Involvement of Ethylene in Physiological Processes Determining the Vase Life of Various Hybrids of Mokara Orchid Cut Flowers
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Treatments
2.2. Measurement of Fresh Weight and Water Uptake
2.3. Measurement of Bud Opening, Floret Senescence, and Vase Life Duration
2.4. Measurement of Ethylene Production and Respiration Rates
2.5. Evaluation of ACC Content and Activities of ACS and ACO Enzymes
2.6. Experimental Design and Statistics Analysis
3. Results
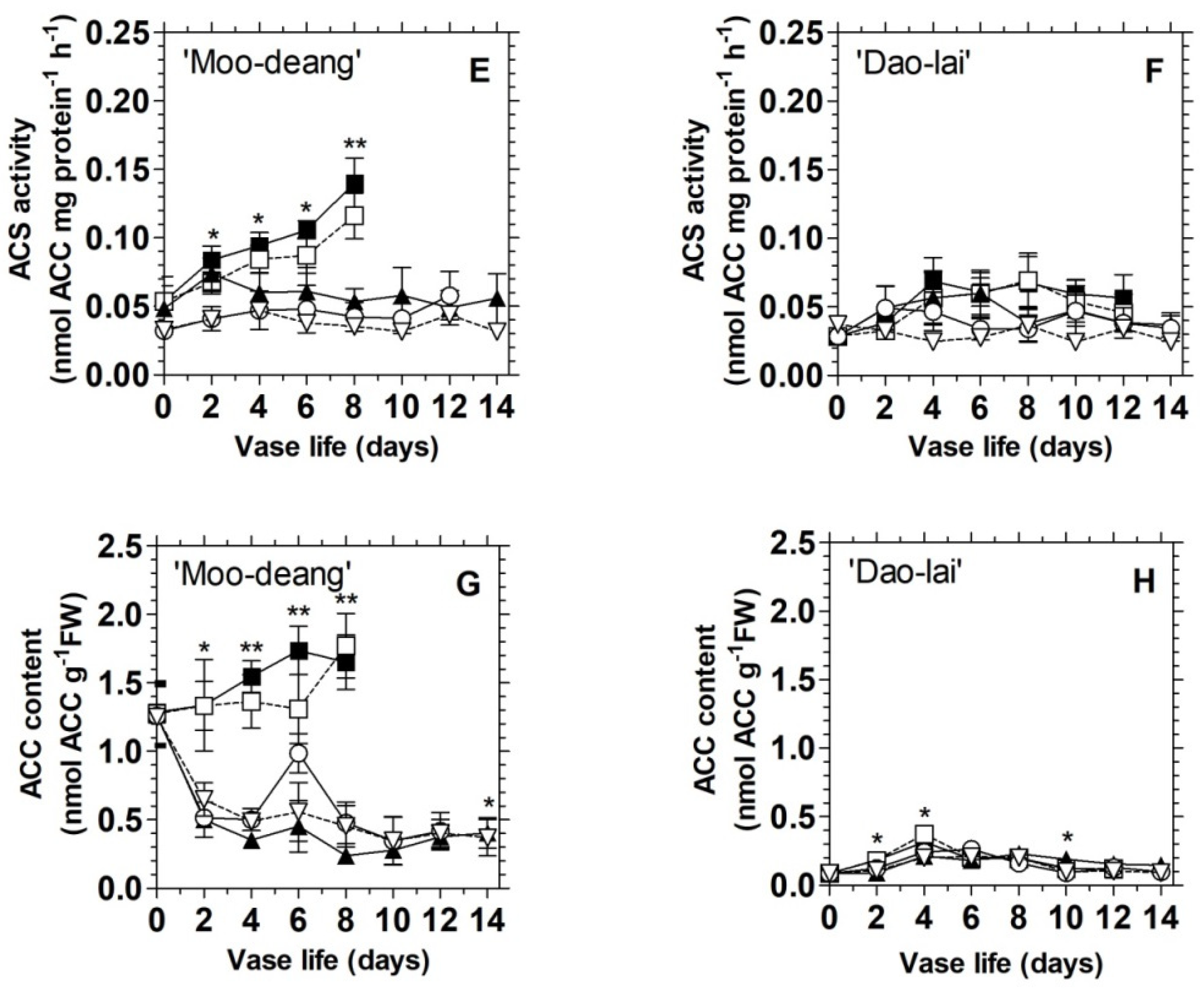
3.1. Characterization of the Different Vase Life Performance among the Five Mokara Hybrids
3.2. Ethylene Regulates Floret Development and Inflorescence Longevity in Two Mokara Hybrids
3.2.1. Effects of Exogenous Ethylene Treatment
3.2.2. Effects of Treatments with Ethylene Inhibitors
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Akamine, E.A. Ethylene production in fading Vanda orchid blossoms. Science 1963, 14, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Burg, S.P.; Dijkman, M.J. Ethylene and auxin participation in pollen-induced fading of Vanda orchid blossoms. Plant Physiol. 1967, 42, 1648–1650. [Google Scholar] [CrossRef] [PubMed]
- Goh, C.J.; Halevy, A.H.; Engelb, R.; Kofranek, A.M. Ethylene evolution and sensitivity in cut orchid flowers. Sci. Hortic. 1985, 26, 56–57. [Google Scholar] [CrossRef]
- Woltering, E.J.; van Doorn, W.G. Role of ethylene in senescence of petals morphological and taxonomical relationships. J. Exp. Bot. 1988, 39, 1605–1616. [Google Scholar] [CrossRef]
