Segregation was observed among populations evaluated after selfing BC
2F
1 generation. Ten percent of the total plants planted per BC
2F
1 populations were screened for performance of traits. A total of six genotypes (NA241, NA307, NA 316, SE408, SE438 and W82) were selected from the best 10% of screened plants (
Table 1).
3.3. Distribution of Chlorophyll Content in Sorghum Genotypes
The evaluation of chlorophyll content distribution indicated that the highest content was found in the middle leaves of sorghum plants in both environments. Results in the water-stressed environment indicated that the donor parent B35 expressed the highest chlorophyll content with 59.04 g
−1 at the top leaf, 60.71 at the middle and 51.4 g
−1 at the bottom leaf (
Table 4). The genotype SE438 expressed the highest chlorophyll content (47.18, 52.14 and 44.96 g
−1) at top, middle and bottom leaves of the plant, respectively. A majority of the genotypes (NA316C, NA307, NA241B, SE408, Seguifa, NACO Mtama 1, Wahi, S438 and B35) had chlorophyll content above 30 g
−1 in all parts of the plant that have been recommended for sorghum. However, the overall chlorophyll content was higher in the donor parent and recurrent parents except for genotype SE438.
Results in the well-watered environment showed greater than 35 g
−1 of chlorophyll content on top, middle and bottom leaves of all sorghum genotypes. The genotypes SE438, NA316C and the parents B35 and Seguifa resulted in greater than 46 g
−1 of chlorophyll content in both parts of the plant. For all genotypes evaluated in the well-watered environment, B35 showed the highest chlorophyll content (66.01, 63.94 and 58.23 g
−1) on the top, middle and bottom plant leaves (
Table 4).
There were significant differences in traits (plant height, leaf rolling, grain weight per plant, panicle weight per plant and STG) of sorghum genotypes at
p < 0.001 under well-watered and water-stress treatments (
Table 5 and
Table 6). The mean performance of plant height was the highest (142.2 cm) in population NA316, followed by NA316B with 139 cm in the stressed environment. The donor parent exhibited the lowest (95.5 cm) plant height in the water-stressed trial; however, there were no significant differences among populations tested. In the well-watered trial, the genotype NA316A and NA316C indicated the high performance of plant height with 143.3 cm and 140.3 cm, while the lowest plant height (94.6 cm) was recorded in the donor parent B35. There were significance differences of leaf rolling in the genotypes NA241B, NA316A, NA316BB, SE408, local check Wahi and the donor parent B35 in the water-stressed trial (
Table 6). The score of leaf rolling for the genotypes NA241A, NA316C, NA307, NACO Mtama 1, Seguifa and SE438 varied but were not significant. The genotypes SE438 and NA316C scored low leaf rolling in both environments. The parent NACO Mtama 1 had the lowest score of leaf rolling under well-watered condition, and the genotypes NA307 and SE438 had the lowest score of leaf rolling for the remaining genotypes under water-stressed condition.
In this study, there were variations (
p < 0.05) for the total number of leaves counted at physiological plant maturity among sorghum genotypes evaluated in water-irrigated and water-stressed conditions. The genotype NA307 showed the highest total number of leaves of backcross genotypes across the environments. The donor parent B35 and check Wahi showed the highest (3.53 and 2.73) total number of green leaves at maturity when compared with backcross genotypes under water-stress environment. A similar trend was recorded in the well-watered environment (
Table 5 and
Table 6).
The genotypes NA316C and NA316A showed 3.87 and 4.2 ratings for STG, similar to the check Wahi and B35 with 3.73 and 4.13 ratings of STG, respectively, in the well-irrigated environment (
Table 5). The genotype NA241B and NA316A had 5.6 and 5.8 ratings for STG, similar to the check Wahi, which showed 5.7 STG under water-stressed environment (
Table 5). The genotype NA316A performed well across the environments. The lowest STG rating (5.8) was recorded from the genotype NA241B in water-irrigated and the lowest STG (7.3) in the genotype NA241A in water-stressed environment.
Based on the root biomass, genotypes NA316C and NA316A produced greater amounts (0.068 and 0.052 kg) than other backcross genotypes, except parents B35 and Seguifa, which produced the greatest root biomass in water-irrigated condition (
Table 5). The lowest (0.04 kg) root biomass was reported on genotype SE408 in unstressed condition. Similarly, there were significant differences among genotypes evaluated in the stressed condition, whereas check Wahi recorded the greatest (0.058 kg) root biomass, followed by B35 (0.048 kg). The genotypes SE 438 and NA307 (45 g) performed well in root biomass accumulation. The genotypes NA241A and SE408 were significantly affected by post-flowering drought, resulting in less than 0.037 kg of root biomass (
Table 6).
