Gram-Negative Bacteria from Organic and Conventional Agriculture in the Hydrographic Basin of Loja: Quality or Pathogen Reservoir?
Abstract
:1. Introduction
2. Materials and Methods
2.1. SamplingArea
- Organic: farms that apply organic fertilizers such as poultry manure and/or hand-processed compost to the agricultural soil and avoid or restrict the use of chemical compounds for soil fertilization and plague control.
- Conventional: farms that apply commercial chemical fertilizers (e.g., N10-P30-K10, urea 46%, ammonium nitrate 36%) and plague control (e.g., metaldehyde; chlorpyrifos) for agricultural management. All conventional farms also use animal manure as a base fertilizer before applying chemical treatments according to the particular needs of each farm.
2.2. Experimental Design and Sampling
2.3. Isolation and Biochemical Identification of Bacterial Strains
2.4. Data Analysis
3. Results
Biochemical Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Uusitalo, V.; Kuokkanen, A.; Grönman, K.; Ko, N.; Mäkinen, H.; Koistinen, K. Environmental Sustainability Assessment from Planetary Boundaries Perspective—A Case Study of an Organic Sheep Farm in Finland. Sci. Total Environ. 2019, 687, 168–176. [Google Scholar] [CrossRef]
- Foley, J.; Ramankutty, N.; Brauman, K.; Cassidy, E.; Gerber, J.; Johnston, M.; Mueller, N.; O’Connell, C.; Ray, D.; West, P.; et al. Solutions for a Cultivated Planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef] [Green Version]
- Nain, L.; Gaind, S.; Pandey, A.; Grover, M.; Sreekanthan, S.; Vasudevan, V. Practical Applications of Bioactive Compost in Organic Agriculture. In Agriculturally Important Microorganisms; Academic Word International: Bhopal, India, 2009; pp. 225–246. [Google Scholar]
- Lans, C. Worldwide Opportunities on Organic Farms (WWOOF) as Part of the Existing Care Economy in Canada. Geoforum 2016, 75, 16–19. [Google Scholar] [CrossRef] [Green Version]
- Fan, K.; Delgado-Baquerizo, M.; Guo, X.; Wang, D.; Zhu, Y.; Chu, H. Microbial Resistance Promotes Plant Production in a Four-Decade Nutrient Fertilization Experiment. Soil Biol. Biochem. 2020, 141, 107679. [Google Scholar] [CrossRef]
- Hellequin, E.; Monard, C.; Quaiser, A.; Henriot, M.; Klarzynski, O.; Binet, F. Specific Recruitment of Soil Bacteria and Fungi Decomposers Following a Biostimulant Application Increased Crop Residues Mineralization. PLoS ONE 2018, 13, e0209089. [Google Scholar] [CrossRef]
- Jiménez, L.; Jiménez, W.; Felicito, D.; Fierro, N.; Quichimbo, P.; Sánchez, D.; Capa-Mora, D. Rediscovering the edaphic knowledge of smallholder farmers in southern Ecuador. Geoderma 2021, 406, 115468. [Google Scholar] [CrossRef]
- Barrowclough, M.; Stehouwer, R.; Alwang, J.; Gallagher, R.; Mosquera, V.H.B.; Dominguez, J.M. Conservation agriculture on steep slopes in the Andes: Promise and obstacles. J. Soil Water Conserv. 2016, 71, 91–102. [Google Scholar] [CrossRef]
- Sanz-Cobena, A.; Lassaletta, L.; Estellés, F.; Del Prado, A.; Guardia, G.; Abalos, D.; Aguilera, E.; Pardo, G.; Vallejo, A.; Sutton, M.; et al. Yield-Scaled Mitigation of Ammonia Emission from N Fertilization: The Spanish Case. Environ. Res. Lett. 2014, 9, 125005. [Google Scholar] [CrossRef]
- Yuan, H.; Ge, T.; Chen, C.; O’Donnell, A.; Wu, J. Significant Role for Microbial Autotrophy in the Sequestration of Soil Carbon. Appl. Environ. Microbiol. 2012, 78, 2328–2336. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.; He, J.; Quan, Z.; Wu, C.; Sheng, R.; Zhang, L.; Geisen, S. Fertilization Changes Soil Microbiome Functioning, Especially Phagotrophic Protists. Soil Biol. Biochem. 2020, 148, 107863. [Google Scholar] [CrossRef]
- Delgado-Baquerizo, M.; Eldridge, D.; Ochoa, V.; Gozalo, B.; Singh, B.; Maestre, F. Soil Microbial Communities Drive the Resistance of Ecosystem Multifunctionality to Global Change in Drylands Across the Globe. Ecol. Lett. 2017, 20, 1295–1305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konopka, A. Ecology microbial. In Encyclopedia of Microbiology, 4th ed.; Schaechter, M., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 97–111. [Google Scholar]
- Nihotimbere, V.; Ongena, M.; Smargiassi, M.; Thonart, P. Beneficial Effect of the Rhizosphere Microbial Community for Plant Growth and Health. Biotechnol. Agron. Soc. Environ. 2011, 15, 327–337. [Google Scholar]
- Staley, Z.; He, D.; Edge, T. Persistence of Fecal Contamination and Pathogenic Escherichia coli O157:H7 In Snow and Snowmelt. J. Great Lakes Res. 2017, 43, 248–254. [Google Scholar] [CrossRef]
- Uprety, S.; Dangol, B.; Nakarmi, P.; Dhakal, I.; Sherchan, S.; Shisler, J.; Jutla, A.; Amarasiri, M.; Sano, D.; Nguyen, T. Assessment of Microbial Risks by Characterization of Escherichia coli Presence to Analyze the Public Health Risks from Poor Water Quality in Nepal. Int. J. Hyg. Environ. Health 2020, 226, 113484. [Google Scholar] [CrossRef]
- Heaton, J.; Jones, K. Microbial Contamination of Fruit and Vegetables and the Behavior of Enteropathogens in the Phyllosphere: A Review. J. Appl. Microbiol. 2008, 104, 613–626. [Google Scholar] [CrossRef]
- Land & Water|Food and Agriculture Organization of the United Nations|FAO. Available online: http://www.fao.org/nr/water/topics_scarc_agri.html (accessed on 4 May 2021).