- Arditti, J.; Jeffrey, D.C.; Flick, B.H. Post-pollination phenomena in orchid flowers. III. Effects and interactions of auxin, kinetin or gibberellin. New Phytol. 1971, 70, 1125–1141. [Google Scholar] [CrossRef]
- Arditti, J.; Hogan, N.M.; Chadwick, A.V. Post-pollination phenomena in orchid flowers: IV. Effects of ethylene. Am. J. Bot. 1973, 60, 883–888. [Google Scholar] [CrossRef]
- Uthaichay, N.; Ketsa, S.; van Doorn, W.G. 1-MCP pretreatment prevents bud and flower abscission in Dendrobium orchids. Postharvest Biol. Technol. 2007, 43, 374–380. [Google Scholar] [CrossRef]
- Khunmuang, S.; Kanlayanarat, S.; Wongs-Aree, C.; Meir, S.; Philosoph-Hadas, S.; Buanong, M. Variability in the response to ethylene of three cultivars of cut Vanda orchid flowers. Acta Hortic. 2019, 1262, 241–249. [Google Scholar] [CrossRef]
- Khunmuang, S.; Kanlayanarat, S.; Wongs-Aree, C.; Meir, S.; Philosoph-Hadas, S.; Oren-Shamir, M.; Ovadia, R.; Buanong, M. Ethylene induces a rapid degradation of petal anthocyanins in cut Vanda ‘Sansai Blue’ orchid flowers. Front. Plant Sci. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Yang, S.F.; Hoffman, N.E. Ethylene biosynthesis and its regulation in higher plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1984, 35, 155–189. [Google Scholar] [CrossRef]
- Kende, H. Ethylene biosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1993, 44, 283–307. [Google Scholar] [CrossRef]
