Soil Factors Effects on the Mineralization, Extractable Residue, and Bound Residue Formation of Aminocyclopyrachlor in Three Tropical Soils
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Sampling
2.2. Herbicide
2.3. Analysis of 14C-Extractable Residue
2.4. Analysis of 14C-Bound Residue
2.5. Measurement of Mineralization to 14CO2
2.6. Aminocyclopyrachlor Mineralization Model
2.7. Statistical Analysis
3. Results and Discussion
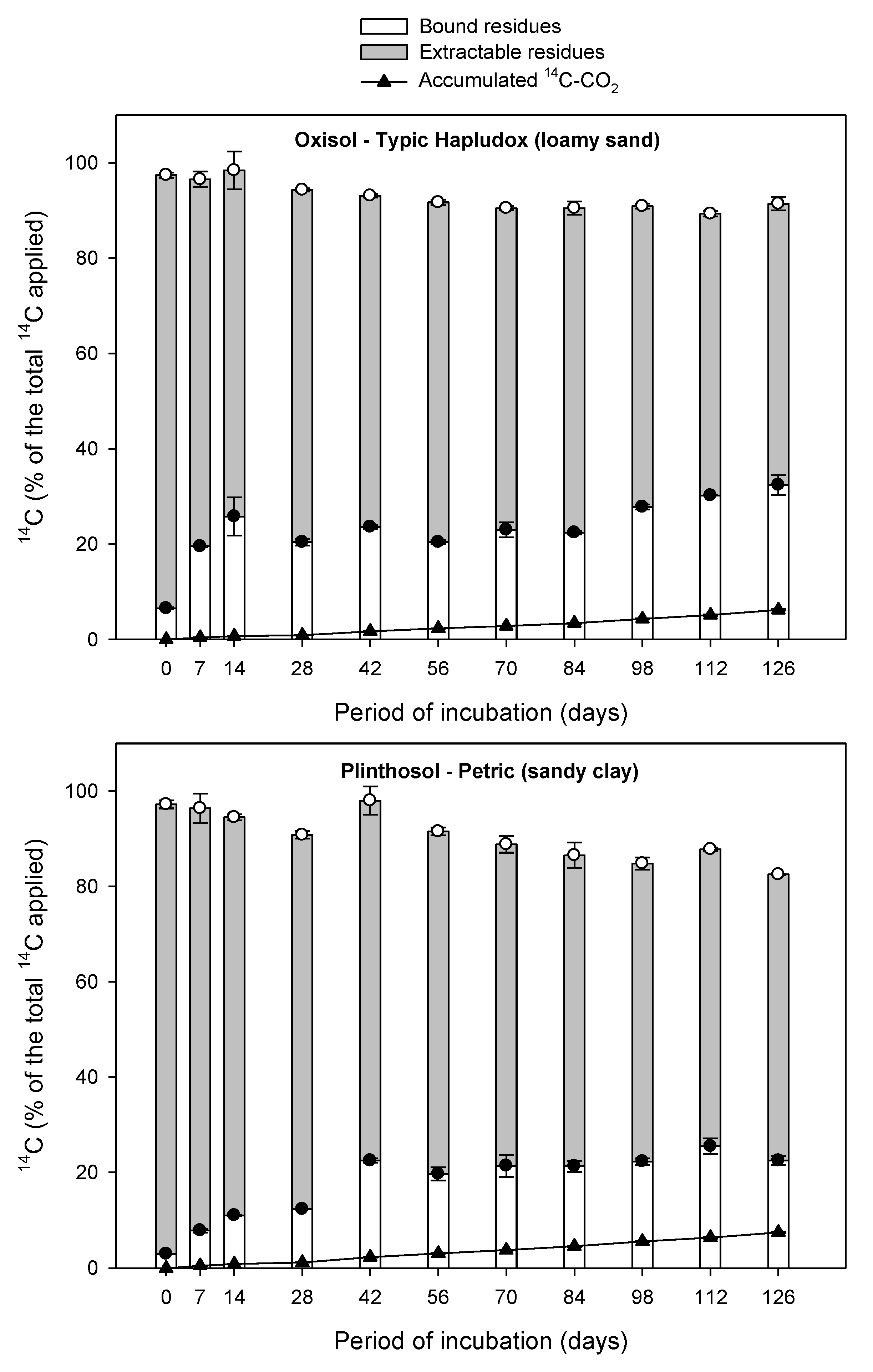
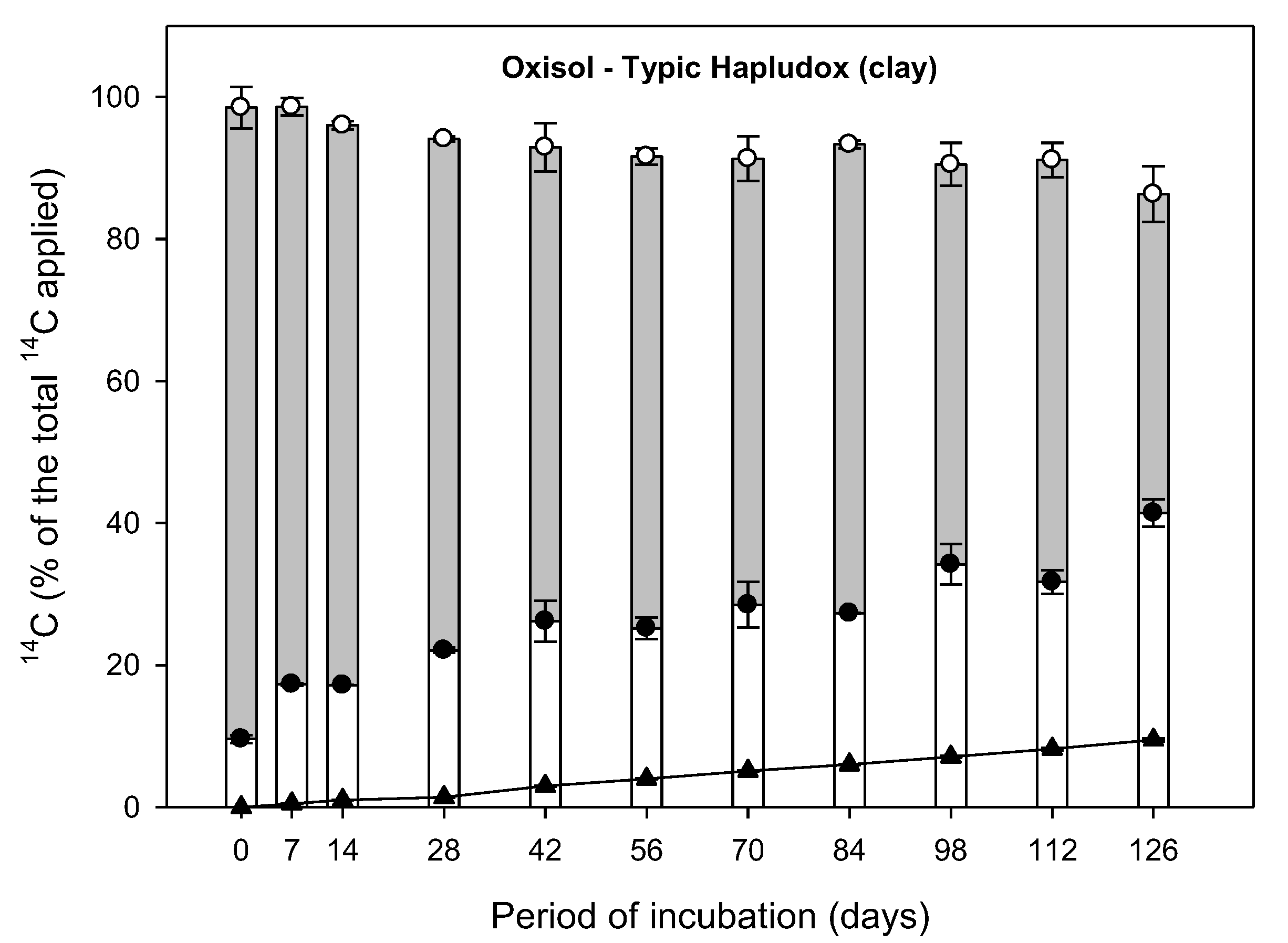
3.1. Mass Balance
3.2. Aminocyclopyrachlor Mineralization to 14CO2
3.3. Dissipation of 14C-Extractable Residue
3.4. 14C-Bound Residue Formation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- USEPA (United States Environmental Protection Agency)/OPPTS (Product Properties Test Guidelines). Ecological Risk Assessment for the Section 3 New Chemical Registration of Aminocyclopyrachlor on Non-Crop Areas and Turf; EPA-HQ-OPP-2009-0789-0004; Off Prevent Pest Tox Sub; Environmental Protection Agency: Washington, DC, USA, 2010. Available online: https://www3.epa.gov/pesticides/chem_search/ppls/000352-00793-20100831.pdf (accessed on 26 September 2017).
