Evaluation of Crossability between Nicotiana benthamiana and Nicotiana excelsior
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Interspecific Crosses
2.3. Analysis of Pollen Viability
2.4. Cytological Analysis of Chromosomes in Meiosis
3. Results
3.1. Reciprocal Crosses between N. benthamiana and N. excelsior
3.2. Fertility of Hybrids between N. benthamiana and N. excelsior
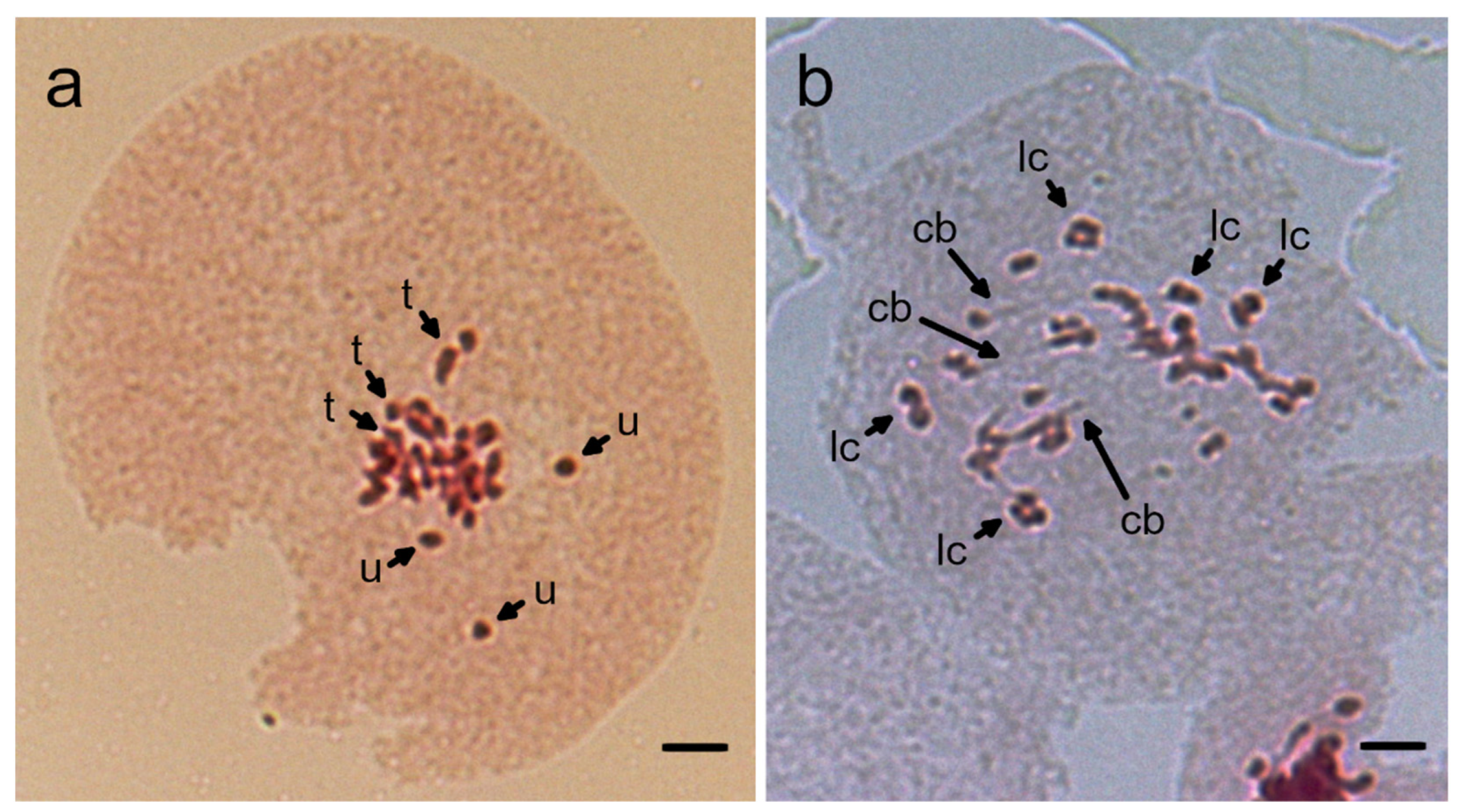
3.3. Chromosomal Instability in Reciprocal Hybrids between N. benthamiana and N. excelsior
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Abbott, R.J. Plant invasions, interspecific hybridization and the evolution of new plant taxa. Trends Ecol. Evol. 1992, 7, 401–405. [Google Scholar] [CrossRef]
- Rieseberg, L.H.; Willis, J.H. Plant Speciation. Science 2007, 317, 910–914. [Google Scholar] [CrossRef]
- Rodionov, A.V.; Amosova, A.V.; Belyakov, E.A.; Zhurbenko, P.M.; Mikhailova, Y.V.; Punina, E.O.; Shneyer, V.S.; Loskutov, I.G.; Muravenko, O.V. Genetic Consequences of Interspecific Hybridization, Its Role in Speciation and Phenotypic Diversity of Plants. Russ. J. Genet. 2019, 55, 278–294. [Google Scholar] [CrossRef]
- Newaskar, G.S.; Chimote, V.P.; Mehetre, S.S.; Jadhav, A.S. Interspecific hybridization in Gossypium L.: Characterization of progenies with different ploidy-confirmed multigenomic backgrounds. Plant Breed. 2013, 132, 211–216. [Google Scholar] [CrossRef]
- Plazas, M.; Vilanova, S.; Gramazio, P.; Rodríguez-Burruezo, A.; Fita, A.; Herraiz, F.J.; Ranil, R.; Fonseka, R.; Niran, L.; Fonseka, H.; et al. Interspecific Hybridization between Eggplant and Wild Relatives from Different Genepools. J. Am. Soc. Hortic. Sci. 2016, 141, 34–44. [Google Scholar] [CrossRef] [Green Version]
- Marasek-Ciolakowska, A.; Nishikawa, T.; Shea, D.J.; Okazaki, K. Breeding of lilies and tulips—Interspecific hybridization and genetic background. Breed. Sci. 2018, 68, 35–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodin, M.M.; Zaitlin, D.; Naidu, R.A.; Lommel, S.A. Nicotiana benthamiana: Its History and Future as a Model for Plant–Pathogen Interactions. Mol. Plant-Microbe Interact. 2008, 21, 1015–1026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bombarely, A.; Rosli, H.G.; Vrebalov, J.; Moffett, P.; Mueller, L.A.; Martin, G.B. A Draft Genome Sequence of Nicotiana benthamiana to Enhance Molecular Plant-Microbe Biology Research. Mol. Plant-Microbe Interact. 2012, 25, 1523–1530. [Google Scholar] [CrossRef] [Green Version]
