Effects of Different Organic Amendments on Soil Improvement, Bacterial Composition, and Functional Diversity in Saline–Sodic Soil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Site
2.2. Experimental Design
2.3. Soil Sampling and Laboratory Analysis
2.4. Bulk Soil DNA Extraction, PCR Amplification, and Sequencing
2.5. Statistical Analysis
3. Results
3.1. Physical and Chemical Properties
3.2. Yield and Yield Components
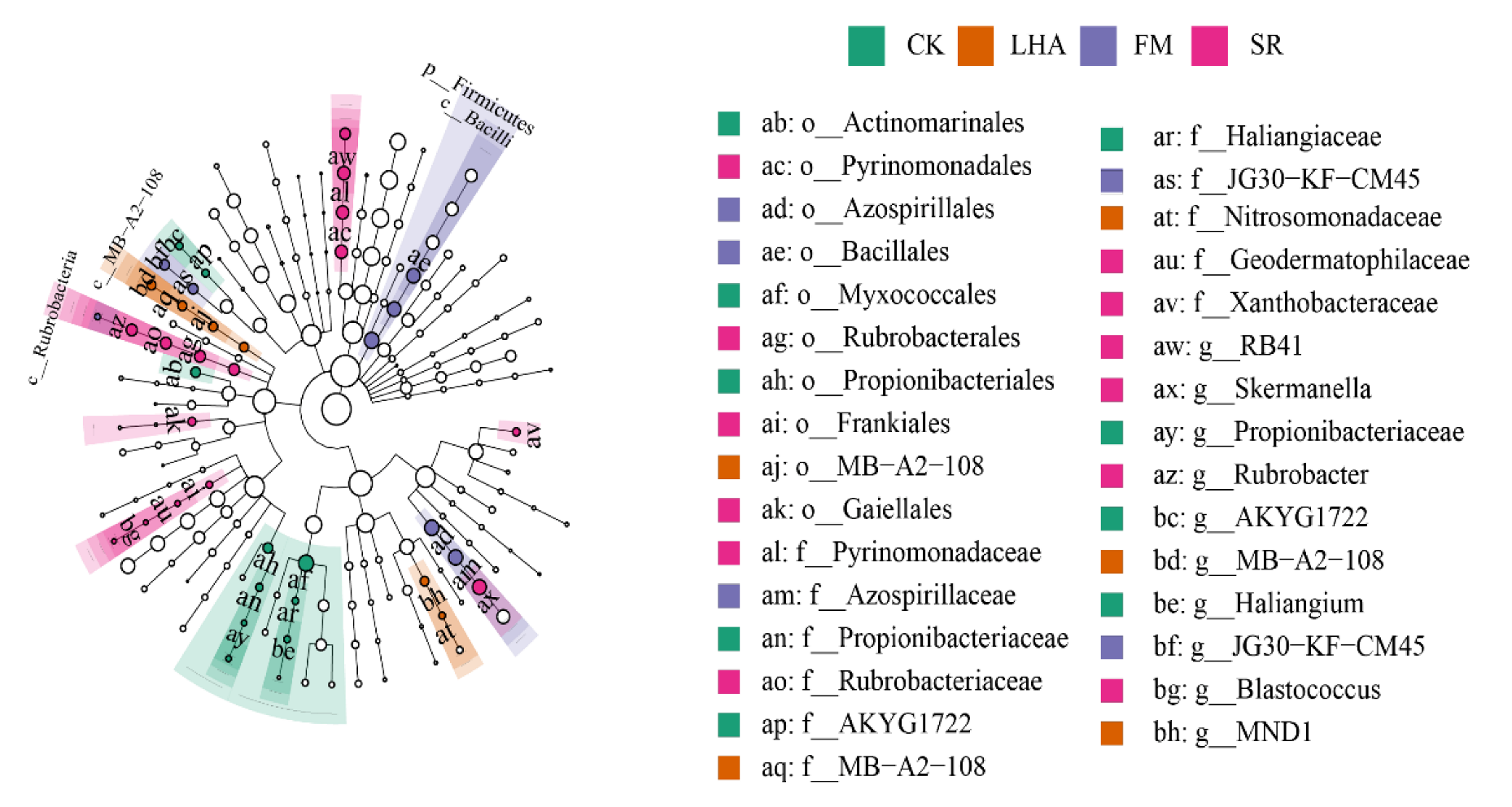
3.3. Microbial Composition: Basic Composition and Specific Species
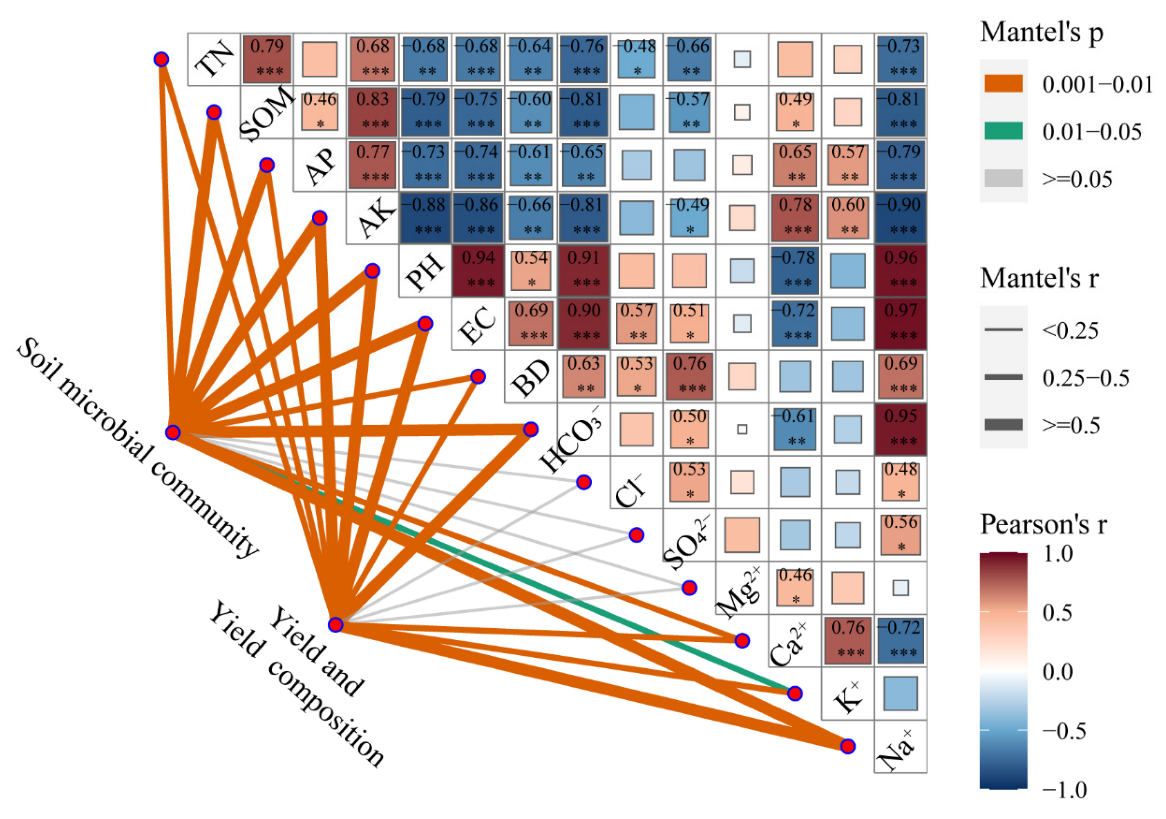
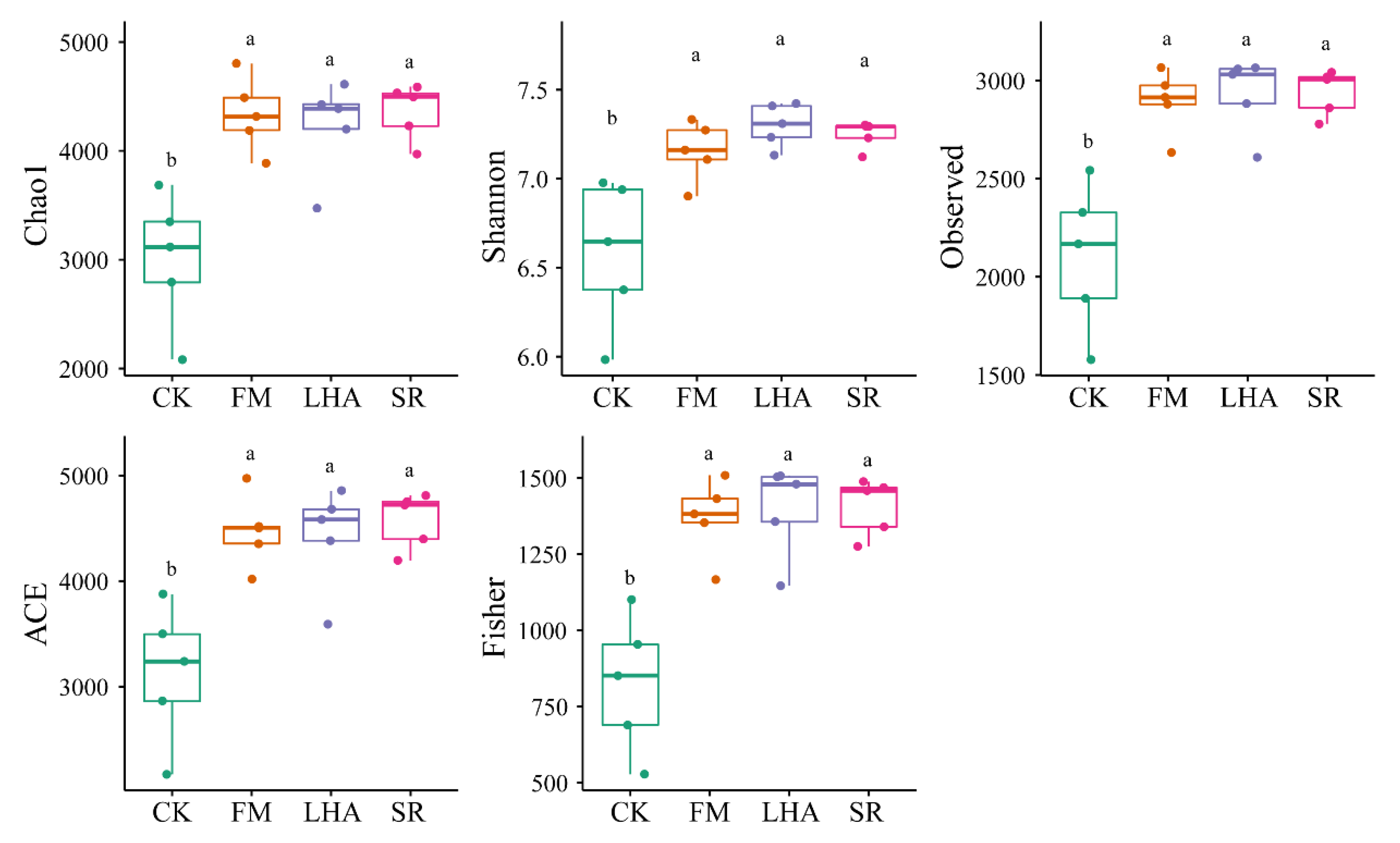
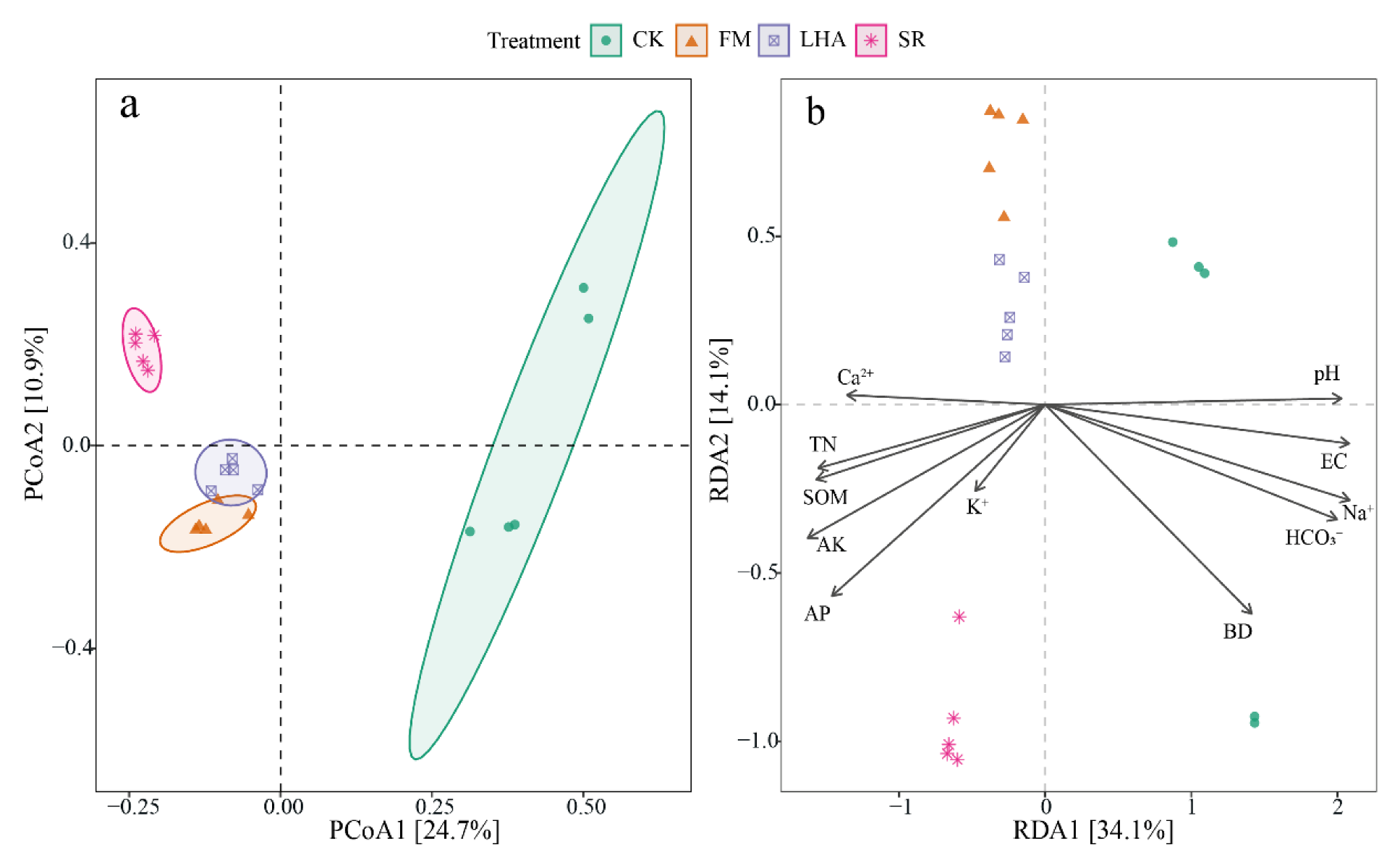
3.4. Soil Bacterial Diversity and Its Influencing Factors
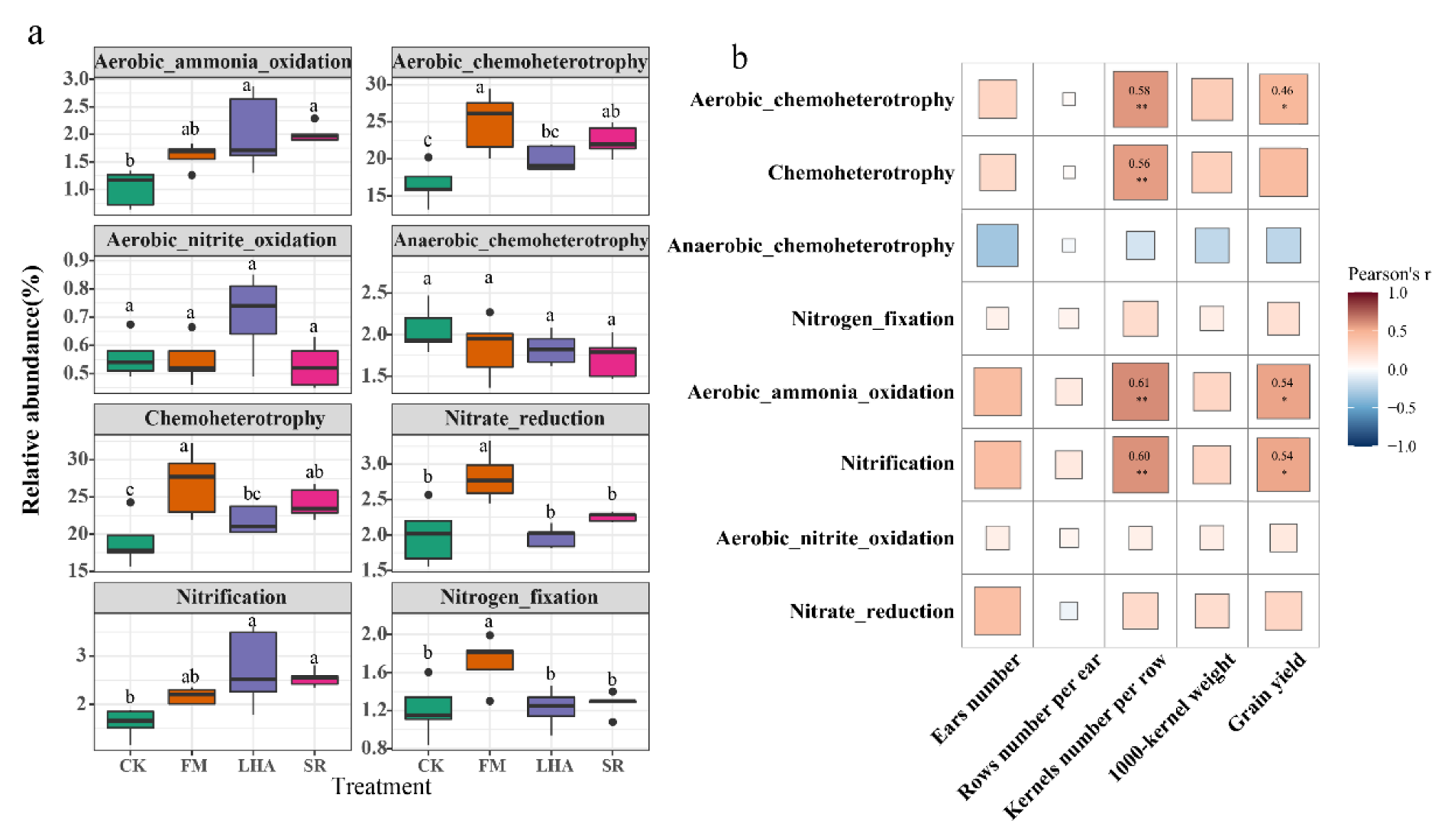
3.5. Relationship between Functional Characteristics of Soil Bacteria and Yield
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Haj-Amor, Z.; Araya, T.; Kim, D.-G.; Bouri, S.; Lee, J.; Ghiloufi, W.; Yang, Y.; Kang, H.; Jhariya, M.K.; Banerjee, A.; et al. Soil salinity and its associated effects on soil microorganisms, greenhouse gas emissions, crop yield, biodiversity and desertification: A review. Sci. Total Environ. 2022, 843, 156946. [Google Scholar] [CrossRef] [PubMed]
- Horn, D.J.V.; Okie, J.G.; Buelow, H.N.; Gooseff, M.N.; Barrett, J.E.; Takacs-Vesbach, C.D.; Kostka, J.E. Soil Microbial Responses to Increased Moisture and Organic Resources along a Salinity Gradient in a Polar Desert. Appl. Environ. Microbiol. 2014, 80, 3034–3043. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Liu, Y.; Ferreira, J.F.S.; Wang, M.; Na, J.; Huang, J.; Liang, Z. Long-term combined effects of tillage and rice cultivation with phosphogypsum or farmyard manure on the concentration of salts, minerals, and heavy metals of saline-sodic paddy fields in Northeast China. Soil Tillage Res. 2022, 215, 105222. [Google Scholar] [CrossRef]
- Zhu, H.; Yang, J.; Yao, R.; Wang, X.; Xie, W.; Zhu, W.; Liu, X.; Cao, Y.; Tao, J. Interactive effects of soil amendments (biochar and gypsum) and salinity on ammonia volatilization in coastal saline soil. CATENA 2020, 190, 104527. [Google Scholar] [CrossRef]
- Kumar, C.; Ramawat, N.; Verma, A.K. Organic fertigation system in saline-sodic soils: A new paradigm for the restoration of soil health. Agron. J. 2022, 114, 317–330. [Google Scholar] [CrossRef]
- Wu, Y.; Li, Y.; Zheng, C.; Zhang, Y.; Sun, Z. Organic amendment application influence soil organism abundance in saline alkali soil. Eur. J. Soil Biol. 2013, 54, 32–40. [Google Scholar] [CrossRef]
