Recovery of Phosphorus from Waste Water Profiting from Biological Nitrogen Treatment: Upstream, Concomitant or Downstream Precipitation Alternatives
Abstract
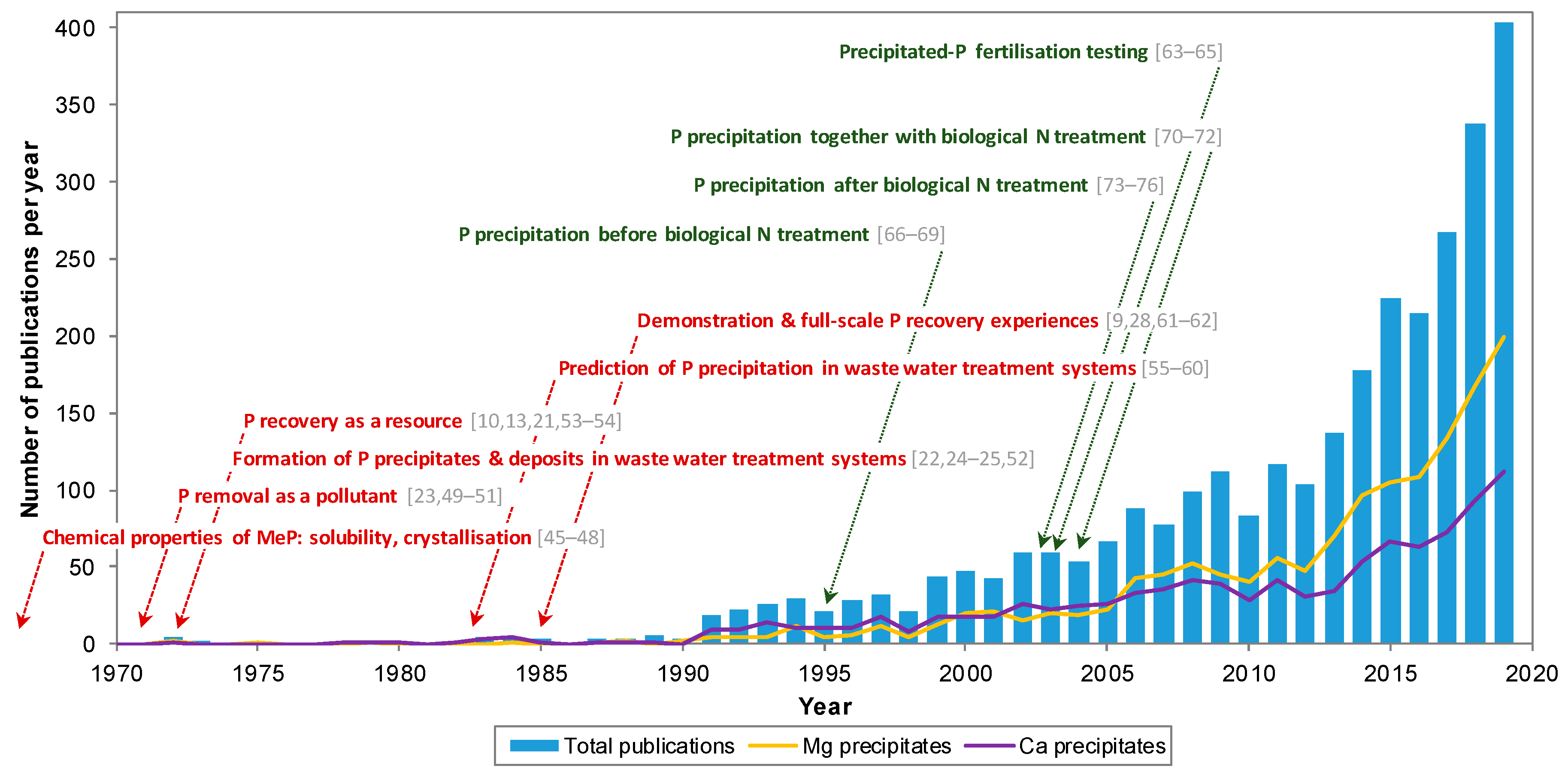
1. Introduction
2. Research Framework
3. Forms of Mineral Phosphate Precipitates in Waste Water Treatment
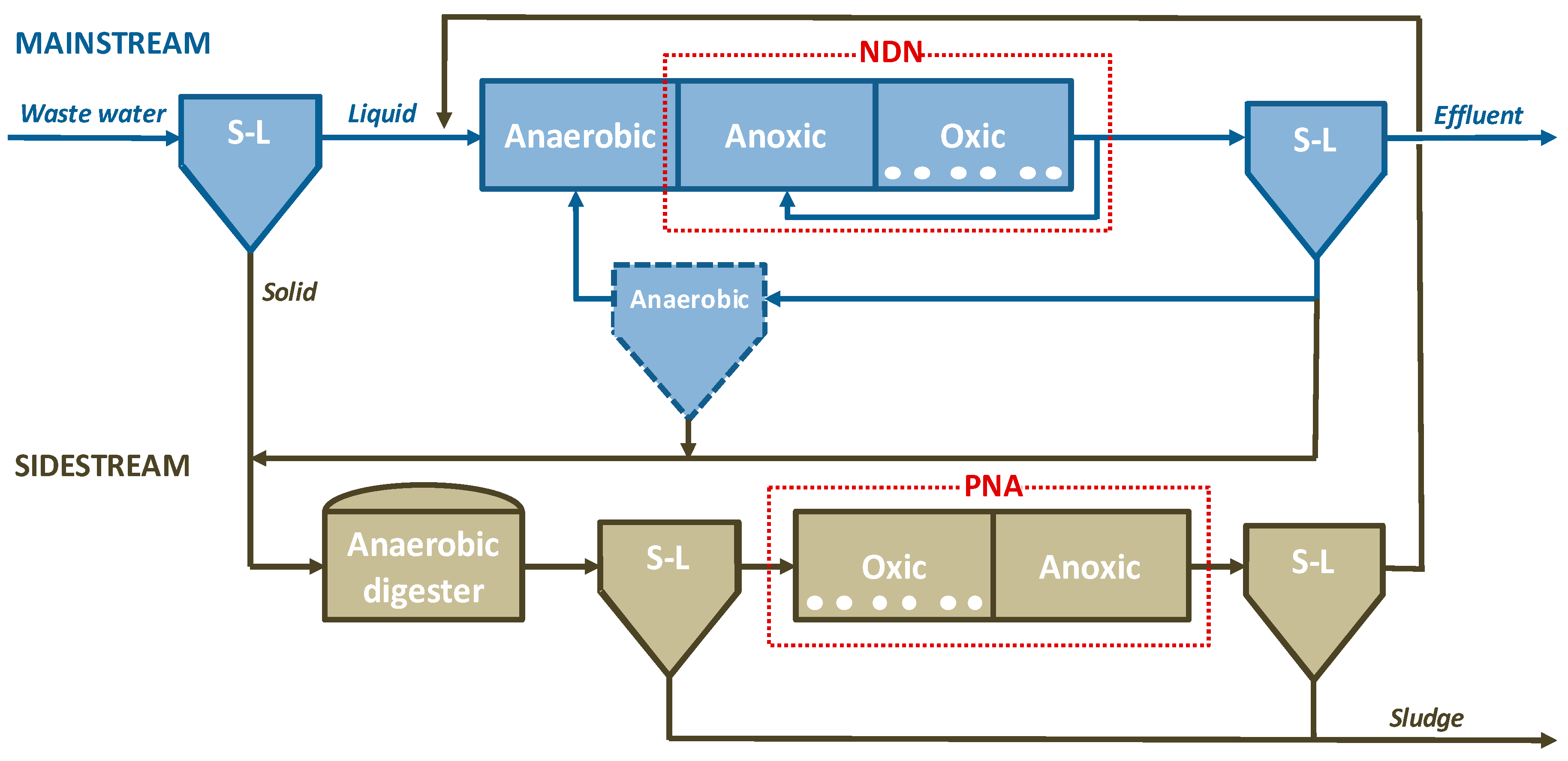
4. Linking Phosphorus Recovery to Biological Nitrogen Treatment
4.1. Phosphorus Recovery before Biological N Treatment (Upstream Configuration)
4.2. Phosphorus Recovery Together with Biological N Treatment (Concomitant Configuration)
4.2.1. Biologically Induced Phosphate Mineralisation
4.2.2. Phosphorus Recovery Experiences in Concomitant Configuration
4.2.3. Interaction between biological N treatment processes and soluble phosphate availability
4.3. Phosphorus Recovery after Biological N Treatment (Downstream Configuration)
5. Use of Recovered Phosphate Products
5.1. Quality Criteria for the Precipitated Phosphate Salts to be Covered by the EU Fertiliser Regulation
5.2. Expected Characteristics of the Precipitated Phosphate Salts Depending on the Production Process
5.3. Agronomic Efficiency Achieved when Using Precipitated Phosphate Salts
6. Conclusions
7. Future Research and Development
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cordell, D.; White, S. Peak phosphorus: Clarifying the key issues of a vigorous debate about long-term phosphorus security. Sustainability 2011, 3, 2027–2049. [Google Scholar] [CrossRef]
- Penuelas, J.; Sardans, J.; Alcaniz, J.M.; Poch, J.M. Increased eutrophication and nutrient imbalances in the agricultural soil of NE Catalonia, Spain. J. Environ. Biol. 2009, 30, 841–846. [Google Scholar] [PubMed]
- Cordell, D.; Drangert, J.-O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Change-Human Policy Dimens. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Gilbert, N. The disappearing nutrient. Nature 2009, 461, 716–718. [Google Scholar] [CrossRef]
- Elser, J.; Bennett, E. A broken biogeochemical cycle. Nature 2011, 478, 29–31. [Google Scholar] [CrossRef]
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions on the 2017 List of Critical Raw Materials for the EU. 2017. COM(2017) 490. Available online: https://ec.europa.eu/transparency/regdoc/rep/1/2017/EN/COM-2017-490-F1-EN-MAIN-PART-1.PDF (accessed on 27 May 2020).