- Matthews, K. Leafy vegetable. In The Produce Contamination Problem. Causes and Solutions, 2nd ed.; Academic Press: San Diego, CA, USA, 2014; pp. 187–206. [Google Scholar]
- Slayton, R.; Turabelidze, G.; Bennett, S.; Schwensohn, C.; Yaffee, A.; Khan, F.; Butler, C.; Trees, E.; Ayers, T.; Davis, M.; et al. Outbreak of Shiga Toxin-Producing Escherichia coli (STEC) O157:H7 Associated with Romaine Lettuce Consumption, 2011. PLoS ONE 2013, 8, e55300. [Google Scholar] [CrossRef] [Green Version]
- Szczech, M.; Kowalska, B.; Smolińska, U.; Maciorowski, R.; Oskiera, M.; Michalska, A. Microbial Quality of Organic and Conventional Vegetables from Polish Farms. Int. J. Food Microbiol. 2018, 286, 155–161. [Google Scholar] [CrossRef]
- Yaron, S. Microbial Attachment and Persistence on Plants. In The Produce Contamination Problem, 2nd ed.; Academic Press: San Diego, CA, USA, 2014; pp. 21–57. [Google Scholar]
- Ruiz-Pico, Á.; Pérez-Cuenca, Á.; Serrano-Agila, R.; Maza-Criollo, D.; Leiva-Piedra, J.; Salazar-Campos, J. Hydrochemical Characterization of Groundwater in the Loja Basin (Ecuador). Appl. Geochem. 2019, 104, 1–9. [Google Scholar] [CrossRef]
- Hualpa, D.; Toledo, Z.; Meneses, M.; Feng, P. Microbiological Quality of Minimally Processed, Ready-to-Eat, Vegetables in Loja, Ecuador. Rev. Politécnica 2018, 41, 1–6. [Google Scholar]
- Villa-Achupallas, M.; Rosado, D.; Aguilar, S.; Galindo-Riaño, M. Water Quality in The Tropical Andes Hotspot: The Yacuambi River (Southeastern Ecuador). Sci. Total Environ. 2018, 633, 50–58. [Google Scholar] [CrossRef]
- Aijuka, M.; Buys, E. Persistence of Foodborne Diarrheagenic Escherichia coli in the Agricultural and Food Production Environment: Implications for Food Safety and Public Health. Food Microbiol. 2019, 82, 363–370. [Google Scholar] [CrossRef]
- Msolo, L.; Iweriebor, B.; Okoh, A. Pervasiveness of Diarrheagenic E. coli Pathotypes and Salmonella Species Among Gastroenteritis Patients in Some Selected Pastoral Hinterlands of the Amathole District Municipality, Eastern Cape, South Africa. Hum. Microbiome J. 2020, 17, 100074. [Google Scholar] [CrossRef]
- Shekar, A.; Babu, L.; Ramlal, S.; Sripathy, M.; Batra, H. Selective and Concurrent Detection of Viable Salmonella spp., E. coli, Staphylococcus aureus, E. coli O157:H7, and Shigella spp. in Low Moisture Food Products By PMA-Mpcr Assay with Internal Amplification Control. LWT 2017, 86, 586–593. [Google Scholar] [CrossRef]
- Zhi, S.; Stothard, P.; Banting, G.; Scott, C.; Huntley, K.; Ryu, K.; Otto, S.; Ashbolt, N.; Checkley, S.; Dong, T.; et al. Characterization of Water Treatment-Resistant and Multidrug-Resistant Urinary Pathogenic Escherichia coli in Treated Wastewater. Water Res. 2020, 182, 115827. [Google Scholar] [CrossRef]
- Ochoa-Cueva, P.; Fries, A.; Montesinos, P.; Rodríguez-Díaz, J.A.; Boll, J. Spatial estimation of soil erosion risk by land-cover change in the Andes of southern Ecuador. Land Degrad. Dev. 2015, 26, 565–573. [Google Scholar] [CrossRef]
- Chuquimarca, L.; Gaona, F.P.; Iñiguez-Armijos, C.; Benítez, Á. Lichen Responses to Disturbance: Clues for Biomonitoring Land-use Effects on Riparian Andean Ecosystems. Diversity 2019, 11, 73. [Google Scholar] [CrossRef] [Green Version]
- MacFaddin, J. Pruebas Bioquímicas para la Identificación de Bacterias de Importancia Clínica, 3rd ed.; Médica Panamericana: Madrid, Spain, 2003. [Google Scholar]
- Mota, R.; da Silva, L.; da Silva, K.; da Silva Neto, J.; da Cunha, A.; do Nascimento Sobrinho, E. Caracterización Bioquímica y Perfil de Sensibilidad Antimicrobiana in Vitro de Muestras de Burkholderia mallei Aisladas de Équidos de la Región Nordeste de Brasil. Arq. Inst. Biol 2005, 72, 7–11. [Google Scholar]
- Mayz, J.; Manzi, L. Bacterias Hidrocarburoclásticas del Género Pseudomonas en la Rizosfera de Samanea Saman (Jacq.) Merr. Rev. Colomb. De Biotecnol. 2017, 19, 29–37. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.; O’Hara, R.; Simpson, G.; Solymos, P.; et al. Package “Vegan”. Community Ecology. 2021. Available online: https://github.com/vegandevs/vegan (accessed on 22 September 2021).