The genotypes NA307, NA316C and S438 performed well in panicle weight (per plant in well-watered condition, with 0.88, 0.8 and 0.74 kg, and 0.79, 0.7 and 0.74 kg in water-stressed environments, respectively (
Table 5 and
Table 6). The panicles per plant showed significant differences among the genotypes at
p < 0.05.
The genotypes NA316C and NA307 produced high yield across the environments. Genotypes NA316C recorded the highest (0.077 kg) grain yield per plant under well-watered condition (
Table 5). The genotype SE438 produced the highest (0.06 kg) grain yield per plant in the water-stress environment (
Table 6).
The mean square values of various traits evaluated between genotypes by environments revealed interactions (at
p < 0.05 and
p < 0.01) (
Table 7). The interaction was recorded from nine traits including days to 50% flowering, leaf rolling, chlorophyll content, total number of green leaves at plant maturity, panicle weight per plant, root biomass and stem biomass. The mean squares for genotypes were significant (at
p < 0.01) for days to 50% flowering, plant height, chlorophyll content, total number of green leaves at plant maturity and 1000-seed grain weight, and at
p < 0.05 for leaf rolling, stem biomass and panicle weight per plant traits. The majority of the traits evaluated for mean square performance between genotypes by environments showed no significant differences except yield of 1000-seed grain weight.
There were positive and negative correlation coefficients among traits contributing to drought tolerance and yield in sorghum populations (
Table 8). The traits of panicle weight, panicle width and panicle length were significantly correlated with grain yield. STG and inflorescence exsertion were negatively correlated with grain yield. Panicle width and panicle weight were strongly correlated to grain yield and each other. Chlorophyll content was positive and significantly correlated with total number