- Withers, P.J.A.; Elser, J.J.; Hilton, J.; Ohtake, H.; Schippere, W.J.; van Dijkf, K.C. Greening the global phosphorus cycle: How green chemistry can help achieve planetary P sustainability. Green Chem. 2015, 17, 2087–2099. [Google Scholar] [CrossRef]
- Rittmann, B.E.; Mayer, B.; Westerhoff, P.; Edwards, M. Capturing the lost phosphorus. Chemosphere 2011, 84, 846–853. [Google Scholar] [CrossRef]
- Desmidt, E.; Ghyselbrecht, K.; Zhang, Y.; Pinoy, L.; Van der Bruggen, B.; Verstraete, W.; Rabaey, K.; Meesschaert, B. Global phosphorus scarcity and full-scale P-recovery techniques: A review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 336–384. [Google Scholar] [CrossRef]
- Egle, L.; Rechberger, H.; Krampe, J.; Zessner, M. Phosphorus recovery from municipal wastewater: An integrated comparative technological, environmental and economic assessment of P recovery technologies. Sci. Total Environ. 2016, 571, 522–542. [Google Scholar] [CrossRef]
- Snoeyink, V.L.; Jenkins, D. Water Chemistry; John Wiley & Sons Inc.: New York, NY, USA, 1980. [Google Scholar]
- Le Corre, K.S.; Valsami-Jones, E.; Hobbs, P.; Parsons, S.A. Phosphorus recovery from wastewater by struvite crystallization: A review. Crit. Rev. Environ. Sci. Technol. 2009, 39, 433–477. [Google Scholar] [CrossRef]
- Kataki, S.; West, H.; Clarke, M.; Baruah, D.C. Phosphorus recovery as struvite from farm, municipal and industrial waste: Feedstock suitability, methods and pre-treatments. Waste Manag. 2016, 49, 437–454. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Hahn, H.H.; Hoffmann, E. The effect of carbonate on the precipitation of calcium phosphate. Environ. Technol. 2002, 23, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Law, K.P.; Pagilla, K.R. Phosphorus recovery by methods beyond struvite precipitation. Water Environ. Res. 2018, 90, 840–850. [Google Scholar] [CrossRef]
- Wilfert, P.; Kumar, P.S.; Korving, L.; Witkamp, G.-J.; van Loosdrecht, M.C.M. The relevance of phosphorus and iron chemistry to the recovery of phosphorus from wastewater: A review. Environ. Sci. Technol. 2015, 16, 9400–9414. [Google Scholar] [CrossRef] [PubMed]
- Wilfert, P.; Dugulan, A.I.; Goubitz, K.; Korving, L.; Witkamp, G.J.; Van Loosdrecht, M.C.M. Vivianite as the main phosphate mineral in digested sewage sludge and its role for phosphate recovery. Water Res. 2018, 144, 312–321. [Google Scholar] [CrossRef]
- Wang, S.; An, J.; Wan, Y.; Du, Q.; Wang, X.; Cheng, X.; Li, N. Phosphorus competition in bioinduced vivianite recovery from wastewater. Environ. Sci. Technol. 2018, 52, 13863–13870. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, J.; Zhang, Q.; Aleem, M.; Fang, F.; Xue, Z.; Cao, J. Potentials and challenges of phosphorus recovery as vivianite from wastewater: A review. Chemosphere 2019, 226, 246–258. [Google Scholar] [CrossRef]
- Prot, T.; Wijdeveld, W.; Ekua Eshun, L.; Dugulan, A.I.; Goubitz, K.; Korving, L.; Van Loosdrecht, M.C.M. Full-scale increased iron dosage to stimulate the formation of vivianite and its recovery from digested sewage sludge. Water Res. 2020, 182, 115911. [Google Scholar] [CrossRef]
- Cornel, P.; Schaum, C. Phosphorus recovery from wastewater: Needs, technologies and costs. Water Sci. Technol. 2009, 59, 1069–1076. [Google Scholar] [CrossRef]
- Borgerding, J. Phosphate deposits in digestion systems. J. Water Pollut. Control Fed. 1972, 44, 813–819. [Google Scholar]
- Buchanan, J.R.; Mote, C.R.; Robinson, R.B. Struvite control by chemical treatment. Trans. ASAE 1994, 37, 1301–1308. [Google Scholar] [CrossRef]
- Doyle, J.D.; Parsons, S.A. Struvite formation, control and recovery. Water Res. 2002, 36, 3925–3940. [Google Scholar] [CrossRef]
- Sharp, R.; Vadiveloo, E.; Fergen, R.; Moncholi, M.; Pitt, P.; Wankmuller, D.; Latimer, R. A theoretical and practical evaluation of struvite control and recovery. Water Environ. Res. 2013, 85, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Geerts, S.; Marchi, A.; Weemaes, M. Full-scale phosphorus recovery from digested wastewater sludge in Belgium—Part II: Economic opportunities and risks. Water Sci. Technol. 2015, 71, 495–502. [Google Scholar] [CrossRef]
- Marchi, A.; Geerts, S.; Weemaes, M.; Wim, S.; Christine, V. Full-scale phosphorus recovery from digested waste water sludge in Belgium—Part I: Technical achievements and challenges. Water Sci. Technol. 2015, 71, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Ueno, Y.; Fujii, M. Three years experience of operating and selling recovered struvite from full scale plant. Environ. Technol. 2001, 22, 1373–1381. [Google Scholar] [CrossRef]
- Van Hulle, S.W.H.; Vandeweyer, H.J.P.; Meesschaert, B.D.; Vanrolleghem, P.A.; Dejans, P.; Dumoulin, A. Engineering aspects and practical application of autotrophic nitrogen removal from nitrogen rich streams. Chem. Eng. J. 2010, 162, 1–20. [Google Scholar] [CrossRef]
- Magrí, A.; Béline, F.; Dabert, P. Feasibility and interest of the anammox process as treatment alternative for anaerobic digester supernatants in manure processing—An overview. J. Environ. Manag. 2013, 131, 170–184. [Google Scholar] [CrossRef]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences—An application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef]
- Kumwimba, M.N.; Lotti, T.; Şenel, E.; Li, X.; Suanon, F. Anammox-based processes: How far have we come and what work remains? A review by bibliometric analysis. Chemosphere 2019, 238, 124627. [Google Scholar] [CrossRef]
- Ndegwa, P.M.; Hristov, A.N.; Arogo, J.; Sheffield, R.E. A review of ammonia emission mitigation techniques for concentrated animal feeding operations. Biosyst. Eng. 2008, 100, 453–469. [Google Scholar] [CrossRef]
- Fumasoli, A.; Etter, B.; Sterkele, B.; Morgenroth, E.; Udert, K.M. Operating a pilot-scale nitrification/distillation plant for complete nutrient recovery from urine. Water Sci. Technol. 2016, 73, 215–222. [Google Scholar] [CrossRef]
- Magrí, A. Research trends on nutrient management from digestates assessed using a bibliometric approach. Front. Sustain. Food Syst. 2018, 2, 40. [Google Scholar] [CrossRef]
- Egle, L.; Rechberger, H.; Zessner, M. Overview and description of technologies for recovering phosphorus from municipal wastewater. Resour. Conserv. Recycl. 2015, 105, 325–346. [Google Scholar] [CrossRef]
- Liu, Y.H.; Kumar, S.; Kwag, J.-H.; Ra, C.S. Magnesium ammonium phosphate formation, recovery and its application as valuable resources: A review. J. Chem. Technol. Biotechnol. 2013, 88, 181–189. [Google Scholar] [CrossRef]
- Reijnders, L. Phosphorus resources, their depletion and conservation, a review. Resour. Conserv. Recycl. 2014, 93, 32–49. [Google Scholar] [CrossRef]
- Kataki, S.; West, H.; Clarke, M.; Baruah, D.C. Phosphorus recovery as struvite: Recent concerns for use of seed, alternative Mg source, nitrogen conservation and fertilizer potential. Resour. Conserv. Recycl. 2016, 107, 142–156. [Google Scholar] [CrossRef]
- Tao, W.; Fattah, K.P.; Huchzermeier, M.P. Struvite recovery from anaerobically digested dairy manure: A review of application potential and hindrances. J. Environ. Manag. 2016, 169, 46–57. [Google Scholar] [CrossRef]
- Sena, M.; Hicks, A. Life cycle assessment review of struvite precipitation in wastewater treatment. Resour. Conserv. Recycl. 2018, 139, 194–204. [Google Scholar] [CrossRef]
- Chrispim, M.C.; Scholz, M.; Nolasco, M.A. Phosphorus recovery from municipal wastewater treatment: Critical review of challenges and opportunities for developing countries. J. Environ. Manag. 2019, 248, 109268. [Google Scholar] [CrossRef]
- Ghosh, S.; Lobanov, S.; Lo, V.K. An overview of technologies to recover phosphorus as struvite from wastewater: Advantages and shortcomings. Environ. Sci. Pollut. Res. 2019, 26, 19063–19077. [Google Scholar] [CrossRef]
- Li, B.; Huang, H.M.; Boiarkina, I.; Yu, W.; Huang, Y.F.; Wang, G.Q.; Young, B.R. Phosphorus recovery through struvite crystallisation: Recent developments in the understanding of operational factors. J. Environ. Manag. 2019, 248, 109254. [Google Scholar] [CrossRef]
- Clark, J.S. Solubility criteria for the existence of hydroxyapatite. Can. J. Chem. 1955, 33, 1696–1700. [Google Scholar] [CrossRef]
- Taylor, A.W.; Frazier, A.W.; Gurney, E.L. Solubility products of magnesium ammonium and magnesium potassium phosphates. Trans. Faraday Soc. 1963, 59, 1580–1584. [Google Scholar] [CrossRef]
- Luff, B.B.; Reed, R.B. Thermodynamic properties of magnesium potassium orthophosphate hexahydrate. J. Chem. Eng. Data 1980, 25, 310–312. [Google Scholar] [CrossRef]
- Ohlinger, K.N.; Young, T.M.; Schroeder, E.D. Predicting struvite formation in digestion. Water Res. 1998, 32, 3607–3614. [Google Scholar] [CrossRef]
- Jenkins, D.; Ferguson, J.F.; Menar, A.B. Chemical processes for phosphate removal. Water Res. 1971, 5, 369–389. [Google Scholar] [CrossRef]
- Yeoman, S.; Stephenson, T.; Lester, J.N.; Perry, R. The removal of phosphorus during wastewater treatment: A review. Environ. Pollut. 1988, 49, 183–233. [Google Scholar] [CrossRef]
- Bunce, J.T.; Ndam, E.; Ofiteru, I.D.; Moore, A.; Graham, D.W. A review of phosphorus removal technologies and their applicability to small-scale domestic wastewater treatment systems. Front. Environ. Sci. 2018, 6, 8. [Google Scholar] [CrossRef]
- Arvin, E. Calcium phosphate precipitation in a denitrifying biofilm—The conceptual basis. In Kinetics of Wastewater Treatment; Jenkins, S.H., Ed.; Pergamon Press: Oxford, UK, 1979; pp. 1–17. [Google Scholar] [CrossRef]
- Salutsky, M.L.; Shapiro, J.J.; Dunseth, M.G.; Ries, K.M. Ultimate disposal of phosphate from waste water by recovery as fertilizer. Effl. Water Treat. J. 1972, 12, 509–519. [Google Scholar]
- Stratful, I.; Brett, S.; Scrimshaw, M.B.; Lester, J.N. Biological phosphorus removal, its role in phosphorus recycling. Environ. Technol. 1999, 20, 681–695. [Google Scholar] [CrossRef]
- Arvin, E.; Kristensen, G.H. Phosphate precipitation in biofilms and flocs. Water Sci. Technol. 1983, 15, 65–85. [Google Scholar] [CrossRef]
- Vereecke, G.; Lemaître, J. Calculation of the solubility diagrams in the system Ca(OH)2-H3PO4-KOH-HNO3-CO2-H2O. J. Cryst. Growth. 1990, 104, 820–832. [Google Scholar] [CrossRef]
- Buchanan, J.R.; Mote, C.R.; Robinson, R.B. Thermodynamics of struvite formation. Trans. ASAE 1994, 37, 617–621. [Google Scholar] [CrossRef]
- Musvoto, E.V.; Wentzel, M.C.; Ekama, G.A. Integrated chemical-physical processes modelling–II. Simulating aeration treatment of anaerobic digester supernatants. Water Res. 2000, 34, 1868–1880. [Google Scholar] [CrossRef]
- Udert, K.M.; Larsen, T.A.; Gujer, W. Estimating the precipitation potential in urine-collecting systems. Water Res. 2003, 37, 2667–2677. [Google Scholar] [CrossRef]
- Gustafsson, J.P. Visual MINTEQ, ver. 3.1. 2018. Available online: https://vminteq.lwr.kth.se/ (accessed on 27 May 2020).