- Inglis, T.; Merritt, A. Burkholderia pseudomallei and Burkholderia mallei. Mol. Med. Microbiol. 2015, 2, 769–791. [Google Scholar]
- Bhar, S.; Edelmann, M.; Jones, M. Characterization and Proteomic Analysis of Outer Membrane Vesicles from a Commensal Microbe, Enterobacter cloacae. J. Proteom. 2021, 231, 103994. [Google Scholar] [CrossRef] [PubMed]
- Bhise, K.; Bhagwat, P.; Dandge, P. Plant Growth-Promoting Characteristics of Salt Tolerant Enterobacter Cloacae Strain KBPD and its Efficacy in Amelioration of Salt Stress in Vigna Radiata L. J. Plant Growth Regul. 2016, 36, 215–226. [Google Scholar] [CrossRef]
- Ji, C.; Liu, Z.; Hao, L.; Song, X.; Wang, C.; Liu, Y.; Li, H.; Li, C.; Gao, Q.; Liu, X. Effects of Enterobacter cloacae HG-1 on the Nitrogen-Fixing Community Structure of Wheat Rhizosphere Soil and on Salt Tolerance. Front. Plant Sci. 2020, 11, 1094. [Google Scholar] [CrossRef]
- Cooney, S.; O’Brien, S.; Iversen, C.; Fanning, S. Bacteria: Other Pathogenic Enterobacteriaceae—Enterobacter and other Genera. Encycl. Food Saf. 2014, 1, 433–441. [Google Scholar]
- Collavino, M.; Sansberro, P.; Mroginski, L.; Aguilar, O. Comparison of in Vitro Solubilization Activity of Diverse Phosphate-Solubilizing Bacteria Native to Acid Soil and their Ability to Promote Phaseolus vulgaris Growth. Biol. Fertil. Soils 2010, 46, 727–738. [Google Scholar] [CrossRef]
- Sindhu, S.; Phour, M.; Choudhary, S.; Chaudhary, D. Phosphorus Cycling: Prospects of Using Rhizosphere Microorganisms for Improving Phosphorus Nutrition of Plants. Geomicrobiol. Biogeochem. 2013, 39, 199–237. [Google Scholar]
- Thapa, L.; Lee, S.; Park, C.; Kim, S. Metabolic Engineering of Enterobacter aerogenes to Improve the Production of 2,3-Butanediol. Biochem. Eng. J. 2019, 143, 169–178. [Google Scholar] [CrossRef]
- Samanta, I.; Bandyopadhyay, S.; Bhatia, R. Antimicrobial Resistance in Agriculture; Academic Press: London, UK; San Diego, CA, USA, 2020; pp. 171–193. [Google Scholar]
- Taabodi, M.; Hashem, F.; Oscar, T.; Parveen, S.; May, E. The Possible Roles of Escherichia coli in the Nitrogen Cycle. Int. J. Environ. Res. 2019, 13, 597–602. [Google Scholar] [CrossRef] [Green Version]
- Pavlova, A.; Leontieva, M.; Smirnova, T.; Kolomeitseva, G.; Netrusov, A.; Tsavkelova, E. Colonization Strategy of the Endophytic Plant Growth-Promoting Strains of Pseudomonas fluorescens and Klebsiella oxytoca on the Seeds, Seedlings and Roots of the Epiphytic Orchid, Dendrobium nobile Lindl. J. Appl. Microbiol. 2017, 123, 217–232. [Google Scholar] [CrossRef] [PubMed]
- Celloto, V.; Oliveira, A.; Gonçalves, J.; Watanabe, C.; Matioli, G.; Gonçalves, R. Biosynthesis of Indole-3-Acetic Acid by New Klebsiella oxytoca free and Immobilized Cells on Inorganic Matrices. Sci. World J. 2012, 2012, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Pongsapipatana, N.; Damrongteerapap, P.; Chantorn, S.; Sintuprapa, W.; Keawsompong, S.; Nitisinprasert, S. Molecular Cloning of Kman Coding for Mannanase from Klebsiella oxytoca KUB-CW2-3 And Its Hybrid Mannanase Characters. Enzym. Microb. Technol. 2016, 89, 39–51. [Google Scholar] [CrossRef]
- Tominaga, T. Rapid Detection of Klebsiella pneumoniae, Klebsiella oxytoca, Raoultella ornithinolytica and Other Related Bacteria in Food by Lateral-Flow Test Strip Immunoassays. J. Microbiol. Methods 2018, 147, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Asis, C.; Adachi, K. Isolation of Endophytic Diazotroph Pantoea agglomerans and Nondiazotroph Enterobacter asburiae from Sweetpotato Stem in Japan. Lett. Appl. Microbiol. 2004, 38, 19–23. [Google Scholar] [CrossRef]
