Effects of Apatite Concentrate in Combination with Phosphate-Solubilizing Microorganisms on the Yield of Ryegrass Cultivar Izorskiy
Abstract
1. Introduction
2. Materials and Methods
2.1. Experiment at the Artificial Climate Facility
Soil Characteristics
2.2. Microorganism Cultures
2.3. Biologized Apatite Concentrates
- -
- Coarse concentrate ground and sifted under standard conditions, “residue on a sieve with mesh No. 016K, no more than 20% (≤20% of particles larger than 0.16 mm)” and used for the production of phosphate fertilizers;
- -
- Apatite crushed in a disintegrator to a particle size of about 20 μm, which should allow PSMs to more effectively decompose hard-to-reach P compounds from apatite.
2.4. Characteristics of Ryegrass Variety Izorsky
2.5. Analytical Methods
2.6. P Utilization Efficiency for Ryegrass Plants
2.7. Experimental Scheme
2.8. Analysis of Plant Growth
2.9. Statistical Analysis
3. Results
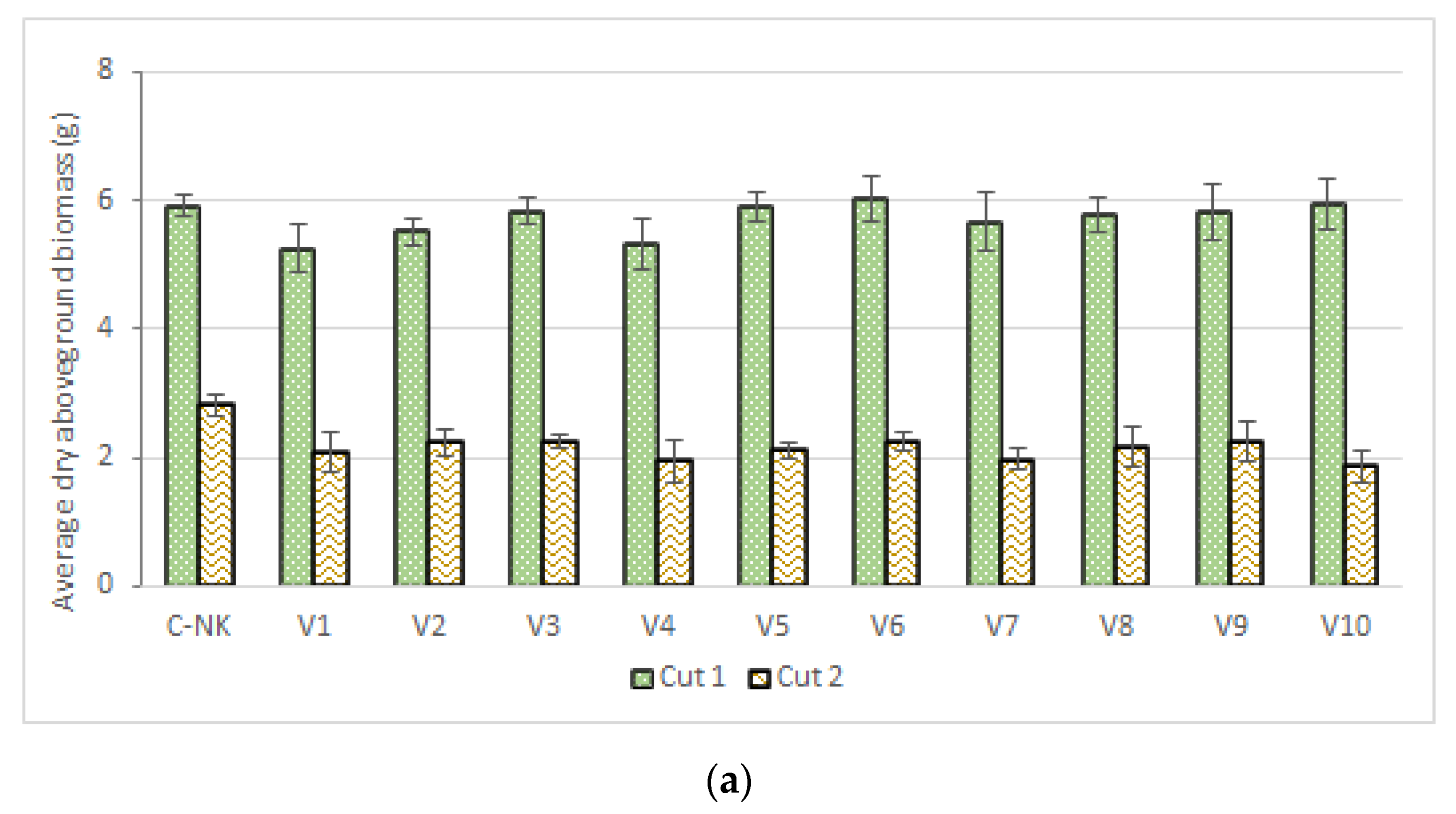
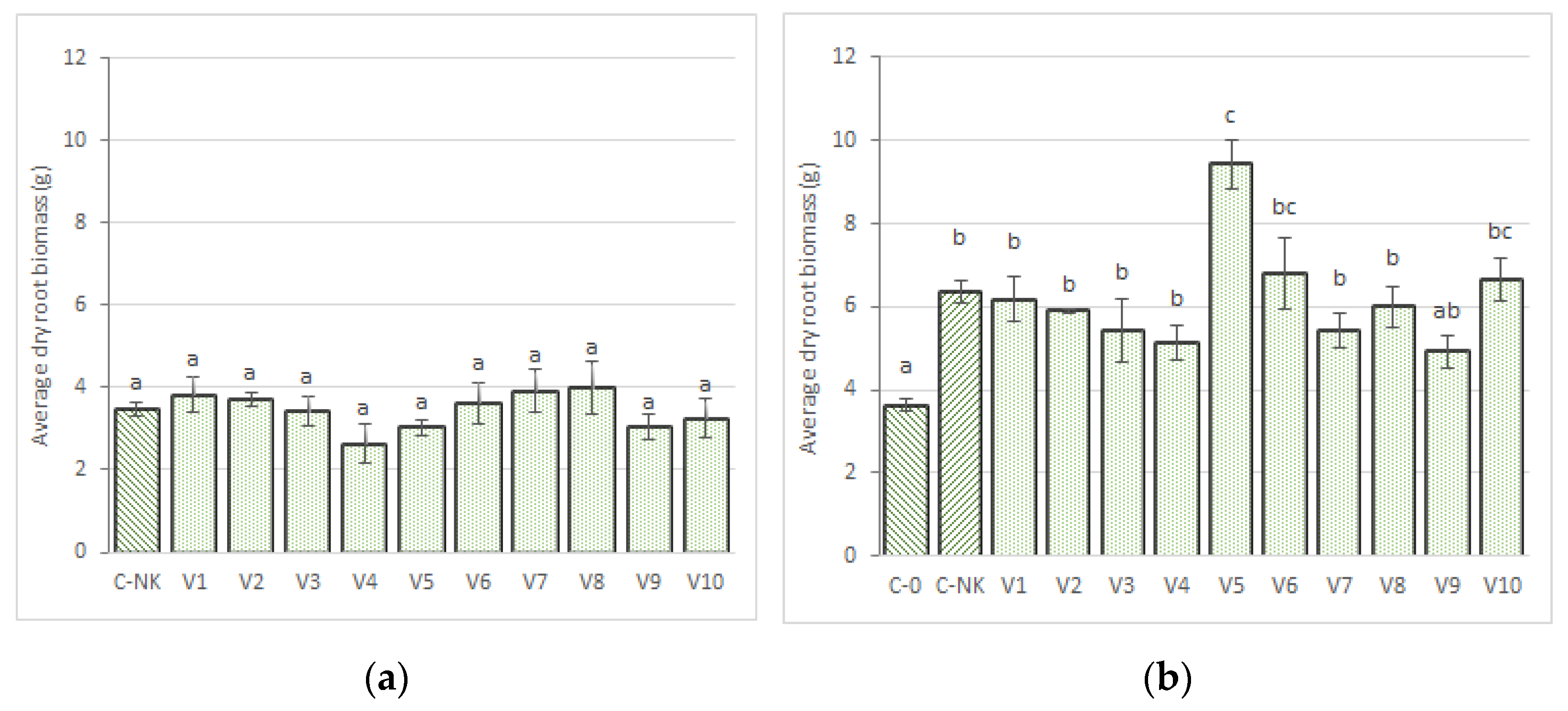
3.1. Biomass Accumulation
3.2. The Effect of Apatite and PSMs on the Accumulation of Microelements in Ryegrass Biomass
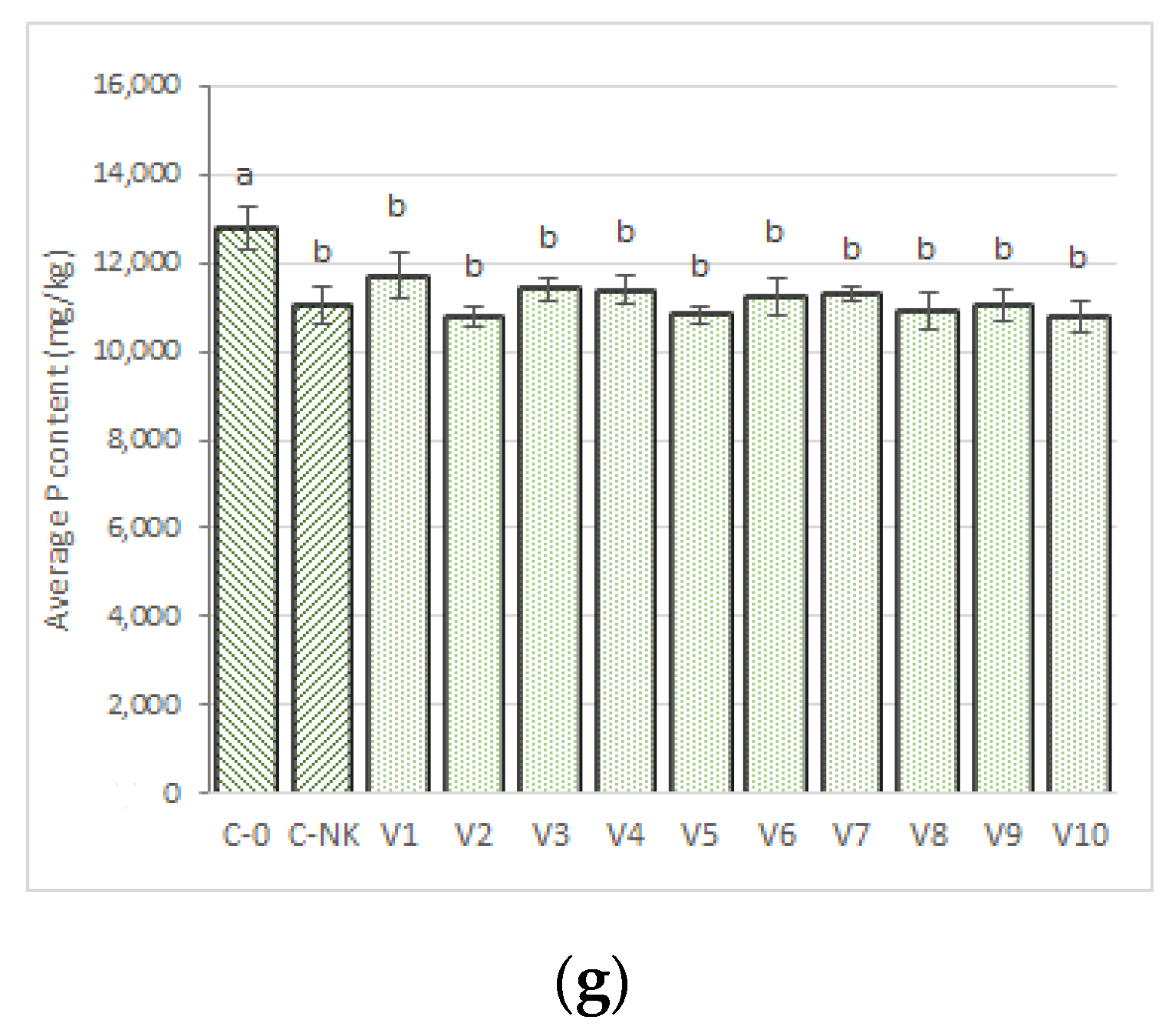
3.2.1. Phosphorus
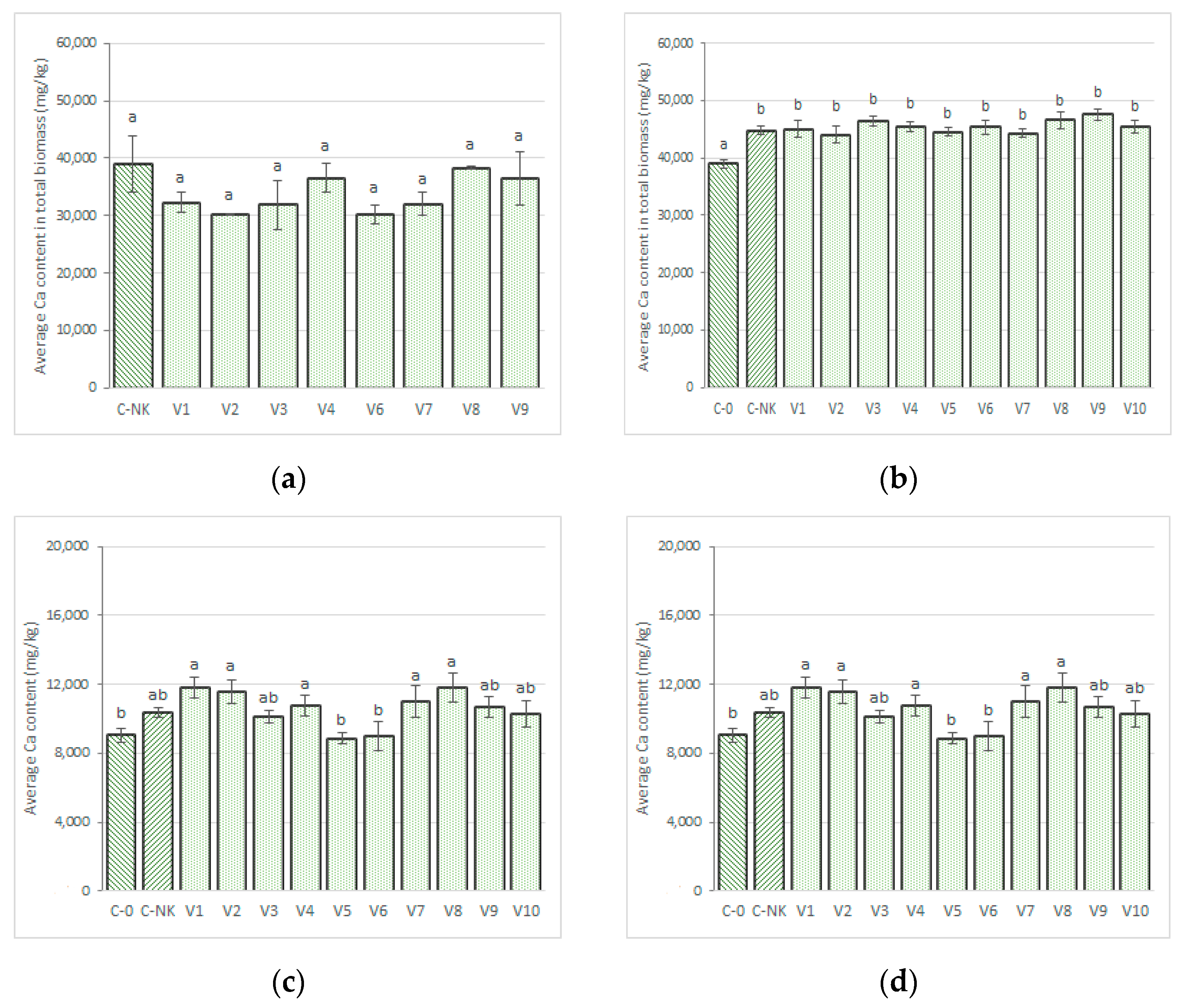
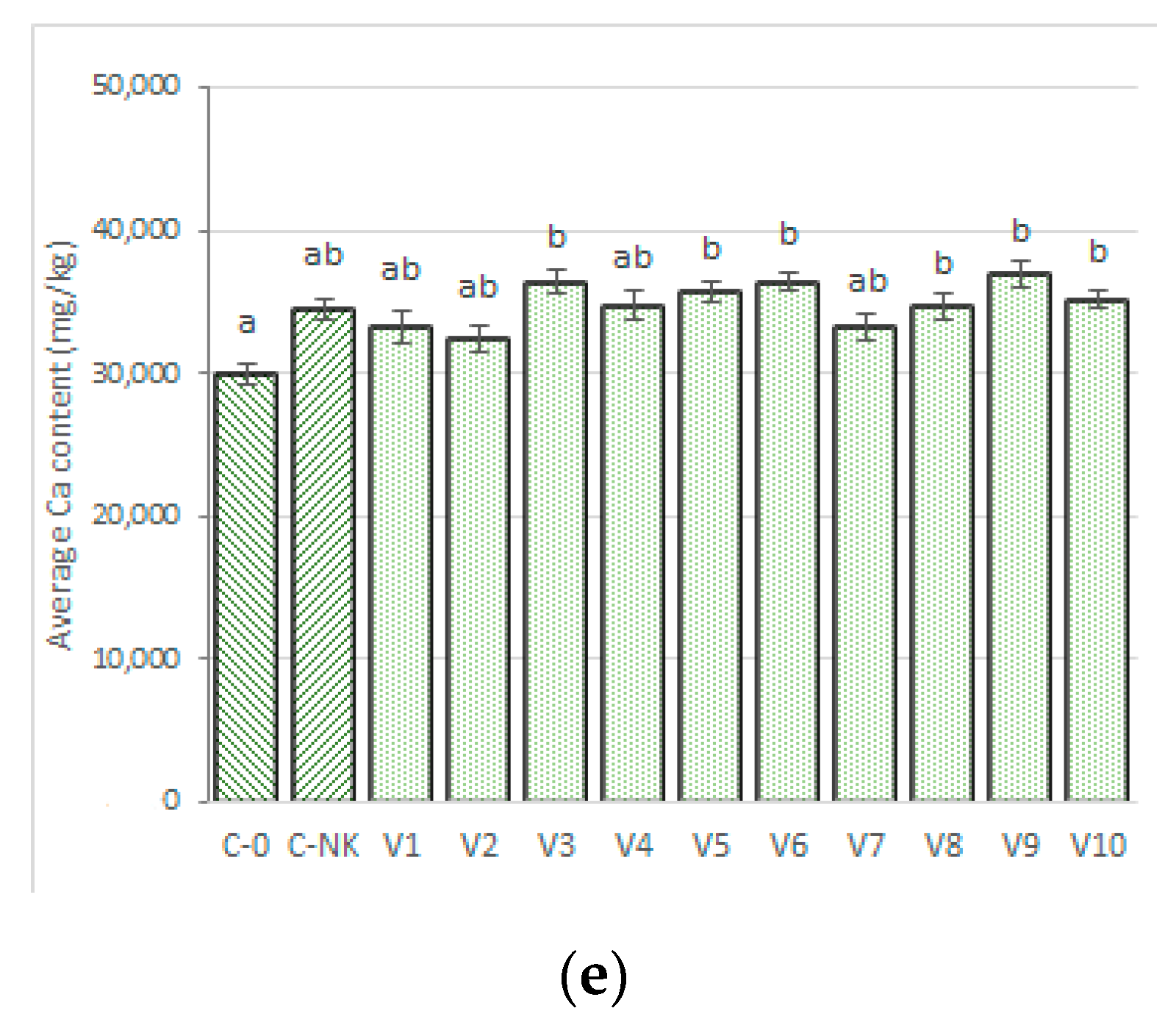
3.2.2. Calcium
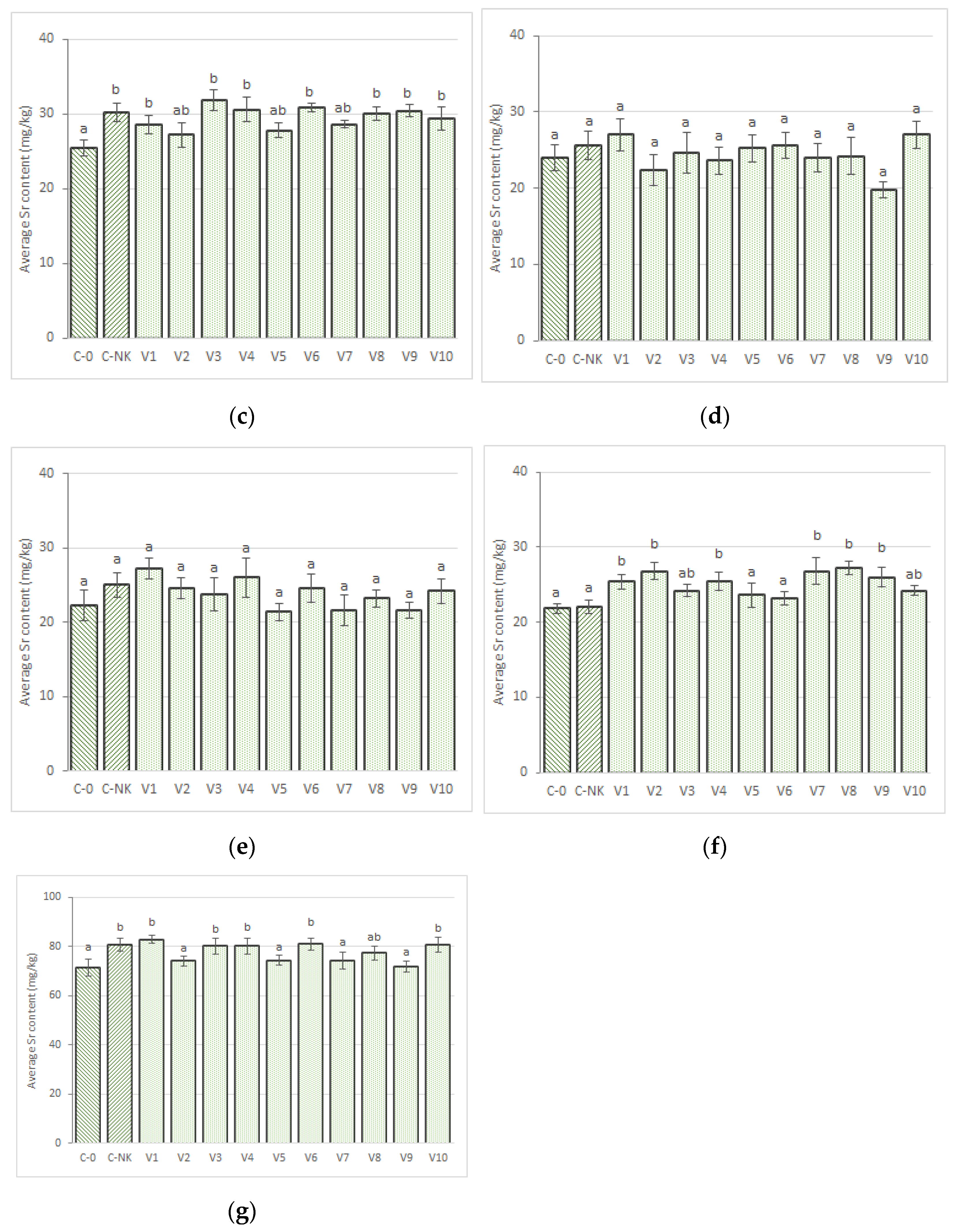
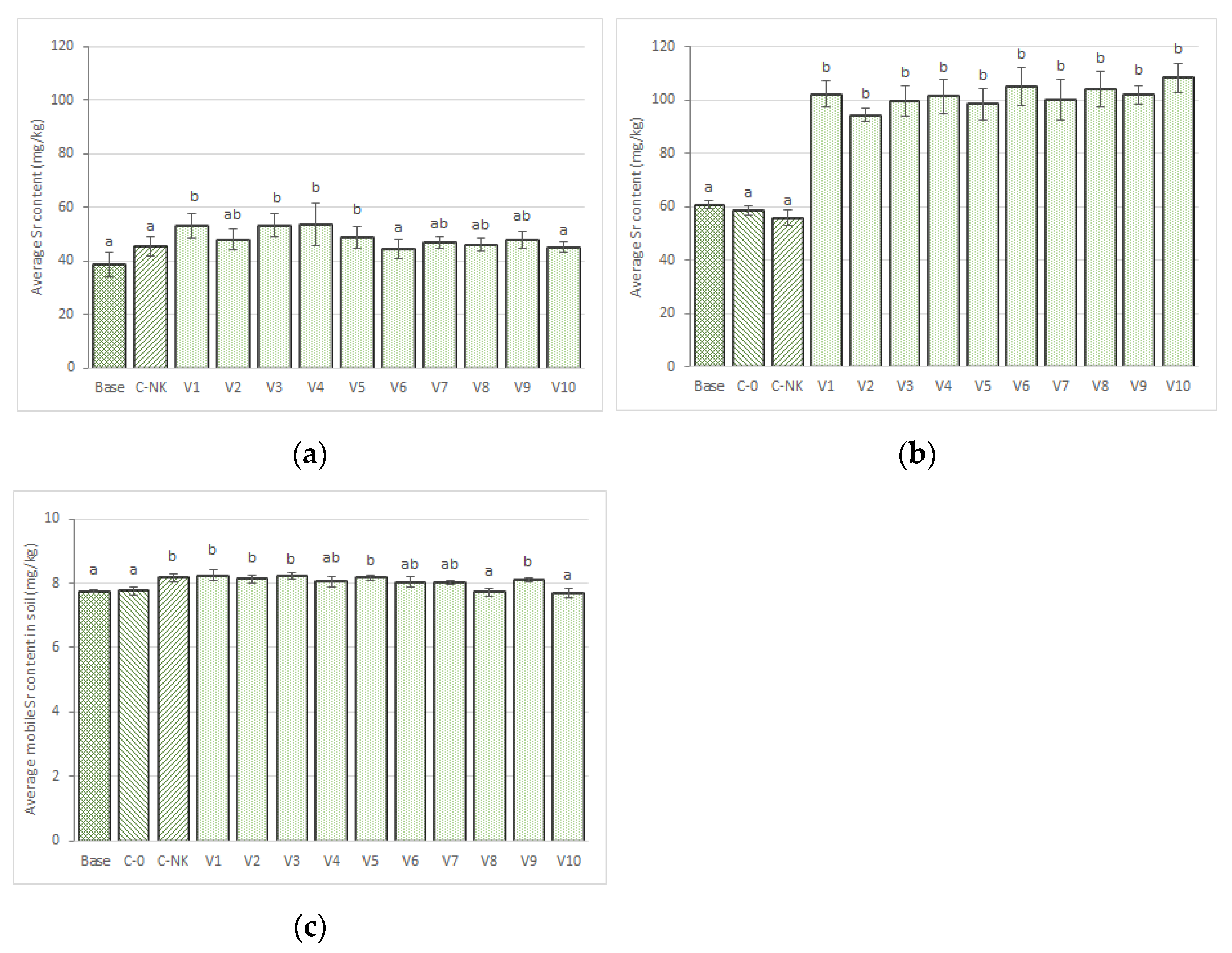
3.2.3. Strontium