of green leaves at maturity. Plant height was correlated with panicle weight; however, it was less correlated with grain yield. STG and inflorescence exsertion showed negative correlation with most traits. However, both showed nonsignificant positive correlation with plant height. The largest negative correlation was between traits STG and total number of green leaves at maturity, and STG and chlorophyll content.
3.6. Drought Stress Indices
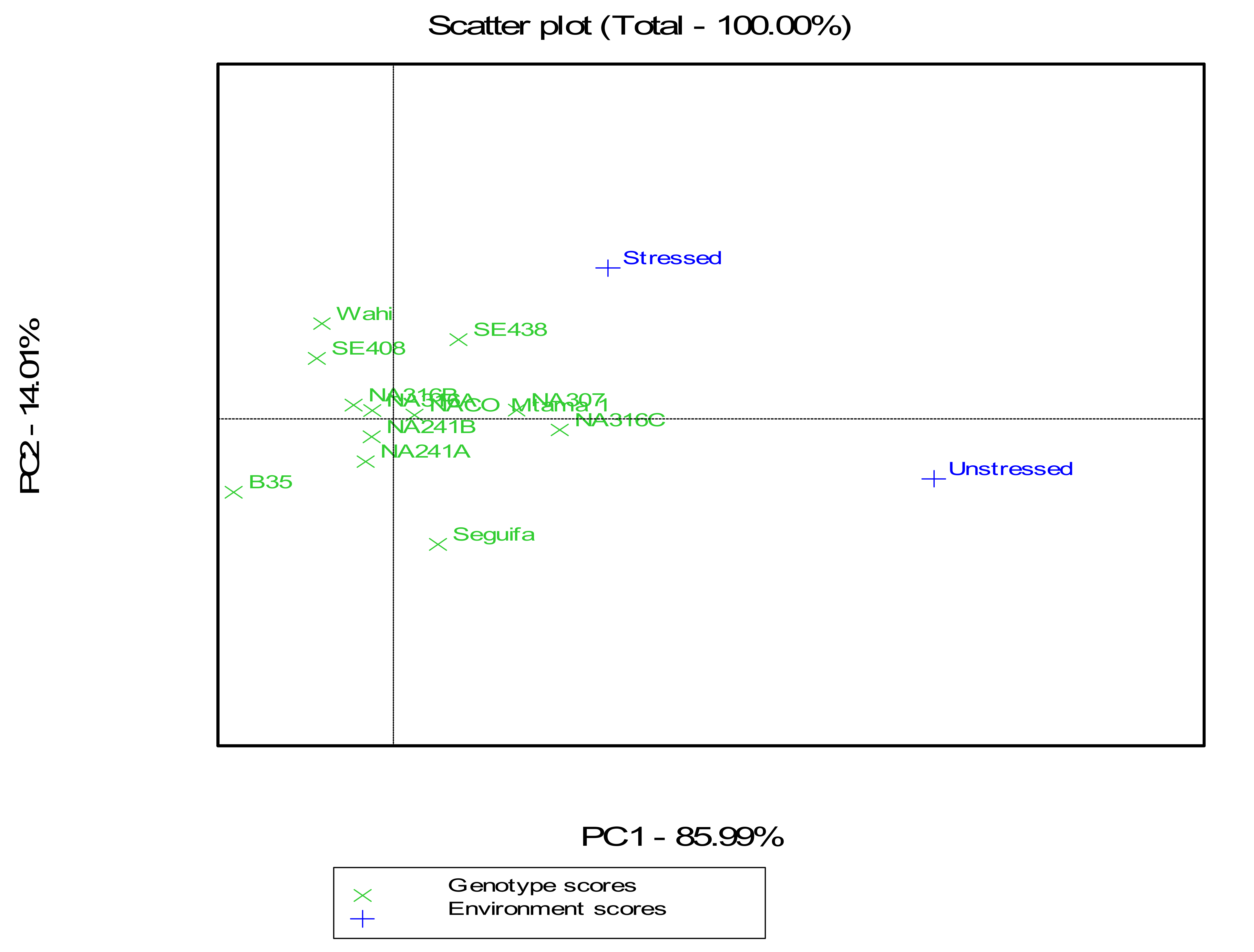
Based on grain yield results, the highest MP and GMP were recorded for populations NA316C (3.42 and 2.59 ton ha
−1) and NA307 (3.16 and 2.56 ton ha
−1), suggesting these genotypes are stable for high yield across the environments (
Table 11). Yield based on STI indicated that the population NA316C (with mean yield 3.42 and 2.59 ton ha
−1), NA307 (3.16 and 2.56 ton ha
−1) and SE438 (2.77 and 2.65 ton ha
−1) performed the best across the environments with the STI values 1.39, 1.27 and 1.15, respectively. The highest values (830, 652 and 607) of TOL were recorded in the populations NA316C (3.42 and 2.59 ton ha
−1), Seguifa (2.63 and 1.98 ton ha
−1) and NA307 (3.16 and 2.56 ton ha
−1). The lowest values (−385, −252 and 66) were recorded in the populations Wahi with mean yield of 2.03 and 2.42 ton ha
−1, SE408 (2.04 and 2.3 ton ha
−1) and NA316B (2.3 and 2.23 ton ha
−1). These populations performed differently across the environments.
Correlation coefficients were used as a tool to determine the promising criteria for drought-tolerant populations (
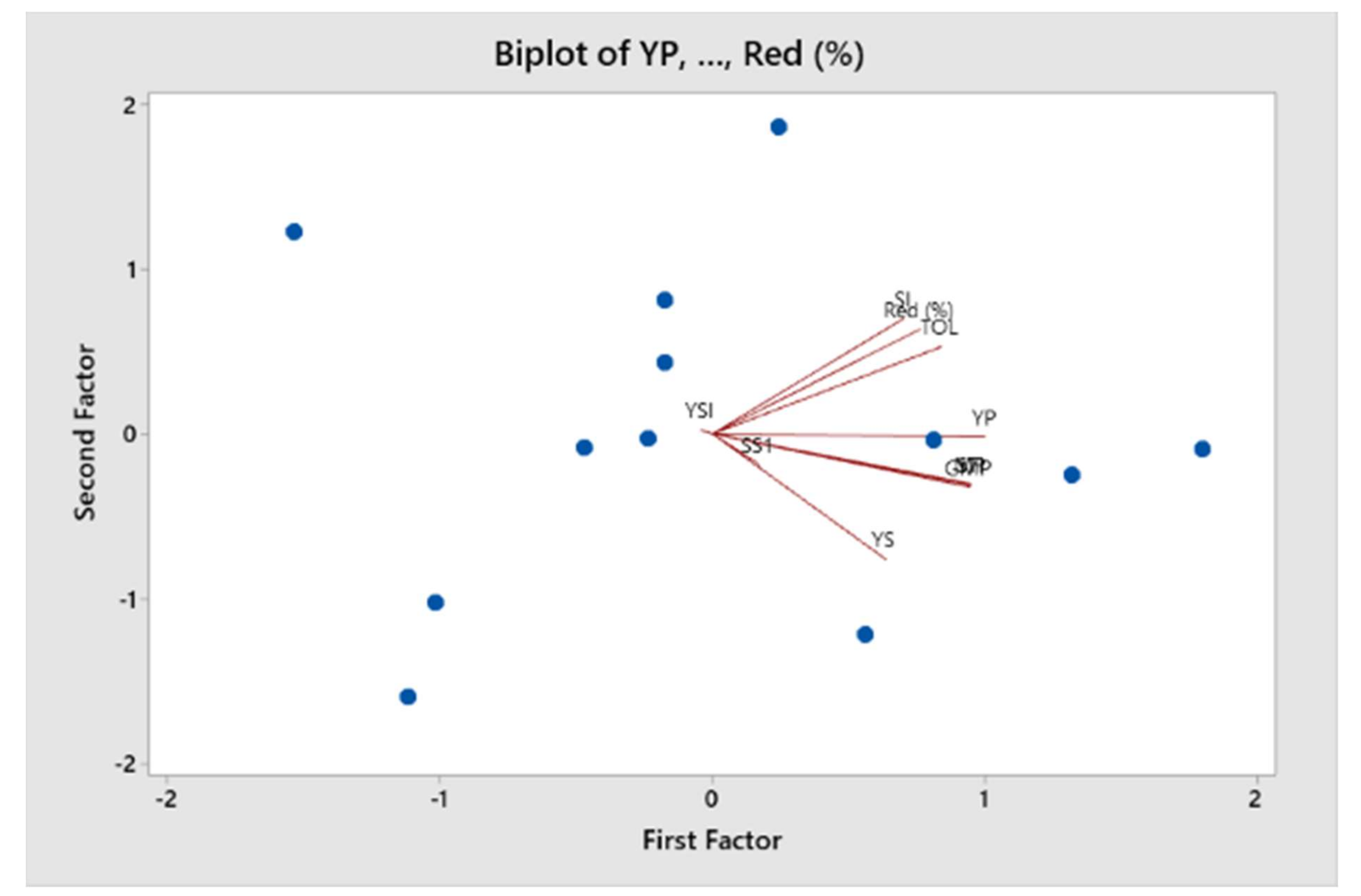
Table 12). The indices GMP and MP were highly correlated with YP and YS and each other. TOL was positively correlated with yield by 0.83 under water-irrigated environment and low correlation with yield by 0.12 under water-stressed condition. The contrasting correlation between TOL and YP and YS suggested the reduction of grain yield under well-watered condition