- Meng, Q.; Ma, X.; Zhang, J.; Yu, Z. The long-term effects of cattle manure application to agricultural soils as a natural-based solution to combat salinization. CATENA 2019, 175, 193–202. [Google Scholar] [CrossRef]
- Medina Litardo, R.C.; García Bendezú, S.J.; Carrillo Zenteno, M.D.; Pérez-Almeida, I.B.; Parismoreno, L.L.; Lombeida García, E.D. Effect of mineral and organic amendments on rice growth and yield in saline soils. J. Saudi Soc. Agric. Sci. 2022, 21, 29–37. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, Y.; Wang, S.; Li, Y.; Liu, J.; Zhuo, Y.; Zhang, W. Combined Application of Flue Gas Desulfurization Gypsum and Straw Pellets to Ameliorate Sodicity, Nutrient Content, and Aggregate Stability of Sodic Soil. J. Soil Sci. Plant Nutr. 2021, 21, 1806–1816. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, H.; Liu, X.; Chen, Y.; Lu, Y.; Shen, M.; Dang, K.; Zhao, Y.; Dong, Y.; Li, Q.; et al. Organic fertilizer enhances rice growth in severe saline–alkali soil by increasing soil bacterial diversity. Soil Use Manag. 2022, 38, 964–977. [Google Scholar] [CrossRef]
- Gregorich, E.G.; Janzen, H.; Ellert, B.H.; Helgason, B.L.; Qian, B.; Zebarth, B.J.; Angers, D.A.; Beyaert, R.P.; Drury, C.F.; Duguid, S.D.; et al. Litter decay controlled by temperature, not soil properties, affecting future soil carbon. Glob. Chang. Biol. 2017, 23, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Horta, C.; Roboredo, M.; Carneiro, J.P.; Duarte, A.C.; Torrent, J.; Sharpley, A. Organic amendments as a source of phosphorus: Agronomic and environmental impact of different animal manures applied to an acid soil. Arch. Agron. Soil Sci. 2018, 64, 257–271. [Google Scholar] [CrossRef]
- Wood, J.D.; VanderZaag, A.C.; Wagner-Riddle, C.; Smith, E.L.; Gordon, R.J. Gas emissions from liquid dairy manure: Complete versus partial storage emptying. Nutr. Cycl. Agroecosystems 2014, 99, 95–105. [Google Scholar] [CrossRef]
- Chen, Z.; Li, Y.; Zhang, X.; Xiong, Y.; Huang, Q.; Jin, S.; Shijun, S.; Daocai, C.; Huang, G. Effects of lignite bioorganic product on sunflower growth, water and nitrogen productivity in saline-sodic farmlands at Northwest China. Agric. Water Manag. 2022, 271, 107806. [Google Scholar] [CrossRef]
