Quadrupole Mass Spectra of Compounds Listed in order of Their Retention Time on DB-5. Identification of Essential Oils Components by Gas Chromatography/Quadrupole Mass Spectroscopy; Allured Publishing Co.: Carol Stream, IL, USA, 2001; p. 456. [Google Scholar]
- Gams, W.; Hoozemans, A.C.M. Cladobotryum-Konidienformen von Hypomyces-Arten. Persoonia 1970, 6, 95–110. [Google Scholar]
- Rogerson, C.T.; Samuels, G.J. Agaricicolous species of Hypomyces. Mycologia 1994, 86, 839–866. [Google Scholar] [CrossRef]
- Robertson, J.L.; Preisler, J.K.; Russell, R.M. Polo Plus. Probit and Logit Analysis. User’s Guide; LeOra Software: Petulama, CA, USA, 2002. [Google Scholar]
- Gea, F.J.; Santos, M.; Diánez, F.; Tello, J.C.; Navarro, M.J. Effect of spent mushroom compost tea on mycelial growth and yield of button mushroom (Agaricus bisporus). World J. Microbiol. Biotechnol. 2012, 28, 2765–2769. [Google Scholar] [CrossRef] [PubMed]
- Chrysayi-Tokousbalides, M.; Kastanias, M.A.; Philippoussis, A.; Diamantopoulou, P. Selective fungitoxicity of famoxadone, tebuconazole and trifloxistrobin between Verticillium fungicola and Agaricus bisporus. Crop Prot. 2007, 26, 469–475. [Google Scholar] [CrossRef]
- Rosenhein, J.A.; Hoy, M. Confidence intervals for Abbott’s Formula correction of bioassay data for control response. J. Econ. Entomol. 1989, 82, 331–335. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Lee, S.O.; Choi, G.J.; Jang, K.S.; Lim, H.K.; Cho, K.Y.; Kim, J.C. Antifungal activity of five plant essential oils as fumigant against postharvest and soil borne plant pathogenic fungi. Plant Pathol. J. 2007, 23, 97–102. [Google Scholar] [CrossRef]
- Stević, T.; Berić, T.; Šavikin, K.; Soković, M.; Goƌevac, D.; Dimkić, I.; Stanković, S. Antifungal activity of selected essential oils against fungi isolated from medicinal plant. Ind. Crops Prod. 2014, 55, 116–122. [Google Scholar] [CrossRef]
