Insecticide Use against Desert Locust in the Horn of Africa 2019–2021 Reveals a Pressing Need for Change
Abstract
1. Introduction
- What changes in the choice of insecticides have been made as compared to previous DL campaigns? What are the differences in insecticide choice among the three countries between August 2019 and December 2021?
- Which environmental side-effects were observed, and how were these impacts assessed?
- How could the control of DL populations, dominated by chemical insecticides, be replaced by biological management using M. acridum and other nature-based solutions, in particular, predators such as locust-eating birds?
2. Materials and Methods
3. Results and Discussion
3.1. Historical Review: The 1986–1989 Plague and the 2003–2005 Upsurge
- The establishment of an independent group of scientists, the PRG, whose role was to advise the FAO on efficacy trials and the choice of insecticides having the least environmental and human health impact;
- A multi-compartment pilot study to quantitatively assess environmental side-effects of the insecticides fenitrothion, chlorpyrifos and diflubenzuron [37] and the subsequent establishment of the Locustox Project (later CERES/Locustox Foundation), which conducted research into the environmental and human health effects of locust control insecticides in West Africa [38], and;
- A consortium called LUBILOSA (Lutte Biologique contre les Locustes et Sauteriaux) was formed to develop a mycoinsecticide, which was selective towards acridids (locusts and grasshoppers) [39].
3.1.1. Protection of Environmental and Human Health and Relevant Regulations
- The Desert Locust Guideline 6: Safety and Environmental Precautions [45] (currently under revision);
- The Manual for the Implementation of Environmental, Health and Safety (EHS) Standards for the Control of Locusts [46], which was based on the Commission for Controlling the Desert Locust in the Western Region (of the DL distribution area) “Cahier des charges environnementales” but was only published after the 2019–2021 upsurge.
3.1.2. Locust Insecticides and International Registration
“Although chlorpyrifos has been phased out in the Union, it appears that it is still used as a pesticide and dispersed in the environment outside the Union. Due to the potential for long-range environmental transport of chlorpyrifos, the measures taken nationally or at Union level are not sufficient to safeguard the high level of protection of the environment and human health. Wider international action is therefore necessary”.
