Abstract
Northeast China (NEC) is a major spring maize (Zea mays L.) growing belt, and the outputs substantially influence national grain production. However, the maize grain yield per unit area has little changes in recent years, partially due to the lack of elite germplasm resources and innovation. Therefore, this study aimed to determine the performance of diverse populations in NEC to propose appropriate strategies for the utilization of elite germplasm to broaden the genetic base of Chinese germplasm. Fifteen diverse maize populations from the International Maize and Wheat Improvement Center (CIMMYT) and the U.S. were crossed to two local tester lines, representing Chinese heterotic groups Reid and Lancaster, for evaluating the combining ability and heterosis in three locations (Gongzhuling, Jilin Province, and Harbin and Suihua, Heilongjiang Province) in NEC over two years. The U.S. (BS13(S)C7 and BS31) and Chinese (Ji Syn A) populations exhibited more favorable alleles for high yield potential in all locations tested. Furthermore, the PH6WC × BS31 and PH6WC × Ji Syn A crosses had higher grain yields, and an appropriate number of days to silking, ear height, and resistance to lodging at Gongzhuling and Harbin in NEC. The best strategies for utilizing these diverse germplasms may be to develop new inbred lines from the existing elite populations or improve the grain yield and resistance to lodging of the elite line PH4CV for broadening the genetic base of the Chinese group Lancaster in NEC.
1. Introduction
Maize (Zea mays L.) is an important grain and feeding crop grown worldwide that plays an important role in food production and has a high economic value [1]. In China, the maize planting area is divided into six regions, which are as follows: the Northeast, Yellow and Huai Rivers, the Northwest, Southwest, South, and Tibetan plateau. The Northeast Region accounts for 39.4% of the national total planting area and contributes 42.8% of the total domestic output [2]. Increasing grain yield in Northeast China (NEC) is, thus, crucial for securing national grain production. However, little changes were shown in maize grain yield per unit area in China [3], and the important factor may be that most commercial varieties have similar genetic backgrounds owing to the overuse of the available elite germplasm [4]. Additionally, the lack of specific germplasm with new favorable alleles, resulting in the upgrading of new elite varieties, is slow [5,6]. Furthermore, extreme climate change events can cause chilling injury during the seeding stage, snowstorms, and frost, and this can also limit maize yields in NEC [7,8]. The introduction and utilization of elite exotic germplasm, including that from tropical, subtropical, and temperate regions, is an effective strategy to broaden the germplasm background [9,10,11,12].
In China, the International Maize and Wheat Improvement Center (CIMMYT) and the U.S. germplasm are the major exotic germplasm sources for maize breeding [13,14]. CIMMYT maize populations have desirable traits, including vigorous seedlings, high biological yield, quick seed dehydration, developed rhizomes, resistance to stalk lodging and drought, and large genetic variability and geographical distance when compared with Chinese temperate germplasm [15]. Thus, CIMMYT populations can be utilized to develop new breeding materials and break the existing heterosis model [6]. In addition, some elite tropical and subtropical maize populations, including Stay green, Pop21, Pop28, Pop32, Pop43, Pop49, and Suwan 1, have been identified and successfully improved using mass selection for adaptation to Chinese temperate areas [5]. The CIMMYT populations Suwan 1, Suwan 2, ETO, and Tuxpeño, are the specific exotic germplasms used for maize breeding in the Chinese Southwest Region [14]. In the Yellow and Huai River regions, the Suwan germplasm has also been used to improve the domestic Sipingtou germplasm by increasing its resistance to disease and lodging [16]. Suwan 1 was found to improve grain yield and resistance to disease in elite inbred lines in NEC [17].
The U.S. Corn Belt germplasm can also provide desirable alleles such as those with early maturation, lower EH, strong resistance to root and stalk lodging, and increased yield potential [11,18,19]. Since the 1980s, several commercial U.S. hybrids have been introduced into China, and the developed inbred lines have been widely used in NEC [20]. This has led to the development of many typical lines, such as the inbred line Zi330, which was derived from the exotic germplasm Oh43 and KeLi67, and the inbred line Ye478, which was developed from U8112 and Shen5003, and these were selected from U.S. hybrids [21]. The elite hybrid in NEC, Xianyu 335, was developed by Tieling Pioneer Seed Research Co., Ltd., and it has a high yield per unit area and high quality [22]. In addition, the extension of the hybrid Xianyu 335 to NEC increased the level of germplasm improvement [23].
General (GCA) and specific combining ability (SCA) are important indexes to evaluate whether germplasm resources are good or not for utilizing germplasm more effectively [14]. Generally, two genetically distant lines with higher GCA effect contain more favorable genes, which also could have greater potential for breeding high-yielding hybrids if they simultaneously show high SCA and heterosis [21]. Although some elite tropical and subtropical and U.S. maize populations were studied in North China, more diverse germplasm has not been investigated and utilized the areas northward in NEC (e.g., Jilin Province and Heilongjiang Province). The objectives of this study were, thus, as follows: (i) to evaluate the effects of GCA and specific combining ability (SCA) on days to silking (DS), ear height (EH), stalk lodging (SL), and grain yield (GY) for 30 crosses (two testers × 15 diverse populations) in three typical locations in NEC, and (ii) to determine the breeding potential of the diverse germplasm to broaden the genetic background of Chinese germplasm groups in NEC.
2. Materials and Methods
2.1. Genetic Materials
Fifteen diverse populations were used in this investigation, and they were sourced from CIMMYT and the U.S.; they were also sourced from China (Table 1). The Tuxpeño (Tuxpeño) and Suwan (Suwan 1) germplasms and high stress-resistant germplasms [Stay green-yellow (SG-Y) and PooL 26] were derived from CIMMYT. These four populations have been improved by five cycles of divergent biparental mass selection for earlier flowering from southern (Guangxi Zhuang Autonomous Region) to northern growing regions in China (Henan, Liaoning, and Heilongjiang Provinces) [24]. Seven of the fifteen populations were introduced into China from the U.S.; including the two-ear synthetic population BS10(FR)C10, the Iowa Stiff Stalk synthetic populations BSSS(R)C10 and BS13(S)C7, Iowa rootworm synthetic population BS20(S)C2, adapted tropical populations BS27 (Antigua composite) and BS31 (Undetermined source), and a broad-based synthetic population BSBB(SRCB)C4. Another four of the fifteen populations were developed in China, including Comp13, a semi-exotic population derived from Suwan 1, and two Chinese domestic populations, Csyn5 and Cysn6. Comp14 was developed from two national populations, Cysn7 and Yu Syn 5, while Ji Syn A is a composite originating from the national germplasm Yinglizi, and Liaoninglv Syn is a synthetic population derived from lines originating from the Chinese domestic germplasm Lvda Red Cob [13,20].

Table 1.
Populations, ecosystem type, and germplasm description.
The check hybrid Xianyu 335 was used in this study, as it has high yields, quality, and extensive adaptability, and has recently been the leading maize hybrid in NEC [23]. The parental lines PH6WC and PH4CV of the hybrid Xianyu 335 were chosen as testers and representatives of the subgroups Reid and Lancaster in the current study. The inbred line PH4CV, derived from the crossing of lines PH7V0 and PHBE2, belongs to the Reid germplasm, and the inbred line PH6WC was developed from the PH01N × PH09B cross and belongs to the Lancaster germplasm [34].
2.2. Experimental Design and Data Collection
The 15 populations were individually crossed with the two tester lines, PH4CV and PH6WC, in a NC Design II scheme at the Chinese Academy of Agriculture Science (CAAS) Hainan Experiment Station in 2011. Bulked pollen samples obtained from a minimum of 100 plants from each population were used to pollinate 100 plants from each of the two tester lines. Thirty crosses and a commercial check (Xianyu 335) were evaluated according to a randomized complete block design with three replicates in 4-row plots that were 5 m in length and 0.6 m in width. The planting space between plants was 0.33 m. The field experiments were performed at Gongzhuling (43.31° N, 124.49° E) in Jilin Province and Harbin (45.8° N, 126.5° E) and Suihua (46.63° N, 126.98° E) in Heilongjiang Province during 2012 and 2013 in NEC. Plots were overplanted and later thinned to obtain a population density of approximately 51,000 plants ha−1. The average annual rainfall in Suihua was 483 mm, which is 86.1 mm lower than that in Harbin and 111.8 mm lower than that in Gongzhuling. The frost-free period in Suihua was 140 days, which is two days less than that of Harbin and four days less than that of Gongzhuling [35]. Soil in the experimental sites is the chernozem soil according to Chinese Soil Classification and Terminology. According to local fertilization recommendations, 450 kg ha−1 of diammonium phosphate before sowing and 150 kg ha−1 of urea during the pulling period were supplied, and timely manual spraying of chemicals and weed control in the field for each location.