- Knapp, S.; Chase, M.W.; Clarkson, J.J. Nomenclatural changes and a new sectional classification in Nicotiana (Solanaceae). Taxon 2004, 53, 73–82. [Google Scholar] [CrossRef]
- Goodspeed, T.H. The Genus Nicotiana; Chronica Botanica Company: Waltham, MA, USA, 1954. [Google Scholar]
- Marks, C.E.; Ladiges, P.Y.; Newbigin, E. Karyotypic variation in Nicotiana section Suaveolentes. Genet. Resour. Crop. Evol. 2011, 58, 797–803. [Google Scholar] [CrossRef]
- He, H.; Iizuka, T.; Maekawa, M.; Sadahisa, K.; Morikawa, T.; Yanase, M.; Yokoi, S.; Oda, M.; Tezuka, T. Nicotiana suaveolens accessions with different ploidy levels exhibit different reproductive isolation mechanisms in interspecific crosses with Nicotiana tabacum. J. Plant Res. 2019, 132, 461–471. [Google Scholar] [CrossRef]
- Dodsworth, S.; Christenhusz, M.J.M.; Conran, J.G.; Guignard, M.S.; Knapp, S.; Struebig, M.; Leitch, A.R.; Chase, M.W. Extensive plastid-nuclear discordance in a recent radiation of Nicotiana section Suaveolentes (Solanaceae). Bot. J. Linn. Soc. 2020, 193, 546–559. [Google Scholar] [CrossRef]
- Kawaguchi, K.; Ohya, Y.; Maekawa, M.; Iizuka, T.; Hasegawa, A.; Shiragaki, K.; He, H.; Oda, M.; Morikawa, T.; Yokoi, S.; et al. Two Nicotiana occidentalis accessions enable gene identification for Type II hybrid lethality by the cross to N. sylvestris. Sci. Rep. 2021, 11, 1–8. [Google Scholar] [CrossRef]
- Clarkson, J.J.; Dodsworth, S.; Chase, M.W. Time-calibrated phylogenetic trees establish a lag between polyploidisation and diversification in Nicotiana (Solanaceae). Plant Syst. Evol. 2017, 303, 1001–1012. [Google Scholar] [CrossRef]
- Burk, L.G.; Heggestad, H.E. The genusNicotiana: A source of resistance to diseases of cultivated tobacco. Econ. Bot. 1966, 20, 76–88. [Google Scholar] [CrossRef]
- Gillham, F.; Wark, D.; Harrigan, E. Disease resistant flue-cured tobacco breeding lines for north Queensland I. Resistance to blue mould, Peronospora tabacina. Aust. J. Exp. Agric. 1977, 17, 652–658. [Google Scholar] [CrossRef]
- Dijk, P.; Cuperus, C. Reactions on Nicotiana species to potato viruses A, X and Y and tobacco mosaic virus in relation to their taxonomy and geographical origin. Neth. J. Plant Pathol. 1989, 95, 343–356. [Google Scholar] [CrossRef]
- Tezuka, T.; Kuboyama, T.; Matsuda, T.; Marubashi, W. Seven of eight species in Nicotiana section Suaveolentes have common factors leading to hybrid lethality in crosses with Nicotiana tabacum. Ann. Bot. 2010, 106, 267–276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tezuka, T. Hybrid lethality in Nicotiana: A review with special attention to interspecific crosses between species in sect. Suaveolentes and N. tabacum. In Herbaceous Plants: Cultivation Methods, Grazing and Environmental Impacts; Wallner, F., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 69–94. [Google Scholar]
