Agricultural Soil Organic Matters and Microbiome Are Shaped by Management and Hedgerows
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Soil Sampling
2.2. Soil DOM Extraction and Data Analysis
2.3. DNA Isolation, Amplification, and Sequence Processing
2.4. Statistical Analysis
3. Results and Discussion
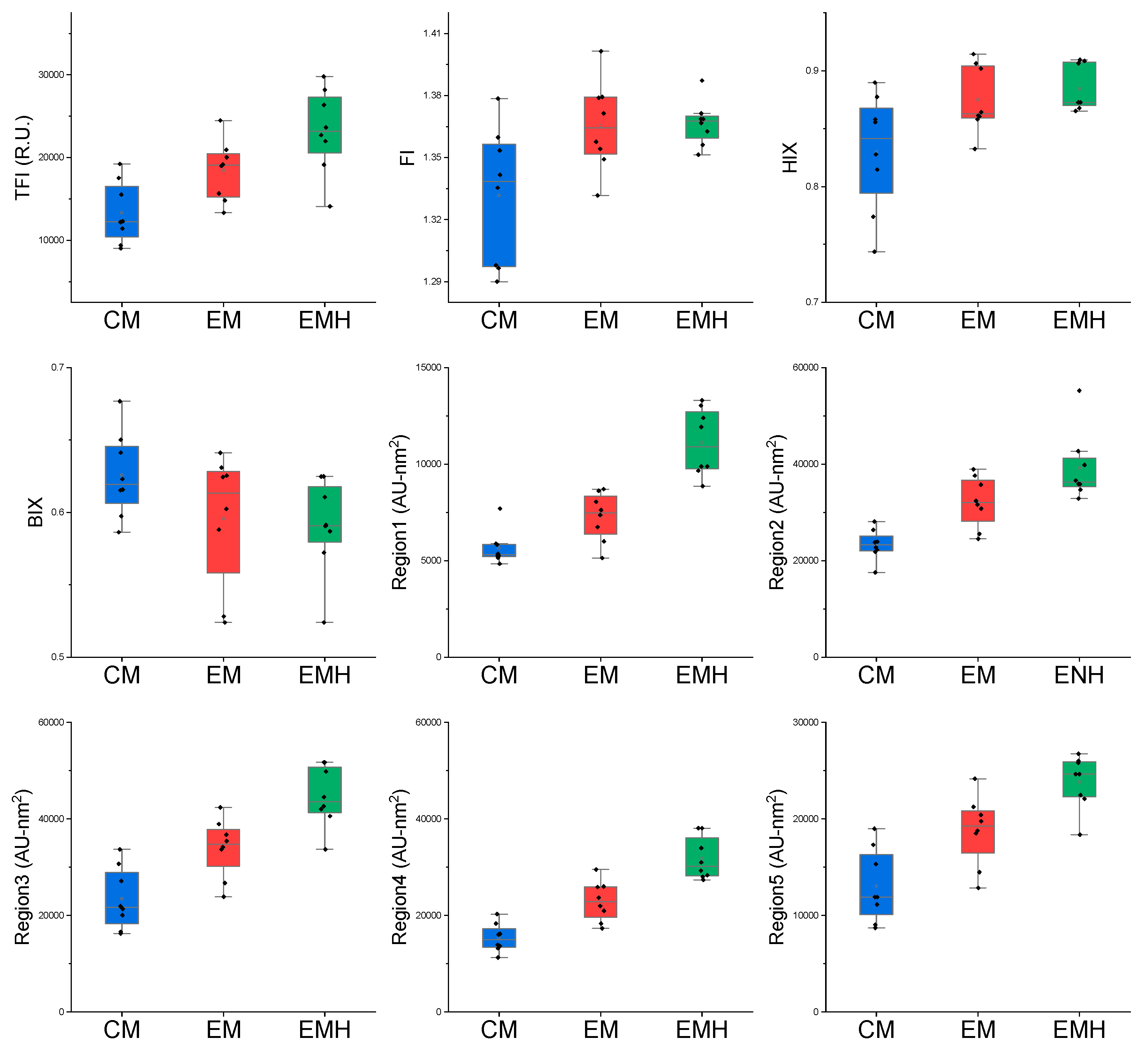
3.1. Variation of Soil Chemical Properties and DOM between the Three Farming Systems
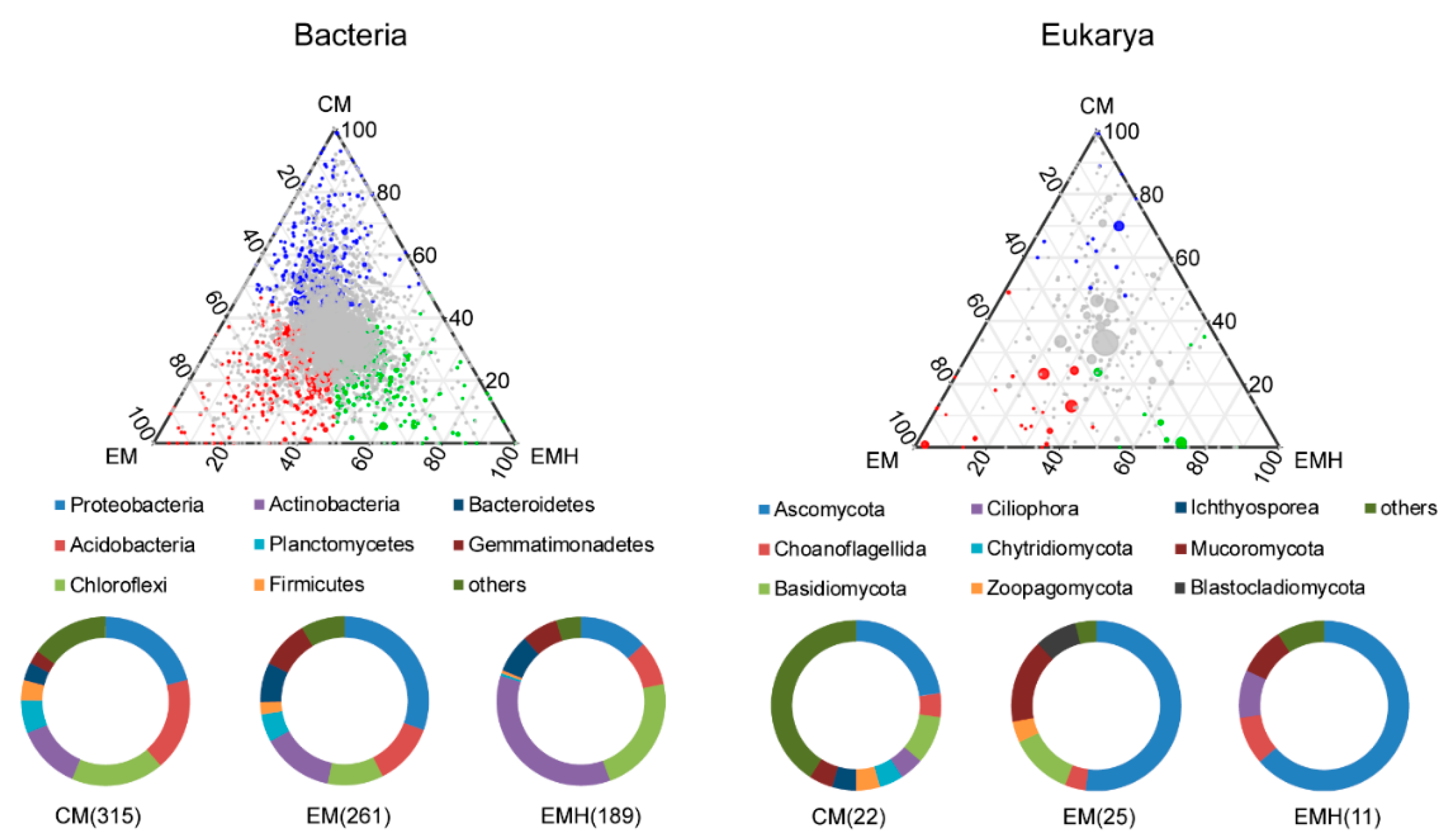
3.2. The Farming System Effects on Bacterial and Eukaryotic Communities
3.3. The Farming System Effects on Predictive Functional Profiling
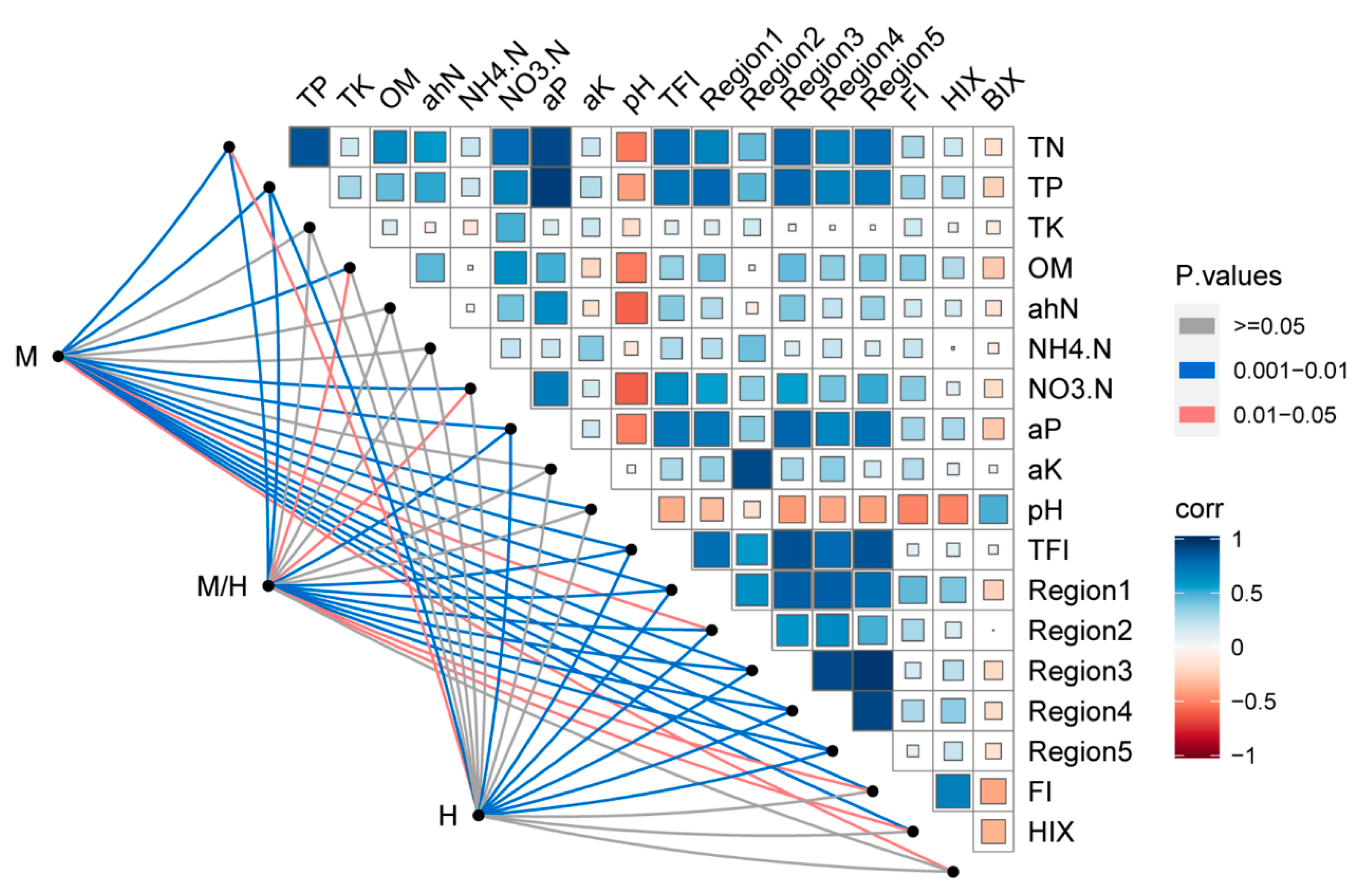
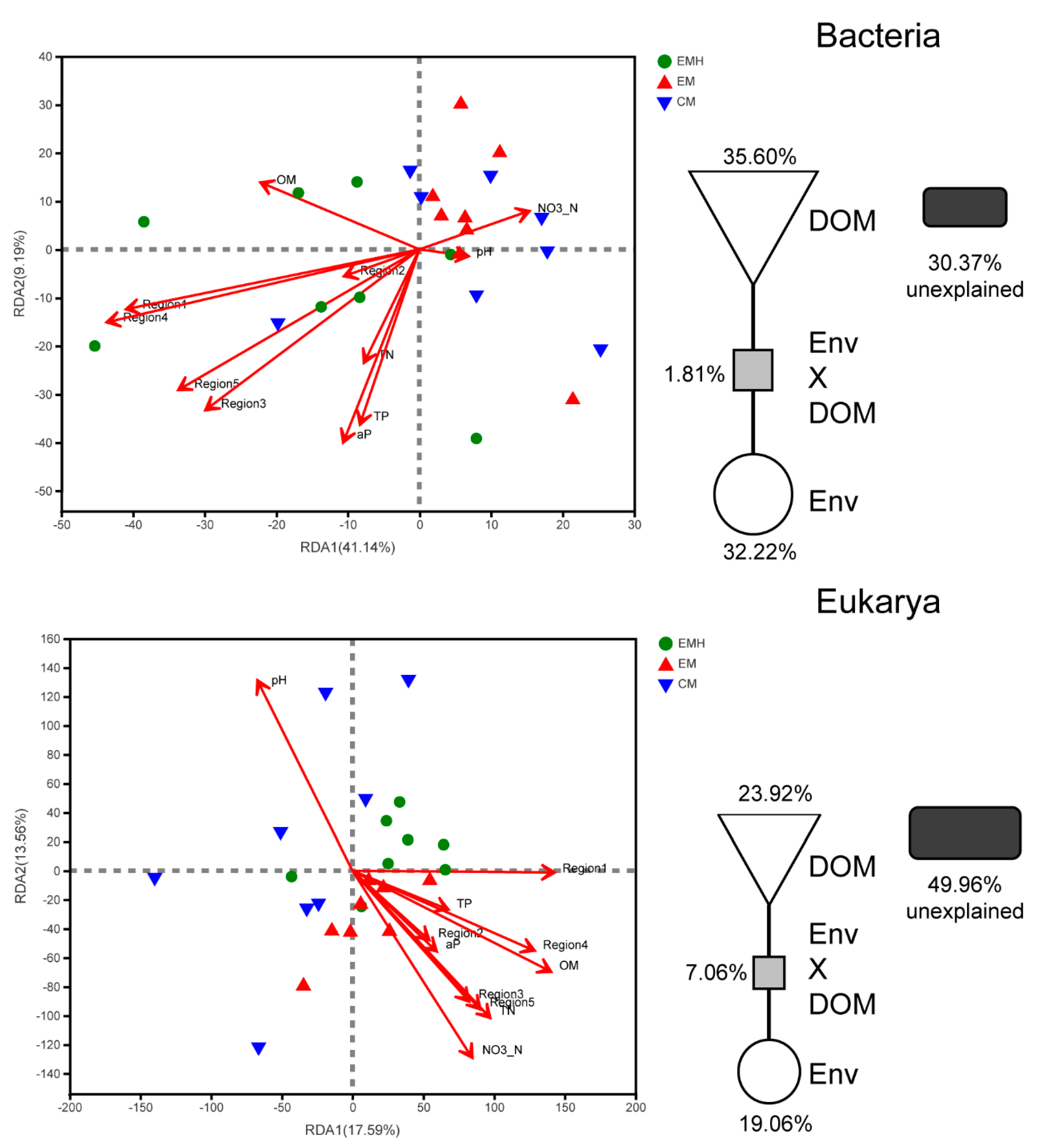
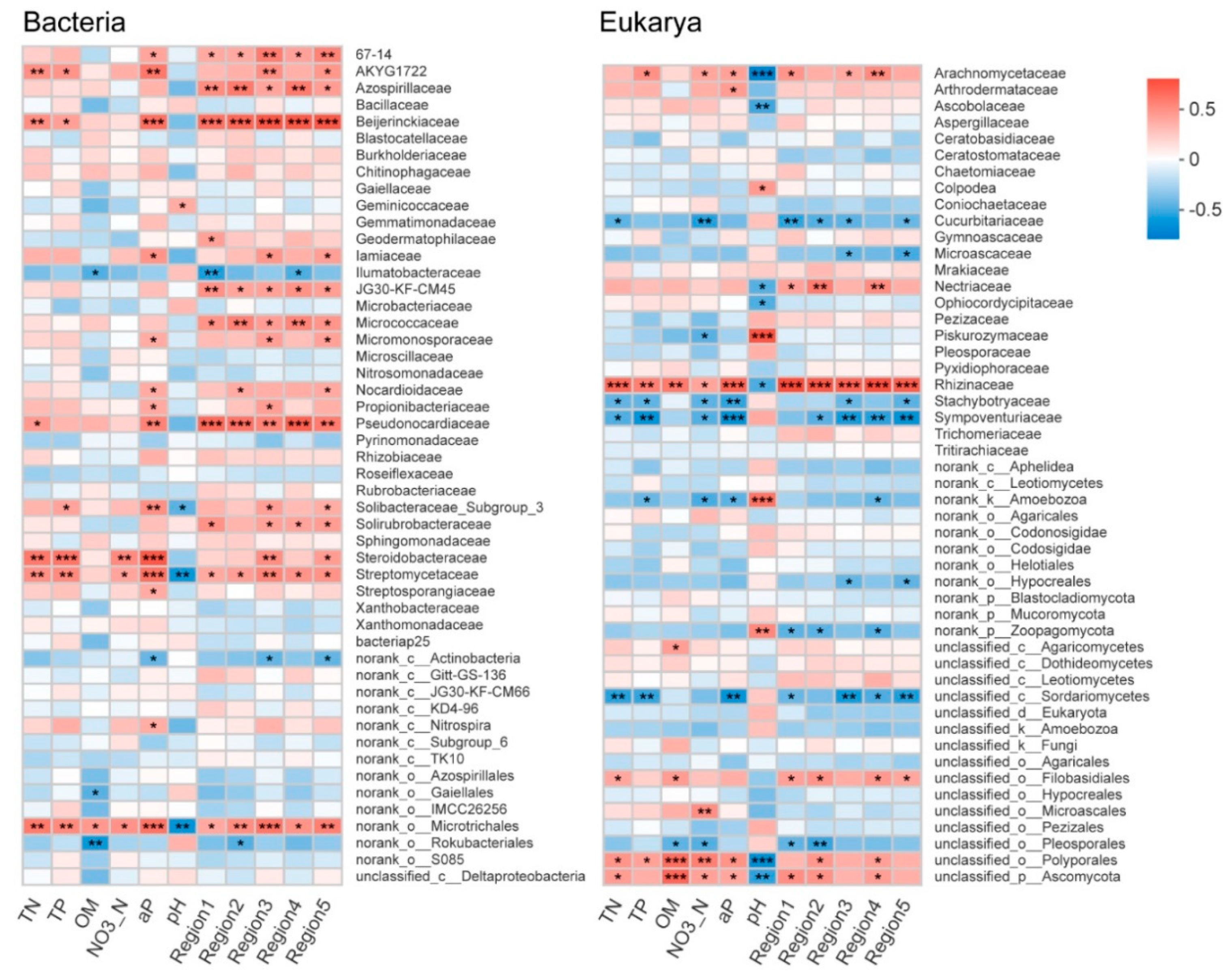
3.4. Relationship between Soil Microbial Communities, Soil Properties, and DOM Composition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polaasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.A.; DeFries, R.; Asner, G.P.; Barford, C.; Bonan, G.; Carpenter, S.R.; Chapin, F.S.; Coe, M.T.; Daily, G.C.; Gibbs, H.K.; et al. Global Consequences of Land Use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C.; et al. Solutions for a Cultivated Planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef]
- Bender, S.F.; Wagg, C.; van der Heijden, M.G.A. An Underground Revolution: Biodiversity and Soil Ecological Engineering for Agricultural Sustainability. Trends Ecol. Evol. 2016, 31, 440–452. [Google Scholar] [CrossRef] [PubMed]
- Gomiero, T.; Pimentel, D.; Paoletti, M.G. Environmental Impact of Different Agricultural Management Practices: Conventional vs. Organic Agriculture. Crit. Rev. Plant Sci. 2011, 30, 95–124. [Google Scholar] [CrossRef]
- Banerjee, S.; Walder, F.; Büchi, L.; Meyer, M.; Held, A.Y.; Gattinger, A.; Keller, T.; Charles, R.; van der Heijden, M.G.A. Agricultural Intensification Reduces Microbial Network Complexity and the Abundance of Keystone Taxa in Roots. ISME J. 2019, 13, 1722–1736. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, M.; Frey, B.; Mayer, J.; Mäder, P.; Widmer, F. Distinct Soil Microbial Diversity under Long-Term Organic and Conventional Farming. ISME J. 2015, 9, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.W.I.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Kleber, M.; Kögel-Knabner, I.; Lehmann, J.; Manning, D.A.C.; et al. Persistence of Soil Organic Matter as an Ecosystem Property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef]