- van Dijk, J.C.; Braakensiek, H. Phosphate removal by crystallization in a fluidized bed. Water Sci. Technol. 1985, 17, 133–142. [Google Scholar] [CrossRef]
- Giesen, A. Crystallisation process enables environmental friendly phosphate removal at low costs. Environ. Technol. 1999, 20, 769–775. [Google Scholar] [CrossRef]
- Johnston, A.E.; Richards, I.R. Effectiveness of different precipitated phosphates as phosphorus sources for plants. Soil Use Manag. 2003, 19, 45–49. [Google Scholar] [CrossRef]
- Römer, W.; Steingrobe, B. Fertilizer effect of phosphorus recycling products. Sustainability 2018, 10, 1166. [Google Scholar] [CrossRef]
- Szymańska, M.; Szara, E.; Wąs, A.; Sosulski, T.; van Pruissen, G.W.P.; Cornelissen, R.L. Struvite–An innovative fertilizer from anaerobic digestate produced in a bio-refinery. Energies 2019, 12, 296. [Google Scholar] [CrossRef]
- Maekawa, T.; Liao, C.-M.; Feng, X.-D. Nitrogen and phosphorus removal for swine wastewater using intermittent aeration batch reactor followed by ammonium crystallization process. Water Res. 1995, 29, 2643–2650. [Google Scholar] [CrossRef]
- Caffaz, S.; Bettazzi, E.; Scaglione, D.; Lubello, C. An integrated approach in a municipal WWTP: Anaerobic codigestion of sludge with organic waste and nutrient removal from supernatant. Water Sci. Technol. 2008, 58, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Gao, Z.; Feng, H.; Zhang, Z.; Li, J.; Wang, A. Efficient nutrient recovery/removal from real source-separated urine by coupling vacuum thermal stripping with activated sludge processes. J. Clean. Prod. 2019, 220, 965–973. [Google Scholar] [CrossRef]
- Yang, S.; Xu, S.; Florentino, A.P.; Mohammed, A.; Ashbolt, N.J.; Liu, Y. Importance of controlling phosphate concentration in nitritation-anammox reactor operation. Environ. Sci. Water Res. Technol. 2019, 5, 1234–1243. [Google Scholar] [CrossRef]
- Johansson, S.; Ruscalleda, M.; Colprim, J. Phosphorus recovery through biologically induced precipitation by partial nitritation-anammox granular biomass. Chem. Eng. J. 2017, 327, 881–888. [Google Scholar] [CrossRef]
- Pambrun, V.; Paul, E.; Spérandio, M. Treatment of nitrogen and phosphorus in highly concentrated effluent in SBR and SBBR processes. Water Sci. Technol. 2004, 50, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Zhang, Y.; Xue, Y.; Li, Y.-Y. A new process for simultaneous nitrogen removal and phosphorus recovery using an anammox expanded bed reactor. Bioresour. Technol. 2018, 267, 201–208. [Google Scholar] [CrossRef]
- Vanotti, M.B.; Szogi, A.; Hunt, P.G. Extraction of soluble phosphorus from swine wastewater. Trans. ASAE 2003, 46, 1665–1674. [Google Scholar] [CrossRef]
- Suzin, L.; Antes, F.G.; Bedendo, G.C.; Bortoli, M.; Kunz, A. Chemical removal of phosphorus from swine effluent: The impact of previous effluent treatment technologies on process efficiency. Water Air Soil Pollut. 2018, 229, 341. [Google Scholar] [CrossRef]
- Tarragó, E.; Ruscalleda, M.; Colprim, J.; Balaguer, M.D.; Puig, S. Towards a methodology for recovering K-struvite from manure. J. Chem. Technol. Biotechnol. 2018, 93, 1558–1562. [Google Scholar] [CrossRef]
- Johansson, S.; Ruscalleda, M.; Saerens, B.; Colprim, J. Potassium recovery from centrate: Taking advantage of autotrophic nitrogen removal for multi-nutrient recovery. J. Chem. Technol. Biotechnol. 2019, 94, 819–828. [Google Scholar] [CrossRef]
- Agrawal, S.; Guest, J.S.; Cusick, R.D. Elucidating the impacts of initial supersaturation and seed crystal loading on struvite precipitation kinetics, fines production, and crystal growth. Water Res. 2018, 132, 252–259. [Google Scholar] [CrossRef]
- Forrest, A.L.; Fattah, K.P.; Mavinic, D.S.; Koch, F.A. Optimizing struvite production for phosphate recovery in WWTP. J. Environ. Eng. 2008, 134, 395–402. [Google Scholar] [CrossRef]
- Udert, K.M.; Larsen, T.A.; Biebow, M.; Gujer, W. Urea hydrolysis and precipitation dynamics in a urine-collecting system. Water Res. 2003, 37, 2571–2582. [Google Scholar] [CrossRef]
- Salutsky, M.L.; Steiger, R.P. Properties of fertilizer materials, metal potassium phosphates. J. Agric. Food Chem. 1964, 12, 486–491. [Google Scholar] [CrossRef]
- Wilsenach, J.A.; Schuurbiers, C.A.H.; van Loosdrecht, M.C.M. Phosphate and potassium recovery from source separated urine through struvite precipitation. Water Res. 2007, 41, 458–466. [Google Scholar] [CrossRef]
- Abbona, F.; Lundager-Madsen, H.E.; Boistelle, R. Crystallization of two magnesium phosphates, struvite and newberyite: Effect of pH and concentration. J. Cryst. Growth. 1982, 57, 6–14. [Google Scholar] [CrossRef]
- Boistelle, R.; Abbona, F.; Lundager-Madsen, H.E. On the transformation of struvite into newberyite in aqueous systems. Phys. Chem. Miner. 1983, 9, 216–222. [Google Scholar] [CrossRef]
- Tung, M.S. Calcium phosphates: Structure, composition, solubility, and stability. In Calcium Phosphates in Biological and Industrial Systems; Amjad, Z., Ed.; Kluwer Academic Publishers: Boston, MA, USA, 1998. [Google Scholar]
- Moreno, E.C.; Gregory, T.M.; Brown, W.E. Preparation and solubility of hydroxyapatite. J. Res. Natl. Bur. Stand. A Phys. Chem. 1968, 72A, 773–782. [Google Scholar] [CrossRef]
- Johnsson, M.S.-A.; Nancollas, G.H. The role of brushite and octacalcium phosphate in apatite formation. Crit. Rev. Oral Biol. Med. 1992, 3, 61–82. [Google Scholar] [CrossRef] [PubMed]
- Drouet, C. Why it may not work as planned, and how to conclusively identify apatite compounds. BioMed Res. Int. 2013, 2013, 490946. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.; Wang, C.; van Loosdrecht, M.C.M.; Hu, Y. Looking beyond struvite for P-recovery. Environ. Sci. Technol. 2013, 47, 4965–4966. [Google Scholar] [CrossRef]
- Taylor, A.W.; Frazier, A.W.; Gurney, E.L.; Smith, J.P. Solubility products of di- and trimagnesium phosphates and the dissociation of magnesium phosphate solutions. Trans. Faraday Soc. 1963, 59, 1585–1589. [Google Scholar] [CrossRef]
- Nriagu, J.O. Stability of vivianite and ion-pair formation in the system Fe3(PO4)2-H3PO4-H2O. Geochim. Cosmochim. Acta 1972, 36, 459–470. [Google Scholar] [CrossRef]
- Vlaeminck, S.E.; De Clippeleir, H.; Verstraete, W. Microbial resource management of one-stage partial nitritation/anammox. Microb. Biotechnol. 2012, 5, 433–448. [Google Scholar] [CrossRef] [PubMed]
- van Rensburg, P.; Musvoto, E.V.; Wentzel, M.C.; Ekama, G.A. Modelling multiple mineral precipitation in anaerobic digester liquor. Water Res. 2003, 37, 3087–3097. [Google Scholar] [CrossRef]
- Fattah, K.P.; Sabrina, N.; Mavinic, D.S.; Koch, F.A. Reducing operating costs for struvite formation with a carbon dioxide stripper. Water Sci. Technol. 2008, 58, 957–962. [Google Scholar] [CrossRef]
- Janus, H.M.; van der Roest, H.F. Don’t reject the idea of treating reject water. Water Sci. Technol. 1997, 35, 27–34. [Google Scholar] [CrossRef]
- van Loosdrecht, M.C.M.; Salem, S. Biological treatment of sludge digester liquids. Water Sci. Technol. 2006, 53, 11–20. [Google Scholar] [CrossRef]
- De Vrieze, J.; Smet, D.; Klok, J.; Colsen, J.; Angenent, L.T.; Vlaeminck, S.E. Thermophilic sludge digestion improves energy balance and nutrient recovery potential in full-scale municipal wastewater treatment plants. Bioresour. Technol. 2016, 218, 1237–1245. [Google Scholar] [CrossRef]
- van der Kooij, S.; van Vliet, B.J.M.; Stomph, T.J.; Sutton, N.B.; Anten, N.P.R.; Hoffland, E. Phosphorus recovered from human excreta: A socio-ecological-technical approach to phosphorus recycling. Resour. Conserv. Recycl. 2020, 157, 104744. [Google Scholar] [CrossRef]
- Shaddel, S.; Ucar, S.; Andreassen, J.-P.; Østerhus, S.W. Enhancing efficiency and economics of phosphorus recovery process by customizing the product based on sidestream characteristics—An alternative phosphorus recovery strategy. Water Sci. Technol. 2019, 79, 1777–1789. [Google Scholar] [CrossRef] [PubMed]
- Vanotti, M.B.; Ro, K.S.; Szogi, A.A.; Loughrin, J.H.; Millner, P.D. High-rate solid-liquid separation coupled with nitrogen and phosphorus treatment of swine manure: Effect on water quality. Front. Sustain. Food Syst. 2018, 2, 49. [Google Scholar] [CrossRef]
- Petzet, S.; Cornel, P. Towards a complete recycling of phosphorus in wastewater treatment—Options in Germany. Water Sci. Technol. 2011, 64, 29–35. [Google Scholar] [CrossRef]
- Piveteau, S.; Picard, S.; Dabert, P.; Daumer, M.-L. Dissolution of particulate phosphorus in pig slurry through biological acidification: A critical step for maximum phosphorus recovery as struvite. Water Res. 2017, 124, 693–701. [Google Scholar] [CrossRef]
- Kok, D.-J.D.; Pande, S.; van Lier, J.B.; Ortigara, A.R.C.; Savenije, H.; Uhlenbrook, S. Global phosphorus recovery from wastewater for agricultural reuse. Hydrol. Earth Syst. Sci. 2018, 22, 5781–5799. [Google Scholar] [CrossRef]
- Hao, X.-D.; van Loosdrecht, M.C.M. A proposed sustainable BNR plant with the emphasis on recovery of COD and phosphate. Water Sci. Technol. 2003, 48, 77–85. [Google Scholar] [CrossRef]
- Solon, K.; Flores-Alsina, X.; Mbamba, C.K.; Ikumi, D.; Volcke, E.I.P.; Vaneeckhaute, C.; Ekama, G.; Vanrolleghem, P.A.; Batstone, D.J.; Gernaey, K.V.; et al. Plant-wide modelling of phosphorus transformations in wastewater treatment systems: Impacts of control and operational strategies. Water Res. 2017, 113, 97–110. [Google Scholar] [CrossRef]
- Orner, K.; Cools, C.; Balaguer-Barbosa, M.; Zalivina, N.; Mihelcic, J.R.; Chen, G.; Cunningham, J.A. Energy recovery and nitrogen management from struvite precipitation effluent via microbial fuel cells. J. Environ. Eng. 2019, 145, 04018145. [Google Scholar] [CrossRef]
- Luo, Z.; Wang, D.; Yang, J.; Huang, H.; Su, G. Nitrogen removal from digested piggery wastewater using fermented superphosphate within the pretreatment stage and an MAP fertilizer pot test. J. Clean. Prod. 2019, 212, 372–380. [Google Scholar] [CrossRef]
- Zhu, Z.; Chen, W.; Tao, T.; Li, Y. A novel AAO-SBSPR process based on phosphorus mass balance for nutrient removal and phosphorus recovery from municipal wastewater. Water Res. 2018, 144, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Yao, S.; Chen, L.; Guan, D.; Zhang, Z.; Tian, X.; Wang, A.; Wang, G.; Yao, Q.; Peng, D.; Li, J. On-site nutrient recovery and removal from source-separated urine by phosphorus precipitation and short-cut nitrification-denitrification. Chemosphere 2017, 175, 210–218. [Google Scholar] [CrossRef]