- Shiragaki, K.; Nakamura, R.; Nomura, S.; He, H.; Yamada, T.; Marubashi, W.; Oda, M.; Tezuka, T. Phenylalanine ammonia-lyase and phenolic compounds are related to hybrid lethality in the cross Nicotiana suaveolens × N. tabacum. Plant Biotechnol. 2020, 37, 327–333. [Google Scholar] [CrossRef]
- Tezuka, T.; Kitamura, N.; Imagawa, S.; Hasegawa, A.; Shiragaki, K.; He, H.; Yanase, M.; Ogata, Y.; Morikawa, T.; Yokoi, S. Genetic Mapping of the HLA1 Locus Causing Hybrid Lethality in Nicotiana Interspecific Hybrids. Plants 2021, 10, 2062. [Google Scholar] [CrossRef] [PubMed]
- Iizuka, T.; Kuboyama, T.; Marubashi, W.; Oda, M.; Tezuka, T. Nicotiana debneyi has a single dominant gene causing hybrid lethality in crosses with N. tabacum. Euphytica 2012, 186, 321–328. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Wheeler, H.-M. A Contribution to the Cytology of the Australian-South Pacific Species of Nicotiana. Proc. Natl. Acad. Sci. USA 1945, 31, 177–185. [Google Scholar] [CrossRef] [Green Version]
- Takenaka, Y. Cytogenetic studies of Nicotiana, V. Reduction divisions in hybrids Between. Bot. Mag. Tokyo 1953, 66, 269–276. [Google Scholar] [CrossRef]
- Goodspeed, T.H.; Thompson, M.C. Cytotaxonomy of Nicotiana. II. Bot. Rev. 1959, 25, 385–415. [Google Scholar] [CrossRef]
- Gopinath, D.M.; Krishnamurthy, K.V.; Krishnamurthy, A.S. Cytological studies on interspecific hybrids in nicotiana involving a new Australian species, Nicotiana amplexicaulis. Can. J. Genet. Cytol. 1965, 7, 328–340. [Google Scholar] [CrossRef]
- Williams, E. Meiotic Chromosome Pairing in Interspecific Hybrids of Nicotiana. N. Z. J. Bot. 1975, 13, 601–609. [Google Scholar] [CrossRef] [Green Version]
- Gerstel, D.U.; Burns, J.A.; Burk, L.G. Interspecific hybridizations with an African tobacco, Nicotiana africana Merxm. J. Hered. 1979, 70, 342–344. [Google Scholar] [CrossRef]
- Gangadevi, T.; Rao, P.N.; Satyanarayana, K.V. Cytogenetic study of an interspecific cross of Nicotiana debneyi X N. umbratica. Theor. Appl. Genet. 1982, 63, 177–181. [Google Scholar] [CrossRef]
- Gangadevi, T.; Rao, P.N.; Satyanarayana, K.V. Morphological and cytological studies of interspecific hybrids in Nicotiana involving N. umbratica Burbidge. Cytologia 1987, 52, 475–486. [Google Scholar] [CrossRef] [Green Version]