- Völksch, B.; Thon, S.; Jacobsen, I.; Gube, M. Polyphasic Study of Plant- and Clinic-Associated Pantoea agglomerans Strains Reveals Indistinguishable Virulence Potential. Infect. Genet. Evol. 2009, 9, 1381–1391. [Google Scholar] [CrossRef]
- Delgado-Bermúdez, A.; Bonet, S.; Yeste, M.; Pinart, E. Long-Term Storage of Boar Seminal Doses Contaminated with Proteus vulgaris: A Dose-Dependent Effect on Sperm Motility and Sperm-Bacteria Interaction. Anim. Reprod. Sci. 2020, 216, 106349. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Sun, Y.; Jiang, M.; Luo, L.; Yu, X.; Yao, W.; Wu, Z. Isolation, Identification and Pathogenicity of Proteus vulgaris from Moribund Common Carp (Cyprinus carpio) Farmed in China. Aquaculture 2020, 525, 735294. [Google Scholar] [CrossRef]
- Yu, S.; Lee, Y. Plant Growth Promoting Rhizobacterium Proteus vulgaris JBLS202 Stimulates the Seedling Growth of Chinese Cabbage through Indole Emission. Plant Soil 2013, 370, 485–495. [Google Scholar] [CrossRef]
- Radhapriya, P.; Ramachandran, A.; Anandham, R.; Mahalingam, S. Pseudomonas aeruginosa RRALC3 Enhances the Biomass, Nutrient and Carbon Contents of Pongamia pinnata Seedlings in Degraded Forest Soil. PLoS ONE 2015, 10, e139881. [Google Scholar] [CrossRef] [PubMed]
- Schinner, S.; Engelhardt, F.; Preusse, M.; Thöming, J.; Tomasch, J.; Häussler, S. Genetic Determinants of Pseudomonas aeruginosa Fitness During Biofilm Growth. Biofilm 2020, 2, 100023. [Google Scholar] [CrossRef]
- Tripathy, S.; Kumar, N.; Mohanty, S.; Samanta, M.; Mandal, R.; Maiti, N. Characterisation of Pseudomonas aeruginosa Isolated from Freshwater Culture Systems. Microbiol. Res. 2007, 162, 391–396. [Google Scholar] [CrossRef]
- Andreani, N.; Carraro, L.; Zhang, L.; Vos, M.; Cardazzo, B. Transposon Mutagenesis in Pseudomonas fluorescens Reveals Genes Involved in Blue Pigment Production and Antioxidant Protection. Food Microbiol. 2019, 82, 497–503. [Google Scholar] [CrossRef]
- Ortiz-Castro, R.; Campos-García, J.; López-Bucio, J. Pseudomonas putida and Pseudomonas fluorescens Influence Arabidopsis Root System Architecture through an Auxin Response Mediated by Bioactive Cyclodipeptides. J. Plant Growth Regul. 2019, 39, 254–265. [Google Scholar] [CrossRef]
- Nkoh, J.; Yan, J.; Xu, R.; Shi, R.; Hong, Z. The Mechanism for Inhibiting Acidification of Variable Charge Soils by Adhered Pseudomonas fluorescens. Environ. Pollut. 2020, 260, 114049. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, I.; Shahid Shaukat, S. Suppression of Root-Knot Disease by Pseudomonas fluorescens CHA0 in Tomato: Importance of Bacterial Secondary Metabolite, 2,4-Diacetylpholoroglucinol. Soil Biol. Biochem. 2003, 35, 1615–1623. [Google Scholar] [CrossRef]
- Patel, A.; Jeyasekaran, G.; Jeyashakila, R.; Anand, T.; Wilwet, L.; Pathak, N.; Malini, A.; Neethiselvan, N. Prevalence of Antibiotic Resistant Salmonella spp. Strains in Shrimp Farm Source Waters of Nagapattinam Region in South India. Mar. Pollut. Bull. 2020, 155, 111171. [Google Scholar] [CrossRef]
