Breeding Amaranth for Biomass: Evaluating Dry Matter Content and Biomass Potential in Early and Late Maturing Genotypes
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
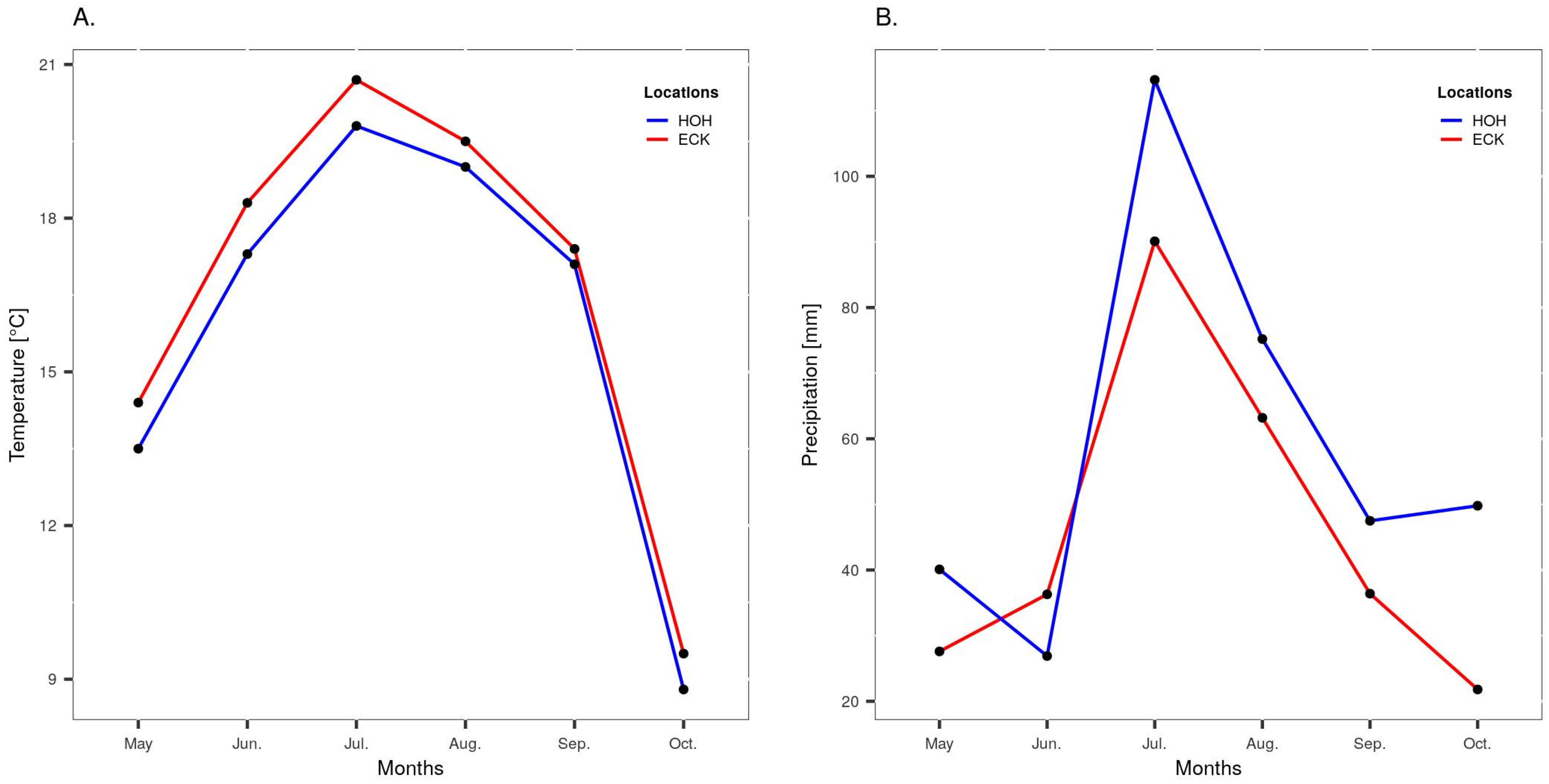
2.2. Experimental Design and Phenotypic Evaluation of Field Trials
2.3. Statistical Analysis
3. Results
3.1. Differences between Biomass Types and Grain Types
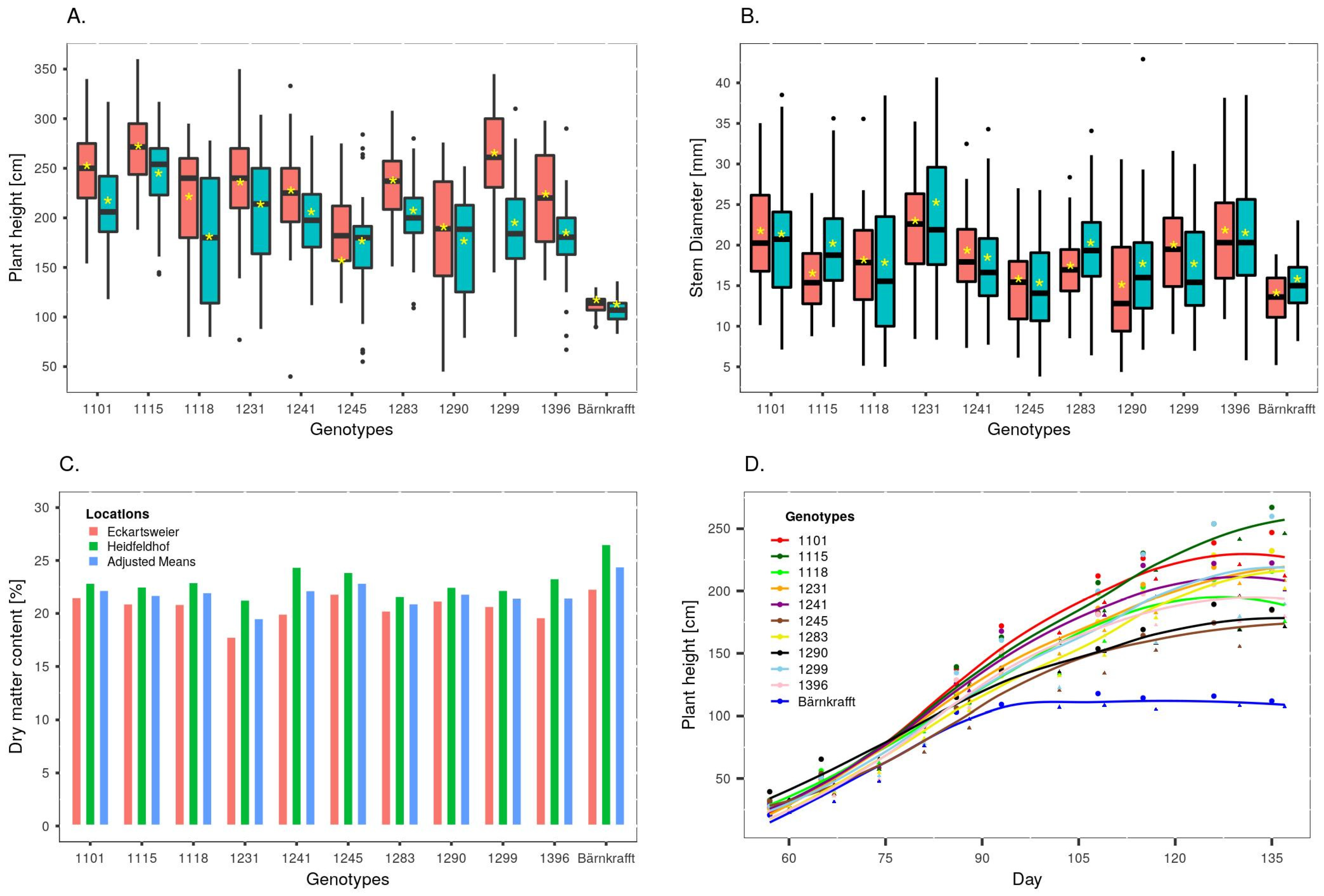
3.2. Variation in Biomass Yield Components
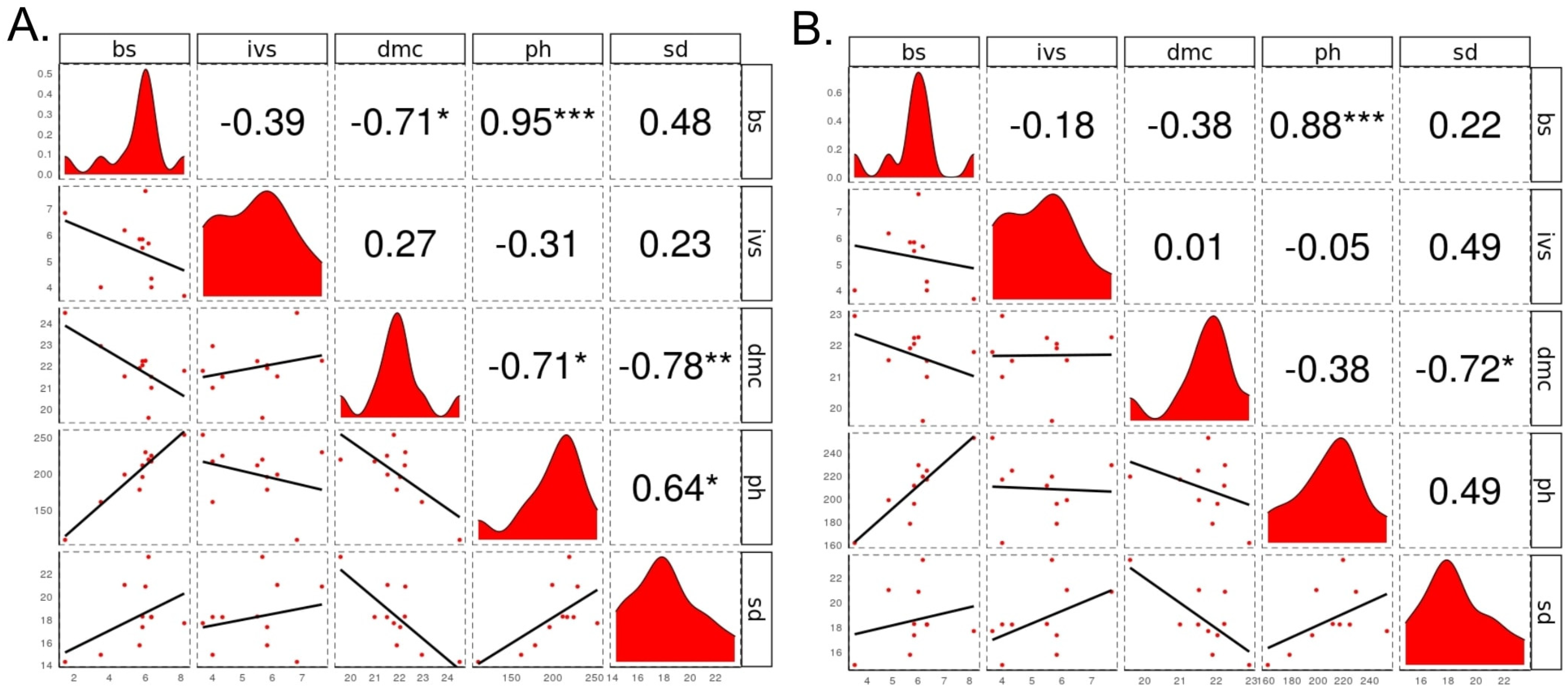
3.3. Quantitative Genetics Parameters and Relationships between Traits
4. Discussion
4.1. Variation in Biomass Yield Components
4.2. Trade-Off between Earliness and Photoperiod Sensitivity