3.3. The Effecst of Apatite and PSMs on Soil-Related Parameters
3.3.1. Soil Acidity
3.3.2. Organic Matter
3.3.3. Nitrogen
3.3.4. Potassium
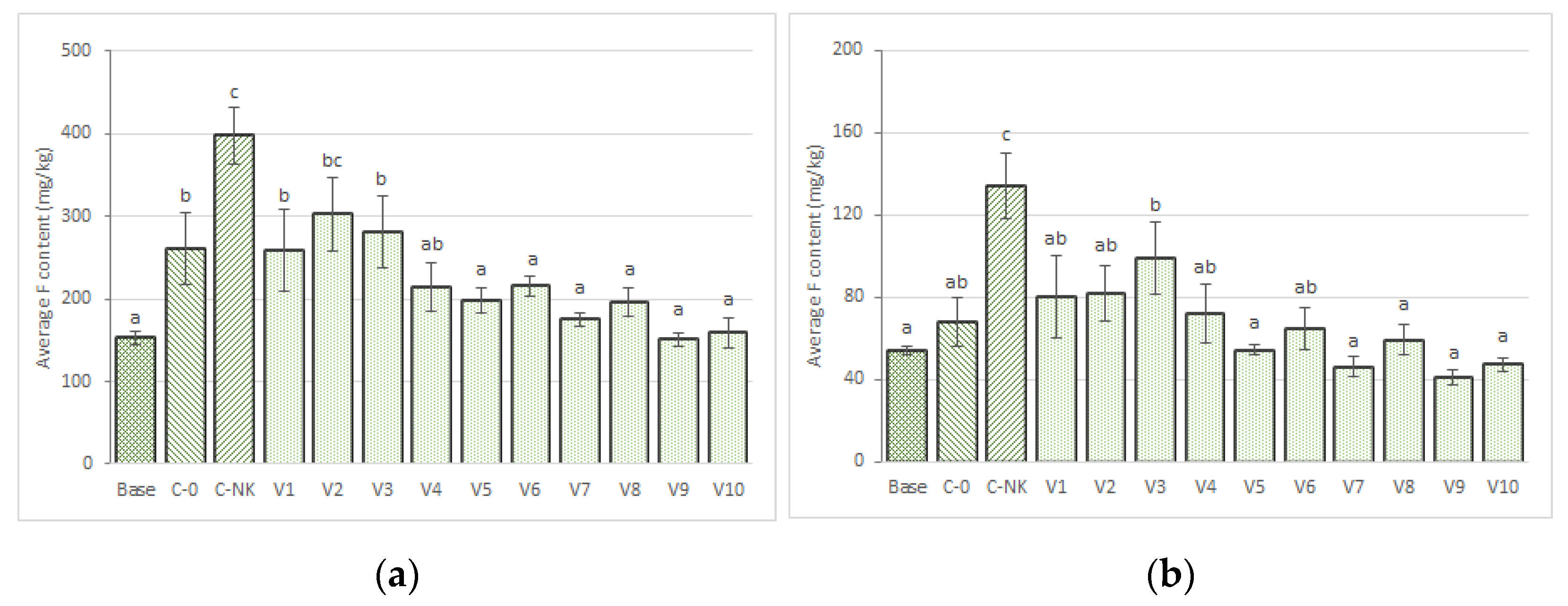
3.3.5. Fluorine
3.3.6. Phosphorus
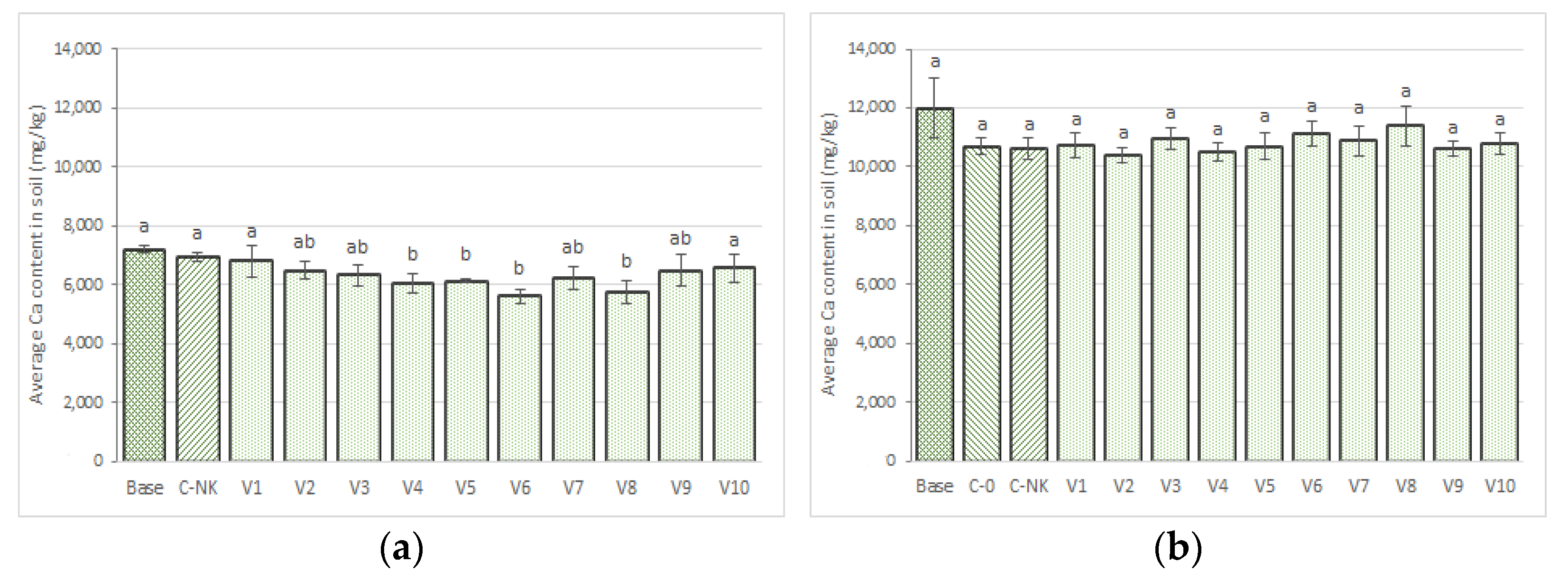
3.3.7. Calcium
3.3.8. Strontium
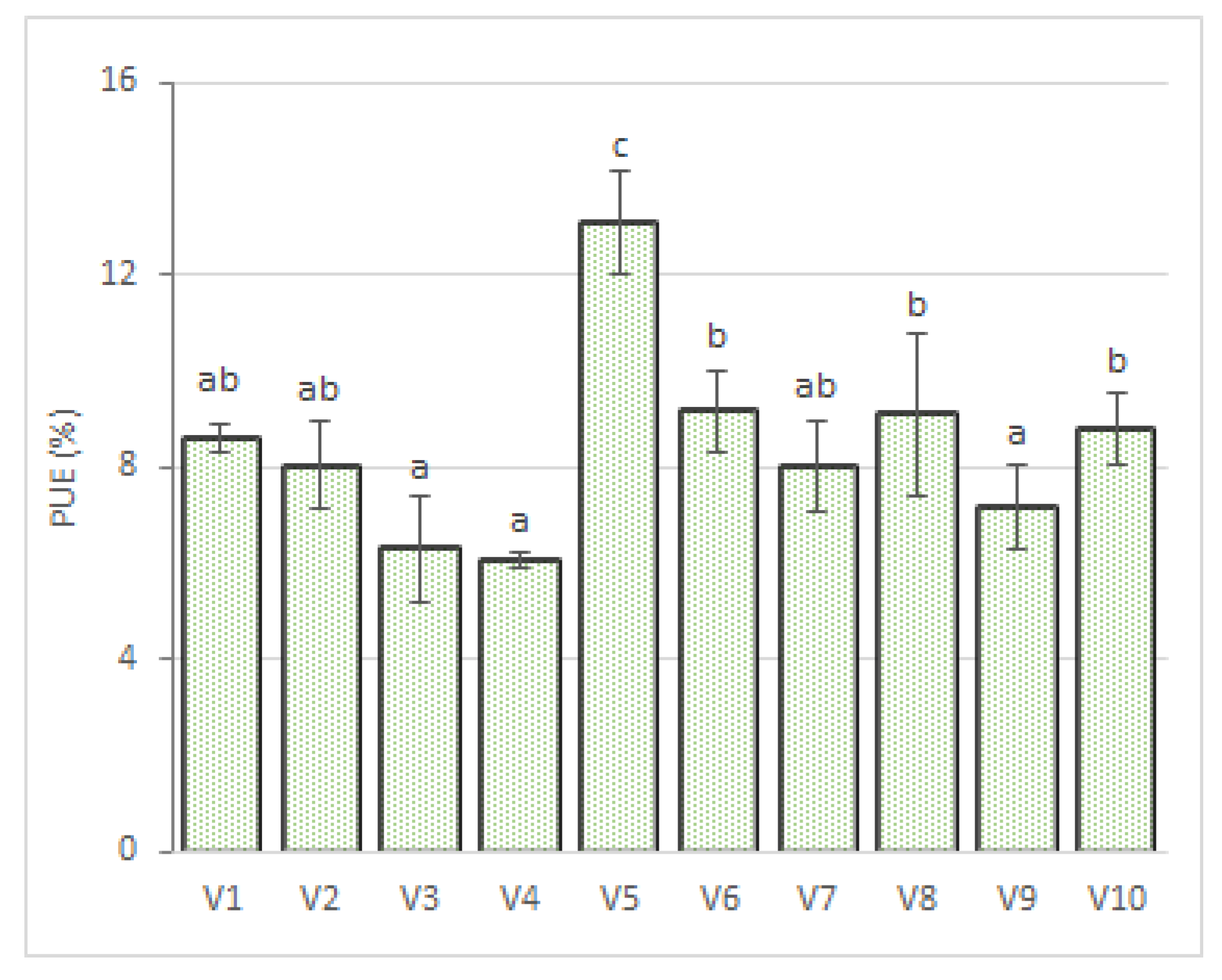
3.4. The Efficiency of P Utilization by Ryegrass
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alori, E.T.; Glick, B.R.; Babalola, O.O. Microbial Phosphorus Solubilization and Its Potential for Use in Sustainable Agriculture. Front. Microbiol. 2017, 8, 971. [Google Scholar] [CrossRef]
- Carmo, T.S.D.; Moreira, F.S.; Cabral, B.V.; Dantas, R.C.C.; de Resende, M.M.; Cardoso, V.L.; Ribeiro, E.J. Phosphorus Recovery from Phosphate Rocks Using Phosphate-Solubilizing Bacteria. Geomicrobiol. J. 2019, 36, 195–203. [Google Scholar] [CrossRef]
- FAO. The International Code of Conduct for the Sustainable Use and Management of Fertilizers; Licence: CC BY-NC-SA 3.0 IGO; FAO: Rome, Italy, 2019. [Google Scholar]
- Alaylar, B. Isolation of PGPR strains with phosphate solubilizing activity from Erzurum and their molecular evaluation by using newly designed specific primer for pqqB gene. IJSER 2018, 9, 103–106. [Google Scholar]
- Alaylar, B.; Güllüce, M.; Karadayi, M.; Isaoglu, M. Rapid Detection of Phosphate-Solubilizing Bacteria from Agricultural Areas in Erzurum. Curr. Microbiol. 2019, 76, 804–809. [Google Scholar] [CrossRef]