- Krzyzanowski, F.; de Souza Lauretto, M.; Nardocci, A.; Sato, M.; Razzolini, M. Assessing the Probability of Infection by Salmonella due to Sewage Sludge Use in Agriculture Under Several Exposure Scenarios for Crops and Soil Ingestion. Sci. Total Environ. 2016, 568, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Kim, D.; Kim, B.; Chi, Y.; Kang, S.; Park, H.; Jung, S.; Lee, J.; Oh, T. Complete Genome Sequencing of Shigella sp. PAMC 28760: Identification of Cazyme Genes and Analysis of their Potential Role in Glycogen Metabolism for Cold Survival Adaptation. Microb. Pathog. 2019, 137, 103759. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Yan, P.; Xiong, W.; Wang, J.; Liu, X. Genomic Characterization of a Novel Virulent Phage Infecting Shigella fiexneri and Isolated from Sewage. Virus Res. 2020, 283, 197983. [Google Scholar] [CrossRef]
- Jansson, J.; Hofmockel, K. The Soil Microbiome—From Metagenomics to Metaphenomics. Curr. Opin. Microbiol. 2018, 43, 162–168. [Google Scholar] [CrossRef]
- Jacoby, R.; Peukert, M.; Succurro, A.; Koprivova, A.; Kopriva, S. The Role of Soil Microorganisms in Plant Mineral Nutrition—Current Knowledge and Future Directions. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [Green Version]
- Shade, A. Diversity is the Question, not the Answer. ISME J. 2016, 11, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Rusiñol, M.; Hundesa, A.; Cárdenas-Youngs, Y.; Fernández-Bravo, A.; Pérez-Cataluña, A.; Moreno-Mesonero, L.; Moreno, Y.; Calvo, M.; Alonso, J.; Figueras, M.; et al. Microbiological Contamination of Conventional and Reclaimed Irrigation Water: Evaluation and Management Measures. Sci. Total Environ. 2020, 710, 136298. [Google Scholar] [CrossRef]
- Soonvald, L.; Loit, K.; Runno-Paurson, E.; Astover, A.; Tedersoo, L. The Role of Long-Term Mineral and Organic Fertilization Treatment in Changing Pathogen and Symbiont Community Composition in Soil. Appl. Soil Ecol. 2019, 141, 45–53. [Google Scholar] [CrossRef]
- Johannessen, G. Use of Manure in Production of Organic Lettuce; Norwegian School of Veterinary Science, National Veterinary Institute: Oslo, Norway, 2005. [Google Scholar]
- De Quadros, R.; Loiko, M.; Minéia Daniel de Paula, C.; Hessel, C.; Jacxsens, L.; Uyttendaele, M.; Bender, R.; Tondo, E. Microbiological Contamination Linked to Implementation of Good Agricultural Practices in The Production of Organic Lettuce in Southern Brazil. Food Control 2014, 42, 152–164. [Google Scholar] [CrossRef]
- Jechalke, S.; Schierstaedt, J.; Becker, M.; Flemer, B.; Grosch, R.; Smalla, K.; Schikora, A. Salmonella Establishment in Agricultural Soil and Colonization of Crop Plants Depend on Soil Type and Plant Species. Front. Microb. 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balkhair, K. Modeling Fecal Bacteria Transport and Retention in Agricultural and Urban Soils Under Saturated and Unsaturated Flow Conditions. Water Res. 2017, 110, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Gerba, C. The Role of Water and Water Testing in Produce Safety. In Microbial Safety of Fresh Produce; Wiley-Blackwell: Hoboken, NJ, USA, 2009; pp. 129–142. [Google Scholar] [CrossRef]
- Jiang, X.; Shepherd, M. The Role of Manure and Compost in Produce Safety. In Microbial Safety of Fresh Produce; Wiley-Blackwell: Hoboken, NJ, USA, 2009; pp. 143–166. [Google Scholar]
- De Corato, U.; Viola, E.; Arcieri, G.; Valerio, V.; Zimbardi, F. Use of Composted Agro-Energy Co-Products and Agricultural Residues Against Soil-Borne Pathogens in Horticultural Soil-Less Systems. Sci. Hortic. 2016, 210, 166–179. [Google Scholar] [CrossRef]
- Drzewiecka, D. Significance and Roles of Proteus spp. Bacteria in Natural Environments. Microb. Ecol. 2016, 72, 741–758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaparro-Acuña, S.; Becerra-Jiménez, M.; Martínez-Zambrano, J.; Rojas-Sarmiento, H. Soil Bacteria that Precipitate Calcium Carbonate: Mechanism and Applications of the Process. Acta Agronómica 2018, 67. [Google Scholar] [CrossRef] [Green Version]