- Ryu, H.-D.; Choo, Y.-D.; Kang, M.-K.; Lee, S.-I. Integrated application of struvite precipitation and biological treatment in treating autothermal thermophilic aerobic digestion supernatant liquid. Environ. Eng. Sci. 2014, 31, 167–175. [Google Scholar] [CrossRef]
- Mackey, H.R.; Zheng, Y.-S.; Tang, W.-T.; Dai, J.; Chen, G.-H. Combined seawater toilet flushing and urine separation for economic phosphorus recovery and nitrogen removal: A laboratory-scale trial. Water Sci. Technol. 2014, 70, 1065–1073. [Google Scholar] [CrossRef]
- Guadie, A.; Xia, S.; Zhang, Z.; Guo, W.; Ngo, H.H.; Hermanowicz, S.W. Simultaneous removal of phosphorus and nitrogen from sewage using a novel combo system of fluidized bed reactor–membrane bioreactor (FBR–MBR). Bioresour. Technol. 2013, 149, 276–285. [Google Scholar] [CrossRef]
- Shi, J.; Lu, X.; Yu, R.; Zhu, W. Nutrient removal and phosphorus recovery performances of a novel anaerobic-anoxic/nitrifying/induced crystallization process. Bioresour. Technol. 2012, 121, 183–189. [Google Scholar] [CrossRef]
- Shi, J.; Lu, X.; Yu, R.; Gu, Q.; Zhou, Y. Influence of wastewater composition on nutrient removal behaviors in the new anaerobic–anoxic/nitrifying/induced crystallization process. Saudi J. Biol. Sci. 2014, 21, 71–80. [Google Scholar] [CrossRef]
- Yuan, Q.; Oleszkiewicz, J.A. Biomass fermentation to augment biological phosphorus removal. Chemosphere 2010, 78, 29–34. [Google Scholar] [CrossRef]
- Ryu, H.-D.; Lee, S.-I. Application of struvite precipitation as a pretreatment in treating swine wastewater. Process Biochem. 2010, 45, 563–572. [Google Scholar] [CrossRef]
- Chen, M.; He, S.; Yi, Q.; Yang, M. Effect of chloride concentration on nitrogen removal from landfill leachate in sequencing batch reactor after MAP pretreatment. Water Sci. Technol. 2010, 62, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yi, L.; Ma, P.; Zhou, L. Industrial wastewater treatment by the combination of chemical precipitation and immobilized microorganism technologies. Environ. Eng. Sci. 2007, 24, 736–744. [Google Scholar] [CrossRef]
- Liao, C.-M.; Maekawa, T.; Feng, X.-D. Nitrogen and phosphorus removal for swine wastewater by ammonium crystallization and intermittent aeration process. J. Environ. Sci. Health Part B Pestic. Contam. Agric. Wastes 1995, 30, 733–758. [Google Scholar] [CrossRef] [PubMed]
- Connan, R.; Dabert, P.; Moya-Espinosa, M.; Bridoux, G.; Béline, F.; Magrí, A. Coupling of partial nitritation and anammox in two- and one-stage systems: Process operation, N2O emission and microbial community. J. Clean. Prod. 2018, 203, 559–573. [Google Scholar] [CrossRef]
- Desmidt, E.; Monballiu, A.; De Clippeleir, H.; Verstraete, W.; Meesschaert, B.D. Autotrophic nitrogen removal after ureolytic phosphate precipitation to remove both endogenous and exogenous nitrogen. Water Sci. Technol. 2013, 67, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Castro-Barros, C.M.; Daelman, M.R.J.; Mampaey, K.E.; van Loosdrecht, M.C.M.; Volcke, E.I.P. Effect of aeration regime on N2O emission from partial nitritation-anammox in a full-scale granular sludge reactor. Water Res. 2015, 68, 793–803. [Google Scholar] [CrossRef]
- Abma, W.R.; Driessen, W.; Haarhuis, R.; van Loosdrecht, M.C.M. Upgrading of sewage treatment plant by sustainable and cost-effective separate treatment of industrial wastewater. Water Sci. Technol. 2010, 61, 1715–1722. [Google Scholar] [CrossRef]
- Fujimoto, N.; Mizuochi, T.; Togami, Y. Phosphorus fixation in the sludge treatment system of a biological phosphorus removal process. Water Sci. Technol. 1991, 23, 635–640. [Google Scholar] [CrossRef]
- Monballiu, A.; Desmidt, E.; Ghyselbrecht, K.; Meesschaert, B. The inhibitory effect of inorganic carbon on phosphate recovery from upflow anaerobic sludge blanket reactor (UASB) effluent as calcium phosphate. Water Sci. Technol. 2018, 78, 2608–2615. [Google Scholar] [CrossRef]
- van der Houwen, J.A.M.; Valsami-Jones, E. The application of calcium phosphate precipitation chemistry to phosphorus recovery: The influence of organic ligands. Environ. Technol. 2001, 22, 1325–1335. [Google Scholar] [CrossRef]
- Taddeo, R.; Honkanen, M.; Kolppo, K.; Lepistö, R. Nutrient management via struvite precipitation and recovery from various agroindustrial wastewaters: Process feasibility and struvite quality. J. Environ. Manag. 2018, 212, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Henze, M.; Harremoës, P.; la Cour Jansen, J.; Arvin, E. Wastewater Treatment, Biological and Chemical Processes; Springer-Verlag: Berlin, Germany, 1995. [Google Scholar]
- Tünay, O.; Kabdasli, I.; Orhon, D.; Kolçak, S. Ammonia removal by magnesium ammonium phosphate precipitation in industrial wastewaters. Water Sci. Technol. 1997, 36, 225–228. [Google Scholar] [CrossRef]
- Kabdaşlı, I.; Tünay, O.; Özcan, P. Application of struvite precipitation coupled with biological treatment to slaughterhouse wastewaters. Environ. Technol. 2009, 30, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Kabdaşlı, I.; Tünay, O.; Çetin, M.Ş.; Ölmez, T. Assessment of magnesium ammonium phosphate precipitation for the treatment of leather tanning industry wastewaters. Water Sci. Technol. 2002, 46, 231–239. [Google Scholar] [CrossRef]
- Tansel, B.; Lunn, G.; Monje, O. Struvite formation and decomposition characteristics for ammonia and phosphorus recovery: A review of magnesium-ammonia-phosphate interactions. Chemosphere 2018, 194, 504–514. [Google Scholar] [CrossRef]
- Qiao, S.; Kanda, R.; Nishiyama, T.; Fujii, T.; Bhatti, Z.; Furukawa, K. Partial nitrification treatment for high ammonium wastewater from magnesium ammonium phosphate process of methane fermentation digester liquor. J. Biosci. Bioeng. 2010, 109, 124–129. [Google Scholar] [CrossRef]
- Pintucci, C.; Carballa, M.; Varga, S.; Sarli, J.; Peng, L.; Bousek, J.; Pedizzi, C.; Ruscalleda, M.; Tarragó, E.; Prat, D.; et al. The ManureEcoMine pilot installation: Advanced integration of technologies for the management of organics and nutrients in livestock waste. Water Sci. Technol. 2017, 75, 1281–1293. [Google Scholar] [CrossRef]
- Tarragó, E.; Sciarria, T.P.; Ruscalleda, M.; Colprim, J.; Balaguer, M.D.; Adani, F.; Puig, S. Effect of suspended solids and its role on struvite formation from digested manure. J. Chem. Technol. Biotechnol. 2018, 93, 2758–2765. [Google Scholar] [CrossRef]
- Hutnik, N.; Stanclik, A.; Piotrowski, K.; Matynia, A. Recovery of phosphates(V) from wastewaters of different chemical composition. Open Chem. 2019, 17, 1071–1079. [Google Scholar] [CrossRef]
- Moragaspitiya, C.; Rajapakse, J.; Millar, G.J. Effect of Ca:Mg ratio and high ammoniacal nitrogen on characteristics of struvite precipitated from waste activated sludge digester effluent. J. Environ. Sci. 2019, 86, 65–77. [Google Scholar] [CrossRef]
- Battistoni, P.; De Angelis, A.; Pavan, P.; Prisciandaro, M.; Cecchi, F. Phosphorus removal from a real anaerobic supernatant by struvite crystallization. Water Res. 2001, 35, 2167–2178. [Google Scholar] [CrossRef]
- Tarragó, E.; Puig, S.; Ruscalleda, M.; Balaguer, M.D.; Colprim, J. Controlling struvite particles’ size using the up-flow velocity. Chem. Eng. J. 2016, 302, 819–827. [Google Scholar] [CrossRef]
- Pastor, L.; Mangin, D.; Barat, R.; Seco, A. A pilot-scale study of struvite precipitation in a stirred tank reactor: Conditions influencing the process. Bioresour. Technol. 2008, 99, 6285–6291. [Google Scholar] [CrossRef] [PubMed]
- Karakashev, D.; Schmidt, J.E.; Angelidaki, I. Innovative process scheme for removal of organic matter, phosphorus and nitrogen from pig manure. Water Res. 2008, 42, 4083–4090. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.-C.; Yang, G.-F.; Yu, J.-J.; Zheng, P. The inhibition of the Anammox process: A review. Chem. Eng. J. 2012, 197, 67–79. [Google Scholar] [CrossRef]
- Carvajal-Arroyo, J.M.; Sun, W.; Sierra-Alvarez, R.; Field, J.A. Inhibition of anaerobic ammonium oxidizing (anammox) enrichment cultures by substrates, metabolites and common wastewater constituents. Chemosphere 2013, 91, 22–27. [Google Scholar] [CrossRef]
- Zhang, Z.-Z.; Hu, H.-Y.; Xu, J.-J.; Shi, Z.-J.; Deng, R.; Ji, Z.-Q.; Shi, M.-L.; Jin, R.-C. Effects of inorganic phosphate on a high-rate anammox system: Performance and microbial community. Ecol. Eng. 2017, 101, 201–210. [Google Scholar] [CrossRef]
- van de Graaf, A.A.; de Bruijn, P.; Robertson, L.A.; Jetten, M.S.M.; Kuenen, J.G. Autotrophic growth of anaerobic ammonium-oxidizing micro-organisms in a fluidized bed reactor. Microbiology 1996, 142, 2187–2196. [Google Scholar] [CrossRef]
- Connan, R.; Dabert, P.; Le Roux, S.; Chapleur, O.; Bridoux, G.; Vanotti, M.B.; Béline, F.; Magrí, A. Characterization of a combined batch-continuous procedure for the culture of anammox biomass. Ecol. Eng. 2017, 106, 231–241. [Google Scholar] [CrossRef]
- Dapena-Mora, A.; Vázquez-Padín, J.R.; Campos, J.L.; Mosquera-Corral, A.; Jetten, M.S.M.; Méndez, R. Monitoring the stability of an Anammox reactor under high salinity conditions. Biochem. Eng. J. 2010, 51, 167–171. [Google Scholar] [CrossRef]
- Lin, Y.M.; Lotti, T.; Sharma, P.K.; van Loosdrecht, M.C.M. Apatite accumulation enhances the mechanical property of anammox granules. Water Res. 2013, 47, 4556–4566. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gil, G.; Sougrat, R.; Behzad, A.R.; Lens, P.N.L.; Saikaly, P.E. Microbial community composition and ultrastructure of granules from a full-scale anammox reactor. Microb. Ecol. 2015, 70, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-Z.; Xu, J.-J.; Hu, H.-Y.; Shi, Z.-J.; Ji, Z.-Q.; Deng, R.; Shi, M.-L.; Jin, R.-C. Insight into the short- and long-term effects of inorganic phosphate on anammox granule property. Bioresour. Technol. 2016, 208, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Mann, S. Biomineralization: Principles and Concepts in Bioinorganic Materials Chemistry; Oxford University Press: New York, NY, USA, 2001. [Google Scholar]
- Rivadeneyra, M.A.; Ramos-Cormenzana, A.; García-Cervigón, A. Bacterial formation of struvite. Geomicrobiol. J. 1983, 3, 151–163. [Google Scholar] [CrossRef]
- Rivadeneyra, M.A.; Pérez-García, I.; Ramos-Cormenzana, A. Struvite precipitation by soil and fresh water bacteria. Curr. Microbiol. 1992, 24, 343–347. [Google Scholar] [CrossRef]
- Omelon, S.; Ariganello, M.; Bonucci, E.; Grynpas, M.; Nanci, A. A review of phosphate mineral nucleation in biology and geobiology. Calcif. Tissue Int. 2013, 93, 382–396. [Google Scholar] [CrossRef]