4.3. Quantitative Genetics Parameters and Relationships between Traits
4.4. Future Prospects
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef]
- Myrna, O.; Odening, M.; Ritter, M. The Influence of Wind Energy and Biogas on Farmland Prices. Land 2019, 8, 19. [Google Scholar] [CrossRef]
- Daniel-Gromke, J.; Rensberg, N.; Denysenko, V.; Stinner, W.; Schmalfuß, T.; Scheftelowitz, M.; Nelles, M.; Liebetrau, J. Current Developments in Production and Utilization of Biogas and Biomethane in Germany. Chem. Ing. Tech. 2017, 90, 17–35. [Google Scholar] [CrossRef]
- Grieder, C.; Dhillon, B.S.; Schipprack, W.; Melchinger, A.E. Breeding maize as biogas substrate in Central Europe: II. Quantitative-genetic parameters for inbred lines and correlations with testcross performance. Theor. Appl. Genet. 2012, 124, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, A.; Rath, J. Biogas Production from Maize: Current State, Challenges, and Prospects. 1. Methane Yield Potential. Bioenergy Res. 2012, 5, 1027–1042. [Google Scholar] [CrossRef]
- Balodis, O.; Bartuševics, J.; Gaile, Z. Biomass Yield of Different Plants for Biogas Production. Environ. Technol. Resour. 2011, 1, 238–245. [Google Scholar]
- Brauer-Siebrecht, W.; Jacobs, A.; Christen, O.; Götze, P.; Koch, H.-J.; Rücknagel, J.; Märländer, B. Silage Maize and Sugar Beet for Biogas Production in Rotations and Continuous Cultivation: Dry Matter and Estimated Methane Yield. Agronomy 2016, 6, 2. [Google Scholar] [CrossRef]
- Vogel, E.; Deumlich, D.; Kaupenjohann, M. Bioenergy maize and soil erosion-Risk assessment and erosion control concepts. Geoderma 2016, 261, 80–92. [Google Scholar] [CrossRef]
- Brenner, D.M.; Baltensperger, D.D.; Kulakow, P.A.; Lehmann, J.W.; Myers, R.L.; Slabbert, M.M.; Sleugh, B.B. Genetic Resources and Breeding of Amaranthus. Plant Breed. Rev. 2010, 19, 227–285. [Google Scholar]
- Sauer, J.D. The grain amaranths and their relatives: A revised taxonomic and geo- graphic survey. Ann. Missouri Bot. Gard. 1967, 54, 102–137. [Google Scholar] [CrossRef]
- National Research Council, Amaranth Modern Prospects for an Ancient Crop; National Academy Press: Washington, DC, USA, 1984.