- Guerra, N.; Oliveira, R.S., Jr.; Constantin, J.; Oliveira Neto, A.M.; Braz, G.B.P. Aminocyclopyrachlor e indaziflam: Seletividade, controle e comportamento no ambiente. Rev. Bras. Herbic. 2013, 12, 285–295. [Google Scholar] [CrossRef]
- Reis, F.C.; Tornisielo, V.L.; Cason, J.B.; Dias, A.C.R.; Freitas, M.M.; Sotomayor, J.F.M.; Barroso, A.A.M.; Victoria, F.R. Uptake, translocation, and control of trumpet flower (Tecoma stans) with aminocyclopyrachlor. J. Environ. Sci. Health Part B 2015, 50, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.S., Jr.; Alonso, D.G.; Koskinen, C.W. Sorption desorption of aminocyclopyrachlor in selected Brazilian soils. J. Agric. Food Chem. 2011, 59, 4045–4050. [Google Scholar] [CrossRef] [PubMed]
- Francisco, J.G.; Mendes, K.F.; Pimpinato, R.F.; Tornisielo, V.L.; Dias, A.C.R. Aminocyclopyrachlor sorption-desorption and leaching from three Brazilian soils. J. Environ. Sci. Health Part B 2017, 53, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Durkin, P.R. Aminocyclopyrachlor: Human Health and Ecological Risk Assessment; Final Report; Syracuse Environmental Research Associates: Manlius, NY, USA, 2012; p. 226, Available online: http://www.fs.fed.us/foresthealth/pesticide/pdfs/Aminocyclopyrachlor.pdf (accessed on 28 March 2017).
- Nanita, S.C.; Stewart, D.J.; Rodgers, C.A.; Tesfai, E.; Schwarz, T.; Robaugh, E.C.; Henze, R.M.; Pentz, A.M.; Moate, T.; Vogl, E.; et al. Analytical method and interlaboratory study for the quantitation of aminocyclopyrachlor residues in vegetation by liquid chromatography/tandem mass spectrometry. J. AOAC Int. 2013, 96, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- PPDB—Pesticide Properties Data Base. Developed by the Agriculture & Environment Research Unit (AERU). University of Hertfordshire. Available online: http://sitem.herts.ac.uk/aeru/ppdb/ (accessed on 17 March 2017).
- Strachan, S.D.; Nanita, S.C.; Ruggiero, M.; Casini, M.S.; Heldreth, K.M.; Hageman, L.H.; Flanigan, H.A.; Ferry, N.M.; Pentz, A.M. Correlation of chemical analysis of residual levels of aminocyclopyrachlor in soil to biological responses of alfalfa, cotton, soybean, and sunflower. Weed Technol. 2011, 25, 239–244. [Google Scholar] [CrossRef]
- Conklin, K.L.; Lym, R.G. Effect of temperature and moisture on aminocyclopyrachlor soil half-life. Weed Technol. 2013, 27, 552–556. [Google Scholar] [CrossRef]
- Guerra, N.; Oliveira, R.S., Jr.; Constantin, J.; Oliveira, N.A.M.; Gemelli, A.; Pereira, D.M., Jr.; Guerra, A. Persistence of biological activity and leaching potential of herbicides aminocyclopyrachlor and indaziflam in soils with different textures. Planta Daninha 2016, 34, 345–356. [Google Scholar] [CrossRef]
- Gevao, B.; Semple, K.T.; Jones, K.C. Bound pesticide residue in soil: A review. Environ. Pollut. 2000, 108, 3–14. [Google Scholar] [CrossRef]
- Wang, H.; Liu, X.; Wu, J.; Huang, P.; Xu, J.; Tang, C. Impact of soil moisture on metsulfuron-methyl residues in Chinese paddy soils. Geoderma 2007, 142, 325–333. [Google Scholar] [CrossRef]
- Barriuso, E.; Benoit, P.; Dubus, I. Formation of pesticide nonextractable (bound) residues in soil: Magnitude, controlling factors and reversibility. Environ. Sci. Technol. 2008, 42, 1845–1854. [Google Scholar] [CrossRef] [PubMed]