- Sánchez-Román, M.; Rivadeneyra, M.A.; Vasconcelos, C.; McKenzie, J.A. Biomineralization of carbonate and phosphate by moderately halophilic bacteria. FEMS Microbiol. Ecol. 2007, 61, 273–284. [Google Scholar] [CrossRef]
- Sinha, A.; Singh, A.; Kumar, S.; Khare, S.K.; Ramanan, A. Microbial mineralization of struvite: A promising process to overcome phosphate sequestering crisis. Water Res. 2014, 54, 33–43. [Google Scholar] [CrossRef]
- Li, H.; Yao, Q.-Z.; Yu, S.-H.; Huang, Y.-R.; Chen, X.-D.; Fu, S.-Q.; Zhou, G.-T. Bacterially mediated morphogenesis of struvite and its implication for phosphorus recovery. Am. Miner. 2017, 102, 381–390. [Google Scholar] [CrossRef]
- Rivadeneyra, M.A.; Ramos-Cormenzana, A.; García-Cervigón, A. Formation of bobierrite (magnesium phosphate) crystal aggregates by Acinetobacter sp. Mineral. J. 1987, 13, 443–447. [Google Scholar] [CrossRef]
- Rivadeneyra, M.A.; Martín-Algarra, A.; Sánchez-Román, M.; Sánchez-Navas, A.; Martín-Ramos, J.D. Amorphous Ca-phosphate precursors for Ca-carbonate biominerals mediated by Chromohalobacter marismortui. ISME J. 2010, 4, 922–932. [Google Scholar] [CrossRef] [PubMed]
- Rivadeneyra, M.A.; Delgado, R.; Párraga, J.; Ramos-Cormenzana, A.; Delgado, G. Precipitation of minerals by 22 species of moderately halophilic bacteria in artificial marine salts media: Influence of salt concentration. Folia Microbiol. 2006, 51, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Rivadeneyra, M.A.; Pérez-García, I.; Ramos-Cormenzana, A. The effect of incubation temperature on struvite formation by bacteria. Folia Microbiol. 1993, 38, 5–9. [Google Scholar] [CrossRef]
- Pérez-García, I.; Rivadeneyra, M.A.; Ramos-Cormenzana, A. The influence of pH on struvite formation by bacteria. Chemosphere 1989, 18, 1633–1638. [Google Scholar] [CrossRef]
- Ben Omar, N.; Entrena, M.; González-Muñoz, M.T.; Arias, J.M.; Huertas, F. Effects of pH and phosphate on the production of struvite by Myxococcus xanthus. Geomicrobiol. J. 1994, 12, 81–90. [Google Scholar] [CrossRef]
- Da Silva, S.; Bernet, N.; Delgenès, J.P.; Moletta, R. Effect of culture conditions on the formation of struvite by Myxococcus xanthus. Chemosphere 2000, 40, 1289–1296. [Google Scholar] [CrossRef]
- Rivadeneyra, M.A.; Pérez-García, I.; Ramos-Cormenzana, A. Influence of ammonium ion on bacterial struvite production. Geomicrobiol. J. 1992, 10, 125–137. [Google Scholar] [CrossRef]
- Pérez-García, I.; Rivadeneyra, M.A.; Quevedo-Sarmiento, J.; Ramos-Cormenzana, A. Struvite formation by Arthrobacter sp. and Pseudomonas sp.: The influence of agitation. Chemosphere 1990, 20, 243–251. [Google Scholar] [CrossRef]
- Soares, A.; Veesam, M.; Simoes, F.; Wood, E.; Parsons, S.A.; Stephenson, T. Bio-struvite: A new route to recover phosphorus from wastewater. Clean-Soil Air Water 2014, 42, 994–997. [Google Scholar] [CrossRef]
- Pratt, C.; Parsons, S.A.; Soares, A.; Martin, B.D. Biologically and chemically mediated adsorption and precipitation of phosphorus from wastewater. Curr. Opin. Biotechnol. 2012, 23, 890–896. [Google Scholar] [CrossRef]
- Bornemann, G.; Waßer, K.; Hauslage, J. The influence of nitrogen concentration and precipitation on fertilizer production from urine using a trickling filter. Life Sci. Space Res. 2018, 18, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Sperandio, M.; Pambrun, V.; Paul, E. Simultaneous removal of N and P in a SBR with production of valuable compounds: Application to concentrated wastewaters. Water Sci. Technol. 2008, 58, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ma, H.; Lin, L.; Cao, W.; Ouyang, T.; Li, Y.-Y. Enhanced simultaneous nitrogen and phosphorus removal performance by anammox-HAP symbiotic granules in the attached film expanded bed reactor. ACS Sustain. Chem. Eng. 2018, 6, 10989–10998. [Google Scholar] [CrossRef]
- Lin, L.; Zhang, Y.; Beckman, M.; Cao, W.; Ouyang, T.; Wang, S.; Li, Y.-Y. Process optimization of anammox-driven hydroxyapatite crystallization for simultaneous nitrogen removal and phosphorus recovery. Bioresour. Technol. 2019, 290, 121779. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Xue, Y.; Zhang, Y.; Kobayashi, T.; Kubota, K.; Li, Y.-Y. Simultaneous nitrogen removal and phosphorus recovery using an anammox expanded reactor operated at 25 °C. Water Res. 2020, 172, 115510. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, Y.; Webeck, E.; Li, Y.-Y. Towards more efficient nitrogen removal and phosphorus recovery from digestion effluent: Latest developments in the anammox-based process from the application perspective. Bioresour. Technol. 2020, 299, 122560. [Google Scholar] [CrossRef]
- Yuan, Z.; Pratt, S.; Batstone, D.J. Phosphorus recovery from wastewater through microbial processes. Curr. Opin. Biotechnol. 2012, 23, 878–883. [Google Scholar] [CrossRef]
- Yilmaz, G.; Lemaire, R.; Keller, J.; Yuan, Z. Simultaneous nitrification, denitrification, and phosphorus removal from nutrient-rich industrial wastewater using granular sludge. Biotechnol. Bioeng. 2008, 100, 529–541. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, A.; Rodriguez-Sanchez, A.; Rivadeneyra, M.A.; Rivadeneyra, A.; Martin-Ramos, D.; Vahala, R.; Gonzalez-Lopez, J. 16S rRNA gene-based characterization of bacteria potentially associated with phosphate and carbonate precipitation from a granular autotrophic nitrogen removal bioreactor. Appl. Microbiol. Biotechnol. 2017, 101, 817–829. [Google Scholar] [CrossRef]
- Simoes, F.; Vale, P.; Stephenson, T.; Soares, A. The role of pH on the biological struvite production in digested sludge dewatering liquors. Sci. Rep. 2018, 8, 7225. [Google Scholar] [CrossRef]
- Leng, Y.; Colston, R.; Soares, A. Understanding the biochemical characteristics of struvite bio-mineralising microorganisms and their future in nutrient recovery. Chemosphere 2020, 247, 125799. [Google Scholar] [CrossRef] [PubMed]
- Rivadeneyra, A.; Gonzalez-Martinez, A.; Gonzalez-Lopez, J.; Martin-Ramos, D.; Martinez-Toledo, M.V.; Rivadeneyra, M.A. Precipitation of phosphate minerals by microorganisms isolated from a fixed-biofilm reactor used for the treatment of domestic wastewater. Int. J. Environ. Res. Public Health 2014, 11, 3689–3704. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, A.; Leyva-Díaz, J.C.; Rodriguez-Sanchez, A.; Muñoz-Palazon, B.; Rivadeneyra, A.; Poyatos, J.M.; Rivadeneyra, M.A.; Martinez-Toledo, M.V. Isolation and metagenomic characterization of bacteria associated with calcium carbonate and struvite precipitation in a pure moving bed biofilm reactor-membrane bioreactor. Biofouling 2015, 31, 333–348. [Google Scholar] [CrossRef]
- Oa, S.W.; Choi, E. Phosphorus removal from nightsoil with sequencing batch reactor (SBR). Water Sci. Technol. 1997, 36, 55–60. [Google Scholar] [CrossRef]
- Daumer, M.L.; Béline, F.; Guiziou, F.; Sperandio, M. Effect of nitrification on phosphorus dissolving in a piggery effluent treated by a sequencing batch reactor. Biosyst. Eng. 2007, 96, 551–557. [Google Scholar] [CrossRef]
- Daumer, M.L.; Béline, F.; Guiziou, F.; Sperandio, M. Influence of pH and biological metabolism on dissolved phosphorus during biological treatment of piggery wastewater. Biosyst. Eng. 2007, 96, 379–386. [Google Scholar] [CrossRef]
- Daumer, M.L.; Beline, F.; Guiziou, F. Fate of phosphorus from biological aerobic treatment of pig slurry. By-products characterization and recovery. Environ. Technol. 2003, 24, 1323–1330. [Google Scholar] [CrossRef]
- Choi, E.; Yu, Y.; Cui, M.; Yun, Z.; Min, K. Effect of biologically mediated pH change on phosphorus removal in BNR system for piggery waste treatment. J. Environ. Sci. Health Part A 2008, 43, 154–160. [Google Scholar] [CrossRef]
- Carlsson, H.; Aspegren, H.; Lee, N.; Hilmer, A. Calcium phosphate precipitation in biological phosphorus removal systems. Water Res. 1997, 31, 1047–1055. [Google Scholar] [CrossRef]
- Maurer, M.; Abramovich, D.; Siegrist, H.; Gujer, W. Kinetics of biologically induced phosphorus precipitation in waste-water treatment. Water Res. 1999, 33, 484–493. [Google Scholar] [CrossRef]
- Maurer, M.; Boller, M. Modelling of phosphorus precipitation in wastewater treatment plants with enhanced biological phosphorus removal. Water Sci. Technol. 1999, 39, 147–163. [Google Scholar] [CrossRef]
- Wong, P.Y.; Cheng, K.Y.; Kaksonen, A.H.; Sutton, D.C.; Ginige, M.P. A novel post denitrification configuration for phosphorus recovery using polyphosphate accumulating organisms. Water Res. 2013, 47, 6488–6495. [Google Scholar] [CrossRef] [PubMed]
- Tomei, M.C.; Stazi, V.; Daneshgar, S.; Capodaglio, A.G. Holistic approach to phosphorus recovery from urban wastewater: Enhanced biological removal combined with precipitation. Sustainability 2020, 12, 575. [Google Scholar] [CrossRef]
- Li, Y.; Zou, J.; Zhang, L.; Sun, J. Aerobic granular sludge for simultaneous accumulation of mineral phosphorus and removal of nitrogen via nitrite in wastewater. Bioresour. Technol. 2014, 154, 178–184. [Google Scholar] [CrossRef]
- Sepúlveda-Mardones, M.; Campos, J.L.; Magrí, A.; Vidal, G. Moving forward in the use of aerobic granular sludge for municipal wastewater treatment: An overview. Rev. Environ. Sci. Bio-Technol. 2019, 18, 741–769. [Google Scholar] [CrossRef]
- de Kreuk, M.K.; Heijnen, J.J.; van Loosdrecht, M.C.M. Simultaneous COD, nitrogen and phosphate removal by aerobic granular sludge. Biotechnol. Bioeng. 2005, 90, 761–769. [Google Scholar] [CrossRef]
- Mañas, A.; Biscans, B.; Spérandio, M. Biologically induced phosphorus precipitation in aerobic granular sludge process. Water Res. 2011, 45, 3776–3786. [Google Scholar] [CrossRef]
- Mañas, A.; Pocquet, M.; Biscans, B.; Sperandio, M. Parameters influencing calcium phosphate precipitation in granular sludge sequencing batch reactor. Chem. Eng. Sci. 2012, 77, 165–175. [Google Scholar] [CrossRef]
- Mañas, A.; Spérandio, M.; Decker, F.; Biscans, B. Location and chemical composition of microbially induced phosphorus precipitates in anaerobic and aerobic granular sludge. Environ. Technol. 2012, 33, 2195–2209. [Google Scholar] [CrossRef]
- Isanta, E.; Suárez-Ojeda, M.E.; Val del Río, A.; Morales, N.; Pérez, J.; Carrera, J. Long term operation of a granular sequencing batch reactor at pilot scale treating a low-strength wastewater. Chem. Eng. J. 2012, 198–199, 163–170. [Google Scholar] [CrossRef]
- Lu, Y.-Z.; Wang, H.-F.; Kotsopoulos, T.A.; Zeng, R.J. Advanced phosphorus recovery using a novel SBR system with granular sludge in simultaneous nitrification, denitrification and phosphorus removal process. Appl. Microbiol. Biotechnol. 2016, 100, 4367–4374. [Google Scholar] [CrossRef] [PubMed]