- Ding, Y.; Shi, Z.; Ye, Q.; Liang, Y.; Liu, M.; Dang, Z.; Wang, Y.; Liu, C. Chemodiversity of Soil Dissolved Organic Matter. Environ. Sci. Technol. 2020, 54, 6174–6184. [Google Scholar] [CrossRef]
- Kellerman, A.M.; Kothawala, D.N.; Dittmar, T.; Tranvik, L.J. Persistence of Dissolved Organic Matter in Lakes Related to Its Molecular Characteristics. Nat. Geosci. 2015, 8, 454–457. [Google Scholar] [CrossRef]
- Lehmann, J.; Kleber, M. The Contentious Nature of Soil Organic Matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.E.; Kent, A.D.; Brisson, V.L.; Gaudin, A.C.M. Agricultural Management and Plant Selection Interactively Affect Rhizosphere Microbial Community Structure and Nitrogen Cycling. Microbiome 2019, 7, 146. [Google Scholar] [CrossRef]
- Fließbach, A.; Oberholzer, H.R.; Gunst, L.; Mäder, P. Soil Organic Matter and Biological Soil Quality Indicators after 21 Years of Organic and Conventional Farming. Agric. Ecosyst. Environ. 2007, 118, 273–284. [Google Scholar] [CrossRef]
- Gattinger, A.; Muller, A.; Haeni, M.; Skinner, C.; Fliessbach, A.; Buchmann, N.; Mäder, P.; Stolze, M.; Smith, P.; Scialabba, N.E.H.; et al. Enhanced Top Soil Carbon Stocks under Organic Farming. Proc. Natl. Acad. Sci. USA 2012, 109, 18226. [Google Scholar] [CrossRef] [PubMed]
- Lal, R. Soil Carbon Management and Climate Change. Carbon Manag. 2013, 4, 439–462. [Google Scholar] [CrossRef]
- Cotrufo, M.F.; Soong, J.L.; Horton, A.J.; Campbell, E.E.; Haddix, M.L.; Wall, D.H.; Parton, W.J. Formation of Soil Organic Matter via Biochemical and Physical Pathways of Litter Mass Loss. Nat. Geosci. 2015, 8, 776–779. [Google Scholar] [CrossRef]
- Doetterl, S.; Stevens, A.; Six, J.; Merckx, R.; Van Oost, K.; Casanova Pinto, M.; Casanova-Katny, A.; Muñoz, C.; Boudin, M.; Zagal Venegas, E.; et al. Soil Carbon Storage Controlled by Interactions between Geochemistry and Climate. Nat. Geosci. 2015, 8, 780–783. [Google Scholar] [CrossRef]