- Kubo, T.; Kumashiro, T.; Saito, Y. Cytoplasmic male sterile lines of a tobacco variety, Tsukuba 1, developed by asymmetric protoplast fusion. Jpn. J. Breed. 1988, 38, 158–164. [Google Scholar] [CrossRef]
- Rieseberg, L.H.; Blackman, B.K. Speciation genes in plants. Ann. Bot. 2010, 106, 439–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stathos, A.; Fishman, L. Chromosomal rearrangements directly cause underdominant F1 pollen sterility in Mimulus lewisii–Mimulus cardinalis hybrids. Evolution 2014, 68, 3109–3119. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.E.Z.; Lin, H.-X. Evolution and Molecular Control of Hybrid Incompatibility in Plants. Front. Plant Sci. 2016, 7, 1208. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Zhou, J.; Zhang, Y.; Yang, Y.; Pu, Q.; Tao, D. New Insights Into the Nature of Interspecific Hybrid Sterility in Rice. Front. Plant Sci. 2020, 11, 555572. [Google Scholar] [CrossRef] [PubMed]
- Kelly, L.J.; Leitch, A.R.; Clarkson, J.J.; Knapp, S.; Chase, M.W. Reconstructing the complex evolutionary origin of wild allopolyploid tobaccos (Nicotiana section Suaveolentes). Evolution 2013, 67, 80–94. [Google Scholar] [CrossRef] [PubMed]
- Gangadevi, T.; Rao, P.N.; Satyanarayana, K.V. Cytogenetic Studies of Some Synthetic Amphiploids of Nicotiana. J. Hered. 1988, 79, 119–122. [Google Scholar] [CrossRef]
- Mariam, A.L.; Zakri, A.H.; Mahani, M.C.; Normah, M.N. Interspecific hybridization of cultivated rice, Oryza sativa L. with the wild rice, O. minuta Presl. Theor. Appl. Genet. 1996, 93, 664–671. [Google Scholar] [CrossRef]
- Chetelat, R.T. Overcoming sterility and unilateral incompatibility of Solanum lycopersicum × S. sitiens hybrids. Euphytica 2016, 207, 319–330. [Google Scholar] [CrossRef]
- Manzoor, A.; Ahmad, T.; Bashir, M.A.; Hafiz, I.A.; Silvestri, C. Studies on Colchicine Induced Chromosome Doubling for Enhancement of Quality Traits in Ornamental Plants. Plants 2019, 8, 194. [Google Scholar] [CrossRef] [Green Version]


| Cross Combination | No. of Flowers Pollinated | No. of Capsules Obtained | No. of Seeds Sown | No. of Hybrids Obtained |
|---|---|---|---|---|
| N. benthamiana JT × N. excelsior JT | 40 | 10 (25% 1) | 178 | 2 (1% 2) |
| N. benthamiana JT × N. excelsior PI 224063 | 11 | 2 (18%) | 48 | 0 (0%) |
| N. benthamiana JT × N. excelsior PI 555685 | 5 | 1 (20%) | 2 | 0 (0%) |
| N. benthamiana PI 555478 × N. excelsior JT | 20 | 0 (0%) | – | – |
| N. benthamiana PI 555478 × N. excelsior PI 224063 | 13 | 1 (8%) | 22 | 0 (0%) |
| N. benthamiana PI 555478 × N. excelsior PI 555685 | 20 | 1 (5%) | 143 | 0 (0%) |