- Hao, W.; Li, Y.; Lv, J.; Chen, L.; Zhu, J. The biological effect of metal ions on the granulation of aerobic granular activated sludge. J. Environ. Sci. 2016, 44, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Barat, R.; Montoya, T.; Seco, A.; Ferrer, J. The role of potassium, magnesium and calcium in the enhanced biological phosphorus removal treatment plants. Environ. Technol. 2005, 26, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.M.; Bassin, J.P.; van Loosdrecht, M.C.M. The contribution of exopolysaccharides induced struvites accumulation to ammonium adsorption in aerobic granular sludge. Water Res. 2012, 46, 986–992. [Google Scholar] [CrossRef] [PubMed]
- Tervahauta, T.; van der Weijden, R.D.; Flemming, R.L.; Hernández-Leal, L.; Zeeman, G.; Buisman, C.J.N. Calcium phosphate granulation in anaerobic treatment of black water: A new approach to phosphorus recovery. Water Res. 2014, 48, 632–642. [Google Scholar] [CrossRef]
- Cunha, J.R.; Tervahauta, T.; van der Weijden, R.D.; Hernández-Leal, L.; Zeeman, G.; Buisman, C.J.N. Simultaneous recovery of calcium phosphate granules and methane in anaerobic treatment of black water: Effect of bicarbonate and calcium fluctuations. J. Environ. Manag. 2018, 216, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Cunha, J.R.; Morais, S.; Silva, J.C.; van der Weijden, R.D.; Leal, L.H.; Zeeman, G.; Buisman, C.J.N. Bulk pH and carbon source are key factors for calcium phosphate granulation. Environ. Sci. Technol. 2019, 53, 1334–1343. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, G.W.; Kunz, A.; Steinmetz, R.L.R.; Szogi, A.; Vanotti, M.; Flores, E.M.d.M.; Dressler, V.L. Chemical phosphorus removal: A clean strategy for piggery wastewater management in Brazil. Environ. Technol. 2012, 33, 1677–1683. [Google Scholar] [CrossRef]
- Monballiu, A.; Desmidt, E.; Ghyselbrecht, K.; Meesschaert, B. Phosphate recovery as hydroxyapatite from nitrified UASB effluent at neutral pH in a CSTR. J. Environ. Chem. Eng. 2018, 6, 4413–4422. [Google Scholar] [CrossRef]
- Monballiu, A.; Ghyselbrecht, K.; Crabeels, X.; Meesschaert, B. Calcium phosphate precipitation in nitrified wastewater from the potato-processing industry. Environ. Technol. 2019, 40, 2250–2266. [Google Scholar] [CrossRef]
- Szogi, A.A.; Vanotti, M.B. Removal of phosphorus from livestock effluents. J. Environ. Qual. 2009, 38, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Son, D.-J.; Kim, W.-Y.; Jung, B.-R.; Hong, K.-H. Removal of nitrogen and phosphate from fertilizer industry wastewater by magnesium ammonium phosphate formation and electrochemical treatment. Int. J. Electrochem. Sci. 2019, 14, 3153–3167. [Google Scholar] [CrossRef]
- Schuiling, R.D.; Andrade, A. Recovery of struvite from calf manure. Environ. Technol. 1999, 20, 765–768. [Google Scholar] [CrossRef]
- Vanotti, M.B.; Millner, P.D.; Hunt, P.G.; Ellison, A.Q. Removal of pathogen and indicator microorganisms from liquid swine manure in multi-step biological and chemical treatment. Bioresour. Technol. 2005, 96, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Mazlum, N.; İkizoğlu, B. Nutrient removal by chemical post treatment with lime following the biological stage. Pol. J. Environ. Stud. 2018, 27, 1187–1195. [Google Scholar] [CrossRef]
- Vanotti, M.B.; Dube, P.J.; Szogi, A.A.; García-González, M.C. Recovery of ammonia and phosphate minerals from swine wastewater using gas-permeable membranes. Water Res. 2017, 112, 137–146. [Google Scholar] [CrossRef]
- Ciceri, D.; Manning, D.A.C.; Allanore, A. Historical and technical developments of potassium resources. Sci. Total Environ. 2015, 502, 590–601. [Google Scholar] [CrossRef]
- Manning, D.A.C. How will minerals feed the world in 2050? Proc. Geol. Assoc. 2015, 126, 14–17. [Google Scholar] [CrossRef]
- Nakao, S.; Nishio, T.; Kanjo, Y. Simultaneous recovery of phosphorus and potassium as magnesium potassium phosphate from synthetic sewage sludge effluent. Environ. Technol. 2017, 38, 2416–2426. [Google Scholar] [CrossRef]
- Le, V.-G.; Vu, C.-T.; Shih, Y.-J.; Bui, X.-T.; Liao, C.-H.; Huang, Y.-H. Phosphorus and potassium recovery from human urine using a fluidized bed homogeneous crystallization (FBHC) process. Chem. Eng. J. 2020, 384. [Google Scholar] [CrossRef]
- Xu, K.; Wang, C.; Wang, X.; Qian, Y. Laboratory experiments on simultaneous removal of K and P from synthetic and real urine for nutrient recycle by crystallization of magnesium–potassium–phosphate–hexahydrate in a draft tube and baffle reactor. Chemosphere 2012, 88, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Li, J.; Li, B.; Zhang, D.; Zhao, N.; Tang, S. Comparison of different K-struvite crystallization processes for simultaneous potassium and phosphate recovery from source-separated urine. Sci. Total Environ. 2019, 651, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Wang, C.; Liu, H.; Qian, Y. Simultaneous removal of phosphorus and potassium from synthetic urine through the precipitation of magnesium potassium phosphate hexahydrate. Chemosphere 2011, 84, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Li, J.; Zheng, M.; Zhang, C.; Xie, T.; Wang, C. The precipitation of magnesium potassium phosphate hexahydrate for P and K recovery from synthetic urine. Water Res. 2015, 80, 71–79. [Google Scholar] [CrossRef]
- Huang, H.; Zhang, D.; Wang, W.; Li, B.; Zhao, N.; Li, J.; Dai, J. Alleviating Na+ effect on phosphate and potassium recovery from synthetic urine by K-struvite crystallization using different magnesium sources. Sci. Total Environ. 2019, 655, 211–219. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, K.-n.; Li, J.-y.; Wang, C.-w.; Zheng, M. Recovery of phosphorus and potassium from source-separated urine using a fluidized bed reactor: Optimization operation and mechanism modeling. Ind. Eng. Chem. Res. 2017, 56, 3033–3039. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, K.; Zheng, M.; Li, J.; Wang, C. Factors affecting the crystal size of struvite-K formed in synthetic urine using a stirred reactor. Ind. Eng. Chem. Res. 2018, 57, 17301–17309. [Google Scholar] [CrossRef]
- Huygens, D.; Saveyn, H.G.M. Agronomic efficiency of selected phosphorus fertilisers derived from secondary raw materials for European agriculture. A meta-analysis. Agron. Sustain. Dev. 2018, 38, 52. [Google Scholar] [CrossRef]
- Schipper, W.J.; Klapwijk, A.; Potjer, B.; Rulkens, W.H.; Temmink, B.G.; Kiestra, F.D.G.; Lijmbach, A.C.M. Phosphate recycling in the phosphorus industry. Environ. Technol. 2001, 22, 1337–1345. [Google Scholar] [CrossRef]
- European Parliament and Council of the European Union. Regulation (EU) 2019/1009, Laying Down Rules on the Making Available on the Market of EU Fertilising Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and Repealing Regulation (EC) No 2003/2003. 2019. OJ L-170. pp. 1–114. Available online: http://data.europa.eu/eli/reg/2019/1009/oj (accessed on 27 May 2020).
- Huygens, S.; Saveyn, H.; Tonini, D.; Eder, P.; Delgado Sancho, L. Technical Proposals for Selected New Fertilising Materials under the Fertilising Products Regulation (Regulation (EU) 2019/1009)—Process and Quality Criteria, and Assessment of Environmental and Market Impacts for Precipitated Phosphate Salts & Derivates, Thermal Oxidation Materials & Derivates and Pyrolysis & Gasification Materials; JRC Science for Policy Report, Publications Office of the European Union: Luxembourg, 2019; EUR 29841 EN. [Google Scholar] [CrossRef]
- Giner Santonja, G.; Georgitzikis, K.; Scalet, B.M.; Montobbio, P.; Roudier, S.; Delgado Sancho, L. Best Available Techniques (BAT) Reference Document for the Intensive Rearing of Poultry or Pigs. Industrial Emissions Directive 2010/75/EU. Integrated Pollution Prevention and Control; JRC Science for Policy Report; Publications Office of the European Union: Luxembourg, 2017; EUR 28674 EN. [Google Scholar] [CrossRef]
- Boxall, A.B.A. New and Emerging Water Pollutants Arising from Agriculture; OECD: Paris, France, 2012; Available online: http://www.oecd.org/greengrowth/sustainable-agriculture/49848768.pdf (accessed on 27 May 2020).
- Lou, Y.; Ye, Z.-L.; Chen, S.; Wei, Q.; Zhang, J.; Ye, X. Influences of dissolved organic matters on tetracyclines transport in the process of struvite recovery from swine wastewater. Water Res. 2018, 134, 311–326. [Google Scholar] [CrossRef]
- Ye, Z.-L.; Deng, Y.; Lou, Y.; Ye, X.; Chen, S. Occurrence of veterinary antibiotics in struvite recovery from swine wastewater by using a fluidized bed. Front. Environ. Sci. Eng. 2018, 12, 7. [Google Scholar] [CrossRef]
- Uysal, A.; Yilmazel, Y.D.; Demirer, G.N. The determination of fertilizer quality of the formed struvite from effluent of a sewage sludge anaerobic digester. J. Hazard. Mater. 2010, 181, 248–254. [Google Scholar] [CrossRef] [PubMed]
- STOWA. Verkenning van de Kwaliteit van Struviet uit de Communale Afvalwaterketen (in dutch). STOWA: Amersfoort, Netherlands. 2015. Report 2015-34. Available online: https://www.stowa.nl/sites/default/files/assets/PUBLICATIES/Publicaties%202015/STOWA%202015-34.pdf (accessed on 27 May 2020).
- Eriksson, J. Concentrations of 61 Trace Elements in Sewage Sludge, Farmyard Manure, Mineral Fertiliser, Precipitation and in Oil and Crops; Swedish Environmental Protection Agency: Stockholm, Sweden, 2001; Available online: http://swedishepa.se/Documents/publikationer/620-6246-8.pdf (accessed on 27 May 2020).
- Gendebien, A.; Ferguson, R.; Brink, J.; Horth, H.; Sullivan, M.; Davis, R.; Brunet, H.; Dalimier, F.; Landrea, B.; Krack, D.; et al. Survey of Wastes Spread on Land—Final Report; European Commission-DG Environment, Office for Official Publications of the European Communities: Luxembourg, 2001; Available online: https://ec.europa.eu/environment/waste/studies/compost/landspreading.pdf (accessed on 27 May 2020).
- Karvelas, M.; Katsoyiannis, A.; Samara, C. Occurrence and fate of heavy metals in the wastewater treatment process. Chemosphere 2003, 53, 1201–1210. [Google Scholar] [CrossRef]
- Ewert, W.; Hermanussen, O.; Kabbe, C.; Mêlè, C.; Niewersch, C.; Paillard, H.; Stössel, E.; Stemann, J. Comparison of Sludge Related Processes. Sustainable Sewage Sludge Management Fostering Phosphorus Recovery and Energy Efficiency, P-REX Project, FP7-Environment. 2014. Deliverable 5.1. Available online: https://zenodo.org/record/242550/files/P-REX%20D5.1%20Comparison%20of%20sludge%20related%20processes.pdf?download=1 (accessed on 27 May 2020).