- Manasa, M.R.K.; Katukuri, N.R.; Darveekaran Nair, S.S.; Haojie, Y.; Yang, Z.; Guo, R.b. Role of biochar and organic substrates in enhancing the functional characteristics and microbial community in a saline soil. J. Environ. Manag. 2020, 269, 110737. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, R.; Peukert, M.; Succurro, A.; Koprivova, A.; Kopriva, S. The role of soil microorganisms in plant mineral nutrition—current knowledge and future directions. Front. Plant Sci. 2017, 8, 1617. [Google Scholar] [CrossRef] [PubMed]
- Sahin, U.; Eroğlu, S.; Sahin, F. Microbial application with gypsum increases the saturated hydraulic conductivity of saline–sodic soils. Appl. Soil Ecol. 2011, 48, 247–250. [Google Scholar] [CrossRef]
- Tian, S.; Zhu, B.; Yin, R.; Wang, M.; Jiang, Y.; Zhang, C.; Li, D.; Chen, X.; Kardol, P.; Liu, M. Organic fertilization promotes crop productivity through changes in soil aggregation. Soil Biol. Biochem. 2022, 165, 108533. [Google Scholar] [CrossRef]
- Chang, C.; Tian, L.; Tian, Z.; McLaughlin, N.; Tian, C. Change of soil microorganism communities under saline-sodic land degradation on the Songnen Plain in northeast China#. J. Plant Nutr. Soil Sci. 2022, 185, 297–307. [Google Scholar] [CrossRef]
- Wu, L.; Zhang, S.; Ma, R.; Chen, M.; Wei, W.; Ding, X. Carbon sequestration under different organic amendments in saline-alkaline soils. CATENA 2021, 196, 104882. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, H.; Lv, J. Evaluation of the applicability of organic amendments from microbially driven carbon and nitrogen transformations. Sci. Total Environ. 2022, 817, 153020. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Zhang, X.-X.; Guo, X.; Wang, D.; Chu, H. Bacterial diversity in soils subjected to long-term chemical fertilization can be more stably maintained with the addition of livestock manure than wheat straw. Soil Biol. Biochem. 2015, 88, 9–18. [Google Scholar] [CrossRef]
- Yang, Y.; Li, G.; Min, K.; Liu, T.; Li, C.; Xu, J.; Hu, F.; Li, H. The potential role of fertilizer-derived exogenous bacteria on soil bacterial community assemblage and network formation. Chemosphere 2022, 287, 132338. [Google Scholar] [CrossRef] [PubMed]
- Chahal, S.S.; Choudhary, O.P.; Mavi, M.S. Organic amendments decomposability influences microbial activity in saline soils. Arch. Agron. Soil Sci. 2017, 63, 1875–1888. [Google Scholar] [CrossRef]
- Blake, G.R.; Hartge, K.H. Bulk Density. In Methods of Soil Analysis; Soil Science Society of America: Madison, WI, USA, 1986; pp. 363–375. [Google Scholar]
- Chi, C.-M.; Wang, Z.-C. Characterizing Salt-Affected Soils of Songnen Plain Using Saturated Paste and 1:5 Soil-to-Water Extraction Methods. Arid. Land Res. Manag. 2010, 24, 1–11. [Google Scholar] [CrossRef]