- Lima, I.O.; de Oliveira Pereira, F.; de Oliveira, W.A.; de Oliveira Lima, E.; Menezes, E.A.; Cunha, F.A.; de Fátima, M.F.M.D. Antifungal activity and mode of action of carvacrol against Candida albicans strains. J. Essent. Oil Res. 2013, 25, 138–142. [Google Scholar] [CrossRef]
- Chavan, P.S.; Tupe, S.G. Antifungal activity and mechanism of action of carvacrol and thymol against vineyard and wine spoilage yeasts. Food Control 2014, 46, 115–120. [Google Scholar] [CrossRef]
- Moon, H.; Rhee, M.S. Synergism between carvacrol or thymol increases the antimicrobial efficacy of soy sauce with no sensory impact. Int. J. Food Microbiol. 2016, 217, 35–41. [Google Scholar] [CrossRef]
- Thomidis, T.; Filotheou, A. Evaluation of five essential oils as bio-fungicides on the control of Pilidiella granati rot in pomegranate. Crop Prot. 2016, 89, 66–71. [Google Scholar] [CrossRef]
- Ultree, A.; Bennink, M.H.J.; Moezelaar, R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar] [CrossRef]
- Tanović, B.; Potočnik, I.; Delibašic, G.; Ristić, M.; Kostić, M.; Marković, M. In vitro effect of essential oils from aromatic and medicinal plants on mushroom pathogens: Verticillium fungicola var. fungicola, Mycogone perniciosa, and Cladobotryum sp. Arch. Biol. Sci. 2009, 61, 231–237. [Google Scholar] [CrossRef]
- Luković, J.; Stepanović, M.; Todorović, B.; Milijašević-Marčić, S.; Duduk, N.; Vico, I.; Potočnik, I. Antifungal activity of cinnamon and clove essential oils against button mushroom pathogens Cladobotryum dendroides (Bull.) W. Gams & Hooz. and Lecanicillium fungicola var. fungicola (Preuss) Hasebrauk. Pestic. I Phytomed. 2018, 33, 19–26. [Google Scholar]
- Savoie, J.M. Changes in enzyme activities during early growth of the edible mushroom, Agaricus bisporus, in compost. Mycol. Res. 1998, 102, 1113–1118. [Google Scholar] [CrossRef]