- Alaylar, B.; Güllüce, M.; Karadayı, G. Detection of the nifH gene in nitrogen-fixing bacteria from agricultural areas in Erzurum. Fresenius Environ. Bull. 2020, 29, 809–814. [Google Scholar]
- Majeed, A.; Farooq, M.; Naveed, M.; Hussain, M. Combined Application of Inorganic and Organic Phosphorous with Inoculation of Phosphorus Solubilizing Bacteria Improved Productivity, Grain Quality and Net Economic Returns of Pearl Millet. Agronomy 2022, 12, 2412. [Google Scholar] [CrossRef]
- Kuzina, E.V.; Rafikova, G.F.; Korshunova, T.Y. Phosphate-solubilizing bacteria of the genus Pseudomonas and the effectiveness of their use to increase the availability of phosphorus. Proc. Ufa Sci. Cent. Russ. Acad. Sci. 2021, 3, 11–16. [Google Scholar] [CrossRef]
- Selvi, K.B.; Paul, J.J.A.; Vijaya, V.; Saraswathi, K. Analyzing the efficacy of phosphate solubilizing microorganisms by enrichment culture techniques. Biochem. Mol. Biol. J. 2017, 3, 1–7. [Google Scholar]
- Timofeeva, A.; Galyamova, M.; Sedykh, S. Prospects for Using Phosphate-Solubilizing Microorganisms as Natural Fertilizers in Agriculture. Plants 2022, 11, 2119. [Google Scholar] [CrossRef]