Data on days to silking (DS), ear height (EH), stalk lodging (SL), and grain yield (GY) were collected at the Gongzhuling, Harbin, and Suihua locations. DS was recorded as the number of days (d) from planting until 50% of the plants had visible silks. EH (cm) was obtained from 20 random plants per plot and was measured from the base of the plant at ground level to the node bearing the uppermost ear. Percentage SL was measured by recording the number of plants with stalks bent below the main ear at an angle greater than 45º and dividing that number by the number of plants evaluated. The grain yield was measured on an area of 12 m2 (5 m × 2.4 m) from each sub plot through manual harvesting and was dried by sunshine and threshed individually for each experimental unit, and then it was converted into GY (t ha−1) at 155 mg kg−1 grain water content.
2.3. Statistical Analysis
For individual and combined locations, analysis of variance was performed using the “aov” function in the R program. Genotypes were treated as fixed effects, and locations were assumed to be random effects. Furthermore, GCA is represented by the sum of squares for the population and testers, and the sum of squares for the tester × population represents the SCA. The GCA and SCA effects were calculated from a joint linear mixed model analysis of crosses in both individual and combined environments with ASReml-R [36], following the general model:
where Yijklm is the mth replication for a cross between the kth female and the lth male in the jth block and ith location, μ is the general mean, and Li is the ith (i = 1 to e) fixed location (environmental) effect. Bj(i) is the jth (j = 1 to b) block within the ith location, and the GCA and SCA effects are assumed to be independently distributed (IND) with variances to individual and combined locations. GCAk and GCAl are the random GCA effects of the kth female and the lth male, respectively, IND following N (0, σ2GCA), k, l = 1 to p, and k < l; SCAkl is the random SCA effect of the kth and lth parents, IND following (0, σ2SCA) (k ≠ l); L × GCAik and L × GCAil are the random GCA by location interaction effect, IND following (0, σ2L*G); L × SCAikl is the random SCA by location interaction effect, IND following (0, σ2L*S); and Eijklm is the random error, IND following (0, σ2E). The significance of GCA and SCA for each cross in this study was tested using a least significant difference (LSD) test, with an estimate of the correctness mean for each measured trait at 99% and 95% probability levels.
Yijklm = μ + Li + Bj(i) + GCAk + GCAl + SCAkl + L × GCAik + L × GCAil + L × SCAikl + Eijklm
3. Results
3.1. Analysis of Variance
Analysis of variance results for DS, EH, SL, and GY in the individual and combined environments are presented in Table A1. Significant differences due to genotype and environment × genotype were observed for all measured traits in the individual and combined environments. Variances attributable to populations and testers are the GCA variances, while those of the crosses based on tester × population interactions are the SCA variances. The GCA variances were significant for all traits measured, and significant differences due to the SCA variances were observed in the individual and combined environments for DS, EH, and GY. In contrast, no significant differences due to the SCA variances for SL were observed in the individual or combined environments of Harbin and Suihua. Estimated variance components of GCA and SCA showed that the additive variance (δ2A) due to the GCA was greater than the dominance variance (δ2D) due to the SCA for all measured traits in the study, which revealed the additive gene action was more important than the dominance gene action for these traits. The broad sense (H2b) and narrow sense (H2n) heritabilities were high for all the studied traits except for DS and GY in the combined environment (Table A1).
3.2. Days to Silking
In Gongzhuling, 4 crosses [PH4CV × Tuxpeño, PH4CV × SG-Y, PH4CV × BS20(S)C2, and PH4CV × Comp13] had more DS than the check Xianyu 335, whereas the other 26 crosses had fewer DS. While in Harbin, 5 crosses [PH4CV × SG-Y, PH4CV × PooL 26, PH6WC × Suwan 1, PH6WC × SG-Y, and PH6WC × Comp13] had more DS than the Xianyu 335, and in Suihua, 8 of the 30 crosses [PH4CV × BS10(FR)C10, PH4CV × BS13(S)C7, PH4CV × BS27, PH4CV × BS31, PH4CV × BSBB(SRCB)C4, PH4CV × BSSS(R)C10, PH4CV × Ji Syn A, and PH4CV × Liaoninglv Syn] had fewer DS than the Xianyu 335 (Table A2). Among the PH4CV × populations, the PH4CV × SG-Y and PH4CV × BSBB(SRCB)C4 crosses had the largest and smallest SCAs, respectively, and the opposite was found for the PH4CV × populations in the individual and combined environments (Figure 1).
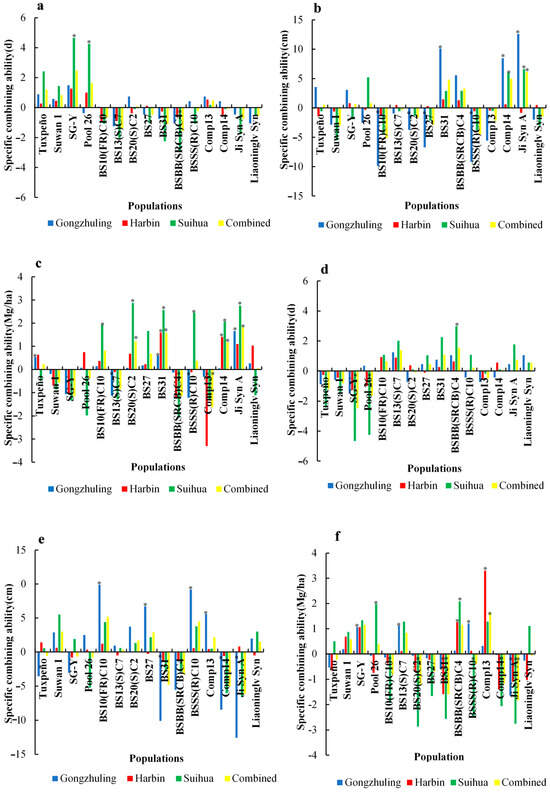
Figure 1.
Specific combining ability for 15 populations crossed with PH4CV (a) for DS, (b) for EH, (c) for GY, and PH6WC (d) for DS, (e) for EH, (f) for GY for days to silking (DS), ear height (EH), and grain yield (GY) in individual and combined environments (* indicate p value of 0.05).
In Gongzhuling, the positive significant GCA effects were found for the SG-Y, BS10(FR)C10, and BS20(S)C2 populations, and the opposite was shown for the BS13(S)C7, BS31, BSBB(SRCB)C4, and Liaoninglv Syn populations. The PooL 26, BS27, and Ji Syn A populations also contributed to the negative and non-significant GCAs (Figure 1). In Harbin, the BS10(FR)C10, BS13(S)C7, BS20(S)C2, and BSBB(SRCB)C4 populations had significantly negative GCAs. While the Tuxpeño, BS31, BSSS(R)C10, Comp14, Ji Syn A, and Liaoninglv Syn populations had negative but non-significant GCAs. In Suihua, the BS31 and BSBB(SRCB)C4 populations exhibited significant negative GCA effects, while Suwan 1, SG-Y, and PooL 26 had significant positive GCA effects. Furthermore, the BS10(FR)C10, BS20 (S)C2, BS27, BSSS(R)C10, Comp14, Ji Syn A, and Liaoninglv Syn populations also had negative and non-significant GCAs. In the combined environment, BS13(S)C7and BSBB(SRCB)C4 had significantly negative GCAs, whereas Suwan 1, SG-Y, and PooL 26 had significantly positive GCAs. Furthermore, BS10(FR)C10, BS27, BS31, BSSS(R)C10, Comp14, Ji Syn A, and Liaoninglv Syn had negative non-significant GCAs. The individual and combined environments for BS13(S)C7 and BSBB(SRCB)C4 all had significant negative GCAs for DS (Figure 2).
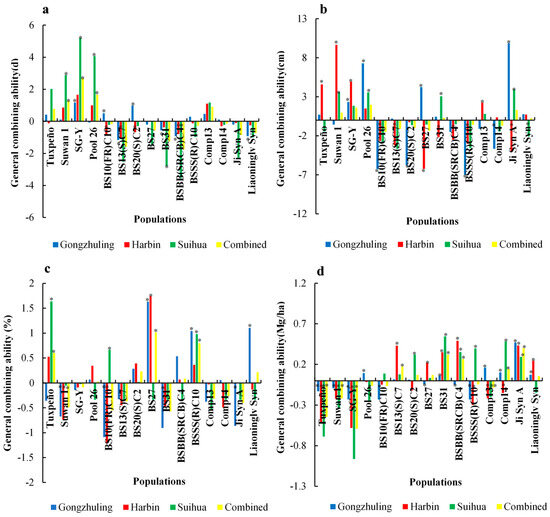
Figure 2.