- Stallknecht, G.F.; Schulz-Schaeffer, J.R. Amaranth Rediscovered; Janick, J., Simon, J.E., Eds.; New Crops Wiley: New York, NY, USA, 1993; pp. 211–218. [Google Scholar]
- Sauer, J.D. The Grain Amaranths: A Survey of Their History and Classification. Ann. Mo. Bot. Gard. 1950, 37, 561. [Google Scholar] [CrossRef]
- Costea, M.; Tardif, F.J. The biology of Canadian weeds. 130. Amaranthus retroflexus L., A. powellii S. Watson and A. hybridus L. (update). Can. J. Plant Sci. 2004, 84, 631–668. [Google Scholar] [CrossRef]
- Assad, R.; Reshi, Z.A.; Jan, S.; Rashid, I. Biology of Amaranths. Bot. Rev. 2017, 83, 382–436. [Google Scholar] [CrossRef]
- Myers, R.L. Amaranth: New Crop Opportunity Progress in New Crops; ASHS Press: Alexandria, VA, USA, 1996; pp. 207–220. [Google Scholar]
- Liu, F.; Stützel, H. Biomass partitioning, specific leaf area, and water use efficiency of vegetable amaranth (Amaranthus spp.) in response to drought stress. Sci. Hortic. (Amst.) 2004, 102, 15–27. [Google Scholar] [CrossRef]
- Von Cossel, M.; Möhring, J.; Kiesel, A.; Lewandowski, I. Methane yield performance of amaranth (Amaranthus hypochondriacus L.) and its suitability for legume intercropping in comparison to maize (Zea mays L.). Ind. Crop. Prod. 2017, 103, 107–121. [Google Scholar] [CrossRef]
- Sleugh, B.B.; Moore, K.J.; Brummer, E.; Knapp, A.D.; Russell, J.; Gibson, L. Forage Nutritive Value of Various Amaranth Species at Different Harvest Dates. Crop. Sci. 2001, 41, 466–472. [Google Scholar] [CrossRef]
- Svirskis, A. Prospects for non-traditional plant species cultivated for forage in Lithuania. Not. Bot. Horti Agrobot. Cluj-Napoca 2009, 37, 215–218. [Google Scholar]
- Seguin, P.; Mustafa, A.F.; Donnelly, D.J.; Gélinas, B. Chemical composition and ruminal nutrient degradability of fresh and ensiled amaranth forage. J. Sci. Food Agric. 2013, 93, 3730–3736. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Sun, M.; Yue, S.; Cai, Y.; Huang, R.; Brenner, D.; Corke, H. Field evaluation of an Amaranthus genetic resource collection in China. Genet. Resour. Crop. Evol. 2000, 47, 43–53. [Google Scholar] [CrossRef]
- Fuller, H.J. Photoperiodic Responses of Chenopodium quinoa Willd. and Amaranthus caudatus L. Am. J. Bot. 1949, 36, 175. [Google Scholar] [CrossRef]
- Jung, C.; Müller, A.E. Flowering time control and applications in plant breeding. Trends Plant Sci. 2009, 14, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Hauptil, H.; Vaidya, K.R. Outcrossing rate in grain amaranths. J. Hered. 1982, 73, 71–72. [Google Scholar] [CrossRef]
- Hauptli, H.; Jain, S. Genetic variation in outcrossing rate and correlated floral traits in a population of grain amaranth (Amaranthus cruentus L.). Genetica 1985, 66, 21–27. [Google Scholar] [CrossRef]
- Lehmann, J.W.; Clark, R.L.; Frey, K.J. Biomass Heterosis and Combining Ability in Interspecific and Intraspecific Matings of Grain Amaranths. Crop. Sci. 1991, 31, 1111–1116. [Google Scholar] [CrossRef]
- Stetter, M.G.; Zeitler, L.; Steinhaus, A.; Kroener, K.; Biljecki, M.; Schmid, K.J. Crossing Methods and Cultivation Conditions for Rapid Production of Segregating Populations in Three Grain Amaranth Species. Front. Plant Sci. 2016, 7, 816. [Google Scholar] [CrossRef]
- Brenner, D.M. Registration of DB 199313, Cytoplasmic Male Sterile Grain Amaranth Genetic Stock. J. Plant Regist. 2019, 13, 251–253. [Google Scholar] [CrossRef]