- Schaller, G.E.; Kieber, J.J. Ethylene. In The Arabidopsis Book; Somerville, C.R., Meyerowitz, E.M., Eds.; American Society Plant Biologists: Rockville, MD, USA, 2002; Volume 1, p. e0071. [Google Scholar] [CrossRef]
- Borochov, A.; Woodson, W.R. Physiology and biochemistry of flower petal senescence. Hortic. Rev. 1989, 11, 15–43. [Google Scholar]
- Wills, R.; McGlasson, B.; Graham, D.; Joyce, D. Postharvest: An Introduction to the Physiology and Handling of Fruit, Vegetables and Ornamentals, 4th ed.; Oxford University Press: Oxford, UK, 1998; 280p. [Google Scholar]
- Halevy, A.H.; Mayak, S. Senescence and post-harvest physiology of cut flowers. Part II. Hortic. Rev. 1981, 1, 212–245. [Google Scholar]
- Wang, H.; Woodson, W.R. Reversible inhibition of ethylene action and interruption of petal senescence in carnation flowers by norbornadiene. Plant Physiol. 1989, 89, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Beyer, E.M., Jr. A potent inhibitor of ethylene action in plants. Plant Physiol. 1976, 58, 268–271. [Google Scholar] [CrossRef]
- Reid, M.S.; Wu, M.J. Ethylene and flower senescence. Plant Growth Regul. 1992, 11, 37–43. [Google Scholar] [CrossRef]
- Veen, H.; Van de Geijn, S.C. Mobility of ionic form of silver as related to longevity of cut carnations. Planta 1978, 140, 93–96. [Google Scholar] [CrossRef]
- Serek, M.; Reid, M.S.; Sisler, E.C. A volatile ethylene inhibitor improves the postharvest life of potted roses. J. Am. Soc. Hortic. Sci. 1994, 119, 572–577. [Google Scholar] [CrossRef]