thus is not recommended for selection of promising populations for drought tolerance indices. There were no significant correlations between SSI and the other indices. SSI is the best index for selection of populations with low yield and drought tolerance to water-stressed condition. YSI was strongly correlated with STI and negatively correlated with YP, TOL, MP, GMP and SSI. Red was only positively correlated with TOL and MP; the remaining indices were negatively correlated and nonsignificant. TOL was positively correlated with MP and GMP but not strongly correlated with YP. Furthermore, SSI indicated nonsignificant correlation with MP and GMP. The lowest correlation was found between Red and YSI and the indices between YSI and TOL.
Ten principal axes were generated in the study of PCA in drought tolerance indices. PC1 showed 59.1% of the total variation and positively correlated with YP, YS, TOL, MP, GMP, SI, SSI, STI and Red (
Table 13). In this study, the first dimension of the principal component may be selected as the best indicator of yield potential and drought tolerance. The population with high values of PC1 can be high yielding in the water-irrigated and water-stressed environments. PC2 showed 20.8% of the variation and explained the positive correlation with TOL, SI, YSI and Red (
Table 13). PC3 indicated 10% variation explaining positive correlation of most of the indices except TOL and SSI. The findings revealed that SSI did not separate the drought-susceptible cultivars. However, the populations with highest PCA1 and lowest PCA2 showed good yield performance in both environments. The remaining of principal axes indicated less than 10% of the total variation.
There was correlation of angles between vectors in the biplot diagram of PCA (
Figure 2). There was similarity between STI and MP and strong correlation with GMP. YP and YS were distantly correlated, indicating that water stress reduced the yield.