- Rivelli, A.R.; Gherbin, P.; De Maria, S.; Pizza, S. Field evaluation of Amaranthus species for seed and biomass yields in southern Italy. Ital. J. Agron. 2008, 3, 225–229. [Google Scholar] [CrossRef]
- Mursec, B.; Vindis, P.; Janzekovic, M.; Brus, M.; Cus, F. Analysis of different substrates for processing into biogas Manufacturing and processing. J. Achiev. Mater. Manuf. Eng. 2009, 37, 652–659. [Google Scholar]
- Pospišil, A. Yield and Quality of Forage Sorghum and Different Amaranth Species (Amaranthus spp.) Biomass. Agric. Conspec. Sci. 2009, 74, 85–89. [Google Scholar]
- Seppälä, M.; Laine, A.; Rintala, J. Screening of novel plants for biogas production in northern conditions. Bioresour. Technol. 2013, 139, 355–362. [Google Scholar] [CrossRef]
- Sitkey, V.; Gaduš, J.; Kliský, Ľ.; Dudák, A. Biogas Production from Amaranth Biomass. Acta Reg. Environ. 2013, 10, 59–62. [Google Scholar] [CrossRef][Green Version]
- Melchinger, A.E.; Gumber, R.K. Overview of Heterosis and Heterotic Groups in Agronomic Crops. In Yield Gains in Major U.S. Field Crops; Wiley: New York, NY, USA, 2015; pp. 29–44. [Google Scholar]
- Duvick, D.N. The Contribution of Breeding to Yield Advances in maize (Zea mays L.). Adv. Agron. 2005, 86, 83–145. [Google Scholar]
- Fu, D.; Xiao, M.; Hayward, A.; Fu, Y.; Liu, G.; Jiang, G.; Zhang, H. Utilization of crop heterosis: A review. Euphytica 2014, 197, 161–173. [Google Scholar] [CrossRef]
- Joshi, D.C.; Sood, S.; Hosahatti, R.; Kant, L.; Pattanayak, A.; Kumar, A.; Yadav, D.; Stetter, M.G. From zero to hero: The past, present and future of grain amaranth breeding. Theor. Appl. Genet. 2018, 131, 1807–1823. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Pandey, V.; Pachauri, G.; Dixit, B.S.; Banerji, R.; Singh, S.P. Nutritional contents of different foliage cuttings of vegetable amaranth. Plant Foods Hum. Nutr. 2003, 58, 1–8. [Google Scholar] [CrossRef]
- Shukla, S.; Bhargava, A.; Chatterjee, A.; Singh, S.P. Estimates of genetic parameters to determine variability for foliage yield and its different quantitative and qualitative traits in vegetable amaranth (A. tricolor) [India]. J. Genet. Breed. 2004, 58, 169–176. [Google Scholar]
- Shukla, S.; Bhargava, A.; Chatterjee, A.; Srivastava, A.; Singh, S.P. Genotypic variability in vegetable amaranth (Amaranthus tricolor L. for foliage yield and its contributing traits over successive cuttings and years. Euphytica 2006, 151, 103–110. [Google Scholar] [CrossRef]
- Bundessortenamt-Deleted Varieties. Available online: http://www.bundessortenamt.de/internet30/index.php?id=141 (accessed on 18 November 2016).
- Bewick, V.; Cheek, L.; Ball, J. Statistics review 10: Further nonparametric methods. Crit. Care 2004, 8, 196–199. [Google Scholar] [CrossRef][Green Version]
- Kenward, M.G.; Roger, J.H. Small Sample Inference for Fixed Effects from Restricted Maximum Likelihood. Biometrics 1997, 53, 983–997. [Google Scholar] [CrossRef]
- Piepho, H.-P.; Möhring, J. Computing heritability and selection response from unbalanced plant breeding trials. Genetics 2007, 177, 1881–1888. [Google Scholar] [CrossRef]
- Pucher, A.; Høgh-Jensen, H.; Gondah, J.; Hash, C.T.; Haussmann, B.I.G. Micronutrient Density and Stability in West African Pearl Millet-Potential for Biofortification. Crop. Sci. 2014, 54, 1709–1720. [Google Scholar] [CrossRef]
- Base SAS® 9.4 Procedures Guide: Statistical Procedures, 2nd ed.; SAS Institute Inc.: Cary, NC, USA, 2013.