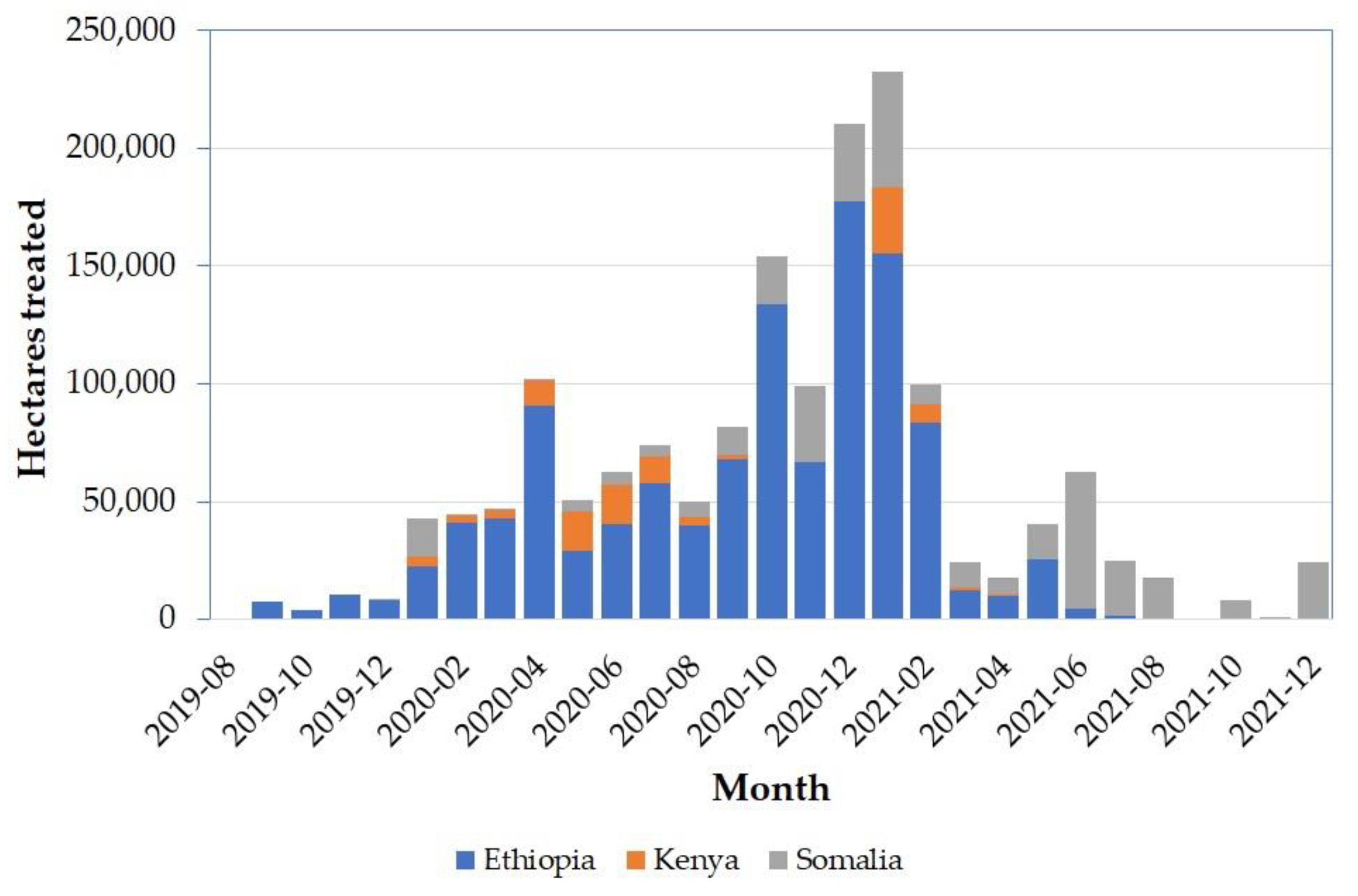
3.2. The 2019–2021 DL Campaign in Ethiopia, Kenya, and Somalia
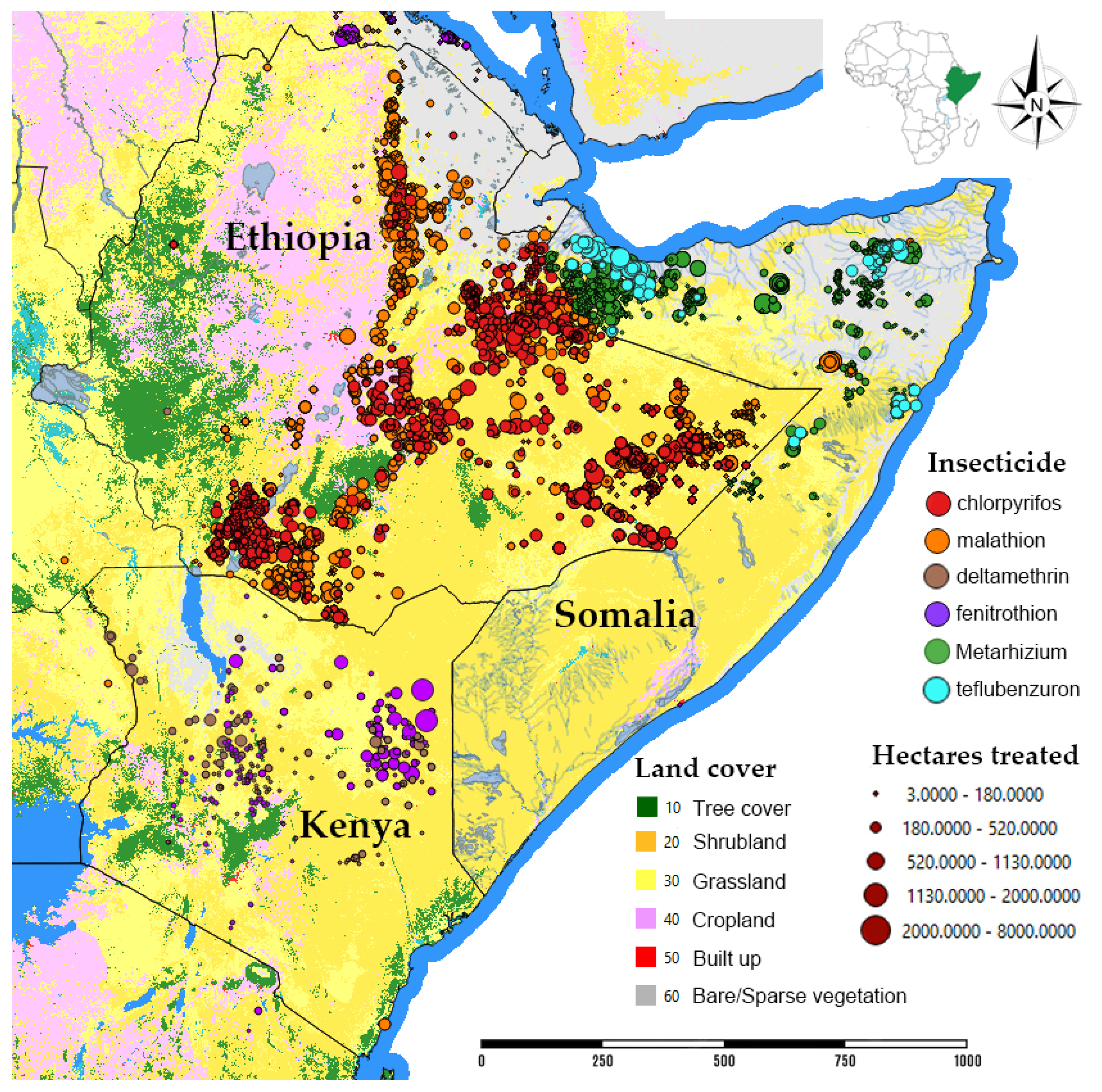
3.2.1. Environmental Monitoring in Kenya and Ethiopia
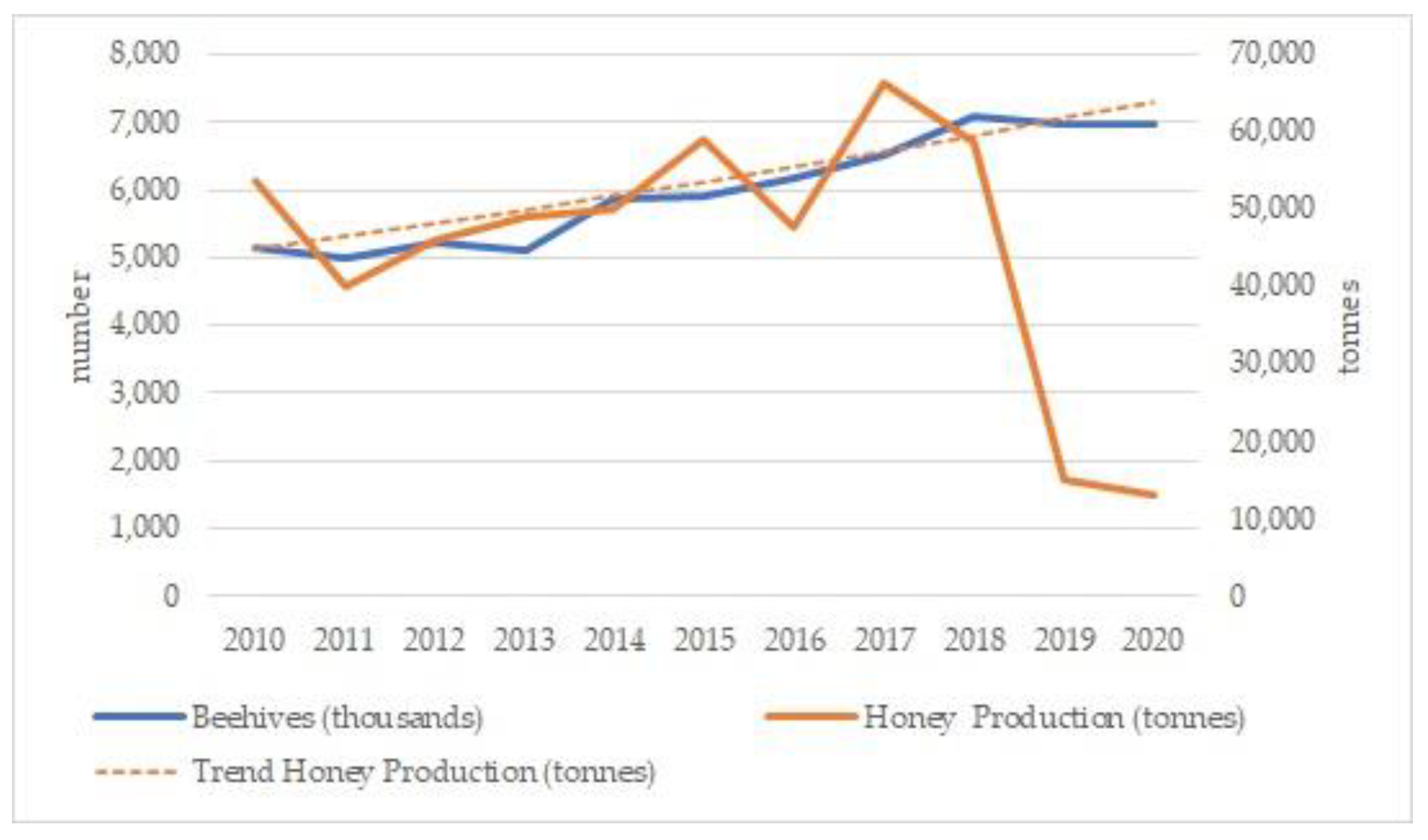
3.2.2. Pollinators and Apiculture
3.2.3. The Case for M. acridum—An Alternative Agent in Locust Control
3.3. Bird Predation and M. acridum: Towards a Novel Approach in Locust Control
4. Conclusions: The Road to Sustainable Locust Management
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

References
- Symmons, P.M.; Cressman, K. Desert Locust Guidelines 1. Biology and Behaviour; Food and Agriculture Organization of the United Nations: Rome, Italy, 2001. [Google Scholar]
- Villarreal, M. Desert Locusts: Can Mathematical Models Help to Control Them? In Imagine Math 8; Springer: Berlin/Heidelberg, Germany, 2022; pp. 405–417. [Google Scholar]
- Brader, L.; Djibo, H.; Faye, F.G.; Ghaout, S.; Lazar, M.; Luzietoso, P.N.; Ould Babah, M.A. Apporter Une Réponse Plus Efficace aux Problèmes Posés par les Criquets Pèlerins et à Leurs Conséquences sur la Sécurité Alimentaire, les Moyens d’Existence et la Pauvreté. Évaluation Multilatérale de la Campagne 2003–05 Contre le Criquet Pèlerin; Food and Agriculture Organization of the United Nations: Rome, Italy, 2006. [Google Scholar]
- FAO. Desert Locust Watch. Available online: https://www.fao.org/ag/locusts/en/info/2094/index.html (accessed on 12 December 2022).
- Showler, A.T.; Ould Babah Ebbe, M.A.; Lecoq, M.; Maeno, K.O. Early Intervention against Desert Locusts: Current Proactive Approach and the Prospect of Sustainable Outbreak Prevention. Agronomy 2021, 11, 312. [Google Scholar] [CrossRef]
- Müller, A.; Nilsson, S.; Prakash, A.; Lazutkaite, E. A Scoping Paper on the Ongoing Desert Locust Crisis 2019–2021+; TMG: Berlin, Germany, 2022. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Pörtner, H.-O., Roberts, D.C., Tignor, M., Poloczanska, E.S., Mintenbeck, K., Alegría, A., Craig, M., Langsdorf, S., Löschke, S., Möller, V., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2022; pp. 37–118. [Google Scholar]
- Roussi, A. Why gigantic locust swarms are challenging governments and researchers. Nature 2020, 579, 330–331. [Google Scholar] [CrossRef] [PubMed]
- World Bank. Factsheet. The Locust Crisis: The World Bank’s Response. Available online: https://www.worldbank.org/en/news/factsheet/2020/04/27/the-locust-crisis-the-world-banks-response (accessed on 15 February 2021).