- Kahiluoto, H.; Kuisma, M.; Ketoja, E.; Salo, T.; Heikkinen, J. Phosphorus in manure and sewage sludge more recyclable than in soluble inorganic fertilizer. Environ. Sci. Technol. 2015, 49, 2115–2122. [Google Scholar] [CrossRef]
- Kratz, S.; Vogel, C.; Adam, C. Agronomic performance of P recycling fertilizers and methods to predict it: A review. Nutr. Cycl. Agroecosyst. 2019, 115, 1–39. [Google Scholar] [CrossRef]
- Weissengruber, L.; Möller, K.; Puschenreiter, M.; Friedel, J.K. Long-term soil accumulation of potentially toxic elements and selected organic pollutants through application of recycled phosphorus fertilizers for organic farming conditions. Nutr. Cycl. Agroecosyst. 2018, 110, 427–449. [Google Scholar] [CrossRef]
- Yli-Halla, M.; Schick, J.; Kratz, S.; Schnug, E. Determination of plant available P in soil. In Phosphorus in Agriculture: 100% Zero; Schnug, E., De Kok, L.J., Eds.; Springer Science & Business Media, B.V.: Dordrecht, Germany, 2016; pp. 63–93. [Google Scholar] [CrossRef]
- Cabeza, R.; Steingrobe, B.; Römer, W.; Claassen, N. Effectiveness of recycled P products as P fertilizers, as evaluated in pot experiments. Nutr. Cycl. Agroecosyst. 2011, 91, 173–184. [Google Scholar] [CrossRef]
- Degryse, F.; Baird, R.; da Silva, R.C.; McLaughlin, M.J. Dissolution rate and agronomic effectiveness of struvite fertilizers—Effect of soil pH, granulation and base excess. Plant Soil 2017, 410, 139–152. [Google Scholar] [CrossRef]
- Möller, K.; Oberson, A.; Bünemann, E.K.; Cooper, J.; Friedel, J.K.; Glæsner, N.; Hörtenhuber, S.; Løes, A.-K.; Mäder, P.; Meyer, G.; et al. Chapter four—Improved phosphorus recycling in organic farming: Navigating between constraints. Adv. Agron. 2018, 147, 159–237. [Google Scholar] [CrossRef]
- Brod, E.; Øgaard, A.F.; Haraldsen, T.K.; Krogstad, T. Waste products as alternative phosphorus fertilisers part II: Predicting P fertilization effects by chemical extraction. Nutr. Cycl. Agroecosyst. 2015, 103, 187–199. [Google Scholar] [CrossRef]
- Antonini, S.; Arias, M.A.; Eichert, T.; Clemens, J. Greenhouse evaluation and environmental impact assessment of different urine-derived struvite fertilizers as phosphorus sources for plants. Chemosphere 2012, 89, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- European Chemicals Agency. Guidance for Identification and Naming of Substances under REACH and CLP, Version 2.1; ECHA: Helsinki, Finland, 2017. [CrossRef]
- Cabeza, R.A. Phosphorus Dynamics in Soil and Plant Availability of Fertilizers from Phosphorus Recycling Evaluated in Field and Pot Experiments; Georg-August-Universität Göttingen: Göttingen, Germany; Cuvillier Verlag: Göttingen, Germany, 2010. [Google Scholar]
- Wollmann, I.; Gauro, A.; Müller, T.; Möller, K. Phosphorus bioavailability of sewage sludge-based recycled fertilizers. J. Plant Nutr. Soil Sci. 2018, 181, 158–166. [Google Scholar] [CrossRef]
- Hall, R.L.; Staal, L.B.; Macintosh, K.A.; McGrath, J.W.; Bailey, J.; Black, L.; Nielsen, U.G.; Reitzel, K.; Williams, P.N. Phosphorus speciation and fertiliser performance characteristics: A comparison of waste recovered struvites from global sources. Geoderma 2020, 362, 114096. [Google Scholar] [CrossRef]
- Ackerman, J.N.; Zvomuya, F.; Cicek, N.; Flaten, D. Evaluation of manure-derived struvite as a phosphorus source for canola. Can. J. Plant Sci. 2013, 93, 419–424. [Google Scholar] [CrossRef]
- El Diwani, G.; El Rafie, S.; El Ibiari, N.N.; El-Aila, H.I. Recovery of ammonia nitrogen from industrial wastewater treatment as struvite slow releasing fertilizer. Desalination 2007, 214, 200–214. [Google Scholar] [CrossRef]
- Vaneeckhaute, C.; Janda, J.; Vanrolleghem, P.A.; Tack, F.M.G.; Meers, E. Phosphorus use efficiency of bio-based fertilizers: Bioavailability and fractionation. Pedosphere 2016, 26, 310–325. [Google Scholar] [CrossRef]
- Vogel, C.; Rivard, C.; Wilken, V.; Muskolus, A.; Adam, C. Performance of secondary P-fertilizers in pot experiments analyzed by phosphorus X-ray absorption near edge structure (XANES) spectroscopy. Ambio 2018, 47, S62–S72. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Nancollas, G.H. Calcium orthophosphates: Crystallization and dissolution. Chem. Rev. 2008, 108, 4628–4669. [Google Scholar] [CrossRef] [PubMed]
- Bauer, P.J.; Szogi, A.A.; Vanotti, M.B. Agronomic effectiveness of calcium phosphate recovered from liquid swine manure. Agron. J. 2007, 99, 1352–1356. [Google Scholar] [CrossRef]
- Meyer, G.; Frossard, E.; Mäder, P.; Nanzer, S.; Randall, D.G.; Udert, K.M.; Oberson, A. Water soluble phosphate fertilizers for crops grown in calcareous soils—An outdated paradigm for recycled phosphorus fertilizers? Plant Soil 2018, 424, 367–388. [Google Scholar] [CrossRef]
- Arai, Y.; Sparks, D.L. Phosphate reaction dynamics in soils and soil components: A multiscale approach. Adv. Agron. 2007, 94, 135–179. [Google Scholar] [CrossRef]
- Vasenko, L.; Qu, H. Novel two-stage oxidation/crystallization technology for high-purity calcium phosphates recovery from digester supernatant. J. Environ. Chem. Eng. 2018, 6, 2975–2982. [Google Scholar] [CrossRef]
- Reuter, H.I.; Lado, L.R.; Hengl, T.; Montanarella, L. Continental-scale digital soil mapping using European soil profile data: Soil pH. In SAGA—Seconds Out; Böhner, J., Blaschke, T., Montanarella, L., Eds.; Universität Hamburg, Institut für Geographie: Hamburg, Germany, 2008. [Google Scholar]
- Díaz, I.; Barrón, V.; del Campillo, M.C.; Torrent, J. Testing the ability of vivianite to prevent iron deficiency in pot-grown grapevine. Sci. Hortic. 2010, 123, 464–468. [Google Scholar] [CrossRef]
- Bisschops, I.; Kjerstadius, H.; Meulman, B.; van Eekert, M. Integrated nutrient recovery from source-separated domestic wastewaters for application as fertilisers. Curr. Opin. Environ. Sustain. 2019, 40, 7–13. [Google Scholar] [CrossRef]
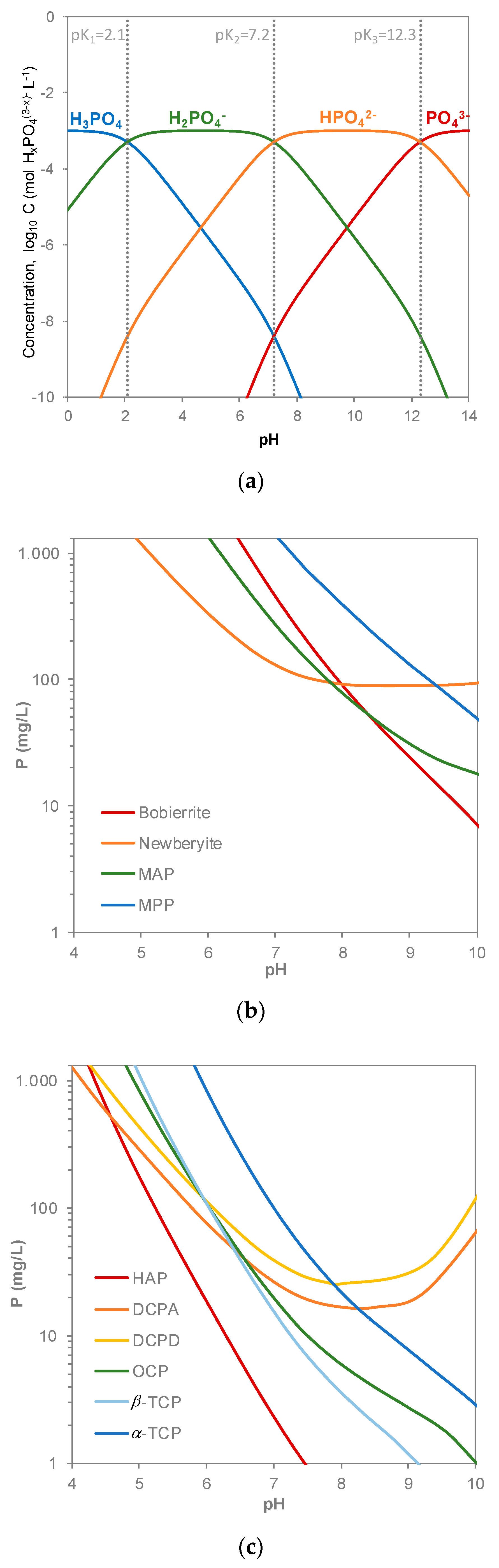

| Name | Empirical Formula | Molecular Weight (g/mol) | P Content (wt %) | Me/P Molar Ratio | pKsp (25 °C) | Requirements |
|---|---|---|---|---|---|---|
| MgP minerals | ||||||
| Magnesium ammonium phosphate (MAP, struvite) | MgNH4PO4·6H2O | 245.41 | 12.62 | 1.00 | 13.26 1 | pH > 7; Mg/Ca molar ratio > 0.6 |
| Magnesium potassium phosphate (MPP, K-struvite) | MgKPO4·6H2O | 266.47 | 11.62 | 1.00 | 11.68 2 | NH4+ unavailable |
| Magnesium hydrogen phosphate trihydrate (newberyite) | MgHPO4·3H2O | 174.33 | 17.77 | 1.00 | 5.51 3 | Acid pH values |
| Trimagnesium phosphate octahydrate (bobierrite) | Mg3(PO4)2·8H2O | 406.98 | 15.22 | 1.50 | 25.20 4 | Basic pH values |
| CaP minerals | ||||||
| Hydroxyapatite (HAP) | Ca10(PO4)6(OH)2 | 1004.6 | 18.50 | 1.67 | 116.8 5 | Long timescale (ripening of precursors); Low Mg/Ca ratio |
| Tricalcium phosphate (α-TCP and β-TCP) | Ca3(PO4)2 | 310.18 | 19.97 | 1.50 | 25.5 (α), 28.9 (β) 5 | - |
| Amorphous calcium phosphate (ACP) | Cax(PO4)y·nH2O | * | * | * | * | Lack of crystallisation nuclei |
| Octacalcium phosphate (OCP) | Ca8H2(PO4)6·5H2O | 982.53 | 18.91 | 1.33 | 96.6 5 | - |
| Dicalcium phosphate (DCPA, monenite) | CaHPO4 | 136.06 | 22.76 | 1.00 | 6.90 5 | Acid pH values |
| Dicalcium phosphate dihydrate (DCPD, brushite) | CaHPO4·2H2O | 172.09 | 18.00 | 1.00 | 6.59 5 | Acid pH values |
| FeP minerals | ||||||
| Ferrous iron phosphate (vivianite) | Fe3(PO4)2·8H2O | 501.61 | 12.35 | 1.50 | 36.0 6 | Stimulated by Fe dosage |
| Waste Water Source | Precipitation Performance | Bioprocess Performance | Precipitate Characteristics | Reference |
|---|---|---|---|---|
| NDN as the bioprocess following chemical P precipitation | ||||
| Hydrolysed urine | MgP precipitated in lab tests simultaneously to vacuum thermal stripping at 25–65 °C by adding MgCl2 | NDN via nitrite; Increased COD/N and ALK/NH4+-N mass ratios after precipitation favoured NDN | MgP mostly precipitated as MAP; Composition of the precipitate: 13.1% P, 8.2% Mg, 5.3% N | [68] |
| Anaerobically digested sludge dewatering centrate from a municipal WWTP | MgP precipitated in a lab batch reactor by adding MgCl2 | Nitrification in a fixed-film reactor, denitrification in a MFC-cathode; MFC-anode fed with raw waste water | No data available for the precipitate | [105] |
| Anaerobic lagoon digester supernatant in a swine farm | MgP precipitated in lab batch tests; Raw vs. (organically) fermented superphosphate fertiliser used as P source; Fermentation increased water-soluble P and provided bCOD | NDN in a SBR; COD/TN mass ratio raised by adding fermented superphosphate favouring NDN | MgP precipitated mostly as MAP; Effectiveness of the precipitate as fertiliser demonstrated in a pot test (increased crop yield) | [106] |
| Side-stream sludge fermenter effluent from an EBPR system treating low-strength synthetic waste water | CaP precipitated in a SBR by adding Ca(OH)2 | EBPR in a continuous flow-activated sludge system; Fermentation of purged sludge + precipitation of released P lead to improved P uptake and N removal in the mainstream | Composition of the precipitate: 13.8% P | [107] |
| Urine | PO4 precipitated in a lab stirred system using RO brine (from a reclaimed WWTP in a thermal power plant) as metal ions source | NDN via nitrite in a continuous flow-activated sludge system using an internal/external COD source | Precipitates produced at different brine/urine mixture ratios with 10–15% P; The amount of brine added affected Mg or Ca in the solid; No presence of hazardous compounds | [108] |
| Ultrafiltered permeate from an(aerobically) digested sludge dewatering centrate in a municipal WWTP | MgP precipitated in lab batch tests; Mg/NH4/PO4 adjusted with MgCl2 + K2HPO4; Use of seeding material | NDN in a continuous flow-activated sludge system for treating raw sewage plus MgP precipitation effluent; Mixing of precipitation effluent with raw sewage decreased COD and N removal efficiency | MgP precipitated mostly as MAP; Quality of the MgP precipitated potentially compromised by the heavy metals content | [109] |
| Urine-seawater mixture | PO4 precipitated in a lab continuous flow reactor incl. a concentric cylinder differentiating hydrolysis-precipitation zone and settling-separation zone | Nitrification in a continuously aerated SBR (for subsequent in-sewer denitrification); ALK added as NaHCO3 based on pH set-point | MgP precipitated mostly as MAP; MAP purity directly correlated with urine content | [110] |
| Low-strength synthetic waste water | MgP precipitated in a lab FBR | NDN in a MBR intermittently aerated; P-recovery in FBR enhanced SND and COD, NH4+ and PO4 removal in MBR | MgP precipitate mostly as MAP | [111] |
| Low-strength synthetic waste water | CaP precipitated in a lab upflow reactor; CaCl2 added after the anaerobic stage of an EBPR system and CO2 stripping by aeration; Calcite as seeding material | EBPR in a continuous flow-activated sludge system for N and P removal; Chemical P precipitation favoured EBPR | CaP precipitate mostly as HAP (or precursors); Composition of the precipitate: 8–13% P; Carbonate co-precipitation | [112,113] |
| Side-stream sludge fermenter effluent from an EBPR system treating low-strength synthetic waste water | MgP precipitated in batch by adding MgO | EBPR in an SBR also involved partial N removal through NDN; Fermentation of purged sludge followed by precipitation of the released N and P lead to higher P separation efficiency and complete nitrification | MgP precipitate mostly as MAP | [114] |
| Swine waste water | MgP precipitated in a lab CSTR; Precipitate recovered from an external settler; MgP precipitate partly recycled to the reaction tank as seeding material | NDN in a 2-stage continuous flow activated sludge system (+ settler); COD/TN mass ratio increased favouring NDN, COD/TP mass ratio increased favouring EBPR | MgP precipitate partly as MAP; Anhydrous precipitate with 25.3% P, 18.3% Mg, 7.1% N, 5.4% K, 2.5% Ca; Quality of the precipitate potentially compromised by the organic content | [115] |