- Ai, P.; Sun, S.; Zhao, J.; Fan, X.; Xin, W.; Guo, Q.; Yu, L.; Shen, Q.; Wu, P.; Miller, A.J.; et al. Two rice phosphate transporters, OsPht1;2 and OsPht1;6, have different functions and kinetic properties in uptake and translocation. Plant J. 2009, 57, 798–809. [Google Scholar] [CrossRef]
- Li, Z.; Bai, T.; Dai, L.; Wang, F.; Tao, J.; Meng, S.; Hu, Y.; Wang, S.; Hu, S. A study of organic acid production in contrasts between two phosphate solubilizing fungi: Penicillium oxalicum and Aspergillus niger. Sci. Rep. 2016, 6, 25313. [Google Scholar] [CrossRef] [PubMed]
- Chebotar, V.K.; Makarova, N.M.; Shaposhnikov, A.I.; Kravchenko, L.V. Antifungal and phytostimulating characteristics of Bacillus subtilis Ch-13 rhizospheric strain, producer of bioprepations. Appl. Biochem. Microbiol. 2009, 45, 419–423. [Google Scholar] [CrossRef]
- Chebotar, V.K.; Petrov, V.B.; Shaposhnikov, A.I.; Kravchenko, L.V. Biochemical criteria for evaluating agronomically significant properties of bacilli used in the creation of microbiological preparations. Agric. Biol. 2011, 3, 119–122. [Google Scholar]
- Chebotar, V.K.; Chizhevskaya, E.P.; Vorobyov, N.I.; Bobkova, V.V.; Pomyaksheva, L.V.; Khomyakov, Y.V.; Konovalov, S.N. The Quality and Productivity of Strawberry (Fragaria × ananassa Duch.) Improved by the Inoculation of PGPR Bacillus velezensis BS89 in Field Experiments. Agronomy 2022, 12, 2600. [Google Scholar] [CrossRef]