- Rayment, G.E.; Lyons, D.J. Soil Chemical Methods: Australasia; CSIRO Publishing: Clayton, Australia, 2011; Volume 3. [Google Scholar]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Caporaso, J.G. Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]
- Liu, C.; Cui, Y.; Li, X.; Yao, M. microeco: An R package for data mining in microbial community ecology. FEMS Microbiol. Ecol. 2020, 97, fiaa255. [Google Scholar] [CrossRef]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef]
- Leogrande, R.; Vitti, C. Use of organic amendments to reclaim saline and sodic soils: A review. Arid. Land Res. Manag. 2019, 33, 1–21. [Google Scholar] [CrossRef]
- Jingkuan, W.; Yingde, X.U.; Fan, D.; Xiaodan, G.A.O.; Shuangyi, L.I.; Liangjie, S.U.N.; Tingting, A.N.; Jiubo, P.E.I.; Ming, L.I.; Yang, W.; et al. Process of Plant Residue Transforming into Soil Organic Matter and Mechanism of its Stabilization: A Review. Acta Pedol. Sin. 2019, 56, 528–540. [Google Scholar]
- De Mastro, F.; Cocozza, C.; Traversa, A.; Savy, D.; Abdelrahman, H.M.; Brunetti, G. Influence of crop rotation, tillage and fertilization on chemical and spectroscopic characteristics of humic acids. PLoS ONE 2019, 14, e0219099. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahman, H.M.; Olk, D.C.; Dinnes, D.; Ventrella, D.; Miano, T.; Cocozza, C. Occurrence and abundance of carbohydrates and amino compounds in sequentially extracted labile soil organic matter fractions. J. Soils Sediments 2016, 16, 2375–2384. [Google Scholar] [CrossRef]
- Abdelrahman, H.; Cocozza, C.; Olk, D.C.; Ventrella, D.; Montemurro, F.; Miano, T. Changes in Labile Fractions of Soil Organic Matter During the Conversion to Organic Farming. J. Soil Sci. Plant Nutr. 2020, 20, 1019–1028. [Google Scholar] [CrossRef]
- Morvan, T.; Nicolardot, B. Role of organic fractions on C decomposition and N mineralization of animal wastes in soil. Biol. Fertil. Soils 2009, 45, 477–486. [Google Scholar] [CrossRef]
- Piccolo, A.; Spaccini, R.; Nieder, R.; Richter, J. Sequestration of a Biologically Labile Organic Carbon in Soils by Humified Organic Matter. Clim. Chang. 2004, 67, 329–343. [Google Scholar] [CrossRef]
- Gonçalo Filho, F.; da Silva Dias, N.; Suddarth, S.R.P.; Ferreira, J.F.S.; Anderson, R.G.; dos Santos Fernandes, C.; de Lira, R.B.; Neto, M.F.; Cosme, C.R. Reclaiming Tropical Saline-Sodic Soils with Gypsum and Cow Manure. Water 2020, 12, 57. [Google Scholar] [CrossRef]