- Boivin, A.; Amellal, S.; Schiavon, M.; van Genuchten, M.T. 2,4-Dichlorophenoxyacetic acid (2,4-d) sorption and degradation dynamics in three agricultural soils. Environ. Pollut. 2005, 138, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Mordaunt, C.J.; Gevao, B.; Jones, K.C.; Semple, K.T. Formation of non-extractable pesticide residues: Observations on compound differences, measurement and regulatory issues. Environ. Pollut. 2005, 133, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Gonod, L.V.; Chenu, C.; Soulas, G. Spatial variability of 2,4-dichlorophenoxyacetic acid (2,4-d) mineralisation potential at a millimeter scale in soil. Soil Biol. Biochem. 2003, 35, 373–382. [Google Scholar] [CrossRef]
- Wang, H.; Ye, Q.; Yue, L.; Han, A.; Yu, Z.; Wang, W.; Yang, Z.; Lu, L. Fate characterization of a novel herbicide ZJ0273 in aerobic soils using multi-position 14C labeling. Sci. Total Environ. 2009, 407, 4134–4139. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yue, L.; Zhang, S.; Ye, Q.; Qi, W.; Wang, H.; Chen, Z. Fate of pyribambenz propyl (ZJ0273) in anaerobic soils revealed by position-specific 14C labeling. J. Hazard. Mater. 2013, 258–259, 151–158. [Google Scholar] [CrossRef] [PubMed]
- EMBRAPA—Empresa Brasileira de Pesquisa Agropecuária. Sistema Brasileiro de Classificação de Solos, 3rd ed.; EMBRAPA Solos: Brasília, Brazil, 2013; 353p. [Google Scholar]
- OECD—Organisation for Economic Co-operation and Development. OECD guidelines for the testing of chemicals. In Test Number 307: Aerobic and Anaerobic Transformation in Soil; OECD: Paris, France, 2002; p. 17. [Google Scholar]
- Nanita, S.C.; Pentz, A.M.; Grant, J.; Vogl, E.; Devine, T.J.; Henze, R.M. Mass spectrometric assessment and analytical methods for quantitation of the new herbicide aminocyclopyrachlor and its methyl analogue in soil and water. Anal. Chem. 2009, 81, 797–808. [Google Scholar] [CrossRef] [PubMed]
- Picton, P.; Farenhorst, A. Factors influencing 2,4-d sorption and mineralization in soil. Environ. Sci. Health Part B 2004, 39, 367–379. [Google Scholar] [CrossRef]
- Mendes, K.F.; Martins, B.A.B.; Reis, M.R.; Pimpinato, R.F.; Tornisielo, V.L. Quantification of the fate of mesotrione applied alone or in a herbicide mixture in two Brazilian arable soils. Environ. Sci. Pollut. Res. 2017, 24, 8425–8435. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.F.; Brighenti, A.M. Comportamento de herbicidas no ambiente. In Biologia e Manejo de Plantas Daninhas; Oliveira, R.S., Jr., Constantin, J., Inoue, M.H., Eds.; Omnipax: Curitiba, Brazil, 2011; pp. 263–304. [Google Scholar]
- Schroll, R.; Becher, H.H.; Dörfler, U.; Gayler, S.; Grundmann, S.; Hartmann, H.P.; Ruoss, J. Quantifying the effect of soil moisture on the aerobic microbial mineralization of selected pesticides in different soils. Environ. Sci. Technol. 2006, 40, 3305–3312. [Google Scholar] [CrossRef] [PubMed]
- Bending, G.G.; Rodriguez-Cruz, M.S. Microbial aspects of the interaction between soil depth and biodegradation of the herbicide isoproturon. Chemosphere 2007, 66, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Patton, A.J.; Ruhl, G.E.; Creswell, T.C.; Wan, P.; Scott, D.E.; Becovitz, J.D.; Weisenberger, D.V. Potential damage to sensitive landscape plants from wood chips of aminocyclopyrachlor damaged trees. Weed Technol. 2013, 27, 803–809. [Google Scholar] [CrossRef]
- Guerra, N.; Oliveira, N.A.M.; Oliveira, R.S., Jr.; Constantin, J.; Takano, H.K. Sensibility of plant species to herbicides aminocyclopyrachlor and indaziflam. Planta Daninha 2014, 32, 609–617. [Google Scholar] [CrossRef]