General combining ability for 15 populations for days to silking (a), ear height (b), stalk lodging (c), and grain yield (d) in individual and combined environments (* indicate p value of 0.05).
3.3. Ear Height
In Gongzhuling, eight crosses [PH4CV × Tuxpeño, PH4CV × Suwan 1, PH4CV × SG-Y, PH4CV × PooL 26, PH4CV × Ji Syn A, PH6WC × PooL 26, PH6WC × BS27, and PH6WC × Ji Syn A] had greater EHs than the check Xianyu 335. The other crosses all had smaller EHs than the control, especially PH6WC × BS20(S)C2 and PH6WC × BSSS(R)C10, which had the smallest EHs. In Harbin, all crosses had smaller EHs than Xianyu 335, except for PH4CV × Tuxpeño, PH4CV × Suwan 1, PH4CV × SG-Y, and PH6WC × Suwan 1. The PH4CV × BS20(S)C2 and PH6WC × BSSS(R)C10 crosses had the smallest EHs among all 30 crosses. In Suihua, six crosses [PH4CV × Suwan 1, PH4CV × SG-Y, PH4CV × PooL 26, PH4CV × Ji Syn A, PH6WC × Suwan 1, and PH6WC × Comp 13] had greater EHs than the check Xianyu 335, whereas the other crosses all had smaller EHs. In particular, the PH4CV × BS13(S)C7and PH4CV × Liaoninglv Syn crosses had the smallest EHs of the 30 crosses (Table A2). Among the PH4CV × populations, the PH4CV × BS10(FR)C10 and PH4CV × Ji Syn A crosses had the greatest and smallest SCAs, respectively, and the opposite was found for the PH6WC × populations in the individual and combined environments (Figure 1).
In Gongzhuling, the GCAs were significantly greater for the SG-Y, PooL 26, BS27, and Ji Syn A populations, whereas significantly negative GCAs were observed for the BS10(FR)C10, BS20(S)C2, BSBB(SRCB)C4, BSSS(R)C10, and Comp14 populations. Suwan 1 and Comp13 had negative but not significant SCAs. In Harbin, seven populations [BS10(FR)C10, BS13(S)C7, BS27, BS31, BSBB(SRCB)C4, BSSS(R)C10, and Ji Syn A] were identified as having significantly negative GCA effects, whereas Tuxpeño, Suwan 1, SG-Y, PooL 26, and Comp13 had significantly positive GCAs. Only one population, BS20(S)C2, had a negative but non-significant GCA. In Suihua, six populations [Tuxpeño, BS10(FR)C10, BS13(S)C7, BSSS(R)C10, Comp14, and Liaoninglv Syn] had significant negative GCAs. Significant positive GCAs were observed in Suwan 1, SG-Y, PooL 26, BS31, and Ji Syn A, while BS20(S)C2, BS27, and BSBB(SRCB)C4, had negative but non-significant GCAs. In the combined environment, significant negative GCAs were identified for BS10(FR)C10, BS13(S)C7, and BS27, whereas SG-Y, PooL 26, and Ji Syn A had non-significant positive GCAs. Five populations [BS20(S)C2, BSBB(SRCB)C4, BSSS(R)C10, Comp13, and Comp14] contributed to the negative but non-significant GCAs (Figure 2).
3.4. Stalk Lodging
Compared to the average performance for the 30 crosses and the check Xianyu 335, in Gongzhuling, 10 of the 30 crosses [PH4CV × BS31, PH4CV × Ji Syn A, PH6WC × Tuxpeño, PH6WC × Suwan 1, PH6WC × BS10(FR)C10, PH6WC × BS31, PH6WC × BSSS(R)C10, PH6WC × Comp13, PH6WC × Comp14, and PH6WC × Ji Syn A] had lower SLs than the check. In Harbin, however, 7 of the crosses, [PH6WC × PooL 26, PH6WC × BS13(S)C7, PH6WC × BS27, PH6WC × BS31, PH6WC × BSBB(SRCB)C4, PH6WC × Comp14, and PH6WC × Ji Syn A] had lower SLs than the check, and in Suihua, 16 of the crosses [PH4CV × PooL 26, PH4CV × BS13(S)C7 PH4CV × BS27, PH4CV × BS31, PH4CV × BSBB(SRCB)C4, PH4CV × Ji Syn A, PH4CV × Liaoninglv Syn, PH6WC × PooL 26, PH6WC × BS13(S)C7, PH6WC × BS27, PH6WC × BS31, PH6WC × BSBB(SRCB)C4, PH6WC × Comp13, PH6WC × Comp14, PH6WC × Ji Syn A, and PH6WC × Liaoning Syn] had lower SLs than the check (Table A2).
In Gongzhuling, significant negative GCAs were observed for the Suwan 1 and BS10(FR)C10 populations, whereas significant positive GCAs were identified for the BS27, BSSS(R)C10, and Liaoninglv Syn populations. In addition, the Tuxpeño, SG-Y, BS13(S)C7, BS31, Comp13, and Ji Syn A populations also contributed to the negative but non-significant GCAs. In Harbin, the BS10(FR)C10 and Comp14 had the expected significant negative GCAs, whereas Tuxpeño and BS27 had significant positive GCAs. Negative but non-significant GCAs were identified in six populations [Suwan 1, SG-Y, BS13(S)C7, BS31, Comp13, and Liaoninglv Syn]. In Suihua, the Tuxpeño, BS10 (FR) C10, and BSSS (R) C10 populations had unexpectedly significant positive GCAs, and only Comp13 exhibited a significantly negative GCA. Furthermore, ten populations, [Suwan 1, SG-Y, PooL 26, BS13(S)C7, BS27, BS31, BSBB(SRCB)C4, Comp14, Ji Syn A, and Liaoninglv Syn] had negative but non-significant GCAs. In the combined environment, significant negative GCA effects were observed in the Suwan 1, BS10(FR)C10, BS13(S)C7, BS31, Comp13, Comp14, and Ji Syn A populations while the Tuxpeño, BS27, and BSSS(R)C10 populations had significant positive effects on the GCA, and only the SG-Y population had a negative but non-significant GCA (Figure 2).
3.5. Grain Yield
The heterosis over the check Xianyu 335 varied from −31.88% (PH4CV × SG-Y) in Suihua to 11.78% (PH6WC × BSBB(SRCB)C4) in Harbin. In Gongzhuling, no cross showed a higher yield than the check Xianyu 335. While in Harbin, 7 crosses [PH6WC × PooL 26 (2.63%), PH6WC × BS13 (11.40%), PH6WC × BS27 (10.93%), PH6WC × BS31 (9.51%), PH6WC × BSBB(SRCB)C4 (11.78%), PH6WC × Ji Syn A (9.42%), and PH6WC × Liaoning Syn (7.72%)] had higher yields, and in Suihua, only the PH4CV × BS31 (1.14%) cross had a higher yield when compared with the Xianyu 335 (Table A2).
According to the GCA effects on GY (Figure 2), in Gongzhuling, significant positive GCA effects were only observed in 6 populations [PooL 26, BS31, Comp13, Comp14, Ji Syn A, and Liaoninglv Syn], and significant negative GCA effects were observed in 7 populations [Tuxpeño, Suwan 1, SG-Y, BS10(FR)C10, BS27, BSBB(SRCB)C4, and BSSS(R)C10]. Positive but non-significant GCAs were also identified for the BS13(S)C7 and BS20(S)C2 populations, with values of 0.02 and 0.01 t/ha, respectively. In Harbin, six populations [BS13(S)C7, BS27, BS31, BSBB(SRCB)C4, Ji Syn A, and Liaoninglv Syn] contributed to the significant positive GCA effects, while significant negative GCA effects were observed in six populations [Tuxpeño, Suwan 1, SG-Y, BSSS(R)C10, Comp13, and Comp14]. In Suihua, the desired significant positive GCA effects were identified in six populations [BS20(S)C2, BS31, BSBB(SRCB)C4, BSSS(R)C10, Comp14, and Ji Syn A], and five populations [Tuxpeño, Suwan 1, SG-Y, PooL 26, and Comp13] had significant negative GCA effects. Furthermore, BS10(FR)C10, BS13(S)C7, and BS27 had positive but non-significant GCAs. In the combined environment as expected, the BS13(S)C7, BS31, BSBB(SRCB)C4, Comp14, and Ji Syn A populations showed significant positive GCA effects, whereas the Tuxpeño, Suwan 1, SG-Y, and Comp13 had significant negative GCA effects. The BS20(S)C3, BS27, and Liaoninglv Syn populations had positive but non-significant GCAs.