- Song, X.; Sun, R.; Chen, W.; & Wang, M. Effects of surface straw mulching and buried straw layer on soil water content and salinity dynamics in saline soils. Can. J. Soil Sci. 2020, 100, 58–68. [Google Scholar] [CrossRef]
- Nan, J.; Chen, X.; Chen, C.; Lashari, M.S.; Deng, J.; Du, Z. Impact of flue gas desulfurization gypsum and lignite humic acid application on soil organic matter and physical properties of a saline-sodic farmland soil in Eastern China. J. Soils Sediments 2016, 16, 2175–2185. [Google Scholar] [CrossRef]
- Chaganti, V.N.; Crohn, D.M. Evaluating the relative contribution of physiochemical and biological factors in ameliorating a saline–sodic soil amended with composts and biochar and leached with reclaimed water. Geoderma 2015, 259, 45–55. [Google Scholar] [CrossRef]
- Maas, E.; Hoffman, G.; Chaba, G.; Poss, J.; Shannon, M. Salt sensitivity of corn at various growth stages. Irrig. Sci. 1983, 4, 45–57. [Google Scholar] [CrossRef]
- Fouladidorhani, M.; Shayannejad, M.; Shariatmadari, H.; Mosaddeghi, M.R.; Arthur, E. Biochar, manure, and super absorbent increased wheat yields and salt redistribution in a saline-sodic soil. Agron. J. 2020, 112, 5193–5205. [Google Scholar] [CrossRef]
- Srivastava, P.K.; Gupta, M.; Shikha; Singh, N.; Tewari, S.K. Amelioration of Sodic Soil for Wheat Cultivation Using Bioaugmented Organic Soil Amendment. Land Degrad. Dev. 2016, 27, 1245–1254. [Google Scholar] [CrossRef]
- Molina-Herrera, S.; Romanyà, J. Synergistic and antagonistic interactions among organic amendments of contrasted stability, nutrient availability and soil organic matter in the regulation of C mineralisation. Eur. J. Soil Biol. 2015, 70, 118–125. [Google Scholar] [CrossRef]
- Wu, Y.; Li, Y.; Zhang, Y.; Bi, Y.; Sun, Z. Responses of Saline Soil Properties and Cotton Growth to Different Organic Amendments. Pedosphere 2018, 28, 521–529. [Google Scholar] [CrossRef]
- Chen, X.; Wu, J.; Opoku-Kwanowaa, Y. Effects of Returning Granular Corn Straw on Soil Humus Composition and Humic Acid Structure Characteristics in Saline-Alkali Soil. Sustainability 2020, 12, 1005. [Google Scholar] [CrossRef]
- Raguraj, S.; Kasim, S.; Jaafar, N.M.; Nazli, M.H.; Amali, R.K.A. A comparative study of tea waste derived humic-like substances with lignite-derived humic substances on chemical composition, spectroscopic properties and biological activity. Environ. Sci. Pollut. Res. 2022, 29, 60631–60640. [Google Scholar] [CrossRef]