- Piepho, H.-P. A SAS macro for generating letter displays of pairwise mean comparisons. Commun. Biometry Crop. Sci. 2021, 13, 456–466. [Google Scholar]
- de Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research. R Ppackage Version 1.3-3. 2020. Available online: https://CRAN.R-project.org/package=agricolae (accessed on 18 November 2016).
- Schloerke, B.; Crowley, C.; Cook, D.; Hofmann, H. Ggally: Extension to ggplot2. R package version 2.0.0. 2020. Available online: https://www.semanticscholar.org/paper/Extension-to-%27ggplot2%27-%5BR-package-GGally-version-Schloerke-Cook/be3249f353c1766493da6f357708aaa00f70754a (accessed on 18 November 2016).
- Lemenkova, P. Regression Models by GRETL and R Statistical Packages for Data Analysis in Marine Geology. Int. J. Environ. Trends 2019, 3, 39–59. [Google Scholar]
- Franco, R.T.; Buffière, P.; Bayard, R. Ensiling for biogas production: Critical parameters. A review. Biomass Bioenergy 2016, 94, 94–104. [Google Scholar] [CrossRef]
- Li, Y.; Xu, M. CCT family genes in cereal crops: A current overview. Crop. J. 2017, 5, 449–458. [Google Scholar] [CrossRef]
- Kulakow, P.A.; Jain, S.K. The inheritance of flowering time in Amaranthus species. J. Genet. 1985, 64, 85–100. [Google Scholar] [CrossRef]
- Jähne, F.; Hahn, V.; Würschum, T.; Leiser, W.L. Speed breeding short-day crops by LED-controlled light schemes. Theor. Appl. Genet. 2020, 133, 2335–2342. [Google Scholar] [CrossRef]
- Fernandez, M.G.S.; Becraft, P.W.; Yin, Y.; Lübberstedt, T. From dwarves to giants? Plant height manipulation for biomass yield. Trends Plant Sci. 2009, 14, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; O’Kiely, P.; Doyle, E.M. Yield and chemical composition of contrasting maize cultivars at sequential stages of maturity. Adv. Anim. Biosci. 2010, 1, 322. [Google Scholar] [CrossRef]
- Windpassinger, S.; Friedt, W.; Frauen, M.; Snowdon, R.J.; Wittkop, B. Designing adapted sorghum silage types with an enhanced energy density for biogas generation in temperate Europe. Biomass Bioenergy 2015, 81, 496–504. [Google Scholar] [CrossRef]
- Schmidt, W. Hybridmaiszüchtung bei der KWS SAAT AG. In Bericht über die 54. Tagung 2003 der Vereinigung der Pflanzenzüchter und Saatgutkaufleute Österreichs; BAL Gumpenstein; 2003; pp. 1–6. Available online: https://raumberg-gumpenstein.at/jdownloads/Tagungen/Saatzuechtertagung/Saatzuechtertagung_2003/2s_2003_schmidt.pdf (accessed on 18 November 2016).