| N. benthamiana PI 555684 × N. excelsior JT | 20 | 2 (%) | 27 | 9 (33%) |
| N. benthamiana PI 555684 × N. excelsior PI 224063 | 5 | 1 (20%) | 48 | 0 (0%) |
| N. benthamiana PI 555684 × N. excelsior PI 555685 | 20 | 0 (0%) | – | – |
| N. excelsior JT × N. benthamiana JT | 18 | 1 (6%) | 32 | 28 (88%) |
| N. excelsior JT × N. benthamiana PI 555478 | 20 | 0 (0%) | – | – |
| N. excelsior JT × N. benthamiana PI 555684 | 9 | 1 (11%) | 17 | 3 (18%) |
| N. excelsior PI 224063 × N. benthamiana JT | 17 | 2 (12%) | 23 | 22 (96%) |
| N. excelsior PI 224063 × N. benthamiana PI 555478 | 17 | 2 (12%) | 20 | 11 (55%) |
| N. excelsior PI 224063 × N. benthamiana PI 555684 | 6 | 2 (33%) | 20 | 17 (85%) |
| N. excelsior PI 555685 × N. benthamiana JT | 3 | 1 (33%) | 31 | 29 (94%) |
| N. excelsior PI 555685 × N. benthamiana PI 555478 | 22 | 0 (0%) | – | – |
| N. excelsior PI 555685 × N. benthamiana PI 555684 | 2 | 1 (50%) | 34 | 32 (94%) |
| Cross Combination | Total no. of Pollens Observed | Percentage of Pollen Viability |
|---|---|---|
| N. benthamiana JT × N. excelsior JT | 368 | 0 |
| N. benthamiana PI 555684 × N. excelsior JT | 326 | 0 |
| N. excelsior JT × N. benthamiana JT | 348 | 0 |
| N. excelsior JT × N. benthamiana PI 555684 | 335 | 0 |
| N. excelsior PI 224063 × N. benthamiana JT | 331 | 0 |
| N. excelsior PI 224063 × N. benthamiana PI 555478 | 331 | 0 |
| N. excelsior PI 224063 × N. benthamiana PI 555684 | 360 | 0 |
| N. excelsior PI 555685 × N. benthamiana JT | 313 | 0 |
| N. excelsior PI 555685 × N. benthamiana PI 555684 | 318 | 0 |
| Cross Combination | No. of Flowers Pollinated | No. of Capsules Obtained |
|---|---|---|
| (N. benthamiana JT × N. excelsior JT) × N. benthamiana JT | 20 | 0 |
| (N. benthamiana JT × N. excelsior JT) × N. excelsior JT | 20 | 0 |
| (N. benthamiana PI 555684 × N. excelsior JT) × N. benthamiana PI 555684 | 20 | 0 |
| (N. benthamiana PI 555684 × N. excelsior JT) × N. excelsior JT | 20 | 0 |
| (N. excelsior JT × N. benthamiana JT) × N. benthamiana JT | 20 | 0 |
| (N. excelsior JT × N. benthamiana JT) × N. excelsior JT | 20 | 0 |
| (N. excelsior JT × N. benthamiana PI 555684) × N. benthamiana PI 555684 | 20 | 0 |
| (N. excelsior JT × N. benthamiana PI 555684) × N. excelsior JT | 20 | 0 |
| (N. excelsior PI 224063 × N. benthamiana JT) × N. benthamiana JT | 20 | 0 |
| (N. excelsior PI 224063 × N. benthamiana JT) × N. excelsior PI 224063 | 20 | 0 |
| (N. excelsior PI 224063 × N. benthamiana PI 555478) × N. benthamiana PI 555478 | 20 | 0 |