- Li, L.; Xu, M.; Eyakub Ali, M.; Zhang, W.; Duan, Y.; Li, D. Factors affecting soil microbial biomass and functional diversity with the application of organic amendments in three contrasting cropland soils during a field experiment. PLoS ONE 2018, 13, e0203812. [Google Scholar] [CrossRef]
- Kielak, A.M.; Barreto, C.C.; Kowalchuk, G.A.; Van Veen, J.A.; Kuramae, E.E. The ecology of Acidobacteria: Moving beyond genes and genomes. Front. Microbiol. 2016, 7, 744. [Google Scholar] [CrossRef]
- Liu, M.; Wang, C.; Wang, F.; Xie, Y. Vermicompost and humic fertilizer improve coastal saline soil by regulating soil aggregates and the bacterial community. Arch. Agron. Soil Sci. 2019, 65, 281–293. [Google Scholar] [CrossRef]
- Panneerselvam, P.; Kumar, U.; Sahu, S.; Mohapatra, S.D.; Dangar, T.K.; Parameswaran, C.; Jahan, A.; Senapati, A.; Govindharaj, G.P.P. Larvicidal potential of Skermanella sp. against rice leaf folder (Cnaphalocrosis medinalis Guenee) and pink stem borer (Sesamia inferens Walker). J. Invertebr. Pathol. 2018, 157, 74–79. [Google Scholar] [CrossRef]
- Velázquez-Becerra, C.; Macías-Rodríguez, L.I.; López-Bucio, J.; Flores-Cortez, I.; Santoyo, G.; Hernández-Soberano, C.; Valencia-Cantero, E. The rhizobacterium Arthrobacter agilis produces dimethylhexadecylamine, a compound that inhibits growth of phytopathogenic fungi in vitro. Protoplasma 2013, 250, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Fan, M.; Wang, E.; Chen, W.; Wei, G. Interactions of plant growth-promoting rhizobacteria and soil factors in two leguminous plants. Appl. Microbiol. Biotechnol. 2017, 101, 8485–8497. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Liu, C.; Song, C.; Wang, X.; Ma, X.; Gao, J.; Gao, S.; Wang, L. Linking soil organic carbon mineralization with soil microbial and substrate properties under warming in permafrost peatlands of Northeastern China. Catena 2021, 203, 105348. [Google Scholar] [CrossRef]
- Condron, L.; Stark, C.; O’Callaghan, M.; Clinton, P.; Huang, Z. The Role of Microbial Communities in the Formation and Decomposition of Soil Organic Matter. In Soil Microbiology and Sustainable Crop Production; Dixon, G.R., Tilston, E.L., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 81–118. [Google Scholar]
- Yang, Y.; Liu, H.; Dai, Y.; Tian, H.; Zhou, W.; Lv, J. Soil organic carbon transformation and dynamics of microorganisms under different organic amendments. Sci. Total Environ. 2021, 750, 141719. [Google Scholar] [CrossRef]