- Zavalin, A.; Chebotar, V.; Alferov, A.; Chernova, L.; Shcherbakova, E.; Chizhevskaya, E. Nitrogen use by plants and nitrogen flows after application of standard and biomodified nitrogen fertilizers on barley. Biol. Commun. 2021, 66, 283–289. [Google Scholar] [CrossRef]
- Nosov, V.V.; Biryukova, O.A.; Bozhkov, D.V. Coefficients of phosphorus and potassium use from fertilizers by corn plants on ordinary chernozem of the Rostov region. Plant Nutr. 2018, Is. 3, 2–5. [Google Scholar]
- Frid, A.S.; Borisochkina, T.I. Fluorine: Migration Mobility in Technogenic Polluted Soils. Agrochemistry 2019, 3, 65–71. [Google Scholar]
- Amri, M.; Mateus, D.; Gatrouni, M.; Rjeibi, M.R.; Asses, N.; Abbes, C. Co-Inoculation with Phosphate-Solubilizing Microorganisms of Rock Phosphate and Phosphogypsum and Their Effect on Growth Promotion and Nutrient Uptake by Ryegrass. Appl. Biosci. 2022, 1, 179–197. [Google Scholar] [CrossRef]
- Mehrvarz, S.; Chaichi, M.R.; Alikhani, H.A. Effects of phosphate solubilizing microorganisms and phosphorus chemical fertilizer on yield and yield components of barely (Hordeum vulgare L.). Am.-Eurasian J. Agric. Environ. Sci. 2008, 3, 822–828. [Google Scholar]
- Chen, S.; Waghmode, T.R.; Sun, R.; Kuramae, E.E.; Hu, C.; Liu, B. Root-associated microbiomes of wheat under the combined effect of plant development and nitrogen fertilization. Microbiome 2019, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kirpichnikov, N.A.; Adrianov, S.N. Action and aftereffects of phosphorus fertilizers on soddy-podzolic heavy loamy soil at various degrees of liming. Agrochemistry 2007, Is. 10, 14–23. [Google Scholar]