| (N. excelsior PI 224063 × N. benthamiana PI 555478) × N. excelsior PI 224063 | 20 | 0 |
| (N. excelsior PI 224063 × N. benthamiana PI 555684) × N. benthamiana PI 555684 | 23 | 0 |
| (N. excelsior PI 224063 × N. benthamiana PI 555684) × N. excelsior PI 224063 | 20 | 0 |
| (N. excelsior PI 555685 × N. benthamiana JT) × N. benthamiana JT | 20 | 0 |
| (N. excelsior PI 555685 × N. benthamiana JT) × N. excelsior PI 555685 | 20 | 0 |
| (N. excelsior PI 555685 × N. benthamiana PI 555684) × N. benthamiana PI 555684 | 20 | 0 |
| (N. excelsior PI 555685 × N. benthamiana PI 555684) × N. excelsior PI 555685 | 20 | 0 |
| Cross Combination | No. of Flowers Pollinated | No. of Capsules Obtained |
|---|---|---|
| N. benthamiana JT × (N. benthamiana JT × N. excelsior JT) | 20 | 0 |
| N. excelsior JT × (N. benthamiana JT × N. excelsior JT) | 14 | 0 |
| N. benthamiana JT × (N. excelsior JT × N. benthamiana JT) | 20 | 0 |
| N. excelsior JT × (N. excelsior JT × N. benthamiana JT) | 20 | 0 |
| N. benthamiana JT × (N. excelsior PI 555685 × N. benthamiana JT) | 20 | 0 |
| N. excelsior PI 555685 × (N. excelsior PI 555685 × N. benthamiana JT) | 20 | 0 |
| N. benthamiana PI 555684 × (N. excelsior PI 555685 × N. benthamiana PI 555684) | 10 | 0 |
| N. excelsior PI 555685 × (N. excelsior PI 555685 × N. benthamiana PI 555684) | 20 | 0 |
| Cross Combination | Mean Chromosome Configuration Per Cell | |||
|---|---|---|---|---|
| Univalent | Bivalent | Trivalent | Tetravalent | |
| N. benthamiana JT × N. excelsior JT | 3.50 ± 0.32 | 12.80 ± 0.25 | 2.80 ± 0.18 | 0.12 ± 0.07 |
| N. excelsior JT × N. benthamiana JT | 2.80 ± 0.21 | 14.04 ± 0.24 | 2.44 ± 0.15 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tezuka, T.; Kitamura, N.; Yanase, M.; Morikawa, T. Evaluation of Crossability between Nicotiana benthamiana and Nicotiana excelsior. Agronomy 2021, 11, 2583. https://doi.org/10.3390/agronomy11122583
Tezuka T, Kitamura N, Yanase M, Morikawa T. Evaluation of Crossability between Nicotiana benthamiana and Nicotiana excelsior. Agronomy. 2021; 11(12):2583. https://doi.org/10.3390/agronomy11122583
Chicago/Turabian StyleTezuka, Takahiro, Naoto Kitamura, Masanori Yanase, and Toshinobu Morikawa. 2021. "Evaluation of Crossability between Nicotiana benthamiana and Nicotiana excelsior" Agronomy 11, no. 12: 2583. https://doi.org/10.3390/agronomy11122583
APA StyleTezuka, T., Kitamura, N., Yanase, M., & Morikawa, T. (2021). Evaluation of Crossability between Nicotiana benthamiana and Nicotiana excelsior. Agronomy, 11(12), 2583. https://doi.org/10.3390/agronomy11122583