- Zhao, S.; Qiu, S.; Xu, X.; Ciampitti, I.A.; Zhang, S.; He, P. Change in straw decomposition rate and soil microbial community composition after straw addition in different long-term fertilization soils. Appl. Soil Ecol. 2019, 138, 123–133. [Google Scholar] [CrossRef]
- Farrell, M.; Kuhn, T.K.; Macdonald, L.M.; Maddern, T.M.; Murphy, D.V.; Hall, P.A.; Singh, B.P.; Baumann, K.; Krull, E.S.; Baldock, J.A. Microbial utilisation of biochar-derived carbon. Sci. Total Environ. 2013, 465, 288–297. [Google Scholar] [CrossRef]
- Chen, Z.; Huang, G.; Li, Y.; Zhang, X.; Xiong, Y.; Huang, Q.; Jin, S. Effects of the lignite bioorganic fertilizer on greenhouse gas emissions and pathways of nitrogen and carbon cycling in saline-sodic farmlands at Northwest China. J. Clean. Prod. 2022, 334, 130080. [Google Scholar] [CrossRef]
- Luo, S.; Wang, S.; Tian, L.; Shi, S.; Xu, S.; Yang, F.; Li, X.; Wang, Z.; Tian, C. Aggregate-related changes in soil microbial communities under different ameliorant applications in saline-sodic soils. Geoderma 2018, 329, 108–117. [Google Scholar] [CrossRef]
- Shu, X.; He, J.; Zhou, Z.; Xia, L.; Hu, Y.; Zhang, Y.; Zhang, Y.; Luo, Y.; Chu, H.; Liu, W.; et al. Organic amendments enhance soil microbial diversity, microbial functionality and crop yields: A meta-analysis. Sci. Total Environ. 2022, 829, 154627. [Google Scholar] [CrossRef]
- Harindintwali, J.D.; Zhou, J.; Muhoza, B.; Wang, F.; Herzberger, A.; Yu, X. Integrated eco-strategies towards sustainable carbon and nitrogen cycling in agriculture. J. Environ. Manag. 2021, 293, 112856. [Google Scholar] [CrossRef]
- Fan, K.; Delgado-Baquerizo, M.; Guo, X.; Wang, D.; Zhu, Y.-g.; Chu, H. Biodiversity of key-stone phylotypes determines crop production in a 4-decade fertilization experiment. ISME J. 2021, 15, 550–561. [Google Scholar] [CrossRef] [PubMed]

| Organic Amendments | |||
|---|---|---|---|
| Parameter | FM | SR | LHA |
| pH. | 8.14 ± 0.09 | 7.63 ± 0.29 | 5.3 ± 0.15 |
| EC1:5 (dS m−1) | 8.34 ± 0.58 | 3.99 ± 0.39 | 2.5 ± 0.26 |
| C (%) | 28.76 ±0.91 | 46.3 ± 1.3 | 42.04 ± 0.8 |
| N (%) | 2.31 ± 0.03 | 1.6 ± 0.1 | 1.10 ± 0.03 |
| P (%) | 3.1 ± 0.06 | 0.8 ± 0.1 | 2.73 ± 0.03 |
| C:N | 12.45 ± 0.25 | 28.93 ± 0.3 | 38.22 ± 0.46 |
| Water content (%) | 29.56 ± 2.33 | 26.7 ± 1.2 | 10.21 ± 0.53 |
| Parameter | Saline–Sodic Soil |
|---|---|
| pH | 9.64 |
| EC1:5 (dS m−1) | 0.41 |
| ESP(%) | 16.82 |
| BD (g/cm3) | 1.68 |
| TN (mg/kg) | 550.36 |
| AP (mg/kg) | 16.35 |
| AK (mg/kg) | 50.88 |
| SOM (g/kg) | 8.23 |