- Kovár, P.; Vozár, Ľ.; Hric, P. Changes in growth-production parameters of Lolium perenne L. turf after application of concentrated polysulfide fertilizer. Acta Fytotechn Zootech. 2020, 23, 155–160. [Google Scholar] [CrossRef]
- Poulton, P.R.; Johnston, A.E. Phosphorus in agriculture: A review of results from 175 years research at Rothamsted, UK. J. Environ. Qual. 2019, 48, 1133–1144. [Google Scholar] [CrossRef]
- Myrvang, M.B.; Hillersøy, M.H.; Heim, M.; Bleken, M.A.; Gjengedal, E. Uptake of macro nutrients, barium, and strontium by vegetation from mineral soils on carbonatite and pyroxenite bedrock at the Lillebukt Alkaline Complex on Stjernøy, Northern Norway. J. Plant Nutr. Soil Sci. 2016, 179, 705–716. [Google Scholar] [CrossRef]





| Compound | Content (%) | |
|---|---|---|
| TiO2 | 0.37 | |
| Fe2O3 | 0.37 | |
| FeO | 1.89 | |
| Tr2O3 | 0.95 | |
| CaO | 0.37 | |
| SrO | 1.89 | |
| MgO | 0.95 | |
| MnO | 0.37 | |
| Na2O | 1.89 | |
| K2O | 0.95 | |
| Cd | 3.1 × 10−6 | |
| Pb | 4.1× 10−4 | |
| F | 2.99 | |
| SiO2 | total | 1.89 |
| residue | 0.95 | |
| Al2O3 | total | 1.89 |
| acid-soluble | 0.95 | |
| Fertilizer Sample | Experimental Variant | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |
| Soil analysis before the experiment | Base | Base | Base | Base | Base |
| Control without fertilizer | C-0 | C-0 | C-0 | C-0 | C-0 |
| Control N + K | C-NK | C-NK | C-NK | C-NK | C-NK |
| Apatite concentrate of standard grinding − control | V.1.1 | V.1.2 | V.1.3 | V.1.4 | V.1.5 |
| Apatite concentrate of standard grinding + strains BI2 and BS89 (6 × 106 CFU/g each) | V.2.1 | V.2.2 | V.2.3 | V.2.4 | V.2.5 |
| Apatite concentrate of standard grinding + strains BI14 and BS89 (6 × 106 CFU/g each) | V.3.1 | V.3.2 | V.3.3 | V.3.4 | V.3.5 |
| Apatite concentrate of standard grinding + strains BI2 and BI14 (6 × 106 CFU/g each) | V.4.1 | V.4.2 | V.4.3 | V.4.4 | V.4.5 |
| Apatite concentrate of standard grinding + BI2, BI14, and BS89 (4 × 106 CFU/g each) | V.5.1 | V.5.2 | V.5.3 | V.5.4 | V.5.5 |
| Apatite concentrate of fine grinding − control 2 | V.6.1 | V.6.2 | V.6.3 | V.6.4 | V.6.5 |
| Apatite concentrate of fine grinding + strains BI2 and BS89 (6 × 106 CFU/g each) | V.7.1 | V.7.2 | V.7.3 | V.7.4 | V.7.5 |
| Apatite concentrate of fine grinding + strains BI14 and BS89 (6 × 106 CFU/g each) | V.8.1 | V.8.2 | V.8.3 | V.8.4 | V.8.5 |
| Apatite concentrate of fine grinding + strains BI2 and BI14 (6 × 106 CFU/g each) | V.9.1 | V.9.2 | V.9.3 | V.9.4 | V.9.5 |