- Ghimire, R.; Lamichhane, S.; Acharya, B.S.; Bista, P.; Sainju, U.M. Tillage, Crop Residue, and Nutrient Management Effects on Soil Organic Carbon in Rice-Based Cropping Systems: A Review. J. Integr. Agric. 2017, 16, 1–15. [Google Scholar] [CrossRef]
- Borisover, M.; Lordian, A.; Levy, G.J. Water-Extractable Soil Organic Matter Characterization by Chromophoric Indicators: Effects of Soil Type and Irrigation Water Quality. Geoderma 2012, 179, 28–37. [Google Scholar] [CrossRef]
- Chantigny, M.H.; Harrison-Kirk, T.; Curtin, D.; Beare, M. Temperature and Duration of Extraction Affect the Biochemical Composition of Soil Water-Extractable Organic Matter. Soil Biol. Biochem. 2014, 75, 161–166. [Google Scholar] [CrossRef]
- Gao, J.; Liang, C.; Shen, G.; Lv, J.; Wu, H. Spectral Characteristics of Dissolved Organic Matter in Various Agricultural Soils throughout China. Chemosphere 2017, 176, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Crits-Christoph, A.; Diamond, S.; Butterfield, C.N.; Thomas, B.C.; Banfield, J.F. Novel Soil Bacteria Possess Diverse Genes for Secondary Metabolite Biosynthesis. Nature 2018, 558, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Peay, K.G.; Kennedy, P.G.; Talbot, J.M. Dimensions of Biodiversity in the Earth Mycobiome. Nat. Rev. Microbiol. 2016, 14, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Huang, Y.; Feng, S.Z.; Ge, Y.H.; Zhang, W.; He, X.; Wang, K. Soil organic carbon mineralization with fresh organic substrate and inorganic carbon additions in a red soil is controlled by fungal diversity along a pH gradient. Geoderma 2018, 321, 79–89. [Google Scholar] [CrossRef]
- Zarraonaindia, I.; Owens, S.M.; Weisenhorn, P.; West, K.; Hampton-Marcell, J.; Lax, S.; Bokulich, N.A.; Mills, D.A.; Martin, G.; Taghavi, S.; et al. The Soil Microbiome Influences Grapevine-Associated Microbiota. mBio 2015, 6. [Google Scholar] [CrossRef]
- de Vries, F.T.; Thébault, E.; Liiri, M.; Birkhofer, K.; Tsiafouli, M.A.; Bjørnlund, L.; Jørgensen, H.B.; Brady, M.V.; Christensen, S.; de Ruiter, P.C.; et al. Soil Food Web Properties Explain Ecosystem Services across European Land Use Systems. Proc. Natl. Acad. Sci. USA 2013, 110, 14296–14301. [Google Scholar] [CrossRef]