- Stoughton, M.; Gibson, J.; Jolley, J.; Kaegi, D.; Neigh, A.; Ongámo, G.; Siegel, S.; Worku, G.; Yussuf, M. Desert Locust Surveillance and Control; USAID: Washington, DC, USA, 2020. [Google Scholar]
- FAO. Real-Time Evaluation of FAO’s Response to Desert Locust Upsurge (2020–2021)–Phase 1; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Pesticide Referee Group. Evaluation of Field Trials Data on the Efficacy and Selectivity of Insecticides on Locusts and Grasshoppers; Food and Agriculture Organization of the United Nations: Rome, Italy, 2014. [Google Scholar]
- Locust Pesticide Referee Group. Evaluation of Field Trials Data on the Efficacy and Selectivity of Insecticides on Locusts and Grasshoppers; Food and Agriculture Organization of the United Nations: Rome, Italy, 2021. [Google Scholar]
- Owuor, A.; McRae, H. The Control of the Desert Locusts (Schistocerca gregaria) in Somalia during the Upsurge Between 2019 and 2021. Outlooks Pest Manag. 2022, 33, 132–136. [Google Scholar] [CrossRef]
- Mutia, T. Environment and Health Monitoring. Final Report Desert Locust Campaign in Kenya; FAO: Rome, Italy, 2020. [Google Scholar]
- Mutia, T. Environment and Health Monitoring Report 7 October 2020–15 January 2021; FAO: Rome, Italy, 2021. [Google Scholar]
- Lemma, H. Final Report on Assessment of Environment and Health Impacts of Insecticides Applied for Desert Control; FAO/DLCO-EA Control Reserve Base: Dire Dawa, Ethiopia, 2021. [Google Scholar]
- Mullié, W.C. Don’t Kill Your Allies: The Impact of Chemical and Biological Locust and Grasshopper Control on Birds; Wageningen University and Research: Wageningen, The Netherlands, 2021. [Google Scholar]
- Mullié, W.C.; Cheke, R.A.; Young, S.; Ibrahim, A.B.; Murk, A.J. Increased and sex-selective avian predation of desert locusts Schistocerca gregaria treated with Metarhizium acridum. PLoS ONE 2021, 16, e0244733. [Google Scholar] [CrossRef] [PubMed]
- Mullié, W.C.; Guèye, Y. Does bird predation enhance the impact of Green Muscle® (Metarhizium acridum) used for grasshopper control? J. Orthoptera Res. 2010, 19, 139–155. [Google Scholar] [CrossRef]
- Mullié, W.C.; Keith, J.O. The effects of aerially applied fenitrothion and chlorpyrifos on birds in the savannah of northern Senegal. J. Appl. Ecol. 1993, 30, 536–550. [Google Scholar] [CrossRef]
- FAO. Locust Hub. Available online: https://locust-hub-hqfao.hub.arcgis.com/ (accessed on 15 December 2022).