The PH4CV × populations in Gongzhuling ranged from −1.21** t/ha in PH4CV × BS13(S)C7 and PH4CV × BSSS(R)C10 to 1.65** t/ha in PH4CV × Ji Syn A. The PH4CV × Tuxpeño, PH4CV × BS31, and PH4CV × Ji Syn A crosses also showed significant positive SCAs. In Harbin, the largest and smallest SCAs were obtained in the PH4CV × BS31 and PH4CV × Comp13 populations, respectively. While a significant positive SCA was also identified in PH4CV × Comp14. In Suihua, the largest and smallest SCAs were identified in PH4CV × BS20(S)C2 and PH4CV × BSBB(SRCB)C4. Furthermore, 6 of the 30 crosses [PH4CV × BS10(FR)C10, PH4CV × BS20(S)C2, PH4CV × BS31, PH4CV × BSSS(R)C10, PH4CV × Comp14, and PH4CV × Ji Syn A] were also found to significantly contribute to the SCA. The combined environment ranged from −1.63** t/ha for the PH4CV × Comp13 cross to 1.83** t/ha for the PH4CV × Ji Syn A cross. Furthermore, the PH4CV × BS20(S)C2, PH4CV × BS31, PH4CV × Comp14, and PH4CV × Ji Syn A crosses exhibited significant positive SCAs (Figure 1).
Among the PH6WC × populations in Gongzhuling, the largest and smallest SCAs were identified for the PH6WC × BSSS(R)C10 and PH6WC × Ji Syn A crosses, respectively. While the PH6WC × SG-Y, PH6WC × BS13(S)C7, and PH6WC × BSSS(R)C10 crosses also contributed to the significant positive SCA. In Harbin, this ranged from −1.59** t/ha in the PH6WC × BS31 cross to 3.30** t/ha in the PH6WC × Comp13 cross. In addition, the PH6WC × BSBB(SRCB)C4 and PH6WC × Comp13 crosses, also exhibited significantly positive SCAs. In Suihua, the largest and smallest SCAs were obtained from the PH6WC × BS20(S)C2 and PH6WC × BSBB(SRCB)C4 crosses, respectively. While the PH6WC × PooL 26 and PH6WC × BSBB(SRCB)C4 crosses also had significant positive SCAs. The combined environment ranged from −1.83** t/ha with the PH6WC × Ji Syn A to 1.63** t/ha with the PH6WC × Comp13. Only the PH6WC × Comp13 cross had a significant positive SCA (Figure 1).
4. Discussion
4.1. Combining Ability and Heterosis of 15 Diverse Populations
The combined results, including mean performance, GCA, SCA, and heterosis from the three NEC locations, showed that the CIMMYT populations Tuxpeño, Suwan 1, SG-Y, and PooL 26, had undesirable alleles for longer DS, higher EH, and lower GY. However, Suwan 1 and SG-Y were identified as resistant to lodging in all environments, and the PH6WC × PooL 26 cross showed a slightly higher GY than the check Xianyu 335 in Harbin. These results indicate that the excellent traits of the CIMMYT populations could be utilized in NEC. Maize is a short sunshine crop, and the germplasm sourced from tropical and subtropical climates is more sensitive to sunlight than temperate germplasm [37]. The sunlight time increases from south to north in NEC, making it difficult for the CIMMYT populations to adapt, and this may be the reason for the undesirable performances in NEC. Previous studies have found that the CIMMYT populations have desirable agronomic traits such as resistance to low temperature, drought, and disease. For example, the SA3 and P147 populations showed resistance to foliar diseases [25], and P147 also showed resistance and tolerance to Maize streak virus, downy mildew, turcicum leaf blight, and drought [14]. In this investigation, the CIMMYT populations Suwan 1 and SG-Y showed resistance to lodging in the multi-environment tests, and this is consistent with previous studies, which have reported that tropical and subtropical germplasms have tough stalks [24].
Among the seven U.S. populations, BS10(FR)C10 exhibited the desired performance in DS (except in Gongzhuling), EH, and SL (except in Suihua). In a previous study, BS10(FR)C10 was also identified as an elite source of favorable alleles for increasing yield and showed resistance to SL in the Yellow and Huai River valleys and Northeast China [5,13]. BS13(S)C7 was considered to have high potential for breeding because of its favorable GCA for DS, EH (except in Gongzhuling), SL, and GY. In addition, BS13(S)C7 also exhibited a high potential for increasing yield and resistance to head smut in Iowa and Northern China [38]. BS20(S)C2 showed a negative GCA for DS (except in Gongzhuling) and EH and a positive GCA for GY (except in Harbin). BS20(S)C2 was also favorable due to its decreasing DS and EH in NEC. BS27 is a tropical germplasm that was adapted in this study for a temperate environment, which had favorable GCA for DS (except in Harbin), EH (except in Harbin), and GY (except in Gongzhuling). BS31 showed a higher GCA for DS, SL, and GY in the 15 populations, and a previous study also found that it had great potential to increase yield and favorable alleles for other agricultural traits [5]. BS31 was originally a synthetic tropical germplasm FS8B developed by mass selection for earliness [30]. BSSS(R)C10 showed the expected GCA for DS (except in Gongzhuling) and EH, consistent with the performance for DS and EH in Northern China. The U.S. germplasm has an abundance of diversity and is characterized by strong stems, high yield potential, and good agricultural traits [18,19]. Yong et al. [5] revealed that U.S. populations could be directly introduced and used in NEC maize breeding programs. Stable and desirable agronomic traits, including DS, EH, and GY, were found in most U.S. populations; however, the unfavorable performance of some populations, especially BS20(S)C2 and BSSS(R)C10 in SL, were also identified. Thus, based on these analyses, most U.S. populations were found to have favorable alleles, and BS13, BS31, and BSBB(SRCB)C4 could be directly used to broaden the domestic germplasm background for the environments in NEC.
Comp13, a composite, had an unexpected GCA for DS, EH, and GY in the Chinese domestic populations but exhibited the desired SL in all environments. While Comp14, another composite, had a favorable GCA for DS (except in Gongzhuling), EH (except in Harbin), SL (except in Gongzhuling), and GY (except in Harbin) in NEC. Ji Syn A had the desired GCA performance for DS, SL (except in Harbin), and GY. The final population, Liaoninglv Syn, had less potential for improving EH and SL but exhibited a favorable GCA for DS and GY (except in Suihua) in NEC. Comp13 was developed from the germplasm background by crossing Cysn5, Suwan 1, and Cysn6. Cysn5 was derived by crossing 38 lines from Sipingtou, and Cysn6 was also a composite population closely related to group NSS [16]. The tropical population Suwan1 is tall and lodging resistant, with a long growth period, ear dysplasia, poor seed maturation, decreased panicle grain and 100-grain weight, and an abundance of genetic variations [15]. The inadaptation of Suwan 1 to NEC may result in the undesirable agronomic traits of Comp13. Comp14 was developed using Cysn7, which was developed from more than 20 lines derived from the PA group [21], and Yu Syn5, which was derived from 16 U.S. lines from the SS and NSS germplasm [33,39]. Jilin SynA and Liaoninglv Syn were developed by crossing 13 and 12 lines derived from Chinese domestic germplasm, respectively.
4.2. Germplasm Improvement and Utilization
Generally, among the CIMMYT and the U.S. populations, and also among the Chinese populations, BS13(S)C7, BS31, and Ji Syn A were found to have high potentials for improving GY in the individual and combined environments. The BS20(S)C2, BS27, BSBB(SRCB)C4, Comp14, and Liaoninglv Syn populations also had yield-enhancing potentials in the three locations. Based on the yield performance of the 30 crosses, only 8crosses had a higher GY than that of the control. PH4CV × BS31 was slightly higher than that of Xianyu 335 in Suihua, and high yields were not observed in any of the other locations. The PH6WC × PooL 26, PH6WC × BS13(S)C7, PH6WC × BS27, PH6WC × BS31, PH6WC × BSBB(SRCB)C4, PH6WC × Ji Syn A, and PH6WC × Liaoninglv Syn crosses exhibited higher GY than Xianyu 335 in Harbin. By comparing other traits, the two crosses, PH4CV × BS31 and PH6WC × BS31 were found to exhibit higher yields, appropriate DS and EH, and resistance to lodging when compared with the check hybrid Xianyu335 (PH6WC × PH4CV) in Gongzhuling and Harbin. Additionally, maize yield is also affected by climate warming. Koimbori et al. [40] documented that increased temperatures and the decreases in both the precipitation level and sunshine hours in the NEC would occur during the 2030s and 2050s based on two climate scenarios, which would shorten the maize growth durations by (1–38 days), and maize yield would result in a reduction by (2.5–26.4%). The use of hybrids containing a diverse germplasm could offset some negative impacts of yield decrease in maize among adaptation measures. In our study, the BS31 population was derived from a highly diverse population, FS8A(T)C4, for earliness improvement of five cycles of mass selection, which composed of 52% of the germplasm from temperate and 48% from tropical regions [30]. The population has been improved for P profile of maize grain and high or low methionine concentrations through breeding and selection [41,42]. The diverse population and both groups, Reid and Lancaster germplasms, showed high heterosis in the study and, thus, the hybrids based on the new heterotic models SS × BS31 and NSS × BS31 could be resist future climate change in NEC more easily.