- Serek, M.; Sisler, E.C.; Reid, M.S. Effect of 1-MCP on the vase life and ethylene response of cut flowers. Plant Growth Regul. 1995, 16, 93–97. [Google Scholar] [CrossRef]
- Lee, H.Y. Genomic and meiotic analysis of Mokara orchids. J. Hered. 1994, 85, 39–43. [Google Scholar]
- Dymek, R.; Adamski, A.; Salachna, P.; Piechocki, R.; Žurawik, P. Asian Mokara (Orchidaceae) hybrids used as cut flowers: Postharvest evaluation. Part 2. World News Nat. Sci. 2019, 26, 11–19. [Google Scholar]
- Rahman, M.M.; Ahmad, S.H.; Mohamed, M.T.M.; Rahman, M.Z. Leaf extracts of Jatropha curcas, Psidium guajava and Andrographis paniculata control microbial population in the vase solution of cut Mokara red orchid flower. Acta Hortic. 2013, 1012, 593–598. [Google Scholar] [CrossRef]
- Rahman, M.Z.; Ahmad, S.H.; Tengku, M.; Mohamed, M.T.M.; Zain, S.M.; Hasan, B.S. Effect of Jatropha curcas, Psidium guajava and Andrographis paniculata leaf extracts on postharvest performance of cut Mokara red orchid flowers. Int. J. Hortic. Floric. 2015, 3, 179–207. [Google Scholar]
- Kato, M.; Hyodo, H. Purification and characterization of ACC oxidase and increase in its activity during ripening of pear fruit. J. Jpn. Soc. Hortic. Sci. 1999, 68, 551–557. [Google Scholar] [CrossRef]
- Lizada, M.C.C.; Yang, S.F. A simple and sensitive assay for 1-aminocyclopropane-1-carboxylic acid. Anal. Biochem. 1979, 100, 140–145. [Google Scholar]
- Hoffman, N.E.; Yang, S.F. Enhancement of wound-induced ethylene synthesis by ethylene in preclimacteric cantaloupe. Plant Physiol. 1982, 69, 317–322. [Google Scholar] [CrossRef]