- Cressman, K.; (Food and Agriculture Organization of the United Nations, Rome, Italy). Personal communication to Lazutkaite, E., 6 January 2022.
- Cressman, K. Desert Locust Survey and Control Data Recording and Transmission by Satellite in Real Time; Food and Agriculture Organization of the United Nations: Rome, Italy, 2014. [Google Scholar]
- McRae, H.; (Food and Agriculture Organization of the United Nations, Rome, Italy). Personal communication to Mullié, W.C., 14 July 2022.
- Pantenius, C. Mission Report Desert Locust Response in the Horn of Africa. 8th December 2019–21st January–31st August 2020; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- The Federal Democratic Republic of Ethiopia; Ministry of Agriculture. Ethiopia Emergency Locust Response Project (EELRP). Environmental and Social Management Framework (ESMF) 2020. Available online: https://documents1.worldbank.org/curated/en/202301598456031352/pdf/Environmental-and-Social-Management-Framework-ESMF-Emergency-Locust-Response-Program-P173702.pdf (accessed on 15 January 2023).
- Pest Control Products Board. Pest Control Products Registered for Use in Kenya. Available online: https://pcpb.go.ke/listofregproducts/List%20of%20Registered%20Products%20%20Version%201_2018.pdf (accessed on 15 January 2023).
- Federal Republic of Somalia. Ministry of Agriculture and Irrigation, Somalia Crisis Recovery Project (Srcp), Pest Management Plan (Pmp). Available online: https://mof.gov.so/sites/default/files/2020-08/Pest%20Management%20Plan%20%28PMP%29%20Somalia%20Crisis%20Recovery%20Project%20%28P173315%29.pdf (accessed on 14 January 2023).
- The National Office of Food Safety. Homologation des Intrants Chimiques. Index Phytosanitaire. Rabat. Available online: http://eservice.onssa.gov.ma/IndPesticide.aspx (accessed on 15 December 2022).
- ESA. ESA WorldCover 2020 Global Land Cover Product at 10 m Resolution for 2020 Based on Sentinel-1 and 2 Data. © European Union, Copernicus Land Monitoring Service 2021, European Environment Agency (EEA). Available online: https://worldcover2020.esa.int/ (accessed on 12 November 2022).
- Lecoq, M.; Cease, A. What Have We Learned after Millennia of Locust Invasions? Agronomy 2022, 12, 472. [Google Scholar] [CrossRef]
- US Congress OTA. Special Report. A Plague of Locusts; Congress of the United States Office of Technology Assessment: Washington, DC, USA, 1990. [Google Scholar]
- Dobson, H.M. Desert Locust Guidelines: Control; Food and Agriculture Organization of the United Nations: Rome, Italy, 2001. [Google Scholar]
- Fitzgerald, L.; Wikoff, D. Persistent organic pollutants. In Encyclopedia of Toxicology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Bennett, L. Review of estimates of the effectiveness of certain control techniques and inecticides against the desert locust. Anti-Locust Bull. 1972, 50, 1–15. [Google Scholar]
- Everts, J.W. Environmental Effects of Chemical Locust and Grasshopper Control. A Pilot Study; Food and Agriculture Organisation of the United Nations: Rome, Italy, 1990. [Google Scholar]
- Everts, J.W.; Mbaye, D.; Barry, O.; Mullié, W. Environmental Side-Effects of Locust and Grasshopper Control; Food and Agriculture Organization of the United Nations: Rome, Italy, 1997; p. 207. [Google Scholar]
- Prior, C.; Lomer, C.; Herren, H.; Paraiso, A.; Kooyman, C.; Smit, J. The IIBC/IITA/DFPV collaborative research programme on the biological control of locusts and grasshoppers. In Proceedings of the Biological Control of Locusts and Grasshoppers: Proceedings of a Workshop Held at the International Institute of Tropical Agriculture, Cotonou, Benin, 29 April–1 May 1991; pp. 8–18. [Google Scholar]
- Pigeon, O.; Ammati, M.; Davis, M.; Diallo, A.; Bernes, A.; De Ryckel, B.; Lecocq, V. Quality control of the remaining stocks of pesticides formulations after the 2003–2005 Desert Locust upsurge in Northern and Western Africa. In Proceedings of the CIPAC Symposium, Ljubljana, Slovenia, 8 June 2010. [Google Scholar]
- Mullié, W.C.; Van de Klashorst, G.; Everts, J.W. Post-campaign assessment of desert locust pesticide pollution at loading sites in Senegal and Mauritania . In The Newsletter of the Orthopterists’ Society. Metaleptea. Special Issue: 2007 African Symposium on Orthoptera, A Symposium on Acridology, Proceedings of the 17th Conference of the African Association of Insect Scientists, Dakar, Senegal, 11–15 June 2007; Université Cheikh Anta Diop de Dakar: Dakar, Senegal, 2007; pp. 16–17. [Google Scholar]
- Carson, R. Silent Spring; Mariner Books: Boston, NY, USA, 1962. [Google Scholar]
- Van der Valk, H.C.; Niassy, A.; Bèye, A.B. Does grasshopper control create grasshopper problems?-Monitoring side-effects of fenitrothion applications in the western Sahel. Crop Prot. 1999, 18, 139–149. [Google Scholar] [CrossRef]
- Lockwood, J.A.; Kemp, W.P.; Onsager, J.A. Long-term, large-scale effects of insecticidal control on rangeland grasshopper populations (Orthoptera: Acrididae). J. Econ. Entomol. 1988, 81, 1258–1264. [Google Scholar] [CrossRef]
- Van der Valk, H.; Everts, J.W. Desert Locust Guidelines 6. Safety and Environmental Precautions; Food and Agriculture Organization of the United Nations: Rome, Italy, 2003. [Google Scholar] [CrossRef]
- Ould Ely, S.; Everts, J.W. Manual for the Implementation of Environmental, Health, and Safety Standards for the Control of Locusts-December 2021; FAO: Rome, Italy, 2022. [Google Scholar]
- McRae, H. Somalia success story. Use of an IGR and the biopesticide Metarhizium in desert locust upsurge control. In Proceedings of the Commission for Controlling the Desert Locust in the Central Region, Jeddah, Saudi Arabia, 5–9 June 2022. [Google Scholar]
- World Health Organization. The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2019. Available online: https://apps.who.int/iris/rest/bitstreams/1278712/retrieve (accessed on 15 January 2023).