- Lehmann, J.W. Rapid cycling of grain amaranths. Legacy 1995, 8, 15–17. [Google Scholar]
- Brenner, D.M.; Widriechner, M.P. Amaranthus seed regeneration in plastic tents in greenhouses. Plant Genet. Resour. Newsl. 1998, 116, 1–4. [Google Scholar]
- Lightfoot, D.J.; Jarvis, D.E.; Ramaraj, T.; Lee, R.; Jellen, E.N.; Maughan, P.J. Single-molecule sequencing and Hi-C-based proximity-guided assembly of amaranth (Amaranthus hypochondriacus) chromosomes provide insights into genome evolution. BMC Biol. 2017, 15, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lübken, M.; Gehring, T.; Wichern, M. Microbiological fermentation of lignocellulosic biomass: Current state and prospects of mathematical modeling. Appl. Microbiol. Biotechnol. 2009, 85, 1643–1652. [Google Scholar] [CrossRef] [PubMed]
| Type 3 Tests for Genotype Effect (Including Bärnkrafft) | Broad-Sense Heritability and Plot-Based Repeatability (Including Bärnkrafft) | ||||||||||||
| Traits | Mean | ± | SE | Range | Num DF | Den DF | F-Value | Pr > F | H2 | wH | wE | ||
| Biomass score | 5.47 | ± | 0.52 | 1.50 | – | 8.17 | 10 | 10 | 13.51 | 0.0002 *** | 0.93 | 0.91 | 0.97 |
| Inflorescence volume score | 5.41 | ± | 0.36 | 3.67 | – | 7.67 | 10 | 10 | 6.08 | 0.0043 ** | 0.84 | 0.74 | 0.76 |
| Dry matter content (%) | 21.93 | ± | 0.38 | 19.59 | – | 24.48 | 10 | 10 | 3.83 | 0.0227 * | 0.74 | 0.78 | 0.79 |
| Plant height (cm) | 199.90 | ± | 11.80 | 109.46 | – | 253.36 | 10 | 10 | 11.87 | 0.0003 *** | 0.92 | 0.90 | 0.95 |
| Stem diameter (mm) | 18.18 | ± | 0.83 | 14.29 | – | 23.46 | 10 | 10 | 8.23 | 0.0013 ** | 0.88 | 0.78 | 0.80 |
| Type 3 Tests for Genotype Effect (Excluding Bärnkrafft) | Broad-Sense Heritability and Plot-Based Repeatability (Excluding Bärnkrafft) | ||||||||||||
| Traits | Mean | ± | SE | Range | Num DF | Den DF | F-Value | Pr > F | H2 | wH | wE | ||
| Biomass score | 5.87 | ± | 0.37 | 3.50 | – | 8.17 | 9 | 9 | 5.94 | 0.007 ** | 0.83 | 0.82 | 0.93 |
| Inflorescence volume score | 5.27 | ± | 0.40 | 3.67 | – | 7.67 | 9 | 9 | 5.60 | 0.0086 ** | 0.82 | 0.74 | 0.72 |
| Dry matter content (%) | 21.67 | ± | 0.28 | 19.59 | – | 22.93 | 9 | 9 | 2.47 | 0.0966 ns | 0.60 | 0.44 | 0.75 |
| Plant height (cm) | 208.95 | ± | 8.37 | 161.36 | – | 253.36 | 9 | 9 | 5.41 | 0.0096 ** | 0.82 | 0.75 | 0.89 |
| Stem diameter (mm) | 18.57 | ± | 0.81 | 14.91 | – | 23.46 | 9 | 9 | 6.63 | 0.0047 ** | 0.85 | 0.75 | 0.74 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baturaygil, A.; Stetter, M.G.; Schmid, K. Breeding Amaranth for Biomass: Evaluating Dry Matter Content and Biomass Potential in Early and Late Maturing Genotypes. Agronomy 2021, 11, 970. https://doi.org/10.3390/agronomy11050970
Baturaygil A, Stetter MG, Schmid K. Breeding Amaranth for Biomass: Evaluating Dry Matter Content and Biomass Potential in Early and Late Maturing Genotypes. Agronomy. 2021; 11(5):970. https://doi.org/10.3390/agronomy11050970
Chicago/Turabian StyleBaturaygil, Ali, Markus G. Stetter, and Karl Schmid. 2021. "Breeding Amaranth for Biomass: Evaluating Dry Matter Content and Biomass Potential in Early and Late Maturing Genotypes" Agronomy 11, no. 5: 970. https://doi.org/10.3390/agronomy11050970
APA StyleBaturaygil, A., Stetter, M. G., & Schmid, K. (2021). Breeding Amaranth for Biomass: Evaluating Dry Matter Content and Biomass Potential in Early and Late Maturing Genotypes. Agronomy, 11(5), 970. https://doi.org/10.3390/agronomy11050970