- Ding, L.J.; Su, J.Q.; Sun, G.X.; Wu, J.S.; Wei, W.X. Increased Microbial Functional Diversity under Long-Term Organic and Integrated Fertilization in a Paddy Soil. Appl. Microbiol. Biotechnol. 2018, 102, 1969–1982. [Google Scholar] [CrossRef]
- Postma-Blaauw, M.B.; de Goede, R.G.M.; Bloem, J.; Faber, J.H.; Brussaard, L. Soil Biota Community Structure and Abundance under Agricultural Intensification and Extensification. Ecology 2010, 91, 460–473. [Google Scholar] [CrossRef]
- Coller, E.; Cestaro, A.; Zanzotti, R.; Bertoldi, D.; Pindo, M.; Larger, S.; Albanese, D.; Mescalchin, E.; Donati, C. Microbiome of Vineyard Soils Is Shaped by Geography and Management. Microbiome 2019, 7, 140. [Google Scholar] [CrossRef]
- Lupatini, M.; Korthals, G.W.; de Hollander, M.; Janssens, T.K.S.; Kuramae, E.E. Soil Microbiome Is More Heterogeneous in Organic than in Conventional Farming System. Front. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Lupatini, M.; Korthals, G.W.; Roesch, L.F.W.; Kuramae, E.E. Long-Term Farming Systems Modulate Multi-Trophic Responses. Sci. Total Environ. 2019, 646, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Bonanomi, G.; De Filippis, F.; Cesarano, G.; La Storia, A.; Ercolini, D.; Scala, F. Organic Farming Induces Changes in Soil Microbiota That Affect Agro-Ecosystem Functions. Soil Biol. Biochem. 2016, 103, 327–336. [Google Scholar] [CrossRef]
- Degrune, F.; Boeraeve, F.; Dufrêne, M.; Cornélis, J.T.; Frey, B.; Hartmann, M. The Pedological Context Modulates the Response of Soil Microbial Communities to Agroecological Management. Front. Ecol. Evol. 2019, 7. [Google Scholar] [CrossRef]
- Holden, J.; Grayson, R.P.; Berdeni, D.; Bird, S.; Chapman, P.J.; Edmondson, J.L.; Firbank, L.G.; Helgason, T.; Hodson, M.E.; Hunt, S.F.P.; et al. The Role of Hedgerows in Soil Functioning within Agricultural Landscapes. Agric. Ecosyst. Environ. 2019, 273, 1–12. [Google Scholar] [CrossRef]
- Marshall, E.J.P.; West, T.M.; Kleijn, D. Impacts of an Agri-Environment Field Margin Prescription on the Flora and Fauna of Arable Farmland in Different Landscapes. Agric. Ecosyst. Environ. 2006, 113, 36–44. [Google Scholar] [CrossRef]