Studies have shown that the BS10(FR)C10 and BS13(S)C7 populations are better donors for the improvement of grain yield and head smut resistance of the hybrid Si287 × Si144 in NEC [13]. Our study also revealed a similar result, and the breeding strategy could be recommended to broaden the germplasm background in NEC to develop new inbred lines from the elite populations, BS13(S)C7, BS31, and Ji Syn A. In the recent years, the parental inbred line, PH4CV, of the elite hybrid Xianyu335 showed poor resistance to lodging for improvement during maize breeding. The alternative approach could be suggested that the BS31 and Ji Syn A populations could be used as donors to further improve the yield and resistance to lodging of PH4CV by developing crosses [(PH4CV × populations) × PH4CV]. The reason is they exhibited a favorable GCA, and the PH4CV × BS31 and PH4CV × Ji Syn A crosses also exhibited low SL and appropriate DS and EH when compared to Xianyu 335 (PH6WC × PH4CV). Another strategy can be that the Comp13 population could also be used as donors to improve resistance to the lodging by backcrossing the PH4CV × populations to the elite line, PH4CV, as it showed a negative GCA for SL in all three NEC environments, although no expected yield potential. Attention should be paid to EH selection during line development as the Comp13 population exhibited an unexpected GCA for EH. Of course, the inbred line PH4CV should be applied for authorization by Tieling Pioneer Seed Research Co., Ltd. before the practical improvement for breeders. Therefore, these elite populations would be more utilized during maize breeding programs in the future.
5. Conclusions
The combining abilities and heterosis of 15 diverse maize populations were evaluated in this study, and the U.S. and Chinese populations were found to possess the most suitable and favorable agronomic traits for NEC. The BS13(S)C7, BS31, and Ji Syn A populations showed high DS, GY, and SL potentials in both the individual and combined environments. Eight crosses, including PH4CV × BS31 in Suihua and PH6WC × PooL 26, PH6WC × BS13(S)C7, PH6WC × BS27, PH6WC × BS31, PH6WC × BSBB(SRCB)C4, PH6WC × Ji SynA, and PH6WC × Liaoninglv Syn in Harbin, showed higher yields than the check, Xianyu 335. Of these, the PH6WC × BS31 and PH6WC × Ji Syn A crosses exhibited higher yields, appropriate DS and EH, and resistance to lodging in Gongzhuling and Harbin. An alternative method to broaden the germplasm background in NEC is the development of new inbred lines from the elite BS13(S)C7, BS31, and Ji Syn A populations. Another approach could utilize the BS31, Ji Syn A, and Comp13 populations as donors to further improve yield and resistance to lodging in Chinese Lancaster germplasm, thus, broadening the genetic base of the heterotic pools in NEC.
Author Contributions
Conceptualization, X.L. and H.Y.; methodology, Y.L., Z.Y., Y.N., M.L., D.Z., Z.H., J.W., X.L. and H.Y.; software, Y.L.; formal analysis, Z.J., L.G. and Y.Y.; investigation, Z.Y., Y.S., Z.J., L.G., Y.Y., F.Z. and Y.Z.; resources, Z.J., L.G., Y.Y., X.L. and H.Y.; data curation, Y.S., F.Z. and Y.Z.; writing—original draft, Y.L., Z.Y. and Y.S.; writing—review and editing, Y.L., Y.N., M.L., D.Z., Z.H., J.W., X.L. and H.Y.; visualization, F.Z. and Y.Z.; project administration, Y.S., X.L. and H.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Key Research and Development Program of China (2022YFD1200802), and the China Agriculture Research System (CARS-02-02, CARS-02-44).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author(s).
Conflicts of Interest
The authors declare no conflicts of interest.
Appendix A

Table A1.
Mean squares from a combined analysis of variance of four traits in factorial crosses of 15 populations to two testers in three individual and combined environments.
Table A1.
Mean squares from a combined analysis of variance of four traits in factorial crosses of 15 populations to two testers in three individual and combined environments.
| Source of Variation | d.f. | DS | EH | SL | GY | Source of Variation | d.f. | DS | EH | SL | GY |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Combined | Harbin | ||||||||||
| Genotypes | 29 | 4.91 *** | 3.08 *** | 2.61 *** | 2.51 *** | Genotypes | 29 | 2.07 ** | 2.62 *** | 1.63 ** | 2.64 *** |
| Environment (Envi.) | 5 | 364.39 * | 47.71 ** | 9.76 ** | 102.19 ** | Environment (Envi.) | 1 | 12.35 * | 81.83 ** | 6.19 | 192.00 * |
| Population | 14 | 3.20 *** | 3.04 *** | 2.18 ** | 3.45 *** | Population | 14 | 2.97 *** | 3.43 *** | 1.84 ** | 2.27 ** |
| Tester | 1 | 3.74 * | 0.91 * | 13.79 *** | 3.63 * | Tester | 1 | 3.24 ** | 14.48 *** | 4.15 * | 13.22 *** |
| Population × Tester | 14 | 3.30 ** | 2.24 *** | 1.12 | 1.15 *** | Population × Tester | 14 | 0.49 *** | 0.43 *** | 1.17 | 0.76 ** |
| Genotypes × Envi. | 145 | 2.72 *** | 2.35 ** | 1.63 ** | 1.65 *** | Genotypes × Envi. | 29 | 1.05 ** | 0.37 *** | 0.45 ** | 1.37 ** |
| Population × Envi. | 70 | 1.40 ** | 0.77 * | 0.79 * | 1.62 ** | Population × Envi. | 14 | 1.02 ** | 0.61 ** | 0.37 ** | 1.35 *** |
| Tester × Envi. | 5 | 18.45 ** | 26.48 ** | 13.93 * | 11.02 *** | Tester × Envi. | 1 | 1.54 ** | 0.44 ** | 1.58 | 4.98 * |
| Population × Tester × Envi. | 14 | 0.70 ** | 0.41 | 0.86 | 0.63 ** | Population × Tester × Envi. | 14 | 0.81 ** | 0.19 ** | 0.43 ** | 0.63 ** |
| δ2A | 6.94 | 3.95 | 15.97 | 7.08 | δ2A | 6.21 | 17.91 | 5.99 | 15.49 | ||
| δ2D | 3.30 | 2.24 | 1.12 | 1.15 | δ2D | 0.49 | 0.43 | 1.17 | 0.76 | ||
| H2b | 0.07 | 0.27 | 0.61 | 0.12 | H2b | 0.83 | 0.48 | 0.88 | 0.29 | ||
| H2n | 0.02 | 0.13 | 0.36 | 0.09 | H2n | 0.62 | 0.40 | 0.56 | 0.17 | ||
| Gongzhuling | Suihua | ||||||||||
| Genotypes | 29 | 3.79 *** | 3.52 *** | 2.31 ** | 1.89 * | Genotypes | 29 | 7.10 *** | 2.18 ** | 1.86 ** | 1.94 ** |
| Environment (Envi.) | 1 | 101.21 * | 90.16 * | 0.05 | 6.44 ** | Environment (Envi.) | 1 | 7.28 ** | 84.74 ** | 65.94 * | 4.00 * |
| Population | 14 | 2.43 ** | 3.22 *** | 1.74 * | 1.92 ** | Population | 14 | 9.15 *** | 3.24 *** | 1.99 ** | 3.00 *** |
| Tester | 1 | 29.77 *** | 19.46 *** | 15.21 * | 10.37 ** | Tester | 1 | 15.29 *** | 1.36 ** | 0.71 * | 0.06 ** |
| Population × Tester | 14 | 0.80 ** | 0.72 * | 1.46 * | 0.69 ** | Population × Tester | 14 | 0.98 ** | 0.13 * | 0.41 | 0.27 ** |
| Genotypes × Envi. | 29 | 3.54 *** | 2.07 * | 1.01 * | 3.56 ** | Genotypes × Envi. | 29 | 1.47 *** | 0.47 * | 1.93 ** | 1.61 *** |
| Population × Envi. | 14 | 0.48 ** | 0.44 ** | 0.32 ** | 3.84 *** | Population × Envi. | 14 | 2.84 ** | 0.30 ** | 2.40 ** | 2.52 ** |
| Tester × Envi. | 1 | 47.91 ** | 29.71 *** | 10.35 ** | 36.91 *** | Tester × Envi. | 1 | 0.12 ** | 0.04 *** | 6.02 ** | 0.04 ** |
| Population × Tester × Envi. | 14 | 1.61 ** | 0.48 ** | 0.98 * | 0.47 ** | Population × Tester × Envi. | 14 | 0.18 ** | 0.71 ** | 0.75 ** | 1.03 ** |
| δ2A | 32.2 | 22.68 | 16.95 | 12.29 | δ2A | 24.44 | 4.60 | 2.70 | 3.06 | ||
| δ2D | 0.80 | 0.72 | 1.46 | 0.69 | δ2D | 0.98 | 0.13 | 0.41 | 0.27 | ||
| H2b | 0.52 | 0.53 | 0.99 | 0.89 | H2b | 0.97 | 0.43 | 0.45 | 0.93 | ||
| H2n | 0.30 | 0.34 | 0.55 | 0.61 | H2n | 0.67 | 0.32 | 0.24 | 0.70 |
*, **, and *** indicate p values of 0.05, 0.01, and 0.001, respectively. DS, days to silking; EH, ear height; SL, stalk lodging; GY, grain yield; δ2A, additive variance; δ2D, dominance variance; H2b, broad sense heritability; H2n, narrow sense heritability.