| CaCO3(%) | 3% |
| Sand (%) | 71% |
| Clay (%) | 11% |
| Silt (%) | 18% |
| CK | SR | LHA | FM | |
|---|---|---|---|---|
| pH | 9.89 ± 0.16 a | 8.65 ± 0.16 c | 8.54 ± 0.09 c | 8.92 ± 0.09 b |
| EC1:5 (dS m−1) | 0.39 ± 0.06 a | 0.12 ± 0.01 bc | 0.10 ± 0.01 c | 0.17 ± 0.05 b |
| BD (g/cm3) | 1.69 ± 0.04 a | 1.62 ± 0.03 ab | 1.60 ± 0.06 b | 1.57 ± 0.04 b |
| TN (mg/kg) | 531.57 ± 91.7 b | 843.07 ± 48.14 a | 861.58 ± 84.62 a | 938.43 ± 232.6 a |
| AP (mg/kg) | 15.77 ± 6.66 c | 24.36 ± 2.4 b | 31.55 ± 5.62 a | 26.23 ± 3.24 ab |
| AK (mg/kg) | 51.77 ± 5.91 c | 98.56 ± 13.63 b | 141.35 ± 5.94 a | 109.18 ± 8.04 b |
| SOM (g/kg) | 8.5 ± 2.67 b | 13.8 ± 1.56 a | 15.9 ± 1.82 a | 15.72 ± 1.66 a |
| Treatments | Ear Number (Ears/m2) | Row Number Per Ear (Rows/Ear) | Kernel Number Per Row (Kernels/Row) | 1000-Kernel Weight (g) | Grain Yield (Ton/ha) |
|---|---|---|---|---|---|
| CK | 6.16 ± 0.11 b | 14.63 ± 0.46 a | 29.28 ± 1.19 b | 313.2 ± 5.45 b | 8.26 ± 0.35 c |
| SR | 6.46 ± 0.23 a | 14.52 ± 1.3 a | 32.78 ± 1.15 a | 324.2 ± 15.11 a | 9.97± 1.08 b |
| LHA | 6.56 ± 0.09 a | 15.1 ± 0.3 a | 33.78 ± 0.74 a | 331.8 ± 6.69 a | 11.10 ± 0.38 a |
| FM | 6.6 ± 0.23 a | 14.84 ± 0.17 a | 33.48 ± 1.17 a | 331.6 ± 8.62 a | 10.86 ± 0.30 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, L.; Nie, Z.; Zhou, J.; Zhang, S.; An, F.; Zhang, L.; Tóth, T.; Yang, F.; Wang, Z. Effects of Different Organic Amendments on Soil Improvement, Bacterial Composition, and Functional Diversity in Saline–Sodic Soil. Agronomy 2022, 12, 2294. https://doi.org/10.3390/agronomy12102294
Guo L, Nie Z, Zhou J, Zhang S, An F, Zhang L, Tóth T, Yang F, Wang Z. Effects of Different Organic Amendments on Soil Improvement, Bacterial Composition, and Functional Diversity in Saline–Sodic Soil. Agronomy. 2022; 12(10):2294. https://doi.org/10.3390/agronomy12102294
Chicago/Turabian StyleGuo, Liangliang, Zhaoyang Nie, Jie Zhou, Shixin Zhang, Fenghua An, Lu Zhang, Tibor Tóth, Fan Yang, and Zhichun Wang. 2022. "Effects of Different Organic Amendments on Soil Improvement, Bacterial Composition, and Functional Diversity in Saline–Sodic Soil" Agronomy 12, no. 10: 2294. https://doi.org/10.3390/agronomy12102294
APA StyleGuo, L., Nie, Z., Zhou, J., Zhang, S., An, F., Zhang, L., Tóth, T., Yang, F., & Wang, Z. (2022). Effects of Different Organic Amendments on Soil Improvement, Bacterial Composition, and Functional Diversity in Saline–Sodic Soil. Agronomy, 12(10), 2294. https://doi.org/10.3390/agronomy12102294