- Morandin, L.A.; Kremen, C. Hedgerow Restoration Promotes Pollinator Populations and Exports Native Bees to Adjacent Fields. Ecol. Appl. 2013, 23, 829–839. [Google Scholar] [CrossRef]
- Morandin, L.A.; Long, R.F.; Kremen, C. Pest Control and Pollination Cost–Benefit Analysis of Hedgerow Restoration in a Simplified Agricultural Landscape. J. Econ. Entomol. 2016, 109, 1020–1027. [Google Scholar] [CrossRef]
- Spaans, F.; Caruso, T.; Hammer, E.C.; Montgomery, I. Trees in Trimmed Hedgerows but Not Tree Health Increase Diversity of Oribatid Mite Communities in Intensively Managed Agricultural Land. Soil Biol. Biochem. 2019, 138, 107568. [Google Scholar] [CrossRef]
- Chai, L.; Huang, M.; Fan, H.; Wang, J.; Jiang, D.; Zhang, M.; Huang, Y. Urbanization Altered Regional Soil Organic Matter Quantity and Quality: Insight from Excitation Emission Matrix (EEM) and Parallel Factor Analysis (PARAFAC). Chemosphere 2019, 220, 249–258. [Google Scholar] [CrossRef]
- Huguet, A.; Vacher, L.; Relexans, S.; Saubusse, S.; Froidefond, J.M.; Parlanti, E. Properties of Fluorescent Dissolved Organic Matter in the Gironde Estuary. Org. Geochem. 2009, 40, 706–719. [Google Scholar] [CrossRef]
- McKnight, D.M.; Boyer, E.W.; Westerhoff, P.K.; Doran, P.T.; Kulbe, T.; Andersen, D.T. Spectrofluorometric Characterization of Dissolved Organic Matter for Indication of Precursor Organic Material and Aromaticity. Limnol. Oceanogr. 2001, 46, 38–48. [Google Scholar] [CrossRef]
- Zsolnay, A.; Baigar, E.; Jimenez, M.; Steinweg, B.; Saccomandi, F. Differentiating with Fluorescence Spectroscopy the Sources of Dissolved Organic Matter in Soils Subjected to Drying. Chemosphere 1999, 38, 45–50. [Google Scholar] [CrossRef]
- Chen, W.; Westerhoff, P.; Leenheer, J.A.; Booksh, K. Fluorescence Excitation−Emission Matrix Regional Integration to Quantify Spectra for Dissolved Organic Matter. Environ. Sci. Technol. 2003, 37, 5701–5710. [Google Scholar] [CrossRef] [PubMed]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science Using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Matson, P.A.; Parton, W.J.; Power, A.G.; Swift, M.J. Agricultural Intensification and Ecosystem Properties. Science 1997, 277, 504–509. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Guo, X.; Feng, J.; Shi, Z.; Zhou, X.; Yuan, M.; Tao, X.; Hale, L.; Yuan, T.; Wang, J.; Qin, Y.; et al. Climate Warming Leads to Divergent Succession of Grassland Microbial Communities. Nat. Clim. Chang. 2018, 8, 813–818. [Google Scholar] [CrossRef]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic Biomarker Discovery and Explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef]
- Louca, S.; Parfrey, L.W.; Doebeli, M. Decoupling Function and Taxonomy in the Global Ocean Microbiome. Science 2016, 353, 1272–1277. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Song, Z.; Bates, S.T.; Branco, S.; Tedersoo, L.; Menke, J.; Schilling, J.S.; Kennedy, P.G. FUNGuild: An Open Annotation Tool for Parsing Fungal Community Datasets by Ecological Guild. Fungal Ecol. 2016, 20, 241–248. [Google Scholar] [CrossRef]
- Williams, A.; Börjesson, G.; Hedlund, K. The Effects of 55 Years of Different Inorganic Fertiliser Regimes on Soil Properties and Microbial Community Composition. Soil Biol. Biochem. 2013, 67, 41–46. [Google Scholar] [CrossRef]
- Xia, L.; Hoermann, G.; Ma, L.; Yang, L. Reducing Nitrogen and Phosphorus Losses from Arable Slope Land with Contour Hedgerows and Perennial Alfalfa Mulching in Three Gorges Area, China. CATENA 2013, 110, 86–94. [Google Scholar] [CrossRef]
- Oshunsanya, S.O.; Li, Y.; Yu, H. Vetiver Grass Hedgerows Significantly Reduce Nitrogen and Phosphorus Losses from Fertilized Sloping Lands. Sci. Total Environ. 2019, 661, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Barthod, J.; Rumpel, C.; Dignac, M.F. Composting with Additives to Improve Organic Amendments. A Review. Agron. Sustain. Dev. 2018, 38, 17. [Google Scholar] [CrossRef]
- He, X.S.; Xi, B.D.; Li, X.; Pan, H.W.; An, D.; Bai, S.G.; Li, D.; Cui, D.Y. Fluorescence Excitation–Emission Matrix Spectra Coupled with Parallel Factor and Regional Integration Analysis to Characterize Organic Matter Humification. Chemosphere 2013, 93, 2208–2215. [Google Scholar] [CrossRef]