| Apatite concentrate of fine grinding + strains BI2, BI14, and BS89 (4 × 106 CFU/g each) | V.10.1 | V.10.2 | V.10.3 | V.10.4 | V.10.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timofeeva, T.A.; Chebotar, V.K.; Demidov, D.V.; Gaidukova, S.E.; Yakovleva, I.V.; Kamionskaya, A.M. Effects of Apatite Concentrate in Combination with Phosphate-Solubilizing Microorganisms on the Yield of Ryegrass Cultivar Izorskiy. Agronomy 2023, 13, 1568. https://doi.org/10.3390/agronomy13061568
Timofeeva TA, Chebotar VK, Demidov DV, Gaidukova SE, Yakovleva IV, Kamionskaya AM. Effects of Apatite Concentrate in Combination with Phosphate-Solubilizing Microorganisms on the Yield of Ryegrass Cultivar Izorskiy. Agronomy. 2023; 13(6):1568. https://doi.org/10.3390/agronomy13061568
Chicago/Turabian StyleTimofeeva, Tatiana A., Vladimir K. Chebotar, Dmitriy V. Demidov, Sofia E. Gaidukova, Irina V. Yakovleva, and Anastasia M. Kamionskaya. 2023. "Effects of Apatite Concentrate in Combination with Phosphate-Solubilizing Microorganisms on the Yield of Ryegrass Cultivar Izorskiy" Agronomy 13, no. 6: 1568. https://doi.org/10.3390/agronomy13061568
APA StyleTimofeeva, T. A., Chebotar, V. K., Demidov, D. V., Gaidukova, S. E., Yakovleva, I. V., & Kamionskaya, A. M. (2023). Effects of Apatite Concentrate in Combination with Phosphate-Solubilizing Microorganisms on the Yield of Ryegrass Cultivar Izorskiy. Agronomy, 13(6), 1568. https://doi.org/10.3390/agronomy13061568