- Chen, Y.; Chefetz, B.; Rosario, R.; Heemst, J.D.H.V.; Romaine, C.P.; Hatcher, P.G. Chemical nature and composition of compost during mushroom growth. Compost Sci. Util. 2000, 8, 347–359. [Google Scholar] [CrossRef]
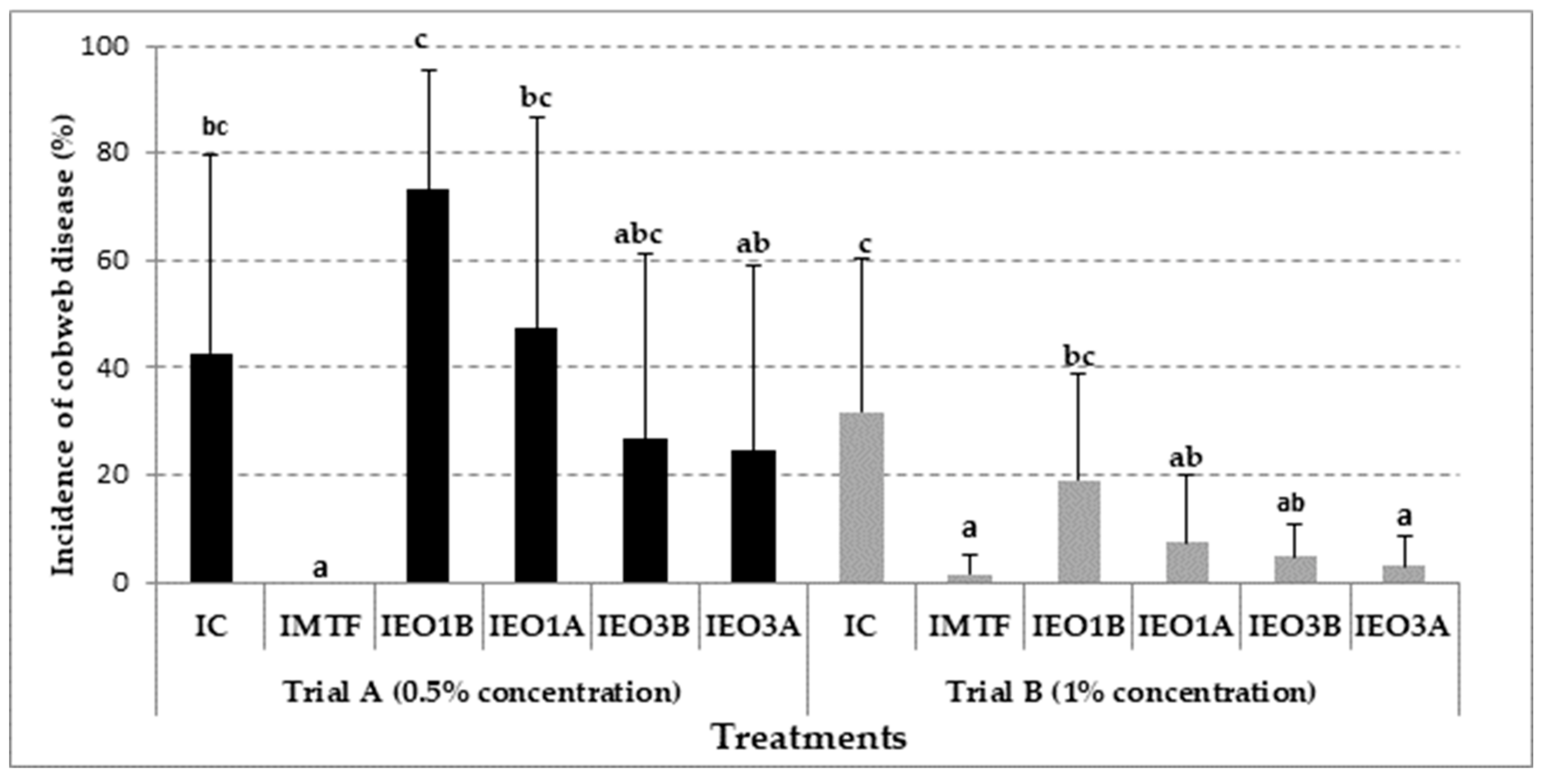
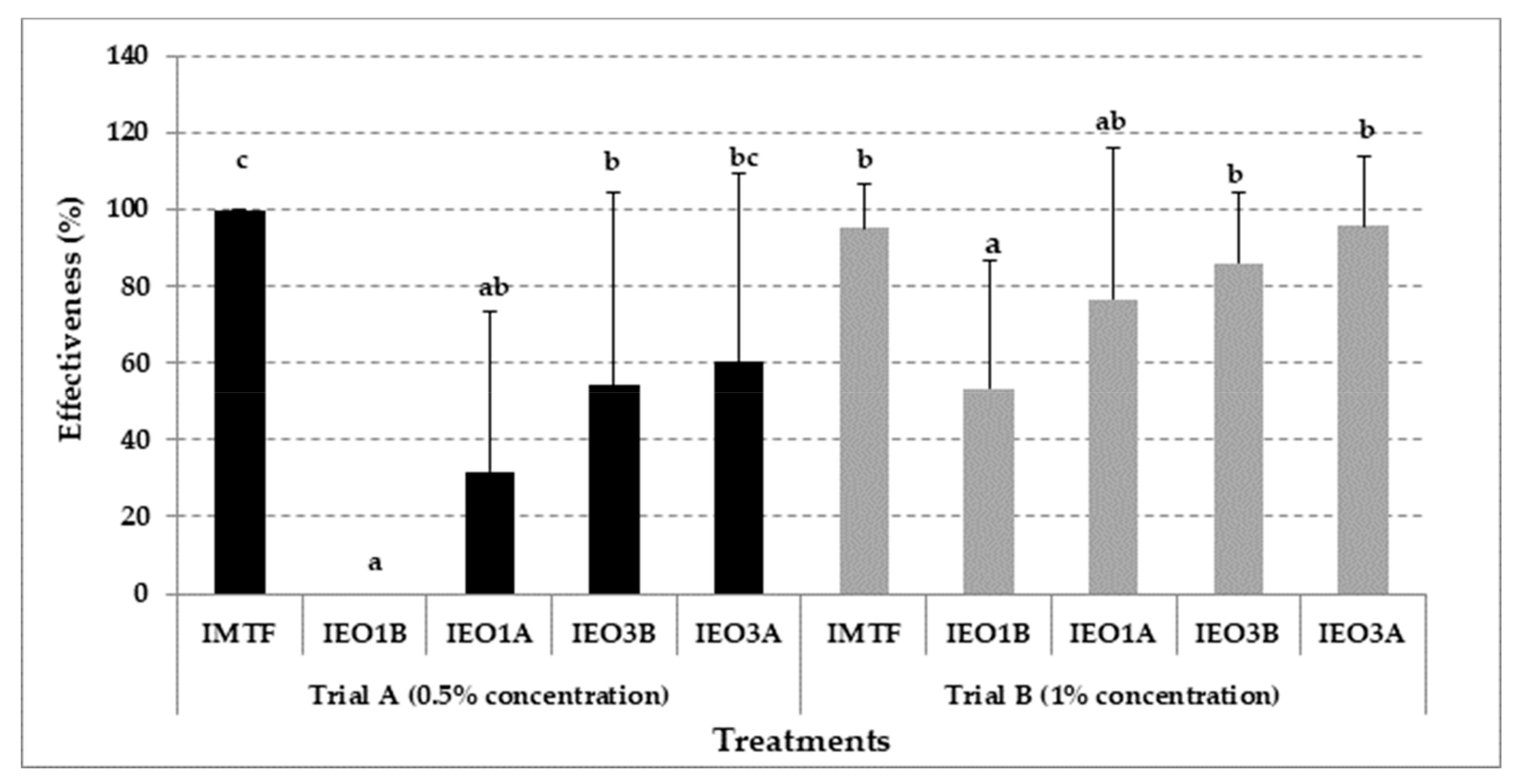
| Trials | Code | Inoculation (106 conidia m−2) a | Treatment b |
|---|---|---|---|
| A, B, and C | C | No | Only tap water. |
| A and B | IMFC | Yes | Vivando® at 1 ml L−1 on day 4. |
| A and B | IC | Yes | Only tap water. |
| A | IEO1B | Yes | Satureja montana essential oil at 0.5% applied 1 day before inoculation with Cladobotryum mycophilum, on day 10. |
| A | IEO1A | Yes | Satureja montana essential oil at 0.5% applied 2 days after inoculation with Cladobotryum mycophilum, on day 13. |
| A | IEO3B | Yes | Thymus vulgaris essential oil at 0.5% applied 1 day before inoculation with Cladobotryum mycophilum, on day 10. |
| A | IEO3A | Yes | Thymus vulgaris essential oil at 0.5% applied 2 days after inoculation with Cladobotryum mycophilum, on day 13. |
| B | IEO1B | Yes | Satureja montana essential oil at 1% applied 1 day before inoculation with Cladobotryum mycophilum, on day 10. |
| B | IEO1A | Yes | Satureja montana essential oil at 1% applied 2 days after inoculation with Cladobotryum mycophilum, on day 13. |
| B | IEO3B | Yes | Thymus vulgaris essential oil at 1% applied 1 day before inoculation with Cladobotryum mycophilum, on day 10. |
| B | IEO3A | Yes | Thymus vulgaris essential oil at 1% applied 2 days after inoculation with Cladobotryum mycophilum, on day 13. |
| C | MFC | No | Vivando® at 1 mL L−1 on day 4. |
| C | EO1B | No | Satureja montana essential oil at 1% applied on day 10. |
| C | EO1A | No | Satureja montana essential oil at 1% applied on day 13. |