- Hagedorn, F.; Kammer, A.; Schmidt, M.W.I.; Goodale, C.L. Nitrogen Addition Alters Mineralization Dynamics of 13C-Depleted Leaf and Twig Litter and Reduces Leaching of Older DOC from Mineral Soil. Glob. Chang. Biol. 2012, 18, 1412–1427. [Google Scholar] [CrossRef]
- Wang, J.J.; Bowden, R.D.; Lajtha, K.; Washko, S.E.; Wurzbacher, S.J.; Simpson, M.J. Long-Term Nitrogen Addition Suppresses Microbial Degradation, Enhances Soil Carbon Storage, and Alters the Molecular Composition of Soil Organic Matter. Biogeochemistry 2019, 142, 299–313. [Google Scholar] [CrossRef]
- Wang, J.J.; Liu, Y.; Bowden, R.D.; Lajtha, K.; Simpson, A.J.; Huang, W.L.; Simpson, M.J. Long-Term Nitrogen Addition Alters the Composition of Soil-Derived Dissolved Organic Matter. ACS Earth Space Chem. 2020, 4, 189–201. [Google Scholar] [CrossRef]
- Lori, M.; Symnaczik, S.; Mäder, P.; Deyn, G.D.; Gattinger, A. Organic Farming Enhances Soil Microbial Abundance and Activity—A Meta-Analysis and Meta-Regression. PLoS ONE 2017, 12, e0180442. [Google Scholar] [CrossRef]
- Fierer, N.; Bradford, M.A.; Jackson, R.B. Toward an Ecological Classification of Soil Bacteria. Ecology 2007, 88, 1354–1364. [Google Scholar] [CrossRef]
- Fierer, N.; Lauber, C.L.; Ramirez, K.S.; Zaneveld, J.; Bradford, M.A.; Knight, R. Comparative Metagenomic, Phylogenetic and Physiological Analyses of Soil Microbial Communities across Nitrogen Gradients. ISME J. 2012, 6, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Crous, P.W.; Schoch, C.L.; Bahkali, A.H.; Guo, L.D.; Hyde, K.D. A Molecular, Morphological and Ecological Re-Appraisal of Venturiales―A New Order of Dothideomycetes. Fungal Divers. 2011, 51, 249–277. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.E.F.; Lopes, A.R.; Cunha-Queda, A.C.; Nunes, O.C. Comparison of the Bacterial Composition of Two Commercial Composts with Different Physicochemical, Stability and Maturity Properties. Waste Manag. 2016, 50, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Sridevi, G.; Minocha, R.; Turlapati, S.A.; Goldfarb, K.C.; Brodie, E.L.; Tisa, L.S.; Minocha, S.C. Soil Bacterial Communities of a Calcium-Supplemented and a Reference Watershed at the Hubbard Brook Experimental Forest (HBEF), New Hampshire, USA. FEMS Microbiol. Ecol. 2012, 79, 728–740. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, B.X.; Ren, H.; Zhang, J. Composition and Functional Diversity of Microbial Community across a Mangrove-Inhabited Mudflat as Revealed by 16S RDNA Gene Sequences. Sci. Total Environ. 2018, 633, 518–528. [Google Scholar] [CrossRef]
- McGee, K.M.; Eaton, W.D.; Shokralla, S.; Hajibabaei, M. Determinants of Soil Bacterial and Fungal Community Composition toward Carbon-Use Efficiency across Primary and Secondary Forests in a Costa Rican Conservation Area. Microb. Ecol. 2019, 77, 148–167. [Google Scholar] [CrossRef]
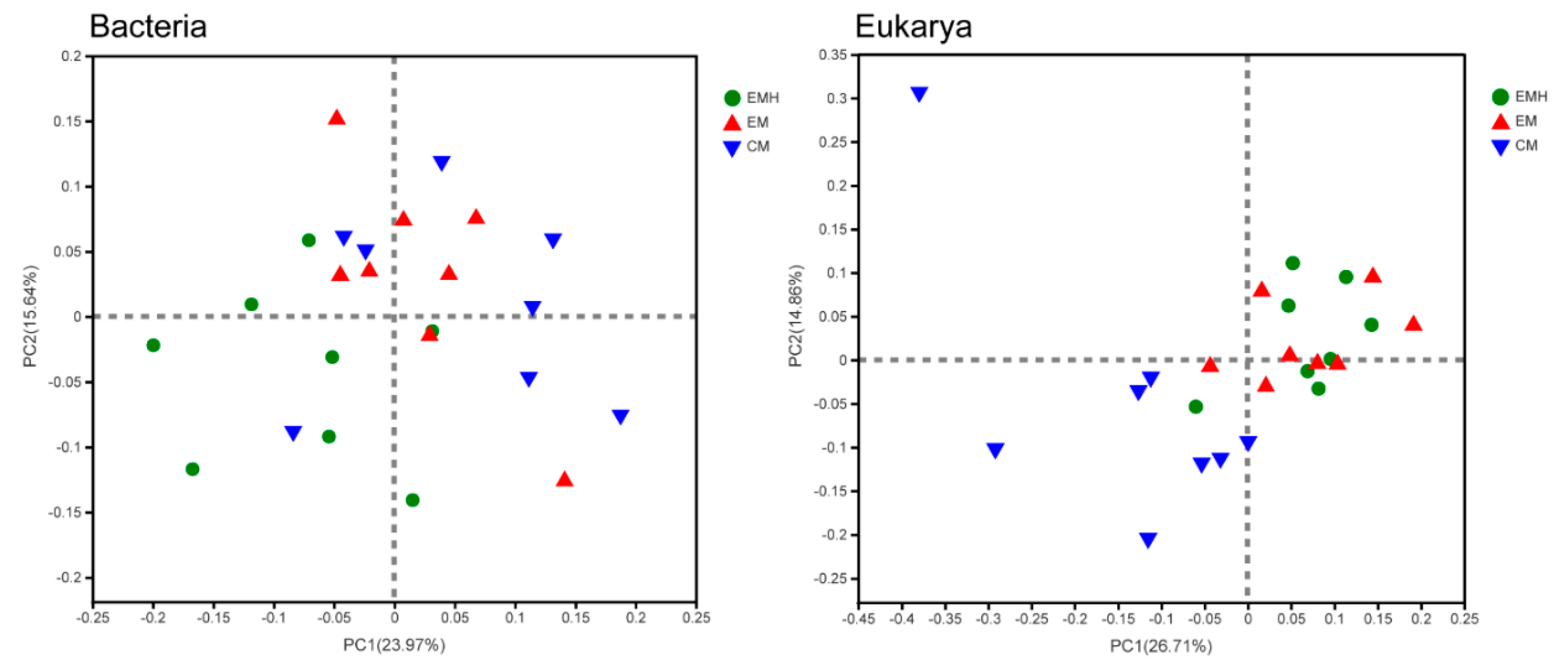
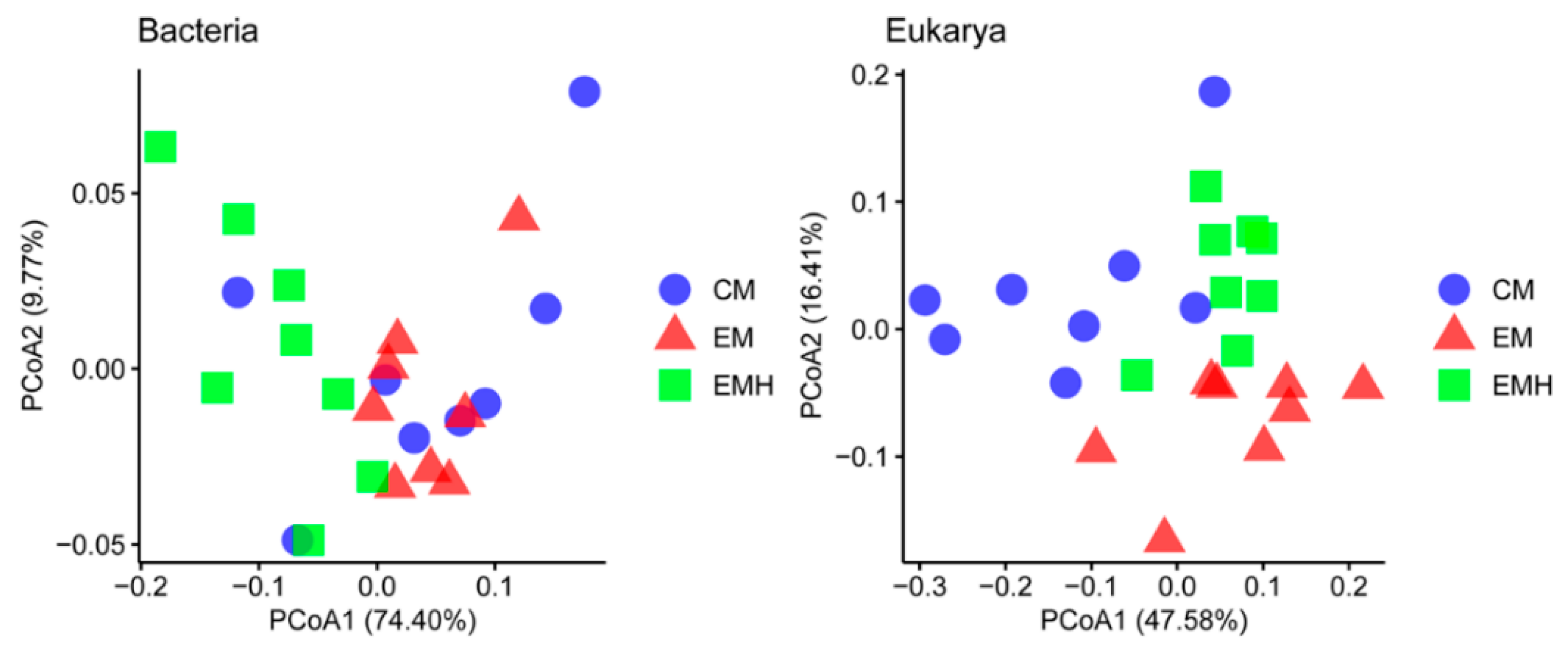
| Adonis | ANOSIM | MRPP | ||||
|---|---|---|---|---|---|---|
| F | P | R | P | δ | P | |
| Bacteria | ||||||
| Management (M) | 2.534 | 0.006 | 0.286 | 0.004 | 0.267 | 0.006 |
| Hedgerow (H) | 3.146 | 0.005 | 0.234 | 0.014 | 0.264 | 0.001 |
| M × H | 2.752 | 0.001 | 0.358 | 0.001 | 0.275 | 0.001 |
| Eukarya | ||||||
| Management (M) | 4.969 | 0.001 | 0.5972 | 0.001 | 0.325 | 0.001 |
| Hedgerow (H) | 2.095 | 0.024 | Neg | 0.517 | 0.338 | 0.005 |
| M × H | 3.673 | 0.001 | 0.367 | 0.001 | 0.312 | 0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, R.; Song, C.; Sun, Y.; Wang, Q.; Liu, H.; Wang, J. Agricultural Soil Organic Matters and Microbiome Are Shaped by Management and Hedgerows. Agronomy 2020, 10, 1316. https://doi.org/10.3390/agronomy10091316
Sun R, Song C, Sun Y, Wang Q, Liu H, Wang J. Agricultural Soil Organic Matters and Microbiome Are Shaped by Management and Hedgerows. Agronomy. 2020; 10(9):1316. https://doi.org/10.3390/agronomy10091316
Chicago/Turabian StyleSun, Renhua, Chengjun Song, Yuanze Sun, Qian Wang, Hongbin Liu, and Jie Wang. 2020. "Agricultural Soil Organic Matters and Microbiome Are Shaped by Management and Hedgerows" Agronomy 10, no. 9: 1316. https://doi.org/10.3390/agronomy10091316
APA StyleSun, R., Song, C., Sun, Y., Wang, Q., Liu, H., & Wang, J. (2020). Agricultural Soil Organic Matters and Microbiome Are Shaped by Management and Hedgerows. Agronomy, 10(9), 1316. https://doi.org/10.3390/agronomy10091316