- Ghobadi Nia, M.; Rahimi, H.; Sohrabi, T.; Naseri, A.; Tofighi, H. Potential Risk of Calcium Carbonate Precipitation in Agricultural Drain Envelopes in Arid and Semi-Arid Areas. Agric. Water Manag. 2010, 97, 1602–1608. [Google Scholar] [CrossRef]
- Cui, H.; Sun, W.; Delgado-Baquerizo, M.; Song, W.; Ma, J.; Wang, K.; Ling, X. The Effects of Mowing and Multi-Level N Fertilization on Soil Bacterial and Fungal Communities in a Semiarid Grassland are Year-Dependent. Soil Biol. Biochem. 2020, 151, 108040. [Google Scholar] [CrossRef]
- Bliven, K.; Lampel, K. Shigella in Foodborne Diseases, 3rd ed.; Dodd, C.E.R., Aldsworth, T., Stein, R.A., Cliver, D.O., Riemann, H., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 171–188. [Google Scholar] [CrossRef]
- Duffy, E.; Lucia, L.; Kells, J.; Castillo, A.; Pillai, S.; Acuff, G. Concentrations of Escherichia coli and Genetic Diversity and Antibiotic Resistance Profiling of Salmonella Isolated from Irrigation Water, Packing Shed Equipment, and Fresh Produce in Texas. J. Food Prot. 2005, 68, 70–79. [Google Scholar] [CrossRef]
- Googoolee, A.; Takooree, S.; Goburdhun, D.; Neetoo, H. Characterizing the Cultivation Practices and Microbiological Quality of Watercress. J. Agric. Food Res. 2020, 2, 100057. [Google Scholar] [CrossRef]
- Schroeder, G.; Hilbi, H. Molecular Pathogenesis of Shigella Spp.: Controlling Host Cell Signaling, Invasion, and Death by Type III Secretion. Clin. Microbiol. Rev. 2008, 21, 134–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saxena, A.; Pal, V.; Tripathi, N.; Goel, A. A Real-Time Loop Mediated Isothermal Amplification Assay for Molecular Detection of Burkholderia mallei, the Aetiological Agent of a Zoonotic and Re-Emerging Disease Glanders. Acta Tropica 2019, 194, 189–194. [Google Scholar] [CrossRef]
- Kanso, S.; Dasri, K.; Tingthong, S.; Watanapokasin, R. Diversity of Cultivable Hydrogen-Producing Bacteria Isolated from Agricultural Soils, Waste Water Sludge and Cow Dung. Int. J. Hydrog. Energy 2011, 36, 8735–8742. [Google Scholar] [CrossRef]
- Danhorn, T.; Fuqua, C. Biofilm Formation by Plant-Associated Bacteria. Annu. Rev. Microbiol. 2007, 61, 401–422. [Google Scholar] [CrossRef] [PubMed]
- Gobbin, D.; Rezzonico, F.; Gessler, C. Quantification of the Biocontrol Agent Pseudomonas fluorescens Pf153 In Soil Using a Quantitative Competitive PCR Assay Unaffected by Variability in Cell Lysis- And DNA-Extraction Efficiency. Soil Biol. Biochem. 2007, 39, 1609–1619. [Google Scholar] [CrossRef]
- Meza, A.; Rojas, P.; Cely-Veloza, W.; Guerrero-Perilla, C.; Coy-Barrera, E. Variation of Isoflavone Content And DPPH• Scavenging Capacity of Phytohormone-Treated Seedlings After In vitro Germination of Cape Broom (Genista Monspessulana). S. Afr. J. Bot. 2020, 130, 64–74. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, J.; Yang, P.; Xu, Z.; He, T.; Gao, Q.; Wang, L.; Li, Q. Interactive Effects Between Cadmium Stabilized by Palygorskite and Mobilized by Siderophores from Pseudomonas fluorescens. Ecotoxicol. Environ. Saf. 2019, 181, 265–273. [Google Scholar] [CrossRef]
- Williamson, A.; Folens, K.; Matthijs, S.; Paz Cortes, Y.; Varia, J.; Du Laing, G.; Boon, N.; Hennebel, T. Selective Metal Extraction by Biologically Produced Siderophores During Bioleaching from Low-Grade Primary and Secondary Mineral Resources. Miner. Eng. 2021, 163, 106774. [Google Scholar] [CrossRef]