- van Doorn, W.G. Categories of petal senescence and abscission: A re-evaluation. Ann. Bot. 2001, 87, 447–456. [Google Scholar] [CrossRef]
- van Doorn, W.G.; Woltering, E.J. Physiology and molecular biology of petal senescence. J. Exp. Bot. 2008, 59, 453–480. [Google Scholar] [CrossRef]
- Porat, R.; Borochov, A.; Halevy, A.H.; O’Neill, S.D. Pollination-induced senescence of Phalaenopsis petals. The wilting process, ethylene production and sensitivity to ethylene. Plant Growth Regul. 1994, 15, 129–136. [Google Scholar] [CrossRef]
- Porat, R.; Halevy, A.H.; Serek, M.; Borochov, A. An increase in ethylene sensitivity following pollination is the initial event triggering an increase in ethylene production and enhanced senescence of Phalaenopsis orchid flowers. Physiol. Plant. 1995, 93, 778–784. [Google Scholar] [CrossRef]
- van Doorn, W.G. Effect of ethylene on flower abscission: A survey. Ann. Bot. 2002, 89, 689–693. [Google Scholar] [CrossRef] [PubMed]
- van Doorn, W.G.; van Meeteren, U. Flower opening and closure: A review. J. Exp. Bot. 2003, 54, 1801–1812. [Google Scholar] [CrossRef] [PubMed]
- Khunmuang, S.; Kanlayanarat, S.; Wongs-Aree, C.; Meir, S.; Philosoph-Hadas, S.; Buanong, M. Effect of ethephon and 1-MCP treatment on the vase life of cut ‘Sansai blue’ Vanda. Acta Hortic. 2016, 1131, 119–125. [Google Scholar] [CrossRef]