Table A2.
Mean performance between 15 populations and two testers for days to silking (DS), ear height (EH), stalk lodging (SL), and grain yield (GY) in Gongzhuling, Harbin, Suihua, and combined environments.
Table A2.
Mean performance between 15 populations and two testers for days to silking (DS), ear height (EH), stalk lodging (SL), and grain yield (GY) in Gongzhuling, Harbin, Suihua, and combined environments.
| Population | DS (d) | EH (cm) | ||||||||||||||
| Gongzhuling | Harbin | Suihua | Combined | Gongzhuling | Harbin | Suihua | Combined | |||||||||
| PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | |
| Tuxpeño | 85.83 # | 81.50 ¶ | 87.83 ¶ | 87.00 ¶ | 96.33 # | 96.00 # | 90.00 # | 88.17 ¶ | 148.28 # | 134.73 ¶ | 146.12 # | 139.42 ¶ | 131.63 ¶ | 136.53 ¶ | 136.68 ¶ | 136.89 ¶ |
| Suwan 1 | 85.16 ¶ | 81.00 ¶ | 88.17 ¶ | 89.33 # | 95.17 # | 99.00 # | 89.50 # | 89.78 # | 151.22 # | 127.82 ¶ | 145.00 # | 152.92 # | 142.62 # | 147.32 # | 142.61 ¶ | 142.69 ¶ |
| SG-Y | 87.16 # | 83.00 ¶ | 89.67 # | 90.00 # | 99.00 # | 100.17 # | 91.94 # | 91.06 # | 156.55 # | 131.65 ¶ | 145.22 # | 135.57 ¶ | 142.53 # | 139.81 ¶ | 144.77 ¶ | 135.68 ¶ |
| PooL 26 | 82.67 ¶ | 81.98 ¶ | 90.00 # | - † | 99.33 # | - | 90.67 # | - | 156.10 # | 158.41 # | 142.67 ¶ | - | 157.33 # | - | 148.70 # | - |
| BS10(FR)C10 | 84.00 ¶ | 83.67 ¶ | 85.67 ¶ | 87.33 ¶ | 92.17 ¶ | 95.33 # | 87.28 ¶ | 88.78 ¶ | 133.08 ¶ | 127.95 ¶ | 132.08 ¶ | 130.75 ¶ | 125.97 ¶ | 136.12 ¶ | 124.38 ¶ | 131.61 ¶ |
| BS13(S)C7 | 79.67 ¶ | 81.02 ¶ | 85.00 ¶ | - | 90.67 ¶ | - | 85.11 ¶ | - | 140.83 ¶ | 143.63 ¶ | 134.67 ¶ | - | 116.43 ¶ | - | 123.98 ¶ | - |
| BS20(S)C2 | 85.50 # | 84.00 ¶ | 86.67 ¶ | 87.33 ¶ | 93.33 # | 94.00 # | 88.50 ¶ | 88.44 ¶ | 140.31 ¶ | 122.27 ¶ | 123.40 ¶ | 124.47 ¶ | 132.98 ¶ | 138.62 ¶ | 132.23 ¶ | 128.45 ¶ |
| BS27 | 82.33 ¶ | 82.12 ¶ | 87.67 ¶ | - | 92.00 ¶ | - | 87.33 ¶ | - | 149.50 # | 149.84 # | 133.00 ¶ | - | 134.07 ¶ | - | 128.86 ¶ | - |
| BS31 | 81.33 ¶ | 80.98 ¶ | 86.67 ¶ | - | 90.33 ¶ | - | 86.11 ¶ | - | 141.33 ¶ | 143.33 ¶ | 128.67 ¶ | - | 131.50 ¶ | - | 138.17 ¶ | - |
| BSBB(SRCB)C4 | 80.33 ¶ | 80.17 ¶ | 85.67 ¶ | - | 89.33 ¶ | - | 85.11 ¶ | - | 137.17 ¶ | 138.37 ¶ | 127.33 ¶ | - | 134.27 ¶ | - | 129.59 ¶ | - |
| BSSS(R)C10 | 84.83 ¶ | 82.00 ¶ | 87.33 ¶ | 87.50 ¶ | 92.17 ¶ | 93.80 # | 88.11 ¶ | 87.77 ¶ | 133.90 ¶ | 124.78 ¶ | 129.40 ¶ | 124.12 ¶ | 127.38 ¶ | 131.62 ¶ | 126.89 ¶ | 126.84 ¶ |
| Comp13 | 85.50 # | 82.00 ¶ | 88.33 ¶ | 89.83 # | 93.67 # | 96.83 # | 89.17 # | 89.55 # | 143.60 ¶ | 133.73 ¶ | 127.60 ¶ | 133.98 ¶ | 134.70 ¶ | 143.20 # | 135.30 ¶ | 136.97 ¶ |
| Comp14 | 84.83 ¶ | 81.33 ¶ | 86.33 ¶ | 88.00 ¶ | 93.33 # | 94.17 # | 88.16 ¶ | 87.83 ¶ | 136.82 ¶ | 132.40 ¶ | 126.07 ¶ | 129.05 ¶ | 131.05 ¶ | 137.08 ¶ | 131.31 ¶ | 132.84 ¶ |
| Ji Syn A | 82.33 ¶ | 81.78 ¶ | 87.33 ¶ | - | 91.00 ¶ | - | 86.89 ¶ | - | 161.67 # | 162.70 # | 121.67 ¶ | - | 159.47 # | - | 144.27 # | - |
| Liaoninglv Syn | 80.33 ¶ | 80.19 ¶ | 87.33 ¶ | 87.00 ¶ | 92.67 ¶ | - | 86.78 ¶ | - | 142.03 ¶ | 144.19 ¶ | 131.00 ¶ | 126.33 ¶ | 125.37 ¶ | - | 132.80 ¶ | - |
| SD | 82.24 ± 2.29 | 82.00 ± 2.69 | 66.99 ± 37.22 | 72.59 ± 38.02 | 87.01 ± 6.34 | 91.04 ± 7.15 | 88.04 ± 1.97 | 88.92 ± 1.14 | 131.02 ± 21.49 | 134.75 ± 21.67 | 123.86 ± 17.77 | 137.59 ± 19.74 | 116.70 ± 16.32 | 126.69 ± 21.01 | 134.70 ± 7.71 | 133.99 ± 5.14 |
| Xianyu 335 | 85.33 | 88.67 | 92.80 | 88.93 | 145.17 | 144.67 | 142.33 | 144.05 | ||||||||
| Population | SL (%) | GY (t/ha) | ||||||||||||||
| Gongzhuling | Harbin | Suihua | Combined | Gongzhuling | Harbin | Suihua | Combined | |||||||||
| PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | PH4CV | PH6WC | |
| Tuxpeño | 5.00 # | 1.00 ¶ | 9.00 # | 8.00 # | 6.00 # | 4.00 # | 8.67 # | 4.67 # | 9.20 ¶ | 9.74 ¶ | 8.20 ¶ | 8.03 ¶ | 6.37 ¶ | 6.64 ¶ | 7.92 ¶ | 8.14 ¶ |
| Suwan 1 | 3.00 # | 1.00 ¶ | 4.00 # | 4.00 # | 3.00 # | 2.00 # | 3.67 # | 2.33 # | 10.01 ¶ | 9.14 ¶ | 8.58 ¶ | 9.55 ¶ | 7.00 ¶ | 6.97 ¶ | 8.53 ¶ | 8.55 ¶ |
| SG-Y | 2.00 # | 3.00 # | 6.00 # | 7.00 # | 4.00 # | 2.00 # | 4.00 # | 4.00 # | 9.61 ¶ | 8.77 ¶ | 8.10 ¶ | 7.79 ¶ | 5.94 ¶ | 6.31 ¶ | 7.88 ¶ | 7.62 ¶ |
| PooL 26 | 4.00 # | 4.00 # | 3.00 # | 0.00 ¶ | 0.00 ¶ | 0.00 ¶ | 4.67 # | 1.33 ¶ | 10.31 ¶ | 10.46 ¶ | 7.97 ¶ | 10.89 # | 6.81 ¶ | - | 8.36 ¶ | - |
| BS10(FR)C10 | 2.00 # | 0.00 ¶ | 3.00 # | 3.00 # | 8.00 # | 3.00 # | 4.33 # | 2.00 # | 10.01 ¶ | 8.36 ¶ | 8.92 ¶ | 9.77 ¶ | 7.03 ¶ | 8.12 ¶ | 8.65 ¶ | 8.75 ¶ |
| BS13(S)C7 | 2.00 # | 2.00 # | 2.00 # | 0.00 ¶ | 0.00 ¶ | 0.00 ¶ | 1.33 ¶ | 0.67 ¶ | 9.11 ¶ | 9.23 ¶ | 9.40 ¶ | 11.82 # | 7.65 ¶ | - | 8.72 ¶ | - |
| BS20(S)C2 | 6.00 # | 2.00 # | 12.00 # | 5.00 # | 3.00 # | 4.00 # | 7.00 # | 3.67 # | 10.19 ¶ | 9.57 ¶ | 9.32 ¶ | 9.40 ¶ | 7.95 ¶ | 7.84 ¶ | 9.15 ¶ | 8.94 ¶ |
| BS27 | 9.00 # | 9.00 # | 23.00 # | 0.00 ¶ | 0.00 ¶ | 0.00 ¶ | 10.67 # | 3.00 # | 9.77 ¶ | 9.87 ¶ | 8.33 ¶ | 11.77 # | 7.54 ¶ | - | 8.55 ¶ | - |
| BS31 | 1.00 ¶ | 1.00 ¶ | 2.00 # | 0.00 ¶ | 0.00 ¶ | 0.00 ¶ | 1.00 ¶ | 0.33 ¶ | 10.05 ¶ | 10.21 ¶ | 9.16 ¶ | 11.62 # | 8.82 # | - | 9.34 ¶ | - |
| BSBB(SRCB)C4 | 5.00 # | 5.00 # | 7.00 # | 0.00 ¶ | 0.00 ¶ | 0.00 ¶ | 4.00 # | 1.67 # | 8.24 ¶ | 8.37 ¶ | 9.67 ¶ | 11.86 # | 8.34 ¶ | - | 8.75 ¶ | - |