- Suresh, P.; Vellasamy, S.; Almaary, K.; Dawoud, T.; Elbadawi, Y. Fluorescent pseudomonads (Fps) as a Potential Biocontrol and Plant Growth Promoting Agent Associated with Tomato Rhizosphere. J. King Saud Univ. Sci. 2021, 33, 101423. [Google Scholar] [CrossRef]
- Gerbal, C.; Smith, J. Sources of Pathogenic Microorganisms and Their Fate During Land Application of Wastes. J. Environ. Qual. 2005, 34, 42–48. [Google Scholar]
- Jiménez, B.; Austin, A.; Cloete, E.; Phasha, C.; Beltrán, N. Biological Risks to Food Crops Fertilized with Ecosan Sludge. Water Sci. Technol. 2007, 55, 21–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pepper, I.; Zerzghi, H.; Brooks, J.; Gerba, C. Sustainability of Land Application of Class B Biosolids. J. Environ. Qual. 2008, 37, S58–S67. [Google Scholar] [CrossRef] [Green Version]
- Navarro, I.; Jiménez, B.; Lucario, S.; Cifuentes, E. Application of Helminth Ova Infection Dose Curve to Estimate the Risks Associated with Biosolid Application on Soil. J. Water Health 2008, 7, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Turner, G.; Green, R.; Alae-Carew, C.; Dangour, A. The Association of Dimensions of Fruit and Vegetable Access in the Retail Food Environment with Consumption; a Systematic Review. Glob. Food Secur. 2021, 29, 100528. [Google Scholar] [CrossRef] [PubMed]
- Hibbing, M.; Fuqua, C.; Parsek, M.; Peterson, S. Bacterial Competition: Surviving and Thriving in the Microbial Jungle. Nat. Rev. Microbiol. 2009, 8, 15–25. [Google Scholar] [CrossRef] [Green Version]

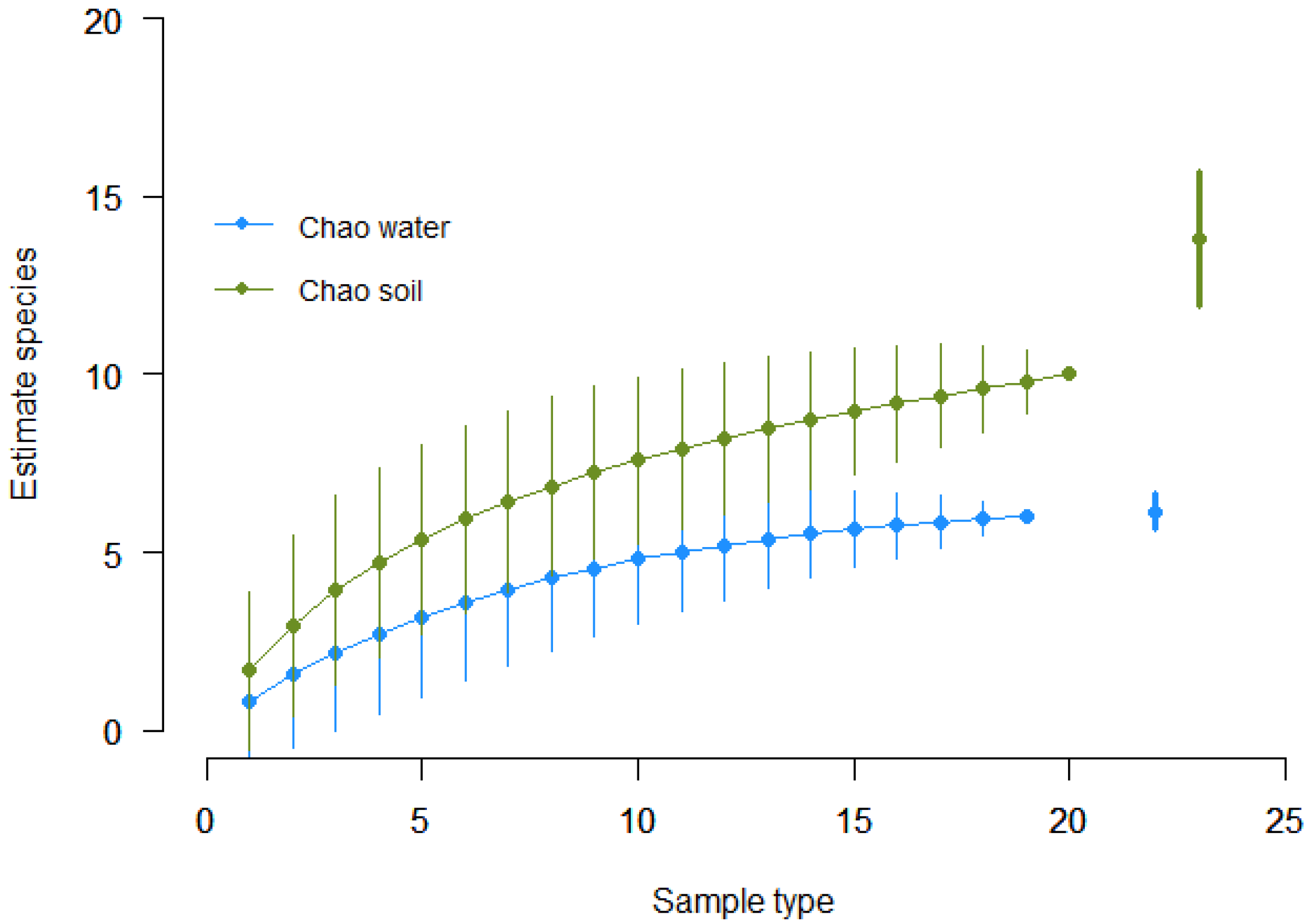
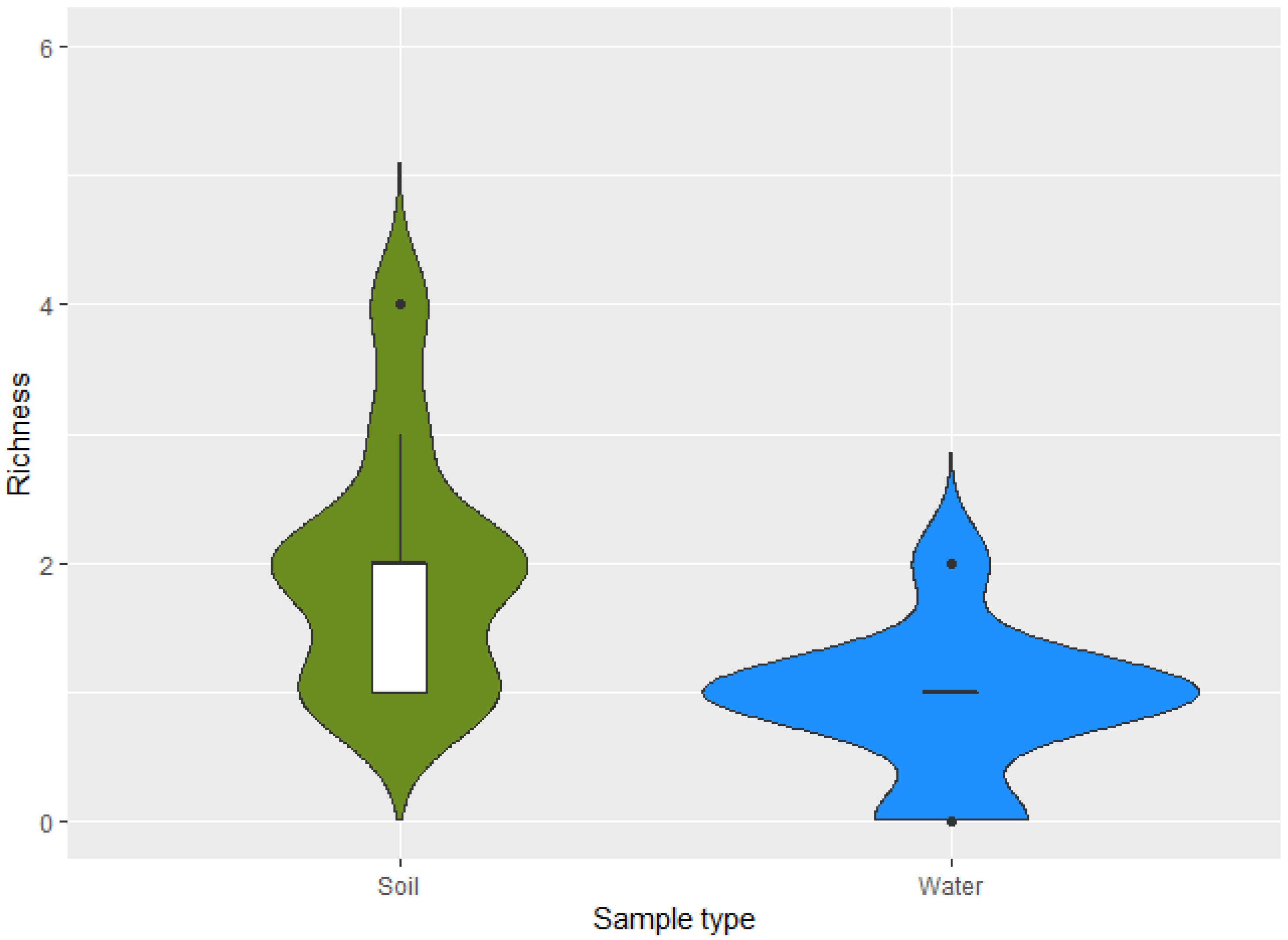
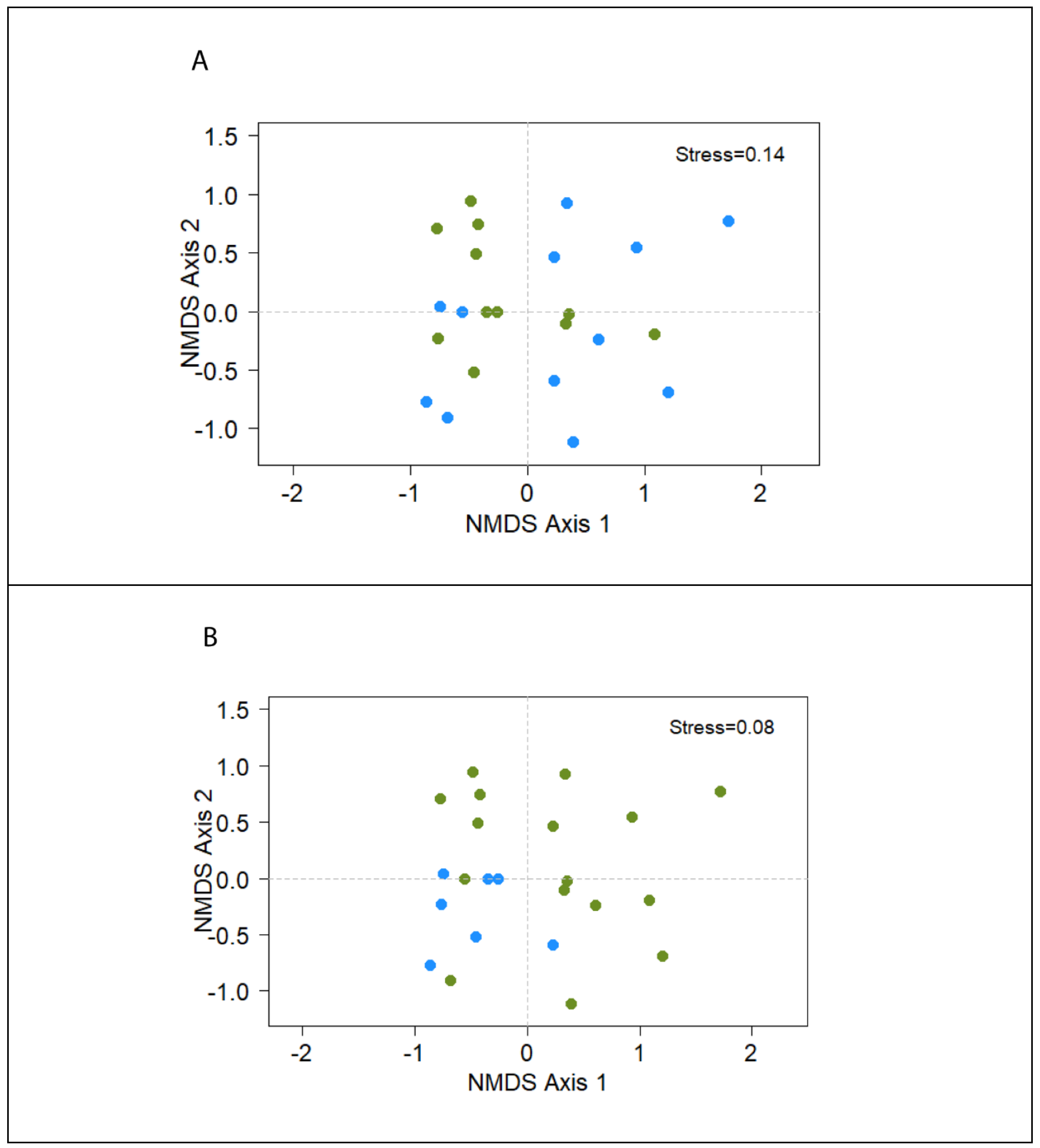
| Bacterial Species | TSI | GAS | H2S | CITRATE | UREA | MOTILITY | INDOL | LYSINE |
|---|---|---|---|---|---|---|---|---|
| Alkalescens dispar | ALC/A | − | − | − | − | − | + | +/− |
| Burkholderia mallei | A/A | − | − | + | − | − | − | − |
| Enterobacter cloacae | A/A | ++ | − | + | − | + | − | − |
| Enterobacter aerogenes | A/A | ++ | − | + | +/− | + | − | + |
| Escherichia coli | A/A | + | − | − | − | +/− | + | + |
| Klebsiella oxytoca | A/A | ++ | − | + | + | − | + | + |
| Pantoea agglomerans | A/A | +/− | − | + | − | + | − | − |
| Proteus vulgaris | ALC/A | +/− | + | +/− | ++ | + | + | − |
| Pseudomonas aeruginosa | K/K | − | − | + | + | + | − | +/− |
| Pseudomonas fluorescens | K/K | − | − | + | − | + | − | + |
| Salmonella spp. | K/A | + | + | + | − | + | − | + |
| Shigella spp. | K/A | − | − | − | − | − | − | − |
| Bacterial Species | Frequency–Number of Isolates by Species, Sample Type and Farm. | Habitat | Plant–Soil Nutrition Cycling or Growth Promotion | Pathogenicity | References | |||
|---|---|---|---|---|---|---|---|---|
| O:W | C:W | O:S | C:S | |||||
| Burkholderia mallei | 1 | Exclusively: intracellular parasite. | Information not found. | Pathogen: equines, mammals including humans. | [36] | |||
| Enterobacter cloacae | 5 | 1 | Ubiquitous: terrestrial and aquatic environments including wastewater and plants. Human intestines. | Some strains alleviating salinity stress and promoting growth in plants due to high nitrogen fixation activity and produced iron carriers. | Nosocomial and opportunistic pathogens. | [37,38,39,40] | ||
| Enterobacter aerogenes | 2 | 2 | 7 | Ubiquitous: terrestrial and aquatic environments, including wastewater and plants. Intestine animals (including humans). | Some strains release high amounts of phosphorus and help their solubilization on soil. | Nosocomial and opportunistic pathogens. | [40,41,42,43] | |