- Khunmuang, S.; Kanlayanarat, S.; Wongchaochant, S.; Wongs-Aree, C.; Meir, S.; Buanong, M. Development of means for delaying senescence and prolonging the vase life of cut flowers of Vanda orchid cv. ‘Sansai blue’. Acta Hortic. 2018, 1213, 581–586. [Google Scholar] [CrossRef]
- Sapbua, D.; Samniangdee, P.; Uthairatanakij, A.; Buanong, M. 1-Methylcyclopropene affected the quality in long vase life of ‘Red Sonia’ Dendrobium Flower. Acta Hortic. 2013, 970, 217–221. [Google Scholar] [CrossRef]
- Yoodee, S.; Obsuwan, K. Effects of 1-MCP on postharvest life of cut Dendrobium ‘Burana Jade’ inflorescences. Acta Hortic. 2013, 970, 261–265. [Google Scholar] [CrossRef]
- Serek, M.; Woltering, E.J.; Sisler, E.C.; Frello, S.; Sriskandarajah, S. Controlling ethylene responses in flowers at the receptor level. Biotechnol. Adv. 2006, 24, 368–381. [Google Scholar] [CrossRef]
- Müller, R.; Anderson, A.S.; Serek, M. Differences in display life of miniature potted roses (Rosa hybrida L.). Sci. Hortic. 1998, 76, 59–71. [Google Scholar] [CrossRef]
- Leonard, R.T.; Nell, T.A.; Hoyer, L. Response of potted rose varieties to short-term ethylene exposure. Acta Hortic. 2005, 669, 373–380. [Google Scholar] [CrossRef]
- Bleecker, A.; Kende, H. Ethylene: A gaseous signal molecule in plants. Ann. Rev. Cell Dev. Biol. 2000, 16, 1–18. [Google Scholar] [CrossRef]
- Kuroda, S.; Hirose, Y.; Shiraishi, M.; Davies, E.; Abe, S. Co-expression of an ethylene receptor gene, ERS1, and ethylene signaling regulator gene, CTR1, in Delphinium during abscission of florets. Plant Physiol. Biochem. 2004, 42, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Sexton, R.; Laird, G.; van Doorn, W.G. Lack of ethylene involvement in tulip petal abscission. Physiol. Plant. 2000, 108, 321–329. [Google Scholar] [CrossRef]