| Attribute | Aminocyclopyrachlor |
|---|---|
| Structural formula | 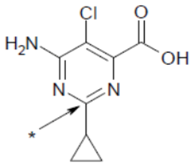 |
| Pesticide type | herbicide |
| Molecular formula | C8H8ClN3O2 |
| IUPAC (International Union of Pure and Applied Chemistry) name | 6-amino-5-chloro-2-cyclopropylpyrimidine-4-carboxylic acid |
| Chemical group | pyrimidine carboxylic acid |
| Molecular weight (g mol−1) | 213.62 |
| Water solubility at 20 °C (mg L−1) | 3130 (high) |
| Log Kow (octanol-water partition coefficient) | −2.48 (low) |
| pKa (dissociation constant) at 25 °C | 4.65 |
| Vapor pressure at 25 °C (mPa) | 6.92 × 10−3 (low volatility) |
| Henry’s law constant at 25 °C (Pa m3 mol−1) | 3.47 × 10−12 (non-volatile) |
| DT50 (soil half-life degradation time) (days) | 31 (moderately persistent) |
| Kd (sorption coefficient) (L Kg−1) | 0.39 (mobile) |
| GUS (Groundwater Ubiquity Score) leaching potential index | 3.91 (high leachability) |
| Origin (City, State) | Soil Classification—Symbology a | pH (CaCl2) | Sand | Silt | Clay | OC | CEC (mmolc dm−3) | Texture Class |
|---|---|---|---|---|---|---|---|---|
| (%) | ||||||||
| Agudos, São Paulo | Oxisol—Typic Hapludox (Latossolo Vermelho distrófico—LVd) | 4.4 | 84.3 | 1.9 | 13.8 | 0.6 | 42.3 | Loamy sand |
| Araguaína, Tocantins | Plinthosol—Petric (Plintossolo Pétrico concrecionário—FFc) | 5.1 | 49.4 | 10.2 | 40.4 | 2.0 | 74.7 | Sandy clay |
| Piracicaba, São Paulo | Oxisol—Typic Hapludox (Latossolo Vermelho eutrófico—LVe) | 4.7 | 13.4 | 12.5 | 74.1 | 1.7 | 96.1 | Clay |
| Soil | Parameter | ||||||
|---|---|---|---|---|---|---|---|
| 14CO2 (%) | k (day−1) | MT50 (days) | R2 | p | |||
| Oxisol—Typic Hapludox (loamy sand) | 7.46 ± 0.20 a | B * | 0.00061 ± 0.000015 | 1128 ± 32 | B | 0.99 | <0.001 |
| Plinthosol—Petric (sandy clay) | 6.15 ± 0.19 | C | 0.00050 ± 0.000017 | 1376 ± 43 | A | 0.98 | <0.001 |
| Oxisol—Typic Hapludox (clay) | 9.48 ± 0.23 | A | 0.00079 ± 0.000019 | 877 ± 22 | C | 0.99 | <0.001 |
| LSD (least significant difference) | 0.50 | 81.17 | |||||
| CV (coefficient of variation) (%) | 3.00 | 3.32 | |||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Francisco, J.G.; Mendes, K.F.; Pimpinato, R.F.; Tornisielo, V.L.; Guimarães, A.C.D. Soil Factors Effects on the Mineralization, Extractable Residue, and Bound Residue Formation of Aminocyclopyrachlor in Three Tropical Soils. Agronomy 2018, 8, 1. https://doi.org/10.3390/agronomy8010001
Francisco JG, Mendes KF, Pimpinato RF, Tornisielo VL, Guimarães ACD. Soil Factors Effects on the Mineralization, Extractable Residue, and Bound Residue Formation of Aminocyclopyrachlor in Three Tropical Soils. Agronomy. 2018; 8(1):1. https://doi.org/10.3390/agronomy8010001
Chicago/Turabian StyleFrancisco, Jeane G., Kassio F. Mendes, Rodrigo F. Pimpinato, Valdemar L. Tornisielo, and Ana Carolina D. Guimarães. 2018. "Soil Factors Effects on the Mineralization, Extractable Residue, and Bound Residue Formation of Aminocyclopyrachlor in Three Tropical Soils" Agronomy 8, no. 1: 1. https://doi.org/10.3390/agronomy8010001
APA StyleFrancisco, J. G., Mendes, K. F., Pimpinato, R. F., Tornisielo, V. L., & Guimarães, A. C. D. (2018). Soil Factors Effects on the Mineralization, Extractable Residue, and Bound Residue Formation of Aminocyclopyrachlor in Three Tropical Soils. Agronomy, 8(1), 1. https://doi.org/10.3390/agronomy8010001