| Anaerobically digested landfill leachate | MgP precipitated in a lab stirred system using MgCl2 and PO4 | NDN in an SBR; BOD5/NH4+-N mass ratio increased favouring NDN; The higher MgCl2 addition the higher NH4+ separation as MAP; High Cl− could limit nitrification | Composition of the MgP formed not determined analytically | [116] |
| Rare-earth smelting waste water | MgP precipitated in lab batch tests; Mg/NH4/PO4 adjusted with MgSO4 + Na2HPO4 (among other P-sources) | SND in a batch system using immobilised microorganisms; COD/NH4+-N mass ratio increased favouring NDN; Rare metal ions removed from the liquid phase favouring biological treatment | MgP precipitate mostly assumed as MAP; Composition: 15.6% P, 9.3% N, 8.3% Mg; Quality of the MgP precipitated compromised by the rare metal content | [117] |
| Raw swine waste water obtained by centrifugation | MgP precipitated in lab batch tests; Mg/NH4/PO4 adjusted with MgCl2 + KH2PO4 | NDN in a fill-and-draw system under intermittent aeration; BOD/TN increased enhancing NDN; Additional PO4 precipitated as CaP by adding CaCl2 once biotreatment ended | Composition of the MgP not determined analytically | [66,118] |
| PNA as the bioprocess following chemical P precipitation | ||||
| Digester sludge thickening lagoon supernatant in a municipal WWTP | MAP precipitated by the Ostara® process at full-scale | PNA in an IFAS-SBR with intermittent aeration; High PO4 inhibited anammox bacteria; Higher tolerance to PO4 stress when biomass grew as biofilm rather than forming flocs | No data available for the precipitate | [69] |
| Anaerobically digested sludge dewatering centrate in a municipal WWTP | MgP precipitated in a lab CSTR with MgO | PNA as 2-step process (PO4 precipitated after PN) and also as 1-step process (with intermittent aeration) using SBRs | Composition of the MgP formed not determined analytically | [119] |
| Anaerobically digested industrial (potato) waste water | MgP precipitated in a lab mixed reactor with MgCl2; Urea added to the crystallisator as N source | PNA as 2-step process (CSTR w/o sludge retention + UFBR) and also as 1-step process (UFBR) | MgP precipitate mostly as MAP | [120] |
| Anaerobically digested industrial (potato) waste water from a UASB reactor + anaerobically digested sludge dewatering centrate in a municipal WWTP | MAP precipitated by the PHOSPAQTM process at full-scale, aeration supplied for bCOD degradation and CO2 stripping; MgO dosed as Mg source | PNA as 1-step process with granular sludge and dynamically controlled continuous aeration at 37 °C | MAP with low heavy metals content with respect to EU standards for fertilisers; Precipitate fertilising effectiveness demonstrated in field tests | [121,122] |
| Anaerobically co-digested sludge dewatering supernatant in a municipal WWTP | MgP precipitated in a CSTR using MgCl2 or MgCO3 | PNA as 2-step process: MBBR + SBR; PO4-P increase implied specific anammox activity loss | Composition of the MgP formed not determined analytically | [67] |
| Waste Water Source | Experimental Conditions | Bioprocess Performance | Precipitate Characteristics | Reference |
|---|---|---|---|---|
| Nitrification as the bioprocess concomitant to P precipitation | ||||
| Synthetic urine | Long-term nitrification in trickling filters partially packed with pumice and mussel shells | PO4 precipitation induced by Ca released from mussel shells; Mussel shells also provided carbonate needed for buffering the system | Composition of the CaP formed not determined analytically | [168] |
| Synthetic waste water mimicking anaerobic digester dewatering centrate | PN in aerated SBR vs. SBBR packed with polyethylene particles | SBR: faster start-up; SBBR: more stable facing disturbances in dissolved oxygen conditions | Suspended solids containing 9–10% P; Complementary work [169] in the SBR transforming PN into NDN showed that PO4 precipitated in different form depending on pH | [71] |
| Anammox as the bioprocess concomitant to P precipitation | ||||
| Synthetic waste water mimicking effluent from a PN reactor | Anammox process in an EBR with internal recirculation; Reactor at 33 ºC and pH 8.6 | NLR 5 g NH4+-N L−1 d−1 and NRE 87%; PLR 0.21 g PO4-P L−1 d−1 and PUE 66%; Granule size 1.3–1.7 mm; Complementary works: [170] attached film EBR (35 °C) reached NLR 50 g NH4+-N L−1 d−1 and NRE 90% + PLR 0.55 g PO4-P L−1 d−1 and PUE 72%; [171] attached film EBR (35 °C) reached NLR 10 g NH4+-N L−1 d−1 and NRE 77% + PLR 0.15 g PO4-P L−1 d−1 and PUE 68%; [172] EBR (25 °C) reached NLR 20 g NH4+-N L−1 d−1 and NRE 84% + PLR 1.32 g PO4-P L−1 d−1 and PUE 94% | CaP recovered as HAP; Granules with 83% mineral content, 28.9% Ca, 13.4% P, 0.9% N, 0.5% Mg | [72] |
| PNA as the bioprocess concomitant to P precipitation | ||||
| Anaerobically digested sludge dewatering centrate in a municipal WWTP | PNA process as 1-stage SBR at 25 °C; No addition of chemicals (w/o pH control) | NLR 0.31 g NH4+-N L−1 d−1 and NRE 66% | CaP recovered as HAP; Granules purged with 71% ashes, 33.5% Ca, 15.8% P; Heavy metals complied with EU standards for P fertilisers and needs from P industry | [70] |
| Waste Water Source | Bioprocess Performance | Precipitation Performance | Precipitate Characteristics | Reference |
|---|---|---|---|---|
| Nitrification as the bioprocess preceding chemical P precipitation | ||||
| Anaerobically digested swine waste water from a UASB reactor | Nitrification in a continuous aerated system at pilot-scale; Similar tests also conducted considering NDN and PNA as the bioprocess preceding P precipitation | CaP precipitated in lab batch tests using hydrated lime; Similar results to those previously given by [205] | Field tests were also performed; The sludge formed contained 30% Ca, 5.7% P, 2.5% Mg; The high Ca/P ratio indicates carbonate co-precipitation | [74] |
| Anaerobically digested industrial (potato) waste water from a UASB reactor | Nitrification in an aerated CSTR | CaP precipitated in a lab CSTR with CaCl2; High pH and Ca/P triggered co-precipitation of carbonates and MgP; Nitrification reduced Ca(HCO3)2 inhibition and carbonate precipitation making Ca available for CaP precipitation | CaP precipitated mostly as HAP; Ca/P/Mg ratio as 1.5/1.0/0.1 | [206] |
| Anaerobically digested industrial (potato) waste water previously processed for urease-induced partial phosphate removal | Nitrification in an aerated MBR; Mg content and pH increased due to preliminary ureolytic phosphate removal | CaP precipitated in lab batch tests; Several chemicals used to raise pH and test effect of TIC; TIC decrease due to nitrification implied shorter dosage of NaOH; NaOH supply w/o nitrification may lead to MgP and CaCO3 formation; pH increase with Ca(OH)2 favoured PO4 precipitation due to extra Ca dosage; pH increase with Na2CO3 hampered Ca and PO4 precipitation | Composition of the precipitate not determined analytically; Similar results than in [207] | [124] |
| Surface lagoon liquids from swine farms | Nitrification in fluidised bed reactors with sludge immobilised in supporting porous medium; >90% ALK reduction and prevention of NH3 emission | CaP precipitated in lab batch tests; Ca(OH)2 added as precipitant; pH increased to ≥9.5 for optimum P precipitation; Similar field test in [73]; P in effluent adjustable for on-farm use according to crop needs | Composition of the precipitate: 2.5–10% P; Nitrification favoured P availability in the precipitate; The recovered material can be exported and reused as P-fertiliser; The high pH value applied in the precipitation process destroys pathogens | [208] |
| NDN as the bioprocess preceding chemical P precipitation | ||||
| Fertiliser industry waste water | NDN in a continuous flow activated sludge system at pilot-scale | MgP precipitated in a continuous pilot-scale unit; Mg/NH4/PO4 adjusted with MgCl2 + H3PO4; Mg and PO4 recovered from the precipitate by heating allowing the recycling within the process | Composition of the precipitate after drying: 24.0% P, 16.6% Mg, 1.1% N | [209] |
| Subfloor slurry from a pig farm, liquid separated by a rotary press | NDN in a continuous flow activated sludge system at full-scale | CaP precipitated in a continuous full-scale unit; Hydrated lime injected to pH 9.5 | Composition of the precipitate not determined analytically; Total faecal coliforms and enterococci reduced | [99] |
| Calf manure | NDN running at full-scale | MPP precipitated in: (1) lab experiments at room temperature, (2) pilot plant running as CSTR, and (3) full-scale plant (3 CSTRs + settler); MgO added as Mg source | Lab: Fine-grained elongated crystals with a max. diameter of several tens of microns; Pilot plant: the recovered product contained 12.5% P, 10.7% Mg, 11.4% K, 1,3% Ca, 0.7% N (5.5% organic-C) | [210] |
| PNA as the bioprocess preceding chemical P precipitation | ||||
| Anaerobically digested sludge dewatering centrate from two municipal WWTPs ‒with chemical vs. biological P removal in the mainstream‒ | PNA as 1-step process at lab- and pilot-scale | MgP precipitated in lab batch tests; K uptake from water line positively correlated with pH value up to 11 | MgP precipitated as MAP and MPP; Precipitate composition (pH 11): 11.4% P, 9.2% Mg, 4.3% K, 3.5% N, 0.9% Ca | [76] |
| Anaerobically digested manure centrate | After biological treatment, Mg/K/P molar ratio adjusted to ca. 2.0/4.5/1.0 | MgP precipitated in lab batch tests targeting MPP recovery; Different dilutions rates tested to decrease interference by TSS | MgP precipitated mostly as MPP with co-precipitation of other salts; Suspended particles favoured aggregation of crystals as star-asterisk forms | [75] |
| Characteristics | Upstream | Concomitant | Downstream |
|---|---|---|---|
| Purity | Presence of solids and other compounds will disturb crystallisation and affect purity of precipitate | Heterogeneous precipitate | Increased effective ion availability favouring crystal formation |
| Organic content | High | High | Low |
| Toxicity | Potential presence of metals, organics and pathogens depending on the waste water origin | - | Pathogen destruction depending on process pH |
| P form (most probable) | MAP and CaP. Carbonate forms may co-precipitate together with phosphates | CaP and MAP | Absence of ammonium will favour formation of alternative MgP salts such as MPP |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magrí, A.; Carreras-Sempere, M.; Biel, C.; Colprim, J. Recovery of Phosphorus from Waste Water Profiting from Biological Nitrogen Treatment: Upstream, Concomitant or Downstream Precipitation Alternatives. Agronomy 2020, 10, 1039. https://doi.org/10.3390/agronomy10071039
Magrí A, Carreras-Sempere M, Biel C, Colprim J. Recovery of Phosphorus from Waste Water Profiting from Biological Nitrogen Treatment: Upstream, Concomitant or Downstream Precipitation Alternatives. Agronomy. 2020; 10(7):1039. https://doi.org/10.3390/agronomy10071039
Chicago/Turabian StyleMagrí, Albert, Mar Carreras-Sempere, Carmen Biel, and Jesús Colprim. 2020. "Recovery of Phosphorus from Waste Water Profiting from Biological Nitrogen Treatment: Upstream, Concomitant or Downstream Precipitation Alternatives" Agronomy 10, no. 7: 1039. https://doi.org/10.3390/agronomy10071039
APA StyleMagrí, A., Carreras-Sempere, M., Biel, C., & Colprim, J. (2020). Recovery of Phosphorus from Waste Water Profiting from Biological Nitrogen Treatment: Upstream, Concomitant or Downstream Precipitation Alternatives. Agronomy, 10(7), 1039. https://doi.org/10.3390/agronomy10071039