| C | EO3B | No | Thymus vulgaris essential oil at 1% applied on day 10. |
| C | EO3A | No | Thymus vulgaris essential oil at 1% applied on day 13. |
| Main Compounds a | KIb | RTc | Lavandula × intermedia | Salvia lavandulifolia | Satureja montana | Thymus mastichina | Thymus vulgaris |
|---|---|---|---|---|---|---|---|
| α-Pinene | 943 | 11.40 | td | 11.39 | 7.86 | 2.33 | 1.73 |
| Camphene | 962 | 12.03 | t | 6.38 | 3.63 | t | t |
| β-Pinene + β-Myrcene | 991 | 12.97 | 1.22 | 13.99 | 8.40 | 4.42 | 2.68 |
| p-Cymene | 1033 | 14.52 | t | t | 11.54 | t | 29.67 |
| Limonene | 1038 | 14.66 | 2.18 | 8.72 | 3.53 | 2.46 | t |
| 1,8-Cineole | 1044 | 14.95 | 6.90 | 20.90 | 4.91 | 44.57 | 1.97 |
| γ-Terpinene | 1066 | 15.68 | -e | 1.4 | 6.79 | t | 7.55 |
| Linalool | 1102 | 17.12 | 28.75 | t | 2.1 | 29.86 | 4.59 |
| Camphor | 1165 | 19.32 | 8.49 | 12.60 | 10.14 | - | 1.54 |
| α-Terpineol | 1206 | 20.84 | t | t | t | 3.60 | t |
| Linalyl acetate | 1254 | 22.45 | 34.20 | - | - | t | t |
| Thymol | 1290 | 24.09 | - | - | 1.87 | - | 25.78 |
| Carvacrol | 1298 | 24.43 | - | - | 17.22 | - | 2.41 |
| trans-caryophyllene | 1441 | 28.71 | 1.86 | 4.45 | 3.36 | t | 2.76 |
| Essential Oil | Cladobotryum mycophilum (n = 12) | Agaricus bisporus (n = 4) | Selectivity Index a |
|---|---|---|---|
| ED50 (mg L−1) | ED50 (mg L−1) | ||
| Satureja montana | 42.8 ± 33.4 ab | 537.4 ± 192.1 b | 0.08 |
| Thymus vulgaris | 35.5 ± 23.0 a | 389.7 ± 131.5 b | 0.09 |
| Salvia lavandulifolia | 598.4 ± 430.9 b | 587.1 ± 177.4 b | 1.02 |
| Thymus mastichina | 627.3 ± 183.9 b | 175.7 ± 60.2 a | 3.57 |
| Lavandula × intermedia | 784.2 ± 460.5 b | 146.6 ± 39.2 a | 5.35 |
| Treatments a | Mushroom Yield (kg m−2) | Biological Efficiency (kg dt−1 Compost) | Earliness (Days from Casing) | ||
|---|---|---|---|---|---|
| 1st Flush | 2nd Flush | Total | |||