- Environmental Protection Agency. Chlorpyrifos; End-Use Products Cancellation Order. Available online: https://www.federalregister.gov/documents/2001/09/12/01-22756/chlorpyrifos-end-use-products-cancellation-order (accessed on 17 December 2022).
- International Agency for Research on Cancer. IARC Monographs Volume 112: Evaluation of Five Organophosphate Insecticides and Herbicides; World Health Organization: Lyon, France, 2015. [Google Scholar]
- Pesticide Action Network International. PAN International List of Highly Hazardous Pesticides; PAN International: Hamburg, Germany, 2021. [Google Scholar]
- Environmental Protection Agency. Fenitrothion Facts. Prevention, Pesticides and Toxic Substances (7508C); EPA: Washington, DC, USA, 2000. [Google Scholar]
- European Food Safety Authority. Conclusion regarding the peer review of the pesticide risk assessment of the active substance fenitrothion. EFSA J. 2006, 4, 59r. [Google Scholar] [CrossRef]
- Peveling, R. Environmental conservation and locust control—Possible conflicts and solutions. J. Orthoptera Res. 2001, 10, 171–187. [Google Scholar] [CrossRef]
- Mamadou, A. An Assessment of the Impact of Chemical Insecticides Used to Control Desert Locust (Schistocerca gregaria)(Orthoptera, Acrididae) on Lacertid Lizard Acanthodactylus spp. (Sauria, Lacertidae) in Niger. Am. Chem. Sci. J. 2015, 7, 193–200. [Google Scholar]
- Sanchez-Bayo, F.; Goka, K. Pesticide residues and bees—A risk assessment. PLoS ONE 2014, 9, e94482. [Google Scholar] [CrossRef]
- European Food Safety Authority. Statement on the available outcomes of the human health assessment in the context of the pesticides peer review of the active substance chlorpyrifos. EFSA J. 2019, 17, e05809. [Google Scholar]
- Gardner, S. EU to Ban Chlorpyrifos Pesticide Starting in February. Available online: https://news.bloomberglaw.com/environment-and-energy/eu-to-ban-chlorpyrifos-pesticide-starting-in-february (accessed on 2 December 2020).
- ECHA. Chlorpyrifos Draft Risk Profile. Available online: https://echa.europa.eu/documents/10162/8a51d7d9-e9a4-2513-e975-492fb70f825c (accessed on 6 May 2022).
- FAO. FAO Response Overview Dashboard. Available online: http://www.fao.org/locusts/response-overview-dashboard/en/ (accessed on 7 March 2021).
- Nasike, C. Toxic Pesticides Employed in the Control of Desert Locusts. Available online: https://www.greenpeace.org/africa/en/blogs/13147/toxic-pesticides-employed-in-the-control-of-desert-locusts/ (accessed on 19 February 2021).
- Worku, Z.; Bihonegn, A.; Begna, D.; Abasimel, S.A.; Wode, A. The Indirect Threats of Desert Locust Infestation on Honeybees in Ethiopia. Adv. Agric. 2022, 2022, 1–7. [Google Scholar] [CrossRef]
- Leach, A.; Mullié, W.; Mumford, J.; Waibel, H. Spatial and Historical Analysis of Pesticide Externalities in Locust Control in Senegal-First Steps; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008. [Google Scholar] [CrossRef]
- Mogaka, D.M.; Muya, S.; Ndwigah, F. Diversity, Abundance, Richness, and Birds of Conservation Interest in Nyando Sugar Belt, Muhoroni Sub-County, Lake Victoria Basin, Western Kenya. Open J. Anim. Sci. 2019, 9, 268–285. [Google Scholar] [CrossRef]
- Fry, C.H.; Keith, S. The Birds of Africa: Volume VI; Bloomsbury Publishing: London, UK, 2020. [Google Scholar]
- UNEP-FAO-CRC. Carbosulfan. Annex to decision CRC-11/4. Rationale for the conclusion by the Chemical Review Committee that the notifications of final regulatory action submitted by the European Union, Burkina Faso, Cabo Verde, Chad, the Gambia, Mauritania, Niger, Senegal and Togo in respect of carbosulfan meet the criteria of Annex II to the Rotterdam Convention. In Proceedings of the Chemical Review Committee Eleventh Meeting, Rome, Italy, 26–28 October 2015. [Google Scholar]
- Mineau, P.; Story, P.; Haroon, W.; Abdalla, A.M.; Lecoq, M.; Luong-Skovmand, M.H. Post-spray pesticide residues in Orthopterans and the risk assessment process for gorgefeeding birds and mammals. In Proceedings of the 10th International Congress of Orthopterology, Antalya, Turkey, 21–25 June 2009. [Google Scholar]
- Mineau, P.; Baril, A.; Collins, B.T.; Duffe, J.; Joerman, G.; Luttik, R. Pesticide acute toxicity reference values for birds. Rev. Env. Contam. Toxicol. 2001, 170, 13–74. [Google Scholar]
- Eardley, C.D.; Gikungu, M.; Schwarz, M.P. Bee conservation in Sub-Saharan Africa and Madagascar: Diversity, status and threats. Apidologie 2009, 40, 355–366. [Google Scholar] [CrossRef]
- Nasike, C.; Mwendwa, C. Bees in Crisis: Toxic Insecticides a Threat to Food Security. Available online: https://www.standardmedia.co.ke/farmkenya/article/2001426542/bees-in-crisis-toxic-insecticides-a-threat-to-food-security (accessed on 19 October 2021).