| BSSS(R)C10 | 10.00 # | 1.00 ¶ | 12.00 # | 4.00 # | 8.00 # | 5.00 # | 10.00 # | 3.33 # | 9.08 ¶ | 9.29 ¶ | 8.14 ¶ | 9.24 ¶ | 7.90 ¶ | 8.11 ¶ | 8.37 ¶ | 8.88 ¶ |
| Comp13 | 4.00 # | 1.00 ¶ | 6.00 # | 7.00 # | 2.00 # | 1.00 ¶ | 4.00 # | 3.00 # | 11.10 ¶ | 9.49 ¶ | 9.43 ¶ | 8.38 ¶ | 7.15 ¶ | 7.21 ¶ | 9.23 ¶ | 8.36 ¶ |
| Comp14 | 7.00 # | 0.00 ¶ | 9.00 # | 1.00 ¶ | 4.00 # | 1.00 ¶ | 6.67 # | 0.67 ¶ | 10.28 ¶ | 9.98 ¶ | 8.64 ¶ | 9.52 ¶ | 8.11 ¶ | 8.13 ¶ | 9.01 ¶ | 9.21 ¶ |
| Ji Syn A | 1.00 ¶ | 1.00 ¶ | 7.00 # | 0.00 ¶ | 0.00 ¶ | 0.00 ¶ | 2.67 # | 0.33 ¶ | 11.27 ¶ | 11.39 ¶ | 9.62 ¶ | 11.61 # | 8.19 ¶ | - | 9.69 ¶ | - |
| Liaoninglv Syn | 7.00 # | 7.00 # | 4.00 # | 8.00 # | 0.00 ¶ | 0.00 ¶ | 3.67 # | 5.00 # | 9.59 ¶ | 10.01 ¶ | 8.82 ¶ | 11.43 # | 7.08 ¶ | - | 8.50 ¶ | - |
| SD | 3.13 ± 4.92 | 1.47 ± 3.06 | 5.86 ± 7.74 | 2.21 ± 5.14 | 5.36 ± 7.75 | 2.20 ± 5.46 | 5.09 ± 2.92 | 2.40 ± 1.56 | 9.36 ± 1.69 | 9.43 ± 1.54 | 8.82 ± 2.15 | 7.53 ± 1.50 | 8.22 ± 1.97 | 7.68 ± 1.51 | 8.71 ± 0.50 | 9.54 ± 1.81 |
| Xianyu 335 | 1.41 | 1.53 | 1.63 | 1.52 | 11.49 | 10.61 | 8.72 | 10.27 | ||||||||
# The value is higher than CK; ¶ The value is lower than CK; † Lack of data.
References
- Kandil, E.E.; Abdelsalam, N.R.; Mansour, M.A.; Ali, H.M.; Siddiqui, M.H. Potentials of organic manure and potassium forms on maize (Zea mays L.) growth and production. Sci. Rep. 2020, 10, 8752. [Google Scholar] [CrossRef]
- National Bureau of Statistics of China (NSBC). China Statistics Yearbook; China Statistics Press: Beijing, China, 2021. [Google Scholar]
- Gong, F.; Wu, X.; Zhang, H.; Chen, Y.; Wang, W. Making better maize plants for sustainable grain production in a changing climate. Front. Plant Sci. 2015, 6, 835. [Google Scholar] [CrossRef] [PubMed]
- Yong, H.J.; Tang, J.; Zhao, X.Z.; Zhang, F.Y.; Yang, Z.Y.; Li, Y.; Li, M.S.; Zhang, D.G.; Hao, Z.F.; Weng, J.F.; et al. Effect of five modified mass selection cycles on combining ability in two Chinese maize populations. Euphytica 2020, 216, 57. [Google Scholar] [CrossRef]
- Yong, H.J.; Zhang, X.C.; Zhang, D.G.; Wang, J.J.; Zhang, H.X.; Li, M.S.; Liu, W.G.; Weng, J.F.; Hao, Z.F.; Ci, X.K.; et al. Breeding potential of U.S. maize germplasm for utilization in Chinese temperate conditions. Euphytica 2013, 192, 435–451. [Google Scholar] [CrossRef]
- Ndoro, O.; Magorokosho, C.; Setimela, P.S.; Kamutando, C.N.; Labuschagne, M.T. Identification of exotic temperate maize inbreds for use in tropical breeding programs. Euphytica 2022, 218, 164. [Google Scholar] [CrossRef]
- Ke, F.; Ma, X. Responses of maize hybrids with contrasting maturity to planting date in Northeast China. Sci. Rep. 2021, 11, 15776. [Google Scholar] [CrossRef]
- Dong, M.; Zhao, J.; Li, E.; Liu, Z.; Guo, S.; Zhang, Z.; Cui, W.; Yang, X. Effects of changing climate extremes on maize grain yield in Northeast China. Agronomy 2023, 13, 1050. [Google Scholar] [CrossRef]
- Glover, M.A.; Willmot, D.B.; Darrah, L.L.; Hibbard, B.E.; Zhu, X. Diallel analyses of agronomic traits using Chinese and U.S. maize germplasm. Crop Sci. 2005, 45, 1096–1102. [Google Scholar] [CrossRef]
- Goodman, M.M. Broadening the U.S. maize germplasm base. Maydica 2005, 50, 203–214. [Google Scholar]
- Cupertino-Rodrigues, M.; Dhliwayo, T.; Trachsel, S.; Guo, R.; San, V.F. Evaluation of U.S. inbred lines with expired plant variety protection for mid-altitude tropical maize breeding. Euphytica 2020, 216, 44. [Google Scholar] [CrossRef]
- Rogers, A.R.; Bian, Y.; Krakowsky, M.; Peters, D.; Turnbull, C.; Nelson, P.; Holland, J.B. Genomic prediction for the germplasm enhancement of maize project. Plant Genome 2022, 15, e20267. [Google Scholar] [CrossRef] [PubMed]
- Yong, H.J.; Jin, Z.; Gao, L.; Zhang, L.; Liu, X.; Zhang, F.; Zhang, X.; Zhang, D.; Li, M.S.; Weng, J.F.; et al. Breeding potential of maize germplasm populations to improve yield and predominant heterotic pattern in Northeast China. Euphytica 2017, 213, 219. [Google Scholar] [CrossRef]
- Fan, X.M.; Zhang, Y.D.; Jeffers, D.P.; Bi, Y.Q.; Kang, M.S.; Yin, X.F. Combining ability of yellow lines derived from CIMMYT populations for use in subtropical and tropical midaltitude maize production environments. Crop Sci. 2018, 58, 169–179. [Google Scholar] [CrossRef]
- Azizdoost, H.; Shiri, M.; Dezhsetan, S. The performance of temperate maize testers for screening tropical and subtropical germplasm. Cereal Res. 2024, 13, 367–384. [Google Scholar] [CrossRef]
- Yong, H.J.; Zhang, F.Y.; Tang, J.; Yang, Z.Y.; Zhao, X.Z.; Li, M.S.; Zhang, D.G.; Hao, Z.F.; Weng, J.F.; Li, X.H. Breeding potential of inbred lines derived from five maize (Zea mays L.) populations. Euphytica 2019, 215, 1. [Google Scholar] [CrossRef]
- Smith, J.S.; Trevisan, W.; McCunn, A.; Huffman, W.E. Global dependence on Corn Belt Dent maize germplasm: Challenges and opportunities. Crop Sci. 2022, 62, 2039–2066. [Google Scholar] [CrossRef]
- Hallauer, A.R.; Carena, M.J. Maize breeding. In Handbook of Plant Breeding: Cereals; Carena, M.J., Ed.; Springer: New York, NY, USA, 2009; pp. 3–98. [Google Scholar]