| C | 11.0 ± 3.8 bb | 5.8 ± 0.9 ab | 16.8 ± 3.4 a | 67.1 ± 13.8 a | 24.0 ± 0.2 a |
| MTF | 12.1 ± 2.6 b | 5.0 ± 0.6 a | 17.1 ± 2.8 a | 68.4 ± 11.1 a | 23.9 ± 0.3 a |
| EO1B | 9.4 ± 1.1 b | 6.6 ± 4.0 ab | 16.0 ± 3.9 a | 64.1 ± 15.4 a | 25.4 ± 0.3 c |
| EO1A | 6.1 ± 2.2 a | 9.1 ± 2.6 bc | 15.1 ± 1.4 a | 60.4 ± 5.4 a | 24.6 ± 0.5 b |
| EO3B | 10.7 ± 1.3 b | 7.2 ± 2.0 ab | 18.0 ± 1.8 a | 71.7 ± 7.2 a | 25.2 ± 0.3 c |
| EO3A | 6.2 ± 2.6 a | 11.3 ± 4.1 c | 17.5 ± 2.0 a | 70.0 ± 8.0 a | 25.0 ± 0.5 bc |
| SED | 1.08 | 1.22 | 1.2 | 4.81 | 0.16 |
| LSD | 4.72 | 5.32 | 5.26 | 21.05 | 0.69 |
| d.f. | 29 | 29 | 29 | 29 | 29 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gea, F.J.; Navarro, M.J.; Santos, M.; Diánez, F.; Herraiz-Peñalver, D. Screening and Evaluation of Essential Oils from Mediterranean Aromatic Plants against the Mushroom Cobweb Disease, Cladobotryum mycophilum. Agronomy 2019, 9, 656. https://doi.org/10.3390/agronomy9100656
Gea FJ, Navarro MJ, Santos M, Diánez F, Herraiz-Peñalver D. Screening and Evaluation of Essential Oils from Mediterranean Aromatic Plants against the Mushroom Cobweb Disease, Cladobotryum mycophilum. Agronomy. 2019; 9(10):656. https://doi.org/10.3390/agronomy9100656
Chicago/Turabian StyleGea, Francisco J., María J. Navarro, Mila Santos, Fernando Diánez, and David Herraiz-Peñalver. 2019. "Screening and Evaluation of Essential Oils from Mediterranean Aromatic Plants against the Mushroom Cobweb Disease, Cladobotryum mycophilum" Agronomy 9, no. 10: 656. https://doi.org/10.3390/agronomy9100656
APA StyleGea, F. J., Navarro, M. J., Santos, M., Diánez, F., & Herraiz-Peñalver, D. (2019). Screening and Evaluation of Essential Oils from Mediterranean Aromatic Plants against the Mushroom Cobweb Disease, Cladobotryum mycophilum. Agronomy, 9(10), 656. https://doi.org/10.3390/agronomy9100656