- Kahungu, E.; Mwongera, V. Implications of Mass Spraying of Pesticides on Pollinators and Food Production While Containing Locusts Invasion in Kenya. Available online: https://africasciencenews.org/environment/15011/ (accessed on 21 May 2022).
- FAO. FAOSTAT. Crops and Livestock Products. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 15 December 2022).
- Gratzer, K.; Wakjira, K.; Fiedler, S.; Brodschneider, R. Challenges and perspectives for beekeeping in Ethiopia. A review. Agron. Sustain. Dev. 2021, 41, 1–15. [Google Scholar] [CrossRef]
- Muhammed, S. Honey bee production common disease and pests in Ethiopia—A review. Int. J. Adv. Res. Biol. Sci. 2021, 8, 47–55. [Google Scholar] [CrossRef]
- Gebremedhn, H.; Amssalu, B.; Smet, L.D.; de Graaf, D.C. Factors restraining the population growth of Varroa destructor in Ethiopian honey bees (Apis mellifera simensis). PLoS ONE 2019, 14, e0223236. [Google Scholar] [CrossRef] [PubMed]
- Roubik, D.W. Ecology and Natural History of Tropical Bees; Cambridge University Press: Cambridge, UK, 1992. [Google Scholar]
- Al-Dobai, S.; (Food and Agriculture Organization of the United Nations, Rome, Italy). Personal communication to Lazutkaite, E., 13 September 2022.
- Hunter, D.M. Credibility of an IPM approach for locust and grasshopper control: The Australian example. J. Orthoptera Res. 2010, 19, 133–137. [Google Scholar] [CrossRef]
- Story, P.G.; Walker, P.W.; McRae, H.; Hamilton, J.G. A case study of the Australian Plague Locust Commission and environmental due diligence: Why mere legislative compliance is no longer sufficient for environmentally responsible locust control in Australia. Integr. Environ. Assess. Manag. Int. J. 2005, 1, 245–251. [Google Scholar] [CrossRef]
- Zhang, L.; Hunter, D.M. Management of locusts and grasshoppers in China. J. Orthoptera Res. 2017, 26, 155–159. [Google Scholar] [CrossRef]
- FAO. Red Locust Disaster in Eastern Africa Prevented. Biopesticides Being Used on a Large Scale. Available online: https://www.fao.org/news/story/en/item/21084/icode/ (accessed on 15 January 2022).
- OEC. Sheep and Goats in Somalia. Available online: https://oec.world/en/profile/bilateral-product/sheep-and-goats/reporter/som#:~:text=About&text=Exports%20In%202020%2C%20Somalia%20exported,most%20exported%20product%20in%20Somalia (accessed on 12 December 2022).
- Greathead, D.J. A Brief Survey of the Effects of Biotic Factors on Populations of the Desert Locust. J. Appl. Ecol. 1966, 3, 239–250. [Google Scholar] [CrossRef]
- Stower, W.J.; Greathead, D.J. Numerical Changes in a Population of the Desert Locust, with Special Reference to Factors Responsible for Mortality. J. Appl. Ecol. 1969, 6, 203–235. [Google Scholar] [CrossRef]
- Smith, K. On birds attacking desert locust swarms in Eritrea. Entomologist 1953, 86, 3–7. [Google Scholar]
- Ashall, C.; Ellis, P.E. Studies on numbers and mortality in field populations of the desert locust (Schistocerca gregaria Forskål). Anti-Locst Bul 1962, 38, 1–59. [Google Scholar]
- Elliot, S.L.; Blanford, S.; Thomas, M.B. Host–pathogen interactions in a varying environment: Temperature, behavioural fever and fitness. Proc. R. Soc. London. Ser. B Biol. Sci. 2002, 269, 1599–1607. [Google Scholar] [CrossRef]
- Wilps, H. Ecology of Schistocerca gregaria (Forskål): Observations in West Africa from 1990 to 1994. In New Strategies in Locust Control; Springer: Berlin/Heidelberg, Germany, 1997; pp. 9–17. [Google Scholar]
- Culmsee, H. The habitat functions of vegetation in relation to the behavior of the desert locust Schistocerca gregaria (Forskål) (Acrididae: Orthoptera): A study in Mauritania (West Africa). Phytocoenologia 2002, 32, 645–664. [Google Scholar] [CrossRef]
- Mullié, W.C. Birds, locusts and grasshoppers. In Living on the Edge; KNNV Publishing: Zeist, The Netherlands, 2009; pp. 202–223. [Google Scholar]
- Kooyman, C.; Godonou, I. Infection of Schistocerca gregaria (Orthoptera: Acrididae) hoppers by Metarhizium flavoviride (Deuteromycotina: Hyphomycetes) conidia in an oil formulation applied under desert conditions. Bull. Entomol. Res. 1997, 87, 105–107. [Google Scholar] [CrossRef]
- Kooyman, C.; Aammati, M.; Moumene, K.; Chaouch, A.; Zeyd, A. Essai de Green Muscle sur les nymphes du criquet pèlerin dans la Wilaya d’El Oued Nord Est Algérie. Rome Food Agric. Organ. United Nations TAC 2005, 715, 2005. [Google Scholar]
- Blanford, S.; Thomas, M.B.; Langewald, J. Behavioural fever in the Senegalese grasshopper, Oedaleus senegalensis, and its implications for biological control using pathogens. Ecol. Entomol. 1998, 23, 9–14. [Google Scholar] [CrossRef]
- Clancy, L.M.; Jones, R.; Cooper, A.L.; Griffith, G.W.; Santer, R.D. Dose-dependent behavioural fever responses in desert locusts challenged with the entomopathogenic fungus Metarhizium acridum. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Dean, G.J.W. Stork and egret as predators of the Red Locust in the Rukwa Valley outbreak area. Ostrich 1964, 35, 95–100. [Google Scholar] [CrossRef]
- Amatobi, C.; Apeji, S.; Oyidi, O. Effect of some insectivorous birds on populations of grasshoppers (Orthoptera) in Kano State, Nigeria. Samaru J. Agric. Res. 1987, 5, 43–50. [Google Scholar]
- Mineau, P. Estimating the probability of bird mortality from pesticide sprays on the basis of the field study record. Environ. Toxicol. Chem. Int. J. 2002, 21, 1497–1506. [Google Scholar] [CrossRef]
- Van der Valk, H. Environmental Impact of Dieldrin Applications in Locust Control—A Hazard Assessment; FAO: Rome, Italy, 1988. [Google Scholar]
- Cressman, K. Desert locust. In Biological and Environmental Hazards, Risks, and Disasters; Shroder, J.F., Sivanpillai, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 87–105. [Google Scholar]
- FAO. Desert Locust Upsurge–Progress Report on the Response in the Greater Horn of Africa and Yemen (September–December 2020); Food and Agriculture Organization of the United Nations: Rome, Italy, 2021. [Google Scholar]
- Word Bank. The World Bank in Ethiopia. Available online: https://www.worldbank.org/en/country/ethiopia/overview#:~:text=With%20about%20117%20million%20people,gross%20national%20income%20of%20%24960 (accessed on 15 February 2023).