- Reid, M.S.; Evans, R.Y.; Dodge, L.L.; Mor, Y. Ethylene and silver thiosulphate influence opening of cut rose flowers. J. Am. Soc. Hortic. Sci. 1989, 114, 436–440. [Google Scholar]
- Yamane, K.; Yamaki, Y.; Fujishige, N. Effect of exogenous ethylene and 1-MCP on ACC oxidase activity, ethylene production and vase life in Cattleya Aliliances. J. Jpn. Soc. Hortic. Sci. 2004, 73, 128–133. [Google Scholar] [CrossRef][Green Version]

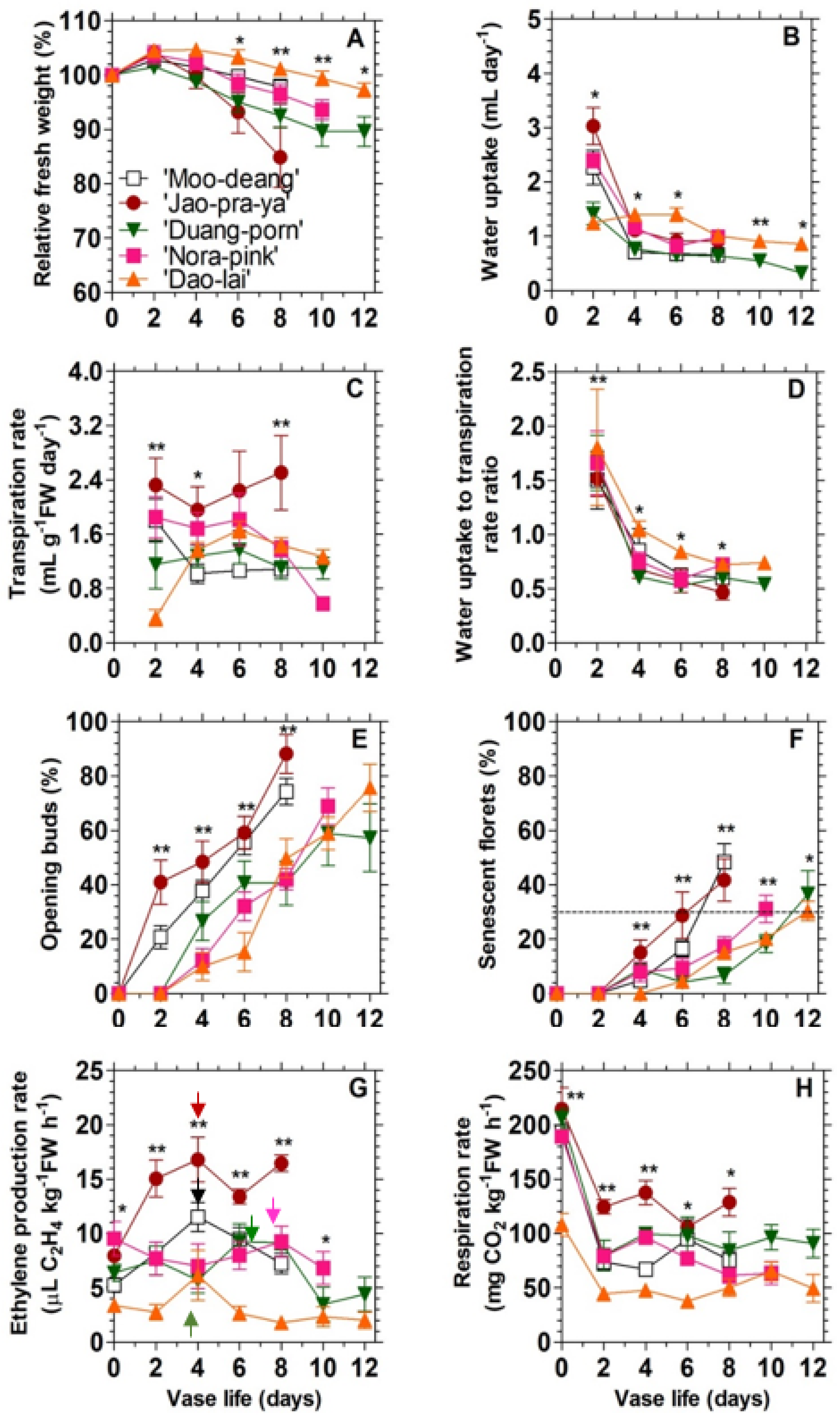
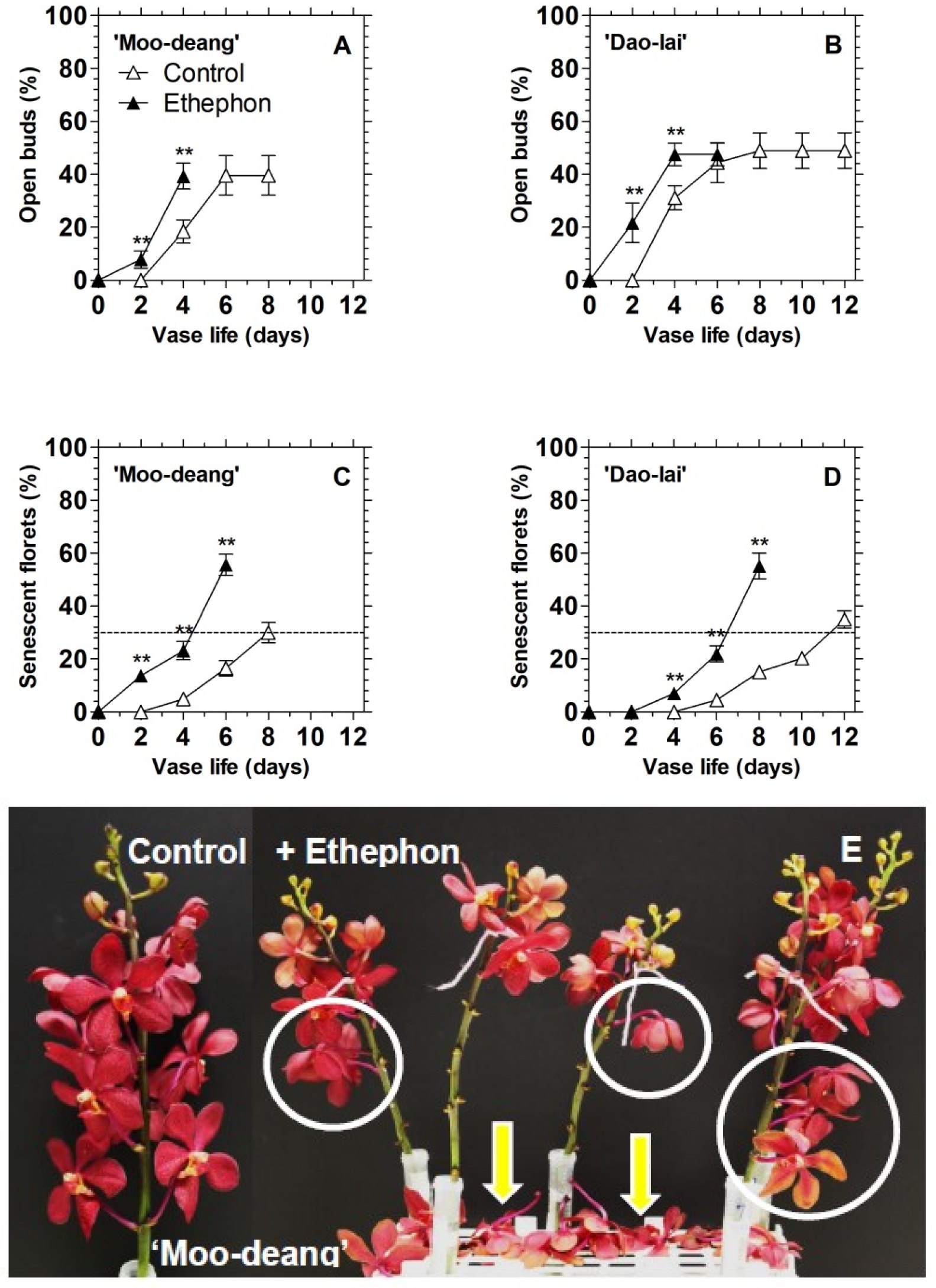

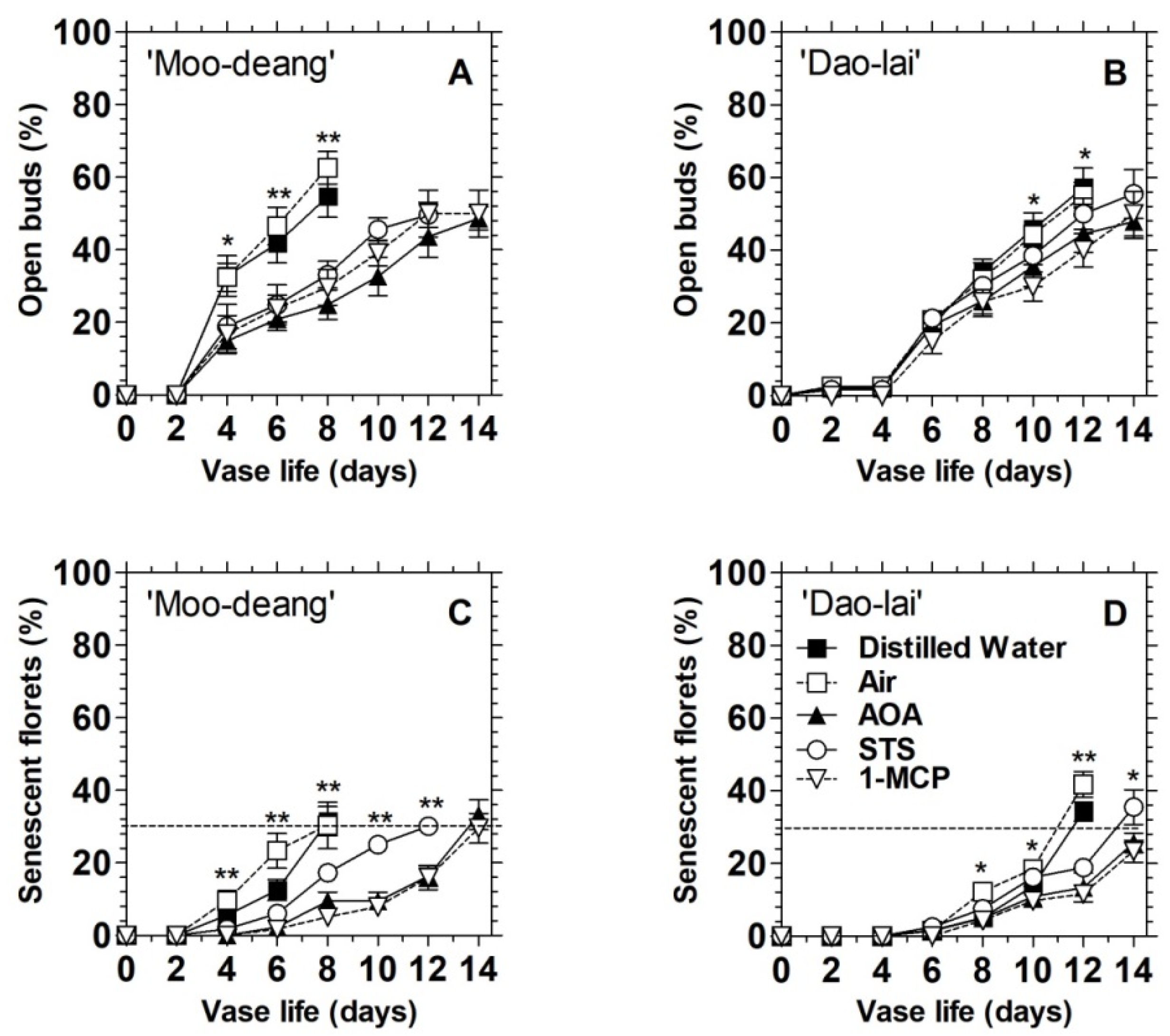
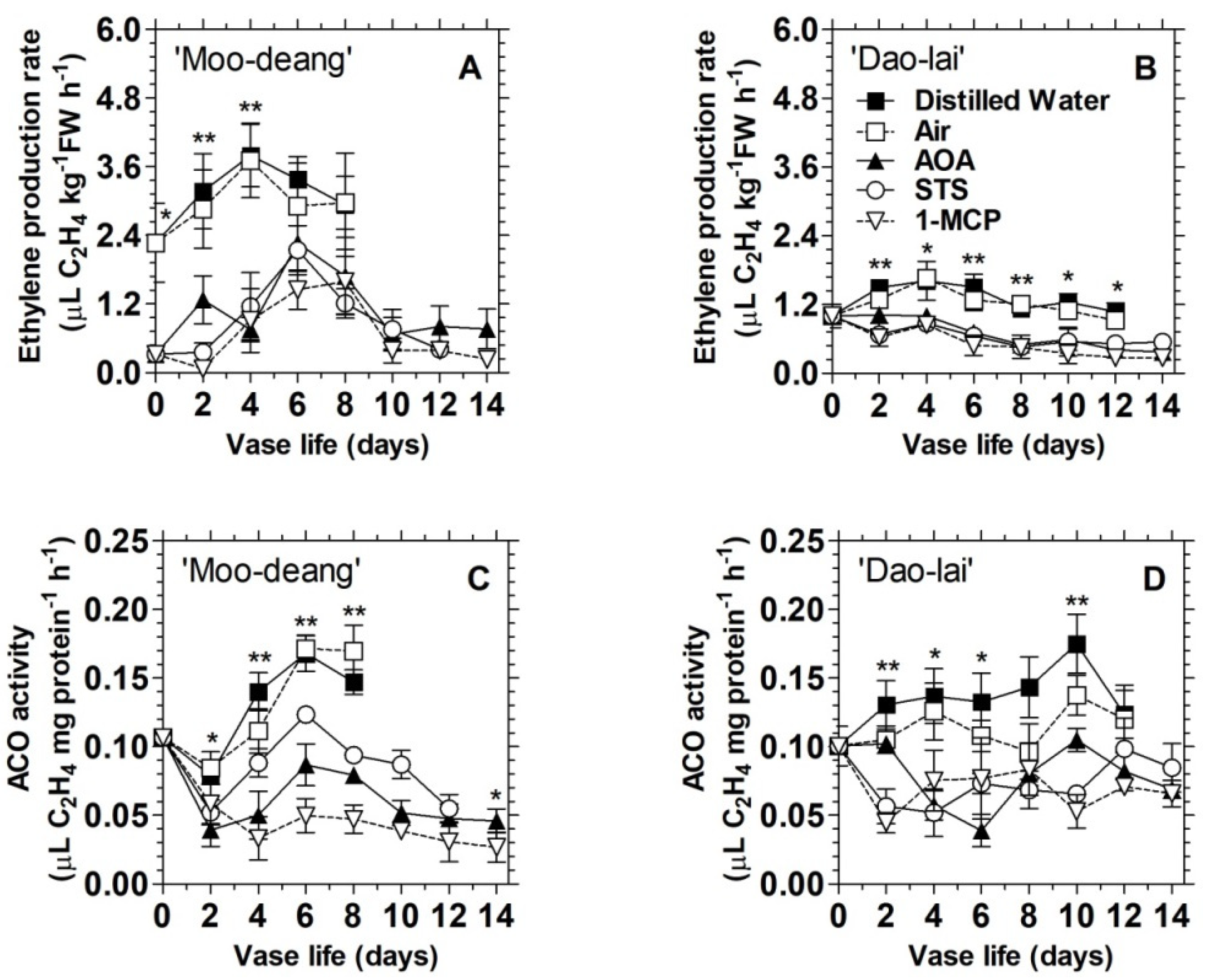
| Hybrids | Vase Life | Ethylene Production Rates (µL C2H4 kg−1 FW h−1) | ||
|---|---|---|---|---|
| Day 2 | Day 4 | Day 6 | ||
| ‘Moo-deang’ | 8.0 c | 8.22 b | 11.54 ab | 9.46 b |
| ‘Jao-pra-ya’ | 7.6 c | 15.06 a | 16.81 a | 13.42 a |
| ‘Duang-porn’ | 11.5 a | 7.52 b | 5.71 b | 9.28 b |
| ‘Nora-pink’ | 10.3 b | 7.70 b | 7.00 b | 7.98 b |
| ‘Dao-lai’ | 11.5 a | 2.77 c | 6.16 b | 2.66 c |
| F-test | ** | ** | ** | ** |
| Coefficient of determination of vase life and ethylene production | y = −0.3191x + 12.354 R2 = 0.5698 NS | y = −0.366x + 13.176 R2 = 0.8656 * | y = −0.3383x + 12.616 R2 = 0.4976 NS | |
| Hybrids | Treatments | Vase Life (Days) |
|---|---|---|
| ‘Moo-deang’ | Control | 8.1 b |
| Ethephon | 4.3 d | |
| ‘Dao-lai’ | Control | 11.3 a |
| Ethephon | 6.6 c | |