- Rizzo, G.; Monzon, J.P.; Tenorio, F.A.; Réka, H.; Cassman, K.G.; Grassini, P. Climate and agronomy, not genetics, underpin recent maize yield gains in favorable environments. Proc. Natl. Acad. Sci. USA 2022, 119, e2113629119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.H.; Bonjean, A.P.A. Maize breeding and production in China. In Cereals in China; He, Z., Bonjean, A.P.A., Eds.; CIMMYT: El Batan, Mexico, 2010; pp. 35–49. [Google Scholar]
- Zhang, R.Y.; Xu, G.; Li, J.S.; Yan, J.B.; Li, H.H.; Yang, X.H. Patterns of genomic variation in Chinese maize inbred lines and implications for genetic improvement. Theor. Appl. Genet. 2018, 131, 1207–1221. [Google Scholar] [CrossRef] [PubMed]
- Alhaj, H.Y.; Shaghaleh, H.; Zhang, K.; Okla, M.K.; Alaraidh, I.A.; AbdElgawad, H.; Sheteiwy, M.S. Calcium lignosulfonate-induced modification of soil chemical properties improves physiological traits and grain quality of maize (Zea mays) under salinity stress. Front. Plant Sci. 2024, 15, 1397552. [Google Scholar] [CrossRef]
- Baloch, N.; Liu, W.M.; Hou, P.; Ming, B.; Xie, R.Z.; Wang, K.R.; Liu, Y.E.; Li, S.K. Effect of latitude on maize kernel weight and grain yield across China. Agron. J. 2020, 113, 1172–1182. [Google Scholar] [CrossRef]
- Yong, H.J.; Zhang, D.G.; Wang, J.J.; Li, M.S.; Liu, W.G.; Zhang, X.C.; Zhao, H.Y.; Weng, J.F.; Hao, Z.F.; Bai, L.; et al. Broadening the genetic base of Chinese maize heterotic pools with exotic germplasm. Crop Sci. 2013, 53, 1907–1916. [Google Scholar] [CrossRef]
- Prasanna, B.M.; Burgueño, J.; Beyene, Y.; Makumbi, D.; Asea, G.; Woyengo, V.; Tarekegne, A.; Magorokosho, C.; Wegary, D.; Ndhlela, T.; et al. Genetic trends in CIMMYT’s tropical maize breeding pipelines. Sci. Rep. 2022, 12, 20110. [Google Scholar] [CrossRef]
- Hallauer, A.R.; Carena, M.J. Adaptation of tropical maize germplasm to temperate environments. Euphytica 2014, 196, 1–11. [Google Scholar] [CrossRef]
- Olmos, S.E.; Lia, V.V.; Eyhérabide, G.H. Genetic diversity and linkage disequilibrium in the Argentine public maize inbred line collection. Plant Genet. Resour. 2017, 15, 515–526. [Google Scholar] [CrossRef]
- Melani, M.D.; Carena, M.J. Alternative maize heterotic patterns for the Northern Corn Belt. Crop Sci. 2005, 45, 2139–2677. [Google Scholar] [CrossRef]
- Hallauer, A.R. Registration of BS27 maize germplasm. Crop Sci. 1992, 32, 1512–1513. [Google Scholar] [CrossRef]
- Sen, H.; Kumar, A.; Janeja, H.S. Biofortification of major crops through conventional and modern biotechnological approaches to fight hidden hunger: An overview. J. Adv. Biol. Biotechnol. 2024, 27, 96–113. [Google Scholar] [CrossRef]
- Jumbo, M.B.; Carena, M.J. Combining ability, maternal, and reciprocal effects of elite early-maturing maize population hybrids. Euphytica 2008, 162, 325–333. [Google Scholar] [CrossRef]
- Edwards, J. Changes in plant morphology in response to recurrent selection in the Iowa stiff stalk synthetic maize population. Crop Sci. 2011, 51, 2352–2361. [Google Scholar] [CrossRef]
- Yong, H.J.; Wang, L.M.; Li, M.S.; Zhang, D.G.; Pan, G.T.; Zhang, S.H.; Li, X.H.; Rong, T.Z. Breeding potential analysis of eight populations in the application of maize breeding in northeast China. J. Nucl. Agric. Sci. 2014, 28, 2139–2147, (With Chinese Abstract). [Google Scholar]
- Yang, K.; Tan, J.; Zhang, Q.; Bai, T.; Zhou, S.; Hao, J.; Yu, X.; Zang, Z.; Zhang, D. Research on the genetic improvement effects of lodging resistance-related traits in maize core germplasm. Agronomy 2025, 15, 17. [Google Scholar] [CrossRef]
- China Meteorological Administration (CMA). Ground Meteorological Data in China. Available online: http://data.cma.cn/ (accessed on 28 August 2022).
- Butler, D.G.; Cullis, B.R.; Gilmour, A.R.; Gogel, B.J.; Thompson, R. ASReml-R Reference Manual, version 4; VSN International Ltd: Hemel Hempstead, UK, 2017; Available online: https://mmade.org/wp-content/uploads/2021/08/asremlRMfinal.pdf (accessed on 18 August 2021).
- Șimon, A.; Moraru, P.I.; Ceclan, A.; Russu, F.; Chețan, F.; Bărdaș, M.; Popa, A.; Rusu, T.; Pop, A.I.; Bogdan, I. The impact of climatic factors on the development stages of maize crop in the transylvanian plain. Agronomy 2023, 13, 1612. [Google Scholar] [CrossRef]
- Rasmussen, C.C.; Hallauer, A.R. Evaluation of heterotic patterns of Iowa stiff stalk synthetic and non-stiff stalk synthetic maize populations. Maydica 2006, 51, 177–186. [Google Scholar]
- Ku, L.X.; Meng, Q.L.; Hou, B.J.; Li, J.F.; Liu, H.J.; Chen, Y.H. Effificiency of recurrent selection for combining ability of yield trait in maize population Yu Syn5. Acta Agron. Sin. 2012, 38, 215–222, (With Chinese Abstract). [Google Scholar] [CrossRef]
- Koimbori, J.K.; Wang, S.; Pan, J.; Guo, L.; Li, K. Yield response of spring maize under future climate and the effects of adaptation measures in Northeast China. Plants 2022, 11, 1634. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, A.J.; Scott, M.P.; Lamkey, K.R. Genetic variation and breeding potential of phytate and inorganic phosphorus in a maize population. Crop Sci. 2008, 48, 79–84. [Google Scholar] [CrossRef]
- Scott, M.P.; Darrigues, A.; Stahly, T.S.; Lamkey, K.R. Recurrent selection to alter grain methionine concentration and improve nutritional value of maize. Crop Sci. 2008, 48, 1705–1713. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).