- Mullié, W.C.; Gueye, Y. Efficacite du Green Muscle (Metarhizium anisopliae var. acridum) en Dose Reduite en Lutte Antiacridienne au Senegal en 2008 et Son Impact sur la Faune Non–Cible et sur la Predation par les Oiseaux; Raport Phase 1. Ministere de l’Agriculture: Dakar, Senegal, 2009. [Google Scholar]
- Mullié, W.C.; Gueye, Y. L’impact du Green Muscle® Metarhizium acridum sur le Peuplement Acridien et ses Predateurs à Khelcom, Sénégal, après deux Traitemments Consécutifs sur Deux Ans; Ministère de l’Agriculture, des Biocarburants et de la Pisciculture: Dakar, Senegal, 2010. [Google Scholar]

| Insecticide | Priority | Dose Rate (g a.i./ha) 1 | Speed of ACTION | Primary Mode of Action | ||
|---|---|---|---|---|---|---|
| Blanket | Intra-Barrier | Overall | ||||
| M. acridum | 1 | 50 | - | - | Slow | Mycosis |
| triflumuron | 2 | 25 | 75.6 | 10.7 | Slow | Chitin synthesis inhibition |
| teflubenzuron | 2 | 30 | n.d. | n.d. 2 | Slow | Chitin synthesis inhibition |
| diflubenzuron | 2 | 30 | 100 | 14.3 | Slow | Chitin synthesis inhibition |
| lambda-cyhalothrin | 3 | 20 | - | - | Fast | Na channel blocking |
| deltametrhrin | 3 | 12.5 or 17.5 | - | - | Fast | Na channel blocking |
| malathion | 3 | 925 | - | - | Medium | AchE inhibition |
| fipronil | 3 | - | 4.2 | 0.6 | Medium | GABA receptor blocking |
| fenitrothion | 3 | 400 | - | - | Medium | AchE inhibition |
| chlorpyrifos | 3 | 240 | - | - | Medium | AchE inhibition |
| Insecticide | Ethiopia | Kenya | Somalia | Total | % |
|---|---|---|---|---|---|
| chlorpyrifos | 440,610 | 440,610 | 27.48 | ||
| malathion | 679,380 | 2050 | 15,732 | 697,162 | 43.49 |
| fenitrothion | 35 | 32,136 | 32,171 | 2.01 | |
| deltamethrin | 72,048 | 72,048 | 4.49 | ||
| M. acridum | 102 | * | 252,689 | 252,791 | 15.77 |
| teflubenzuron | 68,761 | 68,761 | 4.29 | ||
| triflumuron | 400 | 400 | 0.02 | ||
| other | 14,285 | 3187 | 21,705 | 39,177 | 2.44 |
| Total | 1,134,412 | 109,421 | 359,287 | 1,603,120 | 100 |
| Issues | Chemical Insecticides Assessed for Efficacy and Side-Effects by PRG (See [10,11] for Details) | M. acridum (Trade Names Green Muscle® and Novacrid®) |
|---|---|---|
| Legislation and use | Reviewed for efficacy and side-effects by LPRG [10,11] (2014, 2012), widespread registration and use by government agencies to control acridids. | Until 2020, hardly used for locust control (except for in Tanzania and Madagascar) and minor use for grasshopper control in the Sahel despite proven efficacy. Since November 2019, Novacrid® has been registered in nine Sahelian countries (CILSS). Provisional sales authorisations obtained in 2020 in Kenya, Ethiopia and Somalia. |
| Speed of action/meteorological dependency | Rapid knockdown and fatality (hours) post-treatment leaving massive quantities of dead and decaying arthropods strengthening confidence of users. Specific windows for wind speeds (drift), rain and temperature (convection) during and post-spray. Efficacy slightly dependent of ambient temperature. | No knock-down, mortality 80–90% in 2–3 weeks post-treatment, undermining confidence of users. Predators and scavengers remove dead and debilitated insects as they become available. Rains during or post-spray have either no impact or may enhance efficacy, ambient daily temperatures need to be sufficiently long between (20)25 and 35(40) °C for optimal efficacy. |
| Persistence | Rapid degradation (days) on vegetation under field conditions. IGRs and fipronil persist longer and are recommended for barrier treatments only. Chlorpyrifos degrades slowly in soil. | Viable spores persist from seven days to two months providing medium-term effect against re-invasion. Subsequent bird predation may keep populations below economic threshold until next rainy season. Other studies found some carry-over effect of viable spores into the next year. Efficacy when used as barrier treatment inconclusive. |
| Stability, use and storage | Formulations well developed with known shelf-lives, storage, transport and use without problems. Some formulations are corrosive for ULV spray equipment or carriers (planes, vehicles). Training of applicators is a prerequisite. | Much shorter development history of formulations. Formulated product may settle on bottom of containers, clogging Micronair® sprayers leading to frequent cleaning/loss of time, some complaints about short shelf-life of formulated product. Dry spores have known shelf-life and can be stored and formulated in situ. Training of applicators is a prerequisite.Does not require other equipment than already in use to apply chemical insecticides. |
| Non-targets | Non-selective. Most kill non-target arthropods including natural enemies, some also birds, reptiles and other vertebrates and/or deprive them of their arthropod prey. Migration of birds from sprayed plots. Fipronil and neonicotinoids are extremely toxic for social insects. | Selective. No negative impact on non-target species, including honeybees, except on other Orthoptera. Numbers of acridivorous birds remain stable or increase post-spray (immigration). Significant increase of Oedaleus senegalensis eggpod parasitation by diptera (Bombyliidae; cf. Systoechus sp.) found at 18.75 and 37.5 g viable spores/ha. |