| F-test | ** | |
| Treatments | Vase Life (Days) | |
|---|---|---|
| ‘Moo-Deang’ | ‘Dao-Lai’ | |
| Distilled water | 8.1 b | 11.3 b |
| 0.5 mM AOA | 13.4 a | 14.3 a |
| 0.05 mM STS | 12.7 a | 13.8 a |
| Air | 8.4 b | 11.3 b |
| 200 nL·L−1 1-MCP | 13.7 a | 14.8 a |
| F-test | ** | ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wongjunta, M.; Wongs-Aree, C.; Salim, S.; Meir, S.; Philosoph-Hadas, S.; Buanong, M. Involvement of Ethylene in Physiological Processes Determining the Vase Life of Various Hybrids of Mokara Orchid Cut Flowers. Agronomy 2021, 11, 160. https://doi.org/10.3390/agronomy11010160
Wongjunta M, Wongs-Aree C, Salim S, Meir S, Philosoph-Hadas S, Buanong M. Involvement of Ethylene in Physiological Processes Determining the Vase Life of Various Hybrids of Mokara Orchid Cut Flowers. Agronomy. 2021; 11(1):160. https://doi.org/10.3390/agronomy11010160
Chicago/Turabian StyleWongjunta, Melada, Chalermchai Wongs-Aree, Shoshana Salim, Shimon Meir, Sonia Philosoph-Hadas, and Mantana Buanong. 2021. "Involvement of Ethylene in Physiological Processes Determining the Vase Life of Various Hybrids of Mokara Orchid Cut Flowers" Agronomy 11, no. 1: 160. https://doi.org/10.3390/agronomy11010160
APA StyleWongjunta, M., Wongs-Aree, C., Salim, S., Meir, S., Philosoph-Hadas, S., & Buanong, M. (2021). Involvement of Ethylene in Physiological Processes Determining the Vase Life of Various Hybrids of Mokara Orchid Cut Flowers. Agronomy, 11(1), 160. https://doi.org/10.3390/agronomy11010160