| Escherichia coli Escherichia coli inactive (Alkalescens dispar) | 1 | 2 | 3 | Frequently: intestines of animals (including humans). | Possible role in nitrogen cycle using ammonia and nitrate | Pathogen: animals (including humans). | [44,45] | |
| Klebsiella oxytoca | 2 | 1 | Ubiquitous: terrestrial and aquatic environment and intestine of a wide range of animals. | Related to production of auxin (indole-3-acetic acid) and growth promotor for some plants. | Nosocomial and opportunistic pathogens. | [46,47,48,49] | ||
| Pantoea agglomerans | 1 | Frequently: Plants, soil and fecal matter of humans and animals. | Considered as diazotrophic endophyte in the stem of Japanese sweet potato | Pathogen: humans, plants and animals. | [50,51] | |||
| Proteus vulgaris | 1 | Frequently: environmental saprophyte, found on decaying animal matter and in contaminated water. Intestine of animals (including humans). | Volatile organic compounds on growth stimulation of Chinese cabbage | Nosocomial and opportunistic pathogens in animals (including humans). | [52,53,54] | |||
| Pseudomonas aeruginosa | 2 | Ubiquitous: terrestrial and aquatic environments. Part of microbiota in animals (including humans). | Plant growth promoting through nitrogen accumulation, solubilization of phosphate, silicate and zinc. Additional positive for indole acetic acid. | Nosocomial and opportunistic pathogen in humans. | [55,56,57] | |||
| Pseudomonas fluorescens | 1 | 1 | Ubiquitous: terrestrial and aquatic environments. Non-pathogenic rhizobacteria. | Plant growth-promoting capacity through auxin-like phytohormones such as indole acetic acid. | Pathogen: many plants, and fish. Rarely pathogenic to humans. | [46,58,59,60,61] | ||
| Salmonella spp. | 2 | 3 | 15 | 8 | Frequently: intestine animals (including humans); survives in contaminated food and water. | Information not found. | Pathogen: animals (including humans). | [62,63] |
| Shigella spp. | 6 | 1 | 4 | Exclusively: human colon; survives in contaminated food and water. | Information not found. | Pathogen: humans. | [64,65] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz, D.; Cisneros, R.; Benítez, Á.; Zúñiga-Sarango, W.; Peña, J.; Fernández, H.; Jaramillo, A. Gram-Negative Bacteria from Organic and Conventional Agriculture in the Hydrographic Basin of Loja: Quality or Pathogen Reservoir? Agronomy 2021, 11, 2362. https://doi.org/10.3390/agronomy11112362
Cruz D, Cisneros R, Benítez Á, Zúñiga-Sarango W, Peña J, Fernández H, Jaramillo A. Gram-Negative Bacteria from Organic and Conventional Agriculture in the Hydrographic Basin of Loja: Quality or Pathogen Reservoir? Agronomy. 2021; 11(11):2362. https://doi.org/10.3390/agronomy11112362
Chicago/Turabian StyleCruz, Darío, Rodrigo Cisneros, Ángel Benítez, Wilson Zúñiga-Sarango, Jhoan Peña, Heriberto Fernández, and Andrea Jaramillo. 2021. "Gram-Negative Bacteria from Organic and Conventional Agriculture in the Hydrographic Basin of Loja: Quality or Pathogen Reservoir?" Agronomy 11, no. 11: 2362. https://doi.org/10.3390/agronomy11112362
APA StyleCruz, D., Cisneros, R., Benítez, Á., Zúñiga-Sarango, W., Peña, J., Fernández, H., & Jaramillo, A. (2021). Gram-Negative Bacteria from Organic and Conventional Agriculture in the Hydrographic Basin of Loja: Quality or Pathogen Reservoir? Agronomy, 11(11), 2362. https://doi.org/10.3390/agronomy11112362