| Use on locust hopper bands | During large outbreaks and plagues most products (except IGRs and fipronil) are not efficient because of lack of residual action. Therefore, new hatchlings need new treatments, especially as natural enemies may have been reduced by earlier spray. | Very effective and efficient on hoppers, remains infectious during weeks to even months. This way, newly hatched hoppers will be attacked within the infectivity period of M. acridum and by increased natural predation, e.g., by birds. |
| Use on adult locusts and grasshoppers | Widely used for control of acridids under all conditions. Indiscriminate use of chemical insecticides can cause upsurges later in the season or the next year because invertebrate locust egg predators and parasitoids have been killed. | Proven efficacy against adults, can be used in ecologically sensitive areas. First time successful use against (highly mobile) large groups/swarms of adults in Somalia during 2020–2021. |
| Insecticide costs and externalities | Relatively cheap (10–30 USD/L) when supplied through FAO but hidden direct and indirect costs from side-effects (externalities such as reduced pollination, soil contamination or human intoxications not perceived as costs of treatments and hence unmet by society). | Product was available during campaign for FAO at c. 315 USD/kg (15.75 USD/L) excl. diesel fuel as carrier, with no externalities. Therefore “true costs” of biological insecticides are potentially less than economic costs per hectare sprayed of chemical insecticides, without the negative side-effects of the latter. |
| Human toxicity | Slightly to moderately hazardous, sublethal effects and casualties have been reported during campaigns. Should not be sprayed on crops or in the vicinity of human settlements, withholding times apply, killed acridids should not be eaten. Use of PPE compulsory. | Unlikely to present hazard under normal use. Can be sprayed on crops. No withholding time. Use of PPE less stringent, although recommended. |
| Livestock, fisheries and (organic) farming | Slightly to moderately hazardous. Livestock should be removed prior to spraying. Not compatible with organic farming and fisheries. Most products not registered for use on crops. Withholding times and re-entry intervals apply. | Unlikely to present hazard under normal use. No particular safety measures required, although direct spray on livestock should be avoided. Fully compatible with organic farming and fisheries, no withholding times or re-entry intervals. |
| Protected and sensitive areas | Should not be used in protected areas and buffer zones for environmentally sensitive areas, such as wetlands, which limits its use. | Safe to be used in protected areas and near environmentally sensitive areas such as wetlands. No negative impacts of spraying near water bodies known. |
| COUNTRY | STAGE | POPULATION (MILLIONS) | EATEN BY BIRDS (%) | SPECIES | OBSERVATION DAYS | SOURCE |
|---|---|---|---|---|---|---|
| ERITREA | adult | tens | 4 daily | Aquila sp., Ciconia ciconia, F. biarmicus, Leptoptilos crumenifer, Milvus sp. | few | Smith 1953 [85] |
| ERITREA | 1st–5th | 15.2 | 52.6 | Milvus sp., Ciconia abdimii, Motacilla sp. | 14 | Ashall and Ellis 1962 [86] |
| E. AFRICA | adult | up to 5000 | 0.25–6 | all birds | outbreak | Elliott 1962 [87] |
| ERITREA | 2nd–adult | 4 | Oenanthe sp., “kestrels”, | 23 | Greathead 1966 [83] | |
| MAURITANIA | 2nd–4th | 0.13–0.5 | 97.5–99.5 | Cursorius cursor, Passer luteus, P. simplex, Lanius sp. | 4–11 | Wilps 1997 [88] |
| MAURITANIA | 2nd–4th | 1.1 | 95 | Cursorius cursor, Passer luteus, P. simplex, Lanius sp. | 4–11 | Wilps 1997 [88] |
| MAURITANIA | hoppers | 0.02 | >75 | Cursorius cursor, Passer luteus, P. simplex | - | Culmsee 2002 [89] |
| SUDAN | 2nd | 12 | 3 daily, 30–50 in total | Motacilla flava | 21 | Mullié 2009 [90] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mullié, W.C.; Prakash, A.; Müller, A.; Lazutkaite, E. Insecticide Use against Desert Locust in the Horn of Africa 2019–2021 Reveals a Pressing Need for Change. Agronomy 2023, 13, 819. https://doi.org/10.3390/agronomy13030819
Mullié WC, Prakash A, Müller A, Lazutkaite E. Insecticide Use against Desert Locust in the Horn of Africa 2019–2021 Reveals a Pressing Need for Change. Agronomy. 2023; 13(3):819. https://doi.org/10.3390/agronomy13030819
Chicago/Turabian StyleMullié, Wim C., Adam Prakash, Alexander Müller, and Elena Lazutkaite. 2023. "Insecticide Use against Desert Locust in the Horn of Africa 2019–2021 Reveals a Pressing Need for Change" Agronomy 13, no. 3: 819. https://doi.org/10.3390/agronomy13030819
APA StyleMullié, W. C., Prakash, A., Müller, A., & Lazutkaite, E. (2023). Insecticide Use against Desert Locust in the Horn of Africa 2019–2021 Reveals a Pressing Need for Change. Agronomy, 13(3), 819. https://doi.org/10.3390/agronomy